1.1. Applications of Burnt Lime in Sea Water
To combat the negative impact of sea urchins on tare forests in northern coastal waters and blue-green algal blooms in the surrounding of fish-farms (monoculture), Brooks et al. [
1] and Strand et al. [
2] has investigated addition of burnt lime (calcium oxide, CaO) to sea water as a remedial action. The treatment involves a rapid suspension of burnt lime in sea water and immediate discharge into the marine environment. Hence, it becomes of paramount importance to know how CaO particles behaves in contact with sea water and how this substance modifies the water chemistry.
The aim of this treatment is to increase tare growth and population density. This would provide a habitat for many species and will increase the biodiversity of the marine environment.
On a longer term one can imagine harvesting of high-quality sea urchins for the food market as well as a preparedness plan to treat blue-green algal blooms for the same market. In a wider perspective lime can be used to capture CO
2 in sea water as an environmental remediation [
3,
4]. However, designing an efficient treatment strategy to tackle the variable conditions expected in the sea requires increased understanding of the reaction kinetics of CaO in sea water, and this paper is an effort to elucidate this in comparison to fresh water as a novelty.
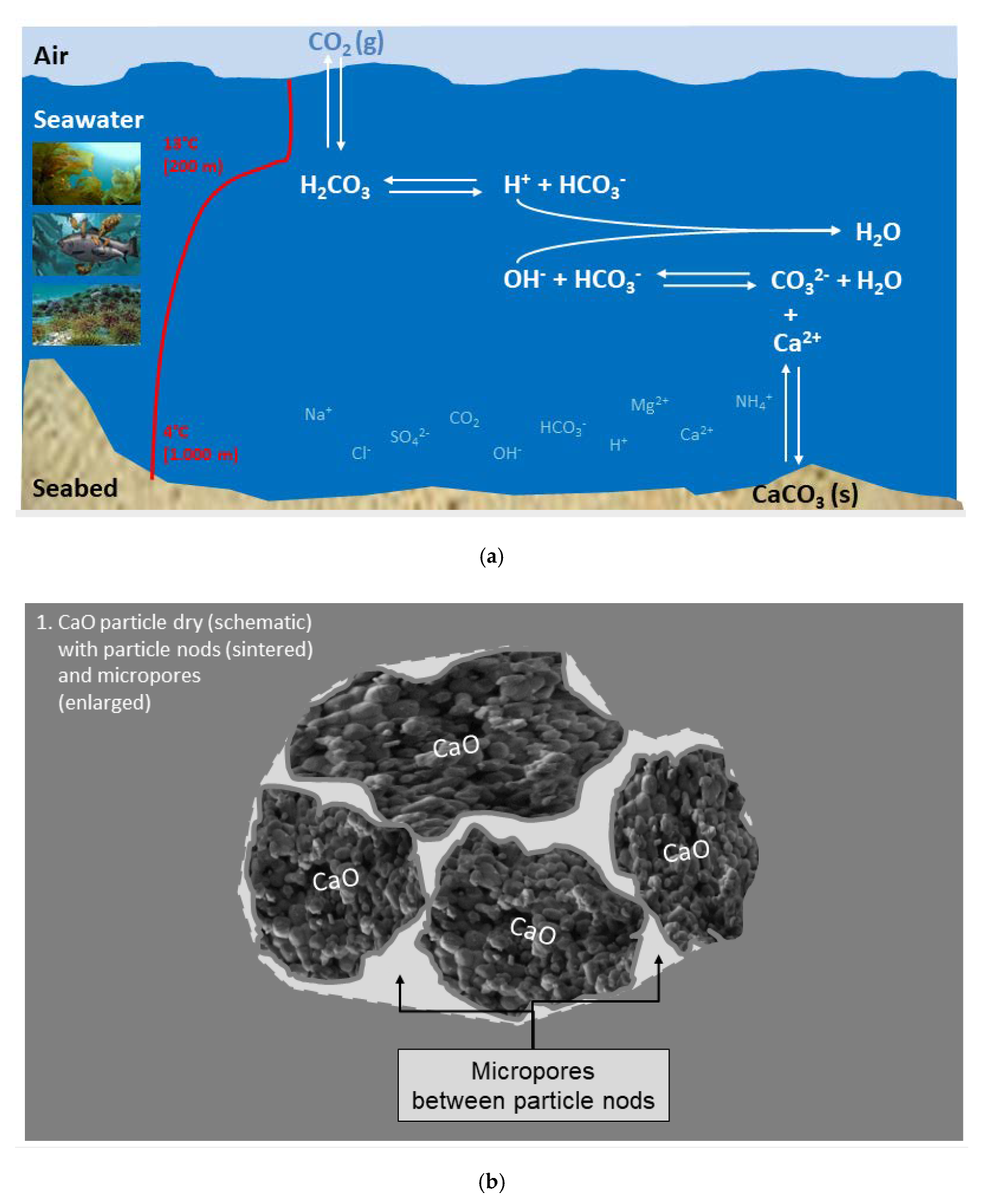
The burnt lime treatment is usually performed by spraying a suspension of CaO particles directly at the surface of the sea and all these particles will interfere with each other in their microenvironment until the gradual dilution is so great that they can be considered as single particles. For instance, the treatment in practice consists of pumping a slurry of 100 kg burnt lime (0.2–0.8 mm diameter) dispersed in 600 litres of sea water onto the surface. This provides an initial CaO concentration in the slurry of 167 g/L, that is taken as a starting point. Assuming that the average particle diameter is 0.5 mm and that they are spherical, the volume of the particle will be
or 0.0654 mm
3. Light-burnt lime (burnt at 1060 °C) has a typical particle density [
5] of ρ
p = 2 g/cm
3. The particle is porous since CaO has solid density ρ
s = 3.35 g/cm
3; porosity = (1 − ρ
p/ρ
s) × 100 vol% = 40 vol%, which is also what Wuhrer [
6] found. This gives for a 167 g/L slurry an outer volume of light-burnt lime of 83.5 cm
3 or in other words about 1275 particles per mL. These particles will however immediately react with water as discussed in the next Section about reaction with fresh water. The reaction with fresh water is evaluated first in order to illustrate the difference to sea water in the Section thereafter.
1.2. Chemistry of Burnt Lime (CaO) in Fresh Water
Burnt lime reacts violently with water to produce calcium hydroxide (so called slaked lime) according to the reaction below:
The reaction in Equation (1) releases a heat of Q = 1160 J/g CaO. The heat capacity of water is c = 4.18 kJ/kg·K. The increase in temperature of a mass (m) of water due to the reaction in Equation (1) can be calculated by the formula in Equation (2);
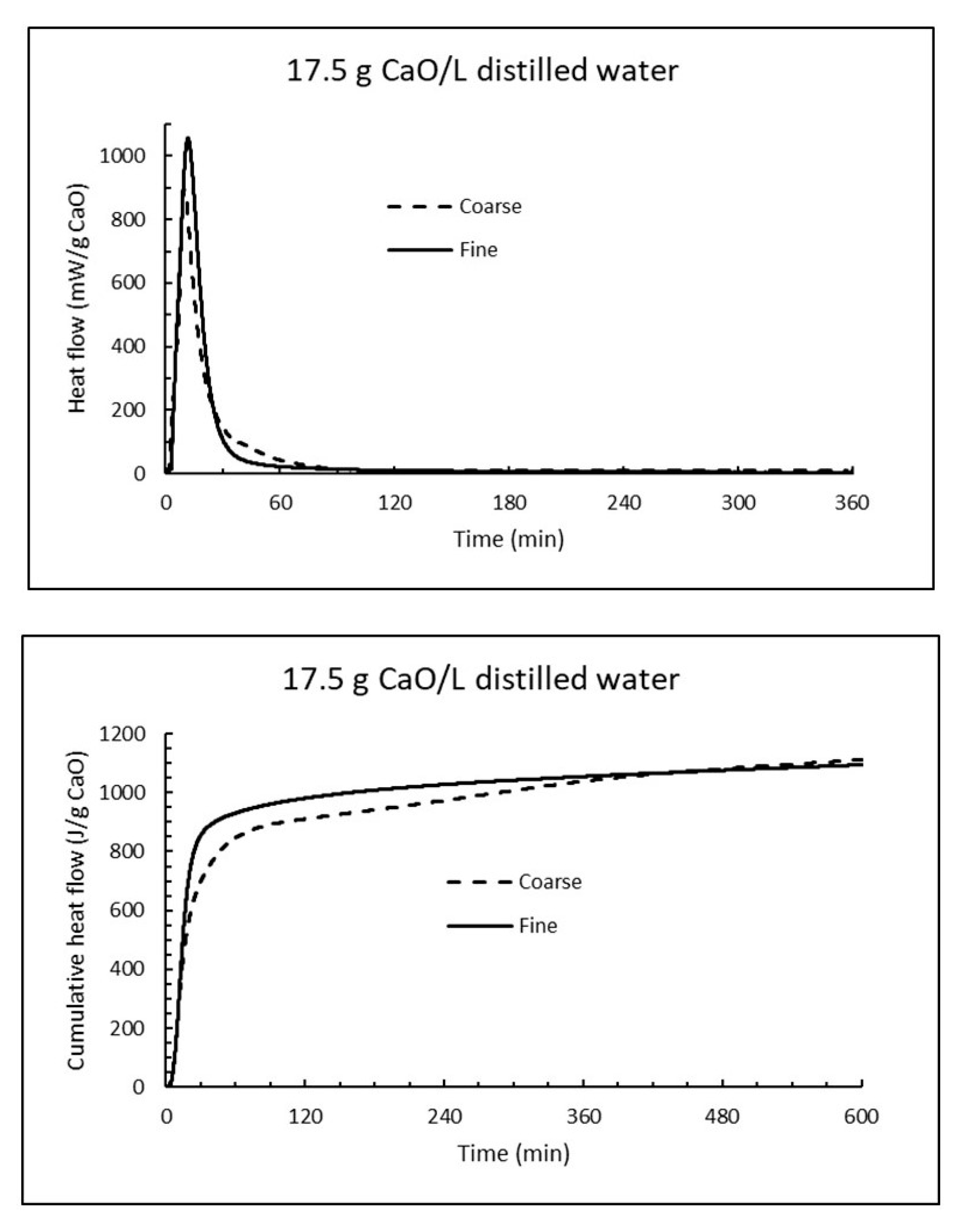
Using the same concentration of particles as in the preceding Section (167 g/L) and a density of water of 1000 g/L, one find that the water will be heated ΔT = 167 g × 1.160 kJ/g/(4.18 kJ/kg·K × 1 kg) = 46 K = 46 °C. This temperature increase is for a closed system (adiabatic, without heat loss) and 100% hydration of the burnt lime particles and will be proportional less for decreasing fractions of reaction. The temperature increase as a function of burnt lime dosage and fraction of hydration is given in
Table 1 as an illustration.
A light-burnt lime particle is not massive, but porous as shown in
Figure 1 [
7].
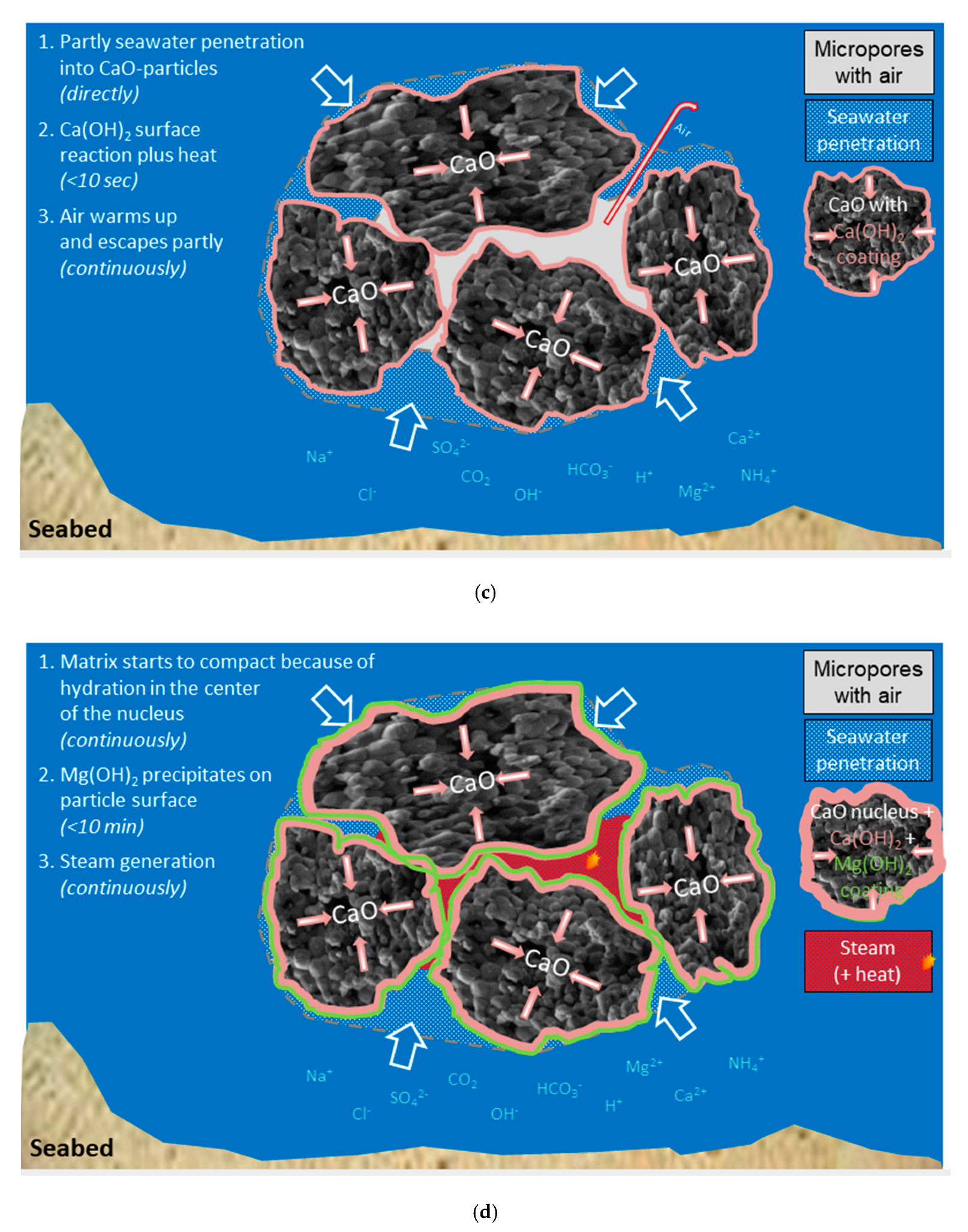
When the lime particle meets water, this will be sucked into the pores and react immediately to calcium hydroxide while the pore water temperature will increase rapidly to above the boiling point since there is a small amount of water relative to the CaO of the pore wall. If the precipitated calcium hydroxide has blocked the pore (Equation (1) doubles the volume of solid matter) the particle will disintegrate due to the thermal expansion of the remaining water and/or its vapor pressure. The reaction can be described as chaotic and will not be less chaotic when components from sea water interfere as described in next Section. Calcium hydroxide has limited solubility and the solubility actually decreases with increasing temperature as shown in Equation (3) (calculated from tabular data [
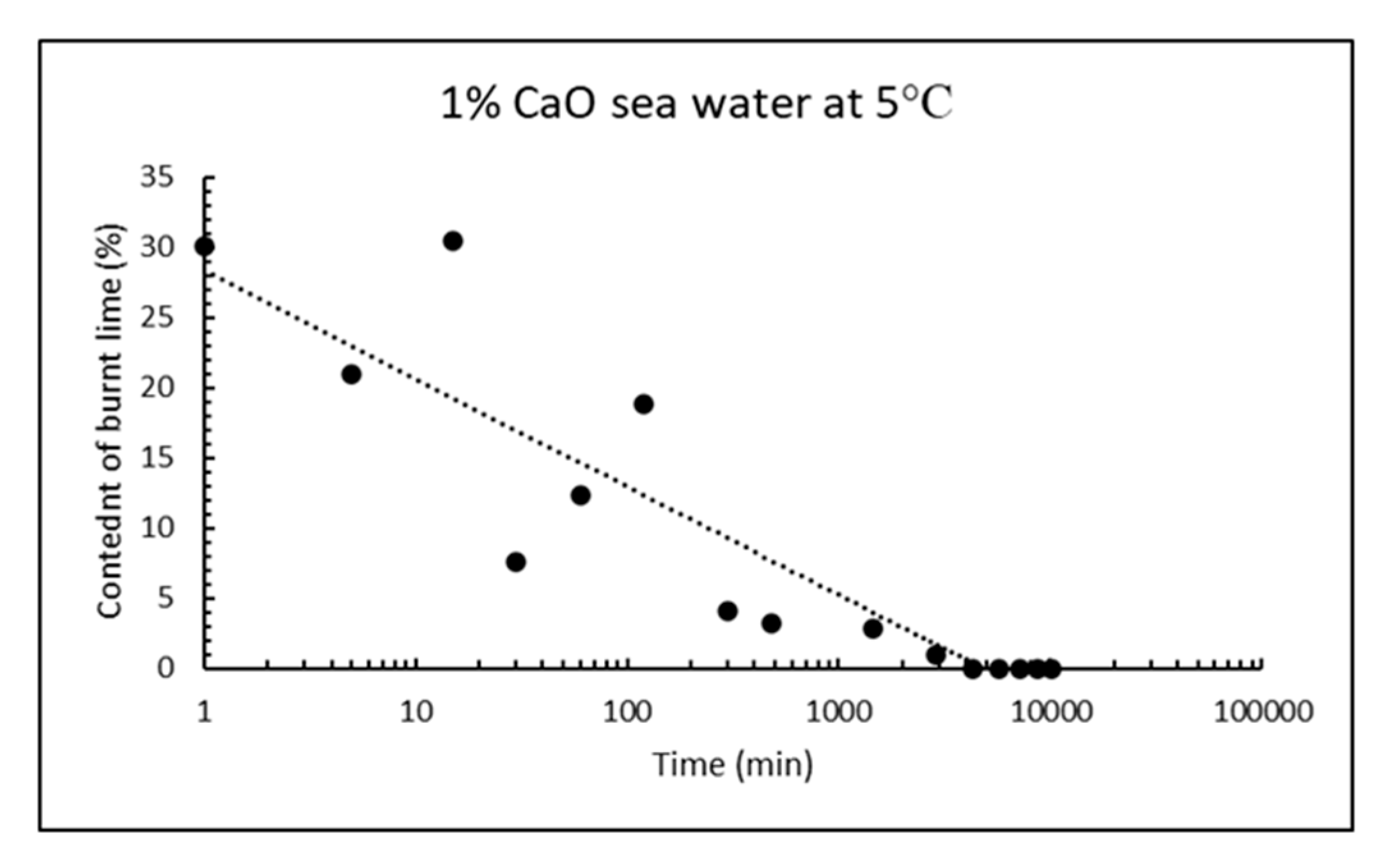
8]), an anomaly compared to most other compounds. The reaction rate of CaO, on the other hand, increases with increasing temperature and the degree of reaction for a light-burnt lime particle may reach 80% after 1 h under isothermal conditions, but after <5 min under adiabatic conditions (time to reach 60 °C according to EN459-2) depending on the fineness of the burnt lime.
In order to calculate the pH of burnt lime slaked in distilled water, one can use the solubility of 1.63 g/L at 25 °C (from Equation (3)) knowing that calcium hydroxide dissociates as written in Equation (4);
where the concentrations of ions in brackets are expressed in mol/L. According to Equation (4) the concentration of hydroxyl ions is twice that of the dissolved calcium hydroxide;
since M
w (Ca(OH)
2) = 74.08 g/mol, [OH
−] = 2 × 1.63 g/L/74.08 g/mol = 0.04405 mol/L
The relation between [OH
−] and pH at 25 °C is;
since
pH = −log [H
+] = 7 for neutral water since [H
+] = [OH
−] and [H
+] = √(1.0 × 10
−14).
[OH−] = 0.04405 M for dissolved calcium hydroxide gives then pH = 12.6 for a solubility of 1.63 g/L at 25 °C.
pH will be a bit lower than this as dissolved calcium hydroxide is not completely dissociated and species like CaOH
+ exists;
The geochemical data program GEMS (gems.web.psi.ch) [
9,
10] with PSI-GEMS thermodynamic data base for aqueous species and solids [
11] minimizes Gibbs free energy and gives the following composition of a saturated solution of calcium hydroxide at 25 °C:
[Ca+2] = 0.016370 M or 0.65611 g/L
[CaOH+] = 0.004265 M or 0.24345 g/L
[OH−] = 0.037006 M or 0.62910 g/L
pH = 12.5
This gives an amount of dissolved calcium hydroxide of 1.53 g/L instead of the 1.63 g/L obtained through Equation (3) from [
8], which explains the difference in calculated pH together with the presence of the species CaOH
+. Due to this species the pH as measured by a pH-meter will be lower (12.5) than the corresponding amount of dissolved calcium hydroxide; pH = 12.6 for 1.63 g/L dissolved Ca(OH)
2, which would have been the result if the solution was titrated rather than measured by a pH-meter. The proceeding evaluations will nevertheless be based on the solubility from Equation (3) (1.63 g/L resulting in pH = 12.6) and ignore the presence of species like CaOH
+ since it will react with the different species in sea water as if it was separated as Ca
2+ and OH
−.
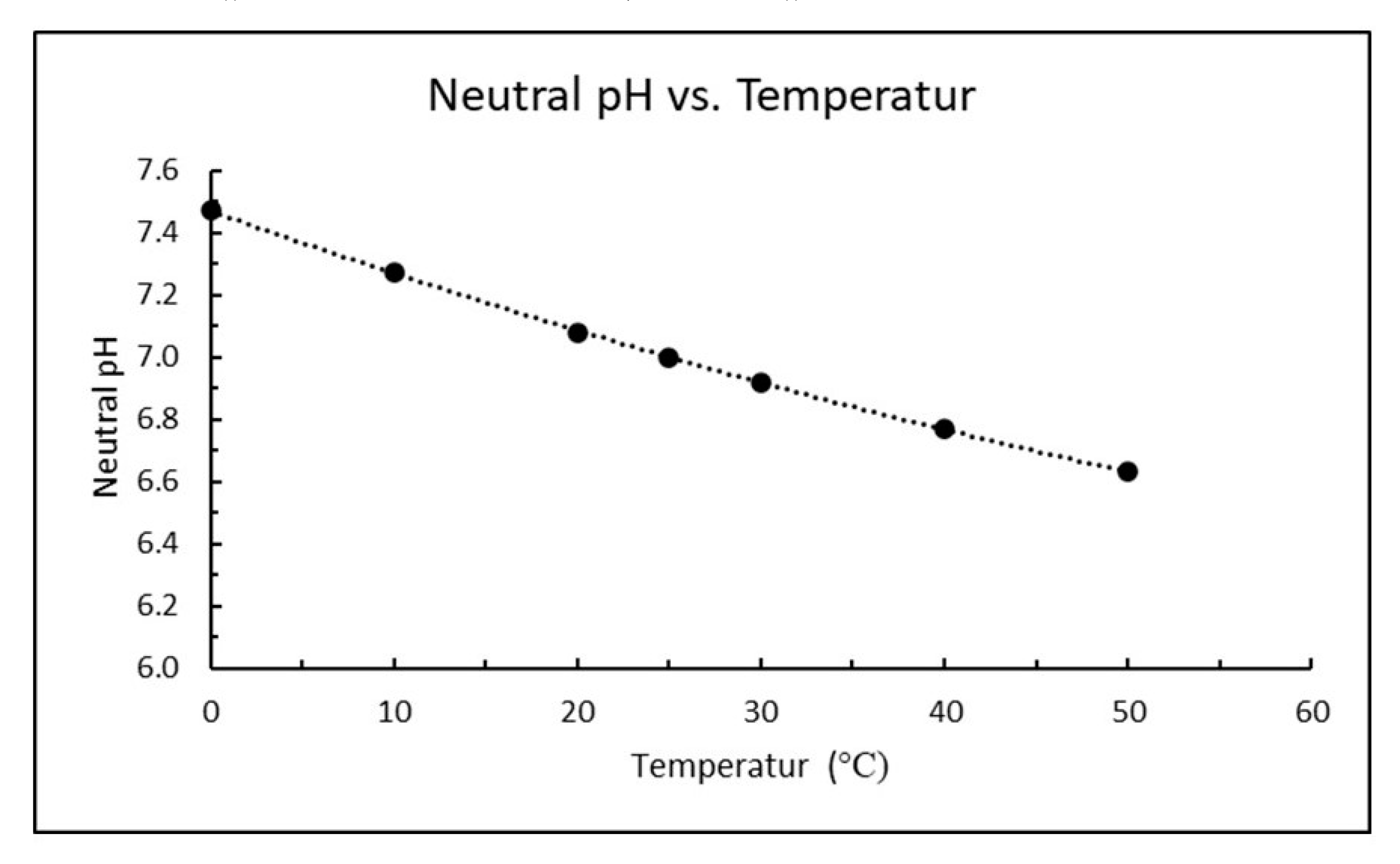
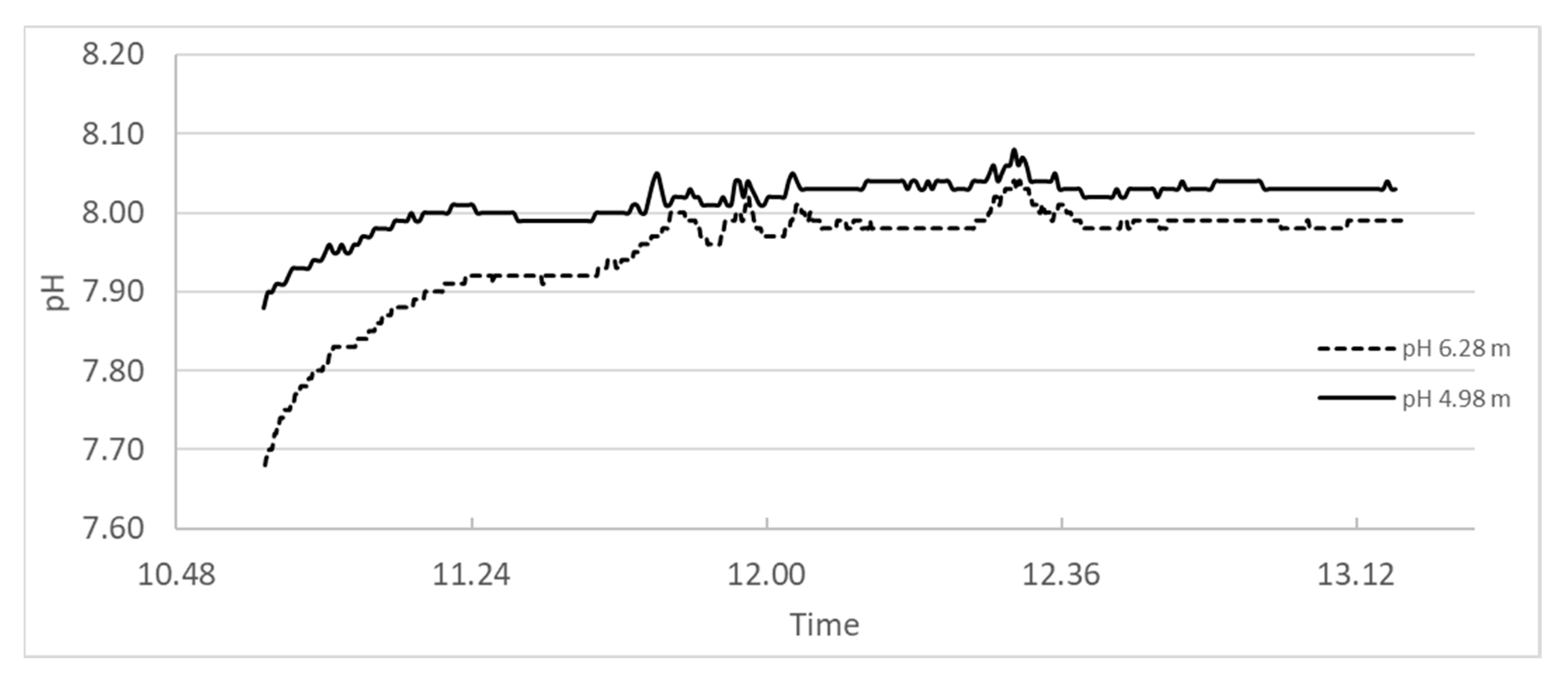
Another complicating factor for correct pH is that the self-dissociation of water in Equation (7) increases with increasing temperature as temperature is just a measure of molecular movement. The pH reference value of 14 will then increase with decreasing temperature, or pH of neutral water will decrease with increasing temperature as plotted in
Figure 2. Neutral pH at 5 and 15 °C would be 7.4 and 7.2, respectively, which corresponds to reference values of 14.8 and 14.4 (rather than 14.0 in Equation (7)) unless the pH-meter is correcting for temperature.
The pH in a suspension of calcium hydroxide will further increase with decreasing temperature since the solubility of calcium hydroxide increases as shown by Equation (3).
Consider distilled water equilibrating with natural air containing 400 ppm or 0.04 vol% carbon dioxide. The CO2 can partly be dissolved as the molecule or react with water to hydrogen carbonate (so called carbonic acid):
The main component of dissolved CO2 will be molecular CO2 (aq), since Kh is so small that the concentration of H2CO3 would be negligible and do not influence pH (i.e., log [H+]). The total solubility of CO2 at 100 kPa CO2 pressure (about 1 atm) and 25 °C is 1.45 g/L.
Hydrogen carbonate dissociates according to 2 sequential steps:
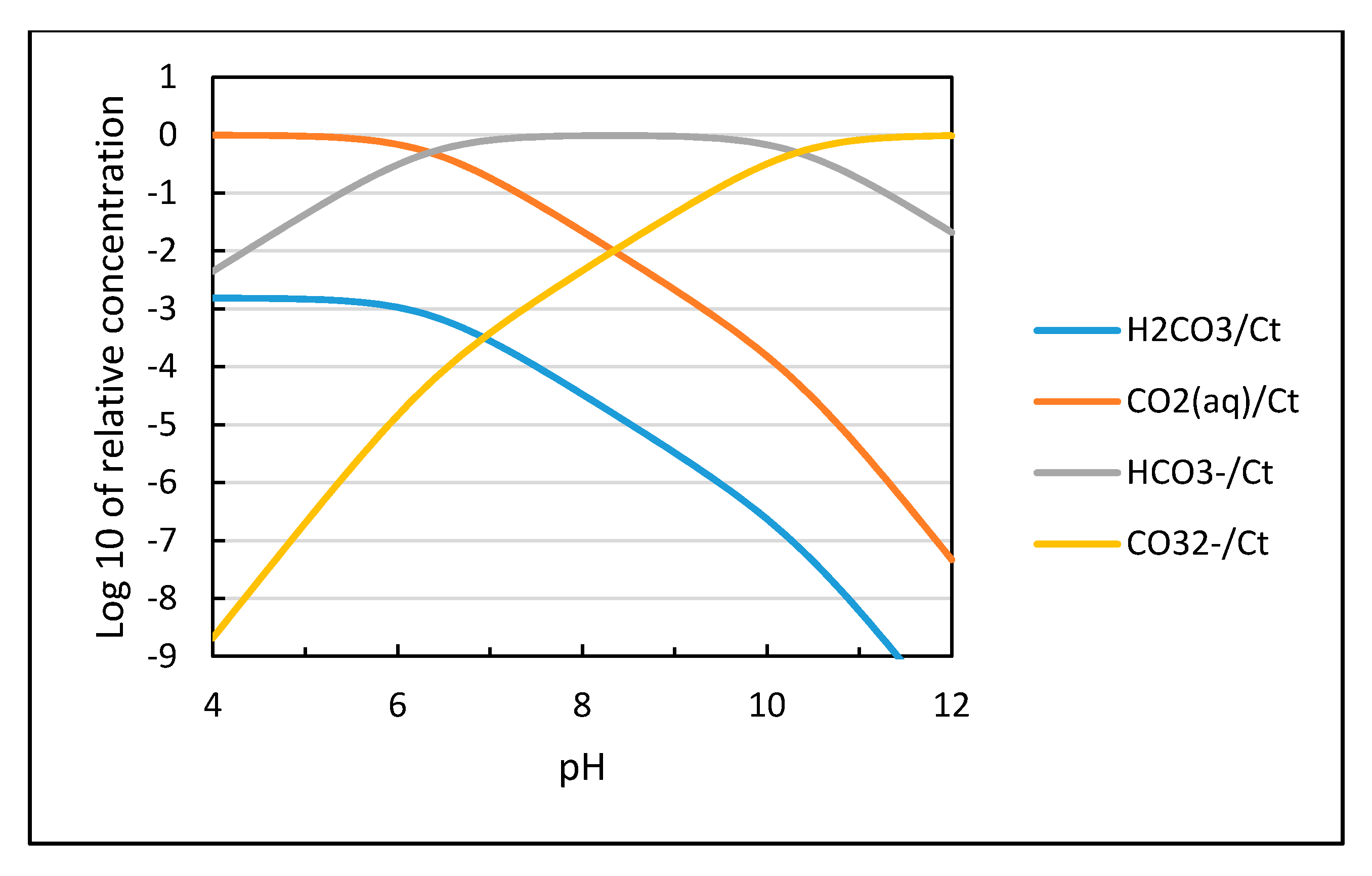
The distribution of the different species for CO
2 dissolved in water as a function of pH is shown in
Figure 3 based on calculations using the equilibrium constants in Equations (9)–(11). The solubility of CO
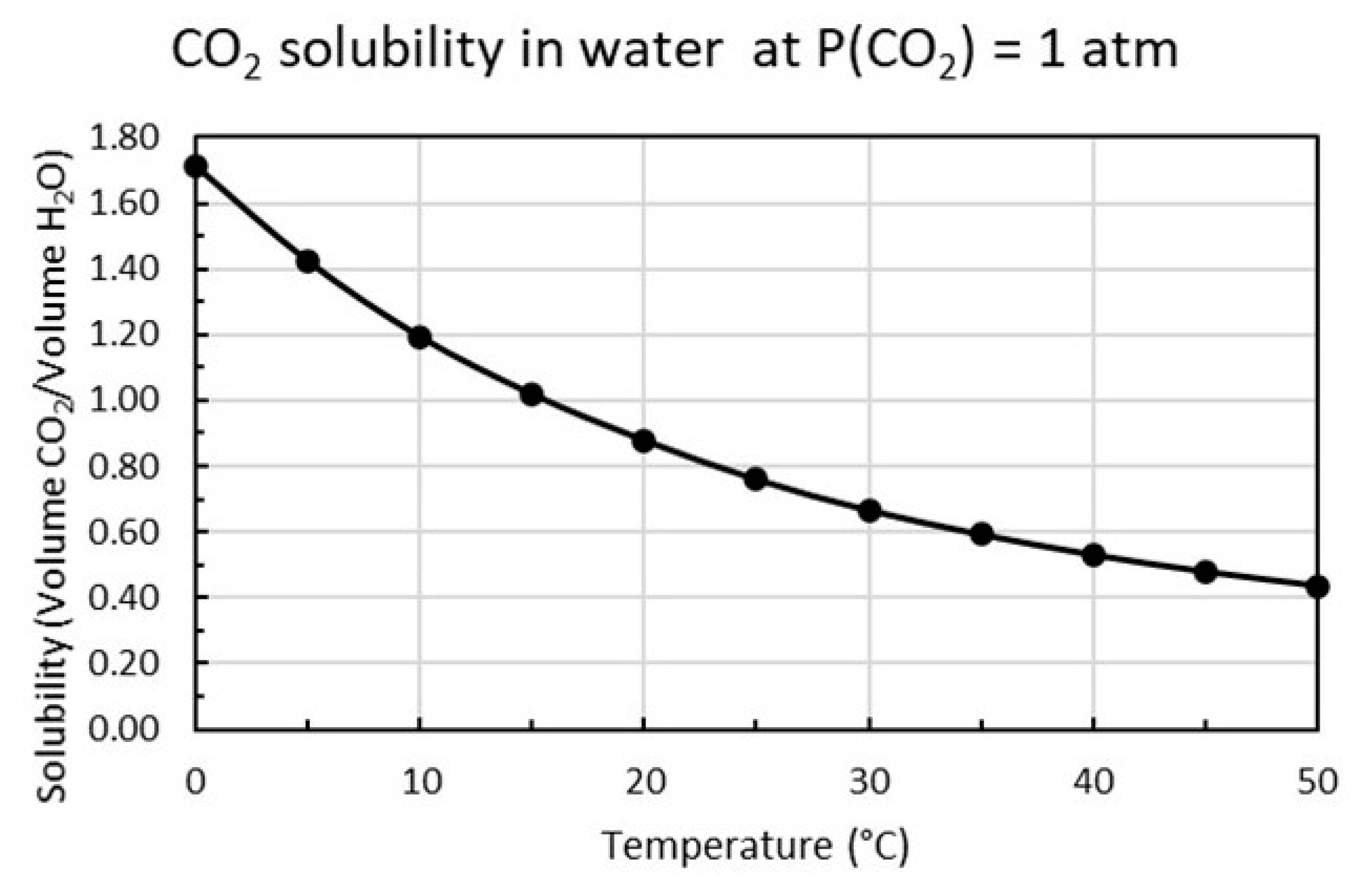
2 is proportional to the partial pressure of carbon dioxide as shown in Equation (12) and increases with decreasing temperature as plotted in
Figure 4.
For two different pressures Equation (12) can be reformulated to
Figure 4 shows that at 15 °C and P
1(CO
2) = 1 atm the concentration of carbon dioxide is [CO
2]
1 = 2 g/L or 45.45 mM. If k is constant over the whole pressure range, the solubility at P
2(CO
2) = 0.04 atm will be [CO
2]
2 = 0.08 g/L or 1.82 mM.
When excess calcium hydroxide with pH = 12.6 interact with fresh water with dissolved CO
2 the equilibria in Equations (9)–(11) will rapidly be displaced to the right and calcium carbonate will precipitate in an amount corresponding to the amount of dissolved CO
2 regardless of species:
If the amount of calcium hydroxide is far above saturation the pH will be unchanged at the same time as calcium carbonate is precipitating since the solubility of calcium carbonate is very small (≈ 0.014 g/L). The calcium concentration for saturated calcium hydroxide is about 22 mM [Ca
2+] and much higher than for saturated calcium carbonate having 0.14 mM [Ca
2+], a difference of about 150 times. This means that for thinning of an exactly saturated calcium hydroxide solution, the pH is controlled by dissolved CO
2:
In natural fresh water with dissolved alkali carbonate (CO
32−), alkali hydrogen carbonate (HCO
3−) and magnesium (Mg
2+) dissolved calcium hydroxide will react as follows:
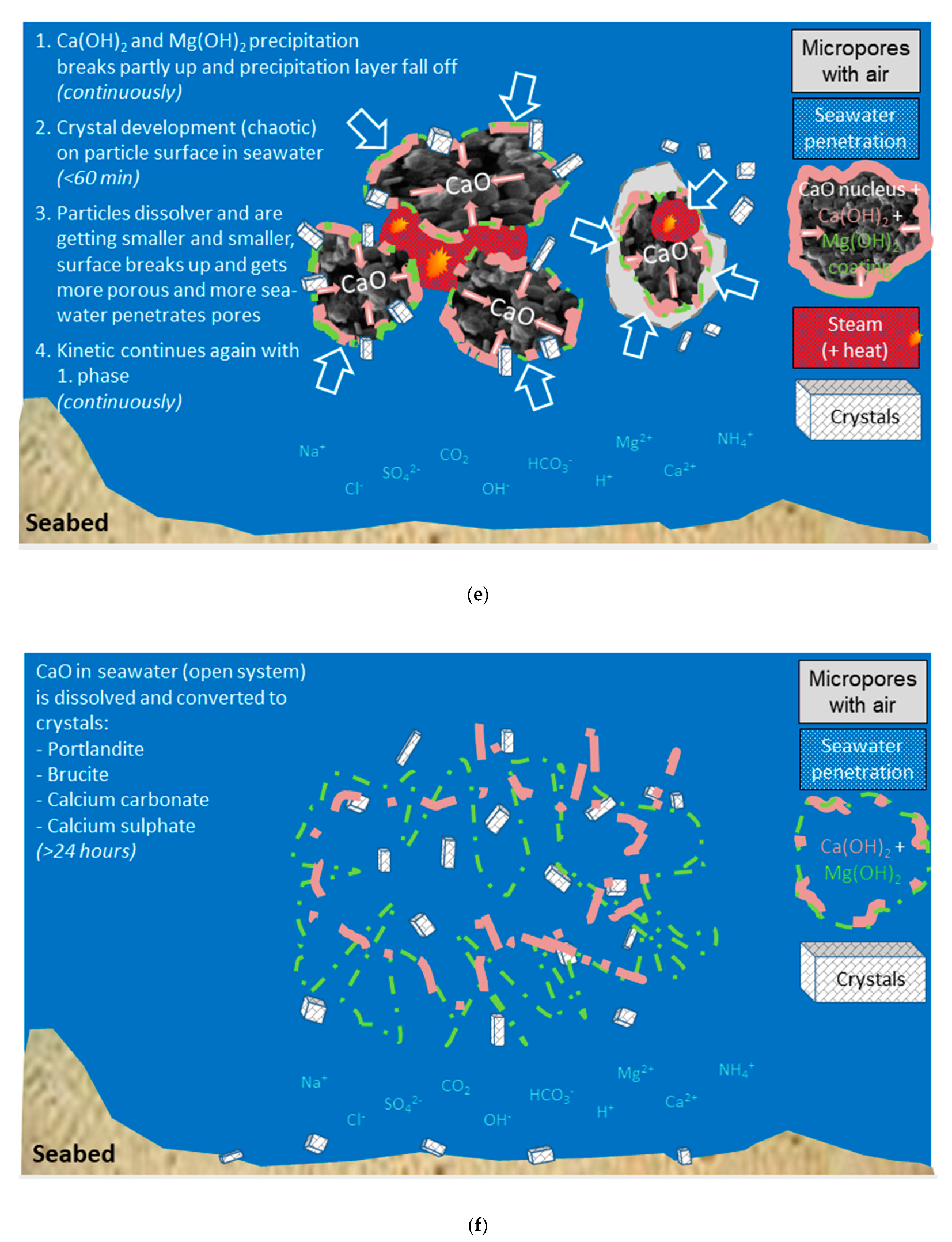
The reactions in Equations (16)–(18) leads to precipitates, since the solubilities at 25 °C for calcite, CaCO3, is 0.014 g/L or 0.14 mM, and for brucite, Mg(OH)2, 0.009 g/L or 0.15 mM. Calcite and brucite are considerably less soluble than portlandite (mineral name for calcium hydroxide), Ca(OH)2, with 1.63 g/L or 22 mM (a factor of about 150 in molarity).
For fresh water saturated with respect to calcium hydroxide, the reactions in Equations (16) and (17) will lead to a pH increase since soluble alkali hydroxides will form if the water contains alkali carbonates or alkali hydrogen carbonates. The reaction in Equation (18) will lead to a weak reduction in pH since it will increase the concentration of calcium ions that in turn will suppress the solubility of calcium hydroxide due to the common ion effect according to Equation (4).
For fresh water undersaturated with respect to calcium hydroxide, reaction 16 will reduce pH, reaction 17 not lead to any change and reaction 18 reduce pH if brucite is precipitated.
On long term, all calcium hydroxide in fresh water will be converted to calcium carbonate as there is an unlimited supply of CO
2 relative to calcium hydroxide while the rate is dependent on the concentration of CO
2. Pure fresh water without dissolved CO
2 in equilibrium with calcite (CaCO
3) has pH 9.9 [
15] but will be somewhat lower if there is dissolved CO
2 present as well. Based on the calcite solubility of 0.14 mM at 25 °C the pH should be about 10, but there is some uncertainty in the solubility.
1.3. Chemistry of Burnt Lime (CaO) in Sea Water
Atlantic sea water has typical composition listed in
Table 2 and the sum of components gives a mass of 3.513%, which is a typical salinity. The sum of positive charge is 0.623 M, while the sum of negative charge is 0.622 M, confirming electroneutrality and that all major ions are accounted for. Since sea water consists of a mixture of dissolved salts, one cation does not belong to a specific anion to make a compound.
The greatest difference between sea water and fresh water is the content of Na
+ and Cl
−. Sodium chloride is not expected to form compounds with calcium hydroxide, but according to Duschesne and Reardon [
16] the solubility of Ca(OH)
2 increases with increasing concentration of NaCl due to ion pair formations in their calculations, which were verified experimentally by Johnston and Grove [
17] and by Yeatts and Marshall [
18]. If a simplified version of sea water is 0.5 M NaCl, the solubility of Ca(OH)
2 will increase from 22 to 28 mM. On the other hand, a number of other ions in sea water will interact with Ca(OH)
2 and disturb this picture.
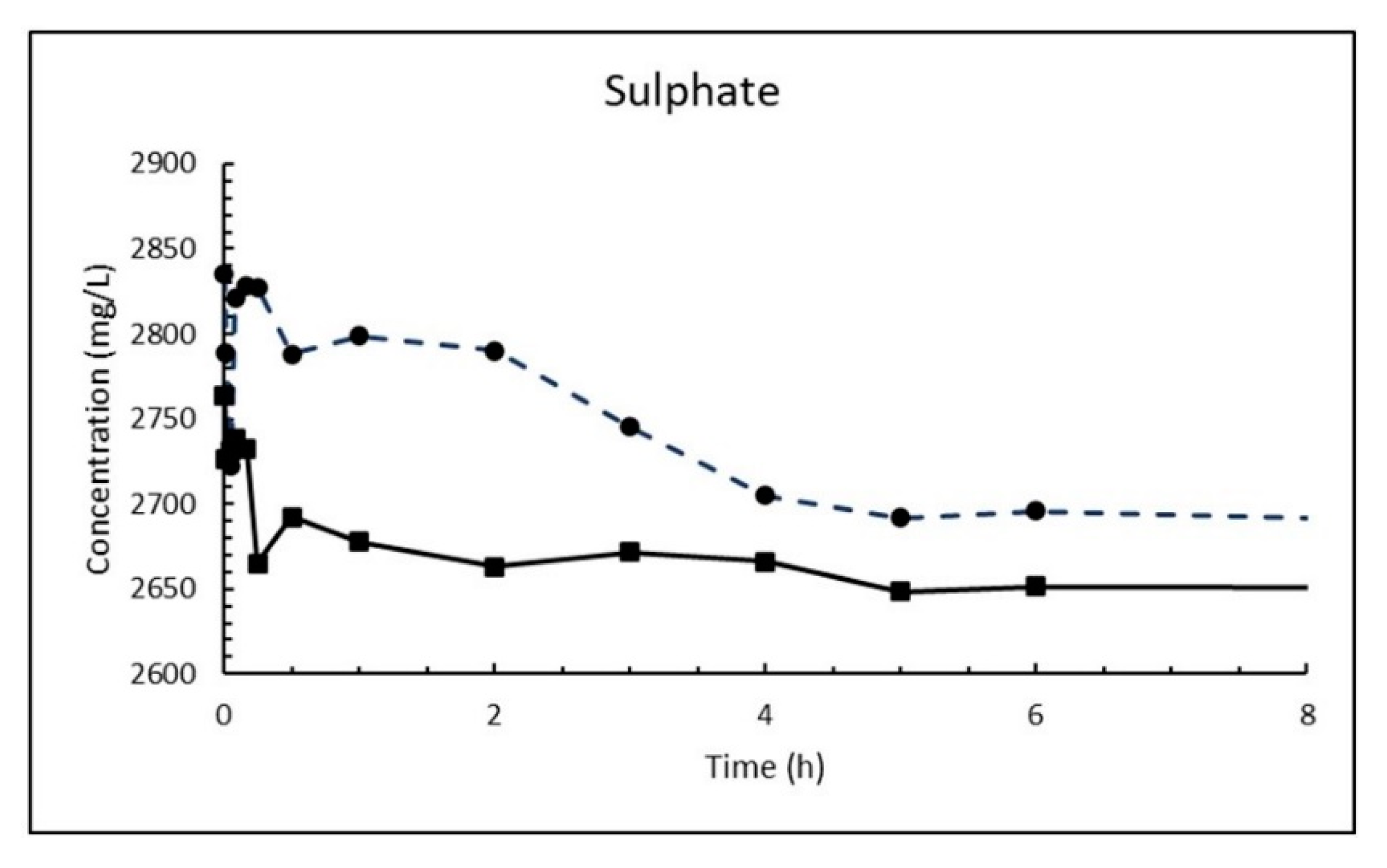
The second biggest difference is the content of sulphate and much higher concentration of magnesium. The magnesium will react as described in Equation (18), while the sulphate ions will form gypsum:
The magnesium reaction (Equation (18)) release calcium ions that suppress hydroxide concentration and gypsum formation (Equation (19)) will release hydroxyl ions that potentially reduce the calcium concentration locally relative to solid calcium hydroxide (Equation (4)), so the overall outcome is complex depending on concentration. According to
Table 2 the molarity of magnesium is 0.0549 M in Atlantic sea water as compared to 0.0289 M for sulphate, so in sea water the magnesium reaction will dominate.
The potential compounds formed by interaction of calcium hydroxide and sea water as listed in
Table 3 are predicted based on possible ion pairs and solubility data [
19].
The magnesium compounds with higher molar solubility than brucite, Mg(OH)2, are not expected to form on its expense. An experimental program was then initiated to measure which compounds that are actually formed when burnt lime is added to sea water.