Preparation of a New Iron-Carbon-Loaded Constructed Wetland Substrate and Enhanced Phosphorus Removal Performance
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Chemicals
2.3. Preparation of NICLS
2.4. Optimization of NICLS Preparation Conditions
2.5. Comparison with Other Constructed Wetland Substrates
2.6. NICLS Removal Experiment of Phosphorus in Simulated Wastewater
2.6.1. Effect of Reaction Time and Initial Phosphorus Concentration
2.6.2. Effect of pH
2.7. Characterization of the NICLS
3. Results and Discussion
3.1. Optimization of NICLS Preparation Conditions
3.1.1. Effect of Adding Varying Amounts of Thickener (Hydroxyethyl Cellulose) on the Removal Capacity of NICLS
3.1.2. Effects of Activated Carbon Types and Activated Carbon Particle Size on the Removal Capacity of NICLS
3.1.3. Effect of Fe/C Ratio of NICLS on Removal Capacity
3.2. Comparison of the Phosphorus Removal Effects of NICLS, Coated Iron, Coated Carbon, and Uncoated Zeolite
3.3. Comparison with Other Constructed Wetland Substrates
3.4. NICLS Phosphorus Degradation Ability
3.4.1. Effect of Contact Time and Initial Phosphorus Concentration
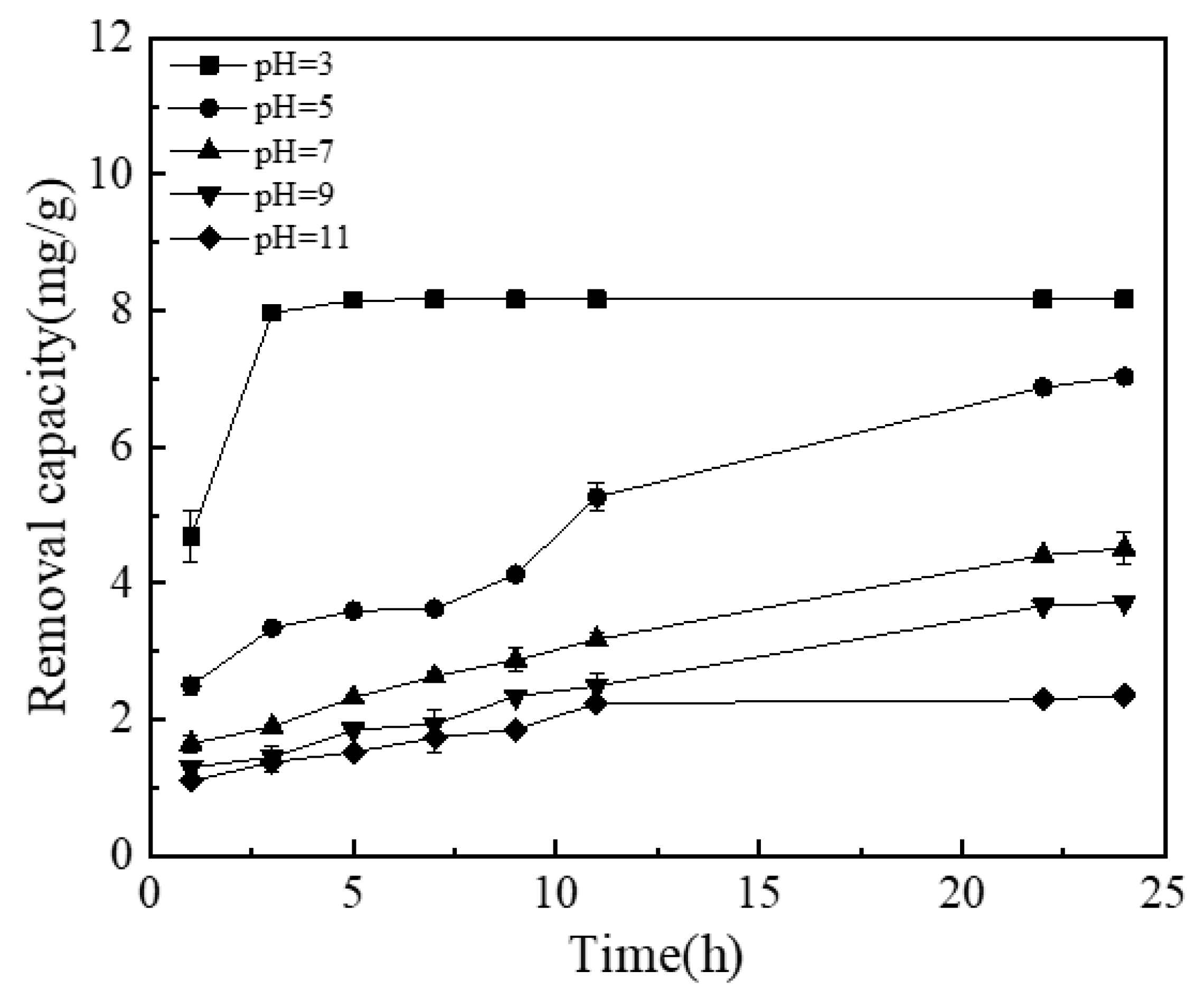
3.4.2. Effect of pH
3.5. Characterization of NICLS before and after the Reaction
3.5.1. Specific Surface Area of NICLS
3.5.2. SEM Analysis of NICLS
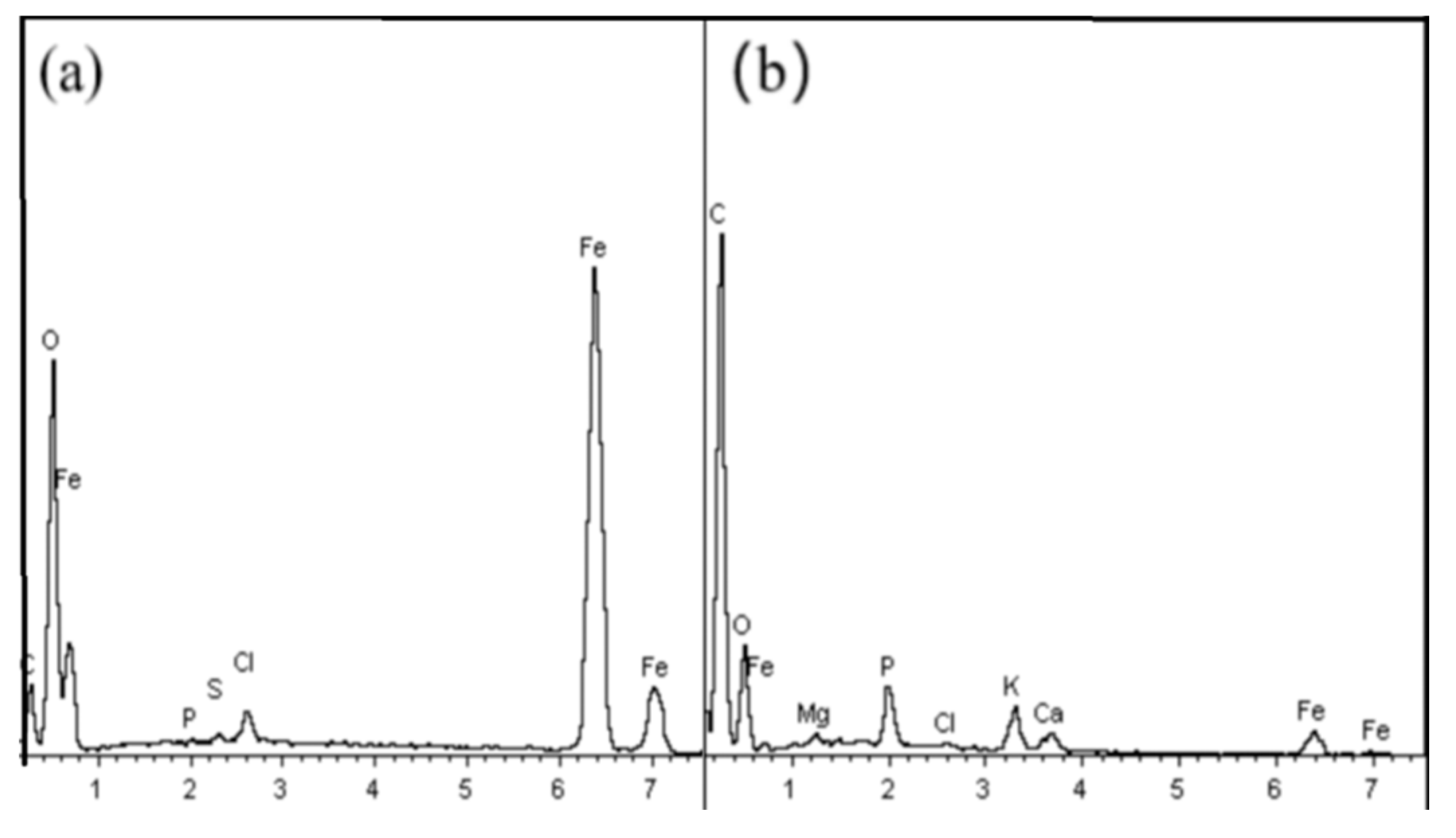
3.5.3. EDS Analysis of NICLS
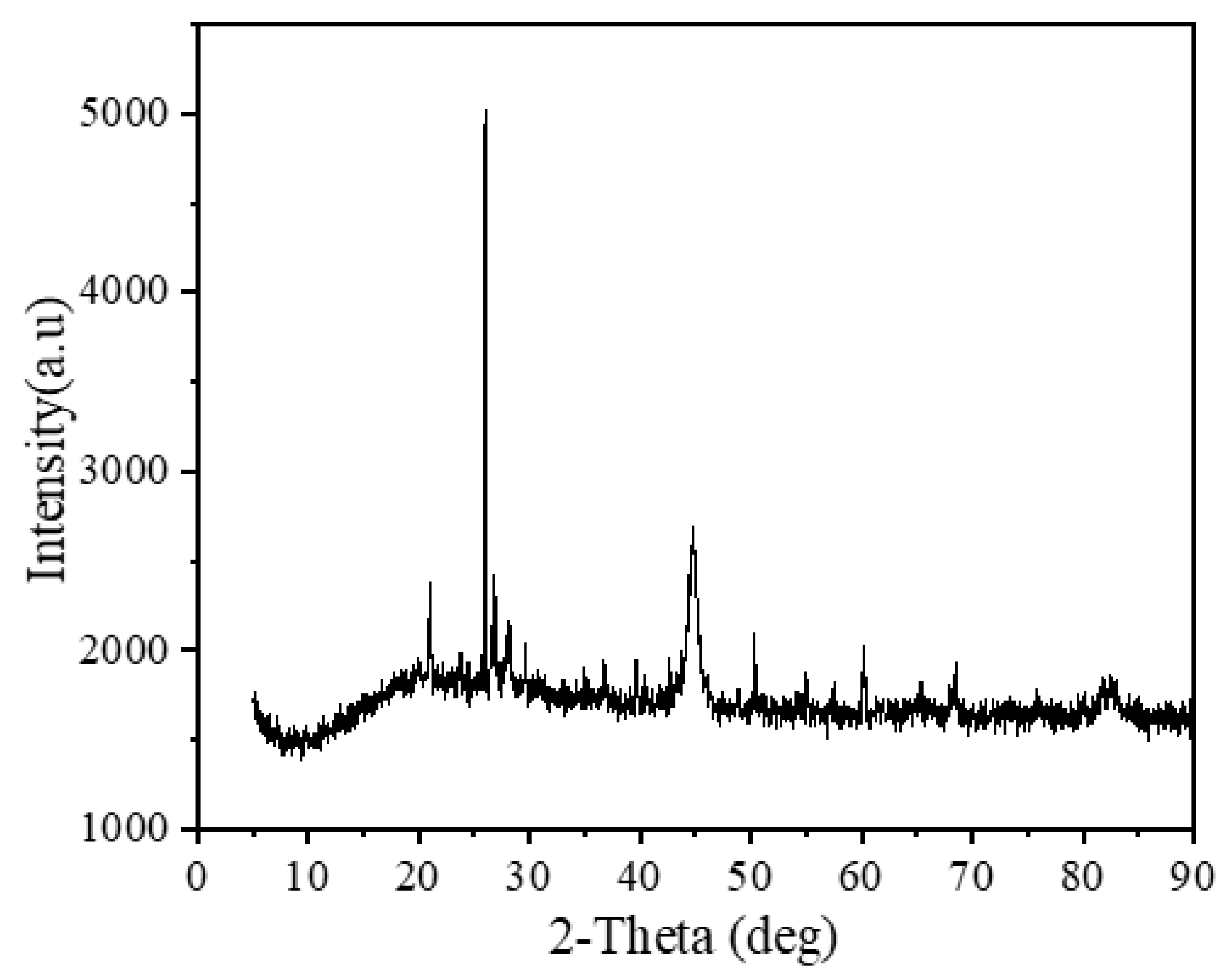
3.5.4. XRD Analysis of NICLS
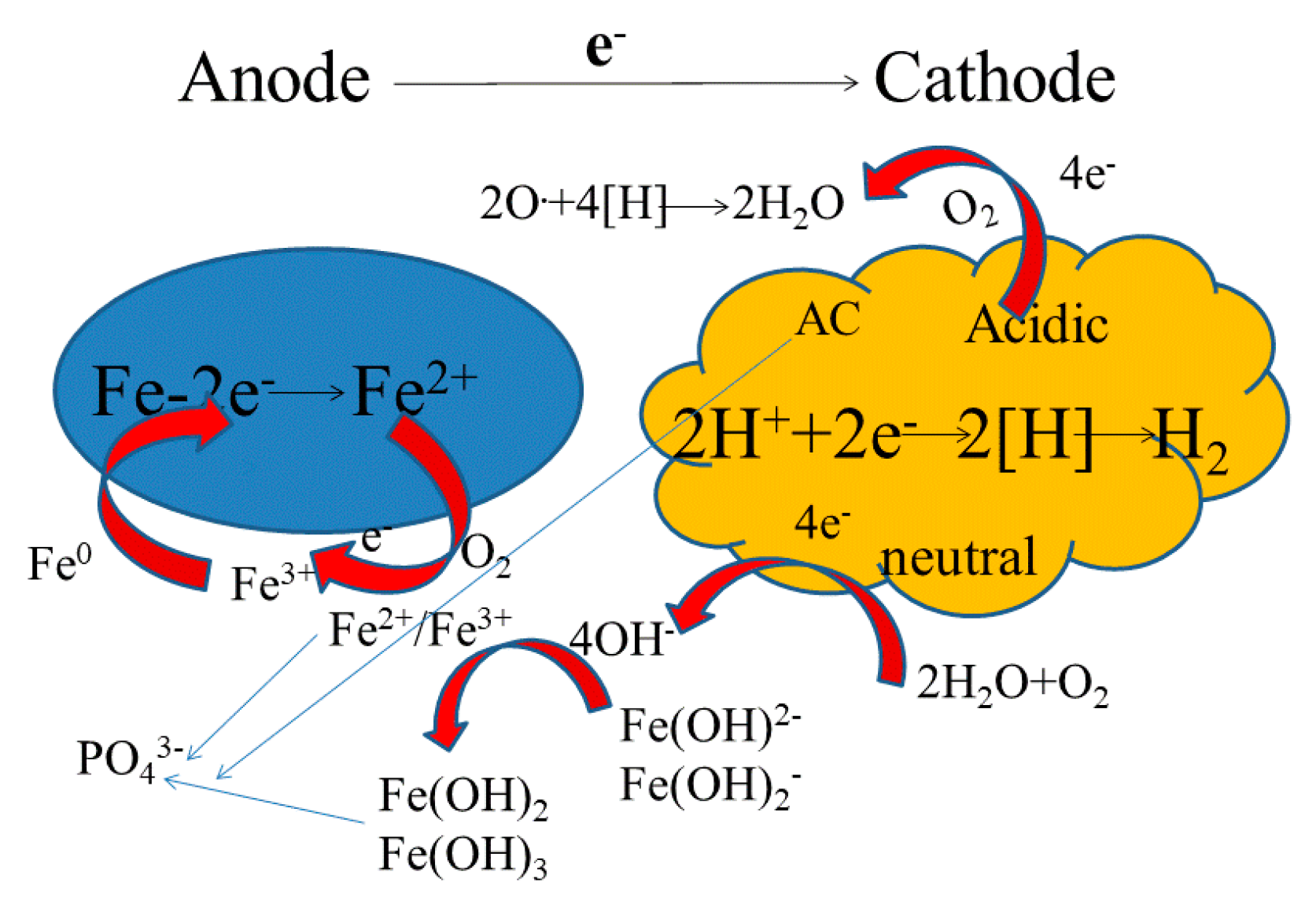
3.6. Mechanism Analysis of NICLS Phosphorus Removal
3.7. Benefit Analysis
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Liu, Y.; Liu, X.; Li, K.; Lu, S.; Guo, X.; Zhang, J.; Xi, B. Removal of nitrogen from low pollution water by long-term operation of an integrated vertical-flow constructed wetland: Performance and mechanism. Sci. Total Environ. 2019, 652, 977–988. [Google Scholar] [CrossRef] [PubMed]
- Guan, Q.; Feng, L.; Hou, X.; Schurgers, G.; Zheng, Y.; Tang, J. Eutrophication changes in fifty large lakes on the Yangtze Plain of China derived from MERIS and OLCI observations. Remote. Sens. Environ. 2020, 246, 111890. [Google Scholar] [CrossRef]
- Pathiratne, K.A.S.; De Silva, O.C.P.; Hehemann, D.; Atkinson, I.; Wei, R. Occurrence and Distribution of Polycyclic Aromatic Hydrocarbons (PAHs) in Bolgoda and Beira Lakes, Sri Lanka. Bull. Environ. Contam. Toxicol. 2007, 79, 135–140. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Li, Y.; Lv, D.; Li, Y.; Wu, J. Nitrogen and phosphorus removal performance and bacterial communities in a multi-stage surface flow constructed wetland treating rural domestic sewage. Sci. Total Environ. 2020, 709, 136235. [Google Scholar] [CrossRef]
- Li, S.; Wang, X.; Qiao, B.; Li, J.; Tu, J. First flush characteristics of rainfall runoff from a paddy field in the Taihu Lake watershed, China. Environ. Sci. Pollut. Res. 2017, 24, 8336–8351. [Google Scholar] [CrossRef]
- Bertanza, G.; Menoni, L.; Capoferri, G.U.; Pedrazzani, R. Promoting biological phosphorus removal in a full scale pre-denitrification wastewater treatment plant. J. Environ. Manag. 2020, 254, 109803. [Google Scholar] [CrossRef]
- Barat, R.; Montoya, T.; Seco, A.; Ferrer, J. Modelling biological and chemically induced precipitation of calcium phosphate in enhanced biological phosphorus removal systems. Water Res. 2011, 45, 3744–3752. [Google Scholar] [CrossRef]
- Santiviago Petzoldt, C.; Peralta Lezcano, J.; López Moreda, I. Removal of orthophosphate and dissolved organic phosphorus from synthetic wastewater in a combined struvite precipitation-adsorption system. J. Environ. Chem. Eng. 2020, 8, 103923. [Google Scholar] [CrossRef]
- Ji, B.; Zhu, L.; Wang, S.; Qin, H.; Ma, Y.; Liu, Y. A novel micro-ferrous dosing strategy for enhancing biological phosphorus removal from municipal wastewater. Sci. Total Environ. 2020, 704, 135453. [Google Scholar] [CrossRef]
- Jiang, C.; Jia, L.; He, Y.; Zhang, B.; Kirumba, G.; Xie, J. Adsorptive removal of phosphorus from aqueous solution using sponge iron and zeolite. J. Colloid Interface Sci. 2013, 402, 246–252. [Google Scholar] [CrossRef]
- Yin, H.; Yan, X.; Gu, X. Evaluation of thermally-modified calcium-rich attapulgite as a low-cost substrate for rapid phosphorus removal in constructed wetlands. Water Res. 2017, 115, 329–338. [Google Scholar] [CrossRef] [PubMed]
- Marchand, L.; Mench, M.; Jacob, D.L.; Otte, M.L. Metal and metalloid removal in constructed wetlands, with emphasis on the importance of plants and standardized measurements: A review. Environ. Pollut. 2010, 158, 3447–3461. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Yue, Q.; Yang, K.; Zhao, P.; Gao, B. Enhanced phosphorus and ciprofloxacin removal in a modified BAF system by configuring Fe-C micro electrolysis: Investigation on pollutants removal and degradation mechanisms. J. Hazard. Mater. 2018, 342, 705–714. [Google Scholar] [CrossRef] [PubMed]
- Hao, S.J.; Jiang, W.F.; Tian, W.; Zhang, Y.Z. Study on Coking Wastewater Treatment by Iron-Carbon Internal Electrolysis Method. Adv. Mater. Res. 2011, 356, 1938–1941. [Google Scholar] [CrossRef]
- Shen, Y.; Zhuang, L.; Zhang, J.; Fan, J.; Yang, T.; Sun, S. A study of ferric-carbon micro-electrolysis process to enhance nitrogen and phosphorus removal efficiency in subsurface flow constructed wetlands. Chem. Eng. J. 2019, 359, 706–712. [Google Scholar] [CrossRef]
- Ma, T.; Zhang, L.; Xi, B.; Xiong, Y.; Yu, P.; Li, G.; Li, J.; Zhao, C. Treatment of farmer household tourism wastewater using iron-carbon micro-electrolysis and horizontal subsurface flow constructed wetlands: A full-scale study. Ecol. Eng. 2018, 110, 192–203. [Google Scholar] [CrossRef]
- Zheng, X.; Jin, M.; Zhou, X.; Chen, W.; Lu, D.; Zhang, Y.; Shao, X. Enhanced removal mechanism of iron carbon micro-electrolysis constructed wetland on C, N, and P in salty permitted effluent of wastewater treatment plant. Sci. Total Environ. 2019, 649, 21–30. [Google Scholar] [CrossRef]
- Cai, L.; Zhang, H.; Feng, Y.; Dong, B.; Wang, Y.; Ge, C. Iron and carbon granules added to anode enhanced the sludge decrement and electrical performance of sludge microbial fuel cell. Chem. Eng. J. 2019, 372, 572–580. [Google Scholar] [CrossRef]
- Zhou, Z.; Qiao, W.; Lin, Y.; Shen, X.; Hu, D.; Zhang, J.; Jiang, L.-M.; Wang, L. Phosphonate removal from discharged circulating cooling water using iron–carbon micro-electrolysis. Water Sci. Technol. 2014, 70, 524–532. [Google Scholar] [CrossRef]
- Shi, Z.; Liu, F.; Yao, S. Adsorptive removal of phosphate from aqueous solutions using activated carbon loaded with Fe (III) oxide. New Carbon Mater. 2011, 26, 299–306. [Google Scholar] [CrossRef]
- Cheng, G.; Li, Q.; Su, Z.; Sheng, S.; Fu, J. Preparation, optimization, and application of sustainable ceramsite substrate from coal fly ash/waterworks sludge/oyster shell for phosphorus immobilization in constructed wetlands. J. Clean. Prod. 2018, 175, 572–581. [Google Scholar] [CrossRef]
- Zhou, H.; Lv, P.; Shen, Y.; Wang, J.; Fan, J. Identification of degradation products of ionic liquids in an ultrasound assisted zero-valent iron activated carbon micro-electrolysis system and their degradation mechanism. Water Res. 2013, 47, 3514–3522. [Google Scholar] [CrossRef] [PubMed]
- Luo, J.; Song, G.; Liu, J.; Qian, G.; Xu, Z.P. Mechanism of enhanced nitrate reduction via micro-electrolysis at the powdered zero-valent iron/activated carbon interface. J. Colloid Interface Sci. 2014, 435, 21–25. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.; Zhao, J.; Zhang, J.; Li, Q.; Gao, J.; Cai, M.; Zhang, J. Preparation of a new low-cost substrate prepared from drinking water treatment sludge (DWTS)/bentonite/zeolite/fly ash for rapid phosphorus removal in constructed wetlands. J. Clean. Prod. 2020, 261, 121110. [Google Scholar] [CrossRef]
- Liu, B.; Liu, L.; Li, W. Effective removal of phosphorus from eutrophic water by using cement. Environ. Res. 2020, 183, 109218. [Google Scholar] [CrossRef]
- Rocha, D.L.; Rocha, F.R.P. Photochemical micro-digestion in a multi-pumping flow system for phosphorus fractionation in cereals. Microchem. J. 2013, 109, 139–144. [Google Scholar] [CrossRef]
- Bao, T.; Chen, T.; Tan, J.; Wille, M.; Zhu, D.; Chen, D.; Xi, Y. Synthesis and performance of iron oxide-based porous ceramsite in a biological aerated filter for the simultaneous removal of nitrogen and phosphorus from domestic wastewater. Sep. Purif. Technol. 2016, 167, 154–162. [Google Scholar] [CrossRef]
- Sizirici, B.; Yildiz, I. Simultaneous removal of organics and metals in fixed bed using gravel and iron oxide coated gravel. Results Eng. 2020, 5, 100093. [Google Scholar] [CrossRef]
- Zhu, X.; Song, T.; Lv, Z.; Ji, G. High-efficiency and low-cost α-Fe 2 O 3 nanoparticles-coated volcanic rock for Cd (II) removal from wastewater. Process. Saf. Environ. Prot. 2016, 104, 373–381. [Google Scholar] [CrossRef]
- Park, T.; Ampunan, V.; Maeng, S.; Chung, E. Application of steel slag coated with sodium hydroxide to enhance precipitation-coagulation for phosphorus removal. Chemosphere 2017, 167, 91–97. [Google Scholar] [CrossRef]
- Jiang, C.; Jia, L.; Zhang, B.; He, Y.; Kirumba, G. Comparison of quartz sand, anthracite, shale and biological ceramsite for adsorptive removal of phosphorus from aqueous solution. J. Environ. Sci. 2014, 26, 466–477. [Google Scholar] [CrossRef]
- Kuzawa, K.; Jung, Y.; Kiso, Y.; Yamada, T.; Nagai, M.; Lee, T. Phosphate removal and recovery with a synthetic hydrotalcite as an adsorbent. Chemosphere 2006, 62, 45–52. [Google Scholar] [CrossRef]
- Wu, F.; Yu, Q.; Gauvin, F.; Brouwers, H.J.H.; Liu, C. Phosphorus removal from aqueous solutions by adsorptive concrete aggregates. J. Clean. Prod. 2021, 278, 123933. [Google Scholar] [CrossRef]
- Gu, S.; Fu, B.; Ahn, J.; Fang, B. Mechanism for phosphorus removal from wastewater with fly ash of municipal solid waste incineration, Seoul, Korea. J. Clean. Prod. 2021, 280, 124430. [Google Scholar] [CrossRef]
- Song, X.; Pan, Y.; Wu, Q.; Cheng, Z.; Ma, W. Phosphate removal from aqueous solutions by adsorption using ferric sludge. Desalination 2011, 280, 384–390. [Google Scholar] [CrossRef]
- Papoulis, D.; Kordouli, E.; Lampropoulou, P.; Rapsomanikis, A.; Kordulis, C.; Panagiotaras, D.; Theophylaktou, K.; Stathatos, E.; Komarneni, S. Synthesis, Characterization and Photocatalytic Activities of Fly Ash-TiO2 Nanocomposites for the Mineralization of Azo Dyes in Water. J. Surfaces Interfaces Mater. 2014, 2, 261–266. [Google Scholar] [CrossRef]
- Li, T.; Tian, H.; Chen, J.; Cheng, L. Application of low pressure gas adsorption to the characterization of pore size distribution of shales: An example from Southeastern Chongqing area, China. J. Nat. Gas Geosci. 2016, 1, 221–230. [Google Scholar] [CrossRef]
- Lin, H.T.; Ferber, M.K.; Becher, P.F.; Price, J.R.; van Roode, M.; Kimmel, J.B.; Jimenez, O.D. Characterization of First-Stage Silicon Nitride Components After Exposure to an Industrial Gas Turbine. J. Am. Ceram. Soc. 2006, 89, 258–265. [Google Scholar] [CrossRef]
- Noh, B.; Yoon, J.; Choi, J.; Jung, S. Effect of thermal treatment on adhesion strength of Cu/Ni–Cr/polyimide flexible copper clad laminate fabricated by roll-to-roll process. Microelectron. Eng. 2011, 88, 718–723. [Google Scholar] [CrossRef]
- Li, M.; Zheng, M.; Zhou, T.; Li, C.; Ma, L.; Shen, W. Fabrication and centeracterization of ordered CuIn(1−x) GaxSe2 nanopore films via template-based electrodeposition. Nanoscale Res. Lett. 2012, 7, 675. [Google Scholar] [CrossRef]
- Wu, X.; Lv, C.; Yu, S.; Li, M.; Ye, J.; Zhang, X.; Liu, Y. Uranium (VI) removal from aqueous solution using iron-carbon micro-electrolysis packing. Sep. Purif. Technol. 2020, 234, 116104. [Google Scholar] [CrossRef]
- Zhu, X.; Chen, X.; Yang, Z.; Liu, Y.; Zhou, Z.; Ren, Z. Investigating the influences of electrode material property on degradation behavior of organic wastewaters by iron-carbon micro-electrolysis. Chem. Eng. J. 2018, 338, 46–54. [Google Scholar] [CrossRef]
- Li, M.; Liu, H.; Chen, T.; Chen, D.; Wang, C.; Wei, L.; Wang, L. Efficient U(VI) adsorption on iron/carbon composites derived from the coupling of cellulose with iron oxides: Performance and mechanism. Sci. Total Environ. 2020, 703, 135604. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Ma, Y.; Liu, Y.; Li, Q.; Zhou, Z.; Ren, Z. Degradation of organic pollutants in near-neutral pH solution by Fe-C micro-electrolysis system. Chem. Eng. J. 2017, 315, 403–414. [Google Scholar] [CrossRef]
- Dou, X.; Li, R.; Zhao, B.; Liang, W. Arsenate removal from water by zero-valent iron/activated carbon galvanic couples. J. Hazard. Mater. 2010, 182, 108–114. [Google Scholar] [CrossRef] [PubMed]
- Arias, M.; Da Silva-Carballal, J.; García-Río, L.; Mejuto, J.; Núñez, A. Retention of phosphorus by iron and aluminum-oxides-coated quartz particles. J. Colloid Interface Sci. 2006, 295, 65–70. [Google Scholar] [CrossRef]
- Chen, F.; Li, H.; Jia, X.; Wang, Z.; Liang, X.; Qin, Y.; Chen, W.; Ao, T. Characteristic and model of phosphate adsorption by activated carbon electrodes in capacitive deionization. Sep. Purif. Technol. 2020, 236, 116285. [Google Scholar] [CrossRef]
- Sun, Z.; Xu, Z.; Zhou, Y.; Zhang, D.; Chen, W. Effects of different scrap iron as anode in Fe-C micro-electrolysis system for textile wastewater degradation. Environ. Sci. Pollut. Res. 2019, 26, 26869–26882. [Google Scholar] [CrossRef]
- Deng, S.; Li, D.; Yang, X.; Xing, W.; Li, J.; Zhang, Q. Iron [Fe (0)]-rich substrate based on iron–carbon micro–electrolysis for phosphorus adsorption in aqueous solutions. Chemosphere 2017, 168, 1486–1493. [Google Scholar] [CrossRef]
- Wen, Z.; Zhang, Y.; Dai, C. Removal of phosphate from aqueous solution using nanoscale zerovalent iron (nZVI). Colloids Surfaces A Physicochem. Eng. Asp. 2014, 457, 433–440. [Google Scholar] [CrossRef]
- Han, Y.; Li, H.; Liu, M.; Sang, Y.; Liang, C.; Chen, J. Purification treatment of dyes wastewater with a novel micro-electrolysis reactor. Sep. Purif. Technol. 2016, 170, 241–247. [Google Scholar] [CrossRef]
- Fida, H.; Guo, S.; Zhang, G. Preparation and characterization of bifunctional Ti–Fe kaolinite composite for Cr (VI) removal. J. Colloid Interface Sci. 2015, 442, 30–38. [Google Scholar] [CrossRef] [PubMed]
- Lai, B.; Zhou, Y.; Yang, P.; Yang, J.; Wang, J. Degradation of 3,3′-iminobis-propanenitrile in aqueous solution by Fe0/GAC micro-electrolysis system. Chemosphere 2013, 90, 1470–1477. [Google Scholar] [CrossRef]
- Che, J.; Wan, J.; Huang, X.; Wu, R.; Liang, K. Pretreatment of piggery digestate wastewater by ferric-carbon micro-electrolysis under alkalescence condition. Korean J. Chem. Eng. 2017, 34, 2397–2405. [Google Scholar] [CrossRef]
- Hou, Q.; Meng, P.; Pei, H.; Hu, W.; Chen, Y. Phosphorus adsorption characteristics of alum sludge: Adsorption capacity and the forms of phosphorus retained in alum sludge. Mater. Lett. 2018, 229, 31–35. [Google Scholar] [CrossRef]
- Wu, L.; Liao, L.; Lv, G.; Qin, F.; He, Y.; Wang, X. Micro-electrolysis of Cr (VI) in the nanoscale zero-valent iron loaded activated carbon. J. Hazard. Mater. 2013, 254, 277–283. [Google Scholar] [CrossRef] [PubMed]
- Peng, S.; Deng, S.; Li, D.; Xie, B.; Yang, X.; Lai, C.; Sun, S.; Yao, H. Iron-carbon galvanic cells strengthened anaerobic/anoxic/oxic process (Fe/C-A2O) for high-nitrogen/phosphorus and low-carbon sewage treatment. Sci. Total Environ. 2020, 722, 137657. [Google Scholar] [CrossRef]
- Zhang, Z.; Feng, Y.; Liu, N.; Zhao, Y.; Wang, X.; Yang, S.; Long, Y.; Qiu, L. Preparation of Sn/Mn loaded steel slag zeolite particle electrode and its removal effect on rhodamine B(RhB). J. Water Process. Eng. 2020, 37, 101417. [Google Scholar] [CrossRef]

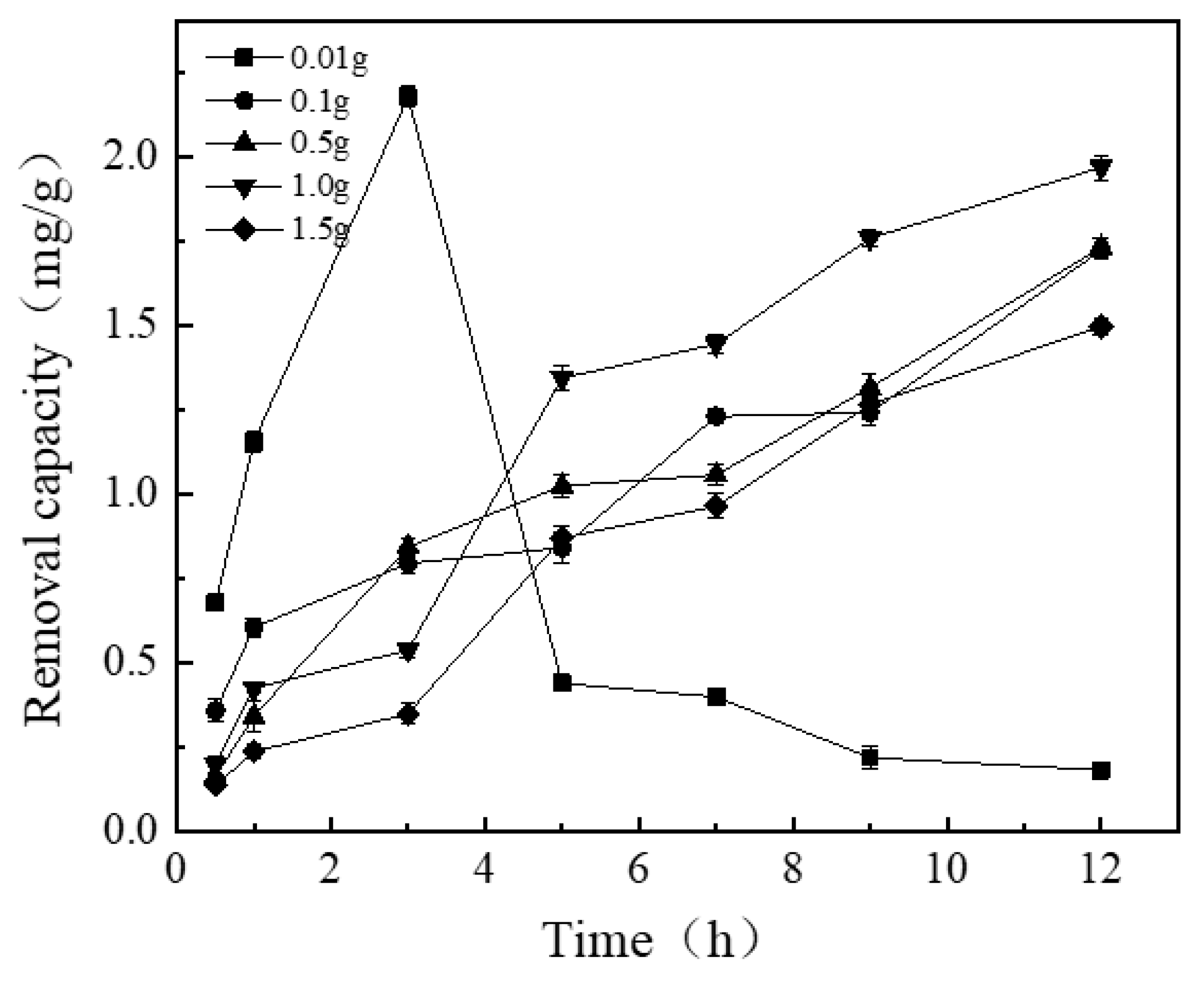
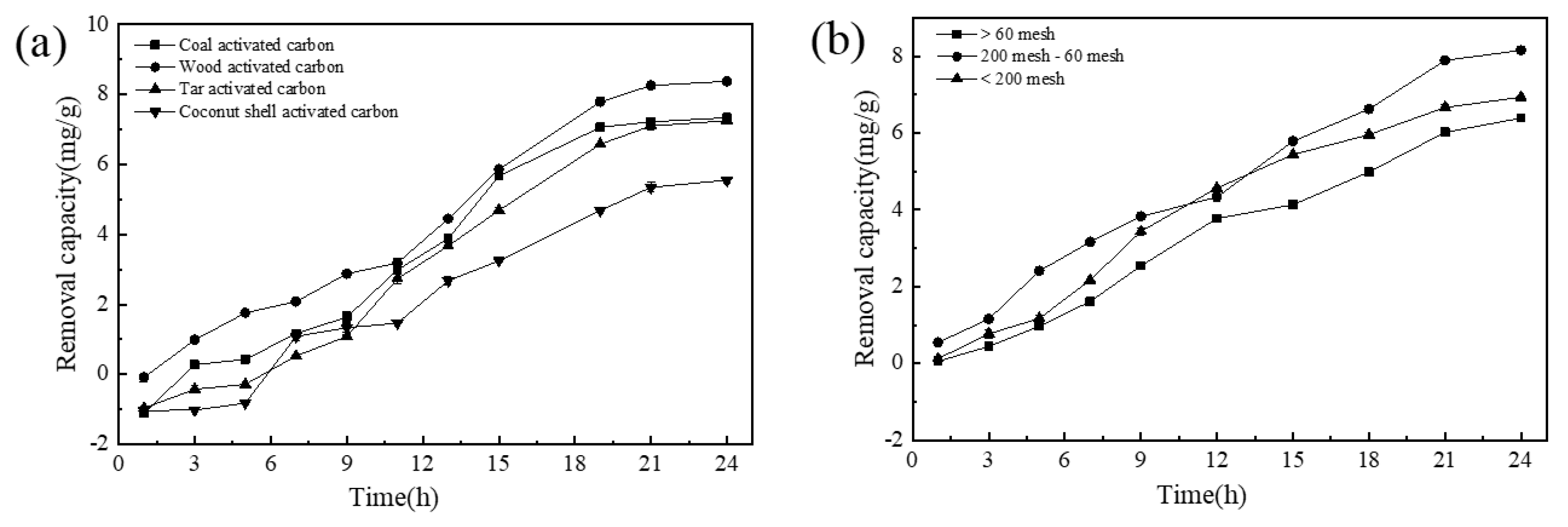

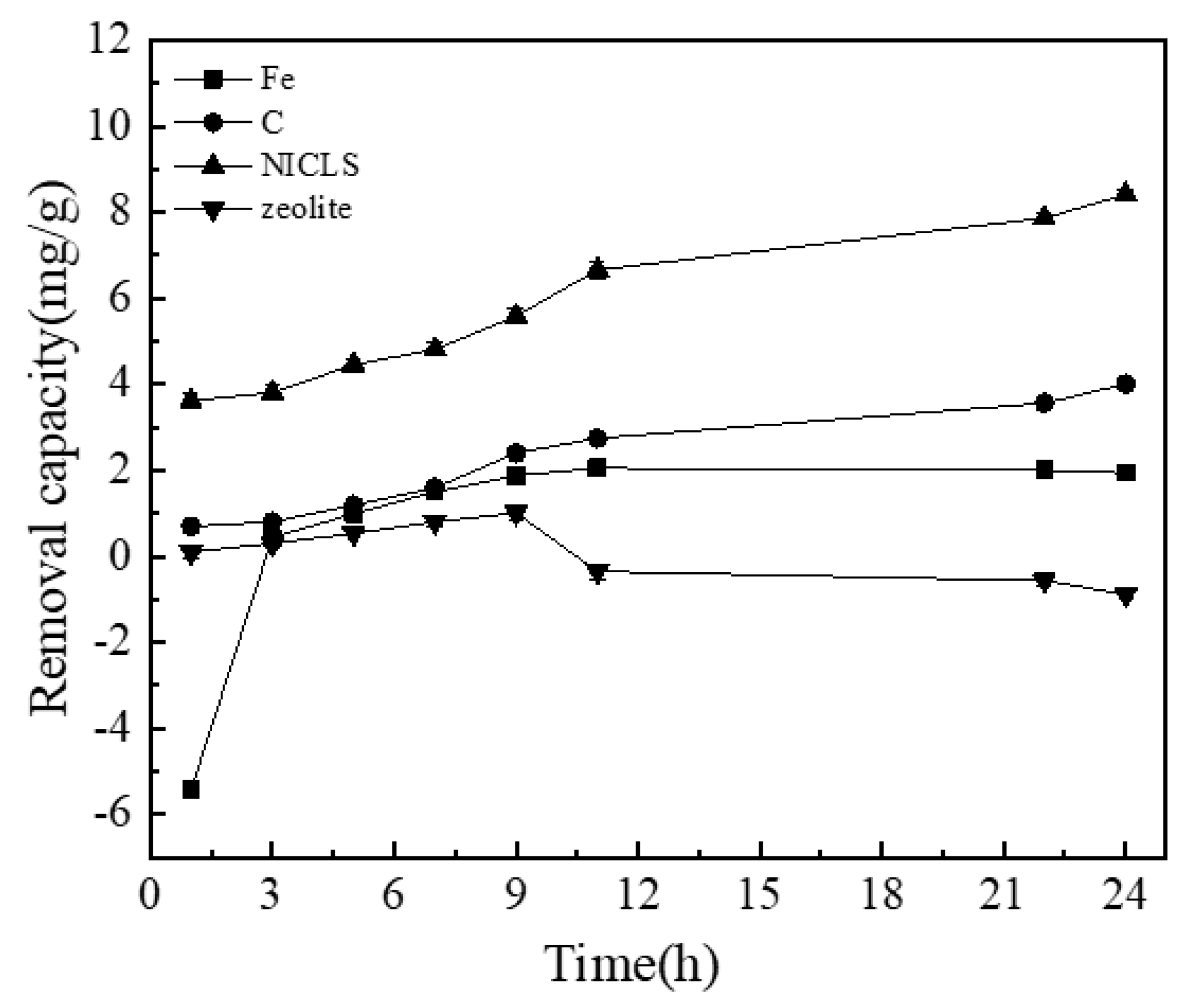
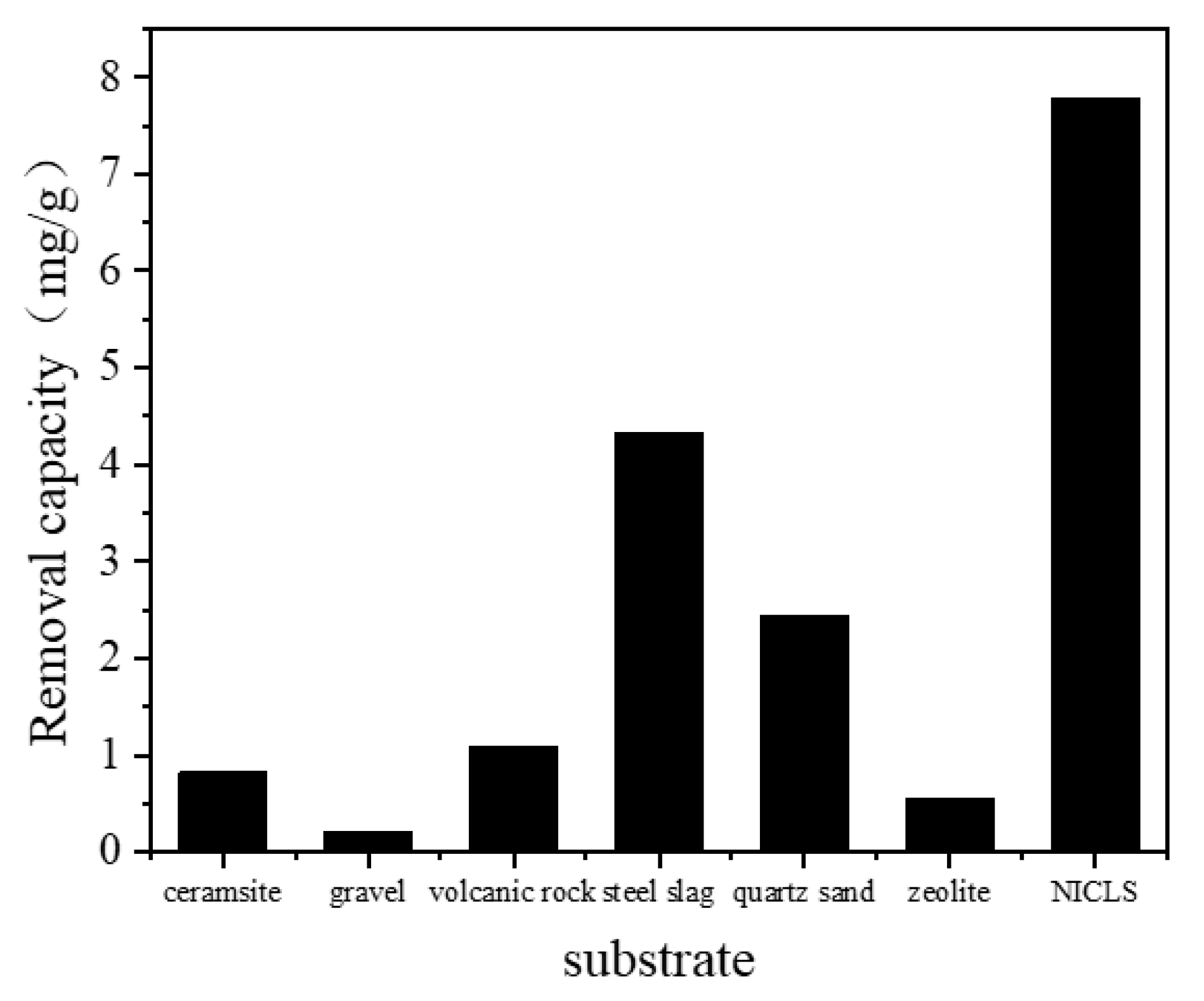


| Time | 0.01 | 0.10 | 0.50 | 1.00 | 1.50 |
|---|---|---|---|---|---|
| 5 h | A | B | C | D | B |
| 7 h | A | B | C | D | E |
| 9 h | A | B | B | C | B |
| 11 h | A | C | C | D | B |
| Time | Coal Activated Carbon | Wood Activated Carbon | Tar Activated Carbon | Coconut Shell Activated Carbon |
|---|---|---|---|---|
| 1 h | A | B | C | C |
| 3 h | C | D | B | A |
| 5 h | C | D | B | A |
| 9 h | C | D | A | B |
| 11 h | C | D | B | A |
| 15 h | C | D | B | A |
| 21 h | B | D | B | A |
| 24 h | B | D | B | A |
| Time | >60 Mesh | 200 Mesh-60 Mesh | <200 Mesh |
|---|---|---|---|
| 1 h | A | B | A |
| 3 h | A | C | B |
| 5 h | A | C | B |
| 7 h | A | C | B |
| 9 h | A | C | B |
| 12 h | A | B | C |
| 15 h | A | C | B |
| 18 h | A | C | B |
| 21 h | A | C | B |
| 24 h | A | C | B |
| Time | Fe/C = 1:4 | Fe/C = 1:3 | Fe/C = 1:2 | Fe/C = 1:1 | Fe/C = 2:1 | Fe/C = 3:1 | Fe/C = 4:1 |
|---|---|---|---|---|---|---|---|
| 1 h | B | B | B | C | B | B | A |
| 6 h | A | A | B | D | C | C | B |
| 12 h | A | B | D | E | D | C | B |
| 18 h | A | C | D | E | C | C | B |
| 24 h | A | A | B | C | B | A | A |
| Time | Fe | C | NICLS | Zeolite |
|---|---|---|---|---|
| 1 h | A | C | D | B |
| 3 h | A | B | C | A |
| 5 h | B | B | C | A |
| 7 h | B | B | C | A |
| 9 h | B | C | D | A |
| 11 h | B | C | D | A |
| 22 h | B | C | D | A |
| 24 h | B | C | D | A |
| Specific Surface Area (before) | Specific Surface Area (after) | Pore Volume | Aperture |
|---|---|---|---|
| 2.6231 m2/g | 3.7107 m2/g | 0.6037 cm3/g | 8.4845 nm |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, J.; Gao, J.; Liu, J. Preparation of a New Iron-Carbon-Loaded Constructed Wetland Substrate and Enhanced Phosphorus Removal Performance. Materials 2020, 13, 4739. https://doi.org/10.3390/ma13214739
Zhao J, Gao J, Liu J. Preparation of a New Iron-Carbon-Loaded Constructed Wetland Substrate and Enhanced Phosphorus Removal Performance. Materials. 2020; 13(21):4739. https://doi.org/10.3390/ma13214739
Chicago/Turabian StyleZhao, Jie, Jingqing Gao, and Junzhao Liu. 2020. "Preparation of a New Iron-Carbon-Loaded Constructed Wetland Substrate and Enhanced Phosphorus Removal Performance" Materials 13, no. 21: 4739. https://doi.org/10.3390/ma13214739
APA StyleZhao, J., Gao, J., & Liu, J. (2020). Preparation of a New Iron-Carbon-Loaded Constructed Wetland Substrate and Enhanced Phosphorus Removal Performance. Materials, 13(21), 4739. https://doi.org/10.3390/ma13214739





