Cytotoxicity and Gene Expression Changes of a Novel Homeopathic Antiseptic Oral Rinse in Comparison to Chlorhexidine in Gingival Fibroblasts
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents and Cells
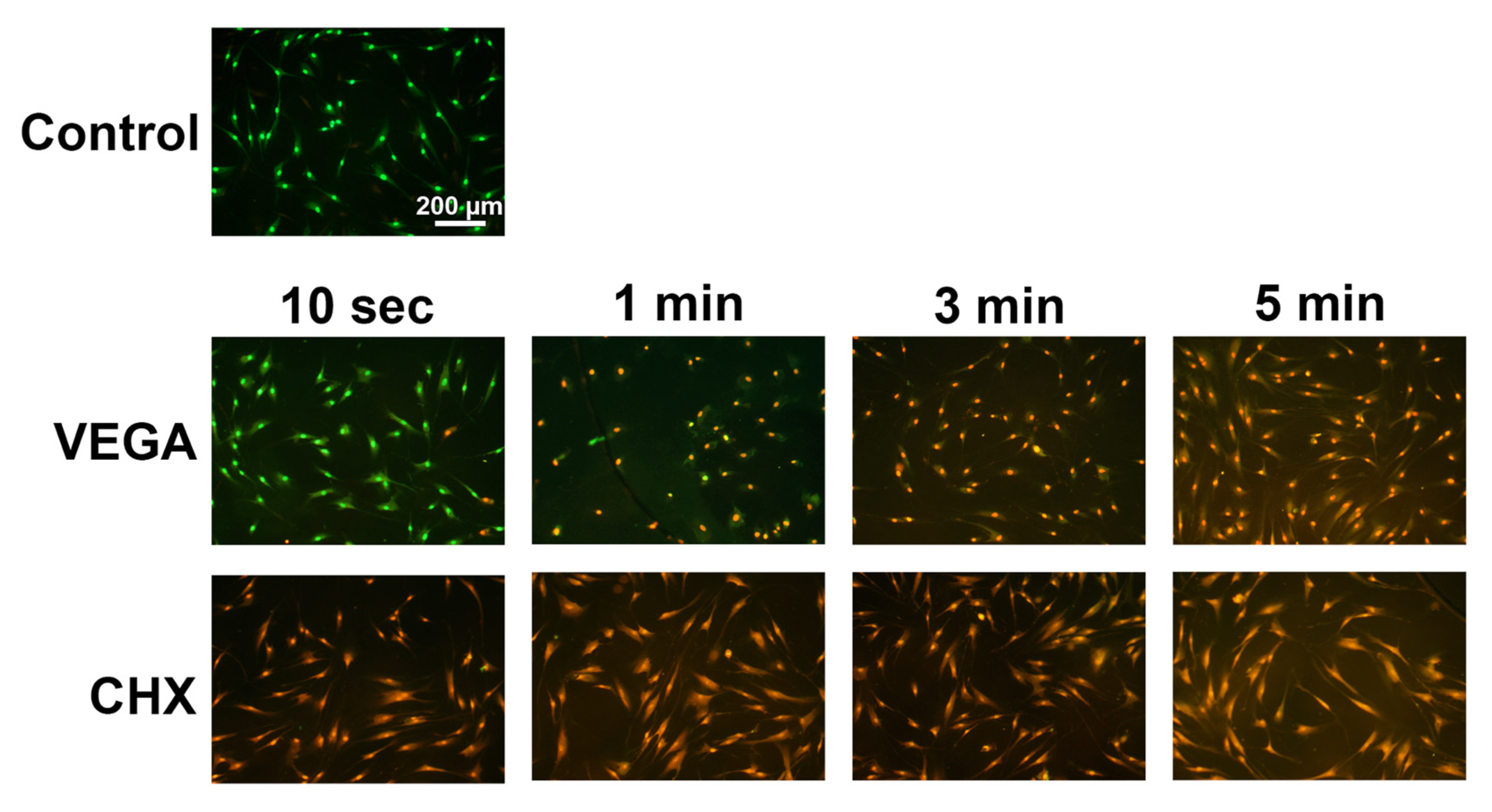
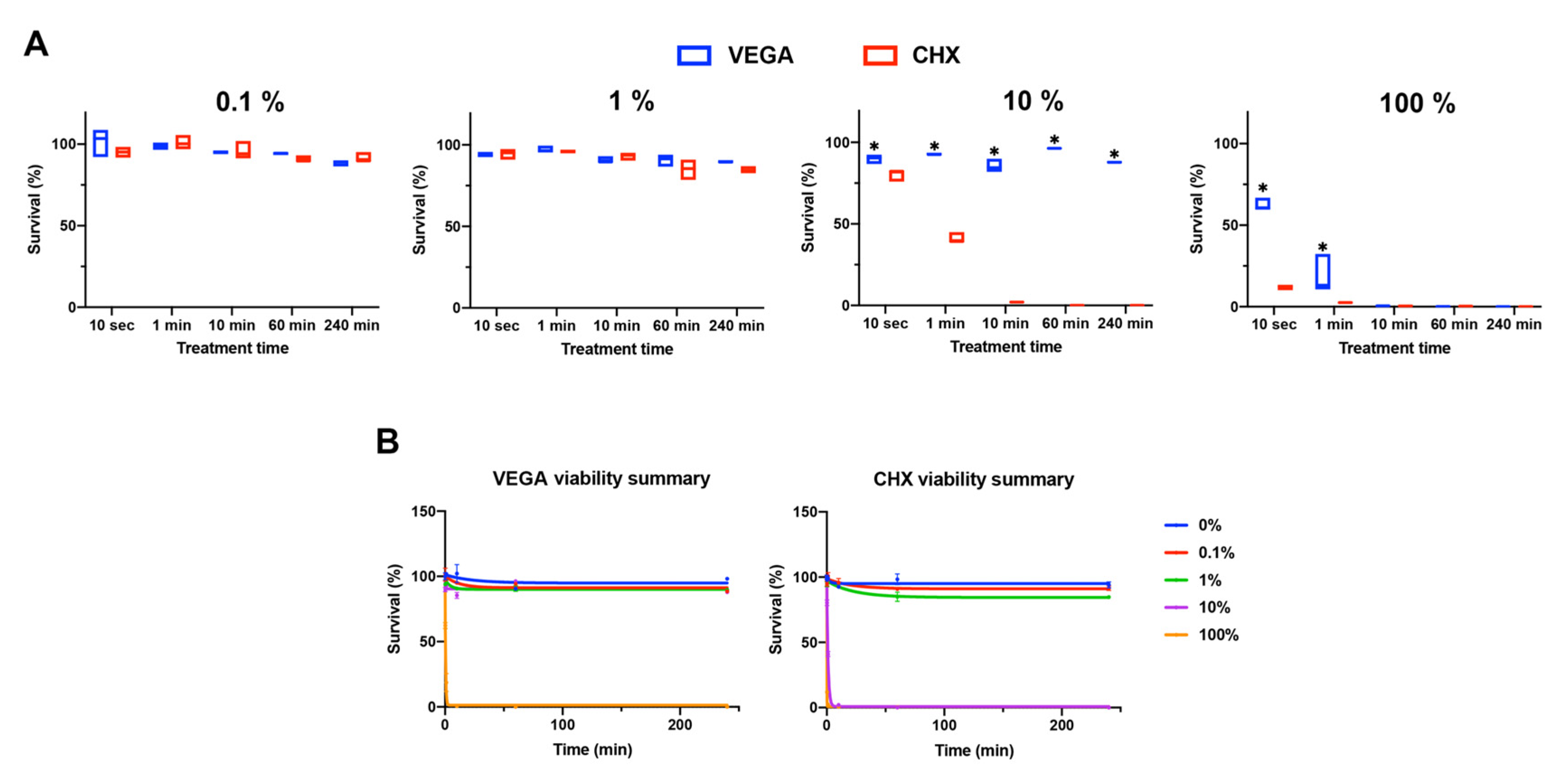
2.2. Cell Viability Assay
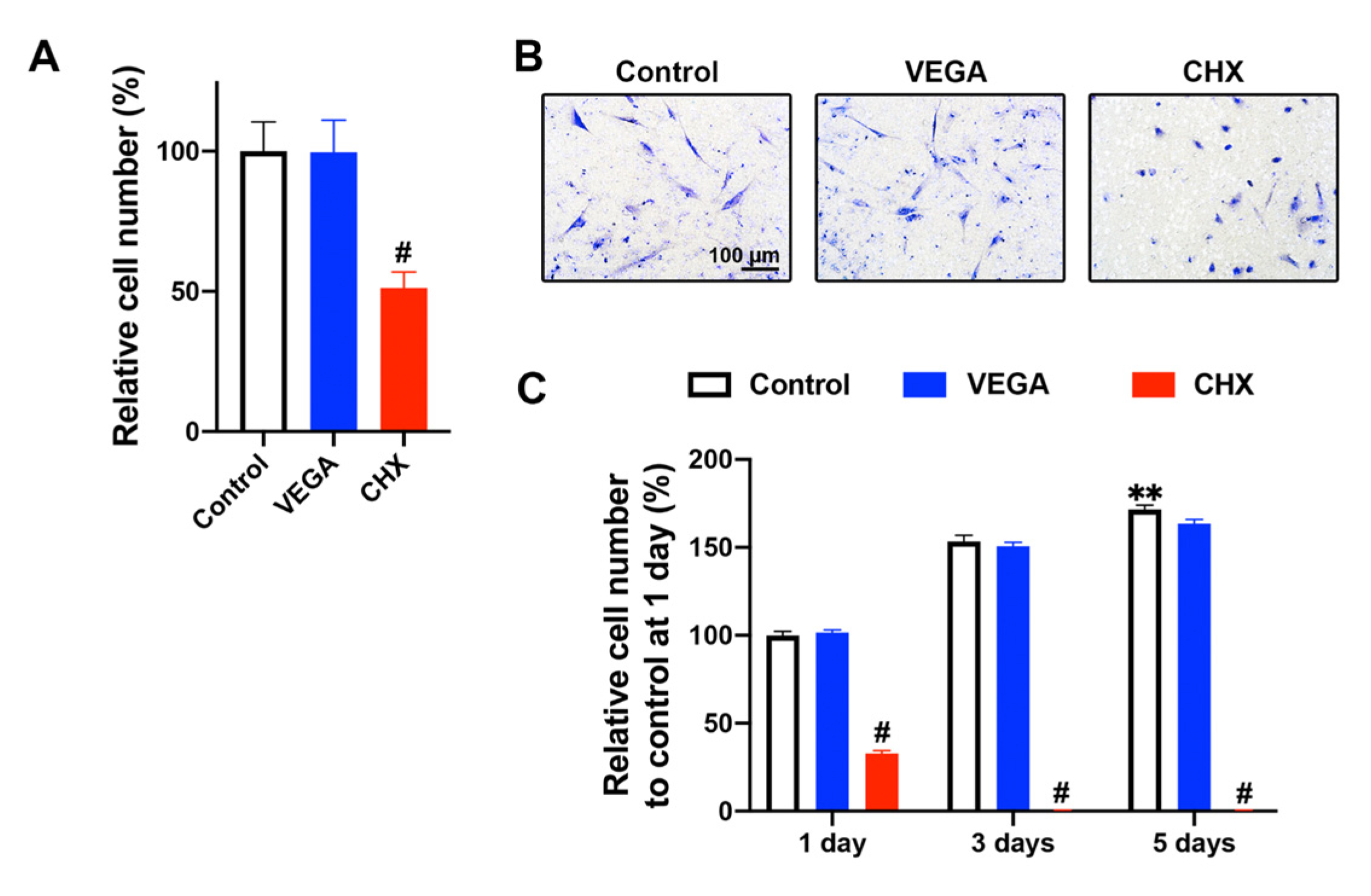
2.3. Cell Migration Assay
2.4. Proliferation Assay
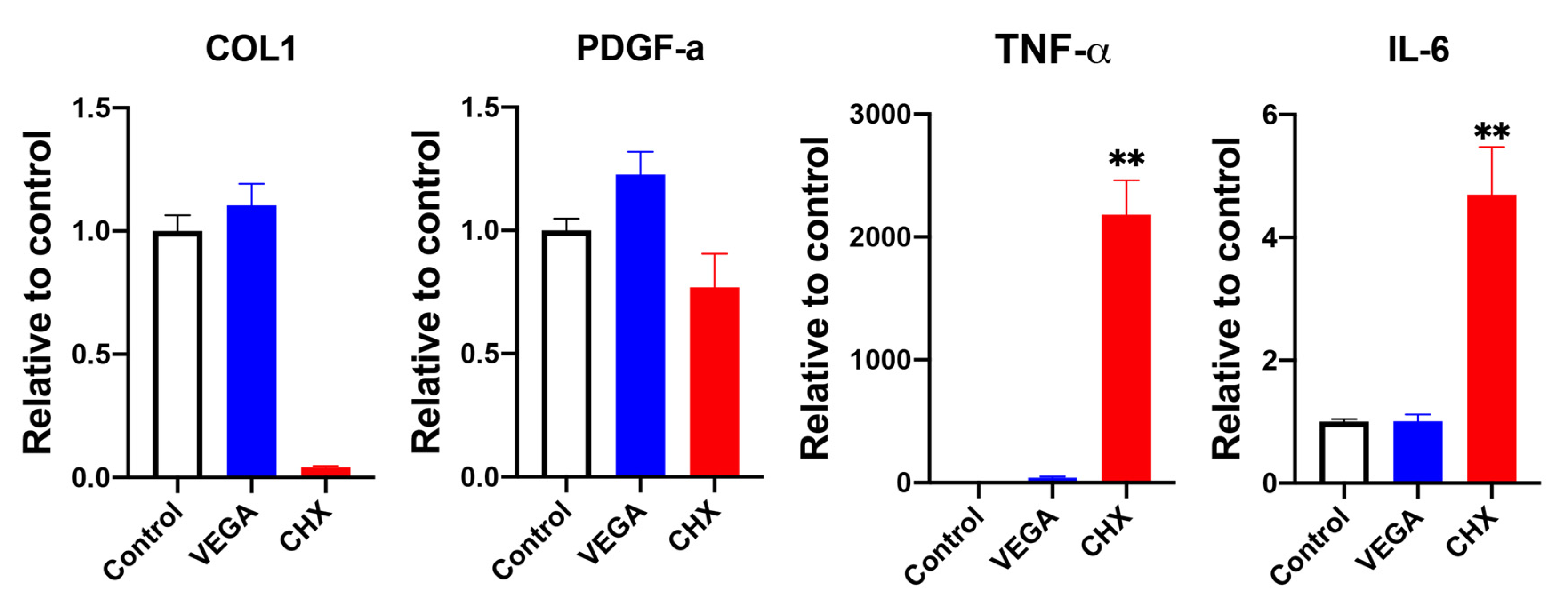
2.5. Real-Time PCR Analysis
2.6. Collagen Immunofluorescent Staining
2.7. Statistical Analysis
3. Results
3.1. Cytotoxity of VEGA and CHX on Human Gingival Fibroblasts
3.2. Influence of VEGA and CHX on Human Gingival Fibroblast Migration and Proliferation
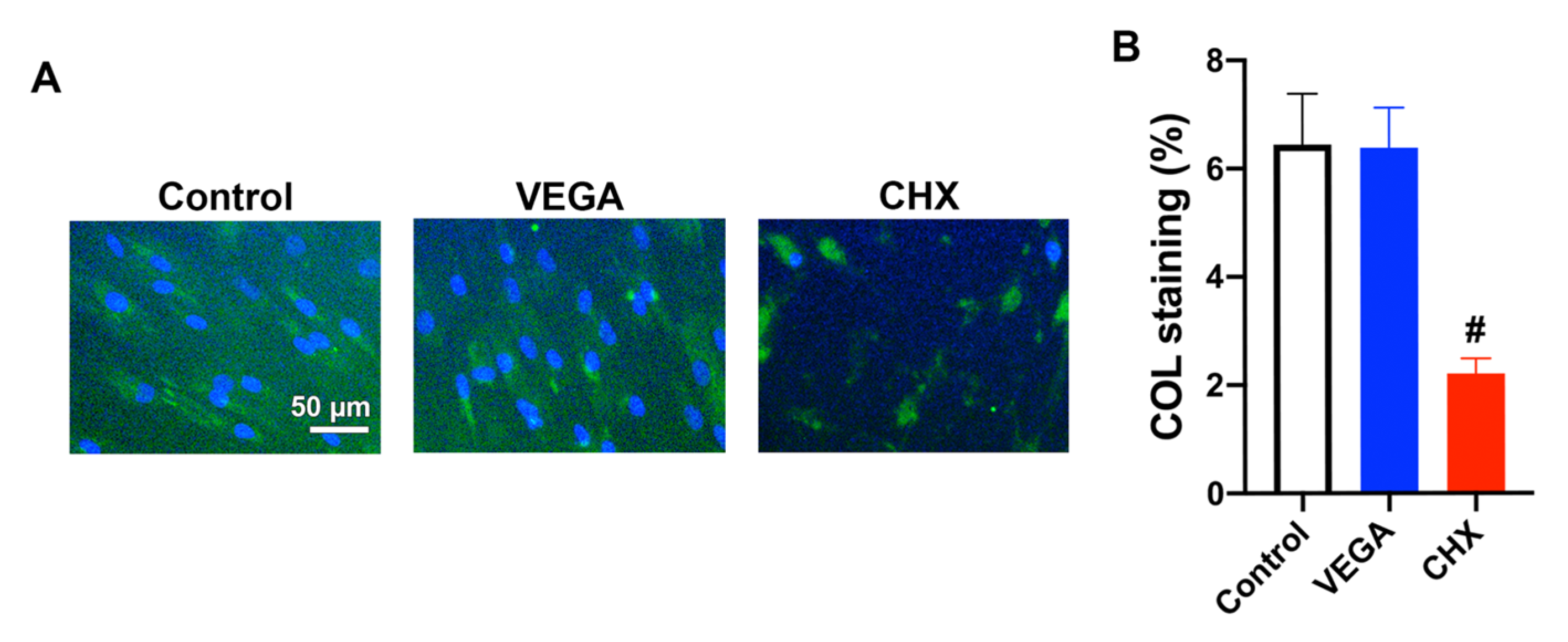
3.3. Influence of VEGA and CHX on Human Gingival Fibroblast Gene Expressions and Collagen Synthesis
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Rudd, R.A.; Aleshire, N.; Zibbell, J.E.; Matthew Gladden, R. Increases in drug and opioid overdose deaths—United States, 2000–2014. Am. J. Transplant. 2016, 16, 1323–1327. [Google Scholar] [CrossRef]
- National Institute of Drug Abuse. Overdose Death Rates; National Institute on Drug Abuse, National Institutes of Health: Hyattsville, MD, USA, 2017.
- Lee, C.Y.; Suzuki, J.B. The efficacy of preemptive analgesia using a non-opioid alternative therapy regimen on postoperative analgesia following block bone graft surgery of the mandible: A prospective pilot study in pain management in response to the opioid epidemic. Clin. J. Pharmacol. Pharmacother. 2019, 1, 1006. [Google Scholar]
- Tatch, W. Opioid prescribing can be reduced in oral and maxillofacial surgery practice. J. Oral Maxillofac. Surg. 2019, 77, 1771–1775. [Google Scholar] [CrossRef] [PubMed]
- Hermesch, C.B.; Hilton, T.J.; Biesbrock, A.R.; Baker, R.A.; Cain-Hamlin, J.; McClanahan, S.F.; Gerlach, R.W. Perioperative use of 0.12% chlorhexidine gluconate for the prevention of alveolar osteitis: Efficacy and risk factor analysis. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 1998, 85, 381–387. [Google Scholar] [CrossRef]
- Rosella, D.; Papi, P.; Giardino, R.; Cicalini, E.; Piccoli, L.; Pompa, G. Medication-related osteonecrosis of the jaw: Clinical and practical guidelines. J. Int. Soc. Prev. Community Dent. 2016, 6, 97. [Google Scholar]
- Curi, M.M.; Lauria, L. Osteoradionecrosis of the jaws: A retrospective study of the background factors and treatment in 104 cases. J. Oral Maxillofac. Surg. 1997, 55, 540–544. [Google Scholar] [CrossRef]
- Rodríguez-Caballero, A.; Torres-Lagares, D.; Robles-García, M.; Pachón-Ibáñez, J.; Gonzalez-Padilla, D.; Gutierrez-Perez, J. Cancer treatment-induced oral mucositis: A critical review. Int. J. Oral. Maxillofac. Surg. 2012, 41, 225–238. [Google Scholar] [CrossRef] [PubMed]
- Daly, B.; Sharif, M.O.; Newton, T.; Jones, K.; Worthington, H.V. Local interventions for the management of alveolar osteitis (dry socket). In Cochrane Database of Systematic Reviews; The Cochrane Library: London, UK, 2012; Volume 12, p. Cd006968. Available online: https://www.cochranelibrary.com/cdsr/doi/10.1002/14651858.CD006968.pub2/abstract (accessed on 16 July 2020).
- Dodson, T. Prevention and treatment of dry socket. Evid.-Based Dent. 2013, 14, 13–14. [Google Scholar] [CrossRef]
- Prag, G.; Falk-Brynhildsen, K.; Jacobsson, S.; Hellmark, B.; Unemo, M.; Soderquist, B. Decreased susceptibility to chlorhexidine and prevalence of disinfectant resistance genes among clinical isolates of Staphylococcus epidermidis. APMIS Acta Pathol. Microbiol. Immunol. Scand. 2014, 122, 961–967. [Google Scholar] [CrossRef]
- Chrcanovic, B.R.; Martins, M.D.; Wennerberg, A. Immediate placement of implants into infected sites: A systematic review. Clin. Implant. Dent. Relat. Res. 2015, 17, e1–e16. [Google Scholar] [CrossRef]
- Pedrazzi, V.; Escobar, E.C.; Cortelli, J.R.; Haas, A.N.; Andrade, A.K.; Pannuti, C.M.; Almeida, E.R.; Costa, F.O.; Cortelli, S.C.; Rode Sde, M. Antimicrobial mouthrinse use as an adjunct method in peri-implant biofilm control. Braz. Oral Res. 2014, 28. [Google Scholar] [CrossRef] [PubMed]
- Anitha, V.; Rajesh, P.; Shanmugam, M.; Priya, B.M.; Prabhu, S.; Shivakumar, V. Comparative evaluation of natural curcumin and synthetic chlorhexidine in the management of chronic periodontitis as a local drug delivery: A clinical and microbiological study. Indian J. Dent. Res. Off. Publ. Indian Soc. Dent. Res. 2015, 26, 53–56. [Google Scholar] [CrossRef] [PubMed]
- Jhinger, N.; Kapoor, D.; Jain, R. Comparison of Periochip (chlorhexidine gluconate 2.5 mg) and Arestin (Minocycline hydrochloride 1 mg) in the management of chronic periodontitis. Indian J. Dent. Res. 2015, 6, 20–26. [Google Scholar]
- Rusu, D.; Stratul, S.I.; Sarbu, C.; Roman, A.; Anghel, A.; Didilescu, A.; Jentsch, H. Evaluation of a hydrophobic gel adhering to the gingiva in comparison with a standard water-soluble 1% chlorhexidine gel after scaling and root planing in patients with moderate chronic periodontitis. A randomized clinical trial. Int. J. Den. Hyg. 2015. [Google Scholar] [CrossRef] [PubMed]
- Sato, S.; Miyake, M.; Hazama, A.; Omori, K. Povidone-iodine-induced cell death in cultured human epithelial HeLa cells and rat oral mucosal tissue. Drug Chem. Toxicol. 2014, 37, 268–275. [Google Scholar] [CrossRef] [PubMed]
- Abbaszadegan, A.; Gholami, A.; Mirhadi, H.; Saliminasab, M.; Kazemi, A.; Moein, M.R. Antimicrobial and cytotoxic activity of Ferula gummosa plant essential oil compared to NaOCl and CHX: A preliminary in vitro study. Restor. Dent. Endod. 2015, 40, 50–57. [Google Scholar] [CrossRef]
- Furukawa, M.; Kaneyama, J.K.; Yamada, M.; Senda, A.; Manabe, A.; Miyazaki, A. Cytotoxic effects of hydrogen peroxide on human gingival fibroblasts in vitro. Oper. Dent. 2015. [Google Scholar] [CrossRef]
- Sawada, K.; Fujioka-Kobayashi, M.; Kobayashi, E.; Schaller, B.; Miron, R.J. Effects of antiseptic solutions commonly used in dentistry on bone viability, bone morphology, and release of growth factors. Int. J. Oral Maxillofac. Surg. Off. J. Am. Assoc. Oral Maxillofac. Surg. 2016, 74, 247–254. [Google Scholar] [CrossRef]
- Sawada, K.; Nakahara, K.; Haga-Tsujimura, M.; Fujioka-Kobayashi, M.; Iizuka, T.; Miron, R.J. Effect of irrigation time of antiseptic solutions on bone cell viability and growth factor release. J. Craniofac. Surg. 2018, 29, 376–381. [Google Scholar] [CrossRef] [PubMed]
- Miron, R.J.; Bosshardt, D.; Hedbom, E.; Zhang, Y.; Haenni, B.; Buser, D.; Sculean, A. Adsorption of enamel matrix proteins to a bovine-derived bone grafting material and its regulation of cell adhesion, proliferation, and differentiation. J. Periodontol. 2012, 83, 936–947. [Google Scholar] [CrossRef] [PubMed]
- Miron, R.J.; Saulacic, N.; Buser, D.; Iizuka, T.; Sculean, A. Osteoblast proliferation and differentiation on a barrier membrane in combination with BMP2 and TGFβ1. Clin. Oral Investig. 2013, 17, 981–988. [Google Scholar] [CrossRef] [PubMed]
- Faria, G.; Celes, M.R.; De Rossi, A.; Silva, L.A.B.; Silva, J.S.; Rossi, M.A. Evaluation of chlorhexidine toxicity injected in the paw of mice and added to cultured l929 fibroblasts. J. Endod. 2007, 33, 715–722. [Google Scholar] [CrossRef] [PubMed]
- Mariotti, A.J.; Rumpf, D.A. Chlorhexidine-induced changes to human gingival fibroblast collagen and non-collagen protein production. J. Periodontol. 1999, 70, 1443–1448. [Google Scholar] [CrossRef] [PubMed]
| Gene | Primer Sequence (5′–3′) | |
|---|---|---|
| Forward | Reverse | |
| hCOL1 | CCCAGCCAAGAACTGGTATAGG | GGCTGCCAGCATTGATAGTTTC |
| hPDGF-A | CACACCTCCTCGCTGTAGTATTTA | GTTATCGGTGTAAATGTCATCCAA |
| hTNF-α | CAGCCTCTTCTCCTTCCTGAT | GCCAGAGGGCTGATTAGAGA |
| hIL-6 | GAAAGGAGACATGTAACAAGAGT | GATTTTCACCAGGCAAGTCT |
| hGAPDH | AGCCACATCGCTCAGACA | GCCCAATACGACCAAATCC |
| hβ-Actin | CCAACCGCGAGAAGATGA | CCAGAGGCGTACAGGGATAG |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fujioka-Kobayashi, M.; Schaller, B.; Pikos, M.A.; Sculean, A.; Miron, R.J. Cytotoxicity and Gene Expression Changes of a Novel Homeopathic Antiseptic Oral Rinse in Comparison to Chlorhexidine in Gingival Fibroblasts. Materials 2020, 13, 3190. https://doi.org/10.3390/ma13143190
Fujioka-Kobayashi M, Schaller B, Pikos MA, Sculean A, Miron RJ. Cytotoxicity and Gene Expression Changes of a Novel Homeopathic Antiseptic Oral Rinse in Comparison to Chlorhexidine in Gingival Fibroblasts. Materials. 2020; 13(14):3190. https://doi.org/10.3390/ma13143190
Chicago/Turabian StyleFujioka-Kobayashi, Masako, Benoit Schaller, Michael A. Pikos, Anton Sculean, and Richard J. Miron. 2020. "Cytotoxicity and Gene Expression Changes of a Novel Homeopathic Antiseptic Oral Rinse in Comparison to Chlorhexidine in Gingival Fibroblasts" Materials 13, no. 14: 3190. https://doi.org/10.3390/ma13143190
APA StyleFujioka-Kobayashi, M., Schaller, B., Pikos, M. A., Sculean, A., & Miron, R. J. (2020). Cytotoxicity and Gene Expression Changes of a Novel Homeopathic Antiseptic Oral Rinse in Comparison to Chlorhexidine in Gingival Fibroblasts. Materials, 13(14), 3190. https://doi.org/10.3390/ma13143190






