Preparation, Characterisation, and Application of Bifunctional BaSO4 Sheets
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Sheet Preparation
2.3. Sheet Characterization
2.3.1. Scanning Electron Microscopy (SEM)
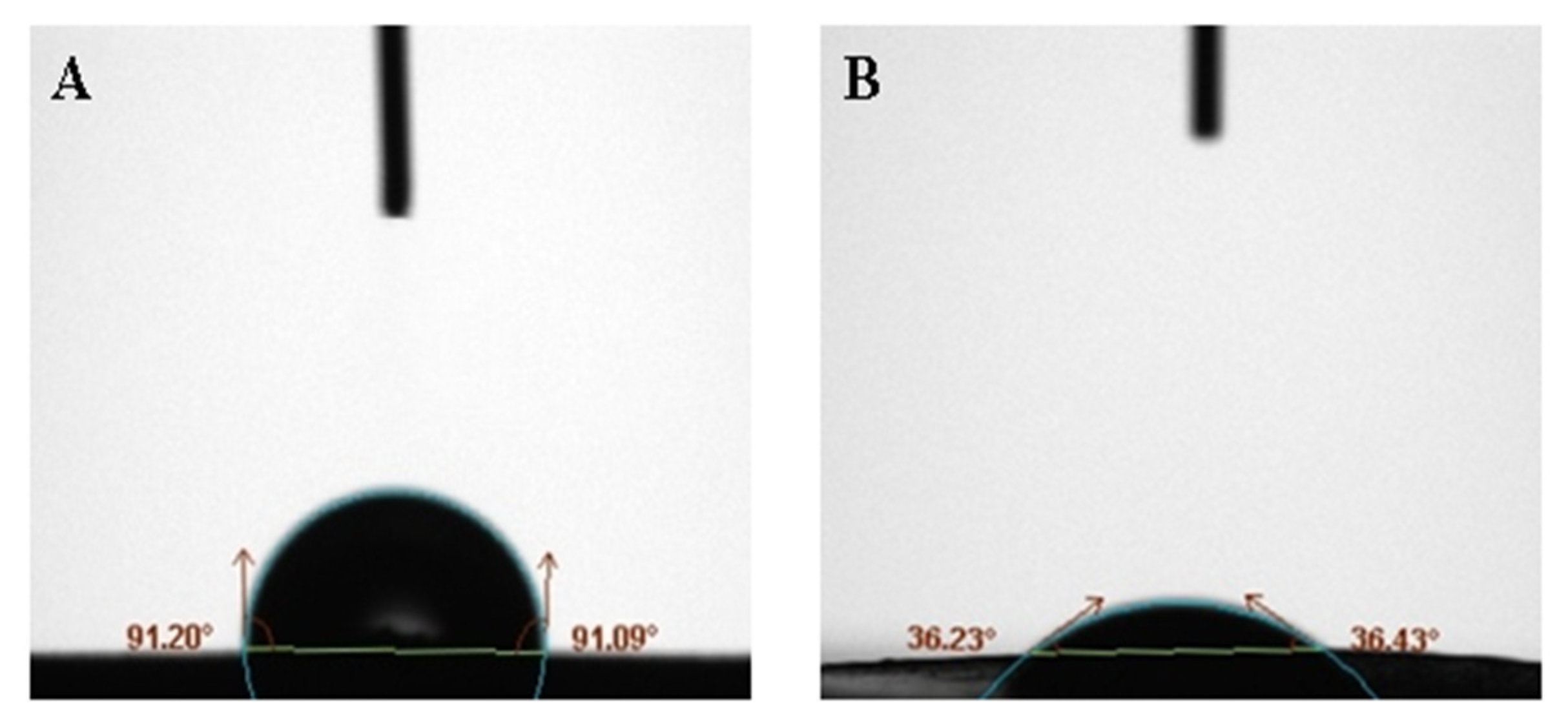
2.3.2. Dynamic Contact Angle Test
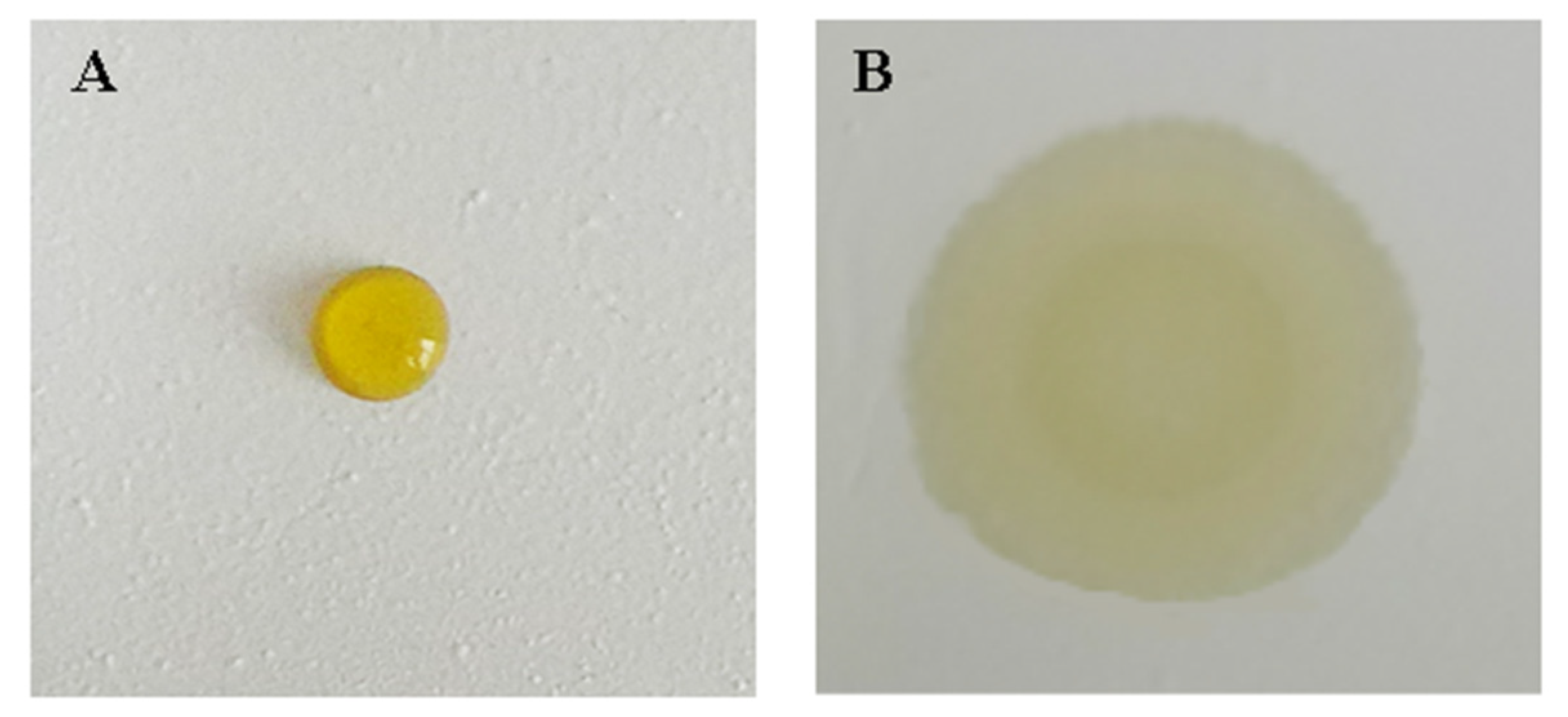
2.3.3. Determination of Solid Content
2.3.4. Determination of Wet Sheet Thickness
2.3.5. Determination of Reflectance
2.3.6. Particle Size Distribution Tests
2.3.7. Determination of Scattering Power
3. Results
3.1. Analysis of BaSO4 Particles
3.2. BaSO4 Morphology
3.3. Sheet Morphology
3.4. Mixing Time
3.5. Surfactant
3.6. Solid Content
3.7. Wet Sheet Thickness
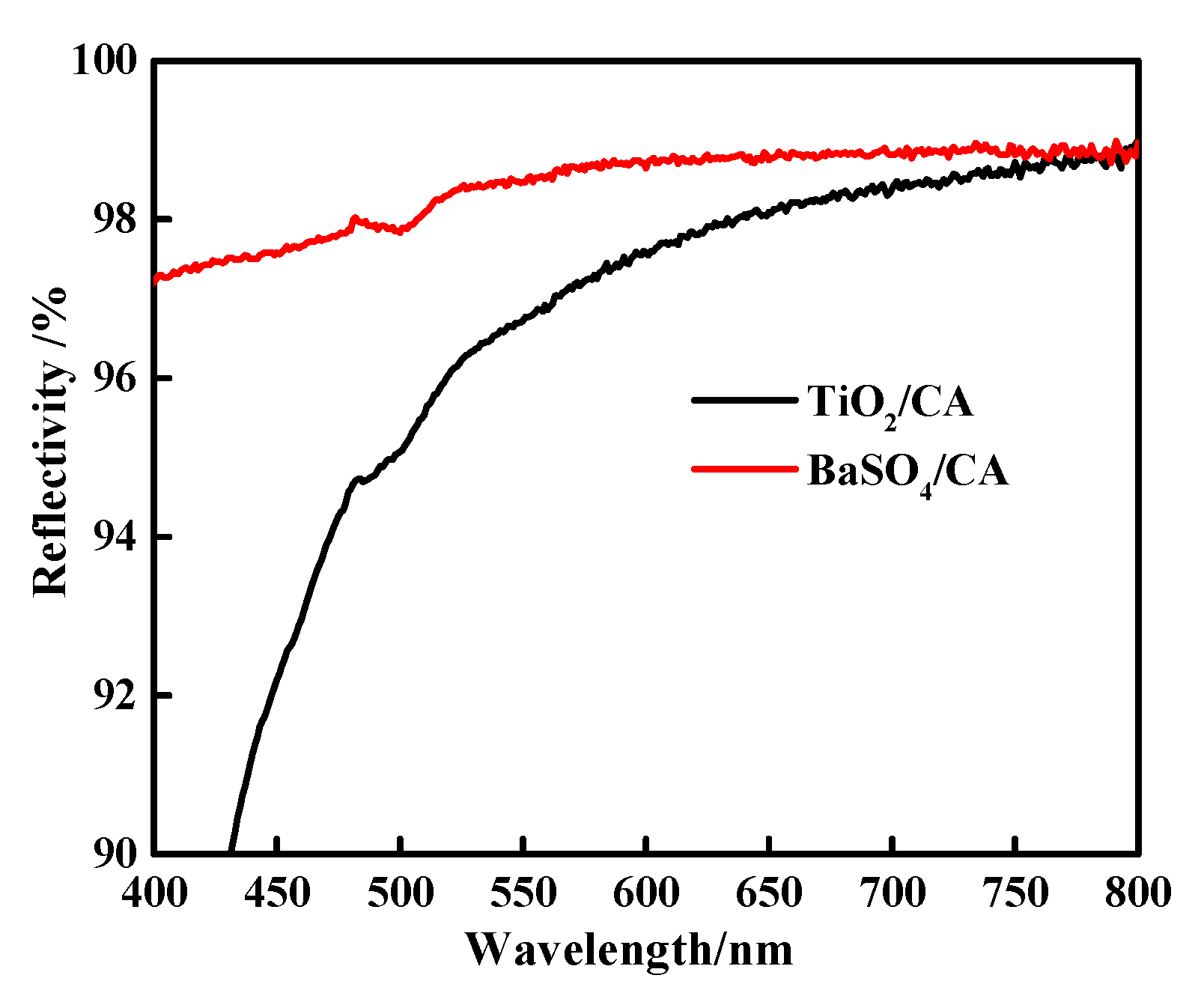
3.8. Reflection Tests
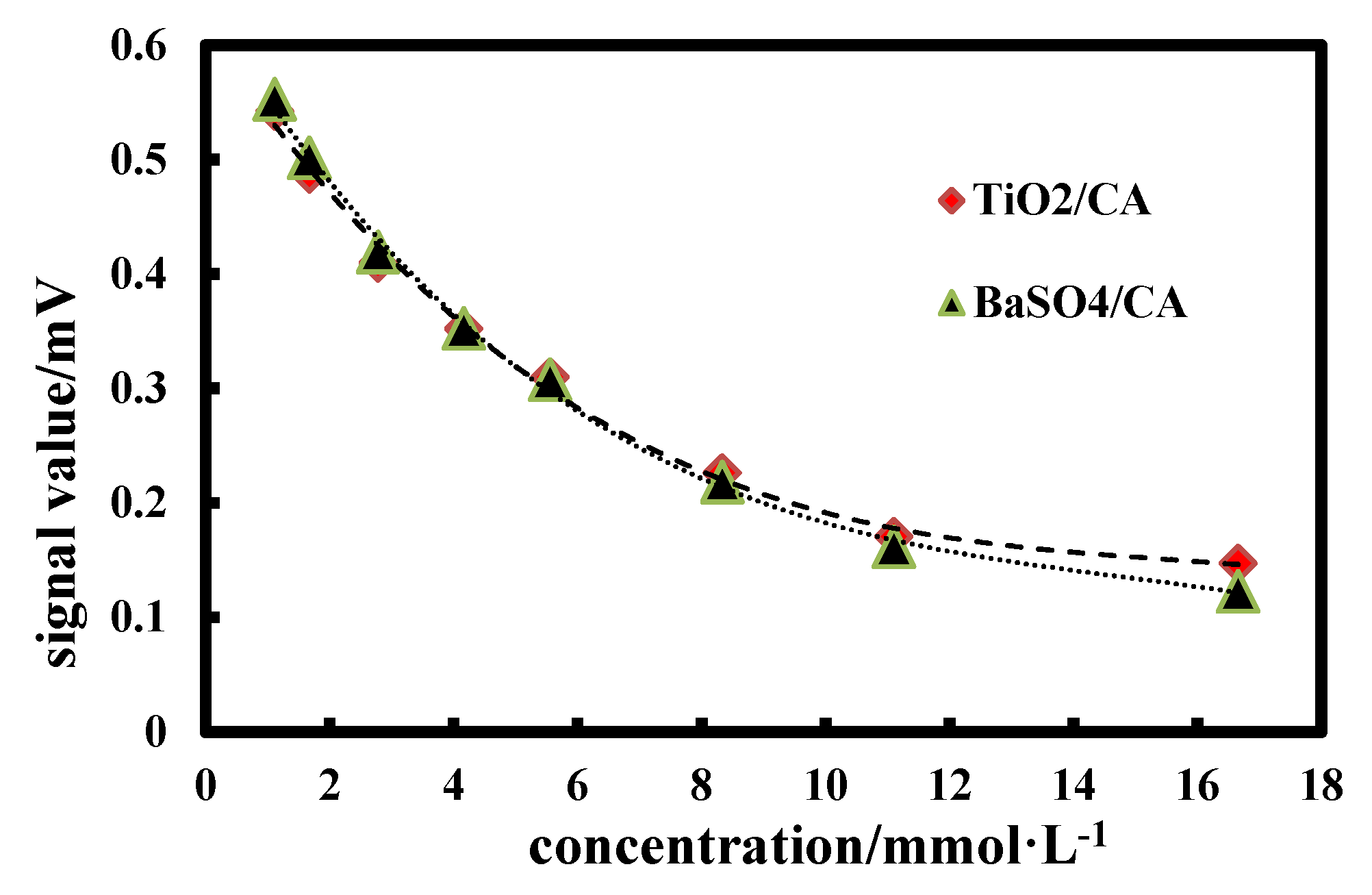
3.9. Scattering PowerTests
3.10. Chip Detection
3.11. Repeatability Tests
3.12. Accuracy Tests
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Yang, S.; Wang, Y.; Luo, S.; Shan, C.; Geng, Y.; Zhang, T.; Sheng, S.-R.; Zan, X. Building polyphenol and gelatin films as implant coating, evaluating from in vitro and in vivo performances. Colloids Surf. B Biointerfaces 2019, 181, 549–560. [Google Scholar] [CrossRef] [PubMed]
- Li, G.X.; Lu, X.J.; Gao, B.X. Comparison of dry chemistry and liquid chemistry in clinical biochemistry. West China Med. J. 2003, 1, 69–70. [Google Scholar]
- Almando, G.; Ernawati, A.G. Synthetic biology-based portable in vitro diagnostic platforms. AJM 2018, 54, 423–428. [Google Scholar]
- Fujishima, A.; Honda, K. Electrochemical photolysis of water at a semiconductor electrode. Nature 1972, 238, 37–38. [Google Scholar] [CrossRef]
- Hovda, K.E.; Gadeholt, G.; Evtodienko, V.; Jacobsen, D. A novel bedside diagnostic test for methanol poisoning using dry chemistry for formate. Scand. J. Clin. Lab. Investig. 2015, 75, 610–614. [Google Scholar] [CrossRef]
- Arai, F.; Kondo, A. Dry-Type Multilayer Analytical Element. U.S. Patent 5,004,685, 18 August 1987. [Google Scholar]
- Liu, D. Establishment of Dry Biochemical Analysis Method and Its Methodological Evaluation. Master’s Thesis, Changchun University of Science and Technology, Changchun, China, 2014. [Google Scholar]
- Sia, S.K.; Linder, V.; Parviz, B.A.; Siegel, A.; Whitesides, G.M. An Integrated approach to a portable and low-cost immunoassay for resource-poor settings. Angew. Chem. Int. Ed. 2004, 43, 498–502. [Google Scholar] [CrossRef]
- Martinez, A.W.; Phillips, S.T.; Whitesides, G.M. From the cover: Three-dimensional microfluidic devices fabricated in layered paper and tape. Proc. Natl. Acad. Sci. USA 2008, 105, 19606–19611. [Google Scholar] [CrossRef] [Green Version]
- Guo, X.Y.; Li, Y.; Zheng, Y.; Cheng, P.L.; He, G.X.; Fan, D.D. Synthesis and characterization of homogeneous titanium dioxide microspheres. Fine Chem. 2017, 34, 1404–1411. [Google Scholar]
- Huang, L.; Du, T.F.; Zhu, B.Y. Novel Quality Control Method Applied to Immunochromatography Test Paper Strip. CN106,353,503, 25 January 2016. [Google Scholar]
- Tong, L.; Ren, X.T.; Su, E.B. Dry Chemical Bigeminal Reagent Strip for Simultaneously Detecting Glutamic Oxalacetic Transaminase and Glutamic-Pyruvic Transaminase. CN106,282,312, 4 January 2017. [Google Scholar]
- Guo, X.; Liu, J.; Liu, L.; Yang, S.; He, G.; Jin, H. Preparation, characteristics, and application of bifunctional TiO2 sheets. Materials 2020, 13, 1615. [Google Scholar] [CrossRef] [Green Version]
- Liu, Y.; Guo, X.; Yang, S.; He, G.; Jin, H. Controllable preparation of uniform micron-sized barium-sulfate spheres. Cryst. Res. Technol. 2018, 53, 1700212. [Google Scholar] [CrossRef]
- Ye, F.F.; Zhang, B.D.; Jin, H.B.; Guo, X.Y.; He, G.X.; Zhang, R.Y.; Gu, Q.Y.; Yang, S.H. Preparation of BaSO4 nanoparticles in microchannel reactor and its application in multifunctional layers of medical slices. CIESC J. 2019, 70, 1179–1187. [Google Scholar]
- Nagaraja, B.M.; Jung, K.-D.; Ahn, B.S.; Abimanyu, H.; Yoo, K.S. Catalytic decomposition of SO3 over Pt/BaSO4 materials in sulfur−iodine cycle for hydrogen production. Ind. Eng. Chem. Res. 2009, 48, 1451–1457. [Google Scholar] [CrossRef]
- Gilman, J.W. Flammability and thermal stability studies of polymer layered-silicate (clay) nanocomposites. Appl. Clay Sci. 1999, 15, 31–49. [Google Scholar] [CrossRef]
- Gao, H.; Lin, J.; Li, W.-Y.; Hu, Z.; Zhang, Y. Formation of shaped barium sulfate-dye hybrids: Waste dye utilization for eco-friendly treatment of wastewater. Environ. Sci. Pollut. Res. 2009, 17, 78–83. [Google Scholar] [CrossRef] [PubMed]
- Ripin, A.; Mohamed, F.; Choo, T.; Yusof, M.; Hashim, S.; Ghoshal, S. X-ray shielding behaviour of kaolin derived mullite-barites ceramic. Radiat. Phys. Chem. 2018, 144, 63–68. [Google Scholar] [CrossRef]
- Hayashida, K.; Takeda, Y.; Yatake, H.; Yamaguchi, M.; Yamamoto, K.; Kuwano, T.; Katsuda, T. Relationship between bone mineral density and body composition estimated by dual-energy X-ray absorptiometry: Comparison between groups aged 20-39 and 40-59 Years. Kobe J. Med Sci. 2015, 61, 97–101. [Google Scholar]
- Yao, C.; Yang, G. Synthesis, thermal, and rheological properties of poly (trimethylene terephthalate)/ BaSO4 nanocomposites. Polym. Adv. Technol. 2009, 20, 768–774. [Google Scholar] [CrossRef]
- Sun, S.; Li, C.; Zhang, L.; Du, H.; Burnell-Gray, J. Effects of surface modification of fumed silica on interfacial structures and mechanical properties of poly(vinyl chloride) composites. Eur. Polym. J. 2006, 42, 1643–1652. [Google Scholar] [CrossRef]
- Mülhaupt, R.; Engelhardt, T.; Sound, N. Nanocomposites-On the way to application. Plastics 2001, 91, 178–190. [Google Scholar]
- Bauer, F.; Flyunt, R.; Czihal, K.; Buchmeiser, M.R.; Langguth, H.; Mehnert, R. Nano/micro particle hybrid composites for scratch and abrasion resistant polyacrylate coatings. Macromol. Mater. Eng. 2006, 291, 493–498. [Google Scholar] [CrossRef]
- Wang, K.; Wu, J.; Ye, L.; Zeng, H. Mechanical properties and toughening mechanisms of polypropylene/barium sulfate composites. Compos. Part A Appl. Sci. Manuf. 2003, 34, 1199–1205. [Google Scholar] [CrossRef]
- Xu, Y.; Zhang, X.Z.; Zhou, C.X.; Wu, W.S.; Shi, L.Y.; Yang, Y. Study on the mechanical properties and structue of PVC/BaSO4 nano-composites. Polym. Eng. Sci. 2007, 23, 144–147. [Google Scholar]
- Li, Z.; Guo, S.; Song, W.; Hou, B. Effect of interfacial interaction on morphology and mechanical properties of PP/POE/BaSO4 ternary composites. J. Mater. Sci. 2003, 38, 1793–1802. [Google Scholar] [CrossRef]
- Li, L.L.; Chai, X.S.; Tian, Y.X.; Chen, R.Q. A new sample pretreatment method of infrared spectrum analysis for barium sulfate identification in paper. China Pulp Pap. 2015, 34, 39–42. [Google Scholar]
- Han, L.; Chen, Q.; Chen, H.; Yu, S.; Xiao, L.; Ye, Z. Synthesis and performance of functionalized α-zirconium phosphate modified with octadecyl isocyanate. J. Nanomater. 2018, 2018, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Liu, L.X. Preparation of Multi-Functional Sheet with Uniform Size TiO2 Microspheres and Application of Dry Chemical Diagnostic Reagents. Master’s Thesis, Beijing University of Chemical Technology, Beijing, China, 2019. [Google Scholar]





| Acetone Volume (mL) | Solid Content (%) | The Surface and Flatness of Bifunctional Sheet |
|---|---|---|
| 8 | 60.1 | uneven |
| 10 | 54.9 | uniform and flat |
| 12 | 50.5 | thin and porous |
| Sheets Code | Wet Sheet Thickness (µm) | Scattering Time (s) | Scattering Diameter (cm) |
|---|---|---|---|
| 1 | 200 | 27 | 2.3 |
| 2 | 250 | 20 | 2.0 |
| 3 | 300 | 17 | 1.9 |
| 4 | 350 | 14 | 1.6 |
| 5 | 400 | 12 | 1.3 |
| 6 | 450 | 11 | 1.2 |
| 7 | 500 | 11 | 1.2 |
| Serial | Single Number (V) | Mean Single Value | Deviation |
|---|---|---|---|
| 1 | 0.3295 | 0.3250 | 0.0045 |
| 2 | 0.3259 | 0.0009 | |
| 3 | 0.3238 | −0.0012 | |
| 4 | 0.3226 | −0.0024 | |
| 5 | 0.3230 | −0.0020 |
| Chip Name | Concentration (mmol·L−1) | SD | CV (%) | |||||
|---|---|---|---|---|---|---|---|---|
| GLU-TiO2/CA | 8.44 | 8.81 | 8.68 | 8.19 | 8.27 | 8.47 | 0.24 | 2.8 |
| GLU-BaSO4/CA | 8.23 | 8.19 | 8.61 | 8.12 | 8.54 | 8.46 | 0.20 | 2.4 |
| Chip Name | Concentration (mmol·L−1) | Target Value (mmol·L−1) | Range of Target Values (mmol·L−1) | ||
|---|---|---|---|---|---|
| GLU-BaSO4/CA | 1.58 | 1.75 | 1.72 | 1.67 | 1.50~1.84 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Luo, J.; Liu, J.; Guo, X.; Liu, Y.; Jin, H.; Fan, D. Preparation, Characterisation, and Application of Bifunctional BaSO4 Sheets. Materials 2020, 13, 2903. https://doi.org/10.3390/ma13132903
Luo J, Liu J, Guo X, Liu Y, Jin H, Fan D. Preparation, Characterisation, and Application of Bifunctional BaSO4 Sheets. Materials. 2020; 13(13):2903. https://doi.org/10.3390/ma13132903
Chicago/Turabian StyleLuo, Jia, Jiaqi Liu, Xiaoyan Guo, Yuejiao Liu, Haibo Jin, and Daidi Fan. 2020. "Preparation, Characterisation, and Application of Bifunctional BaSO4 Sheets" Materials 13, no. 13: 2903. https://doi.org/10.3390/ma13132903
APA StyleLuo, J., Liu, J., Guo, X., Liu, Y., Jin, H., & Fan, D. (2020). Preparation, Characterisation, and Application of Bifunctional BaSO4 Sheets. Materials, 13(13), 2903. https://doi.org/10.3390/ma13132903





