Zinc as a Therapeutic Agent in Bone Regeneration
Abstract
1. Zinc Biodistribution and Main Intracellular Roles
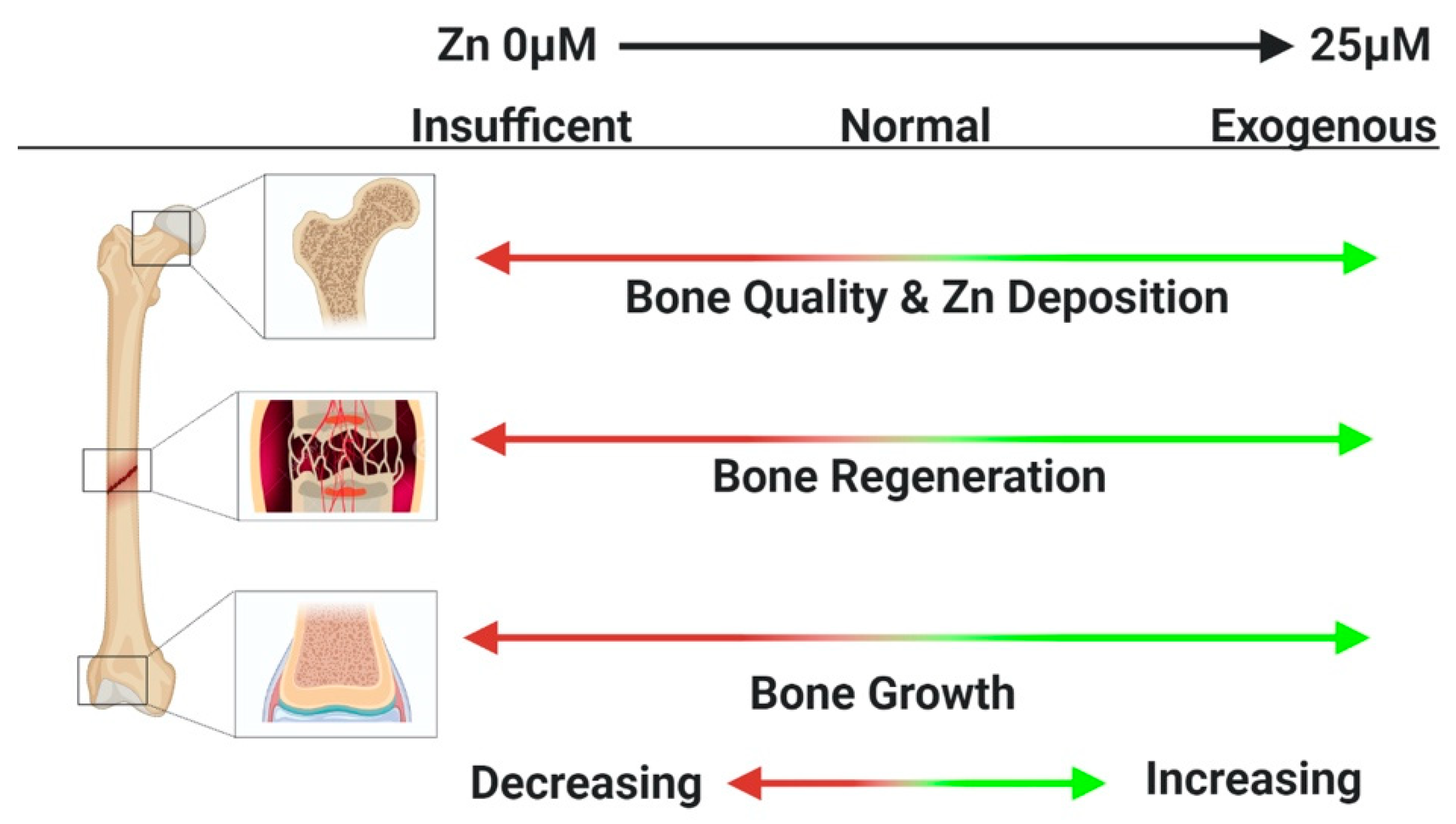
2. Role of Zinc in Skeletal Growth
3. Zinc and Bone Homeostasis
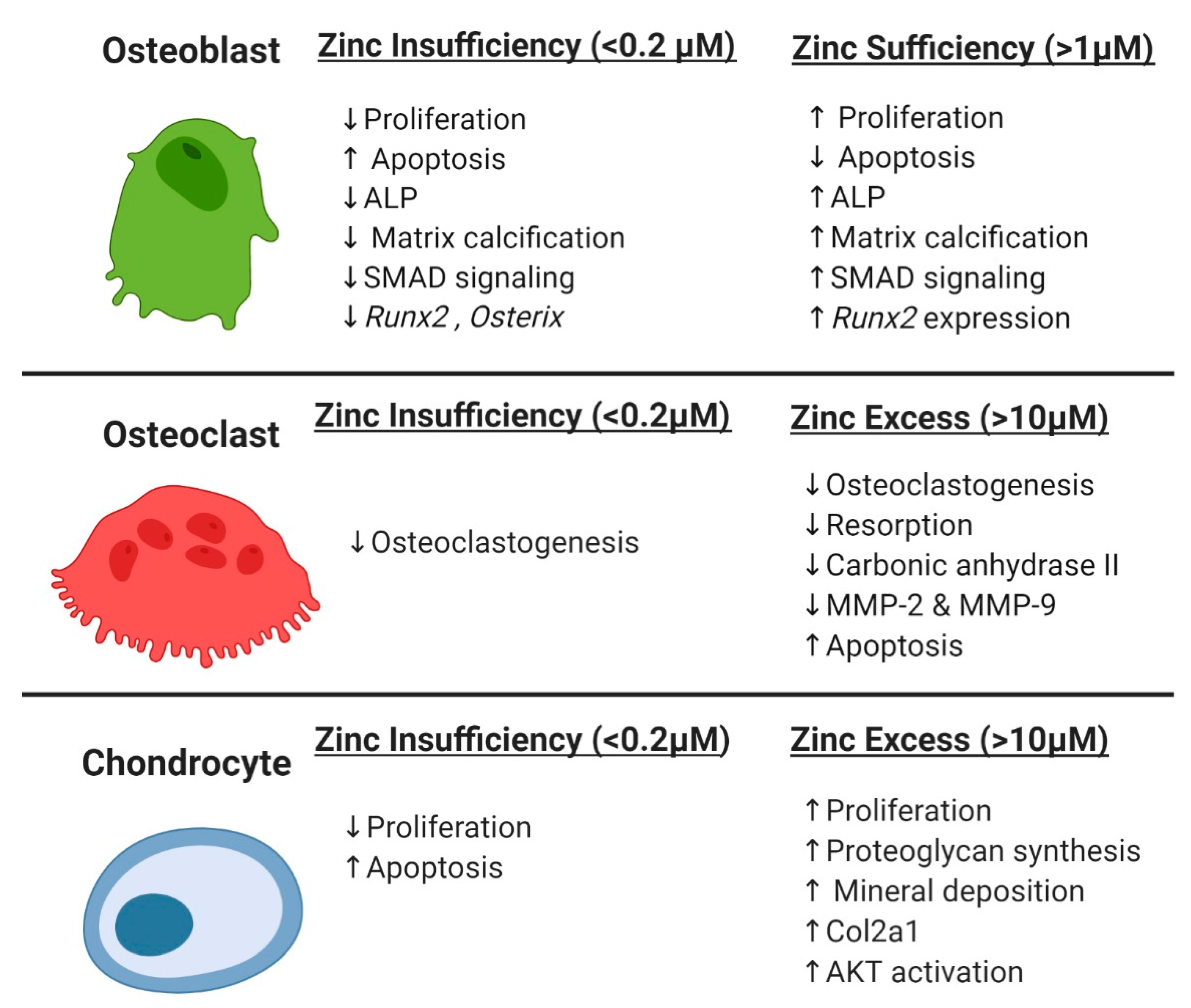
4. Zinc Effects on Osteoblasts
5. Zinc Effects on Osteoclasts
6. Zinc Effects on Chondrocytes
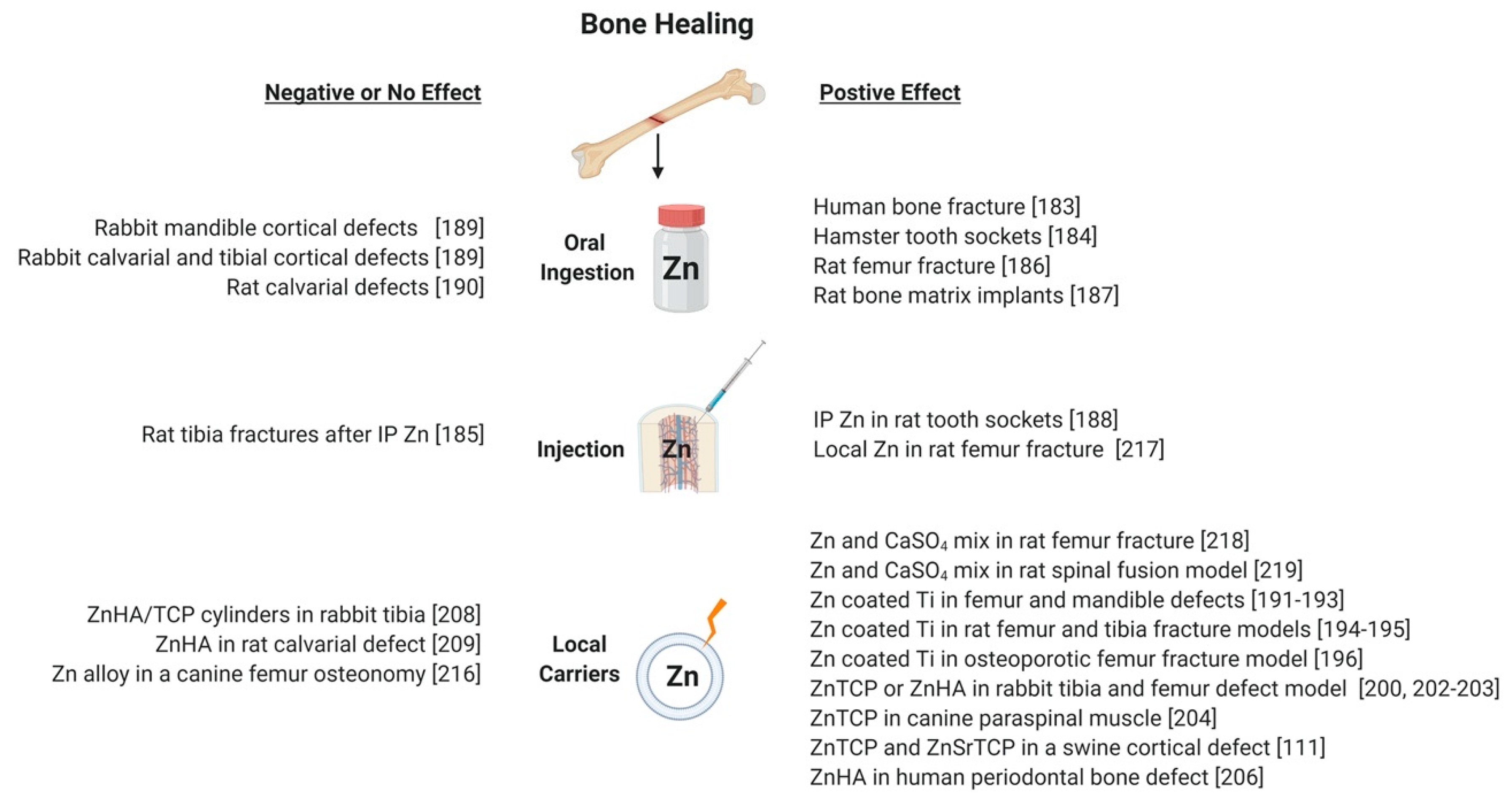
7. Using Zinc to Promote Bone Regeneration
8. Future Directions
Author Contributions
Funding
Conflicts of Interest
References
- Zoroddu, M.A.; Aaseth, J.; Crisponi, G.; Medici, S.; Peana, M.; Nurchi, V.M. The essential metals for humans: A brief overview. J. Inorg. Biochem. 2019, 195, 120–129. [Google Scholar] [CrossRef] [PubMed]
- Barceloux, D.G. Zinc. J. Toxicol. Clin. Toxicol. 1999, 37, 279–292. [Google Scholar] [CrossRef] [PubMed]
- Davies, I.J.T.; Musa, M.; Dormandy, T.L. Measurements of plasma zinc: Part I In health and disease. J. Clin. Pathol. 1968, 21, 359–363. [Google Scholar] [CrossRef] [PubMed]
- Magneson, G.R.; Puvathingal, J.M.; Ray, W.J. The concentrations of free Mg2+ and free Zn2+ in equine blood plasma. J. Boil. Chem. 1987, 262, 11140–11148. [Google Scholar]
- Coverdale, J.P.C.; Barnett, J.; Adamu, A.H.; Griffiths, E.J.; Stewart, A.J.; Blindauer, C.A. A metalloproteomic analysis of interactions between plasma proteins and zinc: Elevated fatty acid levels affect zinc distribution. Met. Integr. Biometal Sci. 2019, 11, 1805–1819. [Google Scholar] [CrossRef] [PubMed]
- Chilvers, D.C.; Dawson, J.B.; Bahreyni-Toosi, M.-H.; Hodgkinson, A. Identification and determination of copper-and zinc-protein complexes in blood plasma after chromatographic separation on DEAE-Sepharose CL-6B. Analyst 1984, 109, 871. [Google Scholar] [CrossRef]
- Foote, J.W.; Delves, H.T. Albumin bound and alpha 2-macroglobulin bound zinc concentrations in the sera of healthy adults. J. Clin. Pathol. 1984, 37, 1050–1054. [Google Scholar] [CrossRef]
- Alker, W.; Schwerdtle, T.; Schomburg, L.; Haase, H. A Zinpyr-1-based Fluorimetric Microassay for Free Zinc in Human Serum. Int. J. Mol. Sci. 2019, 20, 4006. [Google Scholar] [CrossRef]
- Maret, W.; Sandstead, H.H. Zinc requirements and the risks and benefits of zinc supplementation. J. Trace Elements Med. Boil. 2006, 20, 3–18. [Google Scholar] [CrossRef]
- Neldner, K.H.; Hambidge, K.M. Zinc Therapy of Acrodermatitis Enteropathica. N. Engl. J. Med. 1975, 292, 879–882. [Google Scholar] [CrossRef]
- Barnes, P.M.; Moynahan, E.J. Zinc Deficiency in Acrodermatitis Enteropathica: Multiple Dietary Intolerance Treated with Synthetic Diet. Proc. R. Soc. Med. 1973, 66, 327–329. [Google Scholar] [CrossRef] [PubMed]
- Moynahan, E.J. Letter: Acrodermatitis enteropathica: A lethal inherited human zinc-deficiency disorder. Lancet 1974, 2, 399–400. [Google Scholar] [CrossRef]
- Küry, S.; Dréno, B.; Bezieau, S.; Giraudet, S.; Kharfi, M.; Kamoun, R.; Moisan, J.-P. Identification of SLC39A4, a gene involved in acrodermatitis enteropathica. Nat. Genet. 2002, 31, 239–240. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Zhou, B.; Kuo, Y.-M.; Zemansky, J.; Gitschier, J. A Novel Member of a Zinc Transporter Family Is Defective in Acrodermatitis Enteropathica. Am. J. Hum. Genet. 2002, 71, 66–73. [Google Scholar] [CrossRef]
- Kasana, S.; Din, J.; Maret, W. Genetic causes and gene–nutrient interactions in mammalian zinc deficiencies: Acrodermatitis enteropathica and transient neonatal zinc deficiency as examples. J. Trace Elements Med. Boil. 2015, 29, 47–62. [Google Scholar] [CrossRef]
- Geiser, J.; Venken, K.J.T.; De Lisle, R.C.; Andrews, G.K. A Mouse Model of Acrodermatitis Enteropathica: Loss of Intestine Zinc Transporter ZIP4 (Slc39a4) Disrupts the Stem Cell Niche and Intestine Integrity. PLoS Genet. 2012, 8, e1002766. [Google Scholar] [CrossRef]
- Mittermeier, L.; Demirkhanyan, L.; Stadlbauer, B.; Breit, A.; Recordati, C.; Hilgendorff, A.; Matsushita, M.; Braun, A.; Simmons, D.; Zakharian, E.; et al. TRPM7 is the central gatekeeper of intestinal mineral absorption essential for postnatal survival. Proc. Natl. Acad. Sci. USA 2019, 116, 4706–4715. [Google Scholar] [CrossRef]
- Andreini, C.; Banci, L.; Bertini, I.; Rosato, A. Counting the Zinc-Proteins Encoded in the Human Genome. J. Proteome Res. 2006, 5, 196–201. [Google Scholar] [CrossRef]
- Klug, A. The Discovery of Zinc Fingers and Their Applications in Gene Regulation and Genome Manipulation. Annu. Rev. Biochem. 2010, 79, 213–231. [Google Scholar] [CrossRef]
- Westin, G.; Schaffner, W. A zinc-responsive factor interacts with a metal-regulated enhancer element (MRE) of the mouse metallothionein-I gene. EMBO J. 1988, 7, 3763–3770. [Google Scholar] [CrossRef]
- Heuchel, R.; Radtke, F.; Georgiev, O.; Stark, G.; Aguet, M.; Schaffner, W. The transcription factor MTF-1 is essential for basal and heavy metal-induced metallothionein gene expression. EMBO J. 1994, 13, 2870–2875. [Google Scholar] [CrossRef] [PubMed]
- Laity, J.H.; Andrews, G.K. Understanding the mechanisms of zinc-sensing by metal-response element binding transcription factor-1 (MTF-1). Arch. Biochem. Biophys. 2007, 463, 201–210. [Google Scholar] [CrossRef] [PubMed]
- Zhao, W.-J.; Song, Q.; Wang, Y.-H.; Li, K.-J.; Mao, L.; Hu, X.; Lian, H.-Z.; Zheng, W.; Hua, Z.-C. Zn-Responsive Proteome Profiling and Time-Dependent Expression of Proteins Regulated by MTF-1 in A549 Cells. PLoS ONE 2014, 9, e105797. [Google Scholar] [CrossRef] [PubMed]
- Francis, M.; Grider, A. Bioinformatic analysis of the metal response element and zinc-dependent gene regulation via the metal response element-binding transcription factor 1 in Caco-2 cells. BioMetals 2018, 31, 639–646. [Google Scholar] [CrossRef]
- Lindert, U.; Cramer, M.; Meuli, M.; Georgiev, O.; Schaffner, W. Metal-Responsive Transcription Factor 1 (MTF-1) Activity Is Regulated by a Nonconventional Nuclear Localization Signal and a Metal-Responsive Transactivation Domain. Mol. Cell. Boil. 2009, 29, 6283–6293. [Google Scholar] [CrossRef]
- Gunes, C.; Heuchel, R.; Georgiev, O.; Müller, K.; Lichtlen, P.; Blüthmann, H.; Marino, S.; Aguzzi, A.; Schaffner, W. Embryonic lethality and liver degeneration in mice lacking the metal-responsive transcriptional activator MTF-1. EMBO J. 1998, 17, 2846–2854. [Google Scholar] [CrossRef]
- Bouron, A.; Oberwinkler, J. Contribution of calcium-conducting channels to the transport of zinc ions. Pflug. Archiv. Eur. J. Physiol. 2013, 466, 381–387. [Google Scholar] [CrossRef]
- Kambe, T.; Tsuji, T.; Hashimoto, A.; Itsumura, N. The Physiological, Biochemical, and Molecular Roles of Zinc Transporters in Zinc Homeostasis and Metabolism. Physiol. Rev. 2015, 95, 749–784. [Google Scholar] [CrossRef]
- Hojyo, S.; Fukada, T. Roles of Zinc Signaling in the Immune System. J. Immunol. Res. 2016, 2016, 1–21. [Google Scholar] [CrossRef]
- Fukada, T.; Hojyo, S.; Furuichi, T. Zinc signal: A new player in osteobiology. J. Bone Miner. Metab. 2012, 31, 129–135. [Google Scholar] [CrossRef]
- Babula, P.; Masarik, M.; Adam, V.; Eckschlager, T.; Stiborová, M.; Trnkova, L.; Skutkova, H.; Provazník, I.; Hubalek, J.; Kizek, R. Mammalian metallothioneins: Properties and functions. Met. Integr. Biometal Sci. 2012, 4, 739–750. [Google Scholar] [CrossRef] [PubMed]
- Matsui, T.; Yamaguchi, M. Zinc modulation of insulin-like growth factor’s effect in osteoblastic MC3T3-E1 cells. Peptides 1995, 16, 1063–1068. [Google Scholar] [CrossRef]
- Macdonald, R.S. The role of zinc in growth and cell proliferation. J. Nutr. 2000, 130, 1500S–1508S. [Google Scholar] [CrossRef] [PubMed]
- Vardatsikos, G.; Pandey, N.R.; Srivastava, A.K. Insulino-mimetic and anti-diabetic effects of zinc. J. Inorg. Biochem. 2013, 120, 8–17. [Google Scholar] [CrossRef] [PubMed]
- Liang, D.; Yang, M.-W.; Guo, B.; Cao, J.; Yang, L.; Guo, X.; Li, Y.; Gao, Z. Zinc Inhibits H2O2-Induced MC3T3-E1 Cells Apoptosis via MAPK and PI3K/AKT Pathways. Boil. Trace Element Res. 2012, 148, 420–429. [Google Scholar] [CrossRef] [PubMed]
- Sun, W.; Yang, J.; Wang, W.; Hou, J.; Cheng, Y.; Fu, Y.; Xu, Z.; Cai, L. The beneficial effects of Zn on Akt-mediated insulin and cell survival signaling pathways in diabetes. J. Trace Elements Med. Boil. 2018, 46, 117–127. [Google Scholar] [CrossRef]
- Liang, D.; Yang, M.-W.; Guo, B.; Cao, J.; Yang, L.; Guo, X. Zinc Upregulates the Expression of Osteoprotegerin in Mouse Osteoblasts MC3T3-E1 Through PKC/MAPK Pathways. Boil. Trace Element Res. 2011, 146, 340–348. [Google Scholar] [CrossRef]
- Liang, D.; Xiang, L.; Yang, M.-W.; Zhang, X.; Guo, B.; Chen, Y.; Yang, L.; Cao, J. ZnT7 can protect MC3T3-E1 cells from oxidative stress-induced apoptosis via PI3K/Akt and MAPK/ERK signaling pathways. Cell. Signal. 2013, 25, 1126–1135. [Google Scholar] [CrossRef]
- Zhu, D.; Su, Y.; Young, M.L.; Ma, J.; Zheng, Y.; Tang, L. Biological Responses and Mechanisms of Human Bone Marrow Mesenchymal Stem Cells to Zn and Mg Biomaterials. ACS Appl. Mater. Interfaces 2017, 9, 27453–27461. [Google Scholar] [CrossRef]
- Liu, M.J.; Bao, S.; Gálvez-Peralta, M.; Pyle, C.J.; Rudawsky, A.C.; Pavlovicz, R.E.; Killilea, D.W.; Li, C.; Nebert, D.W.; Wewers, M.D.; et al. ZIP8 regulates host defense through zinc-mediated inhibition of NF-kappaB. Cell Rep. 2013, 3, 386–400. [Google Scholar] [CrossRef]
- Park, K.H.; Park, B.; Yoon, D.S.; Kwon, S.-H.; Shin, D.M.; Lee, J.W.; Lee, H.G.; Shim, J.-H.; Park, J.H.; Lee, J.M. Zinc inhibits osteoclast differentiation by suppression of Ca2+-Calcineurin-NFATc1 signaling pathway. Cell Commun. Signal. 2013, 11, 74. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.-J.; Bao, S.; Napolitano, J.R.; Burris, D.L.; Yu, L.; Tridandapani, S.; Knoell, D.L. Zinc Regulates the Acute Phase Response and Serum Amyloid A Production in Response to Sepsis through JAK-STAT3 Signaling. PLoS ONE 2014, 9, e94934. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Elberg, G.; Sekar, N.; He, Z.B.; Shechter, Y. Antilipolytic actions of vanadate and insulin in rat adipocytes mediated by distinctly different mechanisms. Endocrinology 1997, 138, 2274–2279. [Google Scholar] [CrossRef] [PubMed]
- Haase, H.; Maret, W. Protein Tyrosine Phosphatases as Targets of the Combined Insulinomimetic Effects of Zinc and Oxidants. BioMetals 2005, 18, 333–338. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, M.; Fukagawa, M. Role of Zinc in Regulation of Protein Tyrosine Phosphatase Activity in Osteoblastic MC3T3-E1 Cells: Zinc Modulation of Insulin-like Growth Factor-I’s Effect. Calcif. Tissue Int. 2004, 76, 32–38. [Google Scholar] [CrossRef]
- Popovics, P.; Stewart, A.J. GPR39: A Zn2+-activated G protein-coupled receptor that regulates pancreatic, gastrointestinal and neuronal functions. Cell. Mol. Life Sci. 2010, 68, 85–95. [Google Scholar] [CrossRef] [PubMed]
- Holst, B.; Egerod, K.L.; Schild, E.; Vickers, S.P.; Cheetham, S.; Gerlach, L.-O.; Storjohann, L.; Stidsen, C.E.; Jones, R.; Beck-Sickinger, A.G.; et al. GPR39 Signaling Is Stimulated by Zinc Ions But Not by Obestatin. Endocrinology 2007, 148, 13–20. [Google Scholar] [CrossRef]
- Yasuda, S.-I.; Miyazaki, T.; Munechika, K.; Yamashita, M.; Ikeda, Y.; Kamizono, A. Isolation of Zn2+ as an Endogenous Agonist of GPR39 from Fetal Bovine Serum. J. Recept. Signal Transduct. 2007, 27, 235–246. [Google Scholar] [CrossRef]
- Holst, B.; Egerod, K.L.; Jin, C.; Petersen, P.S.; Østergaard, M.V.; Hald, J.; Sprinkel, A.M.E.; Størling, J.; Mandrup-Poulsen, T.; Holst, J.J.; et al. G protein-coupled receptor 39 deficiency is associated with pancreatic islet dysfunction. Endocrinology 2009, 150, 2577–2585. [Google Scholar] [CrossRef]
- Tremblay, F.; Richard, A.-M.T.; Will, S.; Syed, J.; Stedman, N.; Perreault, M.; Gimeno, R.E. Disruption of G Protein-Coupled Receptor 39 Impairs Insulin Secretion in Vivo. Endocrinology 2009, 150, 2586–2595. [Google Scholar] [CrossRef]
- Prasad, A.S.; Halsted, J.A.; Nadimi, M. Syndrome of iron deficiency anemia, hepatosplenomegaly, hypogonadism, dwarfism and geophagia. Am. J. Med. 1961, 31, 532–546. [Google Scholar] [CrossRef]
- Rossi, L.; Migliaccio, S.; Corsi, A.; Marzia, M.; Bianco, P.; Teti, A.M.; Gambelli, L.; Cianfarani, S.; Paoletti, F.; Branca, F. Reduced growth and skeletal changes in zinc-deficient growing rats are due to impaired growth plate activity and inanition. J. Nutr. 2001, 131, 1142–1146. [Google Scholar] [CrossRef] [PubMed]
- Hadley, K.B.; Newman, S.M.; Hunt, J.R. Dietary zinc reduces osteoclast resorption activities and increases markers of osteoblast differentiation, matrix maturation, and mineralization in the long bones of growing rats. J. Nutr. Biochem. 2010, 21, 297–303. [Google Scholar] [CrossRef]
- Hie, M.; Iitsuka, N.; Otsuka, T.; Nakanishi, A.; Tsukamoto, I. Zinc deficiency decreases osteoblasts and osteoclasts associated with the reduced expression of Runx2 and RANK. Bone 2011, 49, 1152–1159. [Google Scholar] [CrossRef]
- Hickory, W.; Catalanotto, F.A.; Nanda, R. Fetal Skeletal Malformations Associated with Moderate Zinc Deficiency During Pregnancy. J. Nutr. 1979, 109, 883–891. [Google Scholar] [CrossRef]
- Eberle, J.; Schmidmayer, S.; Erben, R.; Stangassinger, M.; Roth, H.-P. Skeletal Effects of Zinc Deficiency in Growing Rats. J. Trace Elements Med. Boil. 1999, 13, 21–26. [Google Scholar] [CrossRef]
- Kim, J.-T.; Baek, S.-H.; Lee, S.; Park, E.K.; Kim, E.-C.; Kwun, I.-S.; Shin, H.-I. Zinc-Deficient Diet Decreases Fetal Long Bone Growth through Decreased Bone Matrix Formation in Mice. J. Med. Food 2009, 12, 118–123. [Google Scholar] [CrossRef]
- Laudermilk, M.J.; Manore, M.M.; Thomson, C.A.; Houtkooper, L.B.; Farr, J.N.; Going, S.B. Vitamin C and zinc intakes are related to bone macroarchitectural structure and strength in prepubescent girls. Calcif. Tissue Int. 2012, 91, 430–439. [Google Scholar] [CrossRef][Green Version]
- Hurley, L.S. Teratogenic aspects of manganese, zinc, and copper nutrition. Physiol. Rev. 1981, 61, 249–295. [Google Scholar] [CrossRef]
- Leek, J.C.; Keen, C.L.; Vogler, J.B.; Golub, M.S.; Hurley, L.S.; Hendrickx, A.G.; Gershwin, M.E. Long-term marginal zinc deprivation in rhesus monkeys. IV Effects on skeletal growth and mineralization. Am. J. Clin. Nutr. 1988, 47, 889–895. [Google Scholar] [CrossRef]
- Leek, J.C.; Vogler, J.B.; Gershwin, M.E.; Golub, M.S.; Hurley, L.S.; Hendrickx, A.G. Studies of marginal zinc deprivation in rhesus monkeys. V. Fetal and infant skeletal effects. Am. J. Clin. Nutr. 1984, 40, 1203–1212. [Google Scholar] [CrossRef] [PubMed]
- Rest, J.R. The histological effects of copper and zinc on chick embryo skeletal tissues in organ culture. Br. J. Nutr. 1976, 36, 243. [Google Scholar] [CrossRef] [PubMed]
- Todd, W.R.; Elvehjem, C.A.; Hart, E.B. Zinc in the nutrition of the rat. Am. J. Physiol. Content 1933, 107, 146–156. [Google Scholar] [CrossRef]
- Stirn, F.E.; Elvehjem, C.A.; Hart, E.B. The indispensability of zinc in the nutrition of the rat. J. Biol. Chem. 1935, 109, 347–359. [Google Scholar]
- Sandstead, H.H.; Prasad, A.S.; Schulert, A.R.; Farid, Z.; Miale, A.; Bassilly, S.; Darby, W.J. Human Zinc Deficiency, Endocrine Manifestations and Response to Treatment. Am. J. Clin. Nutr. 1967, 20, 422–442. [Google Scholar] [CrossRef]
- Oner, G.; Bhaumick, B.; Bala, R.M. Effect of Zinc Deficiency on Serum Somatomedin Levels and Skeletal Growth in Young Rats. Endocrinology 1984, 114, 1860–1863. [Google Scholar] [CrossRef]
- Haumont, S. Distribution of zinc in bone tissue. J. Histochem. Cytochem. 1961, 9, 141–145. [Google Scholar] [CrossRef]
- Bergman, B. Concentration of Zinc in Some Hard and Soft Tissues of Rat Determined by Neutron Activation Analysis. Acta Radiol. Ther. Phys. Boil. 1970, 9, 420–432. [Google Scholar] [CrossRef]
- Hove, E.; Elvehjem, C.; Hart, E. The effect of zinc on alkaline phosphatases. J. Boil. Chem. 1940, 134, 425. [Google Scholar]
- Coleman, J.E. Structure and mechanism of alkaline phosphatase. Annu. Rev. Biophys. Biomol. Struct. 1992, 21, 441–483. [Google Scholar] [CrossRef]
- Anderson, H.C. Matrix vesicles and calcification. Curr. Rheumatol. Rep. 2003, 5, 222–226. [Google Scholar] [CrossRef]
- Ciancaglini, P.; Pizauro, J.M.; Grecchi, M.J.; Curti, C.; Leone, F.A. Effect of Zn(II) and Mg(II) on phosphohydrolytic activity of rat matrix-induced alkaline phosphatase. Cell. Mol. Boil. 1989, 35, 503–510. [Google Scholar]
- Starcher, B.; Kratzer, F.H. Effect of Zinc on Bone Alkaline Phosphatase in Turkey Poults. J. Nutr. 1963, 79, 18–22. [Google Scholar] [CrossRef] [PubMed]
- Clancaglini, P.; Plzauro, J.M.; Curti, C.; Tedesco, A.C.; Leone, F.A. Effect of membrane moiety and magnesium ions on the inhibition of matrix-induced alkaline phosphatase by zinc ions. Int. J. Biochem. 1990, 22, 747–751. [Google Scholar] [CrossRef]
- Ciancaglini, P.; Pizauro, J.; Rezende, A.; Rezende, L.; Leone, F. Solubilization of membrane-bound matrix-induced alkaline phosphatase with polyoxyethylene 9-lauryl ether (polidocanol): Purification and metalloenzyme properties. Int. J. Biochem. 1990, 22, 385–392. [Google Scholar] [CrossRef]
- King, J.C.; Shames, D.M.; Woodhouse, L.R. Zinc Homeostasis in Humans. J. Nutr. 2000, 130, 1360S–1366S. [Google Scholar] [CrossRef] [PubMed]
- Taylor, C.M.; Bacon, J.R.; Aggett, P.J.; Bremner, I. Homeostatic regulation of zinc absorption and endogenous losses in zinc-deprived men. Am. J. Clin. Nutr. 1991, 53, 755–763. [Google Scholar] [CrossRef] [PubMed]
- Maares, M.; Haase, H. A Guide to Human Zinc Absorption: General Overview and Recent Advances of In Vitro Intestinal Models. Nutrients 2020, 12, 762. [Google Scholar] [CrossRef]
- Weigand, E.; Kirchgessner, M. Homeostatic Adjustments in Zinc Digestion to Widely Varying Dietary Zinc Intake. Ann. Nutr. Metab. 1978, 22, 101–112. [Google Scholar] [CrossRef]
- Johnson, P.E.; Hunt, C.D.; Milne, D.B.; Mullen, L.K. Homeostatic control of zinc metabolism in men: Zinc excretion and balance in men fed diets low in zinc. Am. J. Clin. Nutr. 1993, 57, 557–565. [Google Scholar] [CrossRef]
- Zhang, S.-Q.; Yu, X.-F.; Zhang, H.-B.; Peng, N.; Chen, Z.-X.; Cheng, Q.; Zhang, X.-L.; Cheng, S.-H.; Zhang, Y. Comparison of the Oral Absorption, Distribution, Excretion, and Bioavailability of Zinc Sulfate, Zinc Gluconate, and Zinc-Enriched Yeast in Rats. Mol. Nutr. Food Res. 2018, 62, 1700981. [Google Scholar] [CrossRef] [PubMed]
- Jackson, M.J.; Jones, D.A.; Edwards, R.H.T. Tissue zinc levels as an index of body zinc status. Clin. Physiol. 1982, 2, 333–343. [Google Scholar] [CrossRef] [PubMed]
- Windisch, W. Development of zinc deficiency in 65Zn labeled, fully grown rats as a model for adult individuals. J. Trace Elements Med. Boil. 2003, 17, 91–96. [Google Scholar] [CrossRef]
- Windisch, W.; Wher, U.; Rambeck, W.; Erben, R. Effect of Zn deficiency and subsequent Zn repletion on bone mineral composition and markers of bone tissue metabolism in 65Zn-labelled, young-adult rats. J. Anim. Physiol. Anim. Nutr. 2002, 86, 214–221. [Google Scholar] [CrossRef] [PubMed]
- Park, J.H.Y.; Grandjean, C.J.; Antonson, D.L.; Vanderhoof, J.A. Effects of Isolated Zinc Deficiency on the Composition of Skeletal Muscle, Liver and Bone during Growth in Rats. J. Nutr. 1986, 116, 610–617. [Google Scholar] [CrossRef] [PubMed]
- Murray, E.J.; Messer, H.H. Turnover of Bone Zinc during Normal and Accelerated Bone Loss in Rats. J. Nutr. 1981, 111, 1641–1647. [Google Scholar] [CrossRef]
- Ovesen, J.; Møller-Madsen, B.; Nielsen, P.T.; Christensen, P.H.; Simonsen, O.; Hoeck, H.C.; Laursen, M.; Thomsen, J. Differences in zinc status between patients with osteoarthritis and osteoporosis. J. Trace Elements Med. Boil. 2009, 23, 1–8. [Google Scholar] [CrossRef]
- Lowe, N.; Fraser, W.D.; Jackson, M.J. Is there a potential therapeutic value of copper and zinc for osteoporosis? Proc. Nutr. Soc. 2002, 61, 181–185. [Google Scholar] [CrossRef]
- Contreras, F.; Simonovis, N.; Fouillioux, C.; Bolívar, A.; Cevallos, J.L.; Lezama, E.; Velasco, M. Zincuria and zincemia in postmenopausal osteoporosis. Int. Congr. Ser. 2002, 1237, 219–229. [Google Scholar] [CrossRef]
- Relea, P.; Revilla, M.; Ripoll, E.; Arribas, I.; Villa, L.F.; Rico, H. Zinc, Biochemical Markers of Nutrition, and Type I Osteoporosis. Age Ageing 1995, 24, 303–307. [Google Scholar] [CrossRef]
- Maret, W. Zinc in Pancreatic Islet Biology, Insulin Sensitivity, and Diabetes. Prev. Nutr. Food Sci. 2017, 22, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Moran, B.M.; Miskelly, M.; Abdel-Wahab, Y.H.A.; Flatt, P.R.; McKillop, A.M. Zinc-induced activation of GPR39 regulates glucose homeostasis through glucose-dependent insulinotropic polypeptide secretion from enteroendocrine K-cells. Boil. Chem. 2019, 400, 1023–1033. [Google Scholar] [CrossRef] [PubMed]
- Gaetke, L.M.; McClain, C.J.; Talwalkar, R.T.; Shedlofsky, S.I. Effects of endotoxin on zinc metabolism in human volunteers. Am. J. Physiol. Content 1997, 272, E952–E956. [Google Scholar] [CrossRef]
- Boosalis, M.G.; Solem, L.D.; McCall, J.T.; Ahrenholz, D.H.; McClain, C.J. Serum zinc response in thermal injury. J. Am. Coll. Nutr. 1988, 7, 69–76. [Google Scholar] [CrossRef] [PubMed]
- Pekarek, R.S.; Beisel, W.R. Effect of Endotoxin on Serum Zinc Concentrations in the Rat. Appl. Microbiol. 1969, 18, 482–484. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Gill, P.S.; Rattan, P.J. Variations in plasma trace-elements concentration during fracture healing in dogs. Indian J. Physiol. Pharmacol. 1991, 35. [Google Scholar]
- Myers, M.A.; Fleck, A.; Sampson, B.; Colley, C.M.; Bent, J.; Hall, G. Early plasma protein and mineral changes after surgery: A two stage process. J. Clin. Pathol. 1984, 37, 862–866. [Google Scholar] [CrossRef]
- Calhoun, N.R.; Campbell, S.; Smith, J.C. Accumulation of Labeled Zinc, Strontium, and Calcium in Bone Injuries. J. Dent. Res. 1970, 49, 1083–1085. [Google Scholar] [CrossRef]
- Savlov, E.D.; Strain, W.H.; Huegin, F. Radiozinc studies in experimental wound healing. J. Surg. Res. 1962, 2, 209–212. [Google Scholar] [CrossRef]
- Calhoun, N.; Smith, J. Uptake of 65Zn in fractured bones. Lancet 1968, 292, 682. [Google Scholar] [CrossRef]
- Senapati, A.; Thompson, R.P.H. Zinc deficiency and the prolonged accumulation of zinc in wounds. BJS 1985, 72, 583–584. [Google Scholar] [CrossRef] [PubMed]
- Iwata, M.; Takebayashi, T.; Ohta, H.; Alcalde, R.E.; Itano, Y.; Matsumura, T. Zinc accumulation and metallothionein gene expression in the proliferating epidermis during wound healing in mouse skin. Histochem. Cell Boil. 1999, 112, 283–290. [Google Scholar] [CrossRef] [PubMed]
- Wessels, I.; Maywald, M.; Rink, L. Zinc as a Gatekeeper of Immune Function. Nutrients 2017, 9, 1286. [Google Scholar] [CrossRef] [PubMed]
- Gao, H.; Dai, W.; Zhao, L.; Min, J.; Wang, F. The Role of Zinc and Zinc Homeostasis in Macrophage Function. J. Immunol. Res. 2018, 2018, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Qi, S.; He, J.; Zheng, H.X.; Chen, C.; Jiang, H.; Lan, S. Zinc Supplementation Increased Bone Mineral Density, Improves Bone Histomorphology, and Prevents Bone Loss in Diabetic Rat. Boil. Trace Element Res. 2019, 194, 493–501. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, M.; Kishi, S.; Hashizume, M. Effect of zinc-chelating dipeptides on osteoblastic MC3T3-E1 cells: Activation of aminoacyl-tRNA synthetase. Peptides 1994, 15, 1367–1371. [Google Scholar] [CrossRef]
- Ito, A.; Ojima, K.; Naito, H.; Ichinose, N.; Tateishi, T. Preparation, solubility, and cytocompatibility of zinc-releasing calcium phosphate ceramics. J. Biomed. Mater. Res. 2000, 50, 178–183. [Google Scholar] [CrossRef]
- Ishikawa, K.; Miyamoto, Y.; Yuasa, T.; Ito, A.; Nagayama, M.; Suzuki, K. Fabrication of Zn containing apatite cement and its initial evaluation using human osteoblastic cells. Biomaterials 2002, 23, 423–428. [Google Scholar] [CrossRef]
- Pina, S.; Vieira, S.I.; Rego, P.A.; Torres, P.M.C.; da Cruz e Silva, O.A.B.; da Cruz e Silva, E.F.; Ferreira, J. Biological responses of brushite-forming Zn- and ZnSr- substituted beta-tricalcium phosphate bone cements. Eur. Cells Mater. 2010, 20, 162–177. [Google Scholar] [CrossRef]
- Seo, H.-J.; Cho, Y.-E.; Kim, T.; Shin, H.-I.; Kwun, I.-S. Zinc may increase bone formation through stimulating cell proliferation, alkaline phosphatase activity and collagen synthesis in osteoblastic MC3T3-E1 cells. Nutr. Res. Pr. 2010, 4, 356–361. [Google Scholar] [CrossRef]
- Alcantara, E.H.; Lomeda, R.-A.R.; Feldmann, J.; Nixon, G.; Beattie, J.H.; Kwun, I.-S. Zinc deprivation inhibits extracellular matrix calcification through decreased synthesis of matrix proteins in osteoblasts. Mol. Nutr. Food Res. 2011, 55, 1552–1560. [Google Scholar] [CrossRef] [PubMed]
- Horiuchi, S.; Hiasa, M.; Yasue, A.; Sekine, K.; Hamada, K.; Asaoka, K.; Tanaka, E. Fabrications of zinc-releasing biocement combining zinc calcium phosphate to calcium phosphate cement. J. Mech. Behav. Biomed. Mater. 2014, 29, 151–160. [Google Scholar] [CrossRef]
- Togari, A.; Arakawa, S.; Arai, M.; Matsumoto, S. Alteration of in vitro bone metabolism and tooth formation by zinc. Gen. Pharmacol. Vasc. Syst. 1993, 24, 1133–1140. [Google Scholar] [CrossRef]
- Kwun, I.-S.; Cho, Y.-E.; Lomeda, R.-A.R.; Shin, H.-I.; Choi, J.-Y.; Kang, Y.-H.; Beattie, J.H. Zinc deficiency suppresses matrix mineralization and retards osteogenesis transiently with catch-up possibly through Runx 2 modulation. Bone 2010, 46, 732–741. [Google Scholar] [CrossRef] [PubMed]
- Dimai, H.P.; Hall, S.L.; Stilt-Coffing, B.; Farley, J.R. Skeletal response to dietary zinc in adult female mice. Calcif. Tissue Int. 1998, 62, 309–315. [Google Scholar] [CrossRef] [PubMed]
- Bortolin, R.H.; da Graça Azevedo Abreu, B.J.; Ururahy, M.A.G.; De Souza, K.S.C.; Bezerra, J.; Loureiro, M.B.; Da Silva, F.S.; da Silva Marques, D.E.; de Sousa Batista, A.A.; Oliveira, G.; et al. Protection against T1DM-Induced Bone Loss by Zinc Supplementation: Biomechanical, Histomorphometric, and Molecular Analyses in STZ-Induced Diabetic Rats. PLoS ONE 2015, 10, e0125349. [Google Scholar] [CrossRef]
- Ito, A.; Kawamura, H.; Otsuka, M.; Ikeuchi, M.; Ohgushi, H.; Ishikawa, K.; Onuma, K.; Kanzaki, N.; Sogo, Y.; Ichinose, N. Zinc-releasing calcium phosphate for stimulating bone formation. Mater. Sci. Eng. C 2002, 22, 21–25. [Google Scholar] [CrossRef]
- Cerovic, A.; Miletić, I.; Sobajic, S.; Blagojevic, D.; Radusinovic, M.; El-Sohemy, A. Effects of zinc on the mineralization of bone nodules from human osteoblast-like cells. Boil. Trace Element Res. 2007, 116, 61–71. [Google Scholar] [CrossRef]
- Suh, K.S.; Lee, Y.S.; Seo, S.H.; Kim, Y.S.; Choi, E.M. Effect of Zinc Oxide Nanoparticles on the Function of MC3T3-E1 Osteoblastic Cells. Boil. Trace Element Res. 2013, 155, 287–294. [Google Scholar] [CrossRef]
- Jin, G.; Cao, H.; Qiao, Y.; Meng, F.; Zhu, H.; Liu, X. Osteogenic activity and antibacterial effect of zinc ion implanted titanium. Colloids Surfaces B Biointerfaces 2014, 117, 158–165. [Google Scholar] [CrossRef]
- Srivastava, S.; Kumar, N.; Thakur, R.S.; Roy, P. Role of Vanadium (V) in the Differentiation of C3H10t1/2 Cells Towards Osteoblast Lineage: A Comparative Analysis with Other Trace Elements. Boil. Trace Element Res. 2013, 152, 135–142. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, M.; Goto, M.; Uchiyama, S.; Nakagawa, T. Effect of zinc on gene expression in osteoblastic MC3T3-E1 cells: Enhancement of Runx2, OPG, and regucalcin mRNA expressions. Mol. Cell. Biochem. 2008, 312, 157–166. [Google Scholar] [CrossRef] [PubMed]
- Hu, D.; Li, K.; Xie, Y.; Pan, H.; Zhao, J.; Huang, L.; Zheng, X. Different response of osteoblastic cells to Mg2+, Zn2+ and Sr2+ doped calcium silicate coatings. J. Mater. Sci. Mater. Electron. 2016, 27, 56. [Google Scholar] [CrossRef] [PubMed]
- Yusa, K.; Yamamoto, O.; Takano, H.; Fukuda, M.; Iino, M. Zinc-modified titanium surface enhances osteoblast differentiation of dental pulp stem cells in vitro. Sci. Rep. 2016, 6, 29462. [Google Scholar] [CrossRef]
- Cho, Y.-E.; Kwun, I.-S. Zinc upregulates bone-specific transcription factor Runx2 expression via BMP-2 signaling and Smad-1 phosphorylation in osteoblasts. J. Nutr. Heal. 2018, 51, 23–30. [Google Scholar] [CrossRef]
- Park, K.H.; Choi, Y.; Yoon, D.S.; Lee, K.-M.; Kim, H.; Lee, J.W. Zinc Promotes Osteoblast Differentiation in Human Mesenchymal Stem Cells Via Activation of the cAMP-PKA-CREB Signaling Pathway. Stem Cells Dev. 2018, 27, 1125–1135. [Google Scholar] [CrossRef]
- Ducy, P.; Zhang, R.; Geoffroy, V.; Ridall, A.L.; Karsenty, G. Osf2/Cbfa1: A transcriptional activator of osteoblast differentiation. Cell 1997, 89, 747–754. [Google Scholar] [CrossRef] [PubMed]
- Komori, T.; Yagi, H.; Nomura, S.; Yamaguchi, A.; Sasaki, K.; Deguchi, K.; Shimizu, Y.; Bronson, R.T.; Gao, Y.-H.; Inada, M.; et al. Targeted Disruption of Cbfa1 Results in a Complete Lack of Bone Formation owing to Maturational Arrest of Osteoblasts. Cell 1997, 89, 755–764. [Google Scholar] [CrossRef]
- Uchiyama, S.; Yamaguchi, M. Genistein and zinc synergistically enhance gene expression and mineralization in osteoblastic MC3T3-E1 cells. Int. J. Mol. Med. 2007, 19, 213–220. [Google Scholar] [CrossRef][Green Version]
- Guo, B.; Yang, M.-W.; Liang, D.; Yang, L.; Cao, J.; Zhang, L. Cell apoptosis induced by zinc deficiency in osteoblastic MC3T3-E1 cells via a mitochondrial-mediated pathway. Mol. Cell. Biochem. 2011, 361, 209–216. [Google Scholar] [CrossRef]
- Wang, C.; Youle, R.J. The role of mitochondria in apoptosis. Annu. Rev. Genet. 2009, 43, 95–118. [Google Scholar] [CrossRef] [PubMed]
- Kirschke, C.P.; Huang, L. ZnT7, a Novel Mammalian Zinc Transporter, Accumulates Zinc in the Golgi Apparatus. J. Boil. Chem. 2002, 278, 4096–4102. [Google Scholar] [CrossRef] [PubMed]
- Kanno, S.; Anuradha, C.D.; Hirano, S. Chemotactic Responses of Osteoblastic MC3T3-E1 Cells Toward Zinc Chloride. Boil. Trace Element Res. 2001, 83, 49–55. [Google Scholar] [CrossRef]
- Kishi, S.; Yamaguchi, M. Inhibitory effect of zinc compounds on osteoclast-like cell formation in mouse marrow cultures. Biochem. Pharmacol. 1994, 48, 1225–1230. [Google Scholar] [CrossRef]
- Yamaguchi, M.; Kishi, S. Differential effects of transforming growth factor-beta on osteoclast-like cell formation in mouse marrow culture: Relation to the effect of zinc-chelating dipeptides. Peptides 1995, 16, 1483–1488. [Google Scholar] [CrossRef]
- Yamaguchi, M.; Kishi, S. Zinc compounds inhibit osteoclast-like cell formation at the earlier stage of rat marrow culture but not osteoclast function. Mol. Cell. Biochem. 1996, 158. [Google Scholar] [CrossRef]
- Kishi, S.; Yamaguchi, M. Characterization of zinc effect to inhibit osteoclast-like cell formation in mouse marrow culture: Interaction with dexamethasone. Mol. Cell. Biochem. 1997, 166, 145–151. [Google Scholar] [CrossRef]
- Yamaguchi, M.; Uchiyama, S. Receptor activator of NF-kappaB ligand-stimulated osteoclastogenesis in mouse marrow culture is suppressed by zinc in vitro. Int. J. Mol. Med. 2004, 14, 81–85. [Google Scholar]
- Khadeer, M.A.; Sahu, S.N.; Bai, G.; Abdulla, S.; Gupta, A. Expression of the zinc transporter ZIP1 in osteoclasts. Bone 2005, 37, 296–304. [Google Scholar] [CrossRef]
- Hie, M.; Tsukamoto, I. Administration of zinc inhibits osteoclastogenesis through the suppression of RANK expression in bone. Eur. J. Pharmacol. 2011, 668, 140–146. [Google Scholar] [CrossRef]
- Yamaguchi, M.; Weitzmann, M.N. Zinc stimulates osteoblastogenesis and suppresses osteoclastogenesis by antagonizing NF-κB activation. Mol. Cell. Biochem. 2011, 355, 179–186. [Google Scholar] [CrossRef] [PubMed]
- Moonga, B.S.; Dempster, D.W. Zinc is a potent inhibitor of osteoclastic bone resorption in vitro. J. Bone Miner. Res. 2009, 10, 453–457. [Google Scholar] [CrossRef] [PubMed]
- Roy, M.; Fielding, G.A.; Bandyopadhyay, A.; Bose, S. Effects of Zinc and Strontium Substitution in Tricalcium Phosphate on Osteoclast Differentiation and Resorption. Biomater. Sci. 2013, 1, 74–82. [Google Scholar] [CrossRef] [PubMed]
- Shepherd, D.; Kauppinen, K.; Brooks, R.A.; Best, S.M. An in vitro study into the effect of zinc substituted hydroxyapatite on osteoclast number and activity. J. Biomed. Mater. Res. Part A 2014, 102, 4136–4141. [Google Scholar] [CrossRef] [PubMed]
- Takayanagi, H.; Kim, S.; Koga, T.; Nishina, H.; Isshiki, M.; Yoshida, H.; Saiura, A.; Isobe, M.; Yokochi, T.; Inoue, J.-I.; et al. Induction and Activation of the Transcription Factor NFATc1 (NFAT2) Integrate RANKL Signaling in Terminal Differentiation of Osteoclasts. Dev. Cell 2002, 3, 889–901. [Google Scholar] [CrossRef]
- Ikeda, F.; Nishimura, R.; Matsubara, T.; Tanaka, S.; Inoue, J.I.; Reddy, S.V.; Hata, K.; Yamashita, K.; Hiraga, T.; Watanabe, T.; et al. Critical roles of c-Jun signaling in regulation of NFAT family and RANKL-regulated osteoclast differentiation. J. Clin. Investig. 2004, 114, 475–484. [Google Scholar] [CrossRef]
- Mohamed, S.G.K.; Sugiyama, E.; Shinoda, K.; Hounoki, H.; Taki, H.; Maruyama, M.; Miyahara, T.; Kobayashi, M. Interleukin-4 inhibits RANKL-induced expression of NFATc1 and c-Fos: A possible mechanism for downregulation of osteoclastogenesis. Biochem. Biophys. Res. Commun. 2005, 329, 839–845. [Google Scholar] [CrossRef]
- Mohamed, S.G.-K.; Sugiyama, E.; Shinoda, K.; Taki, H.; Hounoki, H.; Abdel-Aziz, H.O.; Maruyama, M.; Kobayashi, M.; Ogawa, H.; Miyahara, T. Interleukin-10 inhibits RANKL-mediated expression of NFATc1 in part via suppression of c-Fos and c-Jun in RAW264.7 cells and mouse bone marrow cells. Bone 2007, 41, 592–602. [Google Scholar] [CrossRef]
- Franzoso, G.; Carlson, L.; Xing, L.; Poljak, L.; Shores, E.W.; Brown, K.D.; Leonardi, A.; Tran, T.; Boyce, B.F.; Siebenlist, U. Requirement for NF-kappa B in osteoclast and B-cell development. Genes Dev. 1997, 11, 3482–3496. [Google Scholar] [CrossRef]
- Fong, L.; Tan, K.; Tran, C.; Cool, J.; Scherer, M.; Elovaris, R.; Coyle, P.; Foster, B.K.; Rofe, A.M.; Xian, C.J. Interaction of dietary zinc and intracellular binding protein metallothionein in postnatal bone growth. Bone 2009, 44, 1151–1162. [Google Scholar] [CrossRef]
- Sasaki, S.; Tsukamoto, M.; Saito, M.; Hojyo, S.; Fukada, T.; Takami, M.; Furuichi, T. Disruption of the mouse Slc39a14 gene encoding zinc transporter ZIP14 is associated with decreased bone mass, likely caused by enhanced bone resorption. FEBS Open Bio 2018, 8, 655–663. [Google Scholar] [CrossRef] [PubMed]
- Aydemir, T.B.; Cousins, R.J. The Multiple Faces of the Metal Transporter ZIP14 (SLC39A14). J. Nutr. 2018, 148, 174–184. [Google Scholar] [CrossRef] [PubMed]
- Jovanovic, M.; Schmidt, F.N.; Ram, G.G.; Khayyeri, H.; Hiram-Bab, S.; Orenbuch, A.; Katchkovsky, S.; Aflalo, A.; Isaksson, H.; Busse, B.; et al. Perturbed bone composition and integrity with disorganized osteoblast function in zinc receptor/Gpr39-deficient mice. FASEB J. 2018, 32, 2507–2518. [Google Scholar] [CrossRef] [PubMed]
- Holloway, W.; Collier, F.; Herbst, R.; Hodge, J.; Nicholson, G.C. Osteoblast-mediated effects of zinc on isolated rat osteoclasts: Inhibition of bone resorption and enhancement of osteoclast number. Bone 1996, 19, 137–142. [Google Scholar] [CrossRef]
- Yamada, Y.; Ito, A.; Kojima, H.; Sakane, M.; Miyakawa, S.; Uemura, T.; LeGeros, R.Z. Inhibitory effect of Zn2+ in zinc-containing β-tricalcium phosphate on resorbing activity of mature osteoclasts. J. Biomed. Mater. Res. Part A 2007, 84, 344–352. [Google Scholar] [CrossRef]
- Li, X.; Senda, K.; Ito, A.; Sogo, Y.; Yamazaki, A. Effect of Zn and Mg in tricalcium phosphate and in culture medium on apoptosis and actin ring formation of mature osteoclasts. Biomed. Mater. 2008, 3, 45002. [Google Scholar] [CrossRef]
- Bhardwaj, A.; Swe, K.M.M.; Sinha, N.K.; Osunkwo, I. Treatment for osteoporosis in people with ß-thalassaemia. Cochrane Database Syst. Rev. 2016, 3. [Google Scholar] [CrossRef]
- Bhardwaj, P.; Rai, D.V.; Garg, M.L. Zinc as a nutritional approach to bone loss prevention in an ovariectomized rat model. Menopause 2013, 20, 1184–1193. [Google Scholar] [CrossRef]
- Yamaguchi, M. Role of nutritional zinc in the prevention of osteoporosis. Mol. Cell. Biochem. 2009, 338, 241–254. [Google Scholar] [CrossRef]
- Shepherd, D. Zinc-substituted hydroxyapatite for the inhibition of osteoporosis. Hydroxyapatite (Hap) Biomed. Appl. 2015, 95, 107–126. [Google Scholar]
- Hinton, R.; Jing, Y.; Jing, J.; Feng, J.Q. Roles of Chondrocytes in Endochondral Bone Formation and Fracture Repair. J. Dent. Res. 2016, 96, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Kronenberg, H.M. Developmental regulation of the growth plate. Nature 2003, 423, 332–336. [Google Scholar] [CrossRef]
- Sun, M.M.-G.; Beier, F. Chondrocyte hypertrophy in skeletal development, growth, and disease. Birth Defects Res. Part C Embryo Today Rev. 2014, 102, 74–82. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Fosmire, G.J.; Gay, C.V.; Leach, R.M. Short-Term Zinc Deficiency Inhibits Chondrocyte Proliferation and Induces Cell Apoptosis in the Epiphyseal Growth Plate of Young Chickens. J. Nutr. 2002, 132, 665–673. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez, J.P.; Rosselot, G. Effects of zinc on cell proliferation and proteoglycan characteristics of epiphyseal chondrocytes. J. Cell. Biochem. 2001, 82, 501–511. [Google Scholar] [CrossRef] [PubMed]
- Kirkpatrick, J.; Mohr, W.; Haferkamp, O.; Kirkpatrick, C. Influence of zinc and copper on lapine articular chondrocytes in monolayer culture: Morphology, proliferation and proteoglycan synthesis. Exp. Cell Biol. 1982, 50, 108–114. [Google Scholar] [CrossRef]
- Burgess, D.; Cottrell, J.; Iverson, T. Zinc chloride treatment in ATDC5 cells induces chondrocyte maturation. Int. J. Regen. Med. 2018, 1, 1–11. [Google Scholar] [CrossRef]
- Huang, T.-C.; Chang, W.-T.; Hu, Y.-C.; Hsieh, B.-S.; Cheng, H.-L.; Yen, J.-H.; Chiu, P.-R.; Chang, K.-L. Zinc Protects Articular Chondrocytes through Changes in Nrf2-Mediated Antioxidants, Cytokines and Matrix Metalloproteinases. Nutrients 2018, 10, 471. [Google Scholar] [CrossRef]
- Pasqualicchio, M.; Gasperini, R.; Velo, G.P.; Davies, M.E. Effects of copper and zinc on proteoglycan metabolism in articular cartilage. Mediat. Inflamm. 1996, 5, 95–99. [Google Scholar] [CrossRef]
- Litchfield, T.M.; Ishikawa, Y.; Wu, L.N.Y.; Wuthier, R.E.; Sauer, G.R. Effect of metal ions on calcifying growth plate cartilage chondrocytes. Calcif. Tissue Int. 1998, 62, 341–349. [Google Scholar] [CrossRef]
- Kirsch, T.; Harrison, G.; Worch, K.P.; Golub, E.E. Regulatory Roles of Zinc in Matrix Vesicle-Mediated Mineralization of Growth Plate Cartilage. J. Bone Miner. Res. 2010, 15, 261–270. [Google Scholar] [CrossRef] [PubMed]
- Koyano, Y.; Hejna, M.; Flechtenmacher, J.; Schmid, T.M.; Thonar, E.J.-M.A.; Mollenhauer, J. Collagen and Proteoglycan Production by Bovine Fetal and Adult Chondrocytes Under Low Levels of Calcium and Zinc Ions. Connect. Tissue Res. 1996, 34, 213–225. [Google Scholar] [CrossRef] [PubMed]
- Prasad, A.S. Zinc and immunity. Mol. Cell. Biochem. 1998, 188, 63–69. [Google Scholar] [CrossRef] [PubMed]
- Bao, B.; Prasad, A.S.; Beck, F.W.J.; Fitzgerald, J.T.; Snell, D.; Bao, G.W.; Singh, T.; Cardozo, L.J. Zinc decreases C-reactive protein, lipid peroxidation, and inflammatory cytokines in elderly subjects: A potential implication of zinc as an atheroprotective agent. Am. J. Clin. Nutr. 2010, 91, 1634–1641. [Google Scholar] [CrossRef]
- Bao, B.; Prasad, A.S.; Beck, F.W.; Snell, D.; Suneja, A.; Sarkar, F.H.; Doshi, N.; Fitzgerald, J.T.; Swerdlow, P. Zinc supplementation decreases oxidative stress, incidence of infection, and generation of inflammatory cytokines in sickle cell disease patients. Transl. Res. 2008, 152, 67–80. [Google Scholar] [CrossRef]
- Prasad, A.S.; Bao, B. Molecular Mechanisms of Zinc as a Pro-Antioxidant Mediator: Clinical Therapeutic Implications. Antioxidants 2019, 8, 164. [Google Scholar] [CrossRef]
- Kim, J.-H.; Jeon, J.; Shin, M.; Won, Y.; Lee, M.; Kwak, J.-S.; Lee, G.; Rhee, J.; Ryu, J.-H.; Chun, C.-H.; et al. Regulation of the Catabolic Cascade in Osteoarthritis by the Zinc-ZIP8-MTF1 Axis. Cell 2014, 156, 730–743. [Google Scholar] [CrossRef]
- Agnew, U.M.; Slesinger, T.L. Zinc Toxicity. In StatPearls. Treasure Island (FL); StatPearls Publishing LLC.: Treasure Island, FL, USA, 2020. [Google Scholar]
- Calhoun, N.R.; Smith, J.; Becker, K.L. The effects of zinc on ectopic bone formation. Oral Surgery Oral Med. Oral Pathol. 1975, 39, 698–706. [Google Scholar] [CrossRef]
- Andreen, O.; Larsson, S.-E. Effects of 1,25-dihydroxycholecalciferol on fracture healing calcium, phosphate, and zinc in callus and serum. Arch. Orthop. Trauma Surg. 1984, 103, 257–262. [Google Scholar] [CrossRef]
- Sadighi, A.; Roshan, M.M.; Moradi, A.; Ostadrahimi, A. The effects of zinc supplementation on serum zinc, alkaline phosphatase activity and fracture healing of bones. Saudi Med. J. 2008, 29, 1276–1279. [Google Scholar]
- McCray, L.A.; Higa, L.H.; Soni, N.N. The effect of orally administered zinc sulfate on extraction wound healing in hamsters. Oral Surg. Oral Med. Oral Pathol. 1972, 33, 314–322. [Google Scholar] [CrossRef]
- Battistone, G.C.; Posey, W.R.; Barone, J.J.; Cutright, D.E. Zinc and bone healing: The effect of zinc cysteamine-N-acetic acid on the healing of extraction wounds in rats. Oral Surg. Oral Med. Oral Pathol. 1972, 34, 704–711. [Google Scholar] [CrossRef]
- Igarashi, A.; Yamaguchi, M. Stimulatory effect of zinc acexamate administration on fracture healing of the femoral-diaphyseal tissues in rats. Gen. Pharmacol. Vasc. Syst. 1999, 32, 463–469. [Google Scholar] [CrossRef]
- Belanger, L.F.; Casas-Cordero, M.; Urist, M.R. The Effects of Zinc Deprivation on the Host Response to Intramuscular Bone Matrix Implants in the Rat. Clin. Orthop. Relat. Res. 1977, 208–213. [Google Scholar] [CrossRef]
- Milachowski, K.; Moschinski, D.; Jaeschock, R.; Kaschner, A. The influence of zinc on bone healing in rats. Arch. Orthop. Trauma Surg. 1980, 96, 17–21. [Google Scholar] [CrossRef]
- Abrisham, S.M.; Yaghmaei, M.; Abbas, F.M.; Sharifi, D.; Abrisham, S.M.J. Effect of Oral Zinc Therapy on Osteogenesis in Rabbits. J. Oral Maxillofac. Surg. 2010, 68, 1676–1680. [Google Scholar] [CrossRef]
- Jones, L.; Thomsen, J.; Barlach, J.; Mosekilde, L.; Melsen, B. No influence of alimentary zinc on the healing of calvarial defects filled with osteopromotive substances in rats. Eur. J. Orthod. 2010, 32, 124–130. [Google Scholar] [CrossRef]
- Alvarez, K.; Fukuda, M.; Yamamoto, O. Titanium Implants after Alkali Heating Treatment with a [Zn(OH)4]2− Complex: Analysis of Interfacial Bond Strength Using Push-Out Tests. Clin. Implant. Dent. Relat. Res. 2009, 12, e114–e125. [Google Scholar] [CrossRef]
- He, J.; Feng, W.; Zhao, B.-H.; Zhang, W.; Lin, Z. In Vivo Effect of Titanium Implants with Porous Zinc-Containing Coatings Prepared by Plasma Electrolytic Oxidation Method on Osseointegration in Rabbits. Int. J. Oral Maxillofac. Implant. 2018, 33, 298–310. [Google Scholar] [CrossRef]
- Zhao, S.-F.; Dong, W.-J.; Jiang, Q.-H.; He, F.-M.; Wang, X.-X.; Yang, G.-L. Effects of zinc-substituted nano-hydroxyapatite coatings on bone integration with implant surfaces*. J. Zhejiang Univ. Sci. B 2013, 14, 518–525. [Google Scholar] [CrossRef]
- Qiao, Y.; Zhang, W.; Tian, P.; Meng, F.; Zhu, H.; Jiang, X.; Liu, X.; Chu, P.K. Stimulation of bone growth following zinc incorporation into biomaterials. Biomaterials 2014, 35, 6882–6897. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Xiong, W.; Zhang, C.; Gao, B.; Guan, H.; Cheng, H.; Fu, J.; Li, F. Enhanced osseointegration and antibacterial action of zinc-loaded titania-nanotube-coated titanium substrates:In vitroandin vivostudies. J. Biomed. Mater. Res. Part A 2013, 102, 3939–3950. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Li, Y.; Peng, S.; Ye, B.; Lin, W.; Hu, J. Effect of zinc ions on improving implant fixation in osteoporotic bone. Connect. Tissue Res. 2013, 54, 290–296. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Li, K.; Zheng, X.; He, D.; Ye, X.; Wang, M. In Vitro and In Vivo Evaluation of Zinc-Modified Ca–Si-Based Ceramic Coating for Bone Implants. PLoS ONE 2013, 8, e57564. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Xu, L.; Li, K.; Xie, N.; Xi, Y.; Wang, Y.; Zheng, X.; Chen, X.; Wang, M.; Ye, X. Zinc-modified Calcium Silicate Coatings Promote Osteogenic Differentiation through TGF-β/Smad Pathway and Osseointegration in Osteopenic Rabbits. Sci. Rep. 2017, 7, 3440. [Google Scholar] [CrossRef] [PubMed]
- Tao, Z.-S.; Zhou, W.-S.; He, X.-W.; Liu, W.; Bai, B.-L.; Zhou, Q.; Huang, Z.-L.; Tu, K.-K.; Li, H.; Sun, T.; et al. A comparative study of zinc, magnesium, strontium-incorporated hydroxyapatite-coated titanium implants for osseointegration of osteopenic rats. Mater. Sci. Eng. C 2016, 62, 226–232. [Google Scholar] [CrossRef]
- Chou, J.; Hao, J.; Kuroda, S.; Bishop, D.P.; Ben-Nissan, B.; Milthorpe, B.K.; Otsuka, M. Bone Regeneration of Rat Tibial Defect by Zinc-Tricalcium Phosphate (Zn-TCP) Synthesized from Porous Foraminifera Carbonate Macrospheres. Mar. Drugs 2013, 11, 5148–5158. [Google Scholar] [CrossRef]
- Yu, W.; Sun, T.-W.; Qi, C.; Ding, Z.; Zhao, H.; Zhao, S.; Shi, Z.; Zhu, Y.-J.; Chen, D.; He, Y. Evaluation of zinc-doped mesoporous hydroxyapatite microspheres for the construction of a novel biomimetic scaffold optimized for bone augmentation. Int. J. Nanomed. 2017, 12, 2293–2306. [Google Scholar] [CrossRef]
- Kawamura, H.; Ito, A.; Miyakawa, S.; Layrolle, P.; Ojima, K.; Ichinose, N.; Tateishi, T. Stimulatory effect of zinc-releasing calcium phosphate implant on bone formation in rabbit femora. J. Biomed. Mater. Res. 2000, 50, 184–190. [Google Scholar] [CrossRef]
- Kawamura, H.; Ito, A.; Muramatsu, T.; Miyakawa, S.; Ochiai, N.; Tateishi, T. Long-term implantation of zinc-releasing calcium phosphate ceramics in rabbit femora. J. Biomed. Mater. Res. 2003, 65, 468–474. [Google Scholar] [CrossRef]
- Luo, X.; Barbieri, D.; Davison, N.; Yan, Y.; De Bruijn, J.; Yuan, H. Zinc in calcium phosphate mediates bone induction: In vitro and in vivo model. Acta Biomater. 2014, 10, 477–485. [Google Scholar] [CrossRef] [PubMed]
- Cruz, R.; Calasans-Maia, J.A.; Sartoretto, S.; Moraschini, V.; Rossi, A.M.; Louro, R.S.; Granjeiro, J.M.; Calasans-Maia, M.D. Does the incorporation of zinc into calcium phosphate improve bone repair? A systematic review. Ceram. Int. 2018, 44, 1240–1249. [Google Scholar] [CrossRef]
- Bhardwaj, V.A.; Deepika, P.C.; Basavarajaiah, S. Zinc Incorporated Nano Hydroxyapatite: A Novel Bone Graft Used for Regeneration of Intrabony Defects. Contemp. Clin. Dent. 2018, 9, 427–433. [Google Scholar] [PubMed]
- Resende, R.; Fernandes, G.V.D.O.; Santos, S.R.A.; Rossi, A.M.; Lima, I.; Granjeiro, J.M.; Calasans-Maia, M.D. Long-term biocompatibility evaluation of 0.5 % zinc containing hydroxyapatite in rabbits. J. Mater. Sci. Mater. Electron. 2013, 24, 1455–1463. [Google Scholar] [CrossRef] [PubMed]
- Calasans-Maia, M.; Calasans-Maia, J.A.; Santos, S.; Mavropoulos, E.; Farina, M.; Lima, I.; Lopes, R.T.; Rossi, A.M.; Granjeiro, J.M. Short-term in vivo evaluation of zinc-containing calcium phosphate using a normalized procedure. Mater. Sci. Eng. C 2014, 41, 309–319. [Google Scholar] [CrossRef]
- Fernandes, G.; Calasans-Maia, M.; Mitri, F.; Bernardo, V.G.; Rossi, A.M.; Almeida, G.; Granjeiro, J.M. Histomorphometric Analysis of Bone Repair in Critical Size Defect in Rats Calvaria Treated with Hydroxyapatite and Zinc-Containing Hydroxyapatite 5%. Key Eng. Mater. 2008, 396, 15–18. [Google Scholar] [CrossRef]
- Bowen, P.; Drelich, J.; Goldman, J. Zinc Exhibits Ideal Physiological Corrosion Behavior for Bioabsorbable Stents. Adv. Mater. 2013, 25, 2577–2582. [Google Scholar] [CrossRef]
- Li, G.; Yang, H.; Zheng, Y.; Chen, X.-H.; Yang, J.-A.; Zhu, D.; Ruan, L.; Takashima, K. Challenges in the use of zinc and its alloys as biodegradable metals: Perspective from biomechanical compatibility. Acta Biomater. 2019, 97, 23–45. [Google Scholar] [CrossRef]
- Guo, H.; Xia, D.; Zheng, Y.; Zhu, Y.; Liu, Y.; Zhou, Y. A pure zinc membrane with degradability and osteogenesis promotion for guided bone regeneration: In vitro and in vivo studies. Acta Biomater. 2020, 106, 396–409. [Google Scholar] [CrossRef]
- Lin, J.; Tong, X.; Shi, Z.; Zhang, D.; Zhang, L.; Wang, K.; Wei, A.; Jin, L.; Lin, J.; Li, Y.; et al. A biodegradable Zn-1Cu-0.1Ti alloy with antibacterial properties for orthopedic applications. Acta Biomater. 2020, 106, 410–427. [Google Scholar] [CrossRef]
- Levy, G.K.; Leon, A.; Kafri, A.; Ventura, Y.; Drelich, J.W.; Goldman, J.; Vago, R.; Aghion, E. Evaluation of biodegradable Zn-1%Mg and Zn-1%Mg-0.5%Ca alloys for biomedical applications. J. Mater. Sci. Mater. Electron. 2017, 28, 174. [Google Scholar]
- Li, H.F.; Xie, X.H.; Zheng, Y.; Cong, Y.; Zhou, F.Y.; Qiu, K.J.; Wang, X.; Chen, S.H.; Huang, L.; Tian, L.; et al. Development of biodegradable Zn-1X binary alloys with nutrient alloying elements Mg, Ca and Sr. Sci. Rep. 2015, 5, 10719. [Google Scholar] [CrossRef] [PubMed]
- Pina, M.C.; Villareal, E.; Martin, S.; Leon, B.; Torres-Villasenor, G.; Bosch, P. Bone response to zinalco implants. J. Appl. Biomater. Biomech. 2010, 2. [Google Scholar]
- Wey, A.; Cunningham, C.; Hreha, J.; Breitbart, E.; Cottrell, J.; Ippolito, J.; Clark, D.; Lin, H.-N.; Benevenia, J.; O’Connor, J.P.; et al. Local ZnCl2 accelerates fracture healing. J. Orthop. Res. 2014, 32, 834–841. [Google Scholar] [CrossRef]
- Krell, E.S.; Ippolito, J.A.; Montemurro, N.J.; Lim, P.H.; Vincent, R.A.; Hreha, J.; Cottrell, J.; Sudah, S.Y.; Muñoz, M.F.; Pacific, K.P.; et al. Local Zinc Chloride Release From a Calcium Sulfate Carrier Enhances Fracture Healing. J. Orthop. Trauma 2017, 31, 168–174. [Google Scholar] [CrossRef]
- Koerner, J.D.; Vives, M.J.; O’Connor, J.P.; Chirichella, P.; Breitbart, E.A.; Chaudhary, S.B.; Uko, L.; Subramanian, S.; Fritton, J.; Benevenia, J.; et al. Zinc has insulin-mimetic properties which enhance spinal fusion in a rat model. Spine J. 2016, 16, 777–783. [Google Scholar] [CrossRef]
- Olechnowicz, J.; Tinkov, A.A.; Skalny, A.; Suliburska, J. Zinc status is associated with inflammation, oxidative stress, lipid, and glucose metabolism. J. Physiol. Sci. 2017, 68, 19–31. [Google Scholar] [CrossRef]
- Ono, T.; Takayanagi, H. Osteoimmunology in Bone Fracture Healing. Curr. Osteoporos. Rep. 2017, 15, 367–375. [Google Scholar] [CrossRef]
- Madel, M.-B.; Ibáñez, L.; Wakkach, A.; De Vries, T.J.; Teti, A.; Apparailly, F.; Blin-Wakkach, C. Immune Function and Diversity of Osteoclasts in Normal and Pathological Conditions. Front. Immunol. 2019, 10, 1408. [Google Scholar] [CrossRef]
- Fukada, T.; Civic, N.; Furuichi, T.; Shimoda, S.; Mishima, K.; Higashiyama, H.; Idaira, Y.; Asada, Y.; Kitamura, H.; Yamasaki, S.; et al. The zinc transporter SLC39A13/ZIP13 is required for connective tissue development; its involvement in BMP/TGF-beta signaling pathways. PLoS ONE 2008, 3, e3642. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
O’Connor, J.P.; Kanjilal, D.; Teitelbaum, M.; Lin, S.S.; Cottrell, J.A. Zinc as a Therapeutic Agent in Bone Regeneration. Materials 2020, 13, 2211. https://doi.org/10.3390/ma13102211
O’Connor JP, Kanjilal D, Teitelbaum M, Lin SS, Cottrell JA. Zinc as a Therapeutic Agent in Bone Regeneration. Materials. 2020; 13(10):2211. https://doi.org/10.3390/ma13102211
Chicago/Turabian StyleO’Connor, J. Patrick, Deboleena Kanjilal, Marc Teitelbaum, Sheldon S. Lin, and Jessica A. Cottrell. 2020. "Zinc as a Therapeutic Agent in Bone Regeneration" Materials 13, no. 10: 2211. https://doi.org/10.3390/ma13102211
APA StyleO’Connor, J. P., Kanjilal, D., Teitelbaum, M., Lin, S. S., & Cottrell, J. A. (2020). Zinc as a Therapeutic Agent in Bone Regeneration. Materials, 13(10), 2211. https://doi.org/10.3390/ma13102211





