Nanotechnology Scaffolds for Alveolar Bone Regeneration
Abstract
1. Introduction
- Cells, which represent the fundamental structural unit of any tissue,
- A matrix such as “scaffolds”, as framework material supporting the growth of cells to form a fully organized tissue,
- Biological factors like growth factors (GF) and bone morphogenetic proteins (BMPs) to guide cellular activity and tissue formation,
1.1. Importance of Scaffolds in TE/RM
1.2. Nanotechnology
- Nanoparticle-based techniques for delivering bioactive molecules,
- Nanoparticle-mediated cells tagging and targeting,
- Nanoparticle-based scaffold manufacturing [7].
1.3. Rationale of Nanotechnology Scaffolds in Tissue Engineering
1.4. Nanofibrous Scaffold Systems
1.5. Nanosphere Scaffold Systems
- Mechanically weak scaffolds in load-bearing applications may be reinforced by the adjunct of nanospheres as cross-linking agents,
- The porosity of the materials constituting traditional scaffolds may be significantly enhanced, by adding spheres like porogen. Increased porosity means allowing interior tissue infiltration into the scaffold,
- Nanospheres can stimulate the creation of apatite crystals and the following mineralization of hydrogels through the release of the corresponding minerals, so that self-hardening biomaterials adapted to the regeneration of bone tissue can be produced.
- Injectable and/or moldable materials can be developed thanks to the spherical nature of some nanomaterials, so that their application is possible by means of minimally invasive surgery [17].
1.6. Classification of Nanoparticle (NP) Materials
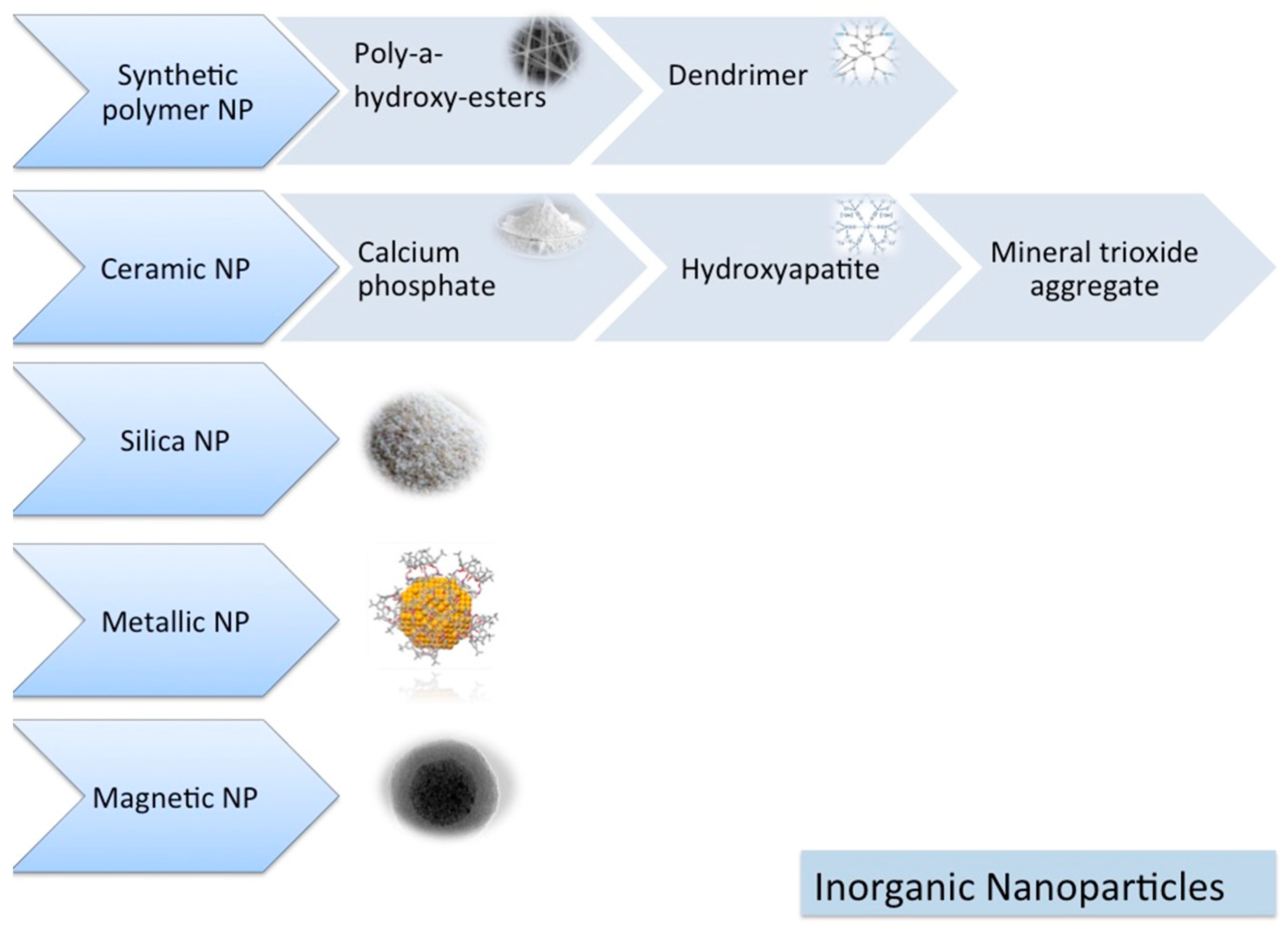
1.6.1. Inorganic NPs
Synthetic Polymers
Ceramic NPs
Silica NPs
Metallic NPs
Magnetic NPs
1.6.2. Organic Nanoparticles
Liposomes
Natural Polymeric NPs
Carbon
1.6.3. Composite Scaffolds
1.7. Manufacturing Methods to Fabricate Scaffolds
1.8. Biphasic and Multiphasic Scaffolds
1.9. Three-Dimensional (3D) Porous Scaffolds
1.10. Three-Dimensional Bio-Printing
1.11. Influence of Magnetic Fields on Biological Systems
2. Nanoscaffold Applications for alveolar Bone Regeneration
3. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| TE/RM | Tissue engineering and regenerative medicine |
| GF | Growth factors |
| BMP | Bone morphogenetic proteins |
| mRNA | Micro ribonucleic acid |
| TCP | Tricalcium phosphate |
| BMMSC | Bone marrow mesenchymal stem cells |
| MSC | Mesenchymal stem cells |
| PRP | Platelet rich plasma |
| ECM | Extracellular matrix |
| nm | Nanometers |
| µm | Micrometers |
| NP | Nanoparticles |
| PLA | Poly-lactic acid |
| PGA | Poly-glycolic acid, |
| PLGA | Poly-lactic-co-glycolic acid |
| PLLA | poly(l-lactic acid) |
| PEG | PLLA/polyethylene glycol |
| PCL | Poly-caprolactone |
| CP | Calcium Phosphate |
| HA | Hydroxyapatite |
| TCP | Tricalcium phosphate |
| nHA | Nano hydroxyapatite |
| nTCP | Nano tricalcium phosphate |
| MTA | Mineral trioxide aggregate |
| MSNP | Mesoporous silica nanoparticle |
| CNT | Carbon nanotube |
| GO | Graphene oxide |
| CD | Carbon dots |
| GO | Graphene oxide |
| rGO | Reduced graphene oxide |
| SLS | Selective laser sintering |
| 3D | 3 dimensional |
| SMF | Static magnetic field |
| FGF-2 | Fibroblast growth factor-2 |
| Ag | Silver |
| GelMA | Gelatin methacrylate |
| hPDLSC | Human periodontal ligament stem cells |
| CEMP1 | Cementum protein 1 |
| PDGF | Platelet derived growth factor |
| DNA | Deoxyribonucleic acid |
References
- Sowmya, S.; Mony, U.; Jayachandran, P.; Reshma, S.; Kumar, R.A.; Arzate, H.; Nair, S.V.; Jayakumar, R. Tri-Layered Nanocomposite Hydrogel Scaffold for the Concurrent Regeneration of Cementum, Periodontal Ligament, and Alveolar Bone. Adv. Healthc. Mater. 2017, 6, 1601251. [Google Scholar] [CrossRef] [PubMed]
- Pilipchuk, S.P.; Plonka, A.B.; Monje, A.; Tau, A.D.; Lanisdro, A.; Kang, B.; Giannobile, W.V. Tissue Engineering for Bone Regeneration and Osseointegration in the Oral Cavity. Dent. Mater. 2015, 31, 317–338. [Google Scholar] [CrossRef] [PubMed]
- Mudda, J.A.; Bajaj, M. Stem cell therapy: A challenge to periodontist. Indian J. Dent. Res. 2011, 22, 132–139. [Google Scholar] [CrossRef] [PubMed]
- Rios, H.F.; Lin, Z.; Oh, B.; Park, C.H.; Giannobile, W.V. Cell- and Gene-Based Therapeutic Strategies for Periodontal Regenerative Medicine. J. Periodontol. 2011, 82, 1223–1237. [Google Scholar] [CrossRef]
- Goker, F.; Ersanlı, S.; Arısan, V.; Cevher, E.; Güzel, E.E.; İşsever, H.; Ömer, B.; Altun, G.D.; Morina, D.; Yılmaz, T.E.; et al. Combined effect of parathyroid hormone and strontiumranelate on bone healing in ovariectomized rats. Oral Dis. 2018, 24, 1255–1269. [Google Scholar] [CrossRef]
- Saiz, E.; Zimmermann, E.A.; Lee, J.S.; Ulrike, G.K.; Wegst, U.G.K.; Tomsia, A.P. Perspectives on the Role of Nanotechnology in Bone Tissue Engineering. Dent. Mater. 2013, 29, 103–115. [Google Scholar] [CrossRef]
- Walmsley, G.G.; Mc Ardle, A.; Tevlin, R.; Momeni, A.; Atashroo, D.; Hu, M.S.; Feroze, A.H.; Wong, V.W.; Lorenz, P.H.; Longaker, M.T.; et al. Nanotechnology in bone tissue engineering. Nanomedicine 2015, 11, 1253–1263. [Google Scholar] [CrossRef]
- Carbone, E.J.; Tao Jiang, T.; Nelson, C.; Henry, N.; Lo, K.W.H. Small molecule delivery through nanofibrous scaffolds for musculoskeletal regenerative engineering. Nanomedicine 2014, 10, 1681–1699. [Google Scholar] [CrossRef]
- Hosseinpour, S.; Ahsaie, M.G.; Rad, M.R.; Baghani, M.; Motamedian, S.R.; Khojasteh, A. Application of selected scaffolds for bone tissue engineering: A systematic review. Oral Maxillofac. Surg. 2017, 21, 109–129. [Google Scholar] [CrossRef]
- D’Amora, U.; Russo, T.; Gloria, A.; Rivieccio, V.; D’Ant, V.; Negri, G.; Ambrosio, L.; De Santis, R. 3D additive-manufactured nanocomposite magnetic scaffolds: Effect of the application mode of a time-dependent magnetic field on hMSCs behavior. Bioact. Mater. 2017, 2, 138–145. [Google Scholar] [CrossRef]
- Ferracane, J.L.; Giannobile, W.V. Novel Biomaterials and Technologies for the Dental, Oral, and Craniofacial Structures. J. Dent. Res. 2014, 93, 1185–1186. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Sun, H.; Song, X.; Gu, X.; Sun, C. Biomaterials for periodontal tissue regeneration. Rev. Adv. Sci. 2015, 40, 209–214. [Google Scholar]
- Deng, M.; James, R.; Laurencin, C.T.; Kumbar, S.G. Nanostructured polymeric scaffolds for orthopaedic regenerative engineering. IEEE Trans. Nanobiosci. 2012, 11, 3–14. [Google Scholar] [CrossRef] [PubMed]
- Laurencin, C.T.; Khan, Y. Regenerative engineering. Sci. Transl. Med. 2012, 14, 160–169. [Google Scholar] [CrossRef]
- Laurencin, C.T.; Ashe, K.M.; Henry, N.; Kan, H.M.; Lo, K.W.H. Delivery of small molecules for bone regenerative engineering: Preclinical studies and potential clinical applications. Drug Discov. Today. 2014, 19, 794–800. [Google Scholar] [CrossRef]
- Vieira, S.; Vial, S.; Reis, R.L.; Oliveira, J.M. Nanoparticles for Bone Tissue Engineering. Biotechnol. Prog. 2017, 33, 590–611. [Google Scholar] [CrossRef]
- Wang, H.; Leeuwenburgh, S.C.G.; Li, Y.; Jansen, J.A. The Use of Micro- and Nanospheres as Functional Components for Bone Tissue Regeneration. Tissue Eng. Part B 2012, 18, 24–39. [Google Scholar] [CrossRef]
- Ionescu, L.C.; Lee, G.C.; Sennett, B.J.; Burdick, J.A.; Mauck, R.L. An anisotropic nanofiber/microsphere composite with controlled release of biomolecules for fibrous tissue engineering. Biomaterials 2010, 31, 4113–4120. [Google Scholar] [CrossRef]
- Wang, X.; Xing, H.; Zhang, G.; Wu, X.; Zou, X.; Feng, L.; Wang, D.; Li, M.; Zhao, J.; Du, J.; et al. Restoration of a critical mandibular bone defect using human alveolar bone-derived stem cells and porous nano-HA/Collagen/PLA Scaffold. Stem Cells Int. 2016, 8741641. [Google Scholar] [CrossRef]
- Wang, X.; Li, W. Biodegradable mesoporous bioactive glass nanospheres for drug delivery and bone tissue regeneration. Nanotechnology 2016, 27, 225102. [Google Scholar] [CrossRef]
- Mundargi, R.C.; Babu, V.R.; Rangaswamy, V.; Patel, P.; Aminabhavi, T.M. Nano/micro technologies for delivering macromolecular therapeutics using poly(d,l-lactide-coglycolide) and its derivatives. J. Control. Release 2008, 125, 193–209. [Google Scholar] [CrossRef] [PubMed]
- Xue, Y.; Hong, X.; Gao, J.; Shen, R.; Ye, Z. Preparation and biological characterization of the mixture of poly(lactic-co-glycolic acid)/chitosan/Ag nanoparticles for periodontal tissue engineering. Int. J. Nanomed. 2019, 14, 483–498. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Chen, X.; Zhang, Z.; Zhang, X.; Saunders, L.; Zhou, Y.; Ma, P.X. Nanofibrous Spongy Microspheres to Distinctly Release miRNA and Growth Factors To Enrich Regulatory T Cells and Rescue Periodontal Bone Loss. ACS Nano 2018, 12, 9785–9799. [Google Scholar] [CrossRef]
- Qian, Y.; Zhou, X.; Zhang, F.; Diekwisch, T.G.H.; Luan, X.; Yang, J. Triple PLGA/PCL Scaffold Modification Including Silver Impregnation, Collagen Coating, and Electrospinning Significantly Improve Biocompatibility, Antimicrobial, and Osteogenic Properties for Orofacial Tissue Regeneration. ACS Appl. Mater. Interfaces 2019, 11, 37381–37396. [Google Scholar] [CrossRef] [PubMed]
- Menjoge, A.R.; Kannan, R.M.; Tomalia, D.A. Dendrimer-based drug and imaging conjugates: Design considerations for nanomedical applications. Drug Discov. Today 2010, 15, 171–185. [Google Scholar] [CrossRef] [PubMed]
- Milhem, O.M.; Myles, C.; McKeown, N.B.; Attwood, D.; D’Emanuele, A. Polyamidoamine Starburst dendrimers as solubility enhancers. Int. J. Pharm. 2000, 197, 239–241. [Google Scholar] [CrossRef]
- Gillies, E.R.; Frechet, J.M.J. Dendrimers and dendritic polymers in drug delivery. Drug Discov. Today. 2005, 10, 35–43. [Google Scholar] [CrossRef]
- Yamamuro, T. Bioceramics. In Biomechanics and Biomaterials in Orthopedics; Poitout, D.G., Ed.; Springer: London, UK, 2004; pp. 22–23. [Google Scholar] [CrossRef]
- Raghavendra, S.S.; Jadhav, G.R.; Gathani, K.M.; Kotadia, P. Bioceramics in endodontics–A review. J. Istanb. Univ. Fac. Dent. 2017, 51, 128–137. [Google Scholar] [CrossRef]
- Pina, S.; Oliveira, J.M.; Reis, R.L. Natural-based nanocomposites for bone tissue engineering and regenerative medicine: A review. Adv. Mater. 2015, 27, 1143–1169. [Google Scholar] [CrossRef]
- Zhang, J.; Chen, Y.; Xu, J.; Wang, J.; Li, C.; Wang, L. Tissue engineering using 3D printed nano-bioactive glass loaded with NELL1 gene for repairing alveolar bone defects. Regen. Biomater. 2018, 5, 213–220. [Google Scholar] [CrossRef]
- Ebrahimi, M.; Botelho, M.; Lu, W.; Monmaturapoj, N. Synthesis and characterization of biomimetic bioceramic nanoparticleswith optimized physicochemical properties for bone tissue engineering. J. Biomed. Mater. Res. A 2019, 27. [Google Scholar] [CrossRef]
- Liu, L.; Liu, J.; Wang, M.; Min, S.; Cai, Y.; Zhu, L.; Yao, J. Preparation and characterization of nano-hydroxyapatite/silk fibroin porous scaffolds. J. Biomater. Sci. Polym. 2008, 19, 325–328. [Google Scholar] [CrossRef] [PubMed]
- Manju, V.; Anitha, A.; Menon, D.; Iyer, S.; Nair, S.V.; Nair, M.B. Nanofibrous yarn reinforced HA-gelatin composite scaffolds promote bone formation in critical sized alveolar defects in rabbit model. Biomed. Mater. 2018, 13, 065011. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.; Wu, Z.; Leung, A.; Chen, X.; Landao-Bassonga, E.; Gao, J.; Chen, L.; Zheng, M.; Yao, F.; Yang, H.; et al. Fabrication of a silver nanoparticle-coated collagen membrane with anti-bacterial and anti-inflammatory activities for guided bone regeneration. Biomed. Mater. 2018, 13, 065014. [Google Scholar] [CrossRef] [PubMed]
- Han, J.; Ma, B.; Liu, H.; Wang, T.; Wang, F.; Xie, C.; Li, M.; Liu, H.; Ge, S. Hydroxyapatite nanowires modified polylactic acid membrane plays barrier/osteoinduction dual roles and promotes bone regeneration in a rat mandible defect model. J. Biomed. Mater. Res. A 2018, 106, 3099–3110. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Shi, Z.; Bai, S.; Li, B.; Huan Liu, H.; Guo, F.; Wu, G.; Liu, S.; Zhao, Y. Fabrication of gelatin methacrylate/nanohydroxyapatite microgel arrays for periodontal tissue regeneration. Int. J. Nanomed. 2016, 11, 4707–4718. [Google Scholar]
- Nie, L.; Zhang, H.; Ren, A.; Li, Y.; Fu, G.; Cannon, R.D.; Ji, P.; Wu, X.; Yang, S. Nano-hydroxyapatite mineralized silk fibroin porous scaffold for tooth extraction site preservation. Dent. Mater. 2019, 35, 1397–1407. [Google Scholar] [CrossRef]
- Dayashankar, C.P.; Deepika, P.C.; Siddaramaiah, B. Clinical and Radiographic Evaluation of Citric Acid-Based Nano Hydroxyapatite Composite Graft in the Regeneration of Intrabony Defects—A Randomized Controlled Trial. Contemp. Clin. Dent. 2017, 8, 380–386. [Google Scholar] [CrossRef]
- Vaquette, C.; Fan, W.; Xiao, Y.; Hamlet, S.; Hutmacher, D.W.; Ivanovski, S. A biphasic scaffold design combined with cell sheet technology for simultaneous regeneration of alveolar bone/periodontal ligament complex. Biomaterials 2012, 33, 5560–5573. [Google Scholar] [CrossRef]
- Ogawa, K.; Miyaji, H.; Kato, A.; Kosen, Y.; Momose, T.; Yoshida, T.; Nishida, E.; Miyata, S.; Murakami, S.; Takita, H.; et al. Periodontal tissue engineering by nano beta-tricalcium phosphate scaffold and fibroblast growth factor-2 in one-wall infrabony defects of dogs. J. Periodontal. Res. 2016, 51, 758–767. [Google Scholar] [CrossRef]
- Camilleri, J. Is mineral trioxide Aggregate a Bioceramic. ODOVTOS Int. J. Dent. Sci. 2016, 18, 13–17. [Google Scholar] [CrossRef]
- Saghiri, M.A.; Orangi, J.; Tanideh, N.; Asatourian, A.; Janghorban, K.; Garcia-Godoy, F.; Sheibani, N. Repair of bone defect by nano-modifed white mineral trioxide aggregates in rabbit: A histopathological study. Med. Oral. Patol. Oral Cir. Bucal 2015, 20, 525–531. [Google Scholar] [CrossRef] [PubMed]
- Lipski, A.M.; Pino, C.J.; Haselton, F.R.; Chen, I.W.; Shastri, V.P. The effect of silica nanoparticle-modified surfaces on cell morphology, cytoskeletal organization and function. Biomaterials 2008, 29, 3836–3846. [Google Scholar] [CrossRef] [PubMed]
- Moller, K.; Kobler, J.; Bein, T. Colloidal suspensions of nanometer-sized mesoporous silica. Adv. Funct. Mater. 2007, 17, 605–612. [Google Scholar] [CrossRef]
- Yeh, Y.C.; Creran, B.; Rotello, V.M. Gold nanoparticles: Preparation, properties, and applications in bionanotechnology. Nanoscale 2012, 4, 1871–1880. [Google Scholar] [CrossRef] [PubMed]
- Vigderman, L.; Khanal, B.P.; Zubarev, E.R. Functional gold nanorods: Synthesis, self-assembly, and sensing applications. Adv. Mater. 2012, 24, 4811–4841. [Google Scholar] [CrossRef]
- Takanche, J.S.; Kim, J.E.; Kim, J.S.; Lee, M.H.; Jeon, J.G.; Park, I.S.; Yi, H.K. Chitosan-gold nanoparticles mediated gene delivery of c-myb facilitates osseointegration of dental implants in ovariectomized rat. Artif. Cells Nanomed. Biotechnol. 2018, 46, S807–S817. [Google Scholar] [CrossRef]
- Colombo, M.; Carregal-Romero, S.; Casula, M.F.; Gutiérrez, L.; Morales, M.P.; Böhm, I.B.; Heverhagen, J.T.; Prosperi, D.; Parak, W.J. Biological applications of magnetic nanoparticles. Chem. Soc. Rev. 2012, 41, 4306–4334. [Google Scholar] [CrossRef]
- Bangham, A.D. Surrogate cells or Trojan horses. The discovery of liposomes. Bioessays 1995, 17, 1081–1088. [Google Scholar] [CrossRef]
- Immordino, M.L.; Dosio, F.; Cattel, L. Stealth liposomes: Review of the basic science, rationale, and clinical applications, existing and potential. Int. J. Nanomed. 2006, 1, 297–315. [Google Scholar]
- Allen, T.M.; Cullis, P.R. Liposomal drug delivery systems: From concept to clinical applications. Adv. Drug Deliv. Rev. 2013, 65, 36–48. [Google Scholar] [CrossRef] [PubMed]
- Nicolas, J.; Mura, S.; Brambilla, D.; Mackiewicz, N.; Couvreur, P. Design, functionalization strategies and biomedical applications of targeted biodegradable/biocompatible polymer-based nanocarriers for drug delivery. Chem. Soc. Rev. 2013, 42, 1147–1235. [Google Scholar] [CrossRef] [PubMed]
- Dhandayuthapani, B.; Yoshida, Y.; Maekawa, T.; Kumar, D.S. Polymeric scaffolds in tissue engineering application: A review. Int. J. Polym. Sci. 2011, 290602. [Google Scholar] [CrossRef]
- Lutolf, M.P.; Weber, F.E.; Schmoekel, H.G.; Schense, J.C.; Kohler, T.; Müller, R.; Hubbell, J.A. Repair of bone defects using synthetic mimetics of collagenous extracellular matrices. Nat. Biotechnol. 2003, 21, 513–518. [Google Scholar] [CrossRef] [PubMed]
- Ye, H.; Zhu, J.; Deng, D.; Jin, S.; Li, J.; Man, Y. Enhanced osteogenesis and angiogenesis by PCL/chitosan/Sr-doped calcium phosphate electrospun nanocomposite membrane for guided bone regeneration. J. Biomater. Sci. Polym. Ed. 2019, 30, 1505–1522. [Google Scholar] [CrossRef] [PubMed]
- Eivazzadeh-Keihan, R.; Maleki, A.; de la Guardia, M.; Bani, M.S.; Chenab, K.K.; Pashazadeh-Panahi, P.; Baradaran, B.; Mokhtarzadeh, A.; Hamblin, M.R. Carbon based nanomaterials for tissue engineering of bone: Building new bone on small black scaffolds: A review. J. Adv. Res. 2019, 18, 185–201. [Google Scholar] [CrossRef]
- La, W.G.; Jin, M.; Park, S.; Yoon, H.H.; Jeong, G.J.; Bhang, S.H.; Park, H.; Char, K.; Kim, B.S. Delivery of bone morphogenetic protein-2 and substance P using graphene oxide for bone regeneration. Int. J. Nanomed. 2014, 9, 107–116. [Google Scholar]
- Zhou, Q.; Yang, P.; Li, X.; Liu, H.; Ge, S. Bioactivity of periodontal ligament stem cells on sodium titanate coated with graphene oxide. Sci. Rep. 2016, 6, 19343. [Google Scholar] [CrossRef]
- Radunovic, M.; De Colli, M.; De Marco, P.; Di Nisio, C.; Fontana, A.; Piattelli, A.; Cataldi, A.; Zara, S. Graphene oxide enrichment of collagen membranes improves DPSCs differentiation and controls inflammation occurrence. J. Biomed. Mater. Res. A. 2017, 105, 2312–2320. [Google Scholar] [CrossRef]
- De Marco, P.; Zara, S.; De Colli, M.; Radunovic, M.; Lazovi’c, V.; Ettorre, V.; Di Crescenzo, A.; Piattelli, A.; Cataldi, A.; Fontana, A. Graphene oxide improves the biocompatibility of collagen membranes in an in vitromodel of human primary gingival fibroblasts. Biomed. Mater. 2017, 12, 055005. [Google Scholar] [CrossRef]
- Liao, C.; Li, Y.; Tjong, S.C. Graphene Nanomaterials: Synthesis, Biocompatibility, and Cytotoxicity. Int. J. Mol. Sci. 2018, 19, 3564. [Google Scholar] [CrossRef] [PubMed]
- Bullock, C.J.; Bussy, C. Biocompatibility Considerations in the Design of Graphene Biomedical Materials. Adv. Mater. Interfaces 2019, 6, 1900229. [Google Scholar] [CrossRef]
- Wang, K.; Ruan, J.; Song, H.; Zhang, J.; Wo, Y.; Guo, S.; Cui, D. Biocompatibility of Graphene Oxide. Nanoscale Res. Lett. 2011, 6, 8. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Lozano, F.J.; García-Bernal, D.; Aznar-Cervantes, S.; Ros-Roca, M.A.; Algueró, M.C.; Atucha, N.M.; Lozano-García, A.A.; Moraleda, J.M.; Cenis, J.L. Effects of composite films of silk fibroin and graphene oxide on the proliferation, cell viability and mesenchymal phenotype of periodontal ligament stem cells. J. Mater. Sci. Mater. Med. 2014, 25, 2731–2741. [Google Scholar] [CrossRef]
- Vera-Sánchez, M.; Aznar-Cervantes, S.; Jover, E.; García-Bernal, D.; Oñate-Sánchez, R.E.; Hernández-Romero, D.; Moraleda, J.M.; Collado-González, M.; Rodríguez-Lozano, F.J.; Cenis, J.L. Silk-Fibroin and Graphene Oxide Composites Promote Human Periodontal Ligament Stem Cell Spontaneous Differentiation into Osteo/Cementoblast-Like Cells. Stem Cells Dev. 2016, 25, 1742–1754. [Google Scholar] [CrossRef]
- Ren, N.; Li, J.; Qiu, J.; Yan, M.; Liu, H.; Ji, D.; Huang, J.; Yu, J.; Liu, H. Growth and accelerated differentiation of mesenchymal stem cells on graphene-oxide-coated titanate with dexamethasone on surface of titanium implants. Dent. Mater. 2017, 33, 525–535. [Google Scholar] [CrossRef]
- Kinoshita, Y.; Maeda, H. Recent developments of functional scaffolds for craniomaxillofacial bone tissue engineering applications. Sci. World J. 2013, 21, 863157. [Google Scholar] [CrossRef]
- Huang, Y.; Ren, J.; Ren, T.; Gu, S.; Tan, Q.; Zhang, L.; Lv, K.; Pan, K.; Jiang, X. Bone marrow stromal cells cultured on poly (lactide-co-glycolide)/nanohydroxyapatite composites with chemical immobilization of Arg-Gly-Asp peptide and preliminary bone regeneration of mandibular defect thereof. J. Biomed. Mater. Res. A 2010, 95, 993–1003. [Google Scholar] [CrossRef]
- Han, X.; Liu, H.; Wang, D.; Su, F.; Zhang, Y.; Zhou, W.; Li, S.; Yang, R. Alveolar bone regeneration around immediate implants using an injectable nHAC/CSH loaded with autogenic blood-acquired mesenchymal progenitor cells: An experimental study in the dog mandible. Clin. Implant Dent. Relat. Res. 2013, 15, 390–401. [Google Scholar] [CrossRef]
- Zhang, J.C.; Lu, H.Y.; Lv, G.Y.; Mo, A.C.; Yan, Y.G.; Huang, C. The repair of critical-size defects with porous hydroxyapatite/polyamide nanocomposite: An experimental study in rabbit mandibles. Int. J. Oral Maxillofac. Surg. 2010, 39, 469–477. [Google Scholar] [CrossRef]
- Du, B.; Liu, W.; Deng, Y.; Li, S.; Liu, X.; Gao, Y.; Zhou, L. Angiogenesis and bone regeneration of porous nano-hydroxyapatite/coralline blocks coated with rhVegF165 in critical-size alveolar bone defects in vivo. Int. J. Nanomed. 2015, 10, 2555–2565. [Google Scholar]
- Su, J.; Xu, H.; Sun, J.; Gong, X.; Zhao, H. Dual delivery of BMP-2 and bFGF from a new nano-composite scaffold, loaded with vascular stents for large size mandibular defect regeneration. Int. J. Mol. Sci. 2013, 14, 12714–12728. [Google Scholar] [CrossRef] [PubMed]
- Zhao, F.; Yin, Y.; Lu, W.W.; Leong, J.C.; Zhang, W.; Zhang, J.; Zhang, M.; Yao, K. Preparation and histological evaluation of biomimetic three-dimensional hydroxyapatite/chitosan-gelatin network composite scaffolds. Biomaterials 2002, 23, 3227–3234. [Google Scholar] [CrossRef]
- Xynos, I.; Hukkanen, M.; Batten, J.; Buttery, L.; Hench, L.; Polak, J. Bioglass® 45S5 stimulates osteoblast turnover and enhances bone formation in vitro: Implications and applications for bone tissue engineering. Calcif. Tissue Int. 2000, 67, 321–329. [Google Scholar] [CrossRef]
- Kim, H.D.; Valentini, R.F. Retention and activity of BMP-2 in hyaluronic acid-based scaffolds in vitro. J. Biomed. Mater. Res. 2002, 59, 573–584. [Google Scholar] [CrossRef] [PubMed]
- El-Ghannam, A.R. Advanced bioceramic composite for bone tissue engineering: Design principles and structure—Bioactivity relationship. J. Biomed. Mater. Res. Part A. 2004, 69, 490–501. [Google Scholar] [CrossRef]
- Iviglia, G.; Saeid Kargozar, S.; Baino, F. Biomaterials, Current Strategies, and Novel Nano-Technological Approaches for Periodontal Regeneration. J. Funct. Biomater. 2019, 10, 3. [Google Scholar] [CrossRef]
- Li, G.; Zhang, T.; Li, M.; Fu, N.; Fu, Y.; Ba, K.; Deng, S.; Jiang, Y.; Hu, J.; Peng, Q.; et al. Electrospun fibers for dental and craniofacial applications. Curr. Stem Cell Res. 2014, 9, 187–195. [Google Scholar] [CrossRef]
- Gmeiner, R.; Deisinger, U.; Schönherr, J.; Lechner, B.; Detsch, R.; Boccaccini, A.; Stampfl, J. Additive manufacturing of bioactive glasses and silicate bioceramics. J. Ceram. Sci. Technol. 2015, 6, 75–86. [Google Scholar]
- Kosachan, N.; Jaroenworaluck, A.; Jiemsirilers, S.; Jinawath, S.; Stevens, R. Hydroxyapatite nanoparticles formed under a wet mechanochemical method. J. Biomed. Mater. Res. Part B Appl. Biomater. 2017, 105, 679–688. [Google Scholar] [CrossRef]
- Ivanovski, S.; Vaquette, C.; Gronthos, S.; Hutmacher, D.W.; Bartold, P.M. Multiphasic Scaffolds for Periodontal Tissue Engineering. J. Dent. Res. 2014, 93, 1212–1221. [Google Scholar] [CrossRef]
- Carter, S.D.; Costa, P.F.; Vaquette, C.; ivanovski, S.; Hutmacher, D.W.; Malda, J. Additive Biomanufacturing: An Advanced Approach for Periodontal Tissue Regeneration. Ann. Biomed. Eng. 2017, 45, 12–22. [Google Scholar] [CrossRef] [PubMed]
- Carlo-Reis, E.C.; Borges, A.P.; Araújo, M.V.; Mendes, V.C.; Guan, L.; Davies, J.E. Periodontal regeneration using a bilayered PLGA/calcium phosphate construct. Biomaterials 2011, 32, 9244–9253. [Google Scholar] [CrossRef] [PubMed]
- Requicha, J.F.; Viegas, C.A.; Muñoz, F.; Azevedo, J.M.; Leonor, I.B.; Reis, R.L.; Gomes, M.E. A tissue engineering approach for periodontal regeneration based on a biodegradable double-layer scaffold and adipose-derived stem cells. Tissue Eng. Part A 2014, 20, 2483–2492. [Google Scholar] [CrossRef] [PubMed]
- Park, C.H.; Rios, H.F.; Jin, Q.; Bland, M.E.; Flanagan, C.L.; Hollister, S.J.; Giannobile, W.V. Biomimetic hybrid scaffolds for engineering human tooth ligament interfaces. Biomaterials 2010, 31, 5945–5952. [Google Scholar] [CrossRef]
- Park, C.H.; Rios, H.F.; Jin, Q.; Sugai, J.V.; Padial-Molina, M.; Taut, A.D.; Flanagan, C.L.; Hollister, S.J.; Giannobile, W.V. Tissue engineering bone-ligament complexes using fiber-guiding scaffolds. Biomaterials 2012, 33, 137–145. [Google Scholar] [CrossRef]
- Vaquette, C.; Cooper-White, J. A simple method for fabricating 3-D multilayered composite scaffolds. Acta Biomater. 2013, 9, 4599–4608. [Google Scholar] [CrossRef]
- Costa, P.F.; Vaquette, C.; Zhang, Q.; Reis, R.L.; Ivanovski, S.; Hutmacher, D.W. Advanced tissue engineering scaffold design for regeneration of the complex hierarchical periodontal structure. J. Clin. Periodontol. 2014, 41, 283–294. [Google Scholar] [CrossRef]
- Lee, C.H.; Hajibandeh, J.; Suzuki, T.; Fan, A.; Shang, P.; Mao, J.J. Three dimensional printed multiphase scaffolds for regeneration of periodontium complex. Tissue Eng. Part A 2014, 20, 1342–1351. [Google Scholar] [CrossRef]
- Murphy, S.V.; Atala, A. 3D bioprinting of tissues and organs. Nat. Biotechnol. 2014, 32, 773–785. [Google Scholar] [CrossRef]
- Dababneh, A.B.; Ozbolat, I.T. Bioprinting technology: A current state-of-the-art review. J. Manuf. Sci. Eng. 2014, 136, 061016. [Google Scholar] [CrossRef]
- Berthiaume, F.; Maguire, T.J.; Yarmush, M.L. Tissue engineering and regenerative medicine: History, progress, and challenges. Annu. Rev. Chem. Biomol. Eng. 2011, 2, 403–430. [Google Scholar]
- Zhang, Y.S.; Yue, K.; Aleman, J.; Mollazadeh-Moghaddam, K.; Bakht, S.M.; Yang, J.; Weitao, J.A.; Dell’Erba, V.; Assawes, P.; Shin, S.R.; et al. 3D Bioprinting for Tissue and Organ Fabrication. Ann. Biomed. Eng. 2017, 45, 148–163. [Google Scholar] [CrossRef] [PubMed]
- Yun, H.M.; Ahn, S.J.; Park, K.R.; Kim, M.J.; Kim, J.J.; Jin, G.Z.; Kim, H.W.; Ahn, S.J.; Kim, H.W.; Kim, E.C. Magnetic nanocomposite scaffolds combined with static magnetic field in the stimulation of osteoblastic differentiation and bone formation. Biomaterials 2016, 85, 88–98. [Google Scholar] [CrossRef] [PubMed]
- Sculean, A.; Nikolidakis, D.; Nikou, G.; Ivanovic, A.; Chapple, I.L.; Stavropoulos, A. Biomaterials for promoting periodontal regeneration in human intrabony defects: A systematic review. Periodontology 2000 2015, 68, 182–216. [Google Scholar] [CrossRef]
- Kargozar, S.; Mozafari, M. Nanotechnology and Nanomedicine: Start small, think big. Mater. Today Proc. 2018, 5, 15492–15500. [Google Scholar] [CrossRef]
- Neel, E.A.A.; Chrzanowski, W.; Salih, V.M.; Kim, H.W.; Knowles, J.C. Review Tissue engineering in dentistry. J. Dent. 2014, 42, 915–928. [Google Scholar] [CrossRef]
- Zhang, Y.; Miron, R.J.; Li, S.; Shi, B.; Sculean, A.; Cheng, X. Novel Meso Porous BioGlass/silk scaffold containing ad PDGF-B and ad BMP 7 for the repair of periodontal defects in beagle dogs. J. Clin. Periodontol. 2015, 42, 262–271. [Google Scholar] [CrossRef]
- Yue, K.; Trujillo-de Santiago, G.; Alvarez, M.M.; Tamayol, A.; Annabi, N.; Khademhosseini, A. Synthesis, properties, and biomedical applications of gelatin methacryloyl (GelMA) hydrogels. Biomaterials 2015, 73, 254–271. [Google Scholar] [CrossRef]
- Chen, X.; Liu, Y.; Miao, L.; Wang, Y.; Ren, S.; Yang, X.; Hu, Y.; Sun, W. Controlled release of recombinant human cementum protein 1 from electrospun multiphasic scaffold for cementum regeneration. Int. J. Nanomed. 2016, 11, 3145–3158. [Google Scholar]
- Guazzo, R.; Gardin, C.; Bellin, G.; Sbricoli, L.; Ferroni, L.; Ludovichetti, F.S.; Piattelli, A.; Antoniac, I.; Bressan, E.; Zavan, B. Graphene-Based Nanomaterials for Tissue Engineering in the Dental Field. Nanomaterials 2018, 8, 349. [Google Scholar] [CrossRef]
- Zhang, Y.; Song, J.; Shia, B.; Wanga, Y.; Chena, X.; Huanga, C.; Yanga, X.; Xua, D.; Cheng, X.; Chen, X. Combination of scaffold and adenovirus vectors expressing bone morphogenetic protein-7 for alveolar bone regeneration at implant defects. Biomaterials 2007, 28, 4635–4642. [Google Scholar] [CrossRef] [PubMed]
- Rasperini, G.; Pilipchuk, S.P.; Flanagan, C.L.; Park, C.H.; Pagni, G.; Hollister, S.J.; Giannobile, W.V. 3D-printed bioresorbable scaffold for periodontal repair. J. Dent. Res. 2015, 94, 153–157. [Google Scholar] [CrossRef] [PubMed]
- Feng, X.; Chen, A.; Zhang, Y.; Wang, J.; Shao, L.; Wei, L. Application of dental nanomaterials: Potential toxicity to the central nervous system. Int. J. Nanomed. 2015, 10, 3547–3565. [Google Scholar]
- Lee, C.M.; Jeong, H.J.; Yun, K.N.; Kim, D.W.; Sohn, M.H.; Lee, J.K.; Jeong, J.; Lim, S.T. Optical imaging to trace near infrared fluorescent zinc oxide nanoparticles following oral exposure. Int. J. Nanomed. 2012, 7, 3203–3209. [Google Scholar]
- Wang, Y.; Chen, Z.; Ba, T.; Pu, J.; Chen, T.; Song, Y.; Gu, Y.; Qian, Q.; Xu, Y.; Xiang, K.; et al. Susceptibility of young and adult rats to the oral toxicity of titanium dioxide nanoparticles. Small 2013, 9, 1742–1752. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Urrusuno, R.; Fattal, E.; Rodrigues, J.M., Jr.; Féger, J.; Bedossa, P.; Couvreur, P. Effect of polymeric nanoparticle administration on the clearance activity of the mononuclear phagocyte system in mice. J. Biomed. Mater. Res. 1996, 31, 401–408. [Google Scholar] [CrossRef]
- Karmakar, A.; Zhang, Q.; Zhang, Y. Neurotoxicity of nanoscale materials. J. Food Drug Anal. 2014, 22, 147–160. [Google Scholar] [CrossRef]
- Ma, L.; Liu, J.; Li, N.; Wang, J.; Duan, Y.; Yan, J.; Liu, H.; Wang, H.; Hong, F. Oxidative stress in the brain of mice caused by translocated nanoparticulate TiO2 delivered to the abdominal cavity. Biomaterials 2010, 31, 99–105. [Google Scholar] [CrossRef]
- Adamcakova-Dodd, A.; Stebounova, L.V.; Kim, J.S.; Vorrink, S.U.; Ault, A.P.; O’Shaughnessy, P.T.; Grassian, V.H.; Thorne, P.S. Toxicity assessment of zinc oxide nanoparticles using sub-acute and sub-chronic murine inhalation models. Part Fibretoxicol. 2014, 1, 11–15. [Google Scholar] [CrossRef]
- Xie, H.; Chua, M.; Islam, I.; Bentini, R.; Cao, T.; Viana-Gomes, J.C.; Castro Neto, A.H.; Rosa, V. CVD-grown monolayer graphene induces osteogenic but not odontoblastic differentiation of dental pulp stem cells. Dent. Mater. 2017, 33, e13–e21. [Google Scholar] [CrossRef]
- Launey, M.E.; Munch, E.; Alsem, D.H.; Barth, H.B.; Saiz, E.; Tomsia, A.P.; Ritchie, R.O. Designing highly toughened hybrid composites through nature-inspired hierarchical complexity. Acta Mater. 2009, 57, 2919–2932. [Google Scholar] [CrossRef]
- Dzenis, Y. Materials science—Structural nanocomposites. Science 2008, 319, 419–420. [Google Scholar] [CrossRef] [PubMed]
- Ritchie, R.O. The quest for stronger, tougher materials. Science 2008, 320, 448. [Google Scholar] [CrossRef] [PubMed]
- Evans, A.G. Perspective on the development of high-toughness ceramics. J. Am. Ceram Soc. 1990, 73, 187–206. [Google Scholar] [CrossRef]

| Material | Reference | Outcome |
|---|---|---|
| 3 layer chitosan/PLGA/nano-sized bioactive glass | Sowmya et al. [1] | Complete periodontal healing and new alveolar bone deposition after three months |
| GO-coating of collagen membranes | Radunovic et al. [60] | Favourable on promoting osteoblastic differentiation process |
| GO-coating of collagen membranes | De Marco et al. [61] | Improved biocompatibility of collagen membranes on in vitro human primary gingival fibroblast model |
| PCL containing ß–TCP | Vaquette et al. [40] | Enhanced mechanical stability of the cell sheets, and mineralization. However, ectopic bone ingrowth was not sufficient |
| nano-ß- TCP/collagen scaffolds | Ogawa et al. [41] | nano-ß-TCP/collagen scaffolds loaded with fibroblast growth factor-2 (FGF-2) improved periodontal tissue wound healing results |
| Chitosan, PLGA, and silver (Ag) nanoparticles complex | Xue et al. [22] | Contributed to cell mineralization without cytotoxicity |
| GelMA/nHAmicrogels | Chen et al. [37] | Promoted in vivo osteogenesis of hPDLSCs encapsulated in microgels |
| nanomaterial-based silk fibroin scaffolds incorporating BMP-7 and/or PDGF-ß | Zhang et al. [103] | Promoted periodontal healing |
| PCLpowder containing HA | Rasperini et al. [104] | Clinical study with failure due exposure of the scaffold |
| Graphene | Xie et al. [112] | Favourable on osteogenic differentiation but not on osteoblastic differentiation |
| Graphene Oxide combined with silk fibroin | Rodríguez-Lozano et al. [65] | Favourable on mechanical resistance and hPDLSC proliferation and showed biocompatibility |
| Graphene Oxide combined with silk fibroin | Vera-Sánchez et al. [66] | PDLSC proliferation rate into osteo/cementoblast like cells improved with these combinations |
| GO-coating of titanium implants | Ren et al. [67] | Improved cell proliferation, osteogenic differentiation and biocompatibility of implants |
| Citric Acid-Based Nano Hydroxyapatite | Dayashankar et al. [39] | Significant bone regeneration |
| Nano-bioactive glass loaded with NELL1 gene | Zhang et al. [31] | Good osteoconductivity for promoting the formation of new alveolar bone tissue |
| Poly(l-lactic acid) (PLLA) nanofibrous spongy microspheres, PLLA/polyethylene glycol (PEG) co-functionalized mesoporous silica nanoparticles, and poly(lactic acid-co-glycolic acid) (PLGA) microspheres | Liu et al. [23] | In a mouse model of periodontitis, the injectable and biomolecule-delivering PLLA lead to Treg enrichment, expansion, and Treg-mediated immune therapy against bone loss |
| Nanofibrous yarn reinforced HA-gelatin | Manju et al. [34] | Promoted bone formation in critical sized alveolar defects in rabbit model |
| Silver nanoparticle-coated collagen membrane | Chen et al. [35] | Induced osteogenic differentiation of mesenchymal stem cells that guide bone regeneration. |
| Chitosan-gold nanoparticles mediated gene delivery | Takanche et al. [48] | Enchanced osseointegration of dental implant even in osteoporotic condition |
| Hydroxyapatite nanowires modified polylactic acid membrane | Han et al. [36] | Promoted bone regeneration in a rat mandible defect model |
| PCL/chitosan/Sr-doped calcium phosphate electrospun nanocomposite | Ye et al. [56] | Higher ALP activity level and a better matrix mineralization |
| Nano hydroxyapatite mineralized silk fibroin | Nie et al. [38] | Improved osteogenesis |
| PLGA/PCL Modification Including Silver Impregnation, Collagen Coating, and Electrospinning | Qian et al. [24] | Enhanced alveolar bone regeneration (31.8%) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Funda, G.; Taschieri, S.; Bruno, G.A.; Grecchi, E.; Paolo, S.; Girolamo, D.; Del Fabbro, M. Nanotechnology Scaffolds for Alveolar Bone Regeneration. Materials 2020, 13, 201. https://doi.org/10.3390/ma13010201
Funda G, Taschieri S, Bruno GA, Grecchi E, Paolo S, Girolamo D, Del Fabbro M. Nanotechnology Scaffolds for Alveolar Bone Regeneration. Materials. 2020; 13(1):201. https://doi.org/10.3390/ma13010201
Chicago/Turabian StyleFunda, Goker, Silvio Taschieri, Giannì Aldo Bruno, Emma Grecchi, Savadori Paolo, Donati Girolamo, and Massimo Del Fabbro. 2020. "Nanotechnology Scaffolds for Alveolar Bone Regeneration" Materials 13, no. 1: 201. https://doi.org/10.3390/ma13010201
APA StyleFunda, G., Taschieri, S., Bruno, G. A., Grecchi, E., Paolo, S., Girolamo, D., & Del Fabbro, M. (2020). Nanotechnology Scaffolds for Alveolar Bone Regeneration. Materials, 13(1), 201. https://doi.org/10.3390/ma13010201







