Properties of Alkali-Activated Slag Paste Using New Colloidal Nano-Silica Mixing Method
Abstract
1. Introduction
2. Materials and Methods
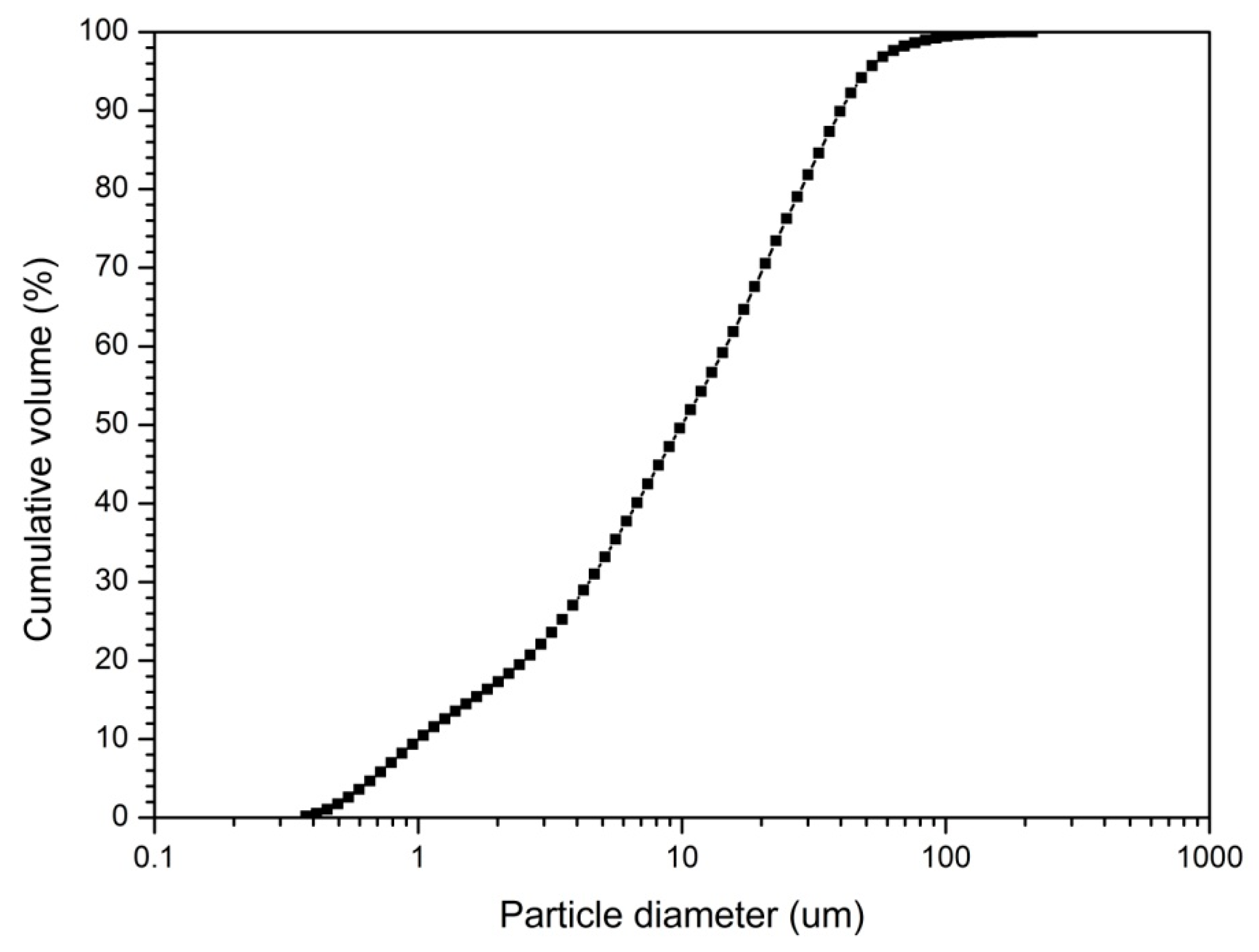
2.1. Materials
2.2. Experimental Details
3. Results and Discussion
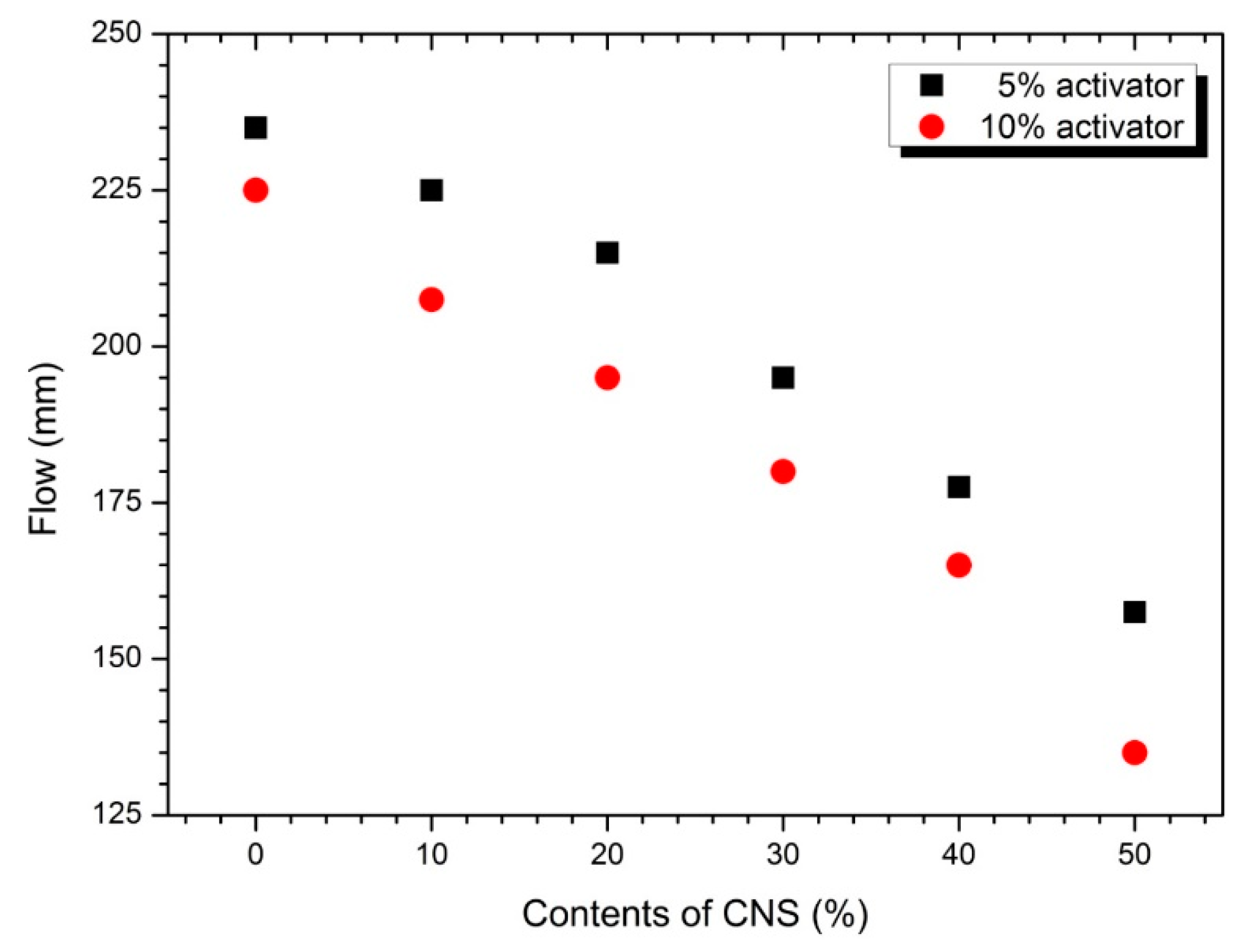
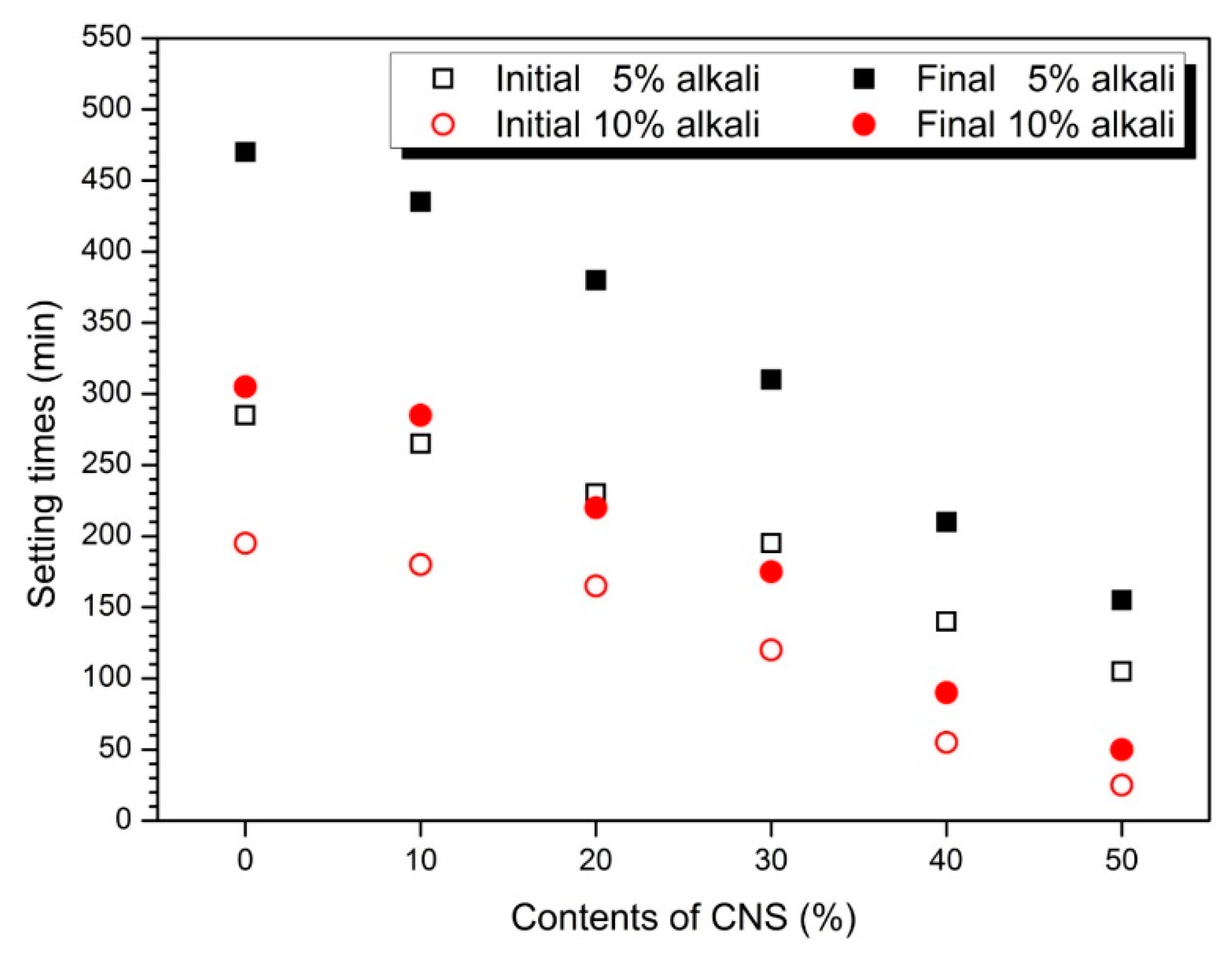
3.1. Flow and Setting Time
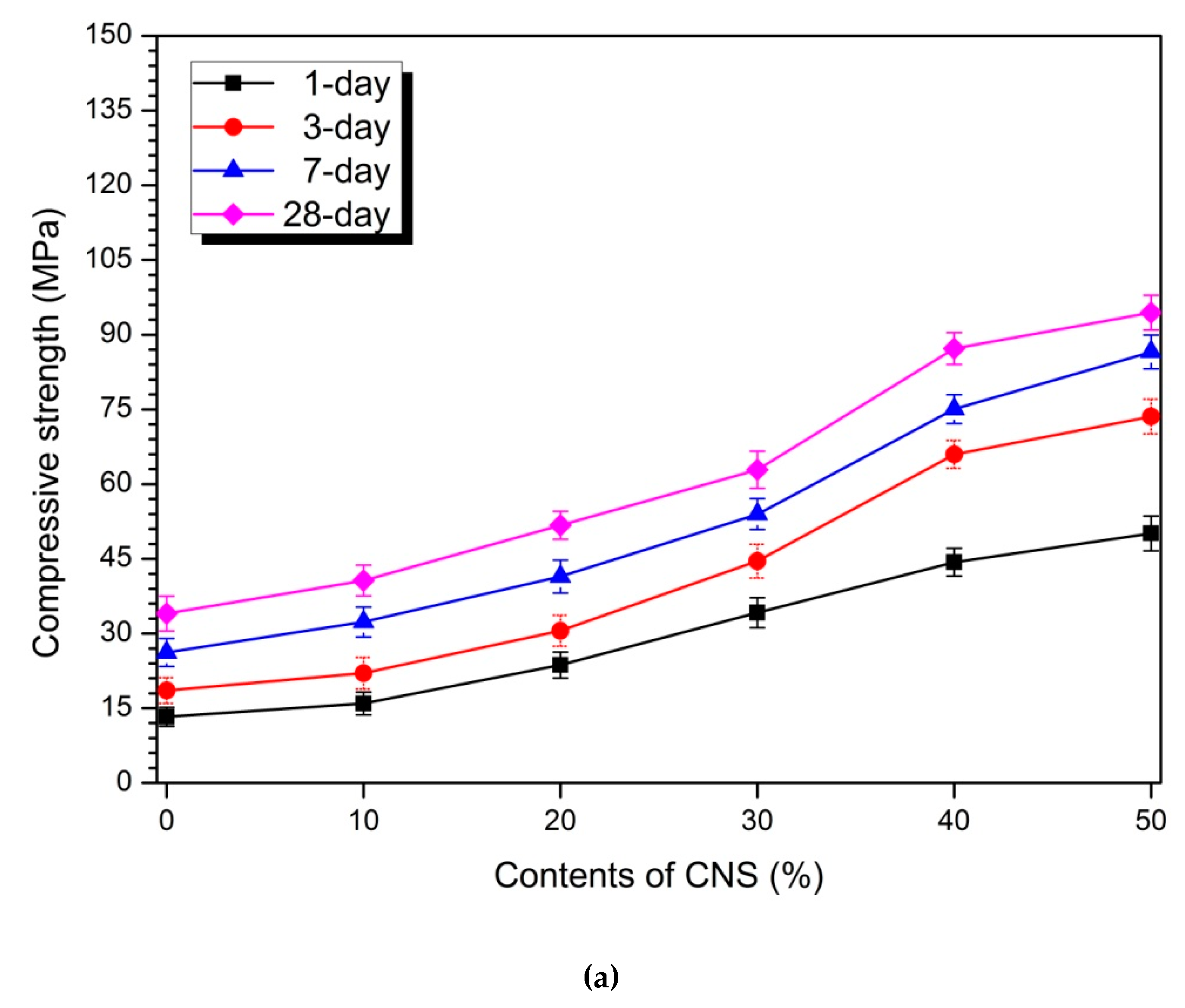
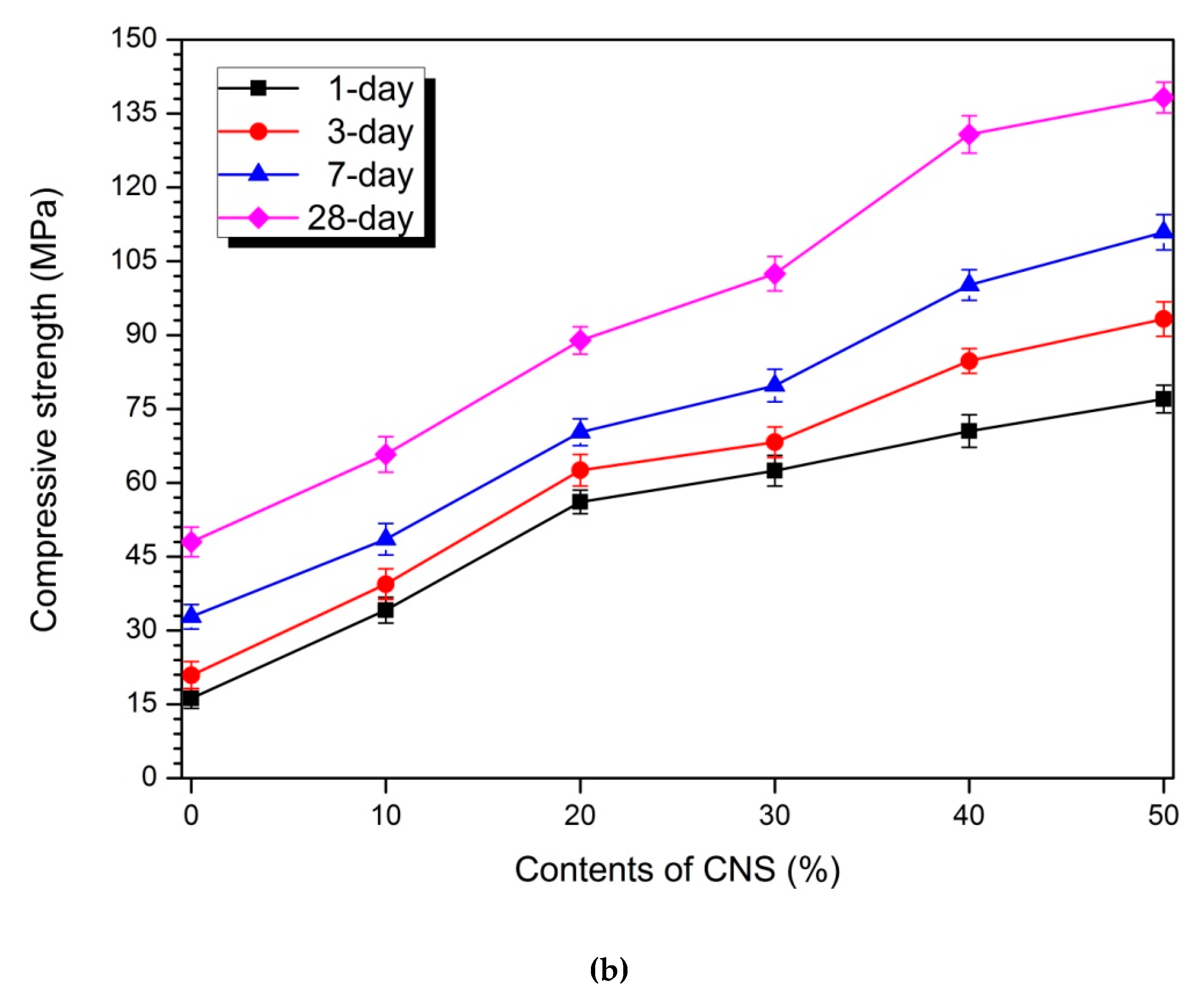
3.2. Compressive Strength
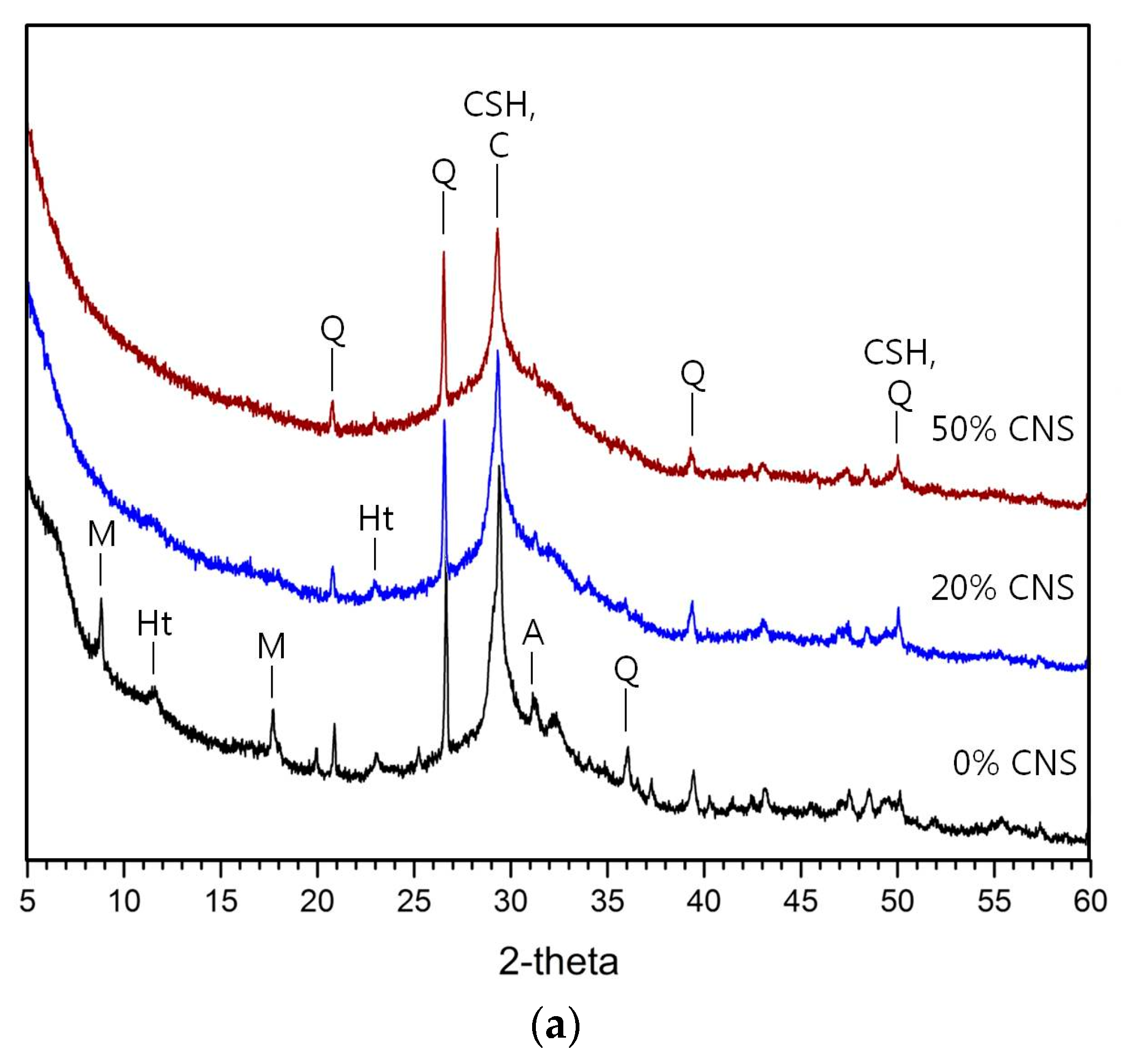
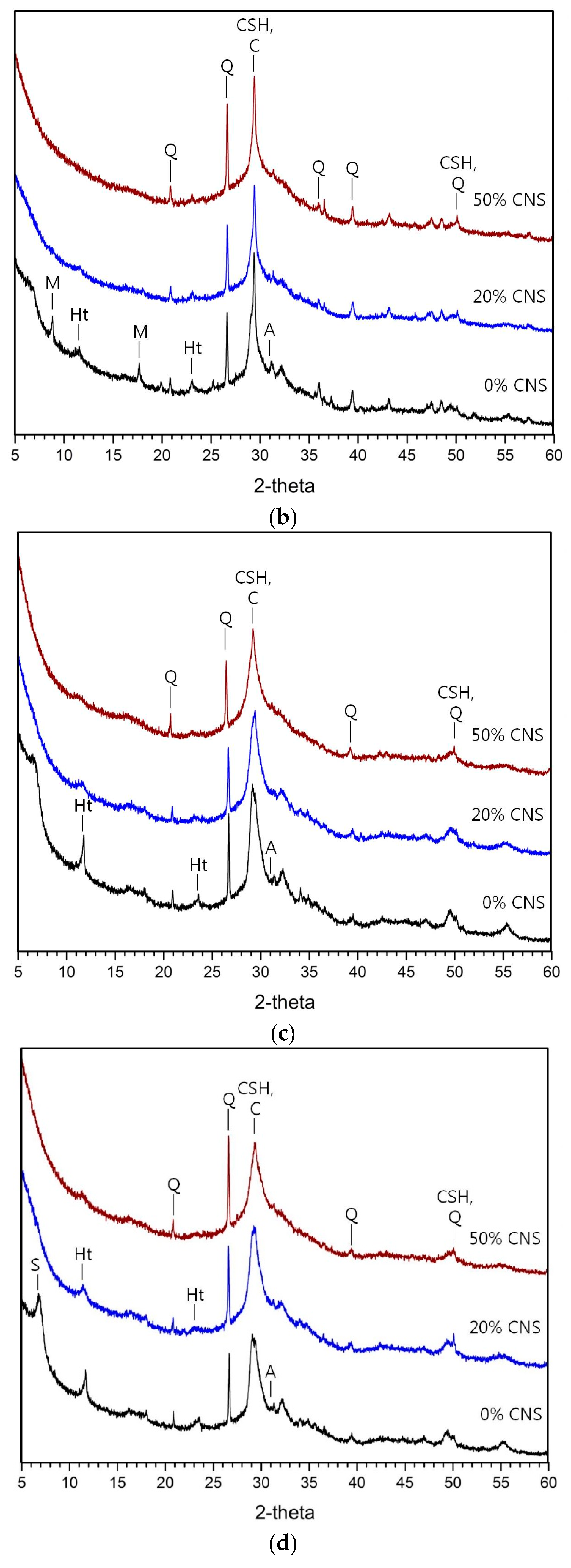
3.3. Hydration Products
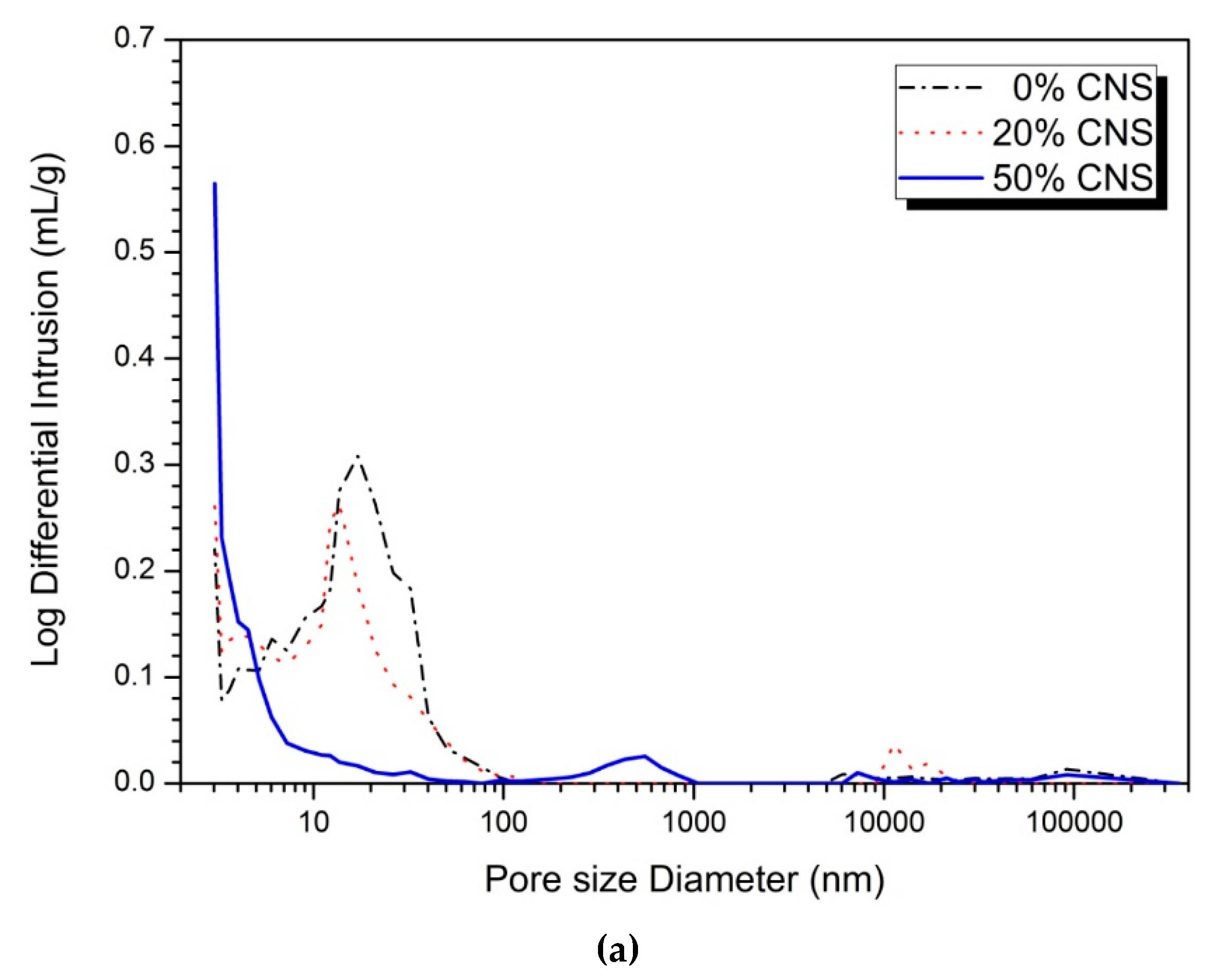
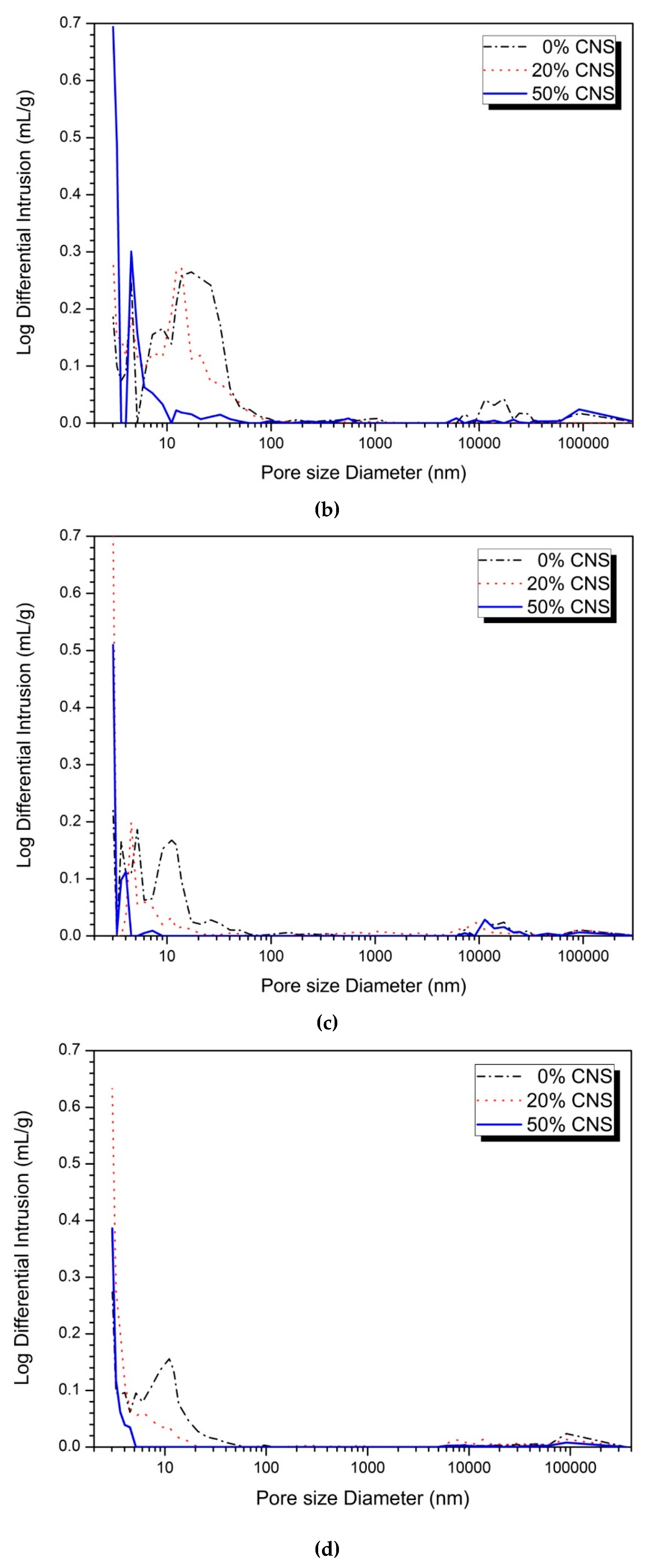
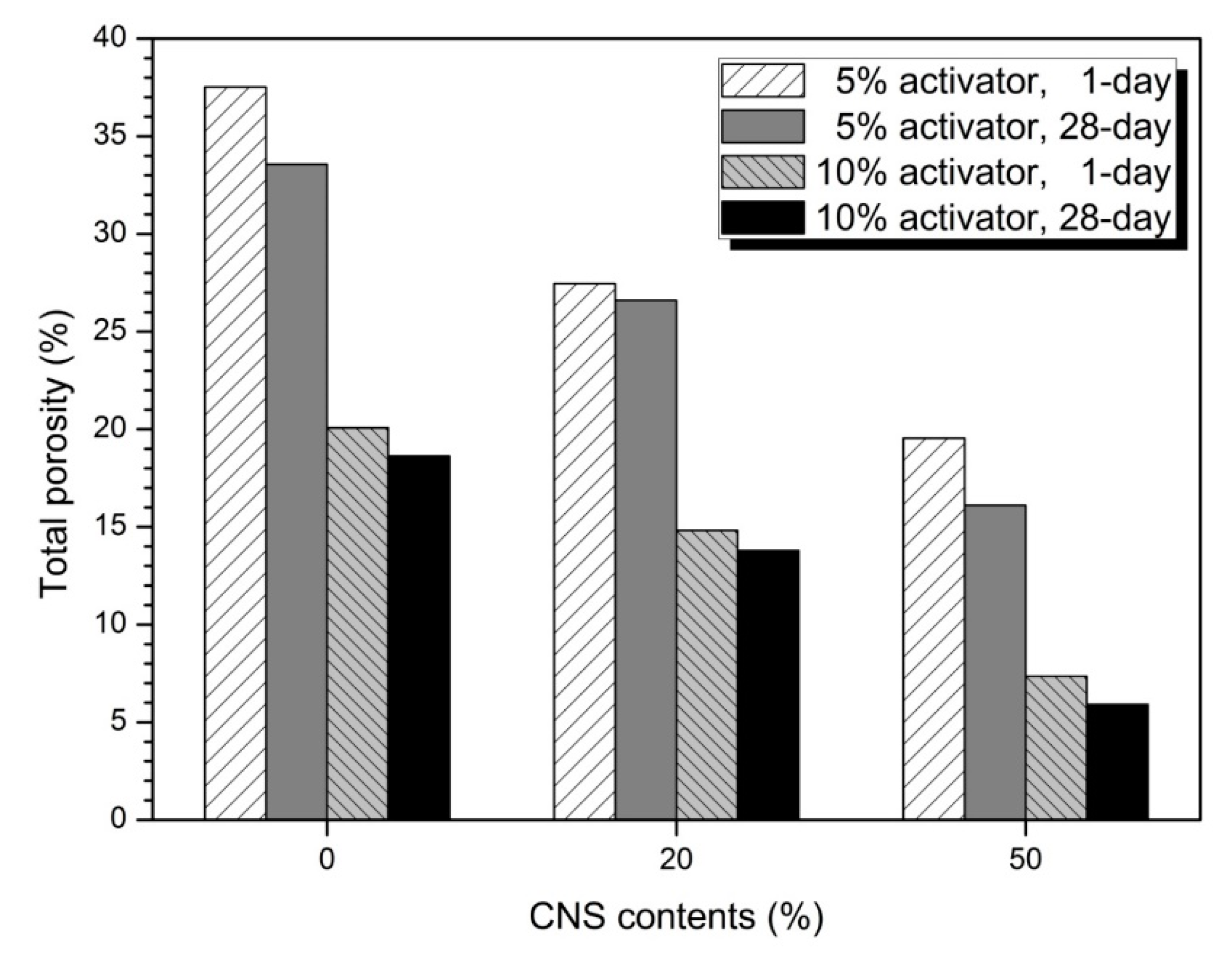
3.4. Pore Structure
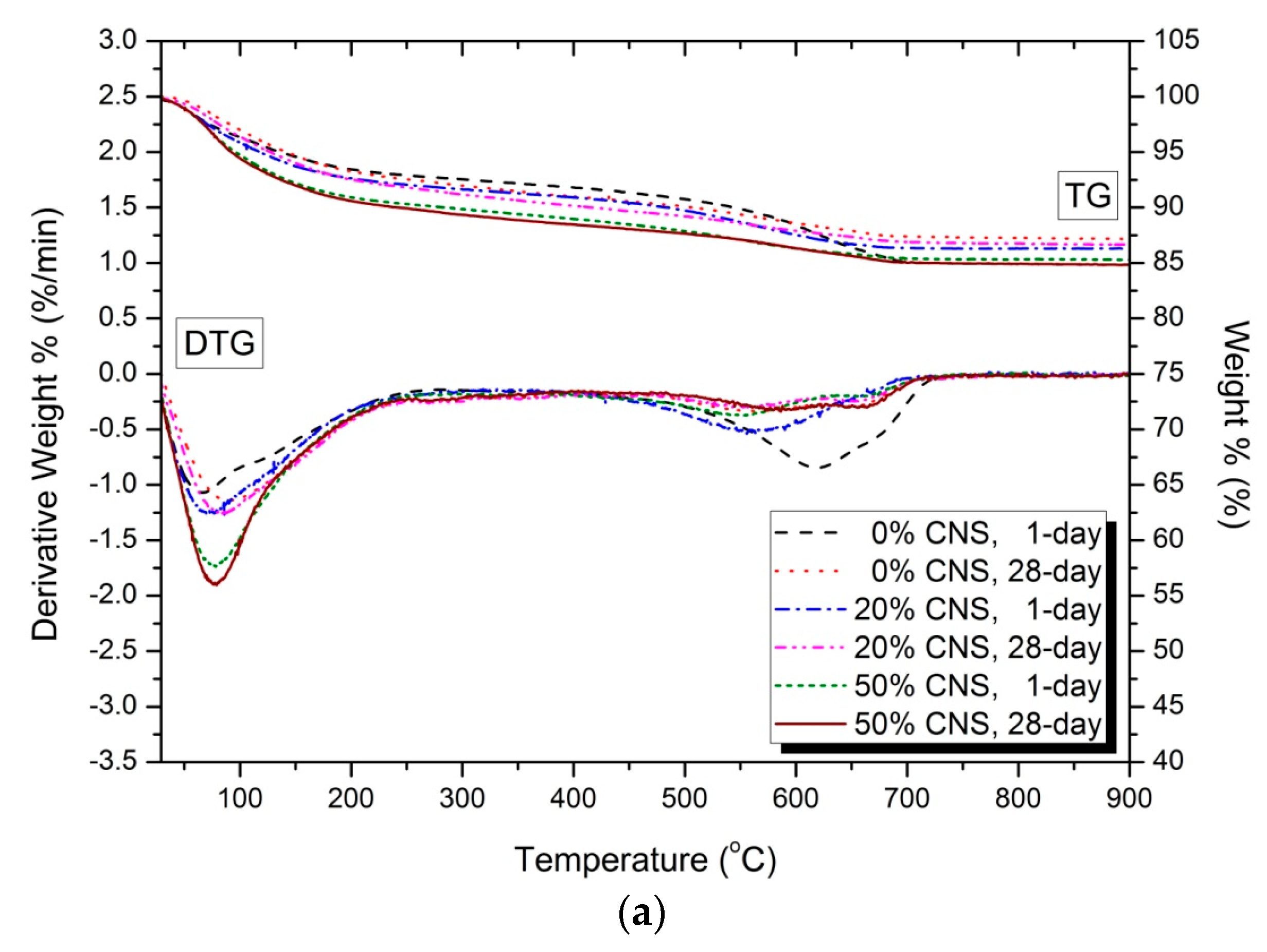
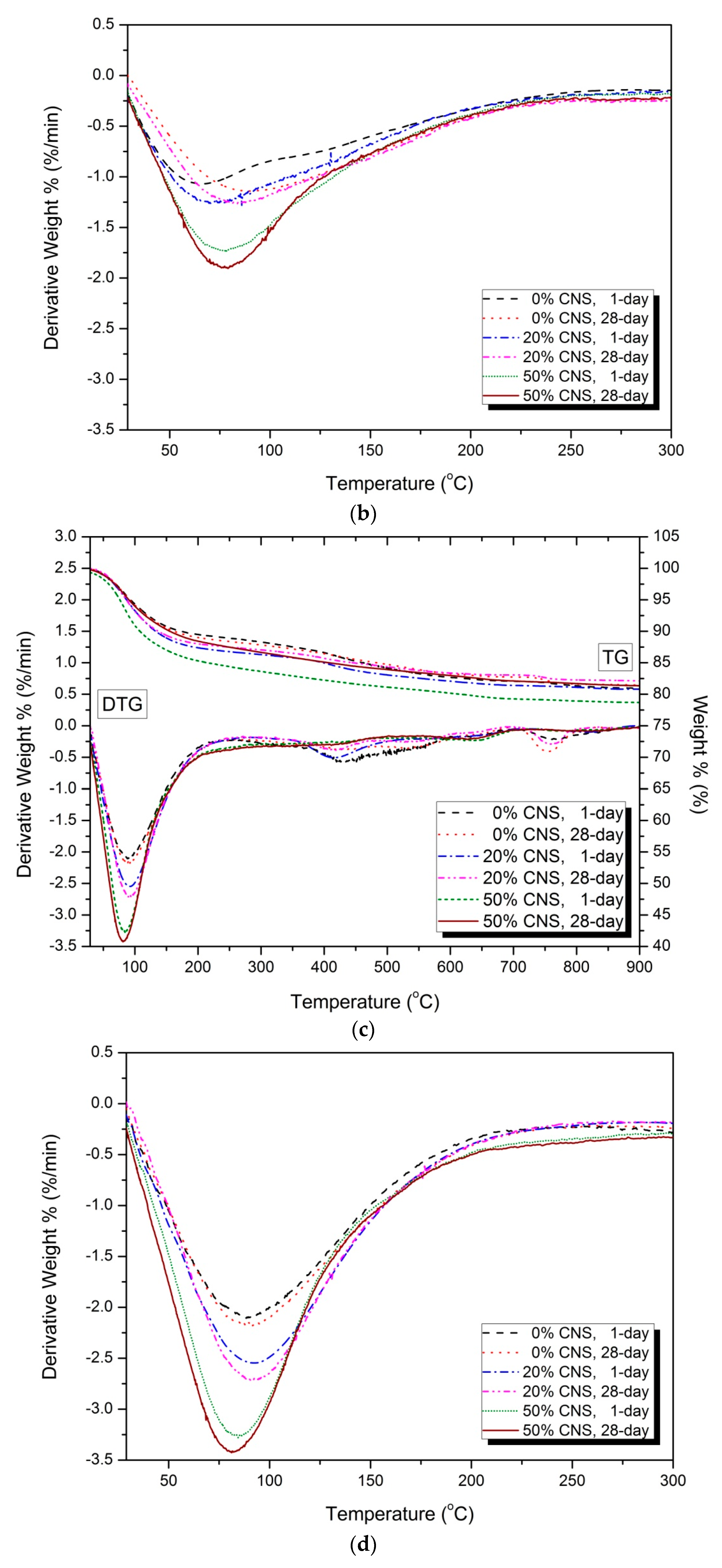
3.5. Thermal Analysis
3.6. Microstructure
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Singh, L.P.; Karade, S.R.; Bhattacharyya, S.K.; Yousuf, M.M.; Ahalawat, S. Beneficial role of nanosilica in cement based materials—A review. Constr. Build. Mater. 2013, 47, 1069–1077. [Google Scholar] [CrossRef]
- Norhasri, M.S.M.; Hamidah, M.S.; Mohd Fadzil, A. Applications of using nano material in concrete: A review. Constr. Build. Mater. 2017, 133, 91–97. [Google Scholar] [CrossRef]
- Sanchez, F.; Sobolev, K. Nanotechnology in concrete—A review. Constr. Build. Mater. 2010, 24, 2060–2071. [Google Scholar] [CrossRef]
- Ardalan, R.B.; Jamshidi, N.; Arabameri, H.; Joshaghani, A.; Mehrinejad, M.; Sharafi, P. Enhancing the permeability and abrasion resistance of concrete using colloidal nano-SiO2 oxide and spraying nanosilicon practices. Constr. Build. Mater. 2017, 146, 128–135. [Google Scholar] [CrossRef]
- Said, A.M.; Zeidan, M.S.; Bassuoni, M.T.; Tian, Y. Properties of concrete incorporating nano-silica. Constr. Build. Mater. 2012, 36, 838–844. [Google Scholar] [CrossRef]
- Shekari, A.H.; Razzaghi, M.S. Influence of nano particles on durability and mechanical properties of high performance concrete. Procedia Eng. 2011, 14, 3036–3041. [Google Scholar] [CrossRef]
- Oltulu, M.; Şahin, R. Effect of nano-SiO2, nano-Al2O3 and nano-Fe2O3 powders on compressive strengths and capillary water absorption of cement mortar containing fly ash: A comparative study. Energy Build. 2013, 58, 292–301. [Google Scholar] [CrossRef]
- Qing, Y.; Zenan, Z.; Deyu, K.; Rongshen, C. Influence of nano-SiO2 addition on properties of hardened cement paste as compared with silica fume. Constr. Build. Mater. 2007, 21, 539–545. [Google Scholar] [CrossRef]
- Xu, Z.; Zhou, Z.; Du, P.; Cheng, X. Effects of nano-silica on hydration properties of tricalcium silicate. Constr. Build. Mater. 2016, 125, 1169–1177. [Google Scholar] [CrossRef]
- Liu, M.; Zhou, Z.; Zhang, X.; Yang, X.; Cheng, X. The synergistic effect of nano-silica with blast-furnace slag in cement based materials. Constr. Build. Mater. 2016, 126, 624–631. [Google Scholar] [CrossRef]
- Wang, F.; Chen, P.; Li, X.; Zhu, B. Effect of colloidal silica on the hydration behavior of calcium aluminate cement. Materials 2018, 11, 1849. [Google Scholar] [CrossRef]
- Najigivi, A.; Khaloo, A.; Rashid, S.A. Investigating the effects of using different types of SiO2 nanoparticles on the mechanical properties of binary blended concrete. Compos. Part B 2013, 54, 52–58. [Google Scholar] [CrossRef]
- Chithra, S.; Kumar, S.R.R.S.; Chinnaraju, K. The effect of colloidal nano-silica on workability, mechanical and durability properties of high performance concrete with copper slag as partial fine aggregate. Constr. Build. Mater. 2016, 113, 794–804. [Google Scholar] [CrossRef]
- Vance, K.; Kumar, A.; Sant, G.; Neithalath, N. The rheological properties of ternary binders containing Portland cement, limestone, and metakaolin or fly ash. Cem. Concr. Res. 2013, 52, 196–207. [Google Scholar] [CrossRef]
- Szeląg, M. Development of cracking patterns in modified cement matrix with microsilica. Materials 2018, 11, 1928. [Google Scholar] [CrossRef]
- Senff, L.; Labrincha, J.A.; Ferreira, V.M.; Hotza, D.; Repette, W.L. Effect of nano-silica on rheology and fresh properties of cement pastes and mortars. Constr. Build. Mater. 2009, 23, 2487–2491. [Google Scholar] [CrossRef]
- Li, L.G.; Huang, Z.H.; Zhu, J.; Kwan, A.K.H.; Chen, H.Y. Synergistic effects of micro-silica and nano-silica on strength and microstructure of mortar. Constr. Build. Mater. 2017, 140, 229–238. [Google Scholar] [CrossRef]
- Quercia, G.; Hüsken, G.; Brouwers, H.J.H. Water demand of amorphous nano silica and its impact on the workability of cement paste. Cem. Concr. Compos. 2012, 42, 344–357. [Google Scholar] [CrossRef]
- Gao, X.; Yu, Q.L.; Brouwers, H.J.H. Characterization of alkali activated slag–fly ash blends containing nano-silica. Constr. Build. Mater. 2015, 98, 397–406. [Google Scholar] [CrossRef]
- Long, W.-J.; Xiao, B.-X.; Gu, Y.-C.; Xing, F. Micro- and macro-scale characterization of nano-SiO2 reinforced alkali activated slag composites. Mat. Charact. 2018, 136, 111–121. [Google Scholar] [CrossRef]
- Behfarnia, K.; Rostami, M. Effects of micro and nanoparticles of SiO2 on the permeability of alkali activated slag concrete. Constr. Build. Mater. 2017, 131, 205–213. [Google Scholar] [CrossRef]
- Gao, K.; Lin, K.-L.; Wang, D.Y.; Hwang, C.-L.; Tuan, B.L.A.; Shiu, H.-S.; Cheng, T.-W. Effect of nano-SiO2 on the alkali-activated characteristics of metakaolin-based geopolymers. Constr. Build. Mater. 2013, 48, 441–447. [Google Scholar] [CrossRef]
- Adak, D.; Sarkar, M.; Mandal, S. Structural performance of nano-silica modified fly-ash based geopolymer concrete. Constr. Build. Mater. 2017, 135, 430–439. [Google Scholar] [CrossRef]
- Lo, K.-W.; Lin, K.-L.; Cheng, T.-W.; Chang, Y.-M.; Lan, J.-Y. Effect of nano-SiO2 on the alkali-activated characteristics of spent catalyst metakaolin-based geopolymers. Constr. Build. Mater. 2017, 143, 455–463. [Google Scholar] [CrossRef]
- Çevika, A.; Alzeebaree, R.; Humur, G.; Niş, A.; Gülşan, M.E. Effect of nano-silica on the chemical durability and mechanical performance of fly ash based geopolymer concrete. Ceram. Int. 2018, 44, 12253–12264. [Google Scholar] [CrossRef]
- Prakasam, G.; Murthy, A.R.; Reheman, M.S. Mechanical, durability and fracture properties of nano-modified FA/GGBS geopolymer mortar. Mag. Concr. Res. 2018. [Google Scholar] [CrossRef]
- Sumesh, M.; Alengaram, U.J.; Jumaat, M.Z.; Kim, H.M.; Alnahhal, M.F. Incorporation of nano-materials in cement composite and geopolymer based paste and mortar—A review. Constr. Build. Mater. 2017, 148, 62–84. [Google Scholar] [CrossRef]
- Jo, B.W.; Kim, C.H.; Tae, G.H.; Park, J.B. Characteristics of cement mortar with nano-SiO2 particles. Constr. Build. Mater. 2007, 21, 1351–1355. [Google Scholar] [CrossRef]
- Gaitero, J.J.; Campillo, I.; Guerrero, A. Reduction of the calcium leaching rate of cement paste by addition of silica nanoparticles. Cem. Concr. Res. 2008, 38, 1112–1118. [Google Scholar] [CrossRef]
- Haha, M.B.; Lothenbach, B.; Le Saout, G.; Winnefeld, F. Influence of slag chemistry on the hydration of alkali-activated blast-furnace slag—Part I: Effect of MgO. Cem. Concr. Res. 2011, 41, 955–963. [Google Scholar] [CrossRef]
- Assaedi, H.; Shaikh, F.U.A.; Low, I.M. Influence of mixing methods of nano silica on the microstructural and mechanical properties of flax fabric reinforced geopolymer composites. Constr. Build. Mater. 2016, 123, 541–552. [Google Scholar] [CrossRef]
- Mukharjee, B.B.; Barai, S.V. Assessment of the influence of nano-silica on the behavior of mortar using factorial design of experiments. Constr. Build. Mater. 2014, 68, 416–425. [Google Scholar] [CrossRef]
- Boshehrian, A.; Hosseini, P. Effect of nano-SiO2 particles on properties of cement mortar applicable for ferrocement elements. Concr. Res. Lett. 2011, 2, 167–180. [Google Scholar]
- Ramezanianpour, A.A.; Moeini, M.A. Mechanical and durability properties of alkali activated slag coating mortars containing nanosilica and silica fume. Constr. Build. Mater. 2018, 163, 611–621. [Google Scholar] [CrossRef]
- Ibrahim, M.; Johari, M.A.M.; Maslehuddin, M.; Rahman, M.K. Influence of nano-SiO2 on the strength and microstructure of natural pozzolan based alkali activated concrete. Constr. Build. Mater. 2018, 173, 573–585. [Google Scholar] [CrossRef]
- Adak, D.; Sarkar, M.; Mandal, S. Effect of nano-silica on strength and durability of fly ash based geopolymer mortar. Constr. Build. Mater. 2014, 70, 453–459. [Google Scholar] [CrossRef]
- Deb, P.S.; Sarker, P.K.; Barbhuiya, S. Sorptivity and acid resistance of ambient-cured geopolymer mortars containing nano-silica. Cem. Concr. Compos. 2016, 72, 235–245. [Google Scholar] [CrossRef]
- Rashad, A.M. Effect of nanoparticles on the properties of geopolymer materials. Mag. Concr. Res. 2019. [Google Scholar] [CrossRef]
- Phoo-ngernkham, T.; Chindaprasirt, P.; Sata, V.; Hanjitsuwan, S.; Hatanaka, S. The effect of adding nano-SiO2 and nano-Al2O3 on properties of high calcium fly ash geopolymer cured at ambient temperature. Mater. Des. 2014, 55, 58–65. [Google Scholar] [CrossRef]
- Wang, J.; Zhou, T.; Xu, D.; Zhou, Z.; Du, P.; Xie, N.; Cheng, X.; Liu, Y. Effect of nano-silica on the efflorescence of waste based alkali-activated inorganic binder. Constr. Build. Mater. 2018, 167, 381–390. [Google Scholar] [CrossRef]
- Deb, P.S.; Sarker, P.K.; Barbhuiya, S. Effects of nano-silica on the strength development of geopolymer cured at room temperature. Constr. Build. Mater. 2015, 101, 675–683. [Google Scholar] [CrossRef]
- Shahrajabian, F.; Behfarnia, K. The effects of nano particles on freeze and thaw resistance of alkali-activated slag concrete. Constr. Build. Mater. 2018, 176, 172–178. [Google Scholar] [CrossRef]
- Yang, L.Y.; Jia, Z.J.; Zhang, Y.M.; Dai, J.G. Effects of nano-TiO2 on strength, shrinkage and microstructure of alkali activated slag pastes. Cem. Concr. Compos. 2015, 57, 1–7. [Google Scholar] [CrossRef]
- Björnström, J.; Martinelli, A.; Matic, A.; Börjesson, L.; Panas, I. Accelerating effects of colloidal nano-silica for beneficial calcium-silicate-hydrate formation in cement. Chem. Phys. Lett. 2004, 392, 242–248. [Google Scholar] [CrossRef]
- ASTM C230. Standard Specification for FLow Table for Use in Tests of Hydraulic Cement; ASTM International: West Conshohocken, PA, USA, 2014. [Google Scholar]
- ASTM C266. Standard Test Method for Time of Setting of Hydraulic Cement Paste by Gillmore Needles; ASTM International: West Conshohocken, PA, USA, 2018. [Google Scholar]
- ASTM 305. Standard Practice for Mechanical Mixing of Hydray;ic Cement Pastes and Mortars of Plastic Consistency; ASTM International: West Conshohocken, PA, USA, 2014. [Google Scholar]
- ASTM C109. Standard Test Method for Compressive Strength of Hydraulic Cement Mortars (Using 2-in. or [50-mm] Cube Specimens); ASTM International: West Conshohocken, PA, USA, 2016. [Google Scholar]
- Mindess, S.; Young, J.F.; Darwin, D. Concrete, 2nd ed.; Prentice Hall: Upper Saddle River, NJ, USA, 2003. [Google Scholar]
- Haha, M.B.; Le Saout, G.; Winnefeld, F.; Lothenbach, B. Influence of activator type on hydration kinetics, hydrate assemblage and microstructural development of alkali activated blast-furnace slags. Cem. Concr. Res. 2011, 41, 301–310. [Google Scholar] [CrossRef]
- Bakolas, A.; Aggelakopoulou, E.; Moropoulou, A.; Anagnostopoulou, S. Evaluation of pozzolanic activity and physicomechanical characteristics in metakaolin‒lime pastes. J. Therm. Anal. Calorim. 2006, 84, 157–163. [Google Scholar] [CrossRef]
- Sha, W.; Pereira, G. Differential scanning calorimetry study of hydrated ground granulated blast-furnace slag. Cem. Concr. Res. 2001, 31, 327–329. [Google Scholar] [CrossRef]
- Wang, S.-D.; Scrivener, K.L. Hydration products of alkali activated slag cement. Cem.Concr. Res. 1995, 25, 561–571. [Google Scholar] [CrossRef]
- Jeon, D.; Jun, Y.; Jeong, Y.; Oh, J.E. Microstructural and strength improvements through the use of Na2CO3 in a cementless Ca(OH)2-activated Class F fly ash system. Cem. Concr. Res. 2015, 67, 215–225. [Google Scholar] [CrossRef]
- Vassileva, C.G.; Vassilev, S.V. Behaviour of inorganic matter during heating of Bulgarian coals: 1. Lignites Fuel Process Tech. 2005, 86, 1297–1333. [Google Scholar] [CrossRef]
- Bernal, S.A.; de Gutiérrez, R.M.; Pedraza, A.L.; Provis, J.L.; Rodriguez, E.D.; Delvasto, S. Effect of binder content on the performance of alkali-activated slag concretes. Cem. Concr. Res. 2011, 41, 1–8. [Google Scholar] [CrossRef]
- Nedeljković, M.; Ghiassi, B.; Melzer, S.; Kooij, C.; van der Laan, S.; Ye, G. CO2 binding capacity of alkali-activated fly ash and slag pastes. Ceram. Inter. 2018, 44, 19646–19660. [Google Scholar] [CrossRef]



| Chemical Components (%) | Density (g/cm3) | Fineness (m2/kg) | LOI (%) | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| SiO2 | Al2O | Fe2O | MgO | CaO | K2O | SO3 | ||||
| slag | 21.32 | 5.61 | 3.12 | 3.94 | 61.72 | 0.79 | 2.51 | 2.84 | 420 | 1.86 |
| w/b Considering Water Including CNS | CNS Replacement Ratios | Ratio of SiO2 in CNS to Binder Weight (%) | Slag (g) | Mixing Water (g) | CNS (g) | |
|---|---|---|---|---|---|---|
| CNS to Mixing Water Ratio (%) | CNS to Binder Ratio (%) | |||||
| 0.500 | 0 | 0 | 0 | 1000 | 500 | 0 |
| 0.485 | 10 | 5 | 1.5 | 1000 | 450 | 50 |
| 0.470 | 20 | 10 | 3.0 | 1000 | 400 | 100 |
| 0.450 | 30 | 15 | 4.5 | 1000 | 350 | 150 |
| 0.440 | 40 | 20 | 6.0 | 1000 | 300 | 200 |
| 0.425 | 50 | 25 | 7.5 | 1000 | 250 | 250 |
| Contents of CNS (%) | 5% Activator | 10% Activator | ||||||
|---|---|---|---|---|---|---|---|---|
| 1-day | 3-day | 7-day | 28-day | 1-day | 3-day | 7-day | 28-day | |
| 10 | 20.10 | 18.69 | 23.41 | 19.42 | 110.54 | 88.47 | 47.95 | 37.10 |
| 20 | 78.31 | 64.65 | 58.22 | 52.01 | 246.14 | 199.18 | 114.26 | 85.23 |
| 30 | 157.45 | 140.08 | 106.07 | 84.78 | 285.25 | 226.35 | 143.17 | 113.44 |
| 40 | 233.73 | 255.44 | 186.63 | 156.24 | 334.97 | 305.30 | 205.35 | 172.43 |
| 50 | 277.25 | 296.44 | 230.43 | 177.52 | 375.07 | 346.11 | 238.04 | 188.00 |
| Activator (%) | CNS Content (%) | Large Capillary Pores (10,000‒50 nm) (%) | Medium Capillary Pores (50‒10 nm) (%) | Gel Pores (<10 nm) (%) | |||
|---|---|---|---|---|---|---|---|
| 1-day | 28-day | 1-day | 28-day | 1-day | 28-day | ||
| 5 | 0 | 3.49 | 3.94 | 57.25 | 56.34 | 39.26 | 39.72 |
| 20 | 3.34 | 3.59 | 46.66 | 44.66 | 50.00 | 51.75 | |
| 50 | 7.56 | 2.17 | 6.98 | 5.02 | 85.46 | 92.80 | |
| 10 | 0 | 3.06 | 1.07 | 32.06 | 31.22 | 64.88 | 67.71 |
| 20 | 8.53 | 2.50 | 6.43 | 5.31 | 85.05 | 92.71 | |
| 50 | 1.65 | 1.16 | 0.00 | 0.00 | 98.35 | 98.84 | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, T.; Kim, J.H.; Jun, Y. Properties of Alkali-Activated Slag Paste Using New Colloidal Nano-Silica Mixing Method. Materials 2019, 12, 1571. https://doi.org/10.3390/ma12091571
Kim T, Kim JH, Jun Y. Properties of Alkali-Activated Slag Paste Using New Colloidal Nano-Silica Mixing Method. Materials. 2019; 12(9):1571. https://doi.org/10.3390/ma12091571
Chicago/Turabian StyleKim, Taewan, Jae Hong Kim, and Yubin Jun. 2019. "Properties of Alkali-Activated Slag Paste Using New Colloidal Nano-Silica Mixing Method" Materials 12, no. 9: 1571. https://doi.org/10.3390/ma12091571
APA StyleKim, T., Kim, J. H., & Jun, Y. (2019). Properties of Alkali-Activated Slag Paste Using New Colloidal Nano-Silica Mixing Method. Materials, 12(9), 1571. https://doi.org/10.3390/ma12091571






