Effect of Al Content in the Mg-Based Alloys on the Composition and Corrosion Resistance of Composite Hydroxide Films Formed by Steam Coating
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
3.1. Surface Characterizations
3.1.1. Surface Morphologies
3.1.2. FESEM-EDS Analyses of the Film Surfaces
3.2. Crystal Phases of Films Prepared on AZ31, AZ61, and AZ91D
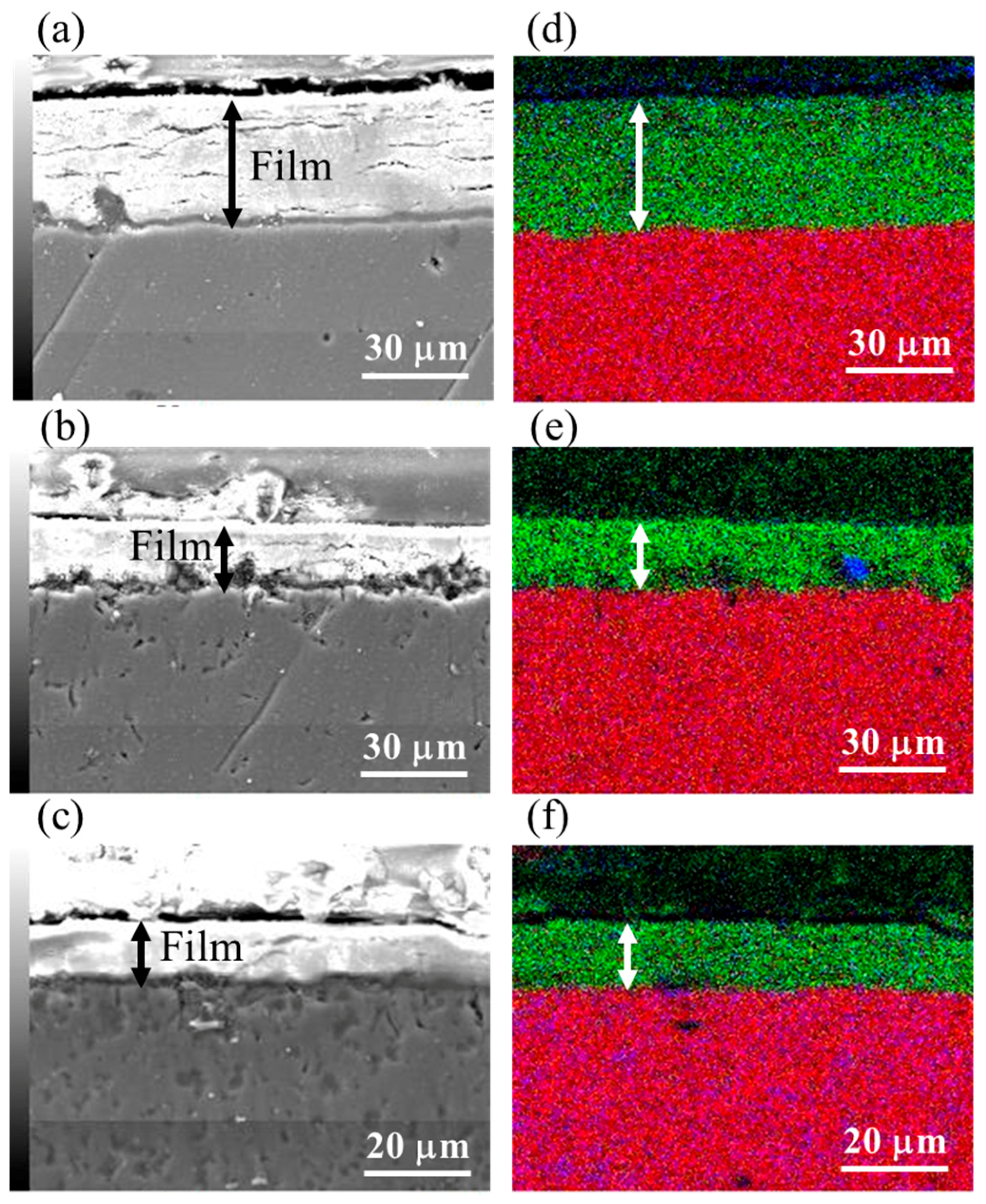
3.3. Film Thickness
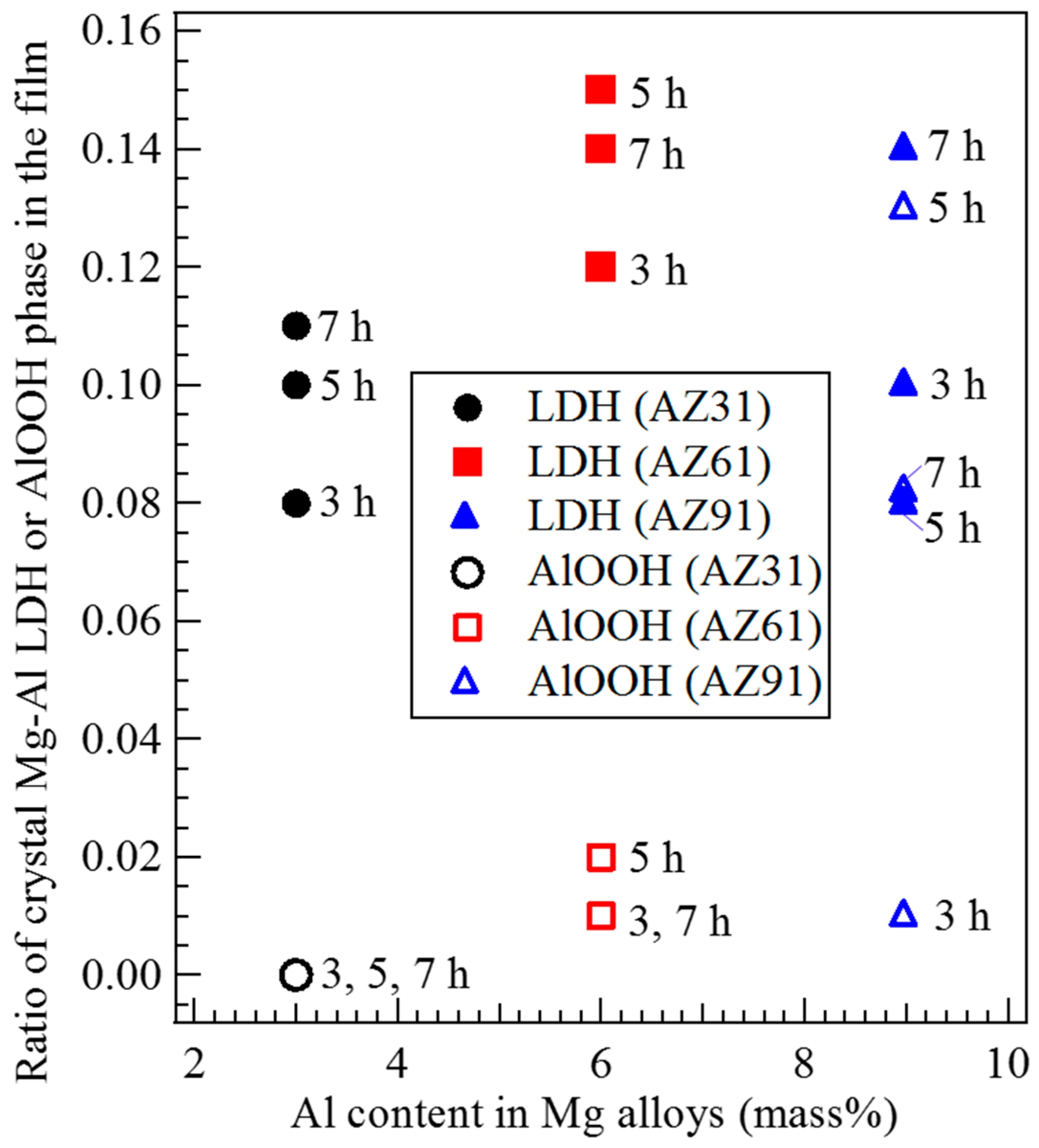
3.4. Effect of Al Content in the Mg Alloys on the Crystal Phases Formed in the Film
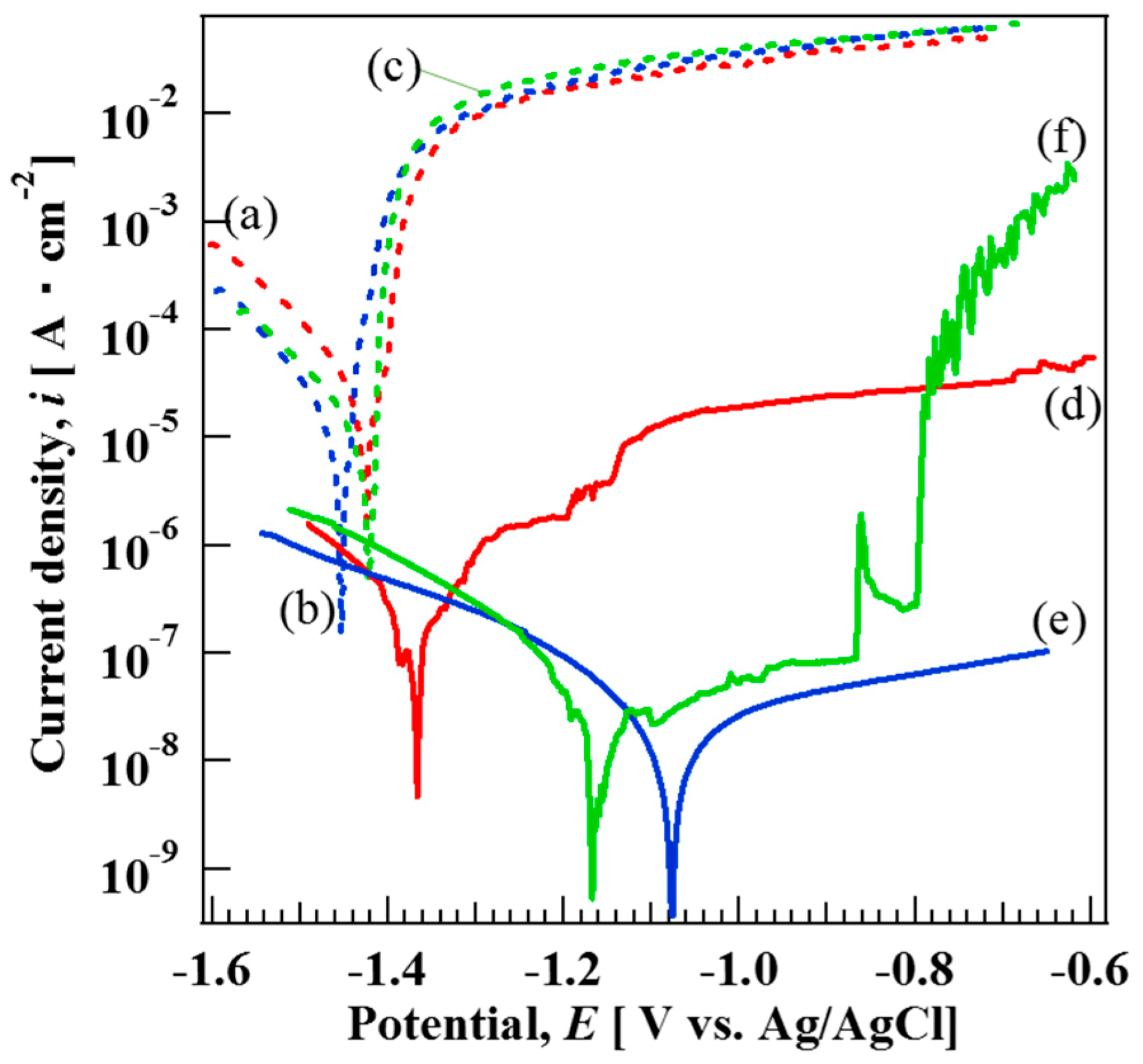
3.5. Corrosion Resistance of AZ31, AZ61, and AZ91D Mg Alloys Coated with and without Films
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Li, G.Y.; Lian, J.S.; Niu, L.Y.; Jiang, Z.H.; Jiang, A.; Black, Q.; White, B. Growth of zinc phosphate coatings on AZ91D magnesium alloy. Surf. Coat. Technol. 2006, 201, 1814–1820. [Google Scholar] [CrossRef]
- Hamdy, A.S.; Farahat, M. Chrome-free zirconia-based protective coatings for magnesium alloys. Surf. Coat. Technol. 2010, 204, 2834–2840. [Google Scholar] [CrossRef]
- Pardo, A.; Merino, M.C.; Coy, A.E.; Arrabal, R.; Viejo, F.; Matykina, E.; Quondam, M.X. Corrosion behaviour of magnesium/aluminium alloys in 3.5 mass% NaCl. Corros. Sci. 2008, 50, 823–834. [Google Scholar] [CrossRef]
- Gray, J.; Luan, B. Protective coatings on magnesium and its alloys—A critical review. J. Alloy. Compd. 2002, 336, 88–113. [Google Scholar] [CrossRef]
- Anık, M.; Körpe, E.; Anik, M. Effect of alloy microstructure on electroless NiP deposition behavior on Alloy AZ91. Surf. Coat. Technol. 2007, 201, 4702–4710. [Google Scholar] [CrossRef]
- Song, Y.; Shan, D.; Chen, R.; Han, E.-H. An environmentally friendly molybdate/phosphate black film on Mg–Zn–Y–Zr alloy. Surf. Coat. Technol. 2010, 204, 3182–3187. [Google Scholar] [CrossRef]
- Hu, J.; Li, Q.; Zhong, X.; Zhang, L.; Chen, B. Composite anticorrosion coatings for AZ91D magnesium alloy with molybdate conversion coating and silicon sol–gel coatings. Prog. Org. Coat. 2009, 66, 199–205. [Google Scholar] [CrossRef]
- Jayaraj, J.; Rajesh, K.; Raj, S.A.; Srinivasan, A.; Ananthakumar, S.; Dhaipule, N.G.K.; Kalpathy, S.K.; Pillai, U.; Mudali, U.K. Investigation on the corrosion behavior of lanthanum phosphate coatings on AZ31 Mg alloy obtained through chemical conversion technique. J. Alloy Compd. 2019, 784, 1162–1174. [Google Scholar] [CrossRef]
- Zhang, C.Y.; Liao, S.J.; Yu, B.X.; Lu, X.P.; Chen, X.B.; Zhang, T.; Wang, F.H. Ratio of total acidity to pH value of coating bath: A new strategy towards phosphate conversion coatings with optimized corrosion resistance for magnesium alloys. Corros. Sci. 2019, 150, 279–295. [Google Scholar]
- An, L.; Ma, Y.; Liu, Y.; Sun, L.; Wang, S.; Wang, Z. Effects of additives, voltage and their interactions on PEO coatings formed on magnesium alloys. Surf. Coat. Technol. 2018, 354, 226–235. [Google Scholar] [CrossRef]
- Shulha, T.N.; Serdechnova, M.; Lamaka, S.V.; Wieland, D.C.F.; Lapko, K.N.; Zheludkevich, M.L. Chelating agent-assisted in situ LDH growth on the surface of magnesium alloy. Sci. Rep. 2018, 8, 16409. [Google Scholar] [CrossRef] [PubMed]
- Gigandet, M.; Faucheu, J.; Tachez, M. Formation of black chromate conversion coatings on pure and zinc alloy electrolytic deposits: Role of the main constituents. Surf. Coat. Technol. 1997, 89, 285–291. [Google Scholar] [CrossRef]
- Anandan, C.; Grips, V.W.; Rajam, K.; Jayaram, V.; Bera, P. Investigation of surface composition of electrodeposited black chrome coatings by X-ray photoelectron spectroscopy. Appl. Surf. Sci. 2002, 191, 254–260. [Google Scholar] [CrossRef]
- Bayati, M.; Shariat, M.; Janghorban, K. Design of chemical composition and optimum working conditions for trivalent black chromium electroplating bath used for solar thermal collectors. Renew. Energy 2005, 30, 2163–2178. [Google Scholar] [CrossRef]
- Budavari, S. The Merck Index: An Encyclopedia of a Chemical, Drugs and Biologicals, 12th ed.; Merck Research Laboratories: Rahway, NJ, USA, 1996; p. 1475. [Google Scholar]
- Choi, Y.-I.; Kuroda, K.; Okido, M. Temperature-dependent corrosion behaviour of flame-resistant, Ca-containing AZX911 and AMX602 Mg alloys. Corros. Sci. 2016, 103, 181–188. [Google Scholar] [CrossRef]
- Li, Y.-G.; Wei, Y.-H.; Hou, L.-F.; Han, P.-J. Atmospheric corrosion of AM60 Mg alloys in an industrial city environment. Corros. Sci. 2013, 69, 67–76. [Google Scholar] [CrossRef]
- Song, G.; Atrens, A. Understanding Magnesium Corrosion—A Framework for Improved Alloy Performance. Adv. Eng. Mater. 2003, 12, 837–858. [Google Scholar] [CrossRef]
- Elsentriecy, H.H.; Azumi, K.; Konno, H. Effect of surface pretreatment by acid pickling on the density of stannate conversion coatings formed on AZ91 D magnesium alloy. Surf. Coat. Technol. 2007, 202, 532–537. [Google Scholar] [CrossRef]
- Chong, K.Z.; Shih, T.S. Conversion-coating treatment for magnesium alloys by a permanganate–phosphate solution. Mater. Chem. Phys. 2003, 80, 191–200. [Google Scholar] [CrossRef]
- Rudd, A.L.; Breslin, C.B.; Mansfeld, F. The corrosion protection afforded by rare earth conversion coatings applied to magnesium. Corros. Sci. 2000, 42, 275–288. [Google Scholar] [CrossRef]
- Takenaka, T.; Ono, T.; Narazaki, Y.; Naka, Y.; Kawakami, M. Improvement of corrosion resistance of magnesium metal by rare earth elements. Electrochim. Acta 2007, 53, 117–121. [Google Scholar] [CrossRef]
- Zucchi, F.; Frignani, A.; Grassi, V.; Trabanelli, G.; Monticelli, C. Stannate and permanganate conversion coatings on AZ31 magnesium alloy. Corros. Sci. 2007, 49, 4542–4552. [Google Scholar] [CrossRef]
- Scholes, F.; Soste, C.; Hughes, A.; Hardin, S.; Curtis, P. The role of hydrogen peroxide in the deposition of cerium-based conversion coatings. Appl. Surf. Sci. 2006, 253, 1770–1780. [Google Scholar] [CrossRef]
- Ishizaki, T.; Chiba, S.; Watanabe, K.; Suzuki, H. Corrosion resistance of Mg–Al layered double hydroxide container-containing magnesium hydroxide films formed directly on magnesium alloy by chemical-free steam coating. J. Mater. Chem. A 2013, 1, 8968. [Google Scholar] [CrossRef]
- Ishizaki, T.; Kamiyama, N.; Watanabe, K.; Serizawa, A. Corrosion resistance of Mg(OH)2/Mg–Al layered double hydroxide composite film formed directly on combustion-resistant magnesium alloy AMCa602 by steam coating. Corros. Sci. 2015, 92, 76–84. [Google Scholar] [CrossRef]
- Kamiyama, N.; Panomsuwan, G.; Yamamoto, E.; Sudare, T.; Saito, N.; Ishizaki, T. Effect of treatment time in the Mg(OH)2/Mg–Al LDH composite film formed on Mg alloy AZ31 by steam coating on the corrosion resistance. Surf. Coatings Technol. 2016, 286, 172–177. [Google Scholar] [CrossRef]
- Zhang, F.; Liu, Z.-G.; Zeng, R.-C.; Li, S.-Q.; Cui, H.-Z.; Song, L.; Han, E.-H. Corrosion resistance of Mg–Al-LDH coating on magnesium alloy AZ31. Surf. Coat. Technol. 2014, 258, 1152–1158. [Google Scholar] [CrossRef]
- Poznyak, S.K.; Tedim, J.; Rodrigues, L.M.; Salak, A.N.; Zheludkevich, M.L.; Dick, L.F.P.; Ferreira, M.G.S.; Poznyak, S.; Salak, A.; Zheludkevich, M.; et al. Novel Inorganic Host Layered Double Hydroxides Intercalated with Guest Organic Inhibitors for Anticorrosion Applications. ACS Appl. Mater. Interfaces 2009, 1, 2353–2362. [Google Scholar] [CrossRef]
- Zheludkevich, M.; Poznyak, S.; Rodrigues, L.; Raps, D.; Hack, T.; Dick, L.F.P.; Nunes, T.; Ferreira, M.; Zheludkevich, M.; Poznyak, S. Active protection coatings with layered double hydroxide nanocontainers of corrosion inhibitor. Corros. Sci. 2010, 52, 602–611. [Google Scholar] [CrossRef]
- Buchheit, R.G.; Guan, H.; Mahajanam, S.; Wong, F. Active corrosion protection and corrosion sensing in chromate-free organic coatings. Prog. Org. Coat. 2003, 47, 174–182. [Google Scholar] [CrossRef]
- Lin, J.; Hsia, C.; Uan, J. Characterization of Mg,Al-hydrotalcite conversion film on Mg alloy and Cl− and CO32- anion-exchangeability of the film in a corrosive environment. Scr. Mater. 2007, 56, 927–930. [Google Scholar] [CrossRef]
- Tedim, J.; Kuznetsova, A.; Salak, A.; Montemor, F.; Snihirova, D.; Pilz, M.; Zheludkevich, M.; Ferreira, M.; Salak, A.; Zheludkevich, M. Zn–Al layered double hydroxides as chloride nanotraps in active protective coatings. Corros. Sci. 2012, 55, 1–4. [Google Scholar] [CrossRef]
- Pardo, A.; Merino, M.; Coy, A.; Viejo, F.; Arrabal, R.; Feliu, S. Influence of microstructure and composition on the corrosion behaviour of Mg/Al alloys in chloride media. Electrochim. Acta 2008, 53, 7890–7902. [Google Scholar] [CrossRef]
- Nakamura, K.; Tsunakawa, M.; Shimada, Y.; Serizawa, A.; Ishizaki, T. Formation mechanism of Mg-Al layered double hydroxide-containing magnesium hydroxide films prepared on Ca-added flame-resistant magnesium alloy by steam coating. Surf. Coat. Technol. 2017, 328, 436–443. [Google Scholar] [CrossRef]
- Digne, M.; Sautet, P.; Raybaud, P.; Toulhoat, H.; Artacho, E. Structure and Stability of Aluminum Hydroxides: A Theoretical Study. J. Phys. Chem. B 2002, 106, 5155–5162. [Google Scholar] [CrossRef]
- Serizawa, A.; Oda, T.; Watanabe, K.; Mori, K.; Yokomizo, T.; Ishizaki, T. Formation of Anticorrosive Film for Suppressing Pitting Corrosion on Al-Mg-Si Alloy by Steam Coating. Coatings 2018, 8, 23. [Google Scholar] [CrossRef]
- Tanaka, D.K.; Long, G.G.; Kruger, J. Structure of the passive films on cast and rapidly solidified Mg alloys. In Proceedings of the 11th International Corrosion Congress, Florence, Italy, 2–9 April 1990. [Google Scholar]
- Baril, G.; Galicia, G.; Deslouis, C.; Pébère, N.; Tribollet, B.; Vivier, V. An Impedance Investigation of the Mechanism of Pure Magnesium Corrosion in Sodium Sulfate Solutions. J. Electrochem. Soc. 2007, 154, C108–C113. [Google Scholar] [CrossRef]
- Muralidharan, V.S. An insight into the origin of corrosion potential—A critical review. Bull. Electrochem. 2002, 18, 179–186. [Google Scholar]


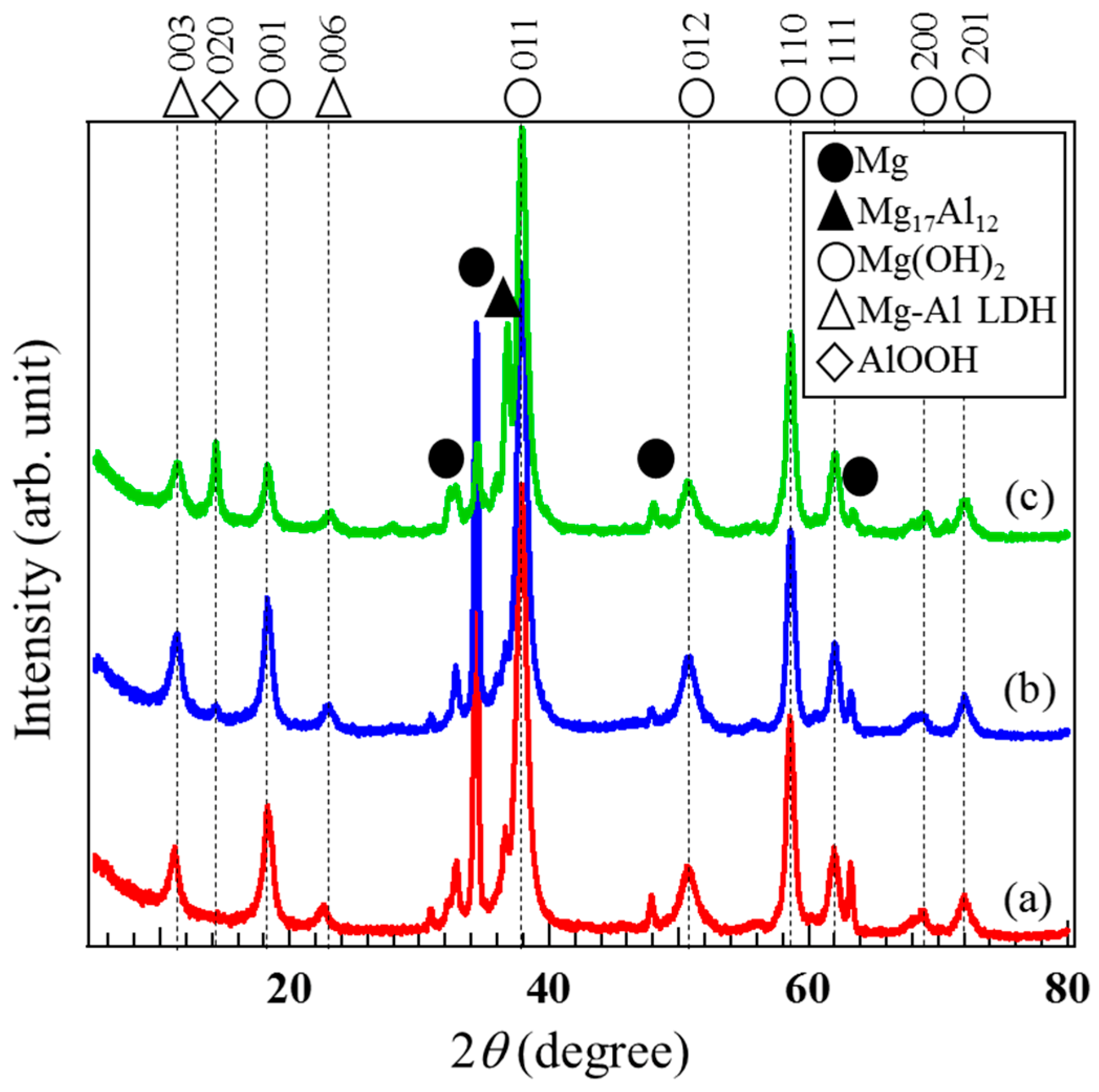


| Types of Mg Alloys | IMg(OH)2-001 | ILDH-003 | IAlOOH-020 |
|---|---|---|---|
| AZ31 | 0.90 | 0.10 | 0 |
| AZ61 | 0.83 | 0.15 | 0.02 |
| AZ91D | 0.79 | 0.08 | 0.13 |
| Types of Mg Alloys | 3 h | 5 h | 7 h |
|---|---|---|---|
| AZ31 | 5.7 | 37.8 | 44.2 |
| AZ61 | 4.8 | 20.0 | 38.5 |
| AZ91D | 4.8 | 11.9 | 26.9 |
| Types of Mg Alloys | Corrosion Potential, Ecorr (V) | Corrosion Current Density, icorr (A cm−2) |
|---|---|---|
| (a) AZ31 bare | −1.42 | - |
| (b) AZ61 bare | −1.45 | - |
| (c) AZ91D bare | −1.42 | - |
| (d) AZ31 | −1.36 | 1.80 × 10−7 |
| (e) AZ61 | −1.07 | 2.37 × 10−8 |
| (f) AZ91D | −1.15 | 2.97 × 10−8 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ishizaki, T.; Miyashita, T.; Inamura, M.; Nagashima, Y.; Serizawa, A. Effect of Al Content in the Mg-Based Alloys on the Composition and Corrosion Resistance of Composite Hydroxide Films Formed by Steam Coating. Materials 2019, 12, 1188. https://doi.org/10.3390/ma12071188
Ishizaki T, Miyashita T, Inamura M, Nagashima Y, Serizawa A. Effect of Al Content in the Mg-Based Alloys on the Composition and Corrosion Resistance of Composite Hydroxide Films Formed by Steam Coating. Materials. 2019; 12(7):1188. https://doi.org/10.3390/ma12071188
Chicago/Turabian StyleIshizaki, Takahiro, Tomohiro Miyashita, Momo Inamura, Yuma Nagashima, and Ai Serizawa. 2019. "Effect of Al Content in the Mg-Based Alloys on the Composition and Corrosion Resistance of Composite Hydroxide Films Formed by Steam Coating" Materials 12, no. 7: 1188. https://doi.org/10.3390/ma12071188
APA StyleIshizaki, T., Miyashita, T., Inamura, M., Nagashima, Y., & Serizawa, A. (2019). Effect of Al Content in the Mg-Based Alloys on the Composition and Corrosion Resistance of Composite Hydroxide Films Formed by Steam Coating. Materials, 12(7), 1188. https://doi.org/10.3390/ma12071188







