A Double-Switch Temperature-Sensitive Controlled Release Antioxidant Film Embedded with Lyophilized Nanoliposomes Encapsulating Rosemary Essential Oils for Solid Food
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Lyophilized Nanoliposomes Containing REOs
2.3. Preparation of TSPU Solution
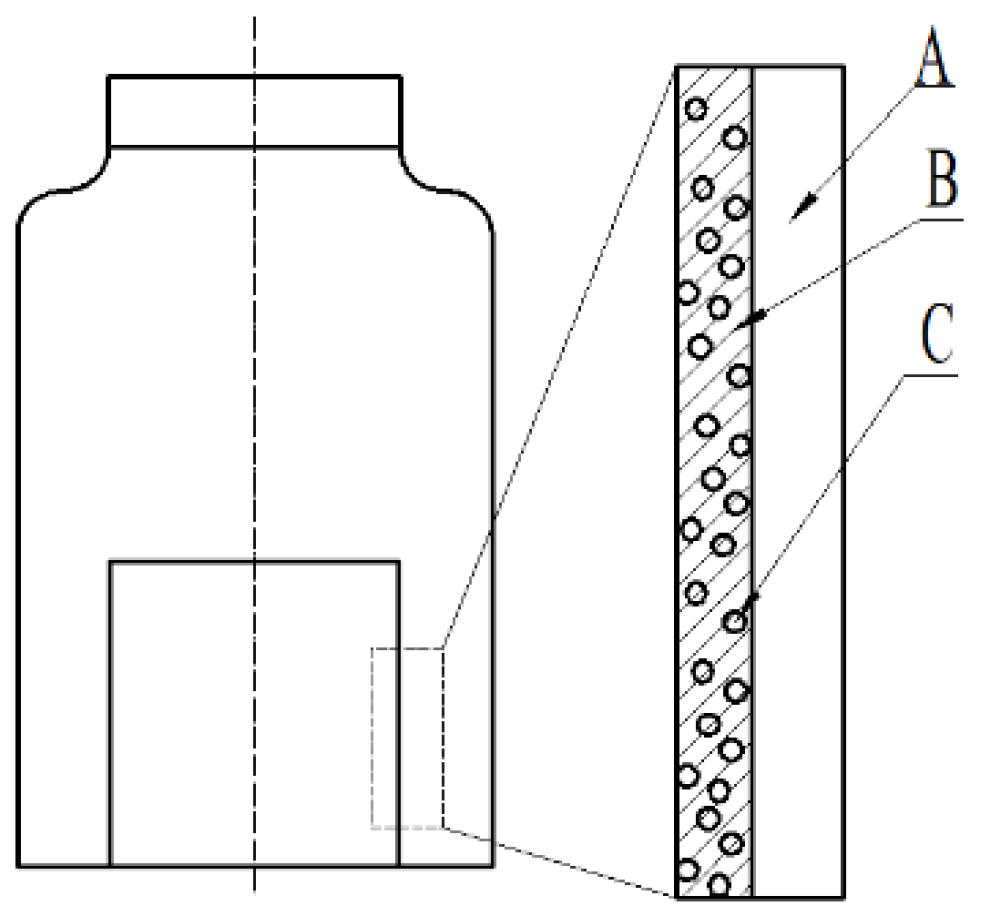
2.4. Preparation of Complex Antioxidant Film
2.5. Quantification of REOs
2.6. Encapsulation Efficiency and Particle Characterization
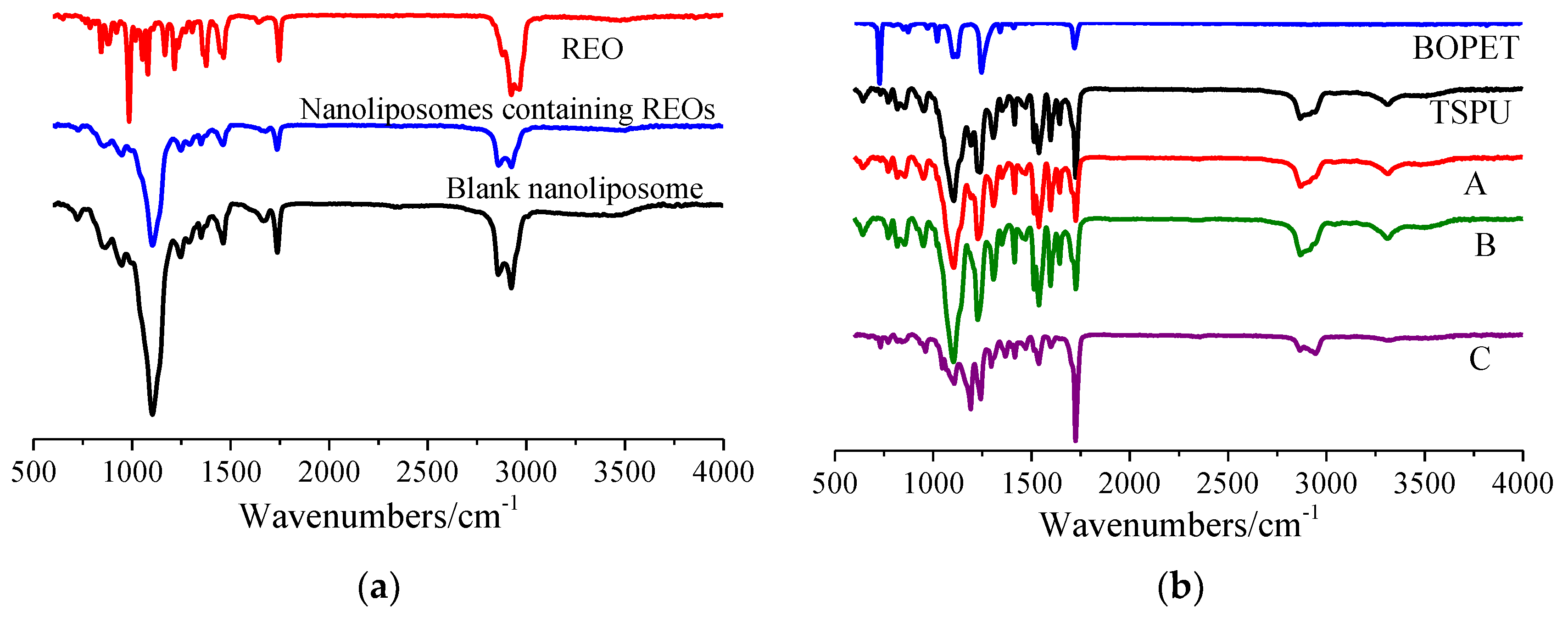
2.7. Fourier Transform Infrared (FT-IR) Spectroscopy
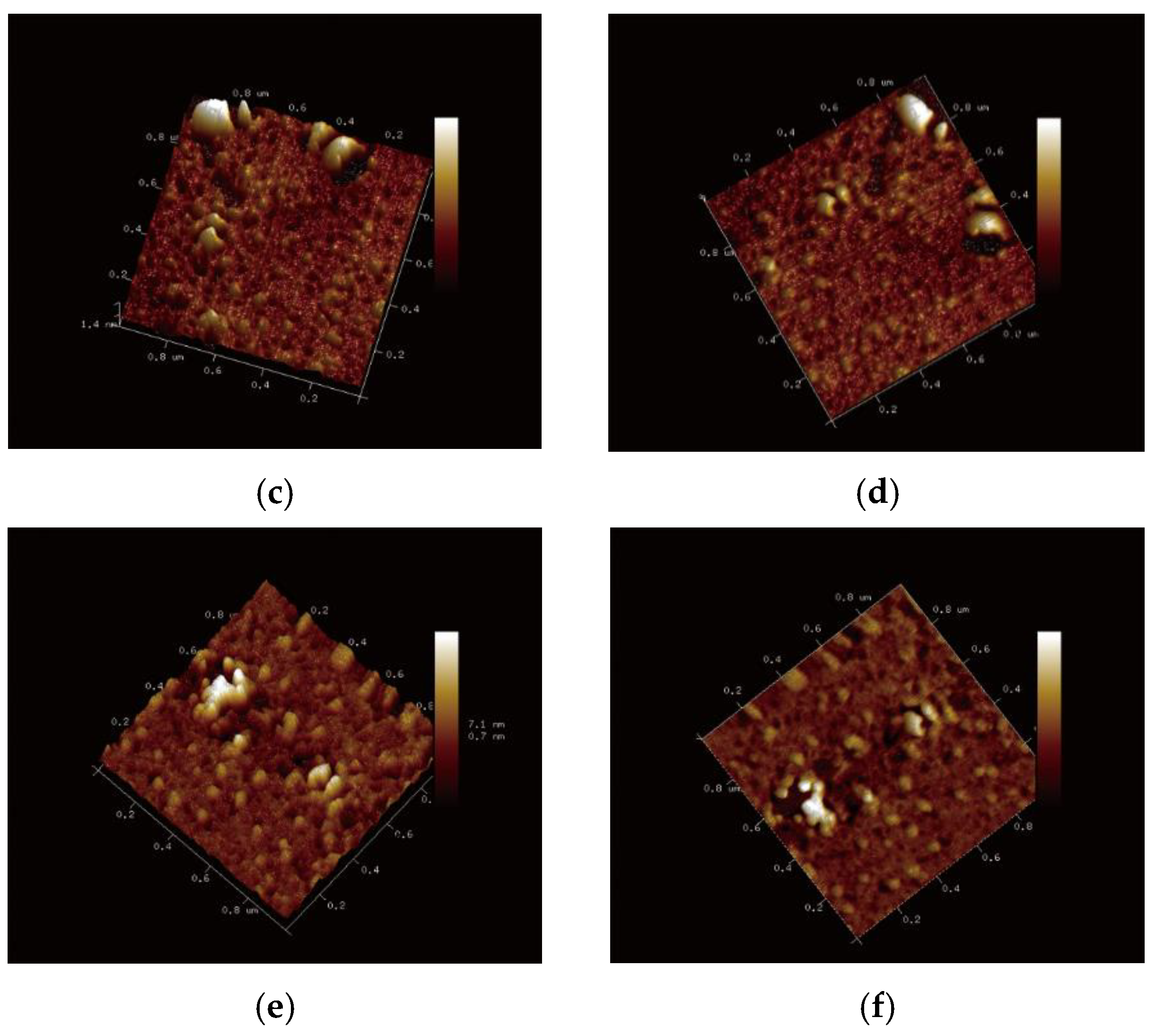
2.8. Micro Structure Analysis
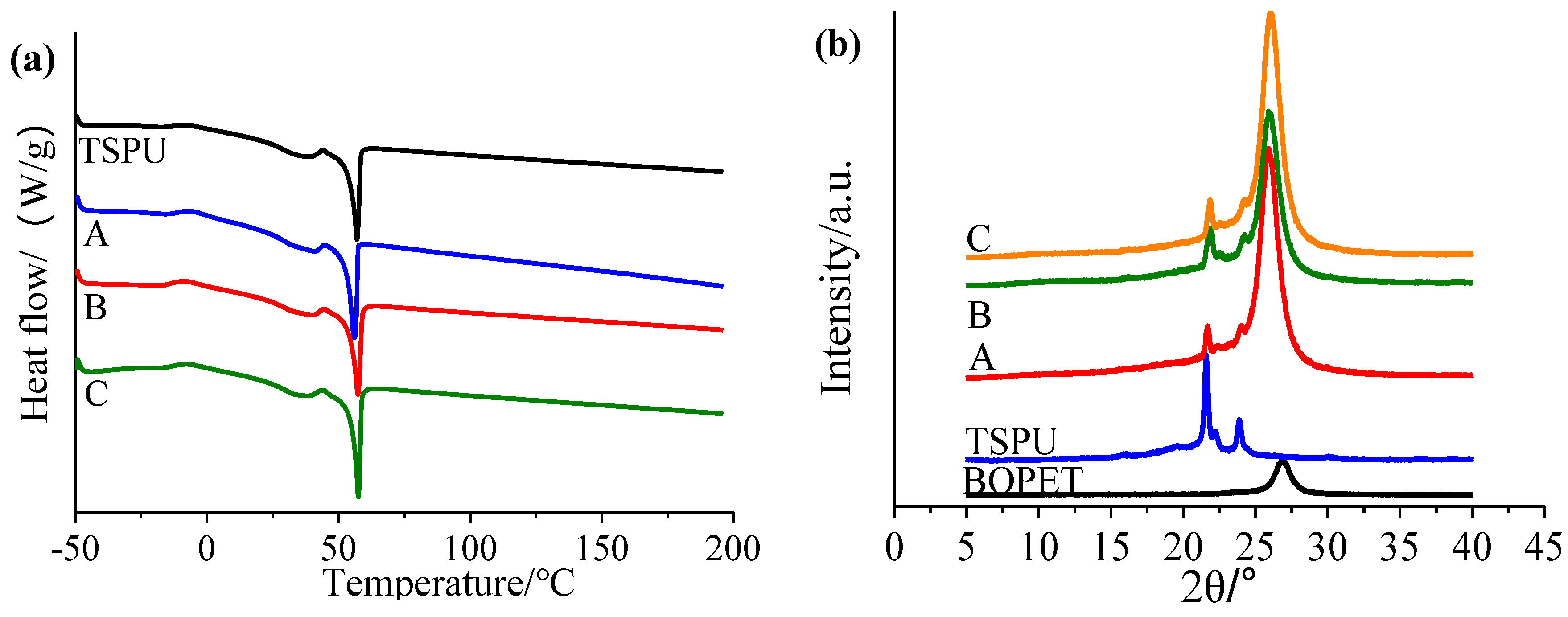
2.9. Differential Scanning Calorimetry (DSC)
2.10. X-ray Diffraction (XRD)
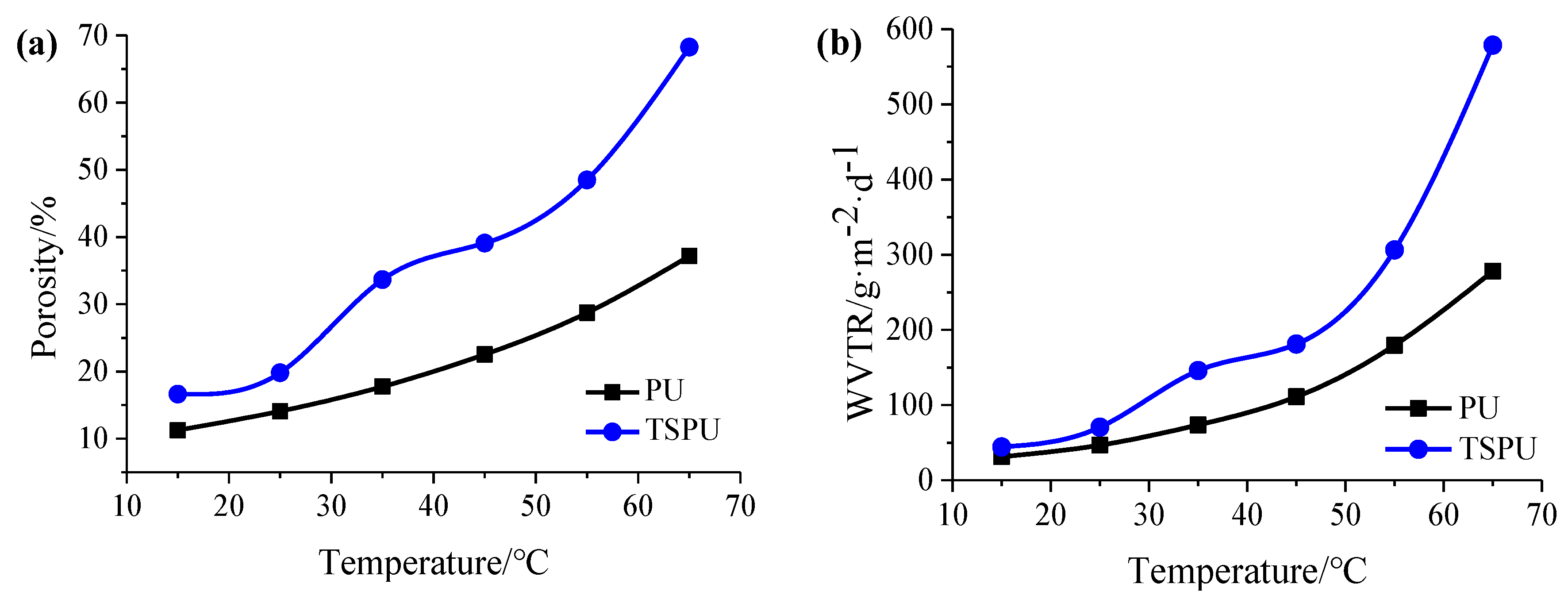
2.11. Porosity Characterization
2.12. Measure of Water Vapor Transmission Rate (WVTR)
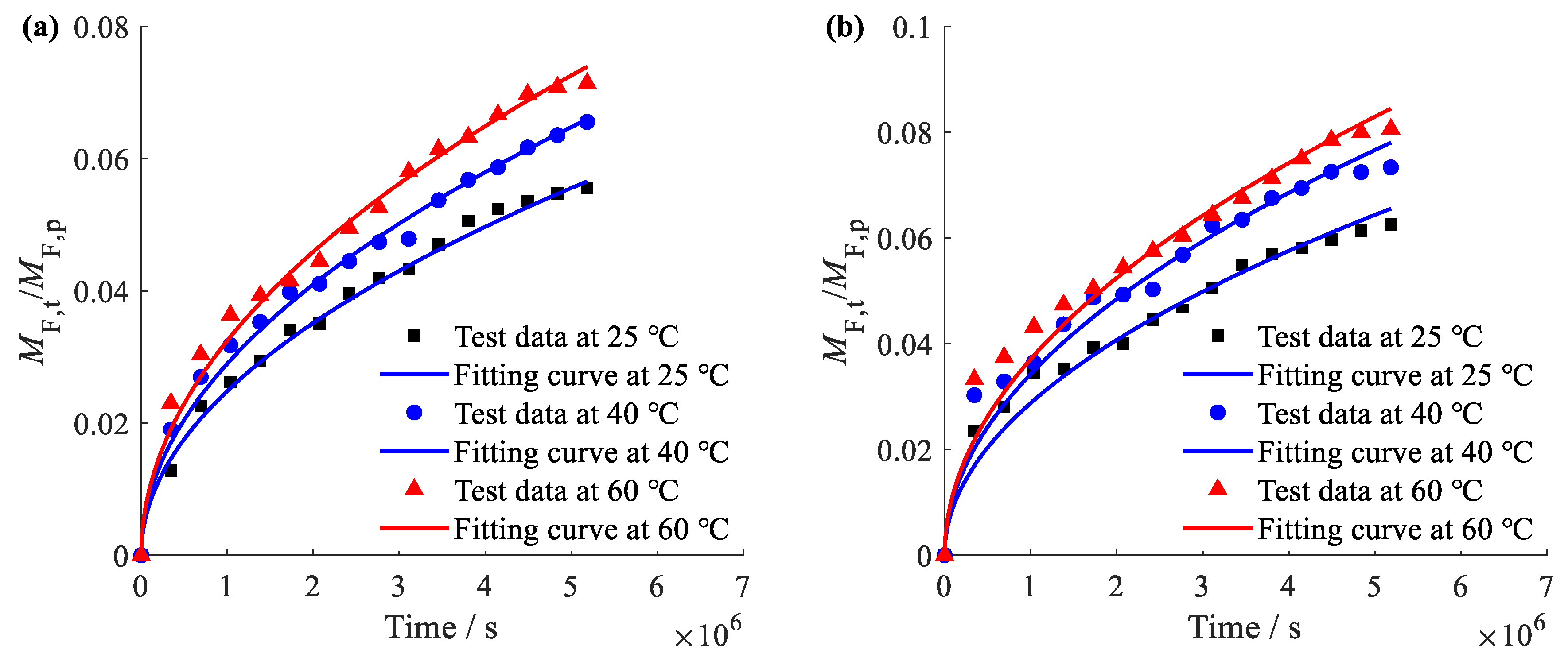
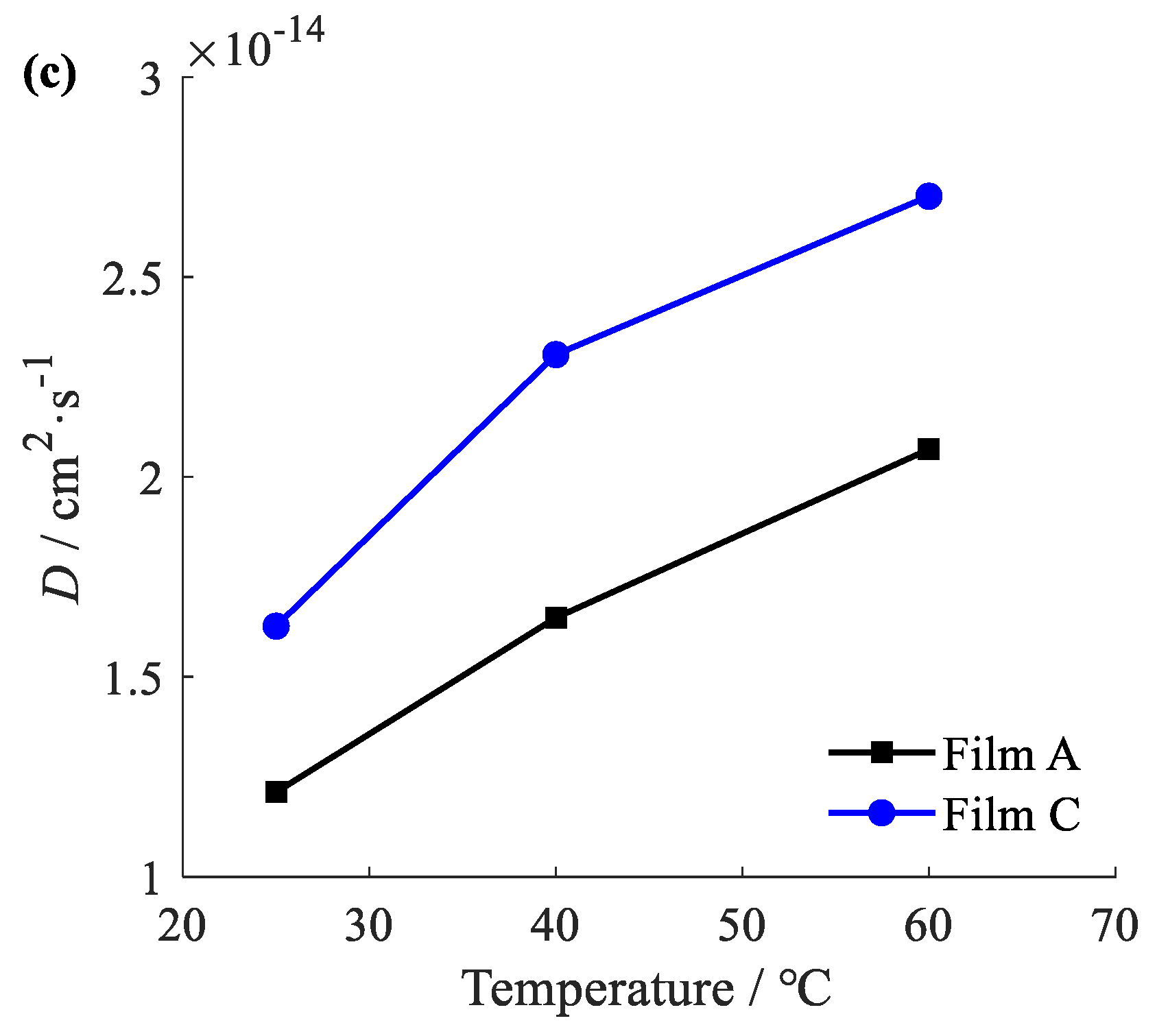
2.13. Release Characterization
2.14. Antioxidant Activity
2.15. Statistical Analysis
3. Results and Discussion
3.1. Characterization of Nanoliposomes Containing REOs
3.2. Double Switch Temperature Sensitive Characterization
3.3. Characterization of Controlled Release Complex Film
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Fang, Z.; Zhao, Y.; Warner, R.D.; Johnson, S.K. Active and intelligent packaging in meat industry. Trends Food Sci. Technol. 2017, 61, 60–71. [Google Scholar] [CrossRef]
- Gómez-Estaca, J.; Lopez-de-Dicastillo, C.; Hernández-Muñoz, P.; Catalá, R.; Gavara, R. Advances in antioxidant active food packaging. Trends Food Sci. Technol. 2014, 35, 42–51. [Google Scholar] [CrossRef]
- Chen, X.; Chen, M.; Xu, C.; Yam, K.L. Critical review of controlled release packaging to improve food safety and quality. Crit. Rev. Food Sci. Nutr. 2019, 59, 2386–2399. [Google Scholar] [CrossRef] [PubMed]
- Valencia, G.A.; Zare, E.N.; Makvandi, P.; Gutiérrez, T.J. Self-assembled carbohydrate polymers for food. Compr. Rev. Food. Sci. Food Saf. 2019. [Google Scholar] [CrossRef]
- Jiang, P.; Wu, Y.Q. Progress in the preparation and application of thermo- and pH-responsive polymer membranes. Sci. Technol. Rev. 2016, 34, 22–30. [Google Scholar]
- Singh, S.; Gaikwad, K.K.; Lee, M.; Lee, Y.S. Temperature sensitive smart packaging for monitoring the shelf life of fresh beef. J. Food Eng. 2018, 234, 41–49. [Google Scholar] [CrossRef]
- Werzer, O.; Tumphart, S.; Keimel, R.; Christian, P.; Coclite, A.M. Drug Release from Thin Films Encapsulated by a temperature-responsive Hydrogel. Soft Matter 2019, 15, 1853–1859. [Google Scholar] [CrossRef]
- Zhou, H.; Xun, R.; Wu, K.; Zhou, Z.; Yu, B.; Tang, Y.; Li, N. Polyurethane membrane with temperature- and pH-Controllable permeability for amino-acids. Macromol. Res. 2015, 23, 94–99. [Google Scholar] [CrossRef]
- Byung, K.K.; Sang, Y.L.; Mao, X. Polyurethanes having shape memory effects. Polymer 1996, 37, 5781–5793. [Google Scholar]
- Turan, D.; Gunes, G.; Seniha Güner, F. Synthesis, characterization and O2 permeability of shape memory polyurethane films for fresh produce packaging. Packag. Technol. Sci. 2016, 29, 415–427. [Google Scholar] [CrossRef]
- Dong, H.; He, J.P.; Xiao, K.J.; Li, C. Temperature-sensitive polyurethane (TSPU) film incorporated with carvacrol and cinnamyl aldehyde: Antimicrobial activity, sustained release kinetics and potential use as food packaging for Cantonese-style moon cake. Int. J. Food Sci. Technol. 2019. [Google Scholar] [CrossRef]
- Ribeiro-Santos, R.; de Melo, N.R.; Andrade, M.; Azevedo, G.; Machado, A.V.; Carvalho-Costa, D.; Sanches-Silva, A. Whey protein active films incorporated with a blend of essential oils: Characterization and effectiveness. Packag. Technol. Sci. 2017, 31, 27–40. [Google Scholar] [CrossRef]
- Richheimer, S.L.; Bernart, M.W.; King, G.A.; Kent, M.C.; Beiley, D.T. Antioxidant activity of lipid-soluble phenolic diterpenes from rosemary. J. Am. Oil Chem. Soc. 1996, 73, 507–514. [Google Scholar] [CrossRef]
- Guo, Y.; Shen, L.X.; Lu, Y.F.; Li, H.Y.; Min, K.; Li, L.F.; Yu, C.Y.; Zheng, X. Preparation of rutin-liposome drug delivery systems and evaluation on their in vitro antioxidant activity. Chin. Herb. Med. 2016, 8, 371–375. [Google Scholar] [CrossRef]
- Valencia-Sullca, C.; Jiménez, M.; Jiménez, A.; Atarés, L.; Vargas, M.; Chiralt, A. Influence of liposome encapsulated essential oils on properties of chitosan films. Polym. Int. 2016, 65, 979–987. [Google Scholar] [CrossRef]
- Cui, H.; Yuan, L.; Lin, L. Novel chitosan film embedded with liposome-encapsulated phage for biocontrol of, Escherichia coli, O157: H7 in beef. Carbohydr. Polym. 2017, 177, 156–164. [Google Scholar] [CrossRef]
- Boelter, J.F.; Brandelli, A. Innovative bionanocomposite films of edible proteins containing liposome-encapsulated nisin and halloysite nanoclay. Colloids Surf. B Biointerfaces 2016, 145, 740–747. [Google Scholar] [CrossRef]
- Khatibi, S.A.; Misaghi, A.; Moosavy, M.H.; Basti, A.A.; Koohi, M.K.; Khosravi, P.; Haghirosadat, F. Encapsulation of Zataria multiflora bioss. Essential oil into nanoliposomes and in vitro antibacterial activity against escherichia coli O157: H7. J. Food Process. Preserv. 2016, 41, e12955. [Google Scholar] [CrossRef]
- Zhou, H.; Zeng, J.; Fan, H.; Liu, Y.; Zhou, J. Thermal sensitive polyurethane membranes with desirable switch temperatures. Macromol. Res. 2010, 18, 1053–1059. [Google Scholar] [CrossRef]
- Cui, H.; Yuan, L.; Li, W.; Lin, L. Antioxidant property of SiO2-eugenol liposome loaded nanofibrous membranes on beef. Food Packag. Shelf Life 2017, 11, 49–57. [Google Scholar] [CrossRef]
- Palacio, L.; Pradanos, P.; Calvo, J.I.; Hernandez, A. Porosity measurements by a gas penetration method and other techniques applied to membrane characterization. Thin Solid Membr. 1999, 348, 22–29. [Google Scholar] [CrossRef]
- Chen, Y.; Liu, Y.; Fan, H.; Li, H.; Shi, B.; Zhou, H.; Peng, B. The polyurethane membranes with temperature sensitivity for water vapor permeation. J. Membr. Sci. 2007, 287, 192–197. [Google Scholar] [CrossRef]
- Chen, X.; Lu, L.X.; Qiu, X.; Tang, Y. Controlled release mechanism of complex bio-polymeric emulsifiers made microspheres embedded in sodium alginate based films. Food Control 2017, 73, 1275–1284. [Google Scholar] [CrossRef]
- Sun, L.N.; Lu, L.X.; Qiu, X.L.; Tang, Y.L. Development of low-density polyethylene antioxidant active films containing α-tocopherol loaded with MCM-41(Mobil Composition of Matter No. 41) mesoporous silica. Food Control 2017, 71, 193–199. [Google Scholar] [CrossRef]
- Sarabandi, K.; Jafari, S.M.; Mohammadi, M.; Akbarbaglu, Z.; Pezeshki, A.; Heshmati, M.K. Production of reconstitutable nanoliposomes loaded with flaxseed protein hydrolysates Stability and characterization. Food Hydrocoll. 2019, 96, 442–450. [Google Scholar] [CrossRef]
- Akhter, R.; Masoodi, F.A.; Wani, T.A.; Rather, S.A. Functional characterization of biopolymer based composite film: Incorporation of natural essential oils and antimicrobial agents. International Int. J. Biol. Macromol. 2019, 137, 1245–1255. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Zhao, P.; Liang, X.; Gong, X.; Song, T.; Niu, R.; Chang, J. Folate-PEG coated cationic modified chitosan—Cholesterol liposomes for tumor-targeted drug delivery. Biomaterials 2010, 31, 4129–4138. [Google Scholar] [CrossRef]
- Pan, L.; Zhang, S.; Gu, K.; Zhang, N. Preparation of astaxanthin-loaded liposomes: Characterization, storage stability and antioxidant activity. CyTA J. Food 2018, 16, 607–618. [Google Scholar] [CrossRef]
- Zhou, H.; Yu, B.; Zhou, J.; Zeng, J.; Zhou, Z.; Liu, G. Synthesis and characterization of thermal-and pH-sensitive polyurethane hydrogels. Polym. Sci. Eng. 2017, 33, 11–16. [Google Scholar]
- Zhou, H.; Shi, H.; Fan, H.; Zhou, J.; Yuan, J. Thermosensitive polyurethane membrane with controllable water vapor permeation for food packaging. Macromol. Res. 2009, 17, 528–532. [Google Scholar] [CrossRef]
- Cinelli, P.; Schmid, M.; Bugnicourt, E.; Coltelli, M.; Lazzeri, A. Recyclability of PET/WPI/PE multilayer films by removal of whey protein isolate-based coatings with enzymatic detergents. Materials 2016, 9, 473. [Google Scholar] [CrossRef] [PubMed]


| Time/d | MD/nm | (s. d.) | PDI | (s. d.) | Zeta Potential/mV | (s. d.) | EE/% | (s. d.) | DPPHs/% | (s. d.) |
|---|---|---|---|---|---|---|---|---|---|---|
| 0 | 60.75 a | 1.079 | 0.230 a | 0.0074 | −31.83 a | 0.7767 | 67.34 a | 1.649 | 61.22 a | 1.427 |
| 10 | 61.15 a | 2.734 | 0.231 a | 0.0099 | −31.76 a | 0.6737 | 67.21 a | 4.695 | 60.57 a | 1.484 |
| 20 | 61.23 a | 2.314 | 0.231 a | 0.0069 | −31.32 a | 0.7263 | 66.70 ab | 2.574 | 59.80 a | 0.5814 |
| 40 | 62.39 a | 2.658 | 0.233 a | 0.0083 | −30.56 a | 0.1227 | 66.52 ab | 0.4148 | 58.93 a | 0.2998 |
| 60 | 64.57 a | 1.22 | 0.236 a | 0.0018 | −30.02 a | 0.3998 | 64.92 b | 0.4344 | 55.77 b | 0.3976 |
| Film Samples | D/×10−14 (cm2·s−1) | |||||
|---|---|---|---|---|---|---|
| 25 °C | RMSE | 40 °C | RMSE | 60 °C | RMSE | |
| A | 1.212 a | 0.001220 | 1.648 a | 0.001509 | 2.069 a | 0.001967 |
| C | 1.627 a | 0.002744 | 2.306 a | 0.003596 | 2.702 a | 0.004054 |
| Film Samples | Absorbance | DPPHs/% | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 0.5 h | (s. d.) | 24 h | (s. d.) | 720 h | (s. d.) | 1440 h | (s. d.) | 0.5 h | (s. d.) | 24 h | (s. d.) | 720 h | (s. d.) | 1440 h | (s. d.) | |
| Control | 0.918 a | 0.0223 | 0.918 a | 0.0026 | 0.917 a | 0.0066 | 0.917 a | 0.0194 | 0.000 a | 0.0000 | 0.000 a | 0.0000 | 0.000 a | 0.0000 | 0.000 a | 0.0000 |
| A | 0.585 b | 0.0142 | 0.525 b | 0.0017 | 0.481 b | 0.0067 | 0.435 b | 0.0085 | 36.27 b | 0.8533 | 42.81 b | 0.2892 | 47.55 b | 1.663 | 52.56 b | 0.1818 |
| B | 0.904 a | 0.0019 | 0.906 a | 0.009 | 0.902 a | 0.0246 | 0.901 a | 0.0637 | 1.530 a | 0.0055 | 1.300 c | 0.0015 | 1.640 a | 0.0055 | 1.740 c | 0.0122 |
| C | 0.512 c | 0.0178 | 0.463 c | 0.0328 | 0.472 b | 0.0028 | 0.526 b | 0.0251 | 44.23 c | 1.019 | 49.57 d | 0.6615 | 48.53 b | 1.01 | 42.64 d | 0.2782 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, X.; Long, Q.; Zhu, L.; Lu, L.-X.; Sun, L.-N.; Pan, L.; Lu, L.-J.; Yao, W.-R. A Double-Switch Temperature-Sensitive Controlled Release Antioxidant Film Embedded with Lyophilized Nanoliposomes Encapsulating Rosemary Essential Oils for Solid Food. Materials 2019, 12, 4011. https://doi.org/10.3390/ma12234011
Chen X, Long Q, Zhu L, Lu L-X, Sun L-N, Pan L, Lu L-J, Yao W-R. A Double-Switch Temperature-Sensitive Controlled Release Antioxidant Film Embedded with Lyophilized Nanoliposomes Encapsulating Rosemary Essential Oils for Solid Food. Materials. 2019; 12(23):4011. https://doi.org/10.3390/ma12234011
Chicago/Turabian StyleChen, Xi, Qing Long, Lei Zhu, Li-Xin Lu, Li-Nan Sun, Liao Pan, Li-Jing Lu, and Wei-Rong Yao. 2019. "A Double-Switch Temperature-Sensitive Controlled Release Antioxidant Film Embedded with Lyophilized Nanoliposomes Encapsulating Rosemary Essential Oils for Solid Food" Materials 12, no. 23: 4011. https://doi.org/10.3390/ma12234011
APA StyleChen, X., Long, Q., Zhu, L., Lu, L.-X., Sun, L.-N., Pan, L., Lu, L.-J., & Yao, W.-R. (2019). A Double-Switch Temperature-Sensitive Controlled Release Antioxidant Film Embedded with Lyophilized Nanoliposomes Encapsulating Rosemary Essential Oils for Solid Food. Materials, 12(23), 4011. https://doi.org/10.3390/ma12234011





