Effect of Silica Fume and Fly Ash Admixtures on the Corrosion Behavior of AISI 304 Embedded in Concrete Exposed in 3.5% NaCl Solution
Abstract
:1. Introduction
2. Materials and Methods
2.1. Characterization of Fresh and Hardened Concrete
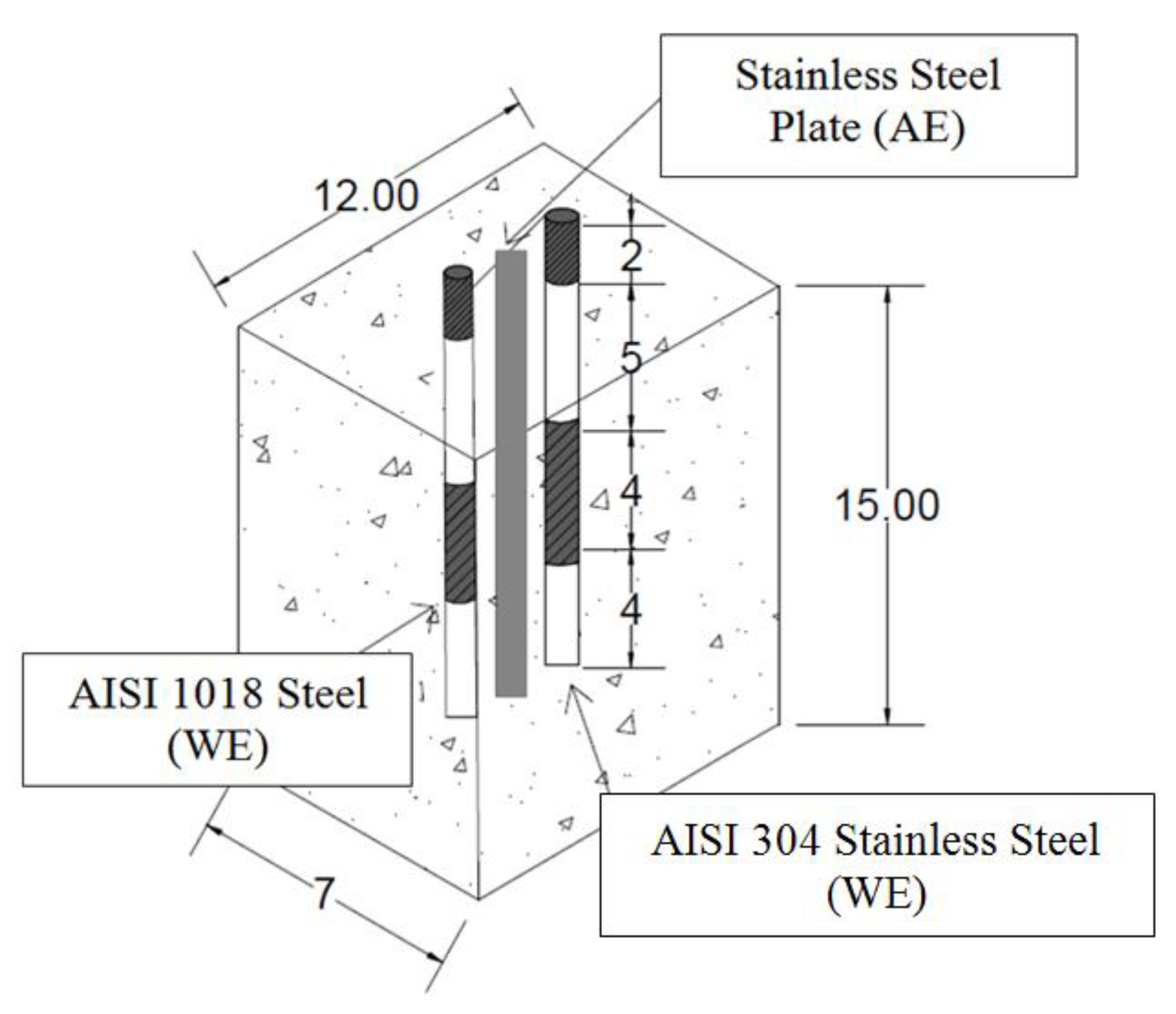
2.2. Concrete Specimens
- 4A indicates the concrete mix with 100% CPC;
- 4B indicates the concrete mix with 80% CPC + 20% SF;
- 4C indicates the concrete mix with 80% CPC + 20% FA;
- N indicates rebars of AISI 1018 carbon steel;
- I indicates rebars of AISI 304 stainless steel.
2.3. Electrochemical Techniques
3. Results and Discussion
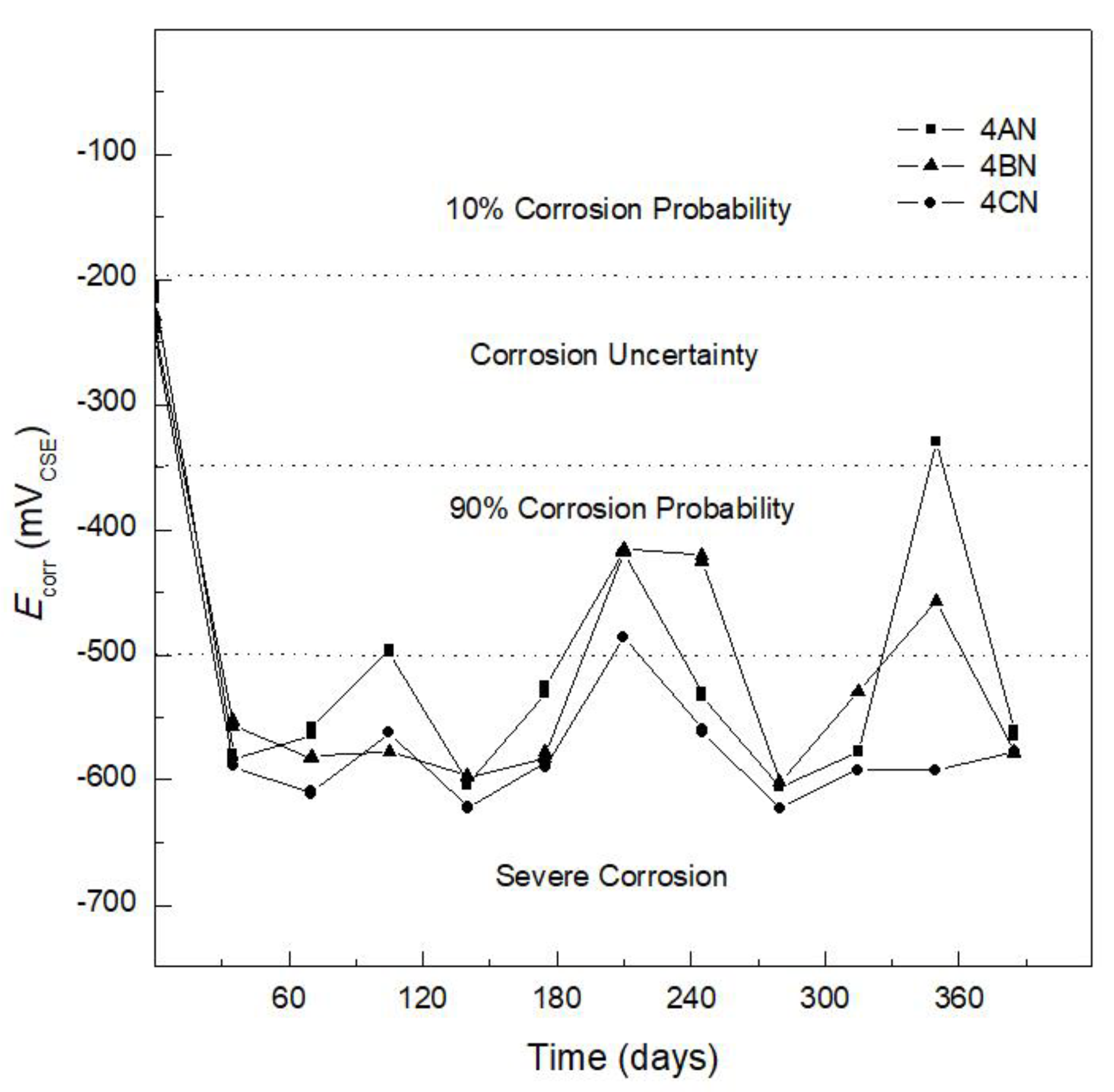
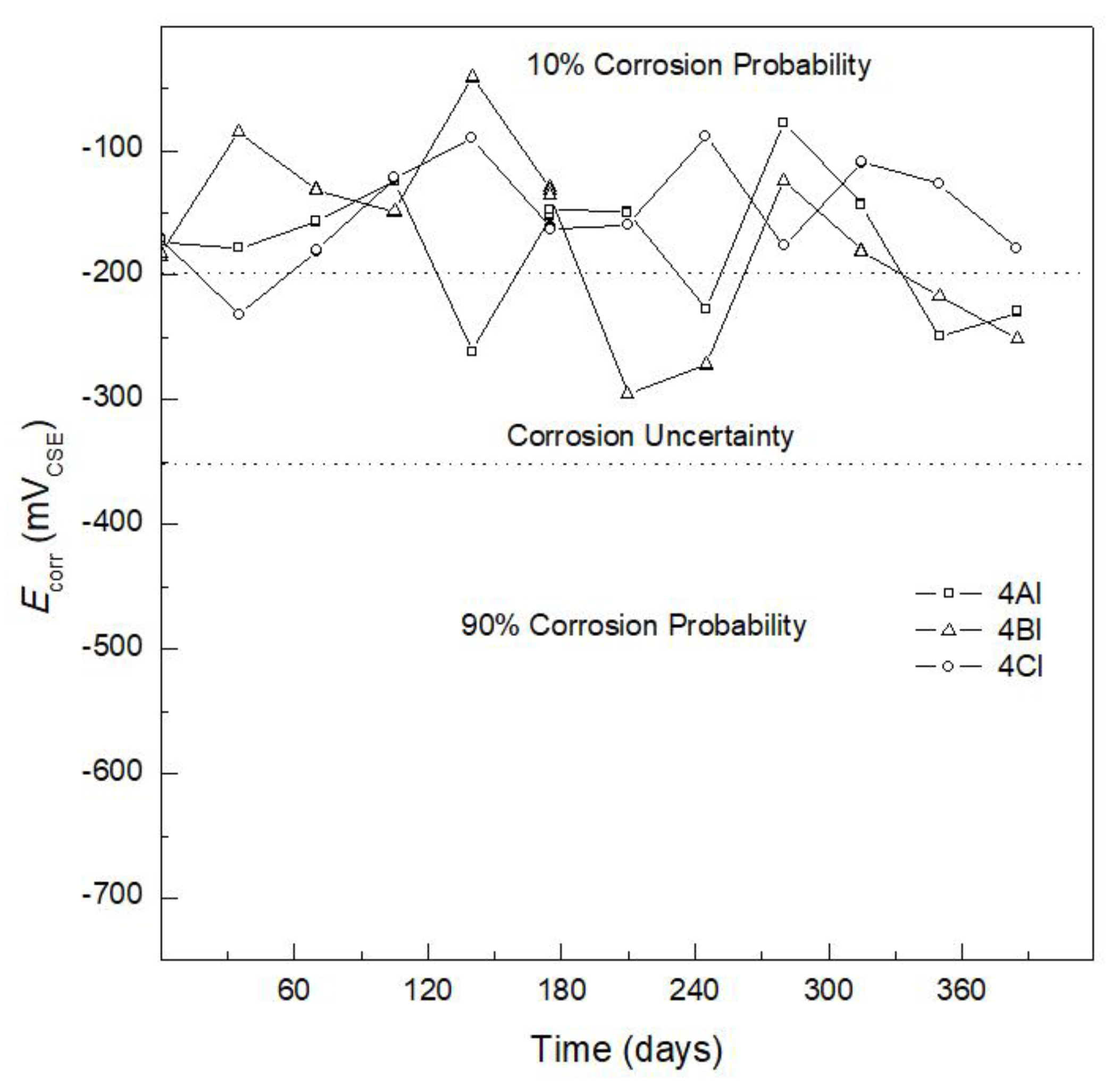
3.1. Corrosion Potential
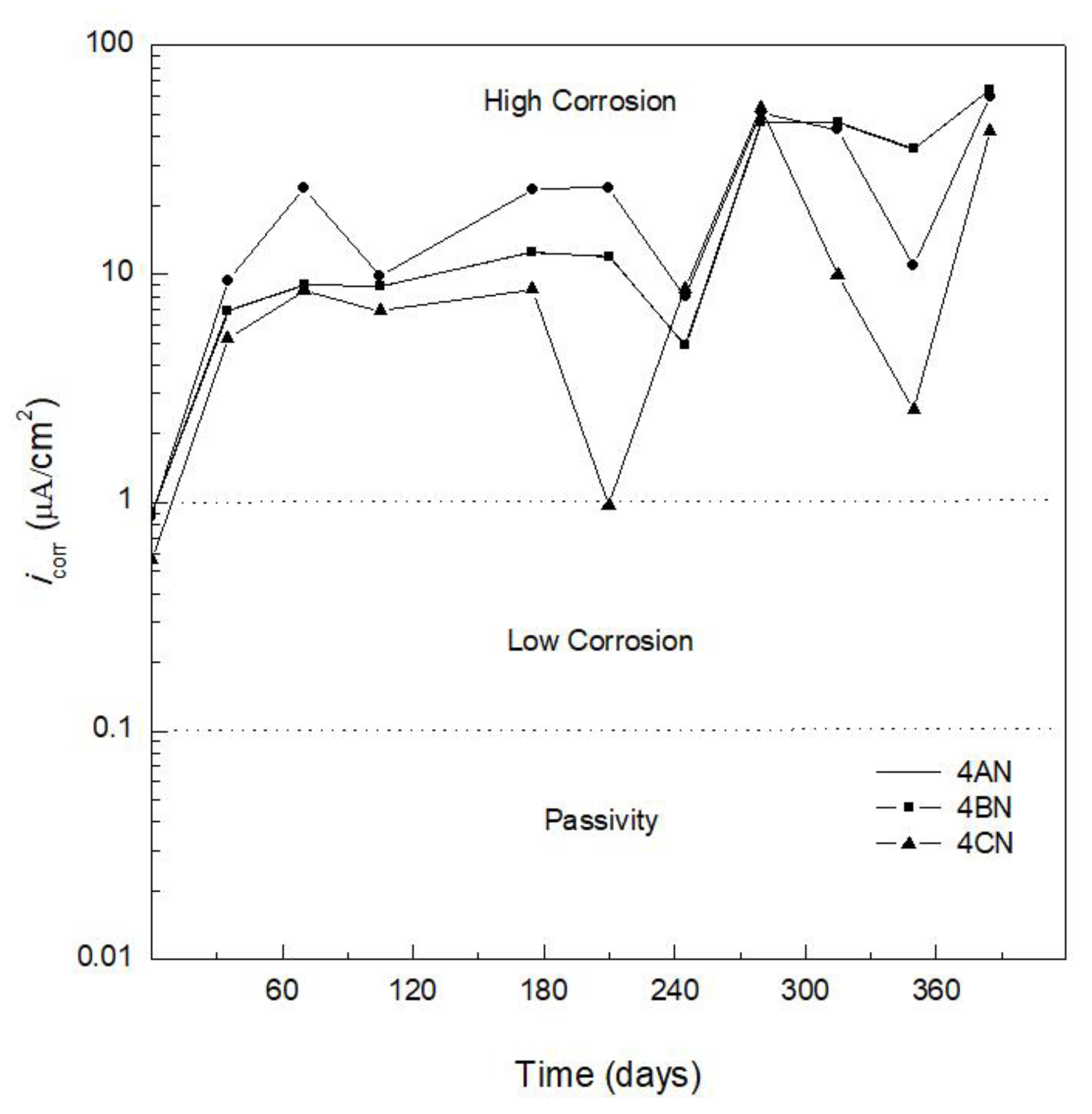
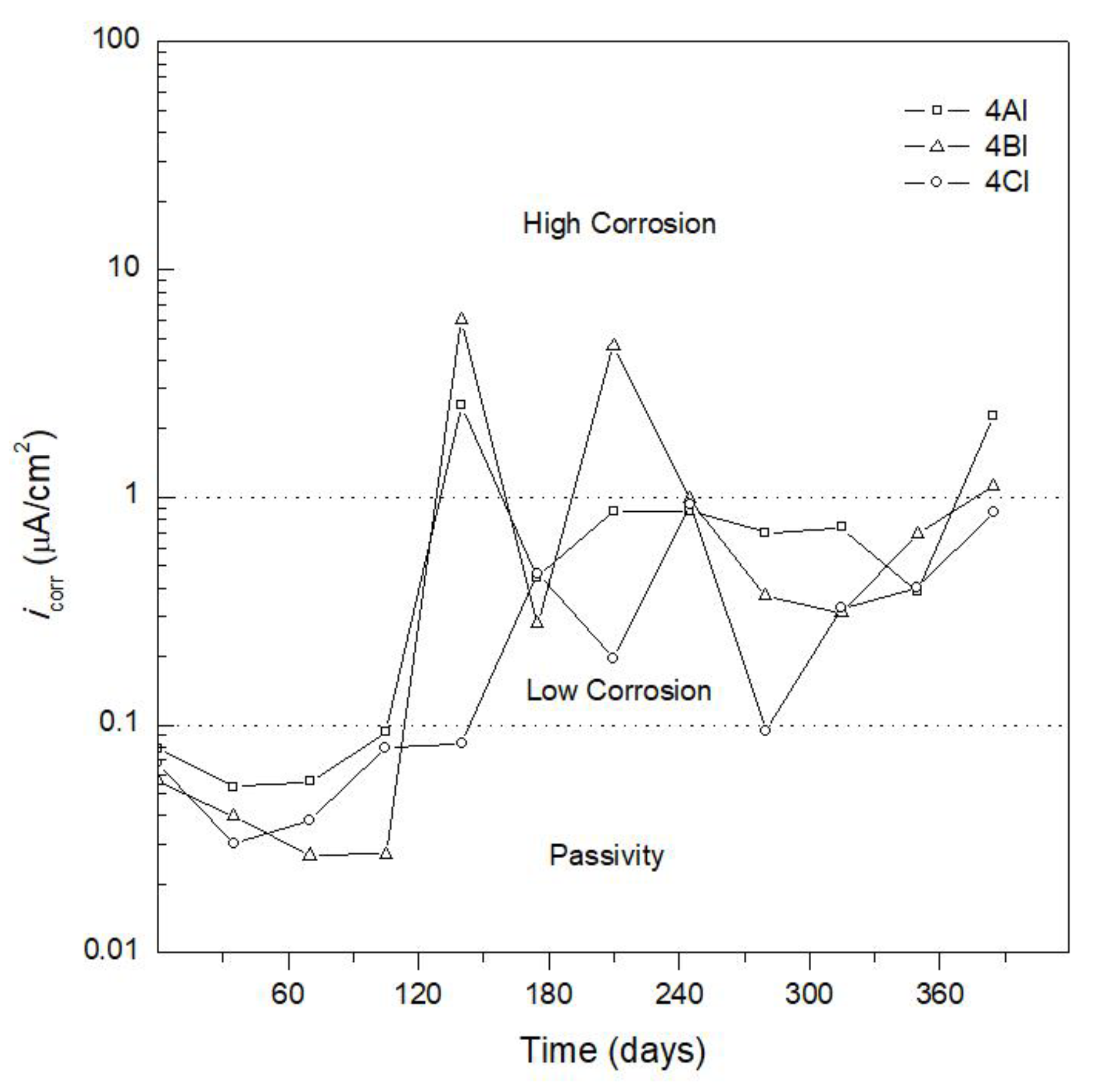
3.2. Corrosion Kinetics
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Campos Silva, H.G.; Garces Terradillos, P.; Zornoza, E.; Mendoza-Rangel, J.M.; Castro-Borges, P.; Juarez Alvarado, C.A. Improving Sustainability through Corrosion Resistance of Reinforced Concrete by Using a Manufactured Blended Cement and Fly Ash. Sustainability 2018, 10, 2004. [Google Scholar] [CrossRef] [Green Version]
- Melchers, R.E.; Li, C.Q. Reinforcement corrosion initiation and activation times in concrete structures exposed to severe marine environments. Cem. Concr. Res. 2009, 39, 1068–1076. [Google Scholar] [CrossRef]
- Baltazar, M.A.; Santiago, G.; Moreno, V.M.; Croche, R.; De la Garza, M.; Estupiñan, F.; Zambrano, P.; Gaona, G. Electrochemical behaviour of galvanized steel embedded in concrete exposed to sand contaminated with NaCl. Int. J. Electrochem. Sci. 2016, 11, 10306–10319. [Google Scholar] [CrossRef]
- Pradhan, B. Corrosion behavior of steel reinforcement in concrete exposed to composite chloride–Sulfate environment. Constr. Build. Mater. 2014, 72, 398–410. [Google Scholar] [CrossRef]
- Santiago, G.; Baltazar, M.A.; Galván, R.; López, L.; Zapata, F.; Zambrano, P.; Gaona, C.; Almeraya, F. Electrochemical Evaluation of Reinforcement Concrete Exposed to Soil Type SP Contaminated with Sulphates. Int. J. Electrochem. Sci. 2016, 11, 4850–4864. [Google Scholar] [CrossRef]
- Troconis de Rincón, O.; Montenegro, J.C.; Vera, R.; Carvajal, A.M.; De Gutiérrez, R.M.; Del Vasto, S.; Saborio, E.; Torres-Acosta, A.; Pérez-Quiroz, J.; Martínez-Madrid, M.; et al. Reinforced Concrete Durability in Marine Environments DURACON Project: Long-Term Exposure. Corrosion 2016, 72, 824–833. [Google Scholar] [CrossRef]
- Caré, S.; Raharinaivo, A. Influence of impressed current on the initiation of damage in reinforced mortar due to corrosion of embedded Steel. Cem. Concr. Res. 2007, 37, 1598–1612. [Google Scholar] [CrossRef]
- Ann, K.Y.; Song, H.W. Chloride threshold level for corrosion of steel in concrete. Corros. Sci. 2007, 49, 4113–4133. [Google Scholar] [CrossRef]
- Medina, E.; Medina, J.M.; Cobo, A.; Bastidas, D.M. Evaluation of mechanical and structural behavior of austenitic duplex stainless steel reinforcements. Constr. Build. Mater. 2015, 78, 1–7. [Google Scholar] [CrossRef]
- Yunovich, M.; Thompson, N.G. Corrosion of Highway Bridges: Economic Impact and Control Methodologies. Concr. Int. 2003, 25, 52–57. [Google Scholar]
- Criado, M.; Fernández-Jiménez, A.; Palomo, A. Alkali activation of fly ash: Effect of SiO2/Na2O ratio Part I: FTIR study. Microporous Mesoporous Mat. 2007, 106, 180–191. [Google Scholar] [CrossRef]
- Santiago-Hurtado, G.; Baltazar-Zamora, M.A.; Olguín-Coca, J.; López, L.D.; Galván-Martínez, R.; Ríos-Juárez, A.; Gaona-Tiburcio, C.; Almeraya-Calderón, F. Electrochemical Evaluation of a Stainless Steel as Reinforcement in Sustainable Concrete Exposed to Chlorides. Int. J. Electrochem. Sci. 2016, 11, 2994–3006. [Google Scholar] [CrossRef]
- Choi, Y.S.; Kim, J.G.; Lee, K.M. Corrosion behavior of steel bar embedded in fly ash concrete. Corros. Sci. 2006, 48, 1733–1745. [Google Scholar] [CrossRef]
- Amorin, R.; Opoku, P.; Osei, E. Evaluation of blended lime-stabilised spent synthetic-based drilling mud and cement for oil well cementing operations. Adv. Geo Energy Res. 2019, 3, 141–148. [Google Scholar] [CrossRef]
- Larki, O.; Norouzi Apourvari, S.; Schaffie, M.; Farazmand, R.A. New formulation for lightweight oil well cement slurry using a natural pozzolan. Adv. Geo Energy Res. 2019, 3, 242–249. [Google Scholar] [CrossRef]
- Broni-Bediako, E.; Amorin, R. Experimental study on the effects of cement contamination in a water based mud. Adv. Geo Energy Res. 2019, 3, 314–319. [Google Scholar] [CrossRef]
- Cordeiro, G.C.; Toledo-Filho, R.D.; Tavares, L.M.; Fairbairn, E.M.R. Pozzolanic activity and filler effect of sugar cane bagasse ash in Portland cement and lime mortars. Cem. Concr. Compos. 2008, 30, 410–418. [Google Scholar] [CrossRef]
- Cordeiro, G.C.; Toledo-Filho, R.D.; Tavares, L.M.; Fairbairn, E.M.R. Ultrafine grinding of sugar cane bagasse ash for application as pozzolanic admixture in concrete. Cem. Concr. Res. 2009, 39, 110–115. [Google Scholar] [CrossRef]
- Landa-Gómez, A.E.; Croche, R.; Márquez-Montero, S.; Villegas Apaez, R.; Ariza-Figueroa, H.A.; Estupiñan López, F.; Gaona Tiburcio, G.; Almeraya Calderón, F.; Baltazar-Zamora, M.A. Corrosion Behavior 304 and 316 Stainless Steel as Reinforcement in Sustainable Concrete Based on Sugar Cane Bagasse Ash Exposed to Na2SO4. ECS Trans. 2018, 84, 179–188. [Google Scholar] [CrossRef]
- Muralidharan, S.; Parande, A.K.; Saraswathy, V.; Kumar, K.; Palaniswamy, N. Corrosion of steel in concrete with and without silica fume. Zaštita Mater. 2008, 49, 3–8. [Google Scholar]
- Cao, H.T.; Sirivivatnanon, V. Corrosion of steel in concrete with and without silica fume. Cem. Concr. Res. 1991, 36, 1922–1930. [Google Scholar] [CrossRef]
- Malhotra, V.M.; Ramezanianpour, A.A. Fly Ash in Concrete, 2nd ed.; Natural Resources CANMET: Hamilton, ON, Canada, 1994. [Google Scholar]
- Franco-Luján, V.A.; Maldonado-García, M.A.; Mendoza-Rangel, J.M.; Montes-García, P. Chloride-induced reinforcing steel corrosion in ternary concretes containing fly ash and untreated sugarcane bagasse ash. Constr. Build. Mater. 2019, 198, 608–618. [Google Scholar] [CrossRef]
- NMX-C-414-ONNCCE-2014—Industria de la Construcción—Cementantes Hidráulicos—Especificaciones y Métodos de Ensayo; ONNCCE: Ciudad de México, Mexico, 2014.
- ACI 211.1-91 Standard. Standard Practice for Selecting Proportions for Normal, Heavyweight, and Mass Concrete; ACI: Farmington Hills, MI, USA, 2002. [Google Scholar]
- NMX-C-083-ONNCCE-2014—Industria de la Construcción—Concreto—Determinación de la Resistencia a la Compresión de Especímenes—Método de Ensayo; ONNCCE: Ciudad de México, Mexico, 2014.
- ASTM C127-15—Standard Test Method for Relative Density (Specific Gravity) and Absorption of Coarse Aggregate; ASTM International: West Conshohocken, PA, USA, 2015.
- ASTM C128-15—Standard Test Method for Relative Density (Specific Gravity) and Absorption of Fine Aggregate; ASTM International: West Conshohocken, PA, USA, 2015.
- ASTM C29/C29M-07—Standard Test Method for Bulk Density (“Unit Weight”) and Voids in Aggregate; ASTM International: West Conshohocken, PA, USA, 2007.
- ASTM C33/C33M-16e1—Standard Specification for Concrete Aggregates; ASTM International: West Conshohocken, PA, USA, 2016.
- ASTM C 1064/C1064M-08—Standard Test Method for Temperature of Freshly Mixed Hydraulic-Cement Concrete; ASTM International: West Conshohocken, PA, USA, 2008.
- NMX-C-156-ONNCCE-2010—Determinación de Revenimiento en Concreto Fresco; ONNCCE: Ciudad de México, Mexico, 2010.
- NMX-C-162-ONNCCE-2014—Industria de la Construcción—Concreto Hidráulico—Determinación de la Masa Unitaria, Cálculo del Rendimiento y Contenido de Aire del Concreto Fresco por el Método Gravimétrico; ONNCCE: Ciudad de México, Mexico, 2014.
- NMX-C-159-ONNCCE-2004, Industria de la Construcción—Concreto—Elaboración y Curado de Especímenes en el Laboratorio; ONNCCE: Ciudad de México, Mexico, 2004.
- ASTM G 59-97 (2014)—Standard Test Method for Conducting Potentiodynamic Polarization Resistance Measurements; ASTM International: West Conshohocken, PA, USA, 2014.
- ASTM C 876-15 (2015)—Standard Test Method for Corrosion Potentials of Uncoated Reinforcing steel in Concrete; ASTM International: West Conshohocken, PA, USA, 2015.
- Song, H.W.; Saraswathy, V. Corrosion Monitoring of Reinforced Concrete Structures—A Review. Int. J. Electrochem. Sci. 2007, 2, 1–28. [Google Scholar]
- Nuñez, R.; Buelna, J.; Barrios, C.; Gaona, C.; Almeraya, F. Corrosion of Modified Concrete with Sugar Cane Bagasse Ash. Int. J. Corrs. 2012, 12, 1–5. [Google Scholar]
- Baltazar, M.A.; Maldonado, M.; Tello, M.; Santiago, G.; Coca, F.; Cedano, A.; Barrios, C.P.; Nuñez, R.; Zambrano, P.; Gaona, C.; et al. Efficiency of Galvanized Steel Embedded in Concrete Previously Contaminated with 2, 3 and 4% of NaCl. Int. J. Electrochem. Sci. 2012, 7, 2997–3007. [Google Scholar]
- Baltazar, M.A.; Santiago, G.; Gaona, C.; Maldonado, M.; Barrios, C.P.; Nunez, R.; Perez, T.; Zambrano, P.; Almeraya, F. Evaluation of the corrosion at early age in reinforced concrete exposed to sulfates. Int. J. Electrochem. Sci. 2012, 7, 588–600. [Google Scholar]
- Feliu, S.; González, J.A.; Andrade, C. Electrochemical methods for on-site determinations of corrosion rates of rebars. In Techniques to Assess the Corrosion Activity of Steel Reinforced Concrete Structures; ASTM STP 1276; Berke, N.S., Escalante, E., Nmai, C.K., Whiting, D., Eds.; ASTM International: West Conshohocken, PA, USA, 1996. [Google Scholar]
- Montemor, M.F.; Simoes, A.M.P.; Salta, M.M. Effect of fly ash on concrete reinforcement corrosion studied by EIS. Cem. Concr. Compos. 2000, 22, 175–185. [Google Scholar] [CrossRef]
- Fajardo, S.; Bastidas, D.M.; Ryan, M.P.; Criado, M.; McPhail, D.S.; Morris, R.J.H.; Bastidas, J.M. Low energy SIMS characterization of passive oxide films formed on a low-nickel stainless steel in alkaline media. Appl. Surf. Sci. 2014, 288, 423–429. [Google Scholar] [CrossRef] [Green Version]
- Fajardo, S.; Bastidas, D.M.; Ryan, M.P.; Criado, M.; McPhail, D.S.; Bastidas, J.M. Low-nickel stainless steel passive film in simulated concrete pore solution: A SIMS study. Appl. Surf. Sci. 2010, 256, 6139–6143. [Google Scholar] [CrossRef]
- Monticelli, C.; Criado, M.; Fajardo, S.; Bastidas, J.M.; Abbottoni, M.; Balbo, A. Corrosion behavior of low Ni austenitic stainless steel in carbonated chloride-polluted alkali-activated fly ash mortar. Cem. Concr. Res. 2014, 55, 49–58. [Google Scholar] [CrossRef]
- Fajardo, S.; Bastidas, D.M.; Criado, M.; Bastidas, J.M. Electrochemical study on the corrosion behavior of a new low-nickel stainless steel in carbonated alkaline solution in the presence of chlorides. Electrochim. Acta 2014, 129, 160–170. [Google Scholar] [CrossRef] [Green Version]
- Criado, M.; Bastidas, D.M.; Fajardo, S.; Fernández-Jiménez, A.; Bastidas, J.M. Corrosion behaviour of a new low-nickel stainless steel embedded in activated fly ash mortars. Cem. Concr. Compos. 2011, 33, 644–652. [Google Scholar] [CrossRef]
- Crouch, L.K.; Hewitt, R.; Byard, B. High Volume Fly Ash Concrete. In Proceedings of the 2007 World of Coal Ash (WOCA), Covington, KY, USA, 7–10 May 2007; pp. 1–14. [Google Scholar]
- Amudhavalli, N.K.; Mathew, J. Effect of silica fume on strength and durability parameters of concrete. Int. J. Eng. Sci. Emerg. Technol. 2012, 3, 28–35. [Google Scholar]
- Baltazar, M.A.; Mendoza, J.M.; Croche, R.; Gaona, C.; Hernández, C.; López, L.; Olguín, F.; Almeraya, F. Corrosion Behavior of Galvanized Steel embedded in concrete exposed to soil type MH contaminated with chlorides. Front. Mater. 2019, 6, 257. [Google Scholar] [CrossRef]
- Bautista, A.; Blanco, G.; Velasco, F. Corrosion behavior of low-nickel austenitic stainless steels reinforcements: A comparative study in simulated pore solutions. Cem. Concr. Res. 2006, 36, 1922–1930. [Google Scholar] [CrossRef]
- Knudsen, A.; Jensen, F.M.; Klinghoffer, O.; Skovsgaard, T. Cost-effective enhancement of durability of concrete structures by intelligent use of stainless steel reinforcement. In Proceedings of the Conference of Corrosion and Rehabilitation of Reinforced Concrete Structures, Orlando, FL, USA, 7–11 December 1998. [Google Scholar]
- Medina, E. Evaluation of Mechanical, Structural and Corrosion Behavior of a New Duplex Low-Nickel Stainless Steel Reinforcement. Ph.D. Thesis, Polytechnic University of Madrid, Madrid, Spain, 2012; pp. 167–180. [Google Scholar]
| Aggregates | Relative Density (Specific Gravity) | Bulk Density (Unit Weight) (kg/m3) | Absorption (%) | Fineness Modulus | Maximum Aggregate Size (mm) |
|---|---|---|---|---|---|
| Coarse (gravel) | 2.32 | 1391 | 5.45 | - - - | 19 |
| Fine (sand) | 2.66 | 1237 | 1.97 | 2.62 | - - - |
| Materials | CPC 30R, kg (4AN, 4AI) | Silica Fume (SF), kg (4BN, 4BI) | Fly Ash (FA), kg (4CN, 4CI) |
|---|---|---|---|
| Cement | 410 | 328 | 328 |
| Partial substitute | 0 | 82 | 82 |
| Water | 205 | 205 | 205 |
| Coarse aggregate | 890 | 890 | 890 |
| Fine aggregate | 838 | 838 | 838 |
| Test | CPC 30R (4AN, 4AI) | Silica Fume (SF) (4BN, 4BI) | Fly Ash (FA) (4CN, 4CI) |
|---|---|---|---|
| Temperature, °C | 24.0 | 21.7 | 22.4 |
| Slump, cm | 4 | 3 | 3 |
| Density, kg/m3 | 2150 | 2188 | 2173 |
| Compressive strength (F’c), MPa (28 days) | 35.9 | 37.1 | 36.6 |
| Steel | Mass, % | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| C | Si | Mn | P | S | Cr | Ni | Mo | Cu | Fe | |
| AISI 1018 | 0.20 | 0.22 | 0.72 | 0.02 | 0.02 | 0.13 | 0.06 | 0.02 | 0.18 | Balance |
| AISI 304 | 0.04 | 0.32 | 1.75 | 0.03 | 0.001 | 18.20 | 8.13 | 0.22 | 0.21 | Balance |
| Corrosion Potential, Ecorr (mVCSE) | |
|---|---|
| >−200 | 10% probability of corrosion |
| −350 < Ecorr < −200 | Uncertainty corrosion |
| −350 < Ecorr < −500 | 90% probability of corrosion |
| <−500 | Severe corrosion |
| Corrosion Rate, icorr (µA/cm2) | Corrosion Level |
|---|---|
| <0.1 | Negligible (passivity) |
| 0.1 < icorr < 0.5 | Low corrosion |
| 0.5 < icorr < 1 | Moderate corrosion |
| >1 | High corrosion |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baltazar-Zamora, M.A.; M. Bastidas, D.; Santiago-Hurtado, G.; Mendoza-Rangel, J.M.; Gaona-Tiburcio, C.; Bastidas, J.M.; Almeraya-Calderón, F. Effect of Silica Fume and Fly Ash Admixtures on the Corrosion Behavior of AISI 304 Embedded in Concrete Exposed in 3.5% NaCl Solution. Materials 2019, 12, 4007. https://doi.org/10.3390/ma12234007
Baltazar-Zamora MA, M. Bastidas D, Santiago-Hurtado G, Mendoza-Rangel JM, Gaona-Tiburcio C, Bastidas JM, Almeraya-Calderón F. Effect of Silica Fume and Fly Ash Admixtures on the Corrosion Behavior of AISI 304 Embedded in Concrete Exposed in 3.5% NaCl Solution. Materials. 2019; 12(23):4007. https://doi.org/10.3390/ma12234007
Chicago/Turabian StyleBaltazar-Zamora, Miguel Angel, David M. Bastidas, Griselda Santiago-Hurtado, José Manuel Mendoza-Rangel, Citlalli Gaona-Tiburcio, José M. Bastidas, and Facundo Almeraya-Calderón. 2019. "Effect of Silica Fume and Fly Ash Admixtures on the Corrosion Behavior of AISI 304 Embedded in Concrete Exposed in 3.5% NaCl Solution" Materials 12, no. 23: 4007. https://doi.org/10.3390/ma12234007
APA StyleBaltazar-Zamora, M. A., M. Bastidas, D., Santiago-Hurtado, G., Mendoza-Rangel, J. M., Gaona-Tiburcio, C., Bastidas, J. M., & Almeraya-Calderón, F. (2019). Effect of Silica Fume and Fly Ash Admixtures on the Corrosion Behavior of AISI 304 Embedded in Concrete Exposed in 3.5% NaCl Solution. Materials, 12(23), 4007. https://doi.org/10.3390/ma12234007










