P3HT Nanofibrils Thin-Film Transistors by Adsorbing Deposition in Suspension
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation
2.3. Polymer Thin-Film Transistors
2.4. Characterization
3. Results
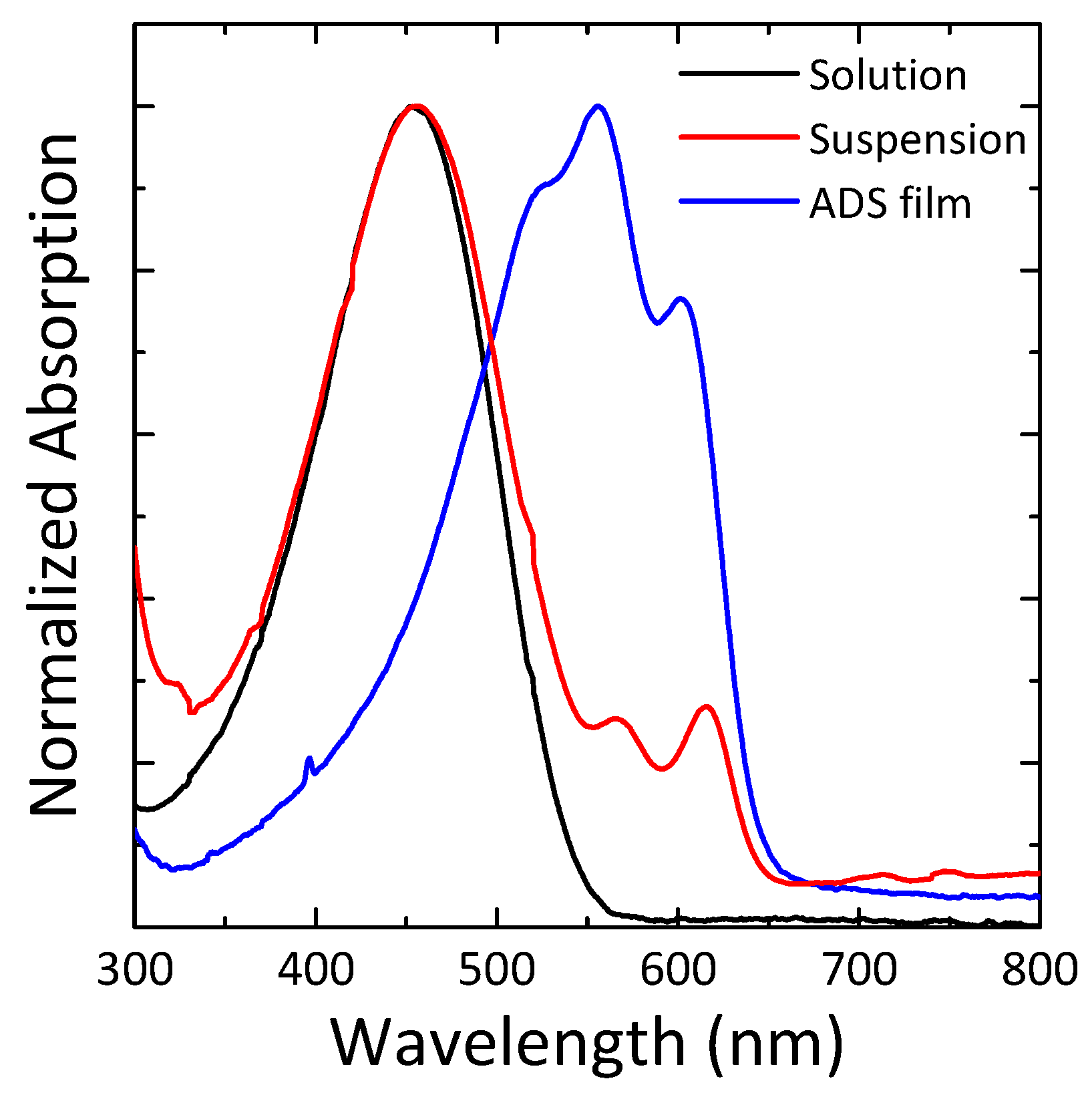
3.1. P3HT Nanofibrils Thin-Film
3.2. ADS P3HT Thin-Film Transistors
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Besra, L.; Liu, M. A review on fundamentals and applications of electrophoretic deposition (EPD). Prog. Mater. Sci. 2007, 52, 1–61. [Google Scholar] [CrossRef]
- Kamat, P.V.; Barazzouk, S.; Thomas, K.G.; Hotchandani, S. Electrodeposition of C60 cluster aggregates on Nanostructured SnO2 films for enhanced photocurrent generation. J. Phys. Chem. B 2000, 104, 4014–4017. [Google Scholar] [CrossRef]
- Tada, K.; Onoda, M. Preparation of smooth and dense composite films consisting of MEHPPV and neat C60 by means of electrophoretic deposition. Sol. Energy Mater. Sol. Cells 2011, 95, 688–692. [Google Scholar] [CrossRef]
- Tada, K.; Onoda, M. Uniform film of conjugated polymer for light-emitting device by electrophoretic deposition. J. Phys. D Appl. Phys. 2008, 41, 1–4. [Google Scholar] [CrossRef]
- Dhand, C.; Arya, S.K.; Datta, M.; Malhotra, B.D. Polyaniline-carbon nanotube composite film for cholesterol biosensor. Anal. Biochem. 2008, 383, 194–199. [Google Scholar] [CrossRef] [PubMed]
- Miyajima, S.; Nagamatsu, S.; Pandey, S.S.; Hayase, S.; Kaneto, K.; Takashima, W. Electrophoretic deposition onto an insulator for thin film preparation toward electronic device fabrication. Appl. Phys. Lett. 2012, 101, 1–4. [Google Scholar] [CrossRef]
- Tripathi, A.S.M.; Sadakata, S.; Gupta, R.K.; Nagamatsu, S.; Ando, Y.; Pandey, S.S. Implication of molecular weight on optical and charge transport anisotropy in PQT-C12 films fabricated by dynamic FTM. ACS Appl. Mater. Interfaces 2019, 11, 28088–280095. [Google Scholar] [CrossRef] [PubMed]
- Pandey, M.; Pandey, S.S.; Nagamatsu, S.; Hayase, S.; Takashima, W. Solvent driven performance in thin floating-films of PBTTT for organic field effect transistor: Role of macroscopic orientation. Org. Electron. 2017, 43, 240–246. [Google Scholar] [CrossRef]
- Yan, H.; Chen, Z.; Zheng, Y.; Newman, C.; Quinn, J.R.; Dotz, F.; Kastler, M.; Facchetti, A. A high-mobility electron-transporting polymer for printed transistors. Nature 2009, 457, 679–686. [Google Scholar] [CrossRef] [PubMed]
- Janasz, L.; Chlebosz, D.; Gradzka, M.; Zajaczkowski, W.; Marszalek, T.; Mullen, K.; Ulanski, J.; Kiersnowski, A.; Pisula, W. Improved charge carrier transport in ultrathin poly(3-hexylthiophene) films via solution aggregation. J. Mater. Chem. C 2016, 4, 11488–11498. [Google Scholar] [CrossRef]


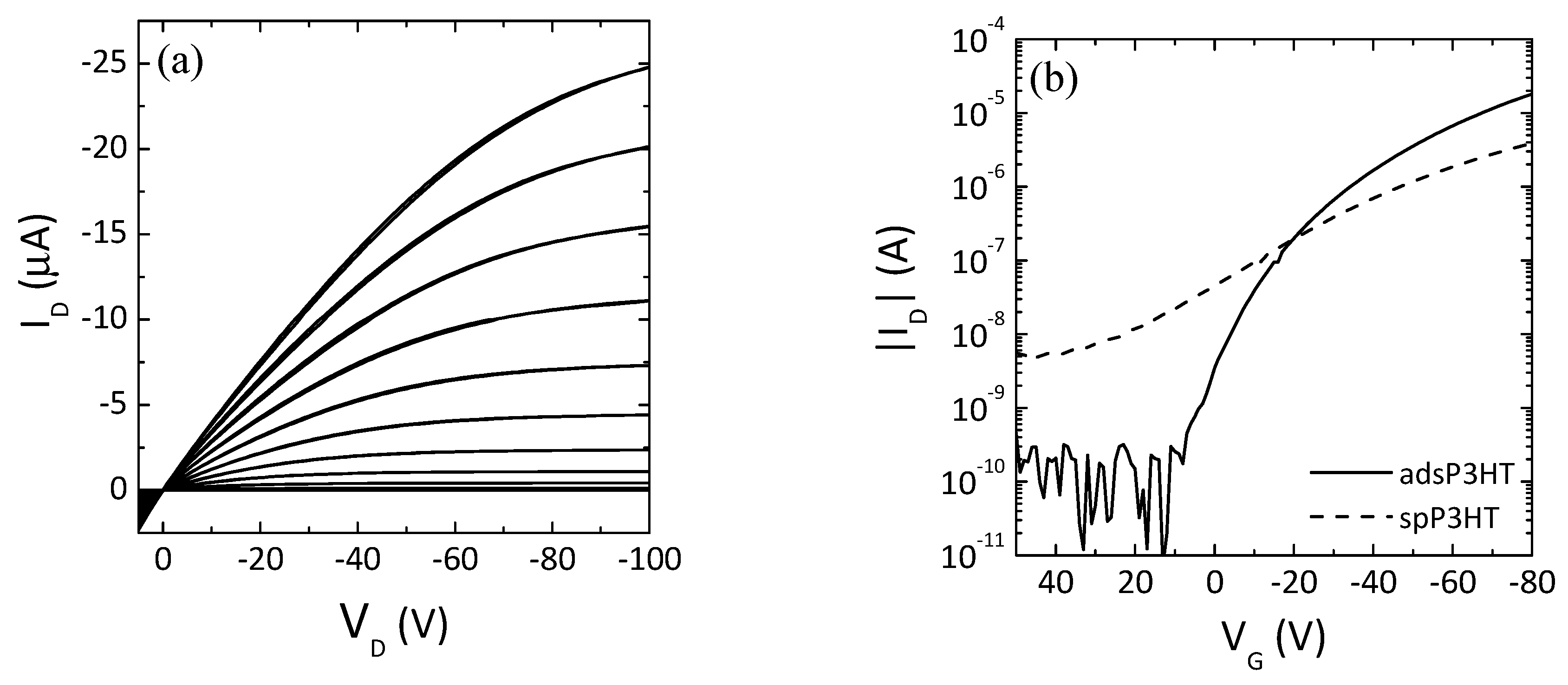
| - | Μ (cm2 V−1 s−1) | VT (V) | On/Off Ratio |
|---|---|---|---|
| adsP3HT | 0.02 | −37 | 500,000 |
| spP3HT | 0.002 | −18 | 800 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nagamatsu, S.; Ishida, M.; Miyajima, S.; Pandey, S.S. P3HT Nanofibrils Thin-Film Transistors by Adsorbing Deposition in Suspension. Materials 2019, 12, 3643. https://doi.org/10.3390/ma12213643
Nagamatsu S, Ishida M, Miyajima S, Pandey SS. P3HT Nanofibrils Thin-Film Transistors by Adsorbing Deposition in Suspension. Materials. 2019; 12(21):3643. https://doi.org/10.3390/ma12213643
Chicago/Turabian StyleNagamatsu, Shuichi, Masataka Ishida, Shougo Miyajima, and Shyam S. Pandey. 2019. "P3HT Nanofibrils Thin-Film Transistors by Adsorbing Deposition in Suspension" Materials 12, no. 21: 3643. https://doi.org/10.3390/ma12213643
APA StyleNagamatsu, S., Ishida, M., Miyajima, S., & Pandey, S. S. (2019). P3HT Nanofibrils Thin-Film Transistors by Adsorbing Deposition in Suspension. Materials, 12(21), 3643. https://doi.org/10.3390/ma12213643





