Design and Development of Ti–Ni, Ni–Mn–Ga and Cu–Al–Ni-based Alloys with High and Low Temperature Shape Memory Effects
Abstract
:1. Introduction
2. Materials and Methods
3. Results and Discussion
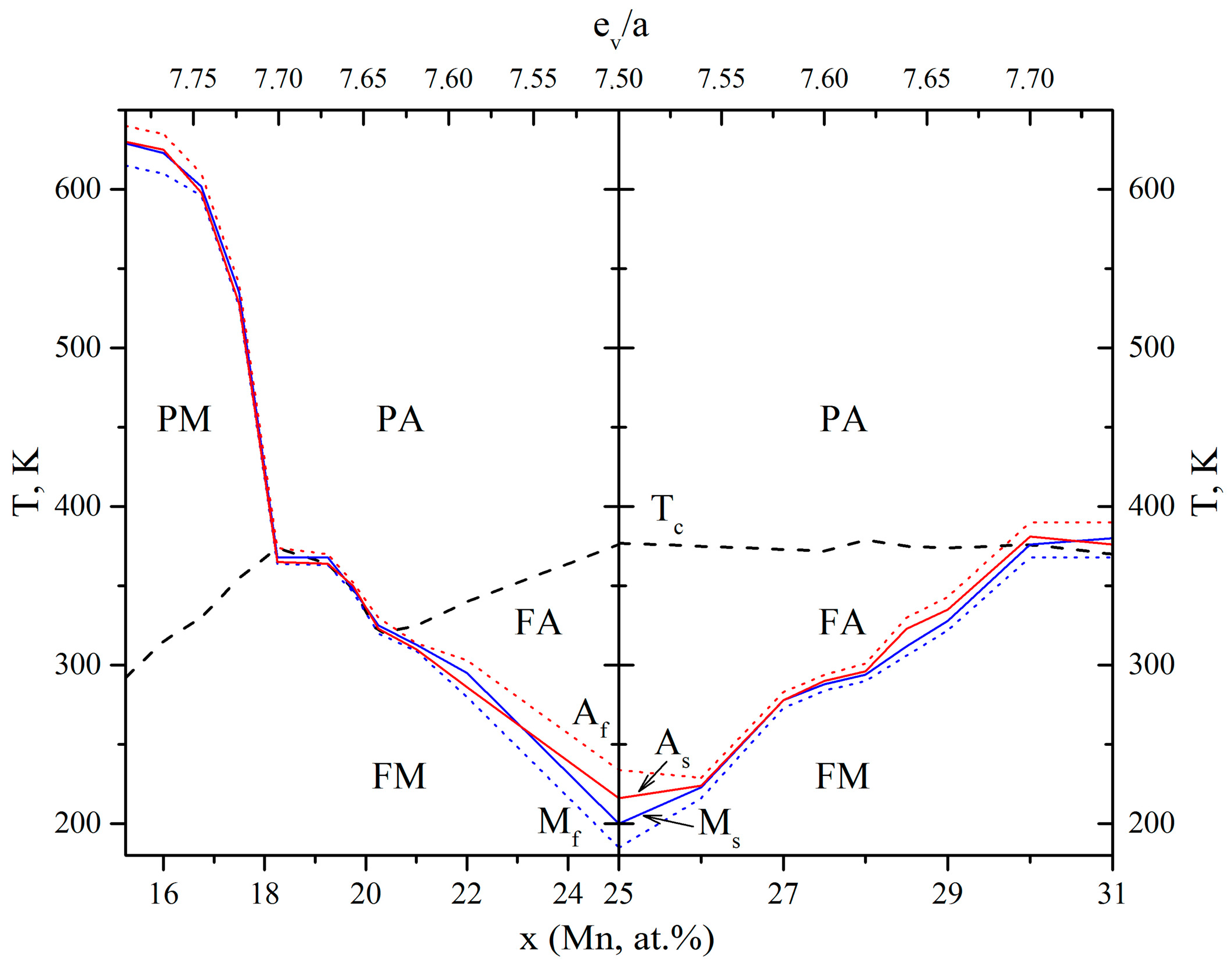
3.1. Temperature and Concentration Dependences of TMT Temperatures
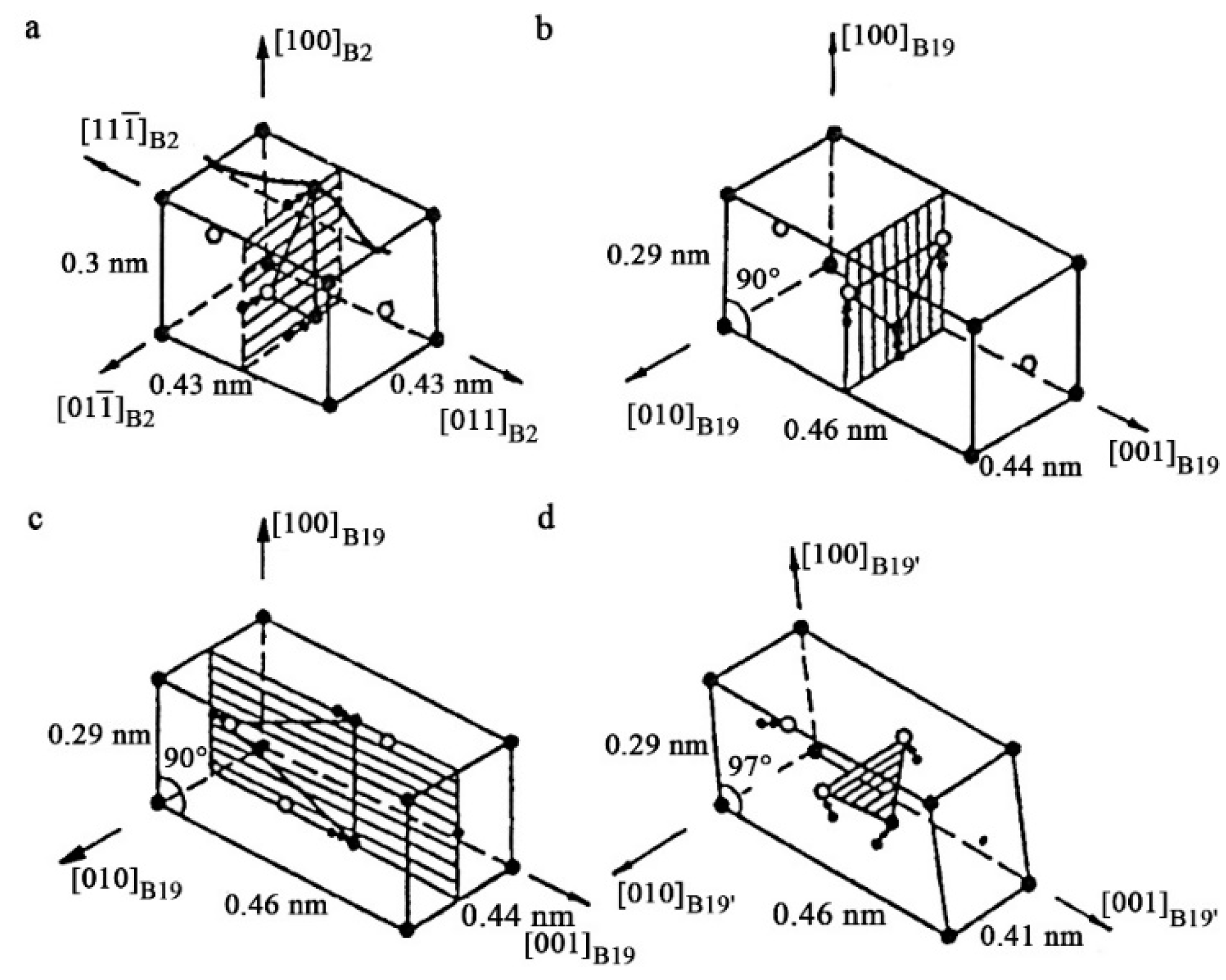
3.2. Crystal Structure and Morphological Regularities of Alloys with TMTs
3.3. Mechanical Properties of Fine- and Ultrafine-Grained Alloys
4. Summary and Conclusions
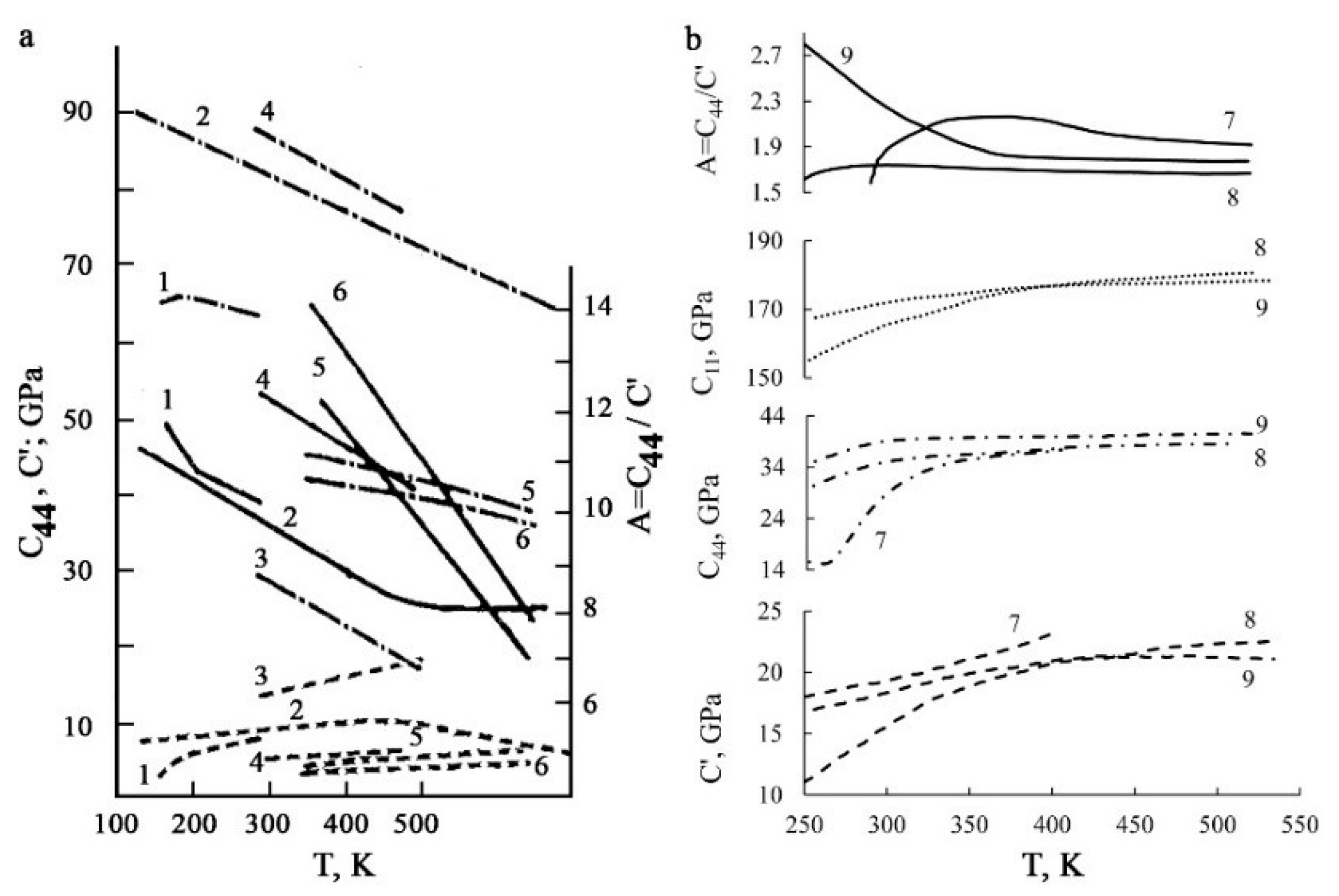
- The small volume effect |ΔV/V| < 1%, typical of TMTs, as well as the presence of pre-martensitic softening of elastic constants and the formation of a special heterogeneous pre-martensitic state was common for all studied alloys with a sufficiently noticeable temperature–concentration changes of crystal-structure parameters of austenite and martensitic phases, structural types of martensite and their metastable long-period variants.
- All studied alloys with TMTs combined the similarity of the pair-twinned packet morphology and microstructural hierarchy, proximity to Bain ratio, the action of several geometrically necessary twinning systems of martensite and the presence of coherent variants of their boundaries and subboundaries. In the condition of low elastic constants this diversity of substructure elements in thermoelastic martensite provided pseudo-elastic planar and volume lattice accommodation, and in the case of external mechanical influences, their mobile favorable reorientation and the effect of pseudoelasticity.
- The preservation of their single-phase high temperature austenitic state capable of TMTs played an important role in the multicomponent doping of these alloys. The chemical elements Zr and Hf from the Ti-period IVB of the periodic table had increased solubility (up to 25–30 at.%) replacing Ti in the quasi-binary alloys NiTi–NiMe. The elements Fe, Co, Pd and Pt from the Ni-period VIIIB had unlimited solubility replacing Ni in the quasi-binary alloys TiNi–TiMe. The solubility of the remained metals in the B2–TiNi compound was relatively low (up to 10 at.%), except for the unlimited solubility of Au and high (up to 25 at.%) one of Cu. It is essential that the high doping by Zr, Pd, Hf, Pt and Au provided a noticeable increase in the temperatures of TMTs, extending the temperature range of the implementation of TMTs above 373 K up to 1000 K. While Fe, Co and a number of other transition metals V, Cr and Mn, located between the elements Ti and Ni, caused them to noticeably decline below room temperature.
- When comparing the concentration changes of TMTs temperatures, average concentrations of valence electrons (cv) and the number of valence electrons (ev/a) for the alloys of the studied doping systems, a wide variety of them were established, which in general did not allow us to correctly determine their correlation. For example, for a number of alloys with high-temperature TMTs high-doped with Zr and Hf (replacing Ti) or Cu, Pd, Pt and Au (replacing Ni), the strong increase in TMTs temperatures was indeed consistent with the opposite decrease in the cv value. There was also the divergence between these factors and doping with V, Cr and Mn. On the contrary, there was a coinciding change in the other TiNi-based alloys moderately doped by the elements Al, Fe, Co, Cu, Pd, Pt and Au replacing Ni. In L21–Ni–Mn–Ga and D03–Cu–Al–Ni alloys, a strong increase in TMTs temperatures was also accompanied by a noticeable increase in cv and ev/a.
- The strong dependence on the grain size characterized the mechanical behavior of the studied alloys during tensile tests, especially for the relative elongation. The strength and the ductile properties of the ternary alloys decreased and the fracture became more brittle intercrystalline, in contrast to the viscous transcrystalline character in the binary TiNi alloy.
- In our opinion, the increase of the elastic anisotropy in high-doped alloys based on TiNi was the main reason for the strong dependence of their mechanical properties on the grain size. All other alloys with TMTs of Ni–Mn–Ga and Cu–Al–Ni systems also were characterized by the utmost high elastic anisotropy. It is because of the coherent accommodation of elastic stresses induced by the volume effect, they were localized at the grain boundaries, which ultimately becomes the general cause of intergranular brittleness of the alloys with large grain size undergoing TMTs.
- The strength and ductility of alloys with TMTs required for the realization of SMEs could be achieved by doping, which would provide a decrease in the elastic anisotropy parameter, and on the other hand, due to the formation of a fine-grained structure. Important especially for alloys with high temperature SMEs was the presence of homogeneous and heterogeneous decomposition with a strong strengthening effect that one should avoid using the quasi-binary precision doping.
Author Contributions
Funding
Conflicts of Interest
References
- Perkins, J. (Ed.) Shape Memory Effects in Alloys; Plenum: London, UK, 1975. [Google Scholar]
- Otsuka, K.; Shimizu, K.; Suzuki, Y.; Sekiguchi, Y.; Tadaki, C.; Honma, T.; Miyazaki, S. Shape Memory Alloys; Funakubo, H., Ed.; Funakubo: Kyoto, Japan, 1984. [Google Scholar]
- Duering, T.W.; Melton, K.L.; Stockel, D.; Wayman, C.M. (Eds.) Engineering Aspects of Shape Memory Alloys; Butterworth-Heineman: London, UK, 1990. [Google Scholar]
- Khachin, V.N.; Pushin, V.G.; Kondratyev, V.V. Titanium Nickelide: Structure and Properties; Nauka: Moscow, Russian, 1992. (In Russian) [Google Scholar]
- Otsuka, K.; Wayman, C.M. Shape Memory Materials; Cambridge University Press: Cambridge, UK, 1999. [Google Scholar]
- Pushin, V.G.; Kondratyev, V.V.; Khachin, V.N. Pretransition Phenomena and Martensitic Transformations; UrO RAN: Yekaterinburg, Russian, 1998. (In Russian) [Google Scholar]
- Yoneyama, T.; Miyazaki, S. Shape Memory Alloys for Medical Applications; Wordhead Publishing: Cambridge, UK, 2009. [Google Scholar]
- Prokoshkin, S.D.; Pushin, V.G.; Ryklina, E.P.; Khmelevskaya, I.Y. Application of Titanium Nickelide–based Alloys in Medicine. Phys. Met. Metallogr. 2004, 97, 56–96. [Google Scholar]
- Wilson, J.; Weselowsky, M. Shape Memory Alloys for Seismic Response Modification: A State-of-the-Art Review. Earthq. Spectra 2005, 21, 569–601. [Google Scholar] [CrossRef]
- Dong, J.; Cai, C.; OKeil, A. Overview of Potential and Existing Applications of Shape Memory Alloys in Bridges. J. Bridg. Eng. 2011, 16, 305–315. [Google Scholar] [CrossRef]
- Khachin, V.N.; Muslov, S.A.; Pushin, V.G.; Chumlyakov, Y.I. Anomalies of the Elastic Properties of Single Crystals of TiNi-TiFe. DAN SSSR 1987, 295, 606–609. (In Russian) [Google Scholar]
- Kuznetsov, A.V.; Muslov, S.A.; Lotkov, A.I.; Pushin, V.G.; Khachin, V.N.; Grishkov, V.N. Elastic Constants Near the TiNi Martensitic Transformations. Izvestia VUZov. Phys. 1987, 30, 98–99. (In Russian) [Google Scholar]
- Muslov, S.A.; Kuznetsov, A.V.; Khachin, V.N.; Lotkov, A.I.; Grishkov, V.N.; Pushin, V.G. Anomalies of the Elastic Constants of Single Crystals Ti50Ni48Fe2 Near Martensitic Transformations. Izvestiya VUZov. Phys. 1987, 30, 104–105. (In Russian) [Google Scholar]
- Khachin, V.N.; Muslov, S.A.; Pushin, V.G.; Kondratyev, V.V. Special elastic properties of B2-compounds of titanium with unstable lattice. Metallophysics 1988, 10, 102–104. (In Russian) [Google Scholar]
- Pushin, V.G.; Khachin, V.N.; Kondratyev, V.V.; Muslov, S.A.; Pavlova, S.P.; Yurchenko, L.I. Structure and Properties of B2 Compounds of Titanium. I. Pre-Martensitic Phenomena. Phys. Met. Metallogr. 1988, 66, 350–358. (In Russian) [Google Scholar]
- Kondratyev, V.V.; Muslov, S.A.; Pushin, V.G.; Khachin, V.N. Structure and Properties of Titanium B2 Compounds. II. Premartensitic Instability of the BCC (B2) Lattice. Phys. Met. Metallogr. 1988, 66, 359–369. (In Russian) [Google Scholar]
- Khachin, V.N.; Pushin, V.G.; Sivokha, V.P.; Kondratyev, V.V.; Muslov, A.S.; Voronin, V.P. Structure and Properties of B2 Titanium Compounds. III. Martensitic Transformations. Phys. Met. Metallogr. 1989, 67, 756–766. (In Russian) [Google Scholar]
- Khachin, V.N.; Sivokha, V.P.; Kondratyev, V.V.; Pushin, V.G. Structure and Properties of B2 Titanium Compounds. IV. Inelastic Behavior. Phys. Met. Metallogr. 1989, 68, 715–722. (In Russian) [Google Scholar]
- Pushin, V.G.; Kondratyev, V.V. Pretransition Phenomena and Martensitic Transformations. Phys. Met. Metallogr. 1994, 78, 40–61. (In Russian) [Google Scholar]
- Pushin, V.G.; Khachin, V.N.; Yurchenko, L.I.; Muslov, S.A.; Ivanova, L.Y.; Sokolova, A.Y. Microstructure and Physical Properties of Ti50Ni50-xFex Alloys with Memory Effects. II. Elastic Properties. Phys. Met. Metallogr. 1995, 79, 70–76. (In Russian) [Google Scholar]
- Lobodyuk, V.A.; Koval’, Y.N.; Pushin, V.G. Crystal-Structural Features of Pretransition Phenomena and Thermoelastic Martensitic Transformations in Alloys of Nonferrous Metals. Phys. Met. Metallogr. 2011, 111, 165–189. [Google Scholar] [CrossRef]
- Satija, S.K.; Shapiro, S.M.; Salamon, M.B.; Wayman, C.M. Phonon Softening in Ni46.8Ti50Fe3.2. Phys. Rev. B 1984, 29, 6031–6035. [Google Scholar] [CrossRef]
- Moine, P.; Allain, J.; Renker, B. Observation of a soft-phonon mode and a premartensitic phase in the intermetallic compound Ti50Ni47Fe3 studied by inelastic neutron scattering. J. Phys. F Met. Phys. 1984, 14, 2517–2529. [Google Scholar] [CrossRef]
- Tietzet, H.; Miillner, M.; Renker, B. Dynamical properties of premartensitic NiTi. J. Phys. C Solid State Phys. 1984, 17, L520–L532. [Google Scholar]
- Salamon, M.B.; Meichle, M.E.; Wayman, C.M. Premartensitic Phases of Ti50Ni47Fe3. Phys. Rev. B 1985, B31, 7306–7315. [Google Scholar] [CrossRef]
- Ren, X.; Otsuka, K. Why does the Martensitic Transformation Temperature Strongly Depend on Composition? Mater. Sci. Forum 2000, 327–328, 429–432. [Google Scholar] [CrossRef]
- Ren, X.; Miura, N.; Zhang, J.; Otsuka, K.; Tanake, K.; Koiwa, M.; Suzuki, N.; Chumlykov, Y.I. A Comparative Study of Elastic Constants of Ti-Ni-based Alloys Prior to Martensitic Transformation. Mater. Sci. Eng. 2001, A312, 196–206. [Google Scholar] [CrossRef]
- Otsuka, K.; Ren, X. Physical Metallurgy of Ti-Ni-based Shape Memory Alloys. Prog. Mater. C 2005, 50, 511–678. [Google Scholar] [CrossRef]
- Nakanishi, N. Elastic Constants as They Relate to Lattice Properties and Martensitic Formation. Prog. Mater. Sci. 1980, 24, 143–265. [Google Scholar] [CrossRef]
- Pushin, V.G.; Kondratyev, V.V.; Khachin, V.N. Pretransition Phenomena and Martensitic Transformations in Alloys Based on Titanium Nickel. Izvestiya VUZov. Phys. 1985, 27, 5–8. (In Russian) [Google Scholar]
- Kondratyev, V.V.; Pushin, V.G. Pretransitional state in Metals, Their Alloys and Compounds: Experimental Results, Structure Models, Classification. Phys. Met. Metallogr. 1985, 60, 629–650. (In Russian) [Google Scholar]
- Pushin, V.G. (Ed.) Shape Memory Alloys of Titanium Nickelide: Structure, Phase Transformations and Properties; UrO RAN: Yekatirinburg, Russian, 2006. (In Russian) [Google Scholar]
- Zheludev, A.; Shapiro, S.M.; Wochner, P.; Schwartz, A.; Wall, M.; Tanner, L.E. Phase Transformation and Phonon Anomalies in Ni2MnGa. J. Phys. IV Fr. 1995, 5, 1139–1144. [Google Scholar] [CrossRef]
- Worgull, J.; Petti, E.; Trivisonno, J. Behavior of the elastic properties near an intermediate phase transition in Ni2MnGa. Phys. Rev. B 1996, 54, 15695–15699. [Google Scholar] [CrossRef] [PubMed]
- Zarinejad, M.; Liu, Y. Dependence of Transformation Temperatures of NiTi-Based Shape Memory Alloys on the Number and Concentration of Valence Electrons. Adv. Funct. Mater. 2008, 18, 2789–2794. [Google Scholar] [CrossRef]
- Zarinejad, M.; Liu, Y.; White, T.J. The Crystal Chemistry of Martensite in NiTiHf Shape Memory Alloys. Intermetallics 2008, 16, 876–883. [Google Scholar] [CrossRef]
- Zarinejad, M.; Liu, Y. Dependence of Transformation Temperatures of Shape Memory Alloys on the Number and Concentration of Valence Electrons. In Shape Memory Alloys: Manufacture, Properties and Applications; Nova Science Publishers: Hauppauge, NY, USA, 2010; pp. 339–360. [Google Scholar]
- Frenzel, J.; Wieczorec, A.; Opahle, B.; MaaB, R.; Drautz, R.; Eggeler, G. On the Effect of Alloy Composition on Martensite Start Temperatures and Latent Heats in Ni–Ti-based Shape Memory Alloys. Acta Mater. 2015, 90, 213–231. [Google Scholar] [CrossRef]
- Reed, R.C.; Bismarck, M.; Skrotzki, B. The Superalloys—Fundamentals and Applications; Cambridge University Press: Cambridge, UK, 2006. [Google Scholar]
- Pushin, V.G.; Kuranova, N.N.; Pushin, A.V.; Uksusnikov, A.N.; Kourov, N.I. Structure and thermoelastic martensitic transformations in ternary Ni–Ti–Hf alloys with a high-temperature shape memory effect. Tech. Phys. 2016, 61, 1009–1014. [Google Scholar] [CrossRef]
- Kuranova, N.N.; Pushin, A.V.; Pushin, V.G.; Kourov, N.I. Structure and Thermoelastic Martensitic Transformations in Ternary Ni–Ti–Zr Alloys with High-Temperature Shape Memory Effects. Phys. Met. Metallogr. 2018, 119, 582–588. [Google Scholar] [CrossRef]
- Cesare, R.; Pons, J.; Santamarta, R.; Segui, C.; Chernenko, V.A. Ferromagnetic Shape Memory Alloys: An Overview. Arch. Metall. Mater. 2004, 49, 779–789. [Google Scholar]
- Nam, T.H.; Saburi, T.; Shimizu, K. Cu-content dependence of shape memory characteristics in Ti-Ni-Cu alloys. Mater. Trans. 1990, 31, 959–967. [Google Scholar] [CrossRef]
- Pushin, A.V.; Popov, A.A.; Pushin, V.G. Effect of the Deviation of the Chemical Composition from the Stoichiometric Composition on the Structural and Phase Transformations and Properties of Rapidly Quenched Ti50+xNi25–xCu25 Alloys. Phys. Met. Metallogr. 2012, 113, 283–294. [Google Scholar] [CrossRef]
- Pushin, A.V.; Popov, A.A.; Pushin, V.G. Effect of Deviations of Composition from the Quasi-Binary Section TiNi–TiCu on Structural and Phase Transformations in Rapidly Quenched Alloys. Phys. Met. Metallogr. 2013, 114, 692–702. [Google Scholar] [CrossRef]
- Nam, T.H.; Saburi, T.; Nakata, Y.; Shimizu, K. Shape Memory Characteristics and Lattice Deformation in Ti-Ni-Cu Alloys. Mater. Trans. 1990, 31, 1050–1056. [Google Scholar] [CrossRef]
- Delville, R.; Schryvers, D.; Zhang, Z.; James, R.D. Transmission Electron Microscopy Investigation of Microstructures in Low-Hysteresis Alloys with Special Lattice Parameters. Scr. Mater. 2019, 60, 293–296. [Google Scholar] [CrossRef]
- Hosoda, H.; Tachi, R.; Inamura, T.; Wakashima, K.; Miyazaki, S. Martensitic Transformation of TiAu Shape Memory Alloys. Mater. Sci. Forum 2007, 561–565, 1541–1544. [Google Scholar] [CrossRef]
- Evirgen, A.; Karaman, I.; Santamarta, R.; Pons, J.; Hayrettin, C.; Noebe, R.D. Relationship between crystallographic compatibility and thermal hysteresis in Ni-rich NiTiHf and NiTiZr high temperature shape memory alloys. Acta Mater. 2016, 121, 374–383. [Google Scholar] [CrossRef] [Green Version]
- Meng, X.L.; Cai, W.; Fu, Y.D.; Zhang, J.X.; Zhao, L.C. Martensitic Structure in Ti-Ni-Hf-Cu quaternary Alloy Ribbons Containing (Ti, Hf)2Ni Precipitates. Acta Mater. 2010, 58, 3751–3763. [Google Scholar] [CrossRef]
- Hu, L.; Jiang, S.; Liu, S.; Zhang, Y.; Zhao, Y.; Zhao, C. Transformation Twinning and Deformation Twinning of NiTi Shape Memory Alloy. Mater. Sci. Eng. A 2016, 660, 1–10. [Google Scholar] [CrossRef]
- Santamarta, R.; Arroyave, R.; Pons, J.; Evirgen, A.; Karaman, I.; Karaca, H.E.; Noebe, R.D. TEM Study of Structural and Microstructural Characteristics of a Precipitate Phase in Ni-rich Ni-Ti-Pa and Ni-Ti-Zr Shape Memory Alloys. Acta Mater. 2013, 61, 6191–6206. [Google Scholar] [CrossRef]
- Yi, X.; Gao, W.; Meng, X.; Gao, Z.; Cai, W.; Zhao, L. Martensitic Transformation Behaviours and Mechanical Properties of (Ti36Ni49Hf15)100-xYx High Temperature Shape Memory Alloys. J. Alloy. Comp. 2017, 705, 98–104. [Google Scholar] [CrossRef]
- Hong, S.H.; Kim, J.T.; Park, H.J.; Kim, Y.S.; Suh, J.Y.; Na, Y.S.; Lim, K.R.; Shim, C.H.; Park, J.M.; Kim, K.B. Influence of Zr Content on Phase Formation, Transition and Mechanical Behavior of Ni-Ti-Hf-Zr High Temperature Shape Memory Alloys. J. Alloy. Comp. 2017, 692, 77–85. [Google Scholar] [CrossRef]
- Fu, Y.; Du, H.; Huang, W.; Zhang, S.; Hu, M. TiNi-based Thin Films in MEMS Applications: A Review. Sens. Actuators A 2004, 112, 395–408. [Google Scholar] [CrossRef]
- Pushin, V.G.; Volkova, S.B.; Matveeva, N.M. Structural and Phase Transformations in Quasi-Binary TiNi-TiCu Alloys Rapidly Quenched from the Melt: III. Mechanisms of Crystallization. Phys. Met. Metallogr. 1997, 83, 435–443. [Google Scholar]
- Matveeva, N.M.; Pushin, V.G.; Shelyakov, A.V.; Bykovskii, Y.A.; Volkova, S.B.; Kraposhin, V.S. Effect of the Conditions of Crystallization of Amorphous TiNi–TiCu Alloys on their Structure and Shape Memory. Phys. Met. Metallogr. 1997, 83, 626–632. [Google Scholar]
- Cesari, E.; Ochin, P.; Portier, R.; Kolomytsev, V.; Koval, Y.; Pasko, A.; Soolshenko, V. Structure and Properties of Ti-Ni-Zr and Ti-Ni-Hf Melt-spun Ribbons. Mater. Sci. Eng. A 1999, 273–275, 738–744. [Google Scholar] [CrossRef]
- Vermaut, P.; Litynska, L.; Portier, R.; Ochin, P.; Dutkiewicz, J. The Microstructure of melt spun Ti-Ni-Cu-Zr Shape Memory Alloys. Mater. Chem. Phys. 2003, 81, 380–382. [Google Scholar] [CrossRef]
- Pasko, A.; Kolomytsev, V.; Vermaut, P.; Prima, F.; Portier, R.; Ochin, P.; Sezonenko, A. Crystallization of the Amorphous Phase and Martensitic Transformations in Multicomponent (Ti,Ni,Zr) (Ni,Cu)-based Alloys. J. Non Crys. Solid 2007, 353, 3062–3068. [Google Scholar] [CrossRef]
- Pushin, V.G.; Kuranova, N.N.; Pushin, A.V.; Uksusnikov, A.N.; Kourov, N.I.; Kuntsevich, T.E. Structural and Phase Transformations, Mechanical Properties and Shape-Memory Effects in Quasibinary Ni50Ti38Hf12 Alloy Obtained by Quenching from the Melt. Phys. Met. Metallogr. 2016, 117, 1251–1260. [Google Scholar] [CrossRef]
- Pushin, A.V.; Pushin, V.G.; Kuranova, N.N.; Kourov, N.I.; Kuntsevich, T.E.; Makarov, V.V.; Uksusnikov, A.N. Structure and Phase Transformations in Copper-Alloyed Rapidly Melt-Quenched Ni50Ti32Hf18-Based Alloys with High-Temperature Shape Memory Effect. Phys. Met. Metallogr. 2017, 118, 997–1005. [Google Scholar] [CrossRef]
- Kuranova, N.N.; Pushin, A.V.; Uksusnikov, A.N.; Belosludtseva, E.S.; Kourov, N.I.; Kuntsevich, T.E.; Pushin, V.G. Fine Structure and Mechanical Properties of the Shape-Memory Ni50Ti32Hf18 Alloy Rapidly Quenched by Spinning. Tech. Phys. 2017, 62, 1189–1193. [Google Scholar] [CrossRef]
- Pushin, A.V.; Pushin, V.G.; Kuntsevich, T.E.; Kuranova, N.N.; Makarov, V.V.; Uksusnikov, A.N.; Kourov, N.I. Features of Crystallization of Fast-Quenched Ni45Ti32Hf18Cu5 and Ni25Ti32Hf18Cu25 Alloys from Melt with High-Temperature Shape Memory Effect. Tech. Phys. 2017, 62, 1843–1847. [Google Scholar] [CrossRef]
- Jiang, B.; Wang, J.; Xu, L.; Qian, C.; Liu, T.; Dai, J.; Hou, X. Tunable Mechanical Properties of Ti-Zr-Ni-Cr-V Amorphous Ribbons via Different Melt Spinning Speeds During Rapid Solidification Process. Materials 2018, 11, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Scudino, S.; Venkataraman, S.; Eckert, J. Thermal Stability, Microstructure and Crystallization Kinetics of Melt-Spun Zr-Ti-Cu-Ni Metallic Glass. J. Alloy. Comp. 2008, 460, 263–268. [Google Scholar] [CrossRef]
- Inamura, T.; Takahashi, Y.; Hosoda, H.; Wakashima, K.; Nagase, T.; Nakano, T.; Umakoshi, Y.; Miyazaki, S. Martensitic Transformation Behavior and Shape Memory Properties of Ti-Ni-Pt Melt-Spun Ribbons. Mater. Trans. 2006, 47, 540–545. [Google Scholar] [CrossRef]
- Badhirappan, G.P. Understanding Structure-Property Relation in Nano-Crystalline Ni-Ti Shape Memory Alloys Thin Film Micro-Actuator. Micro Smart Syst. 2019. [Google Scholar] [CrossRef]
- Yi, X.; Wang, H.; Sun, B.; Sun, K.; Gao, W.; Liu, G.; Meng, X.; Cai, W.; Zhao, L. The Crystallization Process Microstructure, Martensitic Transformation and Mechanical Properties of Ti-Ni-Zr Alloy Ribbons. J. Alloy. Comp. 2019, 778, 542–553. [Google Scholar] [CrossRef]
- Pushin, V.G. Alloys with a Thermo-Mechanical Memory: Structure, Properties, and Application. Phys. Met. Metallogr. 2000, 90, S68–S95. [Google Scholar]
- Pushin, V.G.; Stolyarov, V.V.; Valiev, R.Z.; Kourov, N.I.; Kuranova, N.N.; Prokofiev, E.A.; Yurchenko, L.I. Features of Structure and Phase Transformations in Shape Memory TiNi-Based Alloys after Severe Plastic Deformation. Ann. Chim. Sci. Mat. 2002, 27, 77–88. [Google Scholar] [CrossRef]
- Valiev, R.Z.; Pushin, V.G. Bulk Nanostructured Metallic Materials: Production, Structure, Properties and Functioning. Phys. Met. Metallogr. 2002, 94, S1–S4. [Google Scholar]
- Pushin, V.G.; Stolyarov, V.V.; Valiev, R.Z.; Kourov, N.I.; Kuranova, N.N.; Prokofiev, E.A.; Yurchenko, L.I. Development of Methods of Severe Plastic Deformation for the Production of High-Strength Alloys Based on Titanium Nickelide with a Shape Memory Effect. Phys. Met. Metallogr. 2002, 94, S54–S68. [Google Scholar]
- Pushin, V.G.; Valiev, R.Z. The Nanostructured TiNi Shape-Memory Alloys: New Properties and Applications. Solid State Phenom. 2003, 94, 13–24. [Google Scholar] [CrossRef]
- Pushin, V.G.; Valiev, R.Z.; Yurchenko, L.I. Processing of Nanostructured TiNi-Shape Memory Alloys: Methods, Structures, Properties, Application. J. Phys. IV Fr. 2003, 112, 659–662. [Google Scholar] [CrossRef]
- Pushin, V.G. Historical Development of Titanium Nickelide-Based Alloys with Thermomechanical Memory. Phys. Met. Metallogr. 2004, 9, S1–S2. [Google Scholar]
- Brailovski, V.; Khmelevskaya, I.Y.; Prokoshkin, S.D.; Pushin, V.G.; Ryklina, E.P.; Valiev, R.Z. Foundation of Heat and Thermomechanical Treatments and Their on the Structure and Properties of Titanium Nickelide-Based Alloys. Phys. Met. Metallogr. 2004, 97, S3–S55. [Google Scholar]
- Pushin, V.G.; Valiev, R.Z.; Zhu, Y.T.; Gunderov, D.V.; Kourov, N.I.; Kuntsevich, T.E.; Uksusnikov, A.N.; Yurchenko, L.I. Effect of Severe Plastic Deformation on the Behavior of Ti-Ni Shape Memory Alloys. Mater. Trans. 2006, 47, 694–697. [Google Scholar] [CrossRef]
- Pushin, V.G.; Valiev, R.Z.; Zhu, Y.T.; Gunderov, D.V.; Korolev, A.V.; Kourov, N.I.; Kuntsevich, T.E.; Valiev, E.Z.; Yurchenko, L.I. Severe Plastic Deformation of Melt-Spun Shape Memory Ti2NiCu and Ni2MnGa Alloys. Mater. Trans. 2006, 47, 546–549. [Google Scholar] [CrossRef]
- Pushin, V.G.; Korolev, A.V.; Kourov, N.I.; Gunderov, D.V.; Valiev, R.Z.; Koledov, V.V.; Shavrov, V.G. SPD-Induced Nanocrystallization of Shape Memory Ni2MnGa-Based and NiTi-Based Alloys Quenched from Liquid State. Mater. Sci. Forum 2006, 503–504, 545–550. [Google Scholar] [CrossRef]
- Valiev, R.Z.; Gunderov, D.V.; Pushin, V.G. The New SPD Processing Routes to Fabricate Bulk Nanostructured Materials. Ultrafine Grained Materials IV; Zhu, Y.T., Langdon, T.G., Horita, Z., Zehetbauer, M.J., Semiatin, S.L., Lowe, T.C., Eds.; TMS (The Minerals, Metals and Materials Society): Warrendale, PA, USA, 2006. [Google Scholar]
- Pushin, V.G.; Valiev, R.Z.; Zhu, Y.T.; Prockoshkin, S.; Gunderov, D.V.; Yurchenko, L.I. Effect of Equal Channel Angular Pressing and Repeated Rolling on Structure, Phase Transformation and Properties of TiNi Shape Memory Alloys. Mater. Sci. Forum 2006, 503–504, 539–544. [Google Scholar] [CrossRef]
- Valiev, R.; Gunderov, D.; Prokofiev, E.; Pushin, V.; Zhu, Y. Nanostructuring of TiNi alloy by SPD processing for advanced properties. Mater. Trans. 2008, 49, 97–101. [Google Scholar] [CrossRef]
- Tsuchiya, K.; Hada, Y.; Koyano, T.; Nakajima, K.; Ohnuma, M.; Koike, T.; Todaka, Y.; Umimota, M. Production of TiNi Amorphous/Nanocrystalline Wires with High-Strength and Elastic Modulus by Severe Cold Drawing. Scr. Mater. 2009, 60, 749–752. [Google Scholar] [CrossRef]
- Prokoshkin, S.; Brailivski, V.; Korotitskiy, A.; Inaekyan, K.; Dubinsky, S.; Filonov, M.; Petrzhic, M. Formation of Nanostructures in Thermo-Mechanically-Treated Ti-Ni and Ti-Nb-(Zr, Ta) SMAs and Their Roles in Martensite Crystal Lattice Changes and Mechanical Behavior. Alloy. Comp. 2011, 509, 2066–2075. [Google Scholar]
- Zhang, Y.; Jiang, S.; Hu, L.; Liang, Y. Deformation Mechanism of NiTi Shape Memory Alloy Subjected to Severe Plastic Deformation at Low Temperature. Mater. Sci. Eng. A 2013, 559, 606–614. [Google Scholar] [CrossRef]
- Dye, D.; Azeem, M.A. In Situ Evaluation of the Transformation Behaviour of NiTi-based High Temperature Shape Memory Alloys. Intermetallics 2014, 46, 222–230. [Google Scholar]
- Shahmir, H.; Nili-Ahmadabadi, M.; Huang, Y.; Jung, J.M.; Kim, H.S.; Langdon, T.G. Shape Memory Effect in Nanocrystalline NiTi Alloy Processed by High Pressure Torsion. Mater. Sci. Eng. A 2015, 626, 203–206. [Google Scholar] [CrossRef]
- Prokoshkin, S.; Dubinsky, S.; Brailivski, V.; Korotitskiy, A.; Konopatsky, A.; Sheremetyev, V.; Blinova, E. Nanostructures and Stress-Induced Phase Transformation Mechanism in Titanium Nickelide annealed after Moderate Cold Deformation. Mater. Lett. 2017, 192, 111–114. [Google Scholar] [CrossRef]
- Tulic, S.; Kerber, M.; Matsuda, M.; Waitz, T. Phase Transformations of Severely Plastically Deformed Ti-Ni-Pd High-Temperature Shape Memory Alloys. Funct. Mater. Lett. 2017, 10, 1740012: 1-8. [Google Scholar] [CrossRef]
- Prokoshkin, S.; Dubinsky, S.; Korotitskiy, A.; Konopatsky, A.; Sheremetyev, V.; Schetitin, I.; Gleser, A.; Brailivski, V. Nanostructure Features and Stress-Induced Transformation Mechanisms in Extremely Fine-Grained Titanium Nickelide. J. Alloy. Comp. 2019, 770, 667–686. [Google Scholar] [CrossRef]












| Alloy, at.% | R | B19′ | ev/a | cv | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Ms′, K | Mf′, K | As′, K | Af′, K | Ms, K | Mf, K | As, K | Af, K | |||
| Ti50Ni50 | 343 | 319 | 353 | 373 | 7.00 | 0.280 | ||||
| Ti50Ni49Al1 | 319 | 305 | 301 | 323 | 264 | 245 | 289 | 309 | 6.93 | 0.279 |
| Ti50Ni48Al2 | 295 | 280 | 276 | 301 | 200 | 178 | 227 | 253 | 6.86 | 0.278 |
| Ti50Ni47Al3 | 262 | 247 | 242 | 270 | 130 | 105 | 161 | 188 | 6.79 | 0.277 |
| Ti50Ni46Al4 | 190 | 179 | 171 | 198 | 6.72 | 0.275 | ||||
| Ti50Ni45Al5 | 139 | 131 | 112 | 142 | 6.69 | 0.274 | ||||
| Ni50Ti49Al1 | 321 | 301 | 305 | 324 | 266 | 247 | 288 | 309 | 6.99 | 0.281 |
| Ni50Ti48Al2 | 300 | 272 | 277 | 303 | 199 | 162 | 222 | 251 | 6.98 | 0.281 |
| Ni50Ti48,5Al2,5 | 282 | 253 | 256 | 285 | 109 | 179 | 211 | 6.98 | 0.282 | |
| Ni50Ti47Al3 | 260 | 228 | 232 | 265 | 6.97 | 0.282 | ||||
| Ni50Ti46Al4 | 173 | 134 | 147 | 184 | 6.96 | 0.282 | ||||
| Ti50Ni49Mn1 | 316 | 290 | 285 | 321 | 280 | 237 | 289 | 299 | 6.97 | 0.279 |
| Ti50Ni48Mn2 | 295 | 263 | 259 | 301 | 208 | 164 | 230 | 259 | 6.94 | 0.278 |
| Ti50Ni47Mn3 | 277 | 242 | 237 | 282 | 135 | 89 | 176 | 202 | 6.91 | 0.277 |
| Ti50Ni45Mn5 | 237 | 191 | 189 | 243 | 6.85 | 0.276 | ||||
| Ni50Ti49Mn1 | 304 | 266 | 272 | 305 | 283 | 235 | 298 | 320 | 7.03 | 0.281 |
| Ni50Ti48,5Mn1,5 | 296 | 259 | 266 | 301 | 247 | 191 | 265 | 290 | 7.05 | 0.281 |
| Ni50Ti48Mn2 | 288 | 253 | 259 | 295 | 186 | 122 | 218 | 240 | 7.06 | 0.282 |
| Ni50Ti47Mn3 | 253 | 216 | 223 | 262 | 78 | 112 | 140 | 7.09 | 0.283 | |
| Ni50Ti46Mn4 | 173 | 134 | 146 | 183 | 7.12 | 0.283 | ||||
| Ti50Ni49Fe1 | 307 | 294 | 298 | 313 | 278 | 250 | 289 | 305 | 6.98 | 0.279 |
| Ti50Ni48Fe2 | 287 | 266 | 276 | 297 | 218 | 189 | 235 | 258 | 6.96 | 0.279 |
| Ti50Ni46Fe4 | 264 | 238 | 252 | 280 | 155 | 111 | 186 | 210 | 6.92 | 0.278 |
| Ti50Ni45Fe5 | 230 | 202 | 217 | 240 | 6.90 | 0.277 | ||||
| Ti50Ni49Co1 | 324 | 305 | 313 | 330 | 313 | 265 | 322 | 323 | 6.99 | 0.280 |
| Ti50Ni48Co2 | 301 | 283 | 291 | 311 | 271 | 216 | 289 | 290 | 6.98 | 0.279 |
| Ti50Ni46Co4 | 287 | 266 | 274 | 297 | 237 | 175 | 261 | 267 | 6.96 | 0.279 |
| Ti50Ni45Co5 | 263 | 235 | 244 | 272 | 165 | 101 | 207 | 220 | 6.95 | 0.279 |
| Ti50Ni40Co10 | 218 | 183 | 190 | 226 | 6.90 | 0.277 | ||||
| Ti50Ni35Co15 | 130 | 80 | 89 | 140 | 6.85 | 0.276 | ||||
| Alloy, at.% | B19 | B19′ | ev/a | cv | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Ms′, K | Mf′, K | As′, K | Af′, K | Ms, K | Mf, K | As, K | Af, K | |||
| Ti50Ni45Cu5 | 315 | 222 | 255 | 337 | 7.05 | 0.281 | ||||
| Ti50Ni42,5Cu7,5 | 305 | 209 | 229 | 318 | 7.08 | 0.282 | ||||
| Ti50Ni40Cu10 | 312 | 302 | 307 | 316 | 280 | 201 | 222 | 291 | 7.10 | 0.283 |
| Ti50Ni35Cu15 | 332 | 317 | 322 | 337 | 200 | 181 | 186 | 207 | 7.15 | 0.284 |
| Ti50Ni33Cu17 | 337 | 326 | 331 | 342 | 7.17 | 0.284 | ||||
| Ti50Ni30Cu20 | 337 | 327 | 332 | 345 | 7.20 | 0.285 | ||||
| Ti50Ni25Cu25 | 340 | 326 | 333 | 348 | 7.25 | 0.287 | ||||
| Ti50Ni22Cu28 | 335 | 323 | 328 | 342 | 7.28 | 0.288 | ||||
| Ti50Ni18Cu32 | 331 | 313 | 322 | 339 | 7.32 | 0.289 | ||||
| Ti50Ni16Cu34 | 321 | 307 | 313 | 330 | 7.34 | 0.290 | ||||
| Ti50Ni48Pd2 | 330 | 310 | 335 | 355 | 7.00 | 0.276 | ||||
| Ti50Ni45Pd5 | 320 | 300 | 330 | 350 | 7.00 | 0.27 | ||||
| Ti50Ni40Pd10 | 330 | 320 | 330 | 340 | 270 | 230 | 240 | 280 | 7.00 | 0.261 |
| Ti50Ni25Pd25 | 450 | 410 | 430 | 470 | 7.00 | 0.237 | ||||
| Ti50Ni5Pd45 | 710 | 680 | 710 | 740 | 7.00 | 0.211 | ||||
| Ti50Ni48Pt2 | 305 | 280 | 305 | 330 | 7.00 | 0.270 | ||||
| Ti50Ni42Pt8 | 300 | 275 | 280 | 305 | 270 | 240 | 260 | 290 | 7.00 | 0.245 |
| Ti50Ni25Pt25 | 720 | 670 | 720 | 770 | 7.00 | 0.194 | ||||
| Ti50Ni47Au3 | 315 | 280 | 320 | 350 | 7.03 | 0.265 | ||||
| Ti50Ni45Au5 | 360 | 305 | 320 | 375 | 7.05 | 0.256 | ||||
| Ti50Ni38Au12 | 365 | 340 | 355 | 380 | 300 | 255 | 250 | 310 | 7.12 | 0.229 |
| Ti50Ni25Au25 | 425 | 365 | 400 | 460 | 7.25 | 0.192 | ||||
| Ti50Au50 | 880 | 850 | 870 | 900 | 7.50 | 0.149 | ||||
| Ni50Ti47Zr3 | 350 | 325 | 360 | 385 | 7.00 | 0.274 | ||||
| Ni50Ti45Zr5 | 350 | 325 | 370 | 395 | 7.00 | 0.27 | ||||
| Ni50Ti40Zr10 | 375 | 350 | 400 | 420 | 7.00 | 0.261 | ||||
| Ni50Ti35Zr15 | 455 | 425 | 475 | 500 | 7.00 | 0.253 | ||||
| Ni50Ti32Zr18 | 510 | 480 | 535 | 555 | 7.00 | 0.248 | ||||
| Ni50Ti30Zr20 | 555 | 520 | 570 | 600 | 7.00 | 0.245 | ||||
| Ni50Ti38Hf12 | 405 | 380 | 430 | 450 | 7.00 | 0.226 | ||||
| Ni50Ti35Hf15 | 450 | 430 | 480 | 500 | 7.00 | 0.215 | ||||
| Ni50Ti32Hf18 | 500 | 475 | 530 | 555 | 7.00 | 0.206 | ||||
| Ni50Ti30Hf20 | 520 | 495 | 564 | 585 | 7.00 | 0.200 | ||||
| Alloy, at.% | Ms, K | Mf, K | As, K | Af, K | ev/a | cv |
|---|---|---|---|---|---|---|
| Ni50Mn50 | 970 | 920 | 970 | 1020 | 8.50 | 0.321 |
| Ni50Mn48Ga2 | 907 | 880 | 920 | 954 | 8.42 | 0.316 |
| Ni50Mn46Ga4 | 860 | 830 | 860 | 897 | 8.34 | 0.312 |
| Ni50Mn44Ga6 | 807 | 785 | 830 | 845 | 8.26 | 0.308 |
| Ni50Mn42Ga8 | 755 | 790 | 8.18 | 0.303 | ||
| Ni50Mn40Ga10 | 693 | 720 | 8.10 | 0.299 | ||
| Ni50Mn36Ga14 | 546 | 525 | 540 | 557 | 7.94 | 0.290 |
| Ni50Mn34Ga16 | 457 | 450 | 460 | 469 | 7.86 | 0.286 |
| Ni50Mn32Ga18 | 378 | 375 | 375 | 390 | 7.78 | 0.282 |
| Ni50Mn31Ga19 | 368 | 363 | 363 | 370 | 7.74 | 0.280 |
| Ni50Mn30Ga20 | 362 | 356 | 370 | 374 | 7.70 | 0.278 |
| Ni50Mn29Ga21 | 328 | 322 | 335 | 343 | 7.66 | 0.276 |
| Ni50Mn28.5Ga21.5 | 312 | 306 | 323 | 330 | 7.64 | 0.275 |
| Ni50Mn28Ga22 | 294 | 290 | 296 | 301 | 7.62 | 0.274 |
| Ni50Mn27.5Ga22.5 | 288 | 284 | 290 | 294 | 7.60 | 0.273 |
| Ni50Mn27Ga23 | 278 | 273 | 278 | 283 | 7.58 | 0.272 |
| Ni50Mn26Ga24 | 223 | 216 | 224 | 229 | 7.54 | 0.270 |
| Ni50Mn25Ga25 | 200 | 185 | 216 | 234 | 7.50 | 0.268 |
| Ni53Mn22Ga25 | 295 | 280 | 286 | 303 | 7.59 | 0.270 |
| Ni54Mn21Ga25 | 313 | 309 | 310 | 314 | 7.62 | 0.271 |
| Ni54.75Mn20.25Ga25 | 325 | 320 | 323 | 330 | 7.64 | 0.272 |
| Ni55.25Mn19.75Ga25 | 348 | 346 | 350 | 352 | 7.66 | 0.272 |
| Ni55.75Mn19.25Ga25 | 368 | 363 | 364 | 370 | 7.67 | 0.272 |
| Ni56.75Mn18.25Ga25 | 368 | 364 | 365 | 374 | 7.70 | 0.273 |
| Ni57.5Mn17.5Ga25 | 535 | 526 | 528 | 540 | 7.73 | 0.274 |
| Ni58.25Mn16.75Ga25 | 602 | 596 | 598 | 610 | 7.75 | 0.274 |
| Ni59Mn16Ga25 | 623 | 610 | 625 | 635 | 7.77 | 0.275 |
| Ni60Mn15Ga25 | 629 | 615 | 630 | 640 | 7.79 | 0.276 |
| Alloy, at.% | As, K | Af, K | ev/a | cv | <d>, μm | σm, MPa | σu, MPa | δ, % | Ψ, % |
|---|---|---|---|---|---|---|---|---|---|
| Cu78Al19Ni3 | 903 | 943 | 9.45 | 0.364 | 60 | 280 | 780 | 15 | 0.5 |
| Cu77Al20Ni3 | 893 | 933 | 9.33 | 0.363 | 80 | 260 | 520 | 10 | 0.5 |
| Cu75Al22Ni3 | 793 | 833 | 9.21 | 0.362 | 130 | 260 | 490 | 6 | 0.5 |
| Cu73Al24Ni3 | 673 | 723 | 9.05 | 0.360 | 350 | 260 | 450 | 5 | 0.5 |
| Cu71Al26Ni3 | 513 | 560 | 8.89 | 0.358 | 750 | 200 | 390 | 4 | 0.5 |
| Cu69Al28Ni3 | 283 | 333 | 8.77 | 0.357 | 1000 | 120 | 250 | 3 | 0.5 |
| Alloy, at.% | B19′ | ev/a | cv | |||
|---|---|---|---|---|---|---|
| Ms, K | Mf, K | As, K | Af, K | |||
| Ti50Ni49.5Cr0.5 [38] * | 292 | 264 | 302 | 329 | 6.98 | 0.279 |
| Ti49Ni50Cr1 [38] * | 237 | 215 | 252 | 268 | 7.02 | 0.281 |
| Ti49Ni50V1 [38] * | 318 | 284 | 327 | 348 | 7.01 | 0.280 |
| Ti48Ni50V2 [38] * | 309 | 285 | 316 | 340 | 7.02 | 0.281 |
| Ti47Ni50V3 [38] * | 293 | 271 | 300 | 318 | 7.03 | 0.281 |
| Ti45Ni50V5 [38] * | 283 | 269 | 286 | 297 | 7.05 | 0.281 |
| Ti44Ni50V6 [38] * | 279 | 261 | 282 | 295 | 7.06 | 0.282 |
| Ti50Ni45Cu5 [38] * | 345 | 317 | 340 | 368 | 7.05 | 0.281 |
| Ti50Ni40Cu10 [38] * | 306 | 285 | 300 | 316 | 7.10 | 0.283 |
| Ti50Ni45Cu5 [43] ** | 309 | 295 | 311 | 330 | 7.05 | 0.281 |
| Ti50Ni40Cu10 [43] ** | 292 | 179 | 192 | 200 | 7.10 | 0.283 |
| Alloy, at.% | ΔV/V, % | B2 | Martensite B19′ (B19) | |||
|---|---|---|---|---|---|---|
| a, nm | a, nm | b, nm | c, nm | β, ° | ||
| Ti50Ni50 | −0.13 | 0.3015 | 0.2890 | 0.4120 | 0.4630 | 96.8 |
| Ti49.4Ni50.6 | −0.31 | 0.3013 | 0.2876 | 0.4132 | 0.4622 | 97.0 |
| Ti50Ni49Co1 | −0.24 | 0.3014 | 0.2882 | 0.4115 | 0.4644 | 97.3 |
| Ti50Ni46Co4 | −0.86 | 0.3013 | 0.2874 | 0.4108 | 0.4630 | 97.2 |
| Ti50Ni45Co5 | 0.49 | 0.3012 | 0.2871 | 0.4104 | 0.4697 | 97.1 |
| Ti50Ni45Cu5 [46] | −0.5 | 0.3027 | 0.2903 | 0.4147 | 0.4613 | 96.2 |
| Ti50Ni45Cu7.5 [46] | 0.36 | 0.3030 | 0.2911 | 0.4228 | 0.4517 | 90 |
| Ti50Ni45Cu7.5 [46] | −0.27 | 0.3030 | 0.2907 | 0.4170 | 0.4596 | 95.5 |
| Ti50Ni40Cu10 [46] | −0.36 | 0.3031 | 0.2901 | 0.4249 | 0.4515 | 90 |
| Ti50Ni40Cu10 [46] | −1.44 | 0.3031 | 0.2872 | 0.4192 | 0.4577 | 95.2 |
| Ti50Ni37.5Cu12.5 [46] | −0.14 | 0.3034 | 0.2896 | 0.4252 | 0.4514 | 90 |
| Ti50Ni35Cu15 [46] | −0.44 | 0.3043 | 0.2899 | 0.4260 | 0.4516 | 90 |
| Ti50Ni30Cu20 [46] | −0.53 | 0.3046 | 0.2900 | 0.4264 | 0.4512 | 90 |
| Ti50Ni41Pd9 [47] | −0.78 | 0.3047 | 0.2846 | 0.4304 | 0.4583 | 90 |
| Ti50Ni40Pd11 [47] | −0.93 | 0.3050 | 0.2830 | 0.4314 | 0.4604 | 90 |
| Ti50Ni32Pd18 [47] | −0.68 | 0.3056 | 0.2820 | 0.4343 | 0.4628 | 90 |
| Ti50Ni30Pd20 [47] | −0.58 | 0.3051 | 0.2820 | 0.4340 | 0.4613 | 90 |
| Ti50Ni25Pd25 [47] | −0.53 | 0.3063 | 0.2807 | 0.4361 | 0.4667 | 90 |
| Ti50Au50 [48] | −0.11 | 0.3220 | 0.294 | 0.463 | 0.490 | 90 |
| Alloy, at.% | a, nm | b, nm | c, nm | β, ° |
|---|---|---|---|---|
| Ti50Ni5Pd45 | 0.2803 | 0.454 | 0.4794 | 90 |
| Ti50Ni48Pt2 | 0.2892 | 0.4135 | 0.4643 | 96.6 |
| Ti50Ni42Pt8 | 0.2821 | 0.4292 | 0.4585 | 90 |
| Ti50Ni42Pt8 | 0.2873 | 0.4211 | 0.4627 | 95.6 |
| Ti50Ni25Pt25 | 0.2765 | 0.4483 | 0.4744 | 90 |
| Ti50Ni47Au3 | 0.2907 | 0.4136 | 0.4653 | 96.6 |
| Ti50Ni45Au5 | 0.2907 | 0.4159 | 0.4666 | 96.5 |
| Ti50Ni38Au12 | 0.2876 | 0.4319 | 0.46203 | 90 |
| Ti50Ni25Au25 | 0.2864 | 0.4493 | 0.47492 | 90 |
| Ni50Ti47Zr3 | 0.292 | 0.4114 | 0.467 | 97.8 |
| Ni50Ti45Zr5 | 0.293 | 0.411 | 0.472 | 98.5 |
| Ni50Ti40Zr10 | 0.298 | 0.410 | 0.478 | 100.3 |
| Ni50Ti35Zr15 | 0.303 | 0.409 | 0.487 | 101.8 |
| Ni50Ti32Zr18 | 0.305 | 0.408 | 0.492 | 102.5 |
| Ni50Ti30Zr20 | 0.307 | 0.408 | 0.495 | 103.7 |
| Ni50Ti38Hf12 | 0.3006 | 0.4116 | 0.4803 | 101.2 |
| Ni50Ti35Hf15 | 0.3025 | 0.4096 | 0.4826 | 102 |
| Ni50Ti32Hf18 | 0.3051 | 0.4090 | 0.4850 | 102.5 |
| Ni50Ti30Hf20 | 0.3063 | 0.4083 | 0.4890 | 103 |
| Ni50Mn25Ga25 | 0.555 | 0.555 | 0.670 | 90 |
| Ni53Mn22Ga25 | 0.4142 | 0.556 | 2.953 | 90 |
| Ni54Mn21Ga25 | 0.4232 | 0.550 | 2.937 | 93.3 |
| Ni54.75Mn20.25Ga25 | 0.549 | 0.549 | 0.6478 | 90 |
| Ni55.25Mn19.75Ga25 | 0.548 | 0.548 | 0.653 | 90 |
| Ni56.75Mn18.25Ga25 | 0.546 | 0.546 | 0.653 | 90 |
| Ni57.5Mn17.5Ga25 | 0.542 | 0.542 | 0.660 | 90 |
| Ni58.25Mn16.75Ga25 | 0.543 | 0.543 | 0.663 | 90 |
| Ni59Mn16Ga25 | 0.543 | 0.543 | 0.663 | 90 |
| Ni59.75Mn15.25Ga25 | 0.543 | 0.543 | 0.663 | 90 |
| Alloy, at.% | σm, MPa | σy, MPa | σu, MPa | σr, MPa | δ, % | er, % |
|---|---|---|---|---|---|---|
| Ti50Ni50 | 200 | 850 | 1200 | 650 | 40 | 6 |
| Ti50Ni48Cu2 | 150 | 670 | 850 | 520 | 20 | 5 |
| Ti50Ni45Cu5 | 120 | 580 | 750 | 460 | 18 | 4 |
| Ti50Ni40Cu10 | 100 | 530 | 750 | 430 | 25 | 3 |
| Ti50Ni35Cu15 | 70 | 490 | 720 | 420 | 20 | 3 |
| Ti50Ni30Cu20 | 60 | 500 | 720 | 440 | 20 | 3 |
| Ti50Ni25Cu25 | 60 | 500 | 740 | 440 | 20 | 3 |
| Ti50Ni49Fe1 | 200 | 720 | 1000 | 520 | 24 | 5 |
| Ti50Ni47Fe3 | 650 | 950 | 18 | |||
| Ti50Ni49Co1 | 120 | 740 | 1100 | 620 | 18 | 5 |
| Ti50Ni48Co2 | 160 | 740 | 1100 | 580 | 18 | 4 |
| Ti50Ni47Co3 | 240 | 710 | 1100 | 470 | 16 | 3 |
| Ti50Ni45Co5 | 650 | 1090 | 15 | |||
| Ti50Ni43Co7 | 630 | 1080 | 14 | |||
| Ti50Ni10Pd40 | 380 | 850 | 1010 | 470 | 11 | 5 |
| Alloy, at.% | <d>, m | σm, MPa | σy, MPa | σu, MPa | σr, MPa | δ, % | εr, % |
|---|---|---|---|---|---|---|---|
| Ti49.4Ni50.6 | 60 | 130 | 630 | 1600 | 500 | 75 | 5 |
| Ti49.4Ni50.6 (1) | 0.40 | 250 | 1200 | 1600 | 950 | 60 | 8 |
| Ti49.4Ni50.6 (2) | 0.03 | 460 | 1800 | 2100 | 1340 | 16 | 10 |
| Ti49.4Ni50.6 (2) | 0.05 | 370 | 1500 | 1670 | 1130 | 23 | 13 |
| Ti49.4Ni50.6 (2) | 0.10 | 200 | 1120 | 1250 | 920 | 30 | 16 |
| Ti49.4Ni50.6 (2) | 0.60 | 250 | 900 | 1210 | 650 | 75 | 16 |
| Alloy, at.% | <d>, m | σm, MPa | σy, MPa | σu, MPa | σr, MPa | δ, % | εr, % |
|---|---|---|---|---|---|---|---|
| Ti50Ni25Cu25 | 1.0 | 60 | 680 | 850 | 620 | 12 | 5 |
| Ti50Ni25Cu25 | 0.8 | 70 | 720 | 900 | 650 | 12 | 4 |
| Ti50Ni25Cu25 | 0.5 | 80 | 1070 | 1200 | 990 | 11 | 4 |
| Ti50.5Ni25Cu24.5 | 0.3 | 80 | 1050 | 1170 | 970 | 9 | 3 |
| Ti50.5Ni24.5Cu25 | 0.3 | 90 | 1150 | 1380 | 1060 | 10 | 3 |
| Ti49.5Ni25Cu25.5 | 0.3 | 80 | 950 | 1120 | 870 | 10 | 3 |
| Ti49Ni25Cu26 | 0.2 | 90 | 1150 | 1380 | 1060 | 10 | 3 |
| Ti51Ni24Cu25 | 0.2 | 100 | 1160 | 1300 | 1060 | 9 | 3 |
| Ti51Ni25Cu24 | 0.2 | 90 | 1200 | 1550 | 1110 | 10 | 3 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pushin, V.; Kuranova, N.; Marchenkova, E.; Pushin, A. Design and Development of Ti–Ni, Ni–Mn–Ga and Cu–Al–Ni-based Alloys with High and Low Temperature Shape Memory Effects. Materials 2019, 12, 2616. https://doi.org/10.3390/ma12162616
Pushin V, Kuranova N, Marchenkova E, Pushin A. Design and Development of Ti–Ni, Ni–Mn–Ga and Cu–Al–Ni-based Alloys with High and Low Temperature Shape Memory Effects. Materials. 2019; 12(16):2616. https://doi.org/10.3390/ma12162616
Chicago/Turabian StylePushin, Vladimir, Nataliya Kuranova, Elena Marchenkova, and Artemy Pushin. 2019. "Design and Development of Ti–Ni, Ni–Mn–Ga and Cu–Al–Ni-based Alloys with High and Low Temperature Shape Memory Effects" Materials 12, no. 16: 2616. https://doi.org/10.3390/ma12162616
APA StylePushin, V., Kuranova, N., Marchenkova, E., & Pushin, A. (2019). Design and Development of Ti–Ni, Ni–Mn–Ga and Cu–Al–Ni-based Alloys with High and Low Temperature Shape Memory Effects. Materials, 12(16), 2616. https://doi.org/10.3390/ma12162616




