Toothpaste Composition Effect on Enamel Chromatic and Morphological Characteristics: In Vitro Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Selection
- Healthy patients (n = 15) aged from 14 to 16 years;
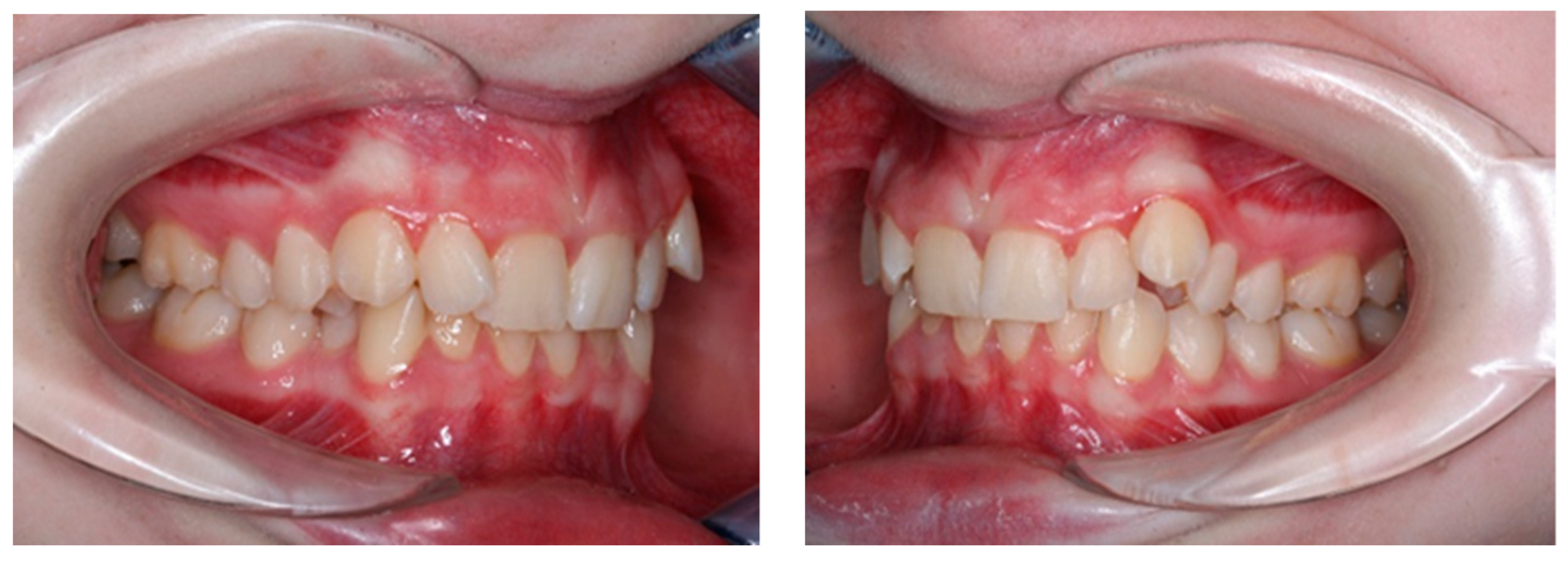
- The patients present malocclusions with arch-size/tooth-size discrepancies, that have to be solved by premolar extractions (first or second premolars) and a normodivergent skeletal pattern (Figure 1);
- Patients must have at least 2 premolars indicated for extraction for orthodontic treatment;
- The teeth were caries free, without stains, fissures, cracks, irregularities, abnormalities hypoplasia or filling observed at inspection, in standard condition. Twenty-five extracted teeth respect these criteria;
- We selected maxillary upper premolars, because these teeth are the most lightened from the lateral group;
- We instructed patients, prior to orthodontic treatment, to initiate toothbrushing with upper arch (vestibular surfaces). Topographical positions of upper teeth allow us to presume limited effect of salivary fluid when compared to lower teeth.
2.2. Sample Preservation and Preparation
2.3. Products Employed and Brushing Protocol
2.3.1. Treatment 1 (T1)
2.3.2. Treatment 2 (T2)
2.3.3. Treatment 3 (T3)
2.4. Colour Evaluation
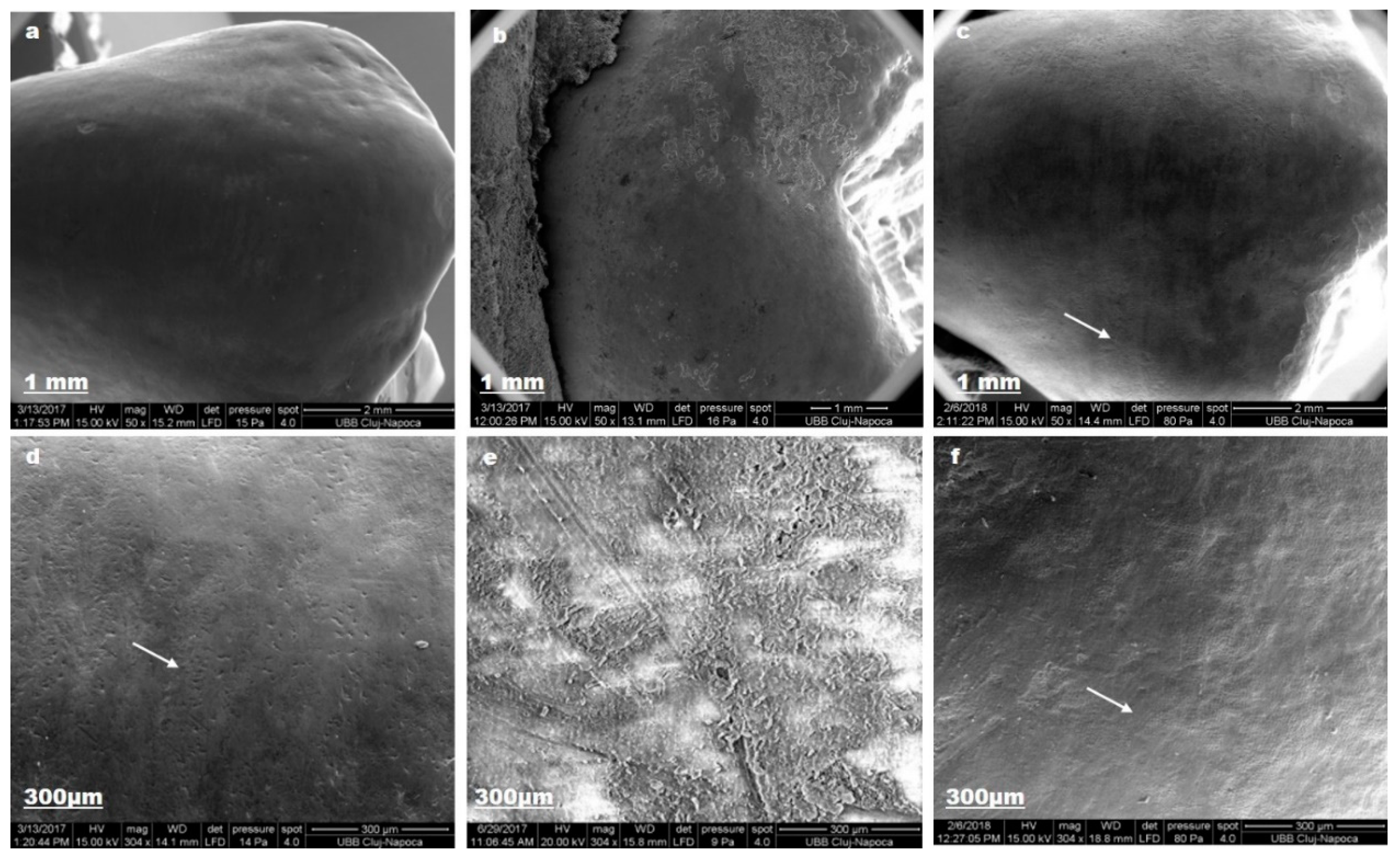
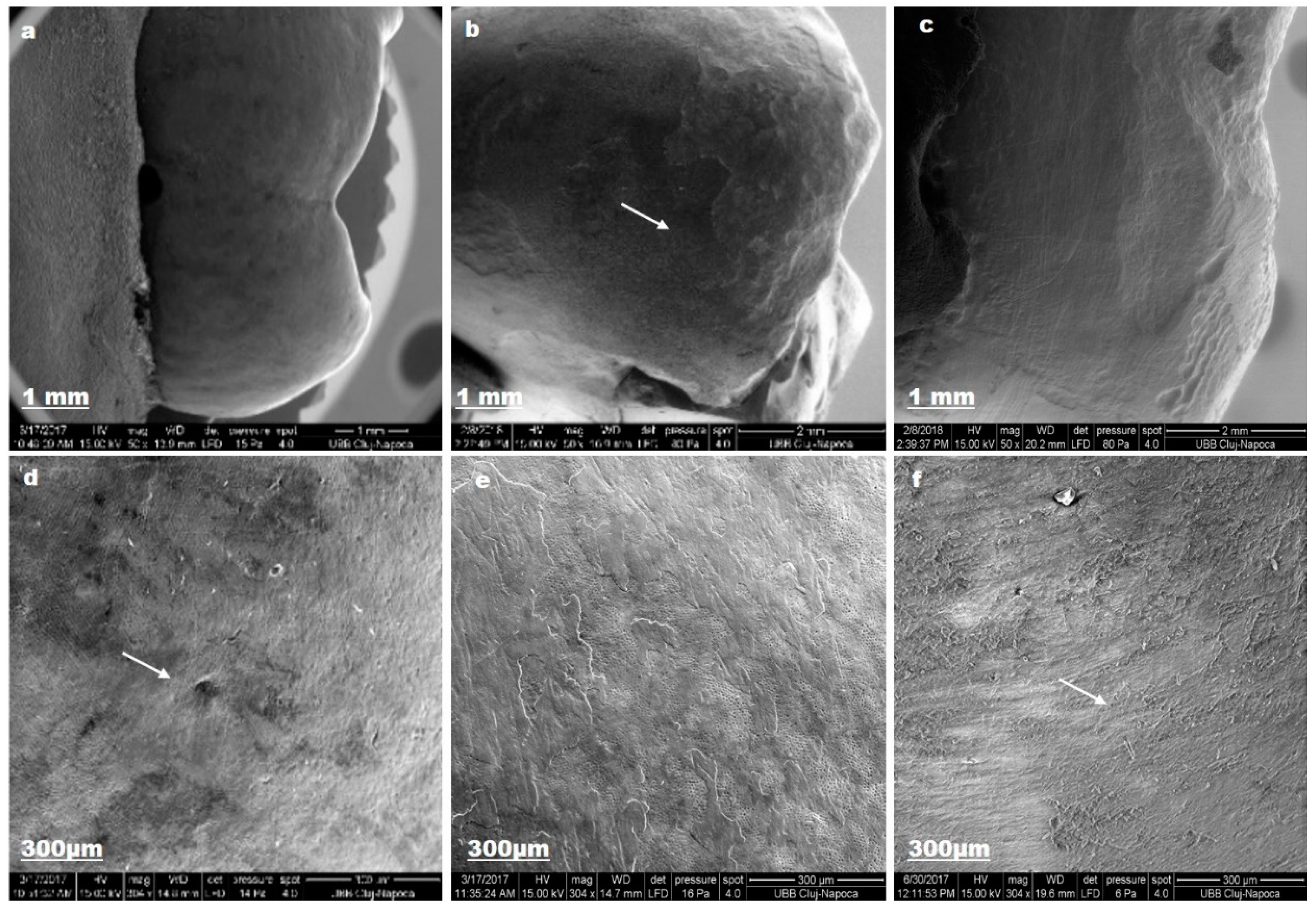
2.5. Morphological Characterization
2.6. Statistical Analysis
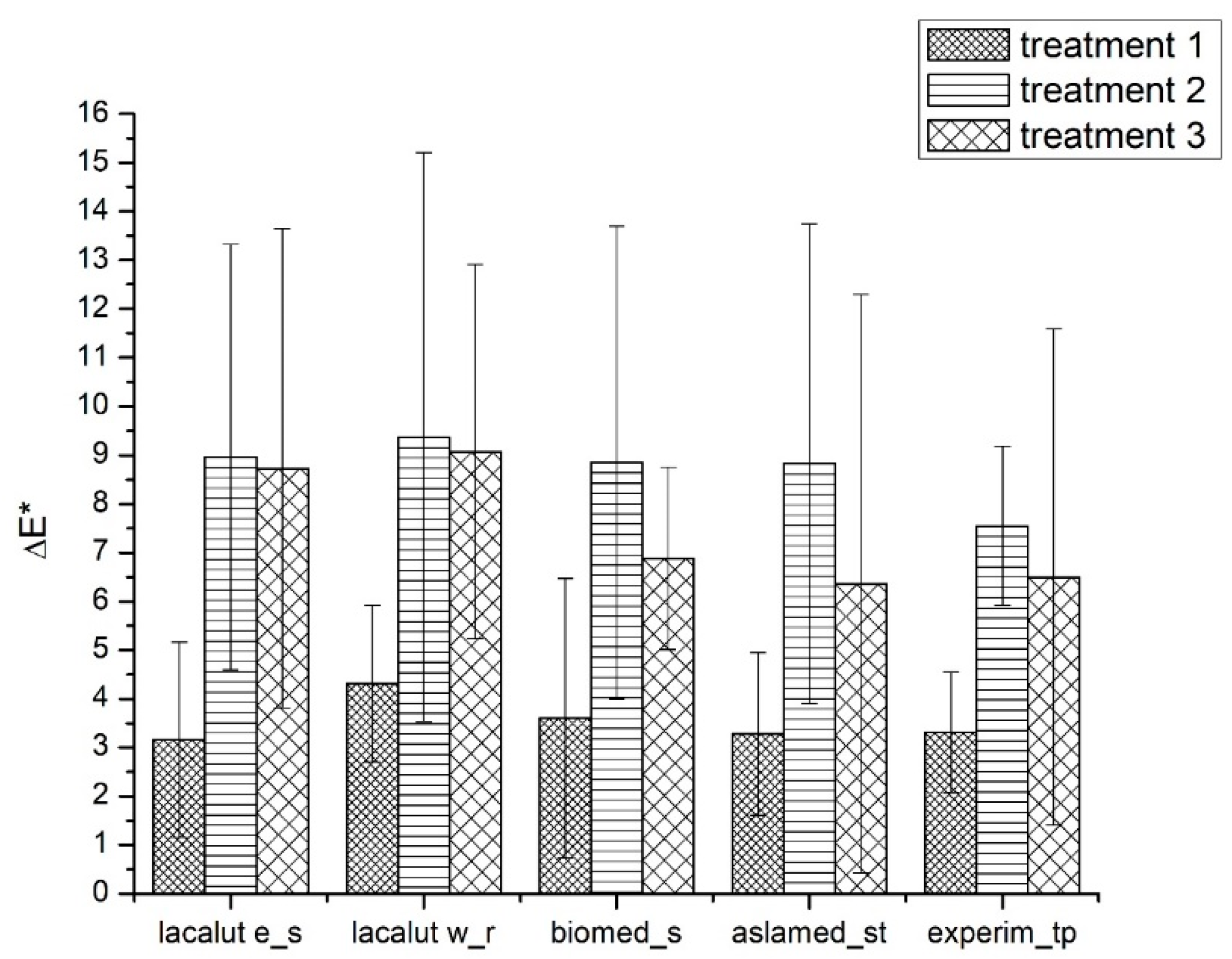
3. Results
3.1. Toothpaste Composition and Tooth Brushing Effect on Enamel Colour
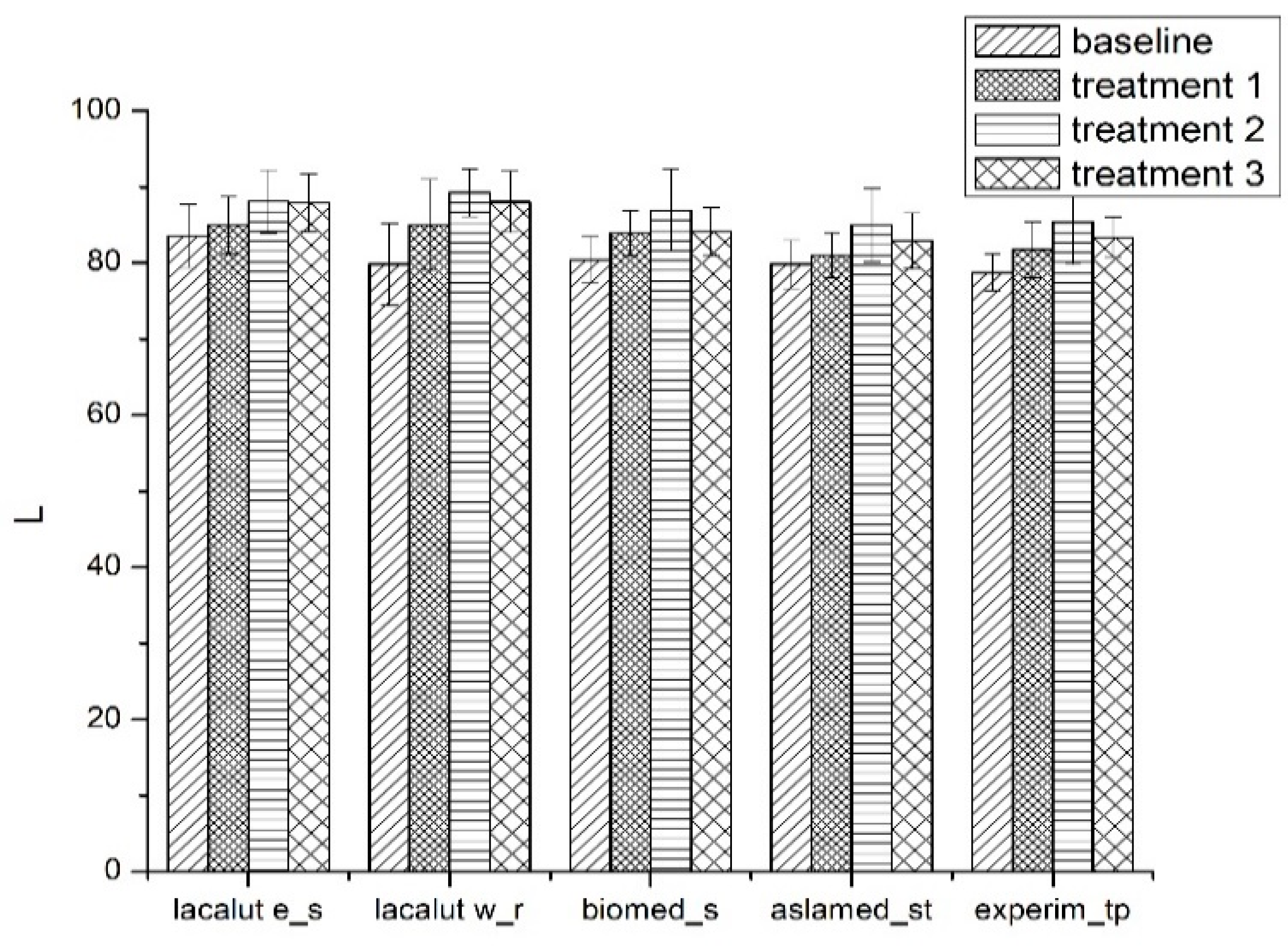
3.1.1. Luminosity (L* and ΔL*)
- The L* value increased after the first brushing protocol T1;
- Significant differences for L* parameter between toothpastes at T0 and T1 (p < 0.05) for lacalut_es, lacalut w_r and biomed_s;
- After demineralization (T2) we notice significant changes for L* parameter between toothpastes (p < 0.05);
- After demineralization and brushing (T3) L* parameter express values less important compared to T2, but the changes are noticeable when compared to T1.
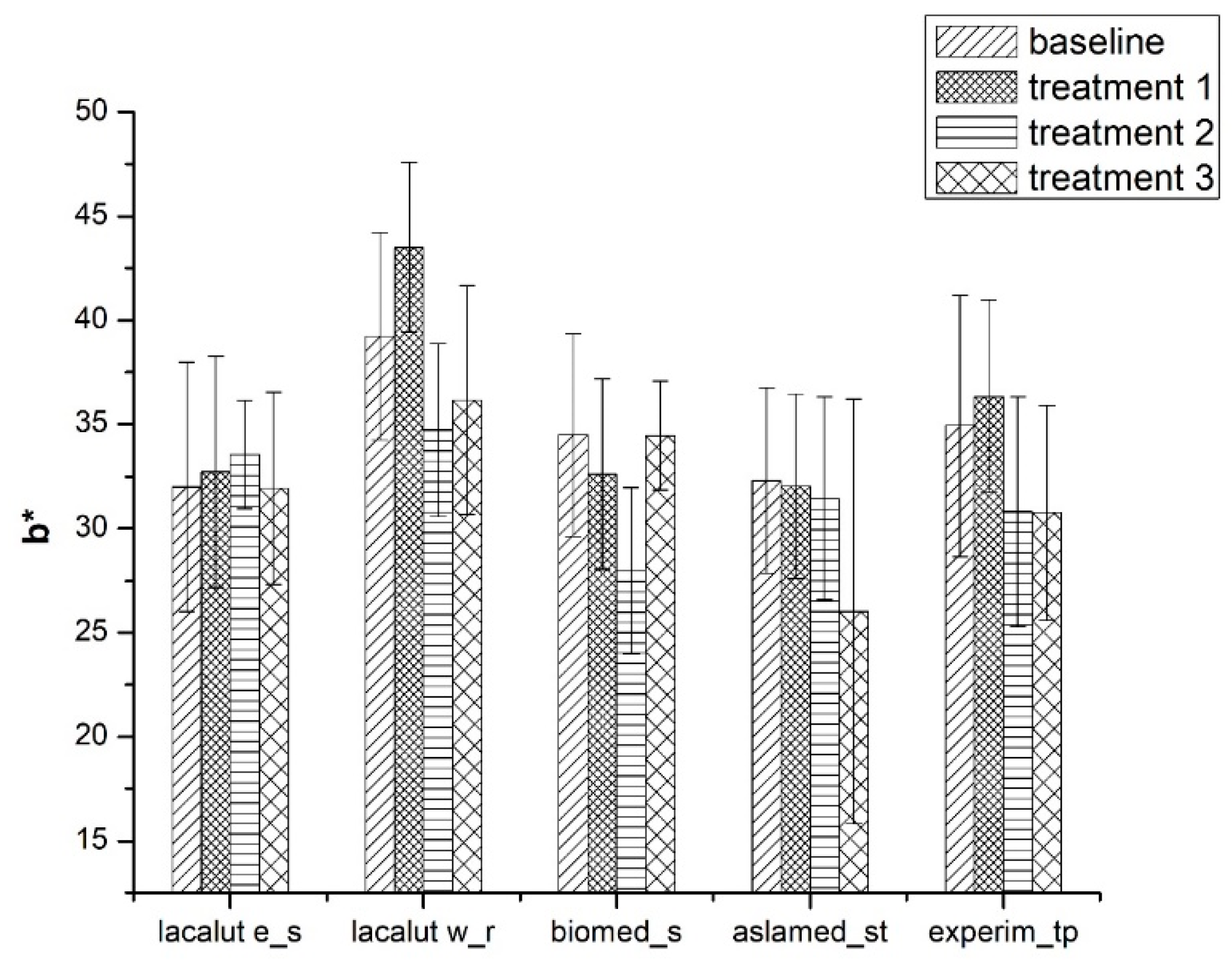
3.1.2. b* Parameter
- b* parameter present significant differences for the duration of the study, between toothpastes (p < 0.05);
- After T1 significant differences for b*parameter were noticed for lacalut_wr and experim_tp (p < 0.05);
- Biomed_s values for b* were significantly different (T2-T1 and T3-T1) (p < 0.05).
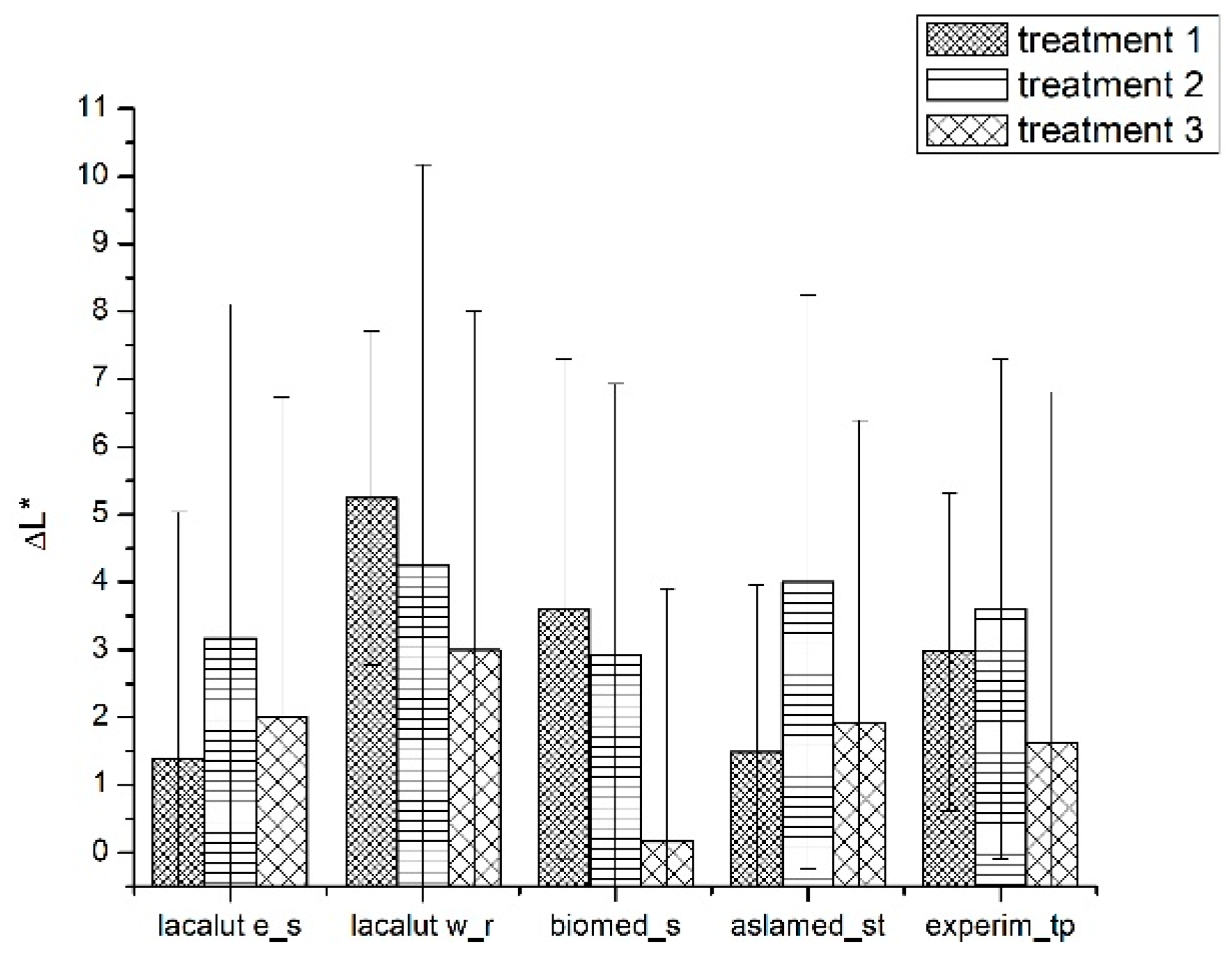
3.1.3. ΔE* Parameter
- After demineralization ΔE* values appear higher than 3 for all evaluated toothpastes, and the differences were statistically significant (T2-T1) (p < 0.05);
- After demineralization and brushing, ΔE* values tend to diminish, but the differences remain clinically acceptable and statistically significant (p < 0.05);
- Lacalut w_r differences were higher and statistically significant when compared to lacalut_s and biomed_s.
3.2. Toothpaste Composition Effect on Enamel Structure
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Burkland, G. Hygiene and the orthodontic patient. J. Clin. Orthod. 1999, 33, 443–446. [Google Scholar] [PubMed]
- Lee, S.M.; Yoo, K.H.; Yoon, S.Y.; Kim, I.R.; Park, B.S.; Son, W.S.; Ko, C.C.; Son, S.A.; Kim, Y.I. Enamel anti-demineralization efect of orthodontic adhesive containing bioactive glass and graphene oxide: An in-vitro study. Materials 2018, 11, 1728. [Google Scholar] [CrossRef] [PubMed]
- Loe, H. Oral hygiene in the prevention of caries and periodontal disease. Int. Dent. J. 2000, 50, 129–139. [Google Scholar] [CrossRef] [PubMed]
- Shellis, R.P.; Finke, M.; Eisenburger, M.; Parker, D.M.; Addy, M. Relationship between enamel erosion and liquid flow rate. Eur. J. Oral Sci. 2005, 113, 232–238. [Google Scholar] [CrossRef] [PubMed]
- Grewal, N.; Sharma, N.; Kaur, N. Surface remineralization potential of nano-hydroxyapatite, sodium monofluorophosphate, and amine fluoride containing dentifrices on primary and permanent enamel surfaces: An in vitrostudy. J. Indian Soc. Pedod. Prev. Dent. 2018, 36, 158–166. [Google Scholar] [CrossRef] [PubMed]
- Rao, A.; Malhotra, N. The role of remineralizing agents in dentistry—A review. J. Am. Dent. Assoc. 2011, 32, 26–33. [Google Scholar]
- Joiner, A. Whitening toothpastes: A review of the literature. J. Dent. 2010, 38, e17–e24. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-H.; Kim, S.-H.; Han, J.-S.; Yeo, I.-S.L.; Yoon, H.-I. Optical and Surface Properties of Monolithic Zirconia after Simulated Toothbrushing. Materials 2019, 12, 1158. [Google Scholar] [CrossRef]
- Goldberg, M.; Grootveld, M.; Lynch, E. Undesirable and adverse effects of tooth-whitening products: A review. Clin. Oral Investig. 2010, 14, 1–10. [Google Scholar] [CrossRef]
- Lee, W.K.; Lu, H.; Oguri, M.; Powers, J.M. Changes in color and staining of dental composite resins after wear simulation. J. Biomed. Mater. Res. Part B Appl. Biomater. 2007, 82, 313–319. [Google Scholar] [CrossRef]
- International Commission on Illumination. Colorimetry: Official Recommendations of the International Commission on Illumination, 2nd ed.; Bureau Central de la CIE: Vienna, Austria, 1986. [Google Scholar]
- Paravina, R.D.; Ghinea, R.; Herrera, L.J.; Bona, A.D.; Igiel, C.; Linninger, M.; Sakai, M.; Takahashi, H.; Tashkandi, E.; Perez, M.D.M. Color Difference Thresholds in Dentistry. J. Esthet. Restor. Dent. 2015, 27, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Kilts, T.; Verdelis, K.; Boskey, A.L.; Young, M.F. Variation in Mineral Properties in Normal and Mutant Bones and Teeth. Cells Tissues Organs 2005, 181, 144–153. [Google Scholar]
- Skucha-Nowak, M.; Gibas, M.; Tanasiewicz, M.; Twardawa, H.; Szklarski, T. Natural and controlled demineralization for study purposes in minimally invasive dentistry. Adv. Clin. Exp. Med. 2015, 24, 891–898. [Google Scholar] [CrossRef] [PubMed]
- Schwendicke, F.; Diederich, C.; Paris, S. Restoration gaps needed to exceed a threshold size to impede sealed lesion arrest in vitro. J. Dent. 2016, 48, 77–80. [Google Scholar] [CrossRef] [PubMed]
- Twetman, S.; Axelsson, S.; Dahlgren, H.; Holm, A.; Källestål, C.; Lagerlöf, F.; Lingström, P.; Mejàre, I.; Nordenram, G.; Norlund, A.; et al. Caries-preventive effect of fluoride toothpaste: A systematic review. Acta Odontol. Scand. 2003, 61, 347–355. [Google Scholar] [CrossRef] [PubMed]
- Hannig, M.; Hannig, C. Nanomaterials in preventive dentistry. Nat. Nanotechnol. 2010, 5, 565–569. [Google Scholar] [CrossRef]
- Demarco, F.F.; Meireles, S.S.; Masotti, A.S. Over-the-counter whitening agents: A concise review. Braz. Oral Res. 2009, 23, 64–70. [Google Scholar] [CrossRef]
- Hattab, F.N.; Qudeimat, M.A.; al-Rimawi, H.S. Dental discoloration: An overview. J. Esthet. Dent. 1999, 11, 291–310. [Google Scholar] [CrossRef]
- Gabasso, S.P.; Pinto, C.F.; Cavalli, V.; Paes-Leme, A.F.; Giannini, M. Effect of fluoride-containing bleaching agents on bovine enamel microhardness. Braz. J. Oral Sci. 2011, 10, 22–26. [Google Scholar]
- Parry, J.; Harrington, E.; Rees, G.D.; McNab, R.; Smith, A.J. Control of brushing variables for the in vitro assessment of toothpaste abrasivity using s novel laboratory model. J. Dent. 2008, 36, 117–124. [Google Scholar] [CrossRef]
- Firoozmand, L.M.; Brandao, J.V.; Fialho, M.P. Influence of mi-crohybrid resin and etching times on bleached enamel for the bonding of ceramic brackets. Braz. Oral Res. 2013, 27, 142–148. [Google Scholar] [CrossRef] [PubMed]
- Al Maaitah, E.F.; Abu Omar, A.A.; Al-Khateeb, S.N. Effect of fixed orthodontic appliances bonded with different etching techniques on tooth color: A prospective clinical study. Am. J. Orthod. Dentofac. Orthop. 2013, 144, 43–49. [Google Scholar] [CrossRef] [PubMed]
- Watts, A.; Addy, M. Tooth discolouration and staining: A review of the literature. Br. Dent. J. 2001, 190, 309–316. [Google Scholar] [CrossRef] [PubMed]
- Borges, A.; Santos, L.; Augusto, M.; Bonfiette, D.; Hara, A.; Torres, C. Toothbrushing abrasion susceptibility of enamel and dentin bleached with calcium-supplemented hydrogen peroxide gel. J. Dent. 2016, 49, 54–59. [Google Scholar] [CrossRef] [PubMed]
- Campos, E.J.; Silva, L.; De Arajo, D.B.; Silva, L.R.; Campos, E.D.J. In vitro study on tooth enamel lesions related to whitening dentifrice. Indian J. Dent. Res. 2011, 22, 770–776. [Google Scholar]
- Odioso, L.; Gibb, R.D.; Gerlach, R.W. Impact of demographic, behavioural, and dental care utilization parameters on tooth colour and personal satisfaction. Compend. Contin. Educ. Dent. 2000, 21, 35–41. [Google Scholar]
- Hilgenberg, S.P.; Pinto, S.C.S.; Farago, P.V.; Santos, F.A.; Wambier, D.S. Physical-chemical characteristics of whitening toothpaste and evaluation of its effects on enamel roughness. Braz. Oral Res. 2011, 25, 288–294. [Google Scholar] [CrossRef] [PubMed]
- Nam, S.N.; Kwun, H.S.; Cheon, S.H.; Kim, H.Y. Effects of whitening toothpaste on color change and mineral contents of dental hard tissues. Biomed. Res. 2017, 28, 3832–3836. [Google Scholar]
- Torres, C.; Perote, L.; Gutierrez, N.; Pucci, C.R.; Borges, A. Efficacy of Mouth Rinses and Toothpaste on Tooth Whitening. Oper. Dent. 2013, 38, 57–62. [Google Scholar] [CrossRef] [PubMed]
- Joiner, A. Review of the extrinsic stain removal and enamel/dentine abrasion by a calcium carbonate and perlite containing whitening toothpaste. Int. Dent. J. 2006, 56, 175–180. [Google Scholar] [CrossRef] [PubMed]
- Yu, O.Y.; Zhao, I.S.; Mei, M.L.; Lo, E.C.-M.; Chu, C.-H. A Review of the Common Models Used in Mechanistic Studies on Demineralization-Remineralization for Cariology Research. J. Dent. 2017, 5, 20. [Google Scholar] [CrossRef] [PubMed]
- Eimar, H.; Marelli, B.; Nazhat, S.N.; Nader, S.A.; Amin, W.M.; Torres, J.; De Albuquerque, R.F.; Taimi, F. The role of enamel crystallography on tooth shade. J. Dent. 2011, 39, e3–e10. [Google Scholar] [CrossRef] [PubMed]
- Novais, R.C.; Toledo, O.A. In vitro study of dental enamel alterations after exposing to a bleaching agent. J. Bras. Clin. Estet. Odontol. 2000, 4, 48–51. [Google Scholar]
- Sharif, N.; Macdonald, E.; Hughes, J.; Newcombe, R.; Addy, M. The chemical stain removal properties of ’whitening’ toothpaste products: Studies in vitro. Br. Dent. J. 2000, 188, 620–624. [Google Scholar] [CrossRef] [PubMed]
- Moran, J.; Claydon, N.C.A.; Addy, M.; Newcombe, R. Clinical studies to determine the effectiveness of a whitening toothpaste at reducing stain (using a forced stain model). Int. J. Dent. Hyg. 2005, 3, 25–30. [Google Scholar] [CrossRef] [PubMed]
- Walsh, T.F.; Rawlinson, A.; Wildgoose, D.; Marlow, I.; Haywood, J.; Ward, J.M. Clinical evaluation of the stain removing ability of a whitening dentifrice and stain controlling system. J. Dent. 2005, 33, 413–418. [Google Scholar] [CrossRef]
- D’Amario, M.; D’Attilio, M.; Baldi, M.; Deangelis, F.; Marzo, G.; Vadini, M.; Varvara, G.; D’Arcangelo, C. Histomorphologic alterations of human enamel after repeated applications of a bleaching agent. Int. J. Immunopath. Pharmacol. 2012, 4, 1021–1027. [Google Scholar] [CrossRef]
- Yu, O.Y.; Mei, M.L.; Zhao, I.S.; Lo, E.C.-M.; Chu, C.-H. Effects of Fluoride on Two Chemical Models of Enamel Demineralization. Materials 2017, 10, 1245. [Google Scholar] [CrossRef]
- Muntean, A.; Mesaros, A.S.; Porumb, A.; Cuc, S.; Moldovan, M.; Balan, A. Enamel Appearance after Orthodontic Attachment Removal In vitro SEM analysis. Rev. Chim. 2017, 68, 125–127. [Google Scholar]
- Antoniac, I.; Sinescu, C.; Antoniac, A. Adhesion aspects in biomaterials and medical devices. J. Adhes. Sci. Technol. 2016, 30, 1711–1715. [Google Scholar]
- Antoniac, I.; Stoia, D.I.; Ghiban, B.; Tecu, C.; Miculescu, F.; Vigaru, C.; Saceleanu, V. Failure analysis of a humeral shaft locking compression plate—Surface investigation and simulation by finite element method. Materials 2019, 12, 1128. [Google Scholar] [CrossRef] [PubMed]



| Toothpaste | Composition | Effect |
|---|---|---|
| Lacalut Extra Sensitive (lacalut e_s) | Sodium fluoride, Aluminium salts, Clorhexidine, KCl, silicium dioxide Sodium fluoride Amine | Potassium chloride: improvement of nerve cells Sodium fluoride: caries prevention |
| Lacalut White and Repair (lacalut w_r) | Hydrated Silica, hydroxyapatite, Pyrophosphate, SLS, Sodium Fluoride(1360 ppm), eugenol | Phosphates: bleach and remove from the surface of the tooth discoloration and sediment Sodium fluoride and hydroxyapatite: remineralisation of enamel |
| Biomed Sensitive (biomed_s) | L-Arginine, Hydroxyapatite, Natural component (Plantain extract, birch leaf polyphenols and red grape seeds) | Calcium hydroxyapatite: enamel strengthening and eliminating the causes of tooth sensitivity Natural component: dental plaque removal, protection against tooth decay, enamel strengthening |
| Aslamed for Sensitive Teeth (aslamed_st) | Sodium fluoride, special clay, potassium nitrate, SLS free | Potassium nitrate: clinically proven desensitising effects Special clay: remineralisation of teeth and strengthens their enamel, astringent effect Sodium fluoride: protects against tooth decay Chamomile extract: antimicrobial and anti-inflammatory effect |
| Experimental toothpaste (experim_tp) | Hydroxyapatite, special clay, potassium nitrate, SLS free | Hydroxyapatite: enamel remineralisation Special clay: remineralisation of teeth and strengthens their enamel, astringent effect |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Muntean, A.; Sava, S.; Delean, A.G.; Mihailescu, A.M.; Dumitrescu, L.S.; Moldovan, M.; Festila, D.G. Toothpaste Composition Effect on Enamel Chromatic and Morphological Characteristics: In Vitro Analysis. Materials 2019, 12, 2610. https://doi.org/10.3390/ma12162610
Muntean A, Sava S, Delean AG, Mihailescu AM, Dumitrescu LS, Moldovan M, Festila DG. Toothpaste Composition Effect on Enamel Chromatic and Morphological Characteristics: In Vitro Analysis. Materials. 2019; 12(16):2610. https://doi.org/10.3390/ma12162610
Chicago/Turabian StyleMuntean, Alexandrina, Sorina Sava, Ada Gabriela Delean, Ana Maria Mihailescu, Laura Silaghi Dumitrescu, Marioara Moldovan, and Dana Gabriela Festila. 2019. "Toothpaste Composition Effect on Enamel Chromatic and Morphological Characteristics: In Vitro Analysis" Materials 12, no. 16: 2610. https://doi.org/10.3390/ma12162610
APA StyleMuntean, A., Sava, S., Delean, A. G., Mihailescu, A. M., Dumitrescu, L. S., Moldovan, M., & Festila, D. G. (2019). Toothpaste Composition Effect on Enamel Chromatic and Morphological Characteristics: In Vitro Analysis. Materials, 12(16), 2610. https://doi.org/10.3390/ma12162610





