Thermal Cycling Effect on Transformation Temperatures of Different Transformation Sequences in TiNi-Based Shape Memory Alloys
Abstract
1. Introduction
2. Experimental Procedures
3. Results
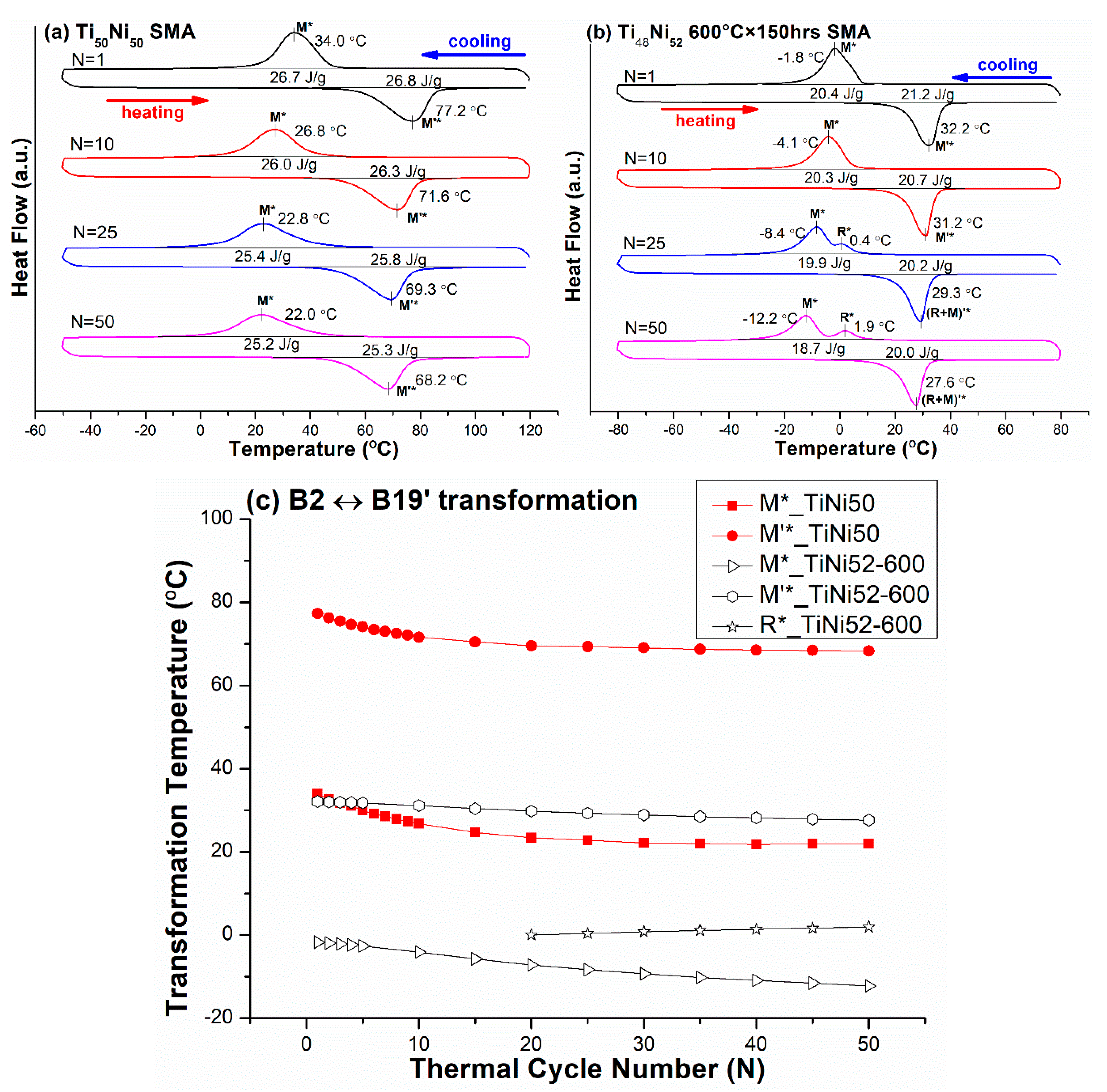
3.1. B2 ↔ B19′ One-Stage Transformation Sequence
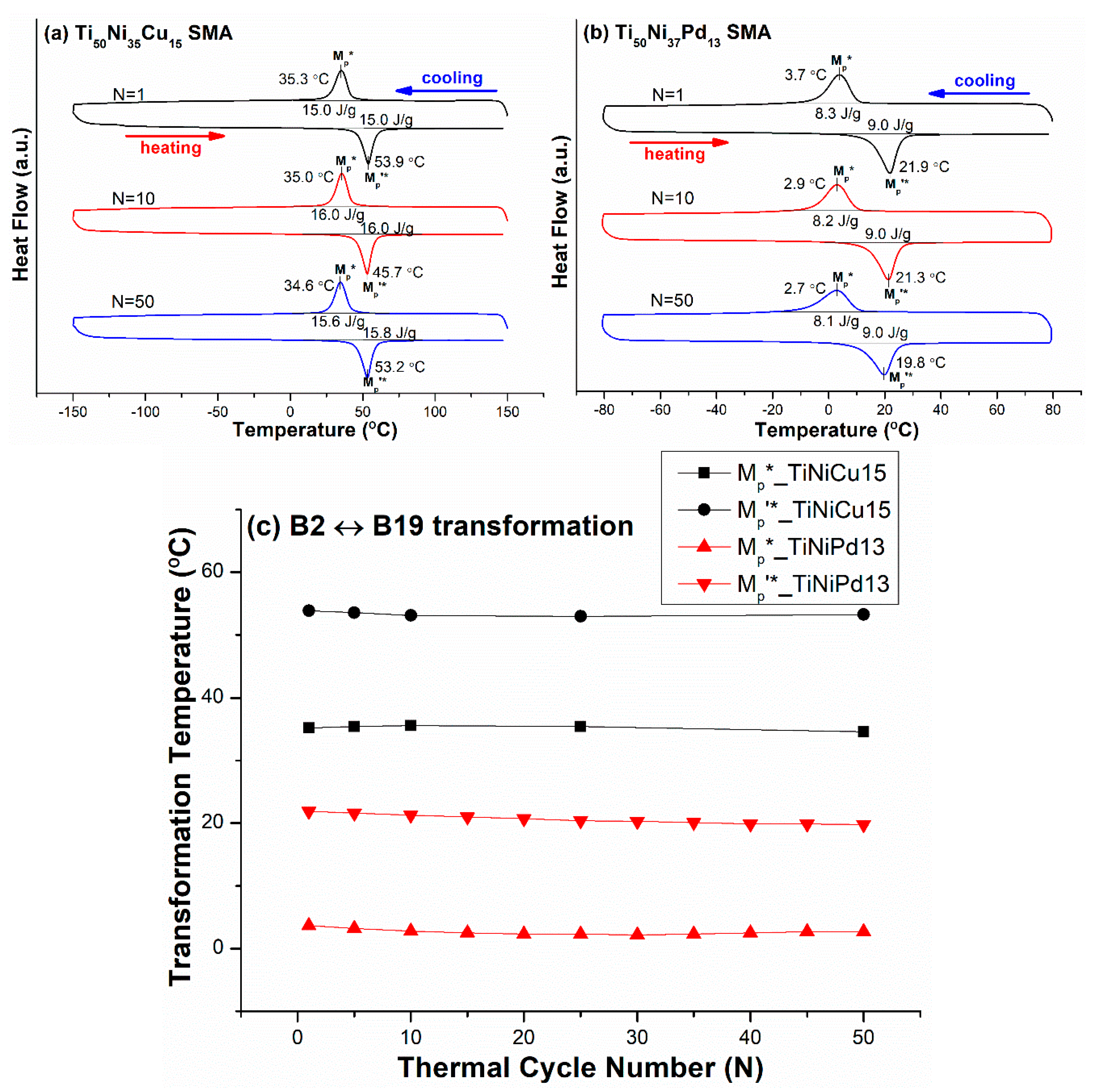
3.2. B2 ↔ R-Phase and B2 ↔ B19 One-Stage Transformation Sequences
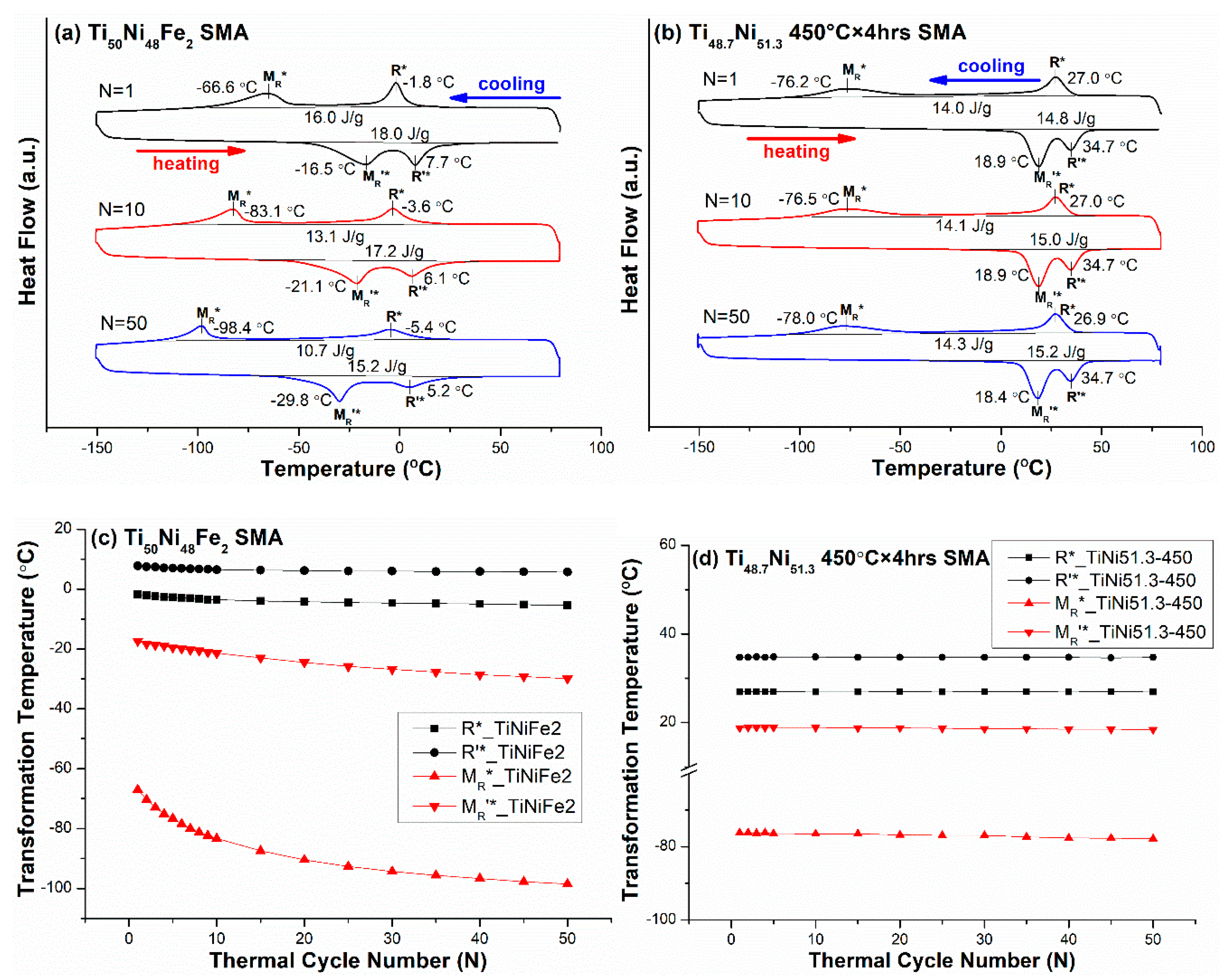
3.3. B2 ↔ R-Phase ↔ B19′ Two-Stage Transformation Sequence
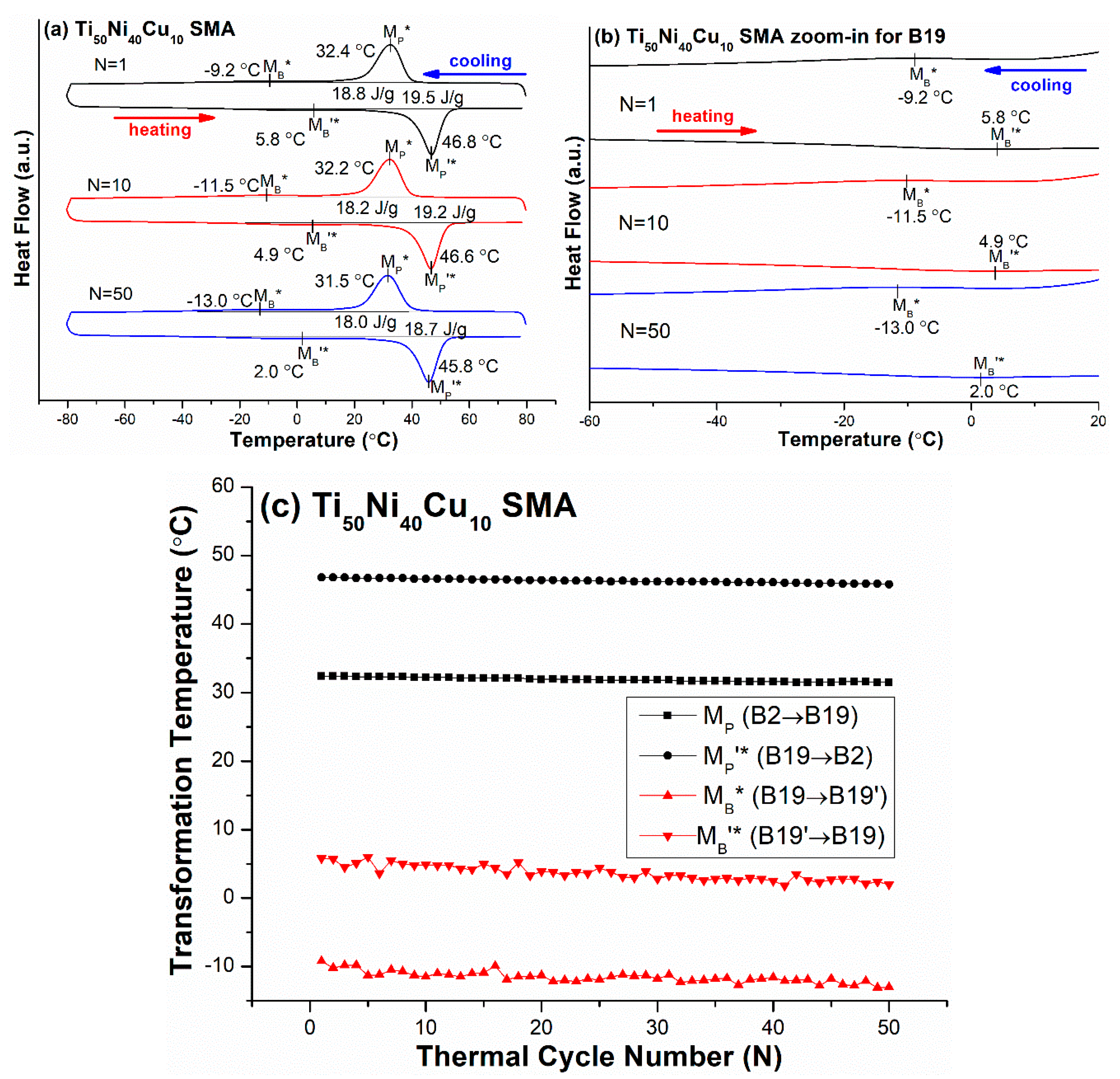
3.4. B2 ↔ B19 ↔B19′ Two-Stage Transformation Sequence
4. Discussion
4.1. Effect of the Hardness of the SMA
4.2. Effect of Shear Strain, s, Associated with Martensitic Transformation
4.3. Characteristics of B2 → R-Phase Transformation Temperature Affected by Thermal Cycling in B2 → R and B2 → R-phase → B19′ Transformations
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Wayman, K.O.A.C.M. Shape Memory Materials; Cambridge University Press: Cambridge, UK, 1998. [Google Scholar]
- Otsuka, K.; Ren, X. Physical metallurgy of Ti-Ni-based shape memory alloys. Prog. Mater. Sci. 2005, 50, 511–678. [Google Scholar] [CrossRef]
- Chang, S.H.; Wu, S.K. Inherent internal friction of B2 -> R and R -> B19 martensitic transformations in equiatomic TiNi shape memory alloy. Scr. Mater. 2006, 55, 311–314. [Google Scholar] [CrossRef]
- Wang, D.; Zhang, Z.; Zhang, J.A.; Zhou, Y.M.; Wang, Y.; Ding, X.D.; Wang, Y.Z.; Ren, X.B. Strain glass in Fe-doped Ti-Ni. Acta Mater. 2010, 58, 6206–6215. [Google Scholar] [CrossRef]
- Ramachandran, B.; Chang, P.C.; Kuo, Y.K.; Chien, C.; Wu, S.K. Characteristics of martensitic and strain-glass transitions of the Fe-substituted TiNi shape memory alloys probed by transport and thermal measurements. Sci. Rep. 2017, 7, 16336. [Google Scholar] [CrossRef]
- Teng, Y.Y.; Zhu, S.L.; Wang, F.H.; Wu, W.T. Electronic structures and shape-memory behavior of Ti50Ni50-xCux (x = 0, 6.25, 12.5, 18.75 and 25.0 at%) by density functional theory. Phys. B 2007, 393, 18–23. [Google Scholar] [CrossRef]
- Ramachandran, B.; Tang, R.C.; Chang, P.C.; Kuo, Y.K.; Chien, C.; Wu, S.K. Cu-substitution effect on thermoelectric properties of the TiNi-based shape memory alloys. J. Appl. Phys. 2013, 113, 203702. [Google Scholar] [CrossRef]
- Wu, S.K.; Wayman, C.M. Martensitic Transformations and the Shape Memory Effect in Ti50Ni10Au40 and Ti50Au50 Alloys. Metallography 1987, 20, 359–376. [Google Scholar] [CrossRef]
- Wu, S.K.; Wayman, C.M. TEM Studies of the Martensitic-Transformation in a Ti50Ni40Au10 Alloy. Scr. Met. Mater. 1987, 21, 83–88. [Google Scholar] [CrossRef]
- Lo, Y.C.; Wu, S.K.; Wayman, C.M. Transformation Heat as a Function of Ternary Pd Additions in Ti50Ni50-xPdx Alloys with x: 20-50 at-Percent. Scr. Met. Mater. 1990, 24, 1571–1576. [Google Scholar] [CrossRef]
- Hsieh, S.F.; Wu, S.K. Martensitic transformation of quaternary Ti50.5-xNi49.5Zrx/2Hfx/2 (X = 0-20 at.%) shape memory alloys. Mater. Charact. 2000, 45, 143–152. [Google Scholar] [CrossRef]
- Firstov, G.S.; Van Humbeeck, J.; Koval, Y. High-temperature shape memory alloys some recent developments. Mat. Sci. Eng. A 2004, 378, 2–10. [Google Scholar] [CrossRef]
- Van Humbeeck, J. Non-medical applications of shape memory alloys. Mat. Sci. Eng. A 1999, 273, 134–148. [Google Scholar] [CrossRef]
- Jani, J.M.; Leary, M.; Subic, A.; Gibson, M.A. A review of shape memory alloy research, applications and opportunities. Mater. Des. 2014, 56, 1078–1113. [Google Scholar] [CrossRef]
- Bonnot, E.; Romero, R.; Manosa, L.; Vives, E.; Planes, A. Elastocaloric effect associated with the martensitic transition in shape-memory alloys. Phys. Rev. Lett. 2008, 100, 125901. [Google Scholar] [CrossRef]
- Miyazaki, S.; Igo, Y.; Otsuka, K. Effect of Thermal Cycling on the Transformation Temperatures of Ti-Ni Alloys. Acta Met. Mater. 1986, 34, 2045–2051. [Google Scholar] [CrossRef]
- Liu, Y.; Mccormick, P.G. Thermodynamic Analysis of the Martensitic-Transformation in NiTi .1. Effect of Heat-Treatment on Transformation Behavior. Acta Met. Mater. 1994, 42, 2401–2406. [Google Scholar] [CrossRef]
- Mccormick, P.G.; Liu, Y.N. Thermodynamic Analysis of the Martensitic-Transformation in NiTi .2. Effect of Transformation Cycling. Acta Met. Mater. 1994, 42, 2407–2413. [Google Scholar] [CrossRef]
- Zarnetta, R.; Takahashi, R.; Young, M.L.; Savan, A.; Furuya, Y.; Thienhaus, S.; Maass, B.; Rahim, M.; Frenzel, J.; Brunken, H.; et al. Identification of Quaternary Shape Memory Alloys with Near-Zero Thermal Hysteresis and Unprecedented Functional Stability. Adv. Funct. Mater. 2010, 20, 1917–1923. [Google Scholar] [CrossRef]
- Wagner, M.F.X.; Dey, S.R.; Gugel, H.; Frenzel, J.; Somsen, C.; Eggeler, G. Effect of low-temperature precipitation on the transformation characteristics of Ni-rich NiTi shape memory alloys during thermal cycling. Intermetallics 2010, 18, 1172–1179. [Google Scholar] [CrossRef]
- Meng, X.L.; Li, H.; Cai, W.; Hao, S.J.; Cui, L.S. Thermal cycling stability mechanism of Ti50.5Ni33.5Cu11.5Pd4.5 shape memory alloy with near-zero hysteresis. Scr. Mater. 2015, 103, 30–33. [Google Scholar] [CrossRef]
- Wang, X.B.; Van Humbeeck, J.; Verlinden, B.; Kustov, S. Thermal cycling induced room temperature aging effect in Ni-rich NiTi shape memory alloy. Scr. Mater. 2016, 113, 206–208. [Google Scholar] [CrossRef]
- Perez-Sierra, A.M.; Pons, J.; Santamarta, R.; Karaman, I.; Noebe, R.D. Stability of a Ni-rich Ni-Ti-Zr high temperature shape memory alloy upon low temperature aging and thermal cycling. Scr. Mater. 2016, 124, 47–50. [Google Scholar] [CrossRef]
- Kuang, C.H.; Chien, C.; Wu, S.K. Multistage martensitic transformation in high temperature aged Ti48Ni52 shape memory alloy. Intermetallics 2015, 67, 12–18. [Google Scholar] [CrossRef]
- Chang, P.C.; Ko, M.L.; Ramachandran, B.; Kuo, Y.K.; Chien, C.; Wu, S.K. Comparative study of R-phase martensitic transformations in TiNi-based shape memory alloys induced by point defects and precipitates. Intermetallics 2017, 84, 130–135. [Google Scholar] [CrossRef]
- Lo, Y.C.; Wu, S.K. Compositional Dependence of Martensitic-Transformation Sequence in Ti50Ni50-xPdx Alloys with X-Less-Than-or-Equal-to-15at-Percent. Scr. Met. Mater. 1992, 26, 1875–1877. [Google Scholar] [CrossRef]
- Otsuka, K.; Shimizu, K. Pseudoelasticity and shape memory effects in alloys. Int. Met. Rev. 1986, 31, 93–114. [Google Scholar] [CrossRef]
- Knowles, K.M.; Smith, D.A. The Crystallography of the Martensitic-Transformation in Equiatomic Nickel-Titanium. Acta Met. Mater. 1981, 29, 101–110. [Google Scholar] [CrossRef]
- Onda, T.; Bando, Y.; Ohba, T.; Otsuka, K. Electron-Microscopy Study of Twins in Martensite in a Ti-50.0 at Percent Ni-Alloy. Mater. T Jim 1992, 33, 354–359. [Google Scholar] [CrossRef]
- Fukuda, T.; Saburi, T.; Doi, K.; Nenno, S. Nucleation and Self-Accommodation of the R-Phase in Ti-Ni Alloys. Mater. T Jim 1992, 33, 271–277. [Google Scholar] [CrossRef]
- Miyazaki, S.; Wayman, C.M. The R-Phase Transition and Associated Shape Memory Mechanism in Ti-Ni Single-Crystals. Acta Met. Mater. 1988, 36, 181–192. [Google Scholar] [CrossRef]
- Watanabe, Y.; Saburi, T.; Nakagawa, Y.; Nenno, S. Self-Accommodation Structure in the Ti-Ni-Cu Orthorhombic Martensite. J. JPN I. Met. 1990, 54, 861–869. [Google Scholar] [CrossRef][Green Version]
- Wu, S.K.; Lin, H.C. The Effect of Precipitation Hardening on the Ms Temperature in a Ti49.2Ni50.8 Alloy. Scr. Met. Mater. 1991, 25, 1529–1532. [Google Scholar] [CrossRef]

| SMAs (in at. %) | DSC Tests for Thermal Cycling at Tmin/Tmax (°C) | Transformation Sequence | Forward Transformation Peak Temperature at N = 1 (°C) |
|---|---|---|---|
| Ti50Ni50 (TiNi50) | −50/120 | B2 ↔ B19′ | 34.0 |
| Ti48Ni52 600 °C × 150 h (TiNi52-600) | −80/80 | B2 ↔ B19′ (N = 1–20) B2 → R → B19′ (N = 21–50) B19′ → B2 (N = 21–50) | −1.8 |
| Ti50Ni46Fe4 (TiNiFe4) | −120/60 | B2 ↔ R | −48.3 |
| TiNi51.3 350 °C × 24 h (TiNi51.3-350) | −80/80 | B2 ↔ R | 35.3 |
| Ti50Ni35Cu15 (TiNiCu15) | −150/150 | B2 ↔ B19 | 35.3 |
| Ti50Ni37Pd13 (TiNiPd13) | −80/80 | B2 ↔ B19 | 3.7 |
| Ti50Ni48Fe2 (TiNiFe2) | −150/80 | B2 ↔ R ↔ B19′ | R*: −1.8, M*: −66.6 |
| TiNi51.3 450 °C × 4 h (TiNi51.3-450) | −150/80 | B2 ↔ R ↔ B19′ | R*: 27.0, M*: −76.2 |
| Ti50Ni40Cu10 (TiNiCu10) | −80/80 | B2 ↔ B19 ↔ B19′ | MP*: 32.4, MB*: −9.2 |
| SMAs (in at. %) | Hardness (Hv) | Phase(s) of Matrix at Room Temperature |
|---|---|---|
| Ti50Ni50 (TiNi50) | 174 ± 6 | B2 and B19′ (with B19′ being the major phase) |
| Ti48Ni52 600 °C × 150 h (TiNi52-600) | 228 ± 7 | B2 and Ti2Ni3 ppts |
| Ti50Ni46Fe4 (TiNiFe4) | 196 ± 5 | only B2 |
| TiNi51.3 350 °C × 24 h (TiNi51.3-350) | 387 ± 8 | (B2 + R) and Ti3Ni4 ppts (with R being the major phase) |
| Ti50Ni35Cu15 (TiNiCu15) | 220 ± 9 | only B19 |
| Ti50Ni37Pd13 (TiNiPd13) | 177 ± 4 | only B2 |
| Ti50Ni48Fe2 (TiNiFe2) | 194 ± 7 | only B2 |
| TiNi51.3 450 °C × 4 h (TiNi51.3-450) | 335 ± 6 | (B2 + R) and Ti3Ni4 ppts (with R-phase being the major phase) |
| Ti50Ni40Cu10 (TiNiCu10) | 177 ± 10 | B2 and B19 (with B19 being the major phase) |
| Martensitic Transformation Type | The Associated Twinning Mode in Martensite | Magnitude of the Shear Strain, s |
|---|---|---|
| B2 ↔ B19′ martensitic transformation | <011 >M type II | 0.2804 [28] |
| (001)M/(100)M compound | 0.2348 [28,29] | |
| {}M type I | 0.30961 [28] | |
| B2 ↔ R premartensitic transformation | {1121}R i.e., {100}B2 | 0.0265 * [30,31] |
| {}R i.e., {011}B2 | 0.0265 * [30,31] | |
| B2 ↔ B19 premartensitic transformation | {111}M type I | 0.17 [32] |
| {011}M compound | 0.11 [32] |
| TiNi-Based SMAs | Transformation Type | s Value | The Decrease of the Forward Transformation Temperature from N = 1 to N = 50 (°C) |
|---|---|---|---|
| Ti50Ni50 (TiNi50) | B2 → B19′ | 0.2804 | −12.0 |
| Ti48Ni52 600 °C × 150 h (TiNi52-600) | B2 → B19′ | 0.2804 | −6.6 § |
| B2 → R | 0.0265 | +1.9 §§ | |
| R → B19′ | <0.2804 *,† | −3.8 §§ | |
| Ti50Ni46Fe4 (TiNiFe4) | B2 → R | 0.0265 | 0 |
| TiNi51.3 350 °C × 24h (TiNi51.3-350) | B2 → R | 0.0265 | 0 |
| Ti50Ni35Cu15 (TiNiCu15) | B2 → B19 {011}M compd. twin + {111}M type I twin | 0.11~0.17 | −0.7 |
| Ti50Ni37Pd13 (TiNiPd13) | B2 → B19 {111}M type I twin | 0.17 | −1.0 |
| Ti50Ni48Fe2 (TiNiFe2) | B2 → R | 0.0265 | −3.6 |
| R → B19′ | <0.2804 *,† | −31.8 | |
| TiNi51.3 450 °C × 4 h (TiNi51.3-450) | B2 → R | 0.0265 | 0 |
| R → B19′ | <0.2804 *,† | −1.8 | |
| Ti50Ni40Cu10 (TiNiCu10) | B2 → B19 | 0.11~0.17 | −0.9 |
| B19 → B19′ | <0.2804 *,† | −3.8 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, S.-K.; Chang, Y.-C. Thermal Cycling Effect on Transformation Temperatures of Different Transformation Sequences in TiNi-Based Shape Memory Alloys. Materials 2019, 12, 2512. https://doi.org/10.3390/ma12162512
Wu S-K, Chang Y-C. Thermal Cycling Effect on Transformation Temperatures of Different Transformation Sequences in TiNi-Based Shape Memory Alloys. Materials. 2019; 12(16):2512. https://doi.org/10.3390/ma12162512
Chicago/Turabian StyleWu, Shyi-Kaan, and Yi-Ching Chang. 2019. "Thermal Cycling Effect on Transformation Temperatures of Different Transformation Sequences in TiNi-Based Shape Memory Alloys" Materials 12, no. 16: 2512. https://doi.org/10.3390/ma12162512
APA StyleWu, S.-K., & Chang, Y.-C. (2019). Thermal Cycling Effect on Transformation Temperatures of Different Transformation Sequences in TiNi-Based Shape Memory Alloys. Materials, 12(16), 2512. https://doi.org/10.3390/ma12162512




