4.1. Grain Refinement Mechanism
In
Figure 1, there is clearly a trend of grain size decreasing by different treated processes. After the addition of 0.05%Fe, the grain size of Mg-3%Al decreases slightly. This means that the constitutional undercooling produced by the Fe element cannot be neglected, especially as Fe has a very high growth restriction factor (GRF = 52.56) [
20]. Therefore, the effect of constitutional undercooling resulting from Fe was determined in this study. The total constitutional undercooling (
) produced at the solid–liquid interface can be calculated by the following equation:
where
fS is the solid mass fraction,
is the slope of the liquids,
is the alloy composition, and
P = 1 −
k,
k is the equilibrium distribution coefficient. The calculation results are shown in
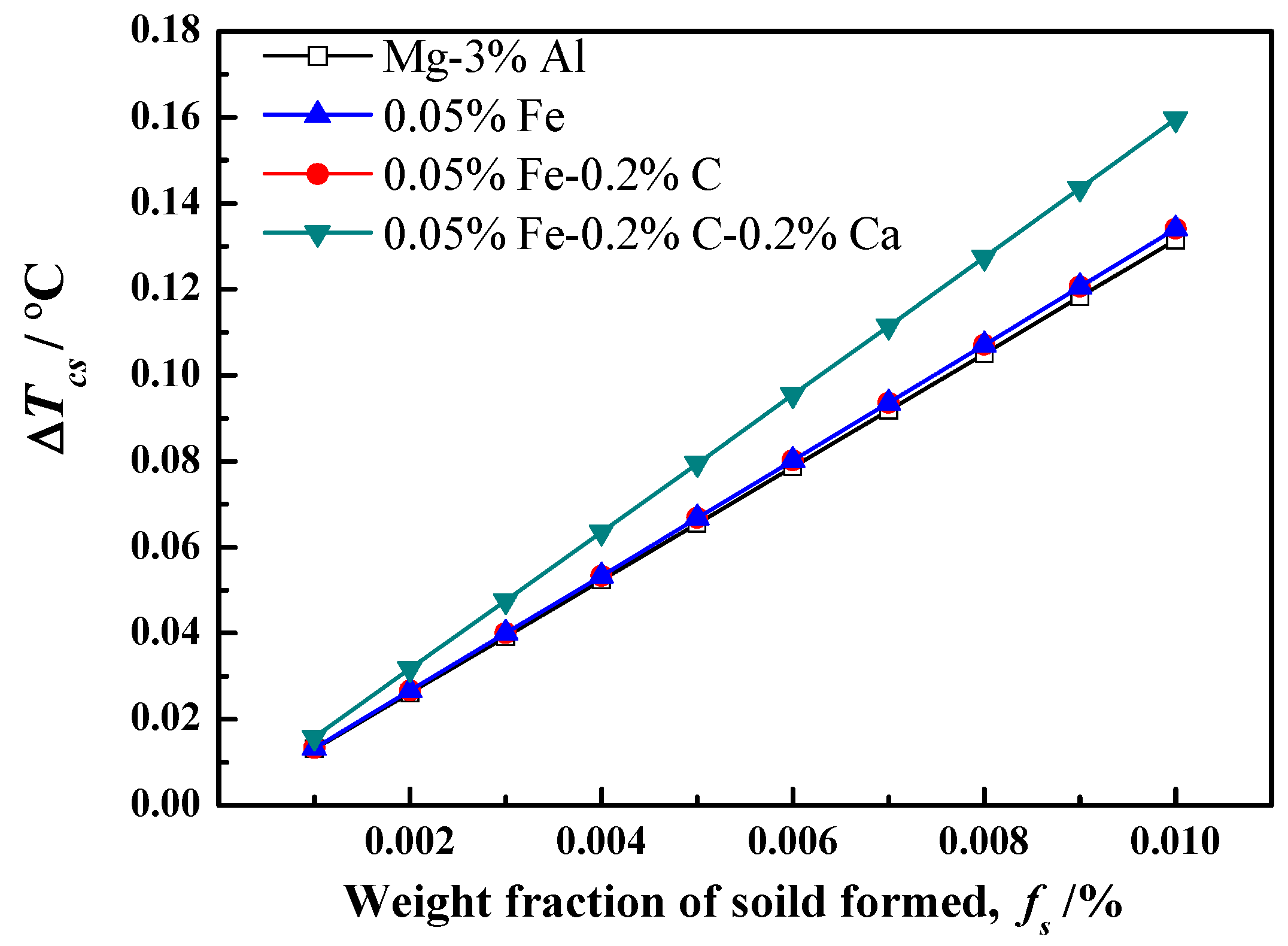
Figure 4, and the relevant parameters are listed in
Table 1. As shown in
Figure 4, 0.05%Fe addition can improve the constitutional undercooling, but the increase is limited. As an inevitable impurity element, the content of Fe in Mg alloy is very low. Even in commercial raw materials, the content of Fe is less than 0.05%. The solubility of carbon in the Mg–Al melt is too small. It has been reported that the solubility of carbon in Mg-3%Al and Mg-9%Al is about 20 ppm [
32]. Therefore, the constitutional undercooling produce by carbon was not taken into calculation, and the curve of carbon addition overlapped with addition of Fe, as shown in
Figure 4. It is worth noting that the constitutional undercooling clearly increased after adding 0.2%Ca. In this research, Ca was added in the Mg-3%Al melt by Mg-10%Ca master alloys. Mg-10%Ca alloys are decomposed completely and dissolved into the melt as a solute, since the solubility of Ca in the Mg melt is about 0.8% at 760 °C [
33]. It is known that Ca has a strong tendency for segregation. Therefore, the solute of Ca could greatly affect the constitutional undercooling and, consequently, restrict grain growth during the solidification process. The results in
Figure 1d also confirm that the grain size is further refined after Ca addition.
The constitutional undercooling shows little change, while the grain size is significantly refined after carbon inoculation, as shown in
Figure 1c and
Figure 4. Therefore, the significant grain refinement after carbon inoculation cannot be explained by the constitutional undercooling. The effect of nucleating particles on grain size must be considered. In this study, there mainly exist two kinds of effective nucleation particles. One is the Al
4C
3 cluster particle, and the other is the duplex-phase particles. The interfacial phases between the Mg melt and these two heterogeneous nucleation particles are both Al
4C
3, which is believed to be a potent nucleating substrate for primary α-Mg by crystallography calculation [
35]. However, the different diameters of these two particles results in different refining efficiencies. The diameter of the nucleating particle is a very important factor for nucleation efficiency. Greer et al. put forward a model establishing the relationship between nucleation particle size and nucleation undercooling (
) [
36], as shown in Equation (2):
where
is the solid-liquid interface energy,
is the entropy of fusion per unit volume, and
is the diameter of the nucleating particle. According to Equation (2), the lager the particle size is, the smaller the nucleation undercooling
is required. Large particles have a higher potency to act as heterogeneous nucleation sites. As shown in
Figure 2, The diameters of duplex-phase particle are about 3–5 μm, while the diameters of Al
4C
3 cluster particle are about 1–2 μm. Deduced from this model, the grain refinement efficiency of the duplex-phase particle is higher than that of the Al
4C
3 cluster particle.
4.2. Formation Process of Duplex-Phase and Al4C3 Cluster Particles
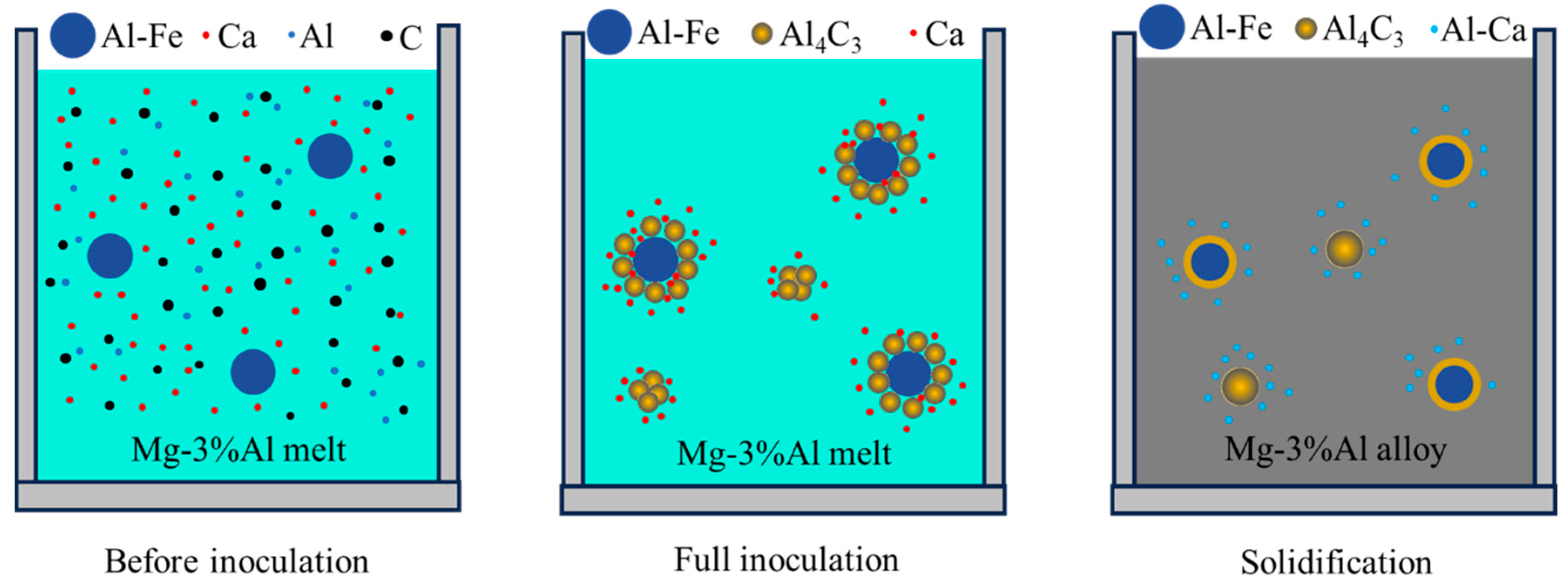
A brief description of the formation process of nucleating particles is given in
Figure 5. In the initial stage of inoculation, Al–Fe particle Ca solutes exist in the Mg-3%Al melt. In the process of inoculation, Al
4C
3 can be formed by the following reaction [
8]:
These thermodynamic data indicate that the formation of Al
4C
3 is thermodynamically possible at a temperature of 760 °C. After full inoculation, some of Al
4C
3 particles are adsorbed onto the surface of Al–Fe particles to form duplex-phase particles. The other Al
4C
3 particles agglomerate to form Al
4C
3 cluster particles. During the inoculation, Ca segregates towards to the interface of the Al–Fe and Al
4C
3 particles, as shown in
Figure 5. Finally, Ca reacts with Al to form Al–Ca particles during solidification.
The exactly microstructure of Al
4C
3 cluster particles are confirmed by TEM as shown in
Figure 6. As can be seen from the TEM image, the diameter of in-situ formation Al
4C
3 particles is very small (nano scale). These tiny particles have a high surface energy and are easily to agglomerate. Therefore, the single phase Al
4C
3 particle is constituted by a cluster of tiny Al
4C
3 particles.
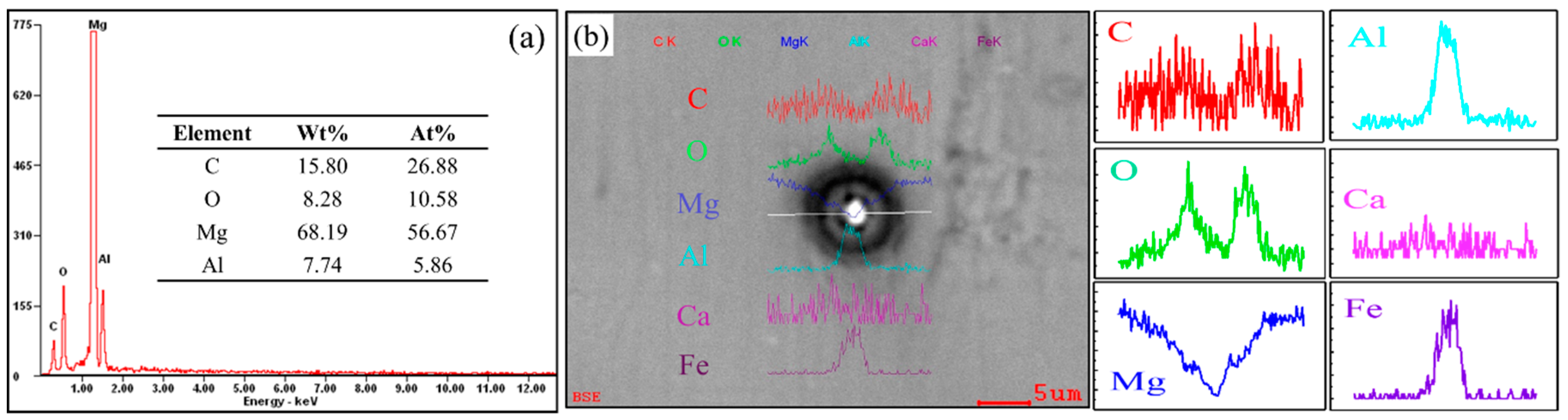
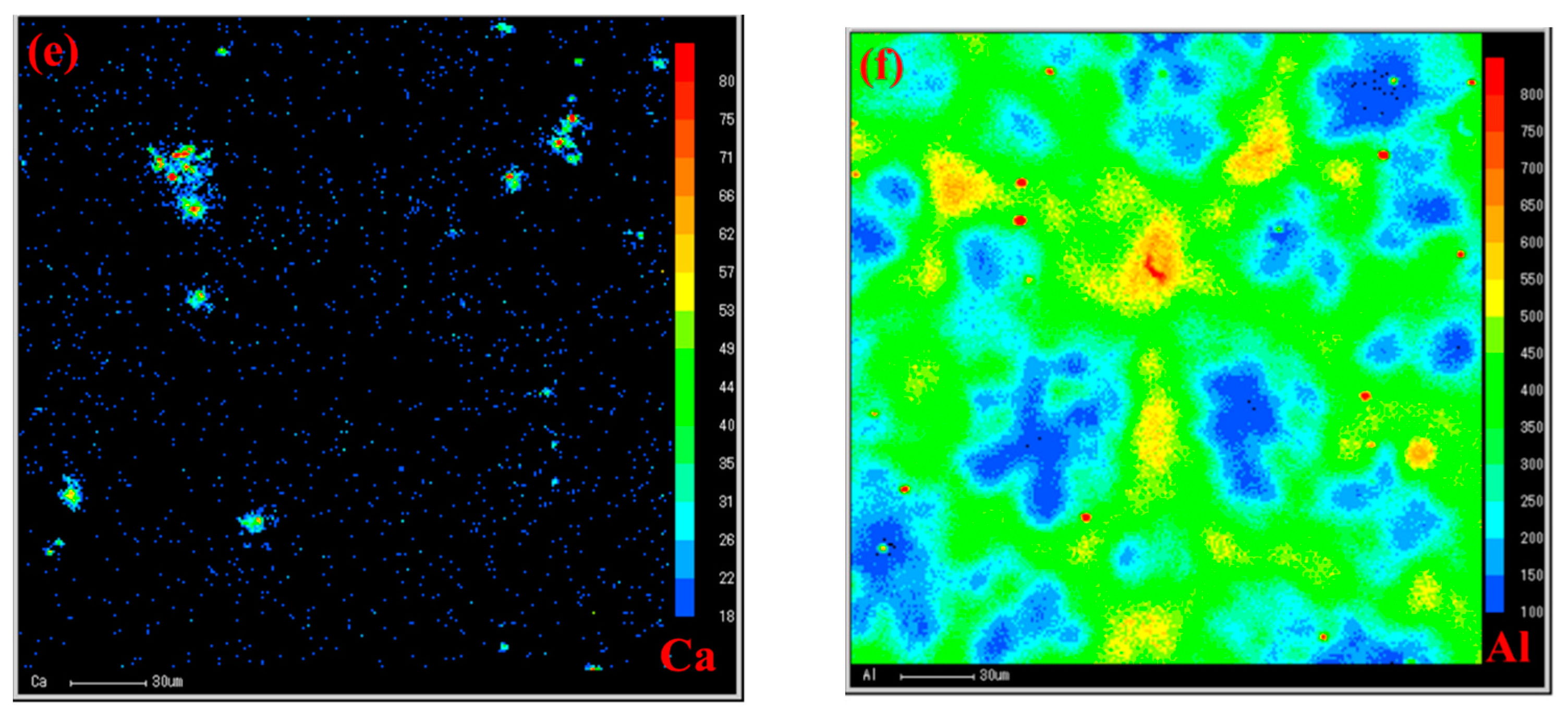
To reveal the relationship between the role of Ca and the duplex-phase particles, an electron probe microanalyzer with a wavelength dispersion spectrometer (EPMA-WDS) map analysis was carried out to identify the element distribution, as shown in
Figure 7. It can be seen from
Figure 7a that there are three typical duplex-phase structure particles and many Al
4C
3 cluster particles. Fe appears at the center of the duplex-phase particles. High concentrations of C and O were found to surround the Fe element and the distribution of these two elements always overlaps. Interestingly, both Al
4C
3 cluster particles and duplex-phase particles were surround by Ca. Unlike the distribution of the C and O elements, the Ca element did not form a complete shell coating on the core of Al–Fe or the Al
4C
3 cluster particles.
Figure 7e clearly shows a trend where the Ca element is segregated towards the duplex-phase and Al
4C
3 cluster particles.
4.3. Establishment of the Thermodynamic Model
In the author’s previous experiment [
37], the amount of tiny Al
4C
3 particles with sizes ranging from 10 to 1000 nm could be found in the melt. Based on the discussion above, these sub-micron particles could be extremely easy to adsorb on relatively large Al–Fe rich particles or cluster together due to their high surface energy. Classical thermodynamic theory can be used to reveal the formation mechanism of two kinds of particles by a change in Gibbs free energy. The Gibbs free energy of the system associated with the formation of a new particle (duplex-phase or Al
4C
3 cluster particles) can be expressed as
where
S is the total surface area per unit mole of particles (m
2/mol) and
γ is the interfacial energies at the boundaries (J/m
2). The element of
i and
j represent the particles in the melt after and before the formation of new particles, respectively.
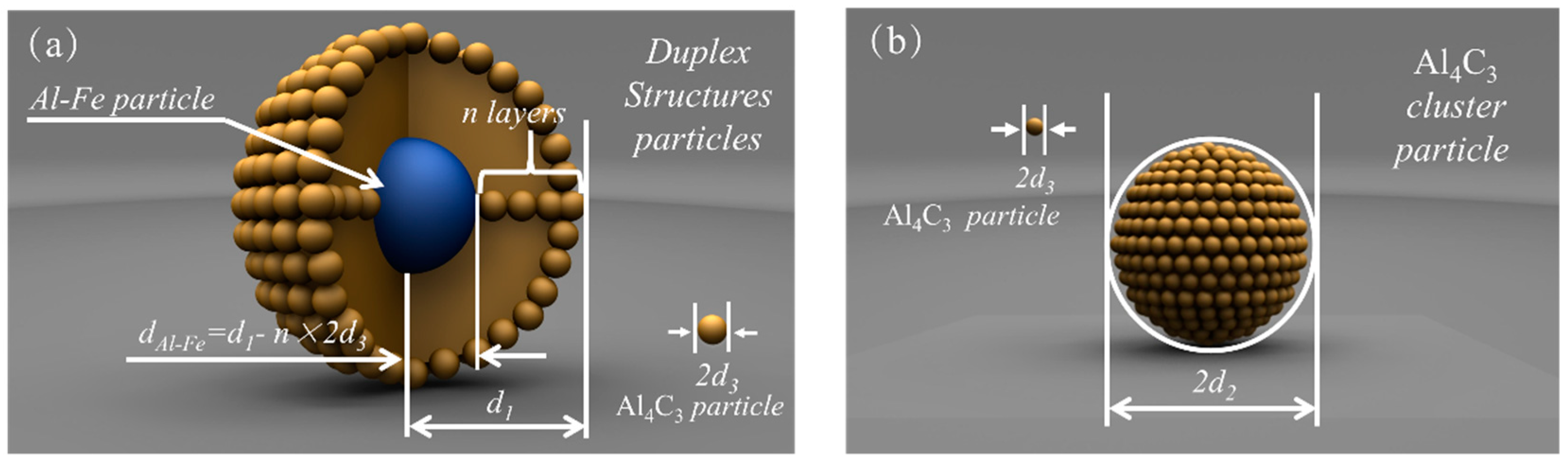
In order to calculate the change in the Gibbs free energy of the system associated with the formation of adsorption and clustering, the assumptions employed are as follows:
(1) All the duplex-phase particles and Al
4C
3 cluster particles are spherical, and their radii are represented by
d1 and
d2, respectively, as shown in
Figure 8a,b.
(2) The radius of all tiny Al4C3 particles is denoted by d3.
(3) Al–Fe particles are completely coated by tiny Al4C3 particles. The thickness of one layer is approximate to a tiny Al4C3 particle’s radius. The number of adsorption layers is represented by n. Therefore, the radius of the Al–Fe particle is dAl-Fe = d1 – n × 2d3.
(4) The tiny Al4C3 particles are distributed uniformly in the Mg–Al melt before adsorbing to the Al–Fe particle or clustering to a relatively large Al4C3 particle.
(5) The total number of the tiny Al4C3 particles in the duplex-phase particle and the Al4C3 cluster particle is the same.
For a duplex-phased structure particle, the volume of the outer Al
4C
3 layer (
) can be expressed as
The total number of the tiny Al
4C
3 particles (N) that adsorbed on the surface of the Al–Fe rich particle can be given as
It is assumed that after carbon containing pellets were plunged into the melt, the total number of tiny Al
4C
3 particles (N) and one Al–Fe particle were uniformly distributed in the melt. In the process of carbon inoculation, tiny Al
4C
3 particles are completely adsorbed onto the surface of Al–Fe particles to form a duplex-phase particle. According to Equation (4), the change in the Gibbs free energy (
) of the system associated with the formation of the duplex-phase particle shown in
Figure 8a can be written as
where the
is the surface area of duplex-phase particle and the
is the interfacial energy between Al
4C
3 phase and Mg melt.
is the surface area of the Al–Fe particle and
is the interfacial energy between the Al
4C
3 phase and the Al–Fe phase, since the Al–Fe particle is surround by tiny Al
4C
3 particles. Before carbon inoculation, tiny Al
4C
3 particles (N) and one Al–Fe particle were uniformly distributed in the melt. Therefore,
is the interfacial energy between Al–Fe phase and the Mg melt.
is the surface area of the all the tiny Al
4C
3 particles. The
,
, and
can be represented by the radii of
d1,
d2, and
d3 via the following function:
Finally, the
be represented by the radius of
d1 and
d2 via the following function:
The Gibbs free energy change of the cluster particles (
) can also be calculated using the same model. The
can be represented by the following function:
Theoretically, the interfacial free energy at the nucleating interface is believed to be a key factor in controlling heterogeneous nucleation efficiency. However, the interfacial energy is usually difficult directly measure for solid–liquid or solid–solid interfaces. As the crystal planes are usually bound with the lowest interface energy the Al
4C
3/Al–Fe interface, it can be regarded as a coherent interface. Therefore, the interface energy
could be taken as 0.1 J/m
2, since the coherent interface energy is generally considered to be in the range from 0 to 0.2 J/m
2 [
38].
The interfacial energy between the two phases can be estimated by Girifalco–Good’s [
38] equation:
where
δA/B is the interfacial energy between the A phase and B phase,
τA and
τB are the surface energy of A phase and B phase respectively, and
ϕAB is the interaction between these two phases which typically ranges around 1.
Pan calculated the surface energy of the FeAl (110) alloy surface by first-principles calculations [
39]. The result shows that the Fe:Al = 1:1, Fe:Al = 1:2, and Fe:Al = 1:3 surface structures are stable, and the surface energy of these three surface structures ranges from 1.28 J/m
2 to 2.4 J/m
2. In our previous study, AlFe
3 was considered as a possible compound with a surface energy
τAl–Fe of 2.24 J/m
2 [
24]. Li analysed slabs of Al
4C
3 (0001) by first-principles calculations [
40]. The calculation shows that the Al-terminated surface is more stable than the C-terminated surface, and the surface energy of C-termination surface is about 2.7 J/m
2, which could be taken as the surface energy of the Al
4C
3 phase. Substituting
τAl–Fe = 2.24 J/m
2,
τAl–C = 1.33 J/m
2, and
τMg(l) = 0.577 J/m
2 into Girifalco–Good’s equation (Equation (13)), the interfacial energy of
and
can be calculated as 0.16 J/m
2 and 0.56 J/m
2, respectively.
For ease of comparison, the radii of the duplex-phase particle and Al
4C
3 cluster particles were taken as the radii of the nucleation particles in
Figure 9 and
Figure 10. In order to established a thermodynamics model close to the real experimental process, the radii of the duplex-phase particle(
d1), the Al
4C
3 cluster particle (
d2), and the tiny Al
4C
3 (
d3) particle range from 0 to 10 μm.
4.4. Calculation Results from Thermodynamic Model
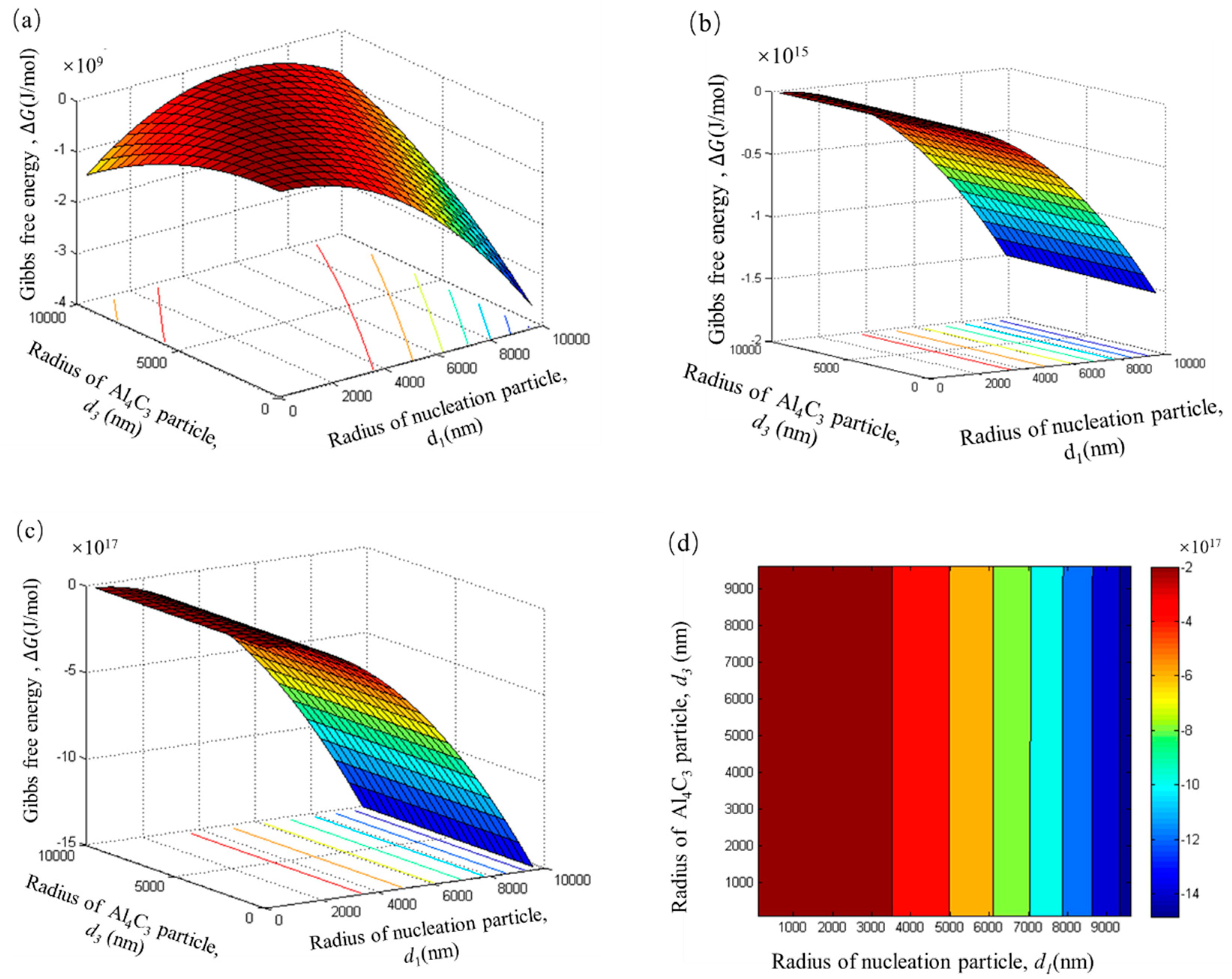
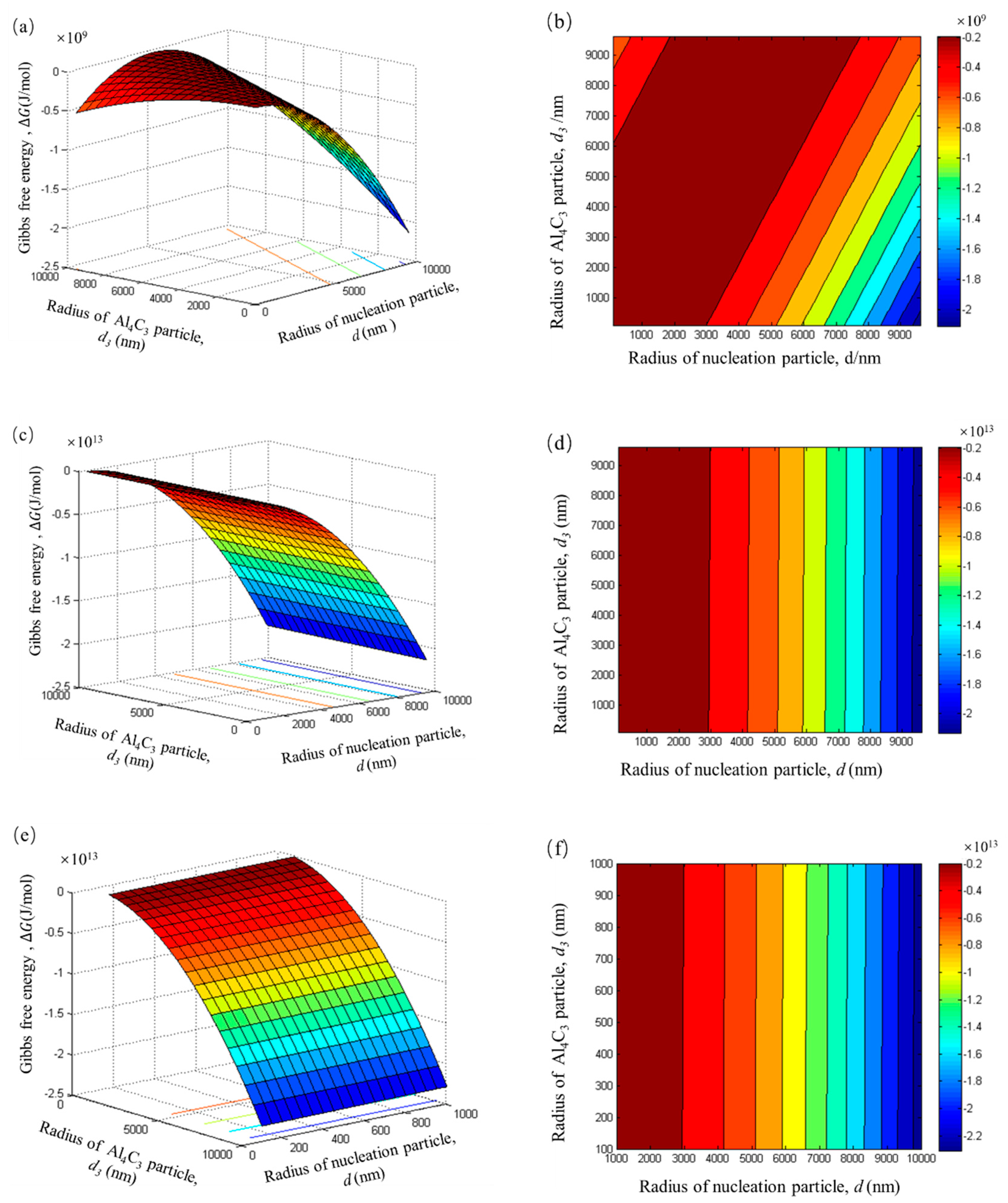
As shown in
Figure 9a, all the Gibbs free energy within the range of
d1 and
d3 is negative, when Al–Fe particles only adsorb a single layer of Al
4C
3 particles. The Gibbs free energy is negative to about a 10
9 J/mol magnitude. This result indicates that the Al–Fe particle adsorbing tiny Al
4C
3 particles to form a duplex-phase particle is a spontaneous process in the initial stage of carbon inoculation. When the number of adsorbed layers increases from 100 to 1000, the Gibbs free energy of the system is more negative, and the order of magnitude increased from 10
15 to 10
17, respectively. These results suggest that the growth of the duplex-phase particle by the Al–Fe particle adsorbing the tiny Al
4C
3 particle is also a spontaneous process. As
Figure 9b,c shows, an increase in the absorption of the layer produced no significant change in the shape of the pattern. In order to predict the trend of Gibbs free energy, which changes alongside the nucleated particles’ radii, the adsorption date of 1000 layers is selected to draw a contour map, as shown in
Figure 9d. The contour map is the region in the same colour expressing equal Gibbs free energy.
It can be seen from
Figure 9d that the Gibbs free energy is significantly reduced, while the radius of the nucleation particles increases. This can be attributed to the larger particles having a larger surface area that can absorb more tiny Al
4C
3 particles. Therefore, the total surface area reduces and makes the Gibbs free energy of the system more negative. It should be noted that the Gibbs free energy has nothing to do with the radius of tiny Al
4C
3 particles.
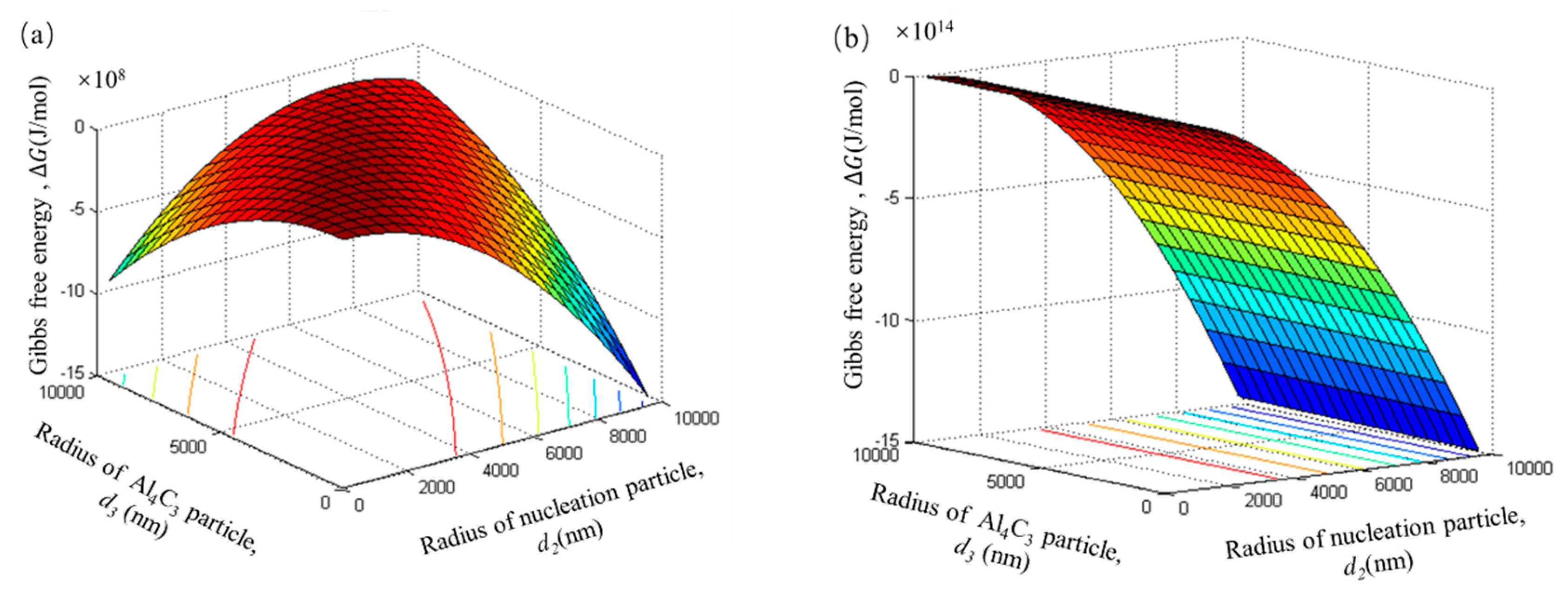
In this thermodynamics model, it is assumed that the number of tiny Al
4C
3 particles in the Al
4C
3 cluster particle is equal to the adsorption layer of duplex-phase particle. The total number of tiny Al
4C
3 particle can be calculated by Equation (6). In the initial stage, the Gibbs free energy change of the Al
4C
3 cluster particle also shows a high negative value of 10
8 J/mol. This indicates that Al
4C
3 cluster particles also have a tendency to form spontaneously. With an increase in the number of tiny Al
4C
3 particles, this spontaneous trend becomes more and more obvious, as shown in
Figure 10b,c. From the decreasing trend of Gibbs free energy (
Figure 10d), it can be seen that the Al
4C
3 cluster particle can grow via tiny Al
4C
3 particle clusters.
From the above experimental results and thermodynamic model analysis, it can be concluded that the two kinds of particles coexist and have a competitive relationship. The data of adsorbing 1, 100, and 1000 layers were used to investigate the competition relationship between these two types particles at different stages of carbon inoculation. The competitive trend between these two types particles can be compared by reducing the Gibbs free energy of the system. The Gibbs free energy of the competitive trend
can be expressed by
In order to compare the formation trend of duplex-phases and Al
4C
3 cluster particles at different radius, the radii of both particles are expressed by
d:
As the relationship of Equation (15) shows, if the is less than 0, the tendency of duplex-structure particle formation is more obvious. Otherwise, the tendency to form Al4C3 cluster particles is more obvious.
The result calculated by Equation (14) is shown in
Figure 11. As can been from
Figure 11a, the Gibbs free energy is always negative. It can be deduced that the adsorption process is more spontaneous than the cluster process and the tiny Al
4C
3 particles prefer to form duplex phase particles rather than gather to form Al
4C
3 cluster particles. However, in the range of a specific particle radius, the
is close to zero, as shown in
Figure 11a (the dark red region). These results suggest that within a certain particle radius, the changes of Gibbs free energy by these two types of particles are almost equal. Thus, if the particle size is within this range, the trend of formation for the two types of particles is equal. This particle size range is accurately reflected in the dark red region of the contour map in
Figure 11b. It is interesting to note that when the layers increased, there is a clear trend showing a decrease of the dark red region area, as shown in
Figure 11d,f. These results indicate that the trend of forming stable duplex-phase particles becomes more and more obvious during the carbon inoculation process. This result is also confirmed by our previous research, which showed that the duplex phase particles kept stable when prolonging the holding time to 80 min and exhibit significant fading resistance [
6].
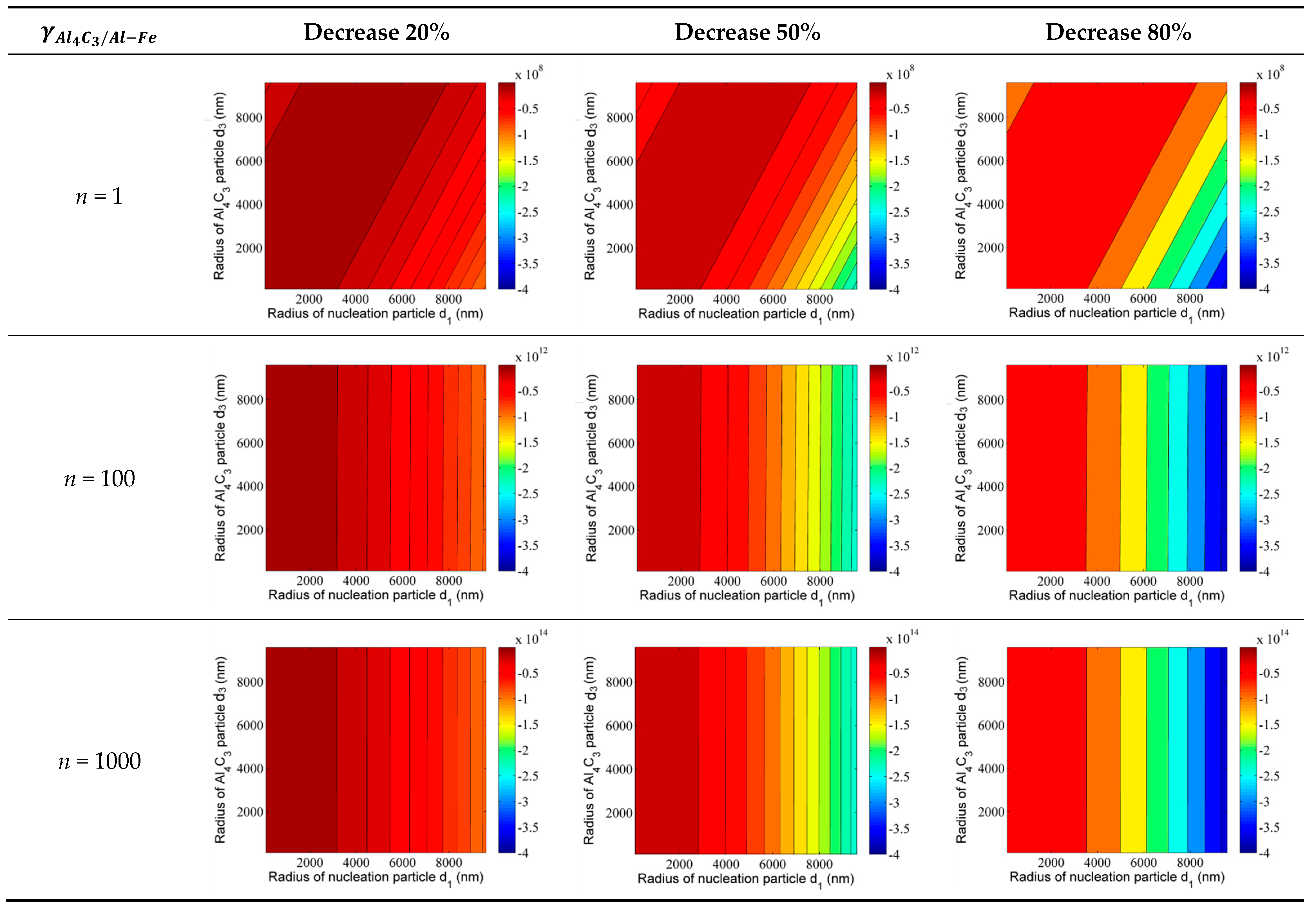
4.5. The Role of Ca Solute in Carbon Inoculated Mg-3%Al Alloy
As discussed above, the solute of Ca can provide constitutional undercooling in front of the nucleus/liquid. Furthermore, Ca is a surface active element that can reduce the interfacial energy effectively between the two phases by segregating around the interface of the two phases. As shown in
Figure 7e, the solute of Ca is segregated around the duplex-phase. It can be inferred that the addition of Ca reduces the interfacial energy between the Al
4C
3 phase and the Al–Fe phase. The change in Gibbs free energy can be used to describe the effect of adding Ca on the formation of the duplex phase particles. The change of the Gibbs free energy after Ca addition can be expressed by the equation:
Unfortunately, there are no accurate data on the reduction of interfacial energy between the Al
4C
3 phase and Al–Fe phase after Ca addition. Therefore, we assume that the interfacial energy
can be reduced by 20%, 50%, and 80%. The changes in the Gibbs free energy after Ca addition are shown in
Figure 12. After adding Ca, all changes in Gibbs free energy with different adsorption Al
4C
3 layers are negative. Furthermore, with the change of Gibbs free energy, the energy becomes more negative with a decrease in the interfacial energy
. As shown in
Figure 12, the color of the contour map changes from dark red to dark blue. This indicates that the addition of Ca can reduce the resistance of forming duplex-phase particles. In other words, the reduction interface energy induced by Ca would promote the formation of duplex phase particles. As the number of adsorption layers increases to 100 and 1000 layers, the trend in Gibbs free energy changes remains unchanged. Furthermore, the orders of magnitude are negative (from 10
12 to 10
14 J/mol), with the adsorption layers increasing from 100 to 1000 layers. This indicates that the size of the duplex phase particle could increase by adsorbing more Al
4C
3 layers after Ca addition. Based on the discussion above, the duplex-phase particles have a higher refinement efficiency due to the larger size of the duplex-phase particle. Therefore, the grain refining efficiency can be further improved by the addition of Ca.