Biocompatibility and Clinical Application of Porous TiNi Alloys Made by Self-Propagating High-Temperature Synthesis (SHS)
Abstract
1. Introduction
2. Fabrication of Porous SHS TiNi
3. Characteristics of Porous SHS TiNi
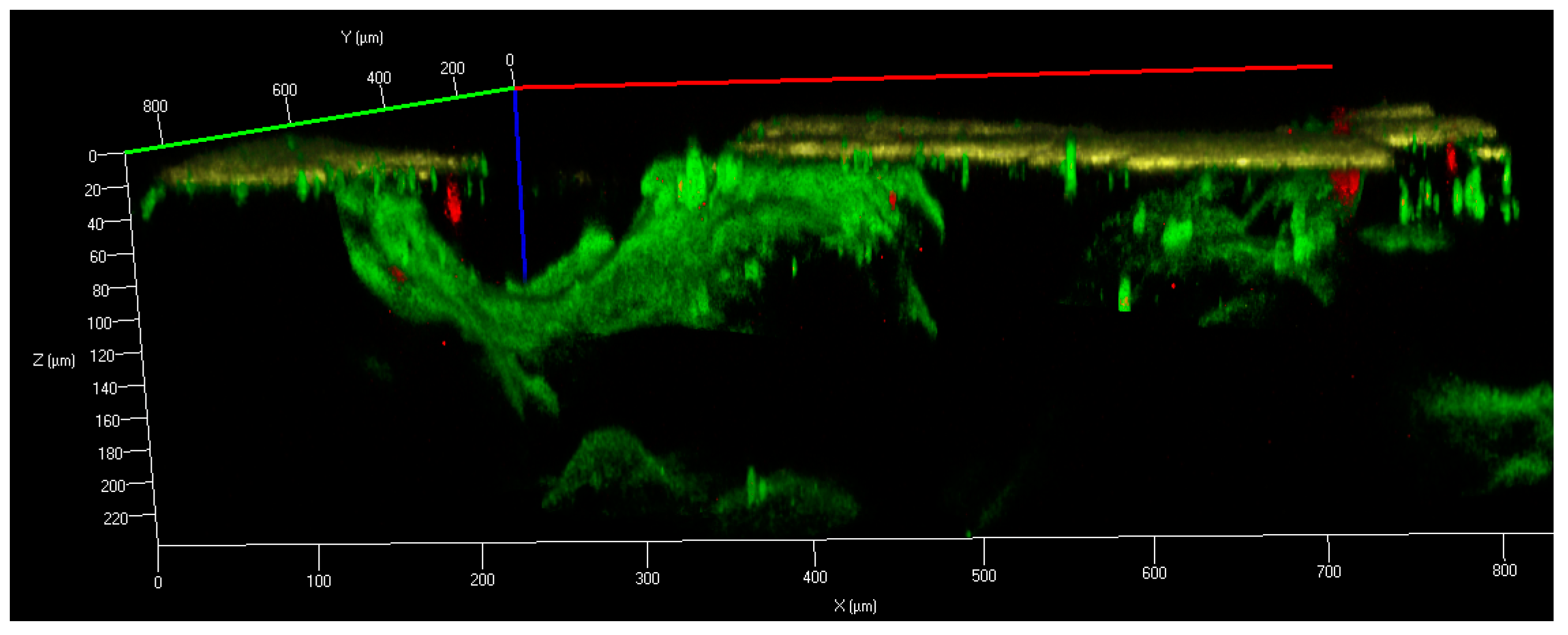
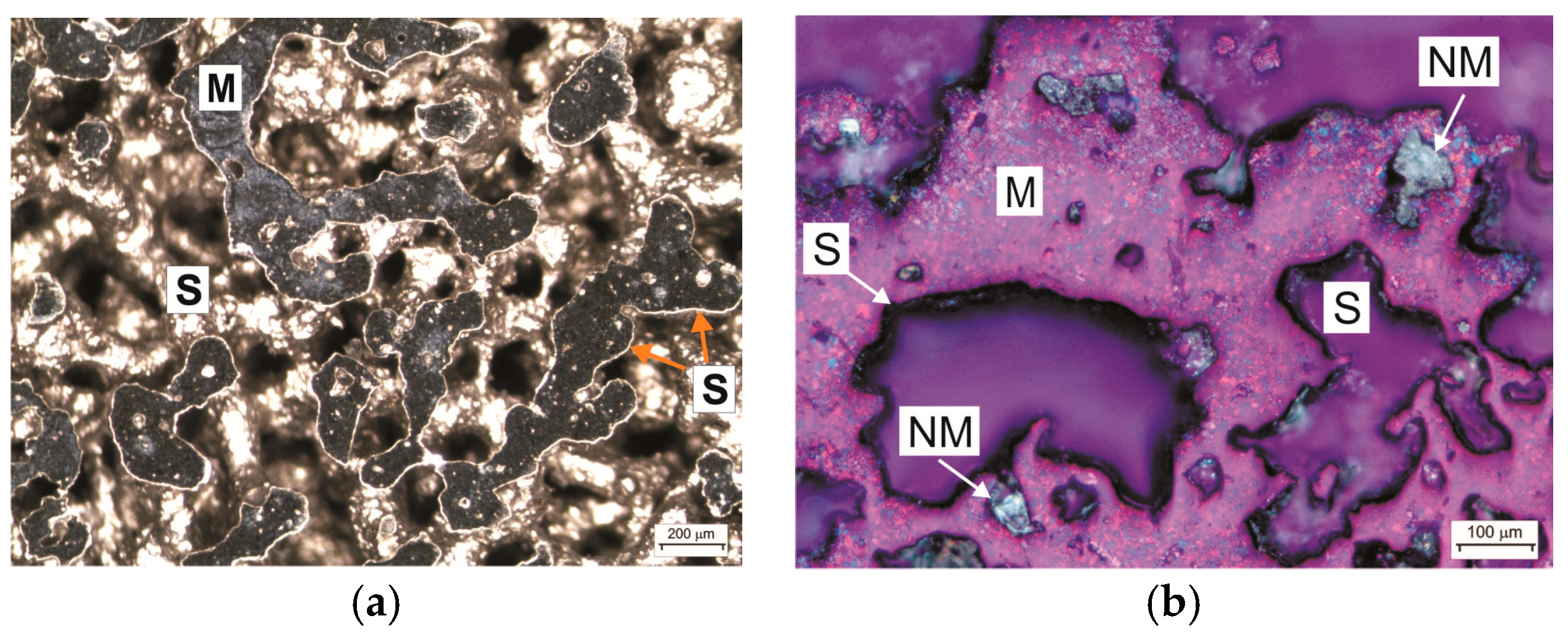
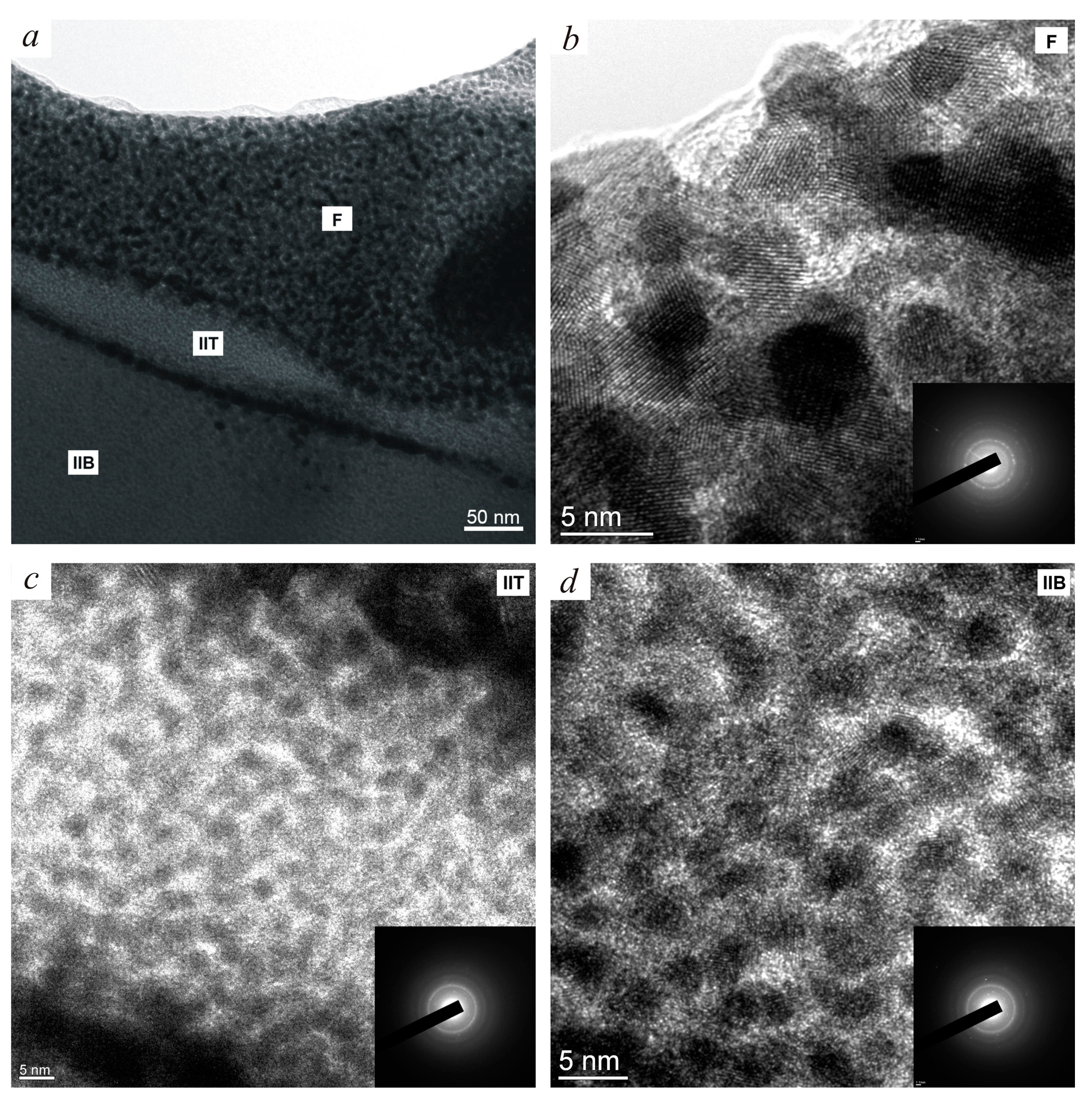
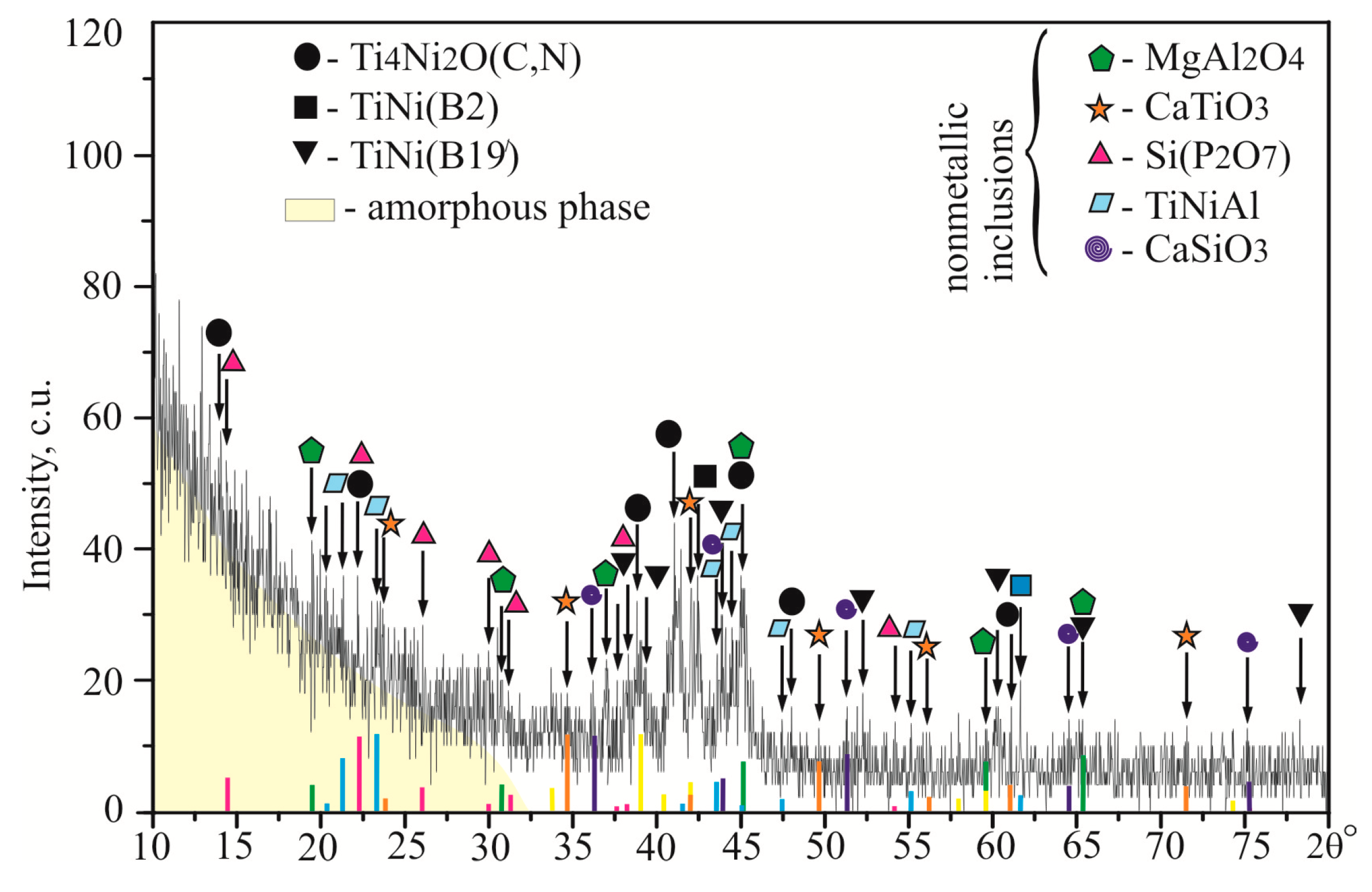
3.1. Structure and Phase Composition of the PTN Surface
3.2. Rheological Resemblance of PTN to Biotissues
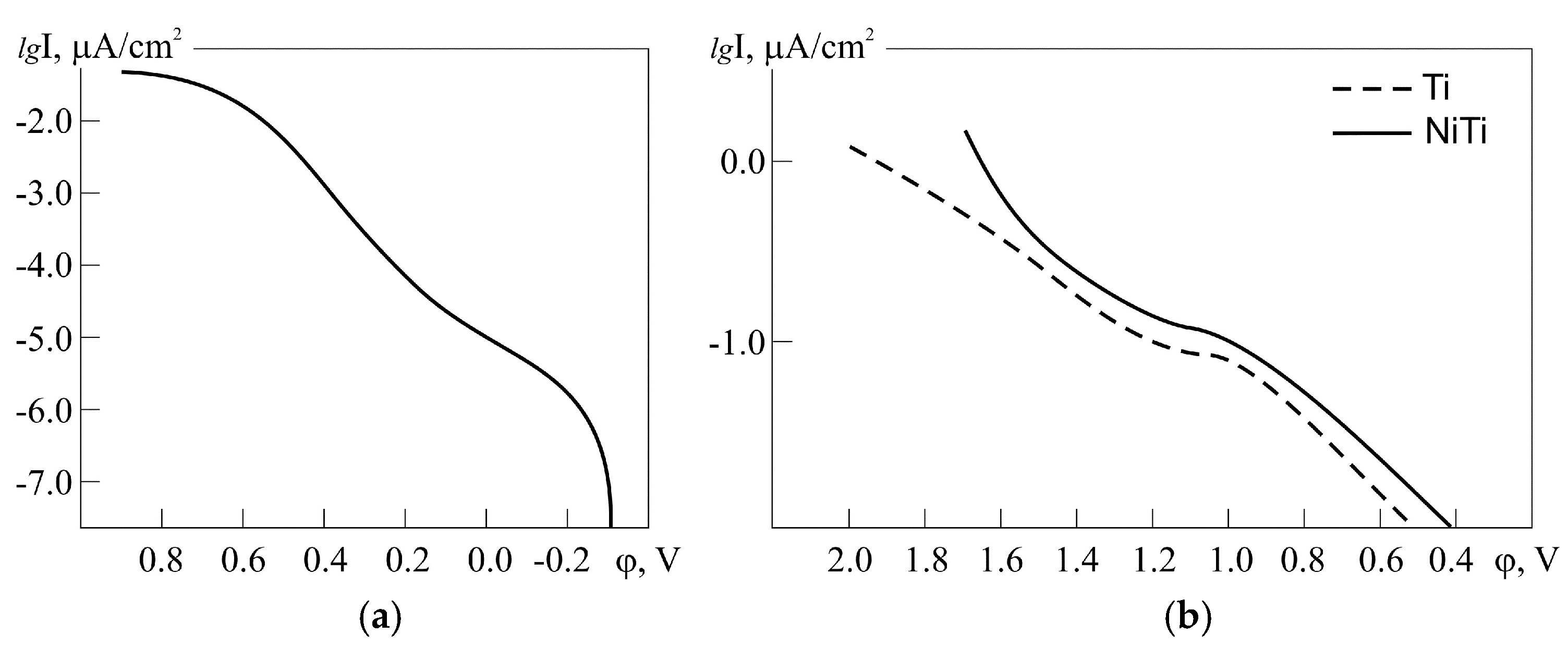
3.3. Polarization Behaviour of PTN
4. Cytocompatibility of the PTN Surface
- -
- A well-developed spitted topography of pore walls (a large number of interconnected small pores and rough interpore partitions), which sustains the initial cell adhesion;
- -
- Wetting ability, which facilitates saturation by water-soluble substances;
- -
- Phase and chemical composition of the superficial strata, which has no inhibitory/toxic effect on cells seeded and growing tissular variants;
- -
- Bio-mechanical behaviour, which is pretty similar to that exhibited by alive tissues.
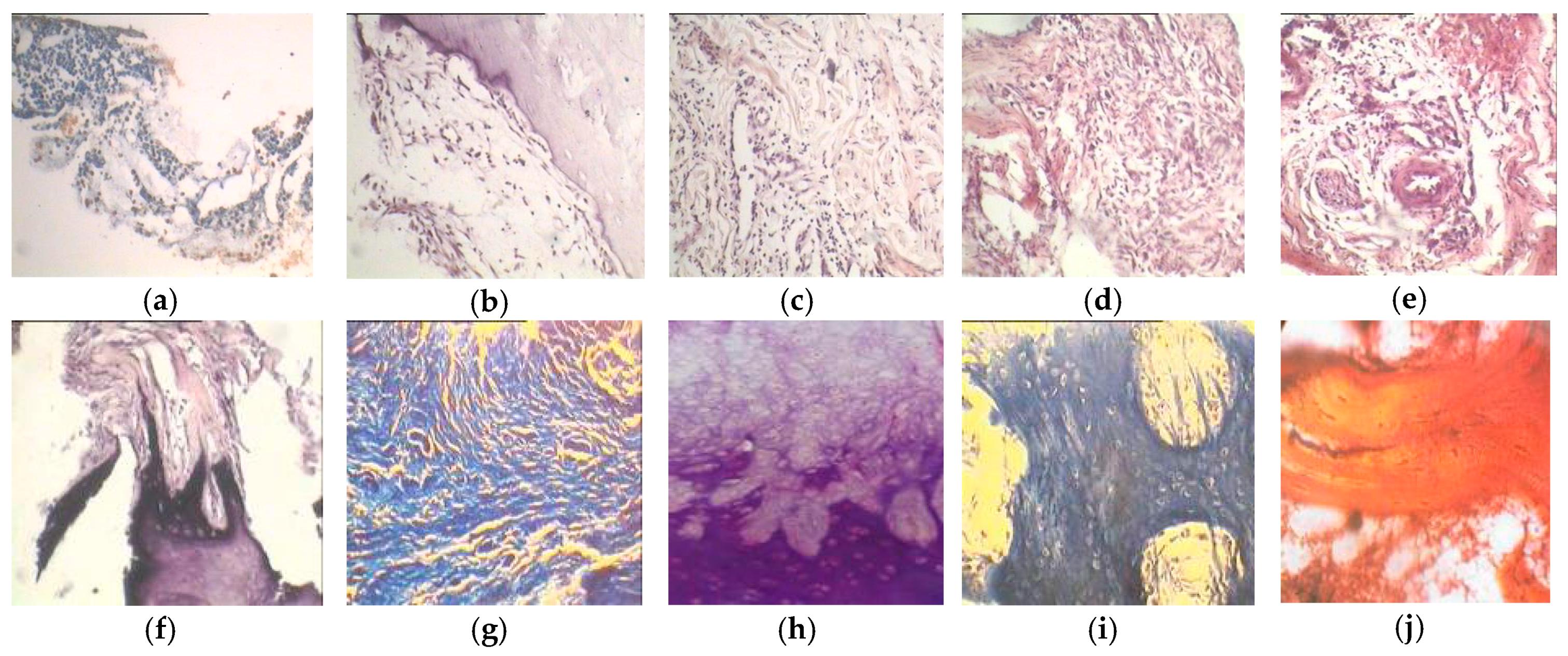
5. Histological Studies of the Implanted PTN Scaffold in Vivo
6. Clinical Application of PTN Implants
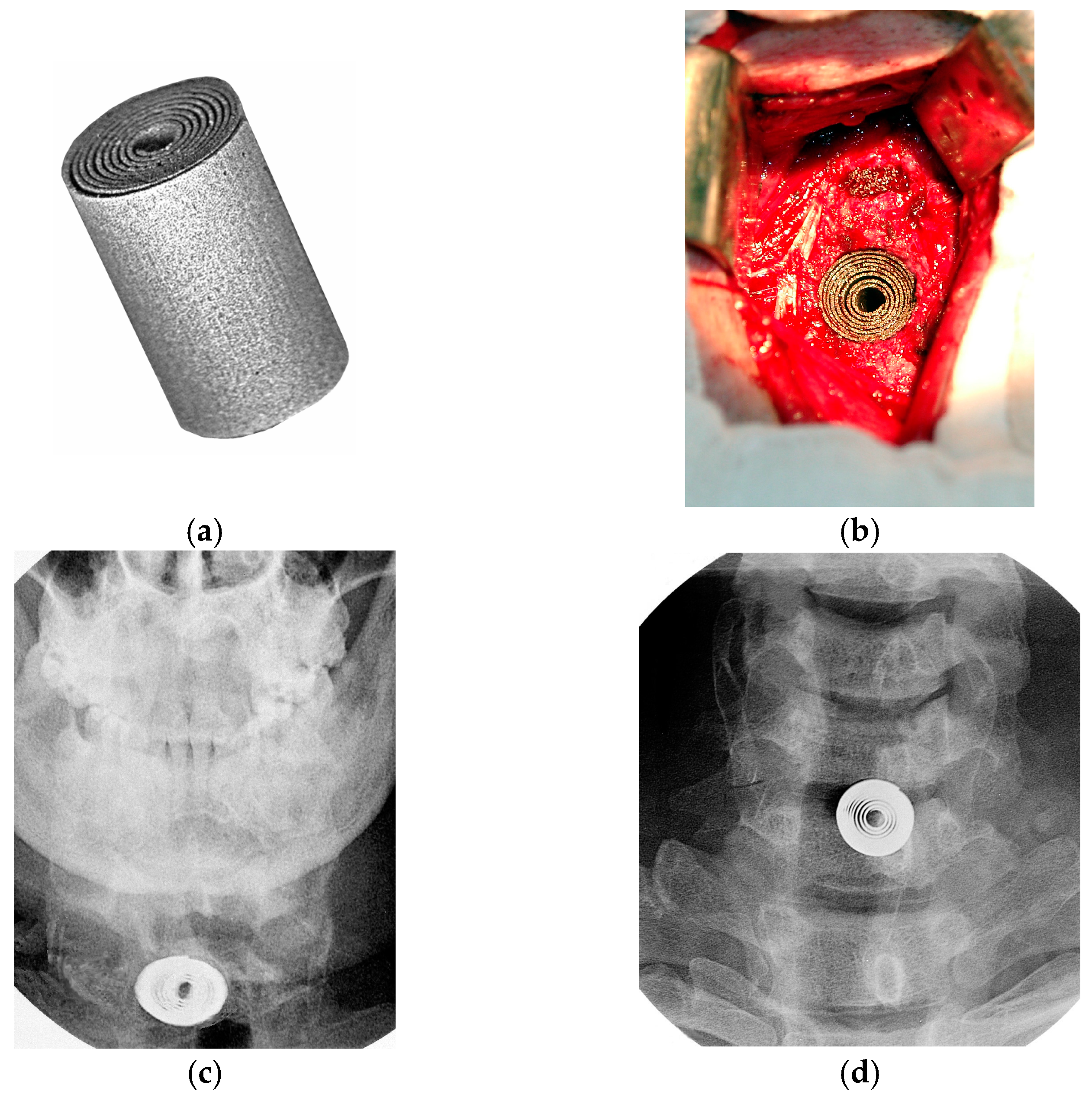
6.1. Cervical Spine Superelastic PTN Cages
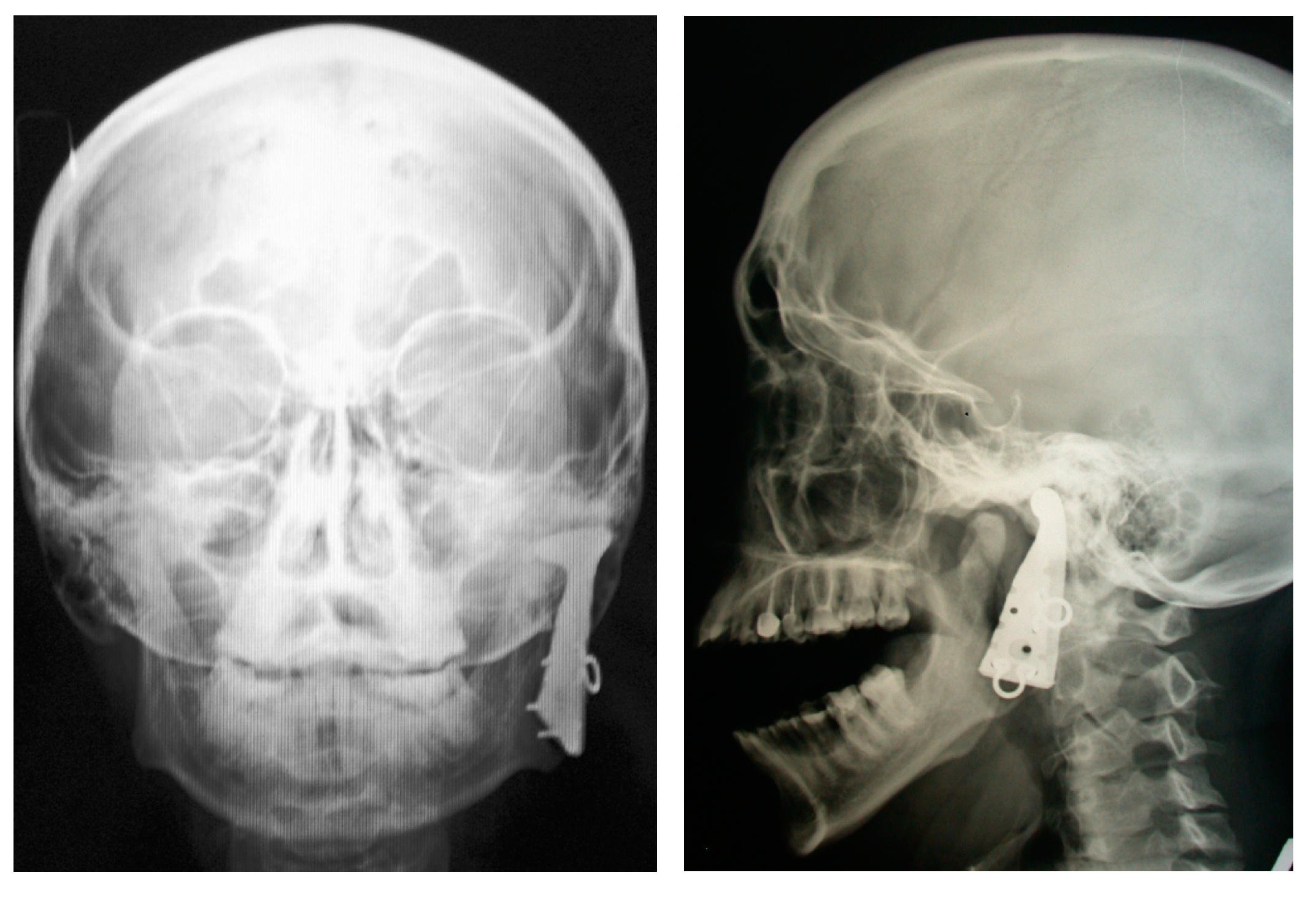
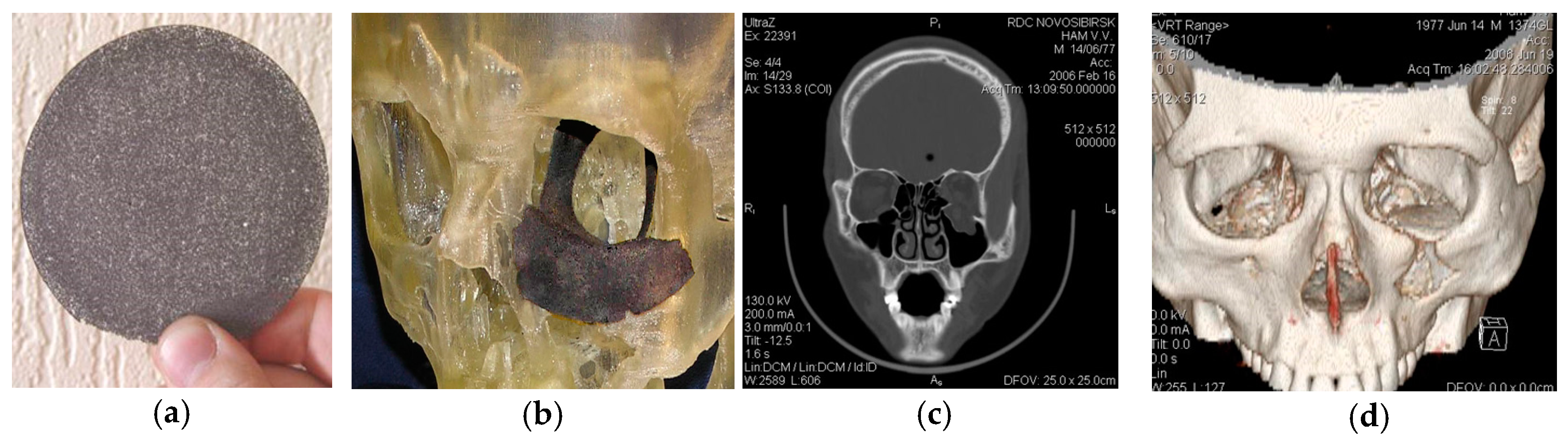
6.2. Customized PTN Endografts in Maxillofacial Surgery
6.3. Customized PTN Endografts in Oncosurgery
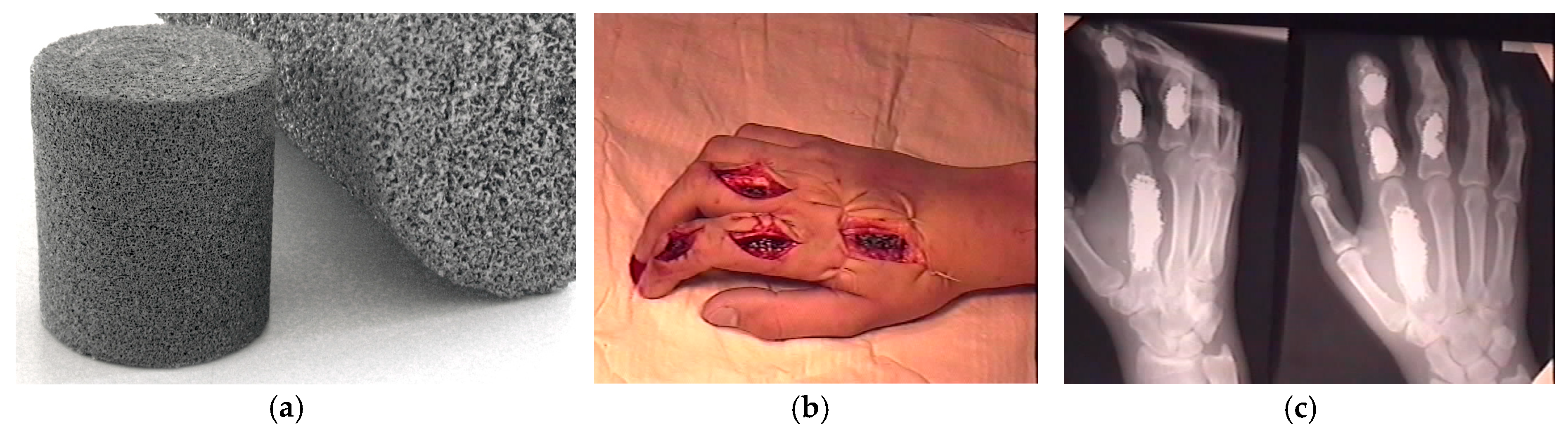
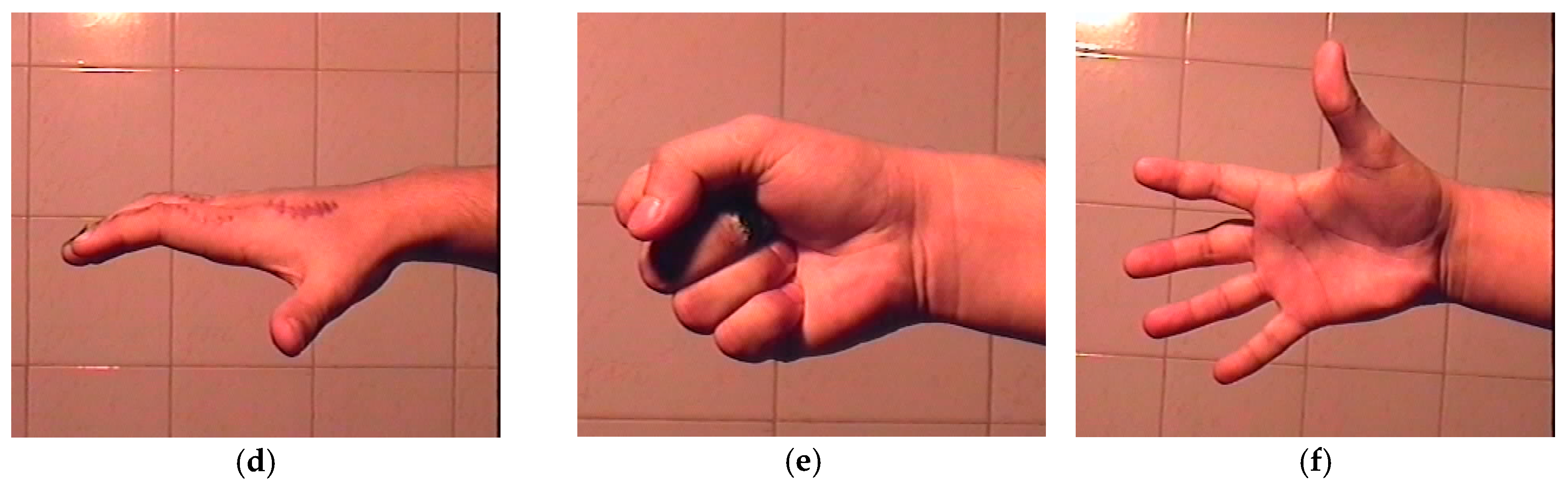
6.4. PTN Implants in Traumatic Surgery
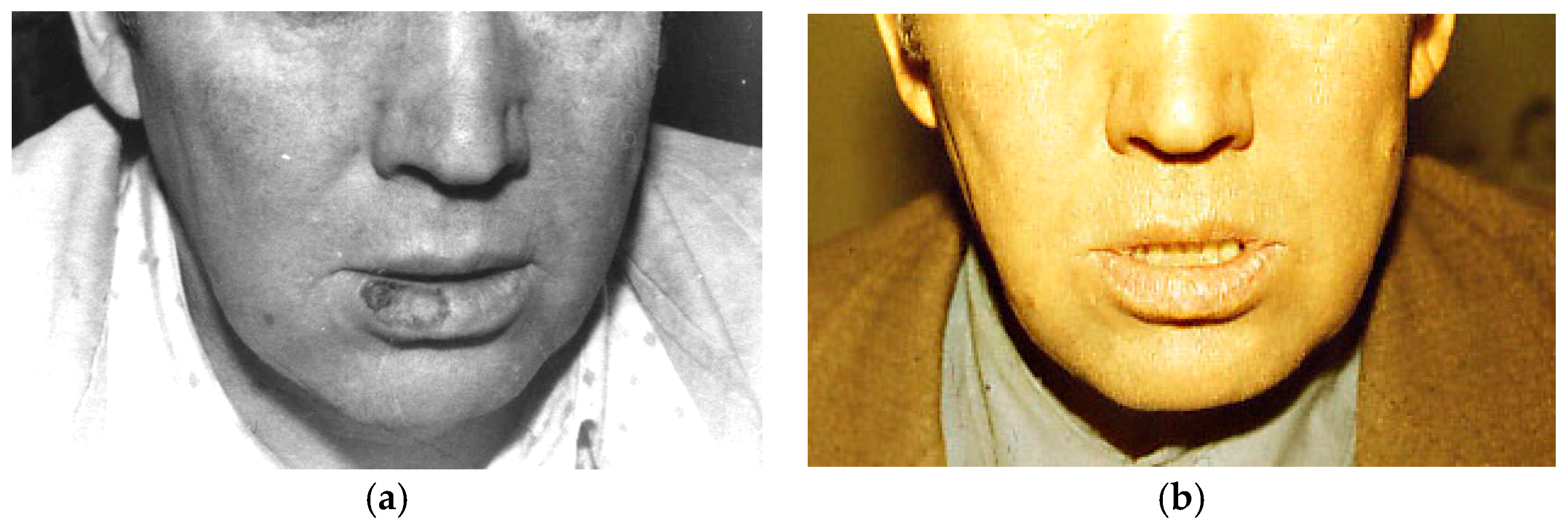
6.5. Cryotools Made of PTN
7. Conclusions
- (i)
- The biomechanical compatibility is referred to as the similarity of viscoelastic rheological characteristics between the PTN implant and host tissues. The combination of toughness inherent in Ti-based alloys, the porous morphology, and the viscoelastic reversible behavior of the porous body emphasizes the potential of PTN alloys to redistribute well physiological loads even in the early postoperative period, allowing to circumvent obstacles faced by existing implants;
- (ii)
- The biochemical compatibility has turned out to be successful as well. The bioinertness of surface and inferential bioactivity evidenced through cytocompatibility and negligible foreign body reaction owe much to self-assembled superficial layers resulted from the SHS process and, as such, the as-received PTN implant does not require further surface modification;
- (iii)
- Multifarious superficial layers demonstrating a complex structure/composition and high corrosion resistance conceal the matrix entirely and can be congruentially deformed without rupture and delamination, withstanding multicycle alternating loads;
- (iv)
- The in vivo performance of PTN bone substitutes is also high. They may go through 107 cycles with no failure due to the fact that chemical-proof layers arrest the surface deterioration, whereas the superelastic behavior of the matrix at alternating load rules out a possibility of the early material’s degradation.
- (v)
- A large number of PTN devices have been clinically applied in traumatic/orthopedic surgery, maxillofacial surgery, spinal surgery, etc., due to a rare combination of structure, mechanical, and physicochemical properties of PTN as a biomaterial. Moreover, bioengineering can consider customized PTN grafts and PTN-based surgical techniques in the context of the next generation implants concerning surgery cost minimization and improved patient tolerance.
- (vi)
- Comparative studies on corrosion fatigue behaviors of porous Ti and TiNi alloys made by both SHS and sintering are further needed to accomplish a complete and systematic understanding of PTN as an advanced biomaterial which can serve multiple clinical purposes.
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Buehler, W.; Gilfrich, J.; Wiley, R. Effect of low-temperature phase changes on mechanical properties of alloys near composition TiNi. J. Appl. Phys. 1963, 34, 1475–1477. [Google Scholar] [CrossRef]
- Gunther, V.; Kotenko, V.; Mirgazizov, M.; Polenichkin, V.; Vitugov, V.; Itin, V.; Ziganshin, R.; Temerhanov, F. Application of Shape Memory Alloys in Medicine; TSU Publishing House: Tomsk, Russia, 1986; 208p, Available online: http://www.sme-implant.com/ccount/click.php?id=48 (accessed on 3 June 2019).
- Gunther, V.; Dambaev, G.; Sysoliatin, P.; Ziganshin, R.; Kornilov, N.; Mirgasisov, M.; Mironov, S.; Fomichev, N.; Hodorenko, V.; Temerkhanov, F.; et al. Delay Law and New Class of Materials and Implants in Medicine; STT: Northampton, UK, 2000; 432p, Available online: http://www.sme-implant.com/ccount/click.php?id=2 (accessed on 3 June 2019).
- Ryhanen, J.; Kallioinen, M.; Tuukkanen, J.; Junila, J.; Niemela, E.; Sandvik, P.; Serlo, W. In vivo biocompatibility evaluation of nickel–titanium shape memory metal alloy: Muscle and perineural tissue responses and encapsule membrane thickness. J. Biomed. Mater. Res. 1998, 41, 481–488. [Google Scholar] [CrossRef]
- Drugacz, J.; Lekston, Z.; Morawiec, H.; Januszewski, K. Use of TiNi shape-memory clamps in the surgical treatment of mandibular fractures. J. Oral. Maxillofac. Surg. 1995, 53, 665–671. [Google Scholar] [CrossRef]
- Ryhanen, J.; Niemi, E.; Serlo, W.; Niemel, E.; Sandvik, P.; Pernu, H.; Salo, T. Biocompatibility of nickel-titanium shape memory metal and its corrosion behavior in human cell cultures. J. BioMed Mater. Res. 1998, 35, 451–457. [Google Scholar] [CrossRef]
- Jani, J.; Leary, M.; Subic, A.; Gibson, M. A review of shape memory alloy research, applications and opportunities. Mater. Des. 2014, 56, 1078–1113. [Google Scholar] [CrossRef]
- Prymak, O.; Bogdanski, D.; Koller, M.; Esenwein, S.A.; Muhr, G.; Beckmann, F.; Donath, T.; Assad, M.; Epple, M. Morphological characterization and in vitro biocompatibility of a porous nickel-titanium alloy. Biomaterials 2005, 26, 5801–5807. [Google Scholar] [CrossRef] [PubMed]
- Rhalmi, S.; Charette, S.; Assad, M.; Coillard, C.; Rivard, C.H. The spinal cord dura mater reaction to nitinol and titanium alloy particles: A 1-year study in rabbits. Eur. Spine J. 2007, 16, 1063–1072. [Google Scholar] [CrossRef]
- Gunther, V.; Sysoliatin, P.; Temerhanov, F.; Mirgazizov, M.; Pushkarev, V.; Tazin, I. Superelastic Shape Memory Implants in Maxillofacial Surgery, Traumatology, Orthopaedics, and Neurosurgery; Tsu Publishing House: Tomsk, Russia, 1995; 224p. [Google Scholar]
- Gunther, V.; Dambaev, G.; Sysoliatin, P.; Ziganshin, R.; Temerhanov, F.; Polenichkin, V.; Mirgazizov, M.; Pahomenko, G.; Savchenko, P.; Staroha, A. et al. Medical Materials and Shape Memory Implants; Tsu Publishing House: Tomsk, Russia, 1998; 487p. [Google Scholar]
- Gunther, V. Shape Memory Implants. Medical Atlas; STT: Northampton, UK, 2002; 234p, Available online: http://www.sme-implant.com/ccount/click.php?id=5 (accessed on 3 June 2019).
- Itin, V.; Gunther, V.; Khachin, V.; Bratchikov, A. Production of titanium nickelide by self-propagating high-temperature synthesis. Powder Metall. Metal. Ceram. 1983, 22, 156–157. [Google Scholar] [CrossRef]
- Li, B.; Rong, L.; Li, Y.; Gjunter, V. Synthesis of porous Ni-Ti shape memory alloys by self-propagating high-temperature synthesis: Reaction mechanism and anisotropy in pore structure. Acta Mater. 2000, 48, 3895–3904. [Google Scholar] [CrossRef]
- Maslov, V.; Borovinskaya, I.; Merzhanov, A. Problem of mechanism of gasless combustion. Combust. Explos. Shock. Waves 1976, 12, 631–636. [Google Scholar] [CrossRef]
- Merzhanov, A. Combustion processes that synthesize materials. J. Mater. Process. Technol. 1996, 56, 222–241. [Google Scholar] [CrossRef]
- Borovinskaya, I.; Gromov, A.; Levashov, E.; Maksimov, Y.; Mukasyan, A.; Rogachev, A. Concise Encyclopedia of Self-Propagating High-Temperature Synthesis History, Theory, Technology, and Products, 1st ed.; Elsevier: Amsterdam, The Netherlands, 2017; 437p. [Google Scholar]
- Li, B.; Rong, L.; Li, Y. Microstructure and superelasticity of porous NiTi alloy. Sci. China Ser. E Technol. Sci. 1999, 42, 94–99. [Google Scholar] [CrossRef]
- Parvizi, S.; Hasannaeimi, V.; Saebnoori, E.; Shahrabi, T.; Sadrnezhaad, S.K. Fabrication of porous NiTi alloy via powder metallurgy and its mechanical characterization by shear punch method. Russ. J. Non Ferr. Met. 2012, 53, 169–175. [Google Scholar] [CrossRef]
- Gunther, V.; Yasenchuk, Y.; Chekalkin, T.; Marchenko, E.; Gunther, S.; Baigonakova, G.; Hodorenko, V.; Kang, J.H.; Weiss, S.; Obrosov, A. Formation of pores and amorphous-nanocrystalline phases in porous TiNi alloys made by self-propagating high-temperature synthesis (SHS). Adv. Powder Technol. 2019, 30, 673–680. [Google Scholar] [CrossRef]
- Yasenchuk, Y.; Gunther, V.; Marchenko, E.; Chekalkin, T.; Baigonakova, G.; Hodorenko, V.; Gunther, S.; Kang, J.H.; Weiss, S.; Obrosov, A. Formation of mineral phases in self-propagating high-temperature synthesis (SHS) of porous TiNi alloy. Mater. Res. Express 2019, 6, 056522. [Google Scholar] [CrossRef]
- Artyukhova, N.; Yasenchuk, Y.; Chekalkin, T.; Gunther, V.; Kim, J.S.; Kang, J.H. Structure and properties of porous TiNi(Co, Mo)-based alloy produced by reaction sintering. Smart Mater. Struct. 2016, 25, 107003. [Google Scholar] [CrossRef]
- Corbin, S.F.; Cluff, D. Determining the rate of (β-Ti) decay and its influence on the sintering behavior of NiTi. J. Alloys Compd. 2009, 487, 179–186. [Google Scholar] [CrossRef]
- Novak, P.; Pokorny, P.; Vojtech, V.; Knaislova, A.; Skolakova, A.; Capek, J.; Karlik, M.; Kopecek, J. Formation of Ni-Ti intermetallics during reactive sintering at 500–650 °C. Mater. Chem. Phys. 2015, 155, 113–121. [Google Scholar] [CrossRef]
- Jasenchuk, Y.; Gjunter, V. Anode polarization of NiTi alloy in HCl solution. In Proceedings of the 1st International Symposium on Advanced Biomaterials (ISAB), Montreal, QC, Canada, 2–5 October 1997; pp. 41–42. [Google Scholar]
- Munroe, N.; Pulletikurthi, C.; Haider, W. Enhanced biocompatibility of porous Nitinol. J. Mater. Eng. Perform. 2009, 18, 765–767. [Google Scholar] [CrossRef]
- Bansiddhi, A.; Sargeant, T.; Stupp, S.; Dunand, D. Porous NiTi for bone implants: A review. Acta Biomater. 2008, 4, 773–782. [Google Scholar] [CrossRef]
- Schrooten, J.; Assad, M.; Humbeeck, J.V.; Leroux, M. In vitro corrosion resistance of porous NiTi intervertebral fusion. Smart Mater. Struct. 2007, 16, S145. [Google Scholar] [CrossRef]
- Gyunter, V.E.; Yasenchuk, Y.F.; Klopotov, A.A.; Khodorenko, V.N. The physicomechanical properties and structure of superelastic porous titanium nickelide-based alloys. Tech. Phys. Lett. 2000, 26, 35–37. [Google Scholar] [CrossRef]
- Bertheville, B.; Neudenberger, M.; Bidaux, J.E. Powder sintering and shape-memory behaviour of NiTi compacts synthesized from Ni and TiH2. Mater. Sci. Eng. A 2004, 384, 143–150. [Google Scholar] [CrossRef]
- Muhamedov, M.; Kulbakin, D.; Gunther, V.; Choynzonov, E.; Chekalkin, T.; Hodorenko, V. Sparing surgery with the use of TiNi-based endografts in larynx cancer patients. J. Surg. Oncol. 2015, 111, 231–236. [Google Scholar] [CrossRef]
- Fomichev, N.; Gunther, V.; Kornilov, N.; Simonovich, A.; Hodorenko, V.; Sizikov, M.; Savchenko, P.; Pahomenko, G.; Yasenchuk, Yu.; Kolumb, V. Novel Techniques in Spinal Care Using Porous Shape Memory Implants; STT: Northampton, UK, 2002; 130p, Available online: http://www.sme-implant.com/ccount/click.php?id=6 (accessed on 3 June 2019).
- Li, J.; Chen, D.; Luan, H.; Zhang, Y.; Fan, Y. Numerical evaluation and prediction of porous implant design and flow performance. BioMed Res. Int. 2018, 12, 1215021. [Google Scholar] [CrossRef]
- Saini, M.; Singh, Y.; Arora, P.; Jain, K. Implant biomaterials: A comprehensive review. World J. Clin. Cases 2015, 3, 52–57. [Google Scholar] [CrossRef]
- Lemons, J.E. (Ed.) Quantitative Characterization and Performance of Porous Implants for Hard Tissue Applications; ASTM International: West Conshohocken, PA, USA, 1987; 410p. [Google Scholar] [CrossRef]
- Novak, P.; Moravec, H.; Salvetr, P.; Prusa, F.; Drahokoupil, J.; Kopecek, J.; Karlik, M.; Kubatiket, T. Preparation of nitinol by non-conventional powder metallurgy techniques. Mater. Sci. Technol. 2015, 31, 1886–1893. [Google Scholar] [CrossRef]
- Abidi, I.H.; Khalid, F.A.; Farooq, M.U.; Hussain, M.U.; Maqbool, A. Tailoring the pore morphology of porous nitinol with suitable mechanical properties for biomedical applications. Mater. Lett. 2015, 154, 17–20. [Google Scholar] [CrossRef]
- Tavadze, G.F.; Shteinberg, A.S. Production of Advanced Materials by Methods of Self-Propagating High-Temperature Synthesis; Springer: Heidelberg, Germany, 2013; 156p. [Google Scholar] [CrossRef]
- Whitney, M.; Corbin, S.F.; Gorbet, R.B. Investigation of the mechanisms of reactive sintering and combustion synthesis of NiTi using differential scanning calorimetry and microstructural analysis. Acta Mater. 2008, 56, 559–570. [Google Scholar] [CrossRef]
- Whitney, M.; Corbin, S.F.; Gorbet, R.B. Investigation of the influence of Ni powder size on microstructural evolution and the thermal explosion combustion synthesis of NiTi. Intermetallics 2009, 17, 894–906. [Google Scholar] [CrossRef]
- Artyukhova, N.; Yasenchuk, Y.; Kim, J.S.; Gunther, V. Reaction sintering of porous shape memory titanium−nickelide-based alloys. Russ. Phys. J. 2015, 57, 1313–1320. [Google Scholar] [CrossRef]
- Yasenchuk, Y.; Gjunter, V. Surface structure of porous titanium nickelide produced by SHS. Russ. Phys. J. 2008, 51, 851–857. [Google Scholar] [CrossRef]
- Yasenchuk, Y.; Artyukhova, N.; Almaeva, K.; Garin, A.; Gunther, V. Segregation in porous NiTi made by SHS in flow reactor. In Proceedings of the Shape Memory Biomaterials and Implants in Medicine (SMBIM), Pusan, South Korea, 1–3 May 2017; KnE Materials Science: Dubai, UAE; pp. 168–175. [Google Scholar] [CrossRef]
- Kokorev, O.; Khodorenko, V.; Baigonakova, G.; Marchenko, E.; Yasenchuk, Y.; Gunther, V.; Anikeev, S.; Barashkova, G. Metal-glass-ceramic phases on the surface of porous TiNi-based SHS-material for carriers of cells. Russ. Phys. J. 2019, 61, 1734–1740. [Google Scholar] [CrossRef]
- Assad, M.; Jarzem, P.; Leroux, M.; Coillard, C.; Chernyshov, A.; Charette, S.; Rivard, C.H. Porous titanium-nickel for intervertebral fusion in a sheep model: Part 1. Histomorphometric and radiological analysis. J. BioMed Mater. Res. 2003, 64, 107–120. [Google Scholar] [CrossRef]
- Kang, S.B.; Yoon, K.S.; Kim, J.S.; Nam, T.H.; Gjunter, V.E. In vivo result of porous TiNi shape memory alloy: Bone response and growth. Mater. Trans. 2002, 43, 1045–1048. [Google Scholar] [CrossRef]
- Kokorev, O.; Hodorenko, V.; Chekalkin, T.; Kim, J.S.; Kang, S.B.; Dambaev, G.; Gunther, V. In vitro and in vivo evaluation of porous TiNi-based alloy as a scaffold for cell tissue engineering. Artif. Cells Nanomed. Biotechnol. 2016, 44, 704–709. [Google Scholar] [CrossRef]
- Gunther, S.; Kokorev, O.; Chekalkin, T.; Hodorenko, V.; Dambaev, G.; Kang, J.H.; Gunther, V. Effects of infrared and ultraviolet radiation on the viability of cells immobilized in a porous TiNi-based alloy scaffold. Adv. Mater. Lett. 2015, 6, 774–778. [Google Scholar] [CrossRef]
- Kokorev, O.; Hodorenko, V.; Chekalkin, T.; Gunther, V.; Kang, S.B.; Chang, M.J.; Kang, J.H. Evaluation of allogenic hepato-tissue engineered in porous TiNi-based scaffolds for liver regeneration in a CCl4-induced cirrhosis rat model. BioMed Phys. Eng. Express 2019, 5, 025018. [Google Scholar] [CrossRef]
- Prasad, K.; Bazaka, O.; Chua, M.; Rochford, M.; Fedrick, L.; Spoor, J.; Symes, R.; Tieppo, M.; Collins, C.; Cao, A.; et al. Metallic biomaterials: Current challenges and opportunities. Materials 2017, 10, 884. [Google Scholar] [CrossRef]
- Prakash, C.; Singh, S.; Pruncu, C.I.; Mishra, V.; Krolczyk, G.; Pimenov, D.Y.; Pramanik, A. Surface modification of Ti-6Al-4V alloy by electrical discharge coating process using partially sintered Ti-Nb electrode. Materials 2019, 12, 1006. [Google Scholar] [CrossRef]
- Mah, D.; Pelletier, M.H.; Lovric, V.; Walsh, W.R. Corrosion of 3D-printed orthopaedic implant materials. Ann. BioMed Eng. 2019, 47, 162–173. [Google Scholar] [CrossRef]
- Yuan, B.; Zhu, M.; Chung, C.Y. Biomedical porous shape memory alloys for hard-tissue replacement materials. Materials 2018, 11, 1716. [Google Scholar] [CrossRef]
- Marchenko, E.S.; Yasenchuk, Y.F.; Gunther, V.E.; Dubovikov, K.M.; Khodorenko, V.N. The comparative structural-phase analysis of the surface of macro and microporous SHS TiNi. In Proceedings of the International Conference. on Shape Memory and Superelastic Technologies (SMST-2019), Konstanz, Germany, 13–17 May 2019; ASM International: Materials Park, OH, USA, 2019; pp. 57–58. [Google Scholar]
- Baigonakova, G.A.; Yasenchuk, Y.F.; Gunther, S.V.; Kokorev, O.V.; Gunther, V.E. Structural-phase and morphological features of the surface layers of the SHS TiNi. In Proceedings of the International Conference on Shape Memory and Superelastic Technologies (SMST-2019), Konstanz, Germany, 13–17 May 2019; ASM International: Materials Park, OH, USA, 2019; pp. 53–54. [Google Scholar]
- Buchwitz, M.; Adlwarth-Dieball, R.; Ryder, P.L. Kinetics of the crystallization of amorphous Ti2Ni. Acta Metall. Mater. 1993, 41, 1885–1892. [Google Scholar] [CrossRef]
- Hashimoto, K.; Kumagai, N.; Yoshioka, H.; Kim, J.H.; Akiyama, E.; Habazaki, H.; Mrowec, S.; Kawashima, A.; Asamia, K. Corrosion-resistant amorphous surface alloys. Corros. Sci. 1993, 35, 363–370. [Google Scholar] [CrossRef]
- Maccabi, A.; Shin, A.; Namiri, N.K.; Bajwa, N.; John, M.S.; Taylor, Z.D.; Grundfest, W.; Saddik, G.N. Quantitative characterization of viscoelastic behavior in tissue-mimicking phantoms and ex vivo animal tissues. PLoS ONE 2018, 13, 0191919. [Google Scholar] [CrossRef]
- Karathanasopoulos, N.; Arampatzis, G.; Ganghoffer, J.F. Unravelling the viscoelastic, buffer-like mechanical behavior of tendons: A numerical quantitative study at the fibril-fiber scale. J. Mech. Behav. BioMed Mater. 2019, 90, 256–263. [Google Scholar] [CrossRef]
- Evans, F.G.; Lebow, M. Regional differences in some of the physical properties of the human femur. J. Appl. Phys. 1951, 3, 563–572. [Google Scholar] [CrossRef]
- Hirsch, C.; Sonnerup, L. Macroscopic rheology in collagen material. J. Biomech. 1968, 1, 13–18. [Google Scholar] [CrossRef]
- Unal, M.; Creecy, A.; Nyman, J.S. The role of matrix composition in the mechanical behavior of bone. Curr. Osteoporos Rep. 2018, 16, 205–215. [Google Scholar] [CrossRef]
- Lu, X.; Wang, C.; Li, G.; Liu, Y.; Zhu, X.; Tu, S. The mechanical behavior and martensitic transformation of porous NiTi alloys based on geometrical reconstruction. Int. J. Appl. Mech. 2017, 9, 1–15. [Google Scholar] [CrossRef]
- Gunther, V.; Hodorenko, V.; Chekalkin, T.; Olesova, V.; Dambaev, G.; Sysoliatin, P.; Fomichev, N.; Mirgazizov, M.; Melnik, D.; Ivchenko, O.; et al. Medical Materials and Shape Memory Implants: Shape Memory Materials; MITS: Tomsk, Russia, 2011; Volume 1, 534p, Available online: http://www.sme-implant.com/ccount/click.php?id=14 (accessed on 3 June 2019).
- Perren, S.M.; Fernandez, A.; Regazzoni, P. Understanding fracture healing biomechanics based on the “strain” concept and its clinical applications. Acta Chir. Orthop. Traumatol. Cech. 2015, 82, 253–260. Available online: https://www.ncbi.nlm.nih.gov/pubmed/26516728 (accessed on 3 June 2019).
- Cole, J.H.; Van der Meulen, M.C. Whole bone mechanics and bone quality. Clin. Orthop. Relat. Res. 2011, 469, 2139–2149. [Google Scholar] [CrossRef]
- Marino, S.; Staines, K.A.; Brown, G.; Howard-Jones, R.A.; Adamczyk, M. Models of ex vivo explant cultures: Applications in bone research. Bonekey Rep. 2016, 5, 818. [Google Scholar] [CrossRef]
- Gyunter, V.; Khodorenko, V.; Monogenov, A.; Yasenchuk, Y. Effect of deformation on the permeability of porous titanium-nickel alloys. Technol. Phys. Lett. 2000, 26, 320–322. [Google Scholar] [CrossRef]
- Bassani, P.; Giuliani, P.; Tuissi, A.; Zanotti, C. Thermomechanical properties of porous NiTi alloy produced by SHS. J. Mater. Eng. Perform. 2009, 18, 594–599. [Google Scholar] [CrossRef]
- Resnina, N.; Belyaev, S.; Voronkov, A.; Gracheva, A. Mechanical behaviour and functional properties of porous Ti-45 at. % Ni alloy produced by self-propagating high-temperature synthesis. Smart Mater. Struct. 2016, 25, 055018. [Google Scholar] [CrossRef]
- Martin, C.L.; Favier, D.; Suery, M. Viscoplastic behaviour of porous metallic materials saturated with liquid part II: Experimental identification on a Sn-Pb model alloy. Int. J. Plast. 1997, 13, 237–259. [Google Scholar] [CrossRef]
- Itin, V.I.; Gyunter, V.E.; Shabalovskaya, S.A.; Sachdeva, L.C. Mechanical properties and shape memory of porous nitinol. Mater. Charact. 1994, 32, 179–187. [Google Scholar] [CrossRef]
- Greiner, C.; Oppenheimer, S.M.; Dunand, D.C. High strength, low stiffness, porous NiTi with superelastic properties. Acta Biomater. 2005, 1, 705–716. [Google Scholar] [CrossRef]
- Robertson, S.W.; Pelton, A.R.; Ritchie, R.O. Mechanical fatigue and fracture of Nitinol. Int. Mater. Rev. 2012, 57, 1–37. [Google Scholar] [CrossRef]
- Chakraborty, R.; Datta, S.; Raza, M.S.; Saha, P. A comparative study of surface characterization and corrosion performance properties of laser surface modified biomedical grade Nitinol. Appl. Surf. Sci. 2019, 469, 753–763. [Google Scholar] [CrossRef]
- Mehta, K.; Gupta, K. Fabrication and Processing of Shape Memory Alloys; Springer: Cham, Switzerland, 2019; 86p. [Google Scholar] [CrossRef]
- Khodaei, M.; Meratian, M.; Savabic, O.; Fathib, M.; Ghomi, H. The side effects of surface modification of porous titanium implant using hydrogen peroxide: Mechanical properties aspects. Mater. Lett. 2016, 178, 201–204. [Google Scholar] [CrossRef]
- Yeung, K.W.; Poon, R.W.; Liu, X.Y.; Ho, J.P.; Chung, C.Y.; Chu, P.K.; Lu, W.W.; Chan, D.; Cheung, K.M. Corrosion resistance, surface mechanical properties, and cytocompatibility of plasma immersion ion implantation–treated nickel-titanium shape memory alloys. J. BioMed Mater. Res. 2005, 75, 256–267. [Google Scholar] [CrossRef]
- Robinson, E.; Gaillard-Campbell, D.; Gross, T.P. Acetabular debonding: An investigation of porous coating delamination in hip resurfacing arthroplasty. Adv. Orthop. 2018, 2018, 5282167. [Google Scholar] [CrossRef]
- Intranuovo, F.; Gristina, R.; Brun, F.; Mohammadi, S.; Ceccone, G.; Sardella, E.; Rossi, F.; Tromba, G.; Favia, P. Plasma modification of pcl porous scaffolds fabricated by solvent-casting/particulate-leaching for tissue engineering. Plasma Process. Polym. 2014, 11, 184–195. [Google Scholar] [CrossRef]
- Wang, Q.; Cheng, M.; He, G.; Zhang, X. Surface modification of porous titanium with microarc oxidation and its effects on osteogenesis activity in vitro. J. Nanomater. 2015, 2015, 408634. [Google Scholar] [CrossRef]
- Seah, K.H.; Thampuran, R.; Chen, X.; Teoh, S.H. A comparison between the corrosion behaviour of sintered and unsintered porous titanium. Corros. Sci. 1995, 35, 1333–1340. [Google Scholar] [CrossRef]
- Sun, X.T.; Kang, Z.X.; Zhang, X.L.; Jiang, X.J.; Guan, R.F.; Zhang, X.P. A comparative study on the corrosion behavior of porous and dense NiTi shape memory alloys in NaCl solution. Electrochim. Acta. 2011, 56, 6389–6396. [Google Scholar] [CrossRef]
- Aihara, H.; Zider, J.; Fanton, G.; Duerig, T. Combustion synthesis porous nitinol for biomedical applications. Int. J. Biomater. 2019, 2019, 4307461. [Google Scholar] [CrossRef]
- Stergioudi, F.; Vogiatzis, C.A.; Pavlidou, E.; Skolianos, S.; Michailidis, N. Corrosion resistance of porous NiTi biomedical alloy in simulated body fluids. Smart Mater. Struct. 2016, 25, 095024. [Google Scholar] [CrossRef]
- Assad, M.; Jarzem, P.; Leroux, M.A.; Coillard, C.; Chernyshov, A.V.; Charette, S.; Rivard, C.H. Porous titanium-nickel for intervertebral fusion in a sheep model: Part 2. Surface analysis and nickel release assessment. J. BioMed Mater. Res. B 2003, 64, 107–120. [Google Scholar] [CrossRef]
- Williams, D.F. Fundamental aspects of biocompatibility. In Tissue Engineering; Elsevier: Amsterdam, The Netherlands, 2008; pp. 255–278. [Google Scholar] [CrossRef]
- Rhalmi, S.; Odin, M.; Assad, M.; Tabrizian, M.; Rivard, C.H.; Yahia, L.H. Hard, soft tissue and in vitro cell response to porous nickel-titanium: A biocompatibility evaluation. BioMed Mater. Eng. 1999, 9, 151–162. Available online: https://www.ncbi.nlm.nih.gov/pubmed/10572619 (accessed on 3 June 2019).
- Mjoberg, B. Theories of wear and loosening in hip prostheses: Wear-induced loosening vs loosening-induced wear—A review. Acta Orthop. Scand. 1994, 65, 361–371. [Google Scholar] [CrossRef]
- Mitra, J.; Tripathi, G.; Sharma, A.; Basu, B. Scaffolds for bone tissue engineering: Role of surface patterning on osteoblast response. RSC Adv. 2013, 3, 11073. [Google Scholar] [CrossRef]
- Hatano, K.; Inoue, H.; Kojo, T.; Matsunaga, T.; Tsujisawa, T.; Uchiyama, C.; Uchida, Y. Effect of surface roughness on proliferation and alkaline phosphatase expression of rat calvarial cells cultured on polystyrene. Bone 1999, 25, 439–445. [Google Scholar] [CrossRef]
- Gu, Y.; Li, H.; Tay, B.; Lim, C.; Yong, M.; Khor, K. In vitro bioactivity and osteoblast response of porous NiTi synthesized by SHS using nanocrystalline Ni-Ti reaction agent. J. BioMed Mater. Res. 2006, 78, 316–323. [Google Scholar] [CrossRef]
- Kim, J.S.; Kang, J.H.; Kang, S.B.; Yoon, K.S.; Kwon, Y.S. Porous TiNi biomaterial by self-propagating high-temperature synthesis. Adv. Eng. Mater. 2004, 6, 403–406. [Google Scholar] [CrossRef]
- Wu, S.; Liu, X.; Chan, Y.; Ho, J.; Chung, C.; Chu, P.K.; Chu, C.; Yeung, K.; Lu, W.; Cheung, K.; et al. Nickel release behavior, cytocompatibility, and superelasticity of oxidized porous single-phase NiTi. J. BioMed Mater. Res. 2007, 81, 948–955. [Google Scholar] [CrossRef]
- Ryan, G.; Pandit, A.; Apatsidis, D. Biomaterials porous titanium scaffolds fabricated using a rapid prototyping and powder metallurgy technique. Biomaterials 2008, 29, 3625–3635. [Google Scholar] [CrossRef]
- Karageorgiou, V.; Kaplan, D. Porosity of 3D biomaterial scaffolds and osteogenesis. Biomaterials 2005, 26, 5474–5491. [Google Scholar] [CrossRef]
- Murphy, C.; Haugh, M.G.; O’Brien, F.J. The effect of mean pore size on cell attachment, proliferation and migration in collagen–glycosaminoglycan scaffolds for bone tissue engineering. Biomaterials 2010, 31, 461–466. [Google Scholar] [CrossRef]
- Radkevich, A.; Hodorenko, V.; Gunther, V. Reparative osteogenesis in bone defectes substituted by porous TiNi. Shape Memory Implant. 2005, 2, 30–35. Available online: http://www.sme-implant.com/ccount/click.php?id=38 (accessed on 3 June 2019).
- Radkevich, A.A. Reconstructive Surgery of Alveolar Process. Ph.D. Thesis, Irkutsk State University, Irkutsk Oblast, Russia, 6 May 2002. Available online: http://medical-diss.com/docreader/479398/a#?page=2 (accessed on 3 June 2019).
- Oxland, T.R. Fundamental biomechanics of the spine—What we have learned in the past 25 years and future directions. J. Biomech. 2016, 49, 817–832. [Google Scholar] [CrossRef]
- Fomichev, N.; Gunther, V.; Lutsik, A.; Sergeev, K.; Kornilov, N.; Shevtsov, V.; Durov, M.; Pahomenko, G.; Savchenko, P.; Ratkin, I.; et al. Medical Materials and Shape Memory Implants: Shape Memory Implants in Spinal Surgery; MITS: Tomsk, Russia, 2011; Volume 3, 374p, Available online: http://www.sme-implant.com/ccount/click.php?id=16 (accessed on 3 June 2019).
- Sysoliatin, P.; Gunther, V.; Sysoliatin, S.; Mirgazizov, M.; Radkevich, A.; Olesova, V.; Hodorenko, V.; Duriagin, N.; Melnik, D.; Tazin, I.; et al. Medical Materials and Shape Memory Implants: Shape Memory Implants in Maxillo-Facial Surgery; MITS: Tomsk, Russia, 2012; Volume 4, 384p, Available online: http://www.sme-implant.com/ccount/click.php?id=17 (accessed on 3 June 2019).
- Mirgazizov, M.; Gunther, V.; Galonskiy, V.; Olesova, V.; Radkevich, A.; Hafizov, R.; Mirgazizov, R.; Yudin, P.; Starosvetskiy, S.; Zvigintsev, M.; et al. Medical Materials and Shape Memory Implants: Shape Memory Implants in Dentistry; MITS: Tomsk, Russia, 2011; Volume 5, 220p, Available online: http://www.sme-implant.com/ccount/click.php?id=18 (accessed on 3 June 2019).
- Choinzonov, E.; Gunther, V.; Muhamedov, M.; Novikov, V.; Anisenia, I.; Cherdyntseva, N.; Kolomiets, L.; Chernysheva, A.; Slonimskaya, E.; Tuzikov, S.; et al. Medical Materials and Shape Memory Implants: Shape Memory Implants in Oncology; MITS: Tomsk, Russia, 2013; Volume 13, 336p, Available online: http://www.sme-implant.com/ccount/click.php?id=26 (accessed on 3 June 2019).
- Shtin, V.I. Repair of Sub-Cranial Lesions in Cancer Patients Using Nitinol Implants and Radiotherapy. Ph.D. Thesis, Tomsk Cancer Research Institute, Tomsk, Russia, 9 December 2010. Available online: https://search.rsl.ru/ru/record/01004616721 (accessed on 3 June 2019).
- Lanshakov, V.; Gunther, V.; Plotkin, G.; Fomichev, N.; Savchenko, P.; Fominyh, A.; Maslikov, V.; Petrov, L.; Melnik, D.; Baranov, G.; et al. Medical Materials and Shape Memory Implants: Shape Memory Implants in Traumatic and Orthopaedic Surgery; MITS: Tomsk, Russia, 2010; Volume 2, 384p, Available online: http://www.sme-implant.com/ccount/click.php?id=15 (accessed on 3 June 2019).
- Fominyh, A.; Goryachev, A. Reconstructive hand surgery using shape memory materials. In Biocompatible Materials and Shape Memory Implants; STT: Northampton, UK, 2001; pp. 142–156. Available online: http://www.sme-implant.com/ccount/click.php?id=2 (accessed on 3 June 2019).
- Melnik, D.; Gunther, V.; Dambaev, G.; Chuguy, E.; Hodorenko, V.; Sysoliatin, P.; Mahnev, A.; Tokmakova, S.; Musin, V.; Shkuratov, S.; et al. Medical Materials and Shape Memory Implants: Porous NiTi Tools in Clinical Practice; MITS: Tomsk, Russia, 2010; Volume 9, 306p, Available online: http://www.sme-implant.com/ccount/click.php?id=22 (accessed on 3 June 2019).
- Shkuratov, S.; Gunther, V.; Dambaev, G.; Malkova, E.; Isaenko, V.; Feofilov, I.; Erkovich, I.; Shkuratov, S.; Davydov, A.; Yarin, G.; et al. Medical Materials and Shape Memory Implants: Shape Memory Implants in Urology; MITS: Tomsk, Russia, 2009; Volume 7, 248p, Available online: http://www.sme-implant.com/ccount/click.php?id=20 (accessed on 3 June 2019).
- Tlish, M.M.; Kolesnikova, V.N.; Gunter, V.E.; Steblyuk, A.N.; Marchenko, E.S.; Shavilova, M.E.; Tserkovnaya, A.A. Possibilities of using cryotherapy in patients with ocular rosacea. Ophthalmol. J. 2018, 11, 7–14. [Google Scholar] [CrossRef]
- Shtofin, S.; Gunther, V.; Anishenko, V.; Dambaev, G.; Shtofin, G.; Kulikova, L.; Merzlikin, N.; Volodos, N.; Sadovskiy, A.; Hodorenko, V.; et al. Medical Materials and Shape Memory Implants: Shape Memory Implants in Urology; MITS: Tomsk, Russia, 2013; Volume 12, 126p, Available online: http://www.sme-implant.com/ccount/click.php?id=25 (accessed on 3 June 2019).







| Property | Value |
|---|---|
| Specific weight, g/cm3 | 5.85 |
| Porosity, % | 60 to 75 |
| Pore size, μm | 0.1 to 200 |
| Permeability coefficient (water/glycerin), m2 | (0.27/62) × 10−9 |
| Melting point, °C | 1310 |
| Ultimate tensile strength, MPa | 100 to 500 |
| Stretch at breaking point, % | 5 to 7 |
| Loading plato stress, MPa | 50 to 200 |
| Total elongation, % | 2.5 to 4.5 |
| Permanent set, % | 5 to 20 |
| SME recovery stress, MPa | 200 to 400 |
| SME temperature hysteresis, degree | 30 to 100 |
| Transformation temperature range, °C | −180 to 50 |
| Clinical Field | Embodiment of PTN | Number of Cases | Note |
|---|---|---|---|
| Traumatic and orthopedic surgery | Plates, Bars, Round bars, Stripes, Tapers, Customized endografts, Pellets | 621 | Open/closed bone fracture—361 Traumatic bone/joint lesion—127 Posttraumatic joint contracture—62 False joint—38 Congenital bone abnormality—23 Ankylosis—10 |
| Spinal surgery | Customized cages (cylinder, bar, wedge etc.) | 1983 | Lumbar anterior/posterior interbody fusion: L5-S1—959; L4-5—791; L3-4—233 |
| 641 | Cervical anterior/posterior interbody fusion: C2-4—190; C4-7—451 | ||
| 257 | Spinal stenosis surgery: Lumbar—214; Cervical—43 | ||
| Maxillofacial surgery | Plates, Bars, Round bars, Tapers, Stripes, Customized endografts, Pellets | 409 | Radicular cyst—175 Ameloblastoma—81 Odontoma—56 Osteoma—33 Condylar joint replacement—29 Total/subtotal mandibular replacement—25 Giant-cell tumor—10 |
| Cryo-surgery/therapy | Cryotools having different size/shape/surface of the working part | 1314 | Cryodestruction of cutaneous and subcutaneous tumors (malignant, premalignant, non-malignant) |
| 200 | Cryodestruction of hemorrhoidal boluses | ||
| 138 | Cryotherapy of the obstructed urethra (urethral patency restoration) | ||
| <9000 | Cryotherapy of hemangioma in infants and pediatric patients | ||
| Oncosurgery | Plates, Disks, Round bars, Stripes, Customized endografts, Pellets | 617 | Bone/joint post-resection repair—322 Head and neck sparing surgery—295 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yasenchuk, Y.; Marchenko, E.; Gunther, V.; Radkevich, A.; Kokorev, O.; Gunther, S.; Baigonakova, G.; Hodorenko, V.; Chekalkin, T.; Kang, J.-h.; et al. Biocompatibility and Clinical Application of Porous TiNi Alloys Made by Self-Propagating High-Temperature Synthesis (SHS). Materials 2019, 12, 2405. https://doi.org/10.3390/ma12152405
Yasenchuk Y, Marchenko E, Gunther V, Radkevich A, Kokorev O, Gunther S, Baigonakova G, Hodorenko V, Chekalkin T, Kang J-h, et al. Biocompatibility and Clinical Application of Porous TiNi Alloys Made by Self-Propagating High-Temperature Synthesis (SHS). Materials. 2019; 12(15):2405. https://doi.org/10.3390/ma12152405
Chicago/Turabian StyleYasenchuk, Yuri, Ekaterina Marchenko, Victor Gunther, Andrey Radkevich, Oleg Kokorev, Sergey Gunther, Gulsharat Baigonakova, Valentina Hodorenko, Timofey Chekalkin, Ji-hoon Kang, and et al. 2019. "Biocompatibility and Clinical Application of Porous TiNi Alloys Made by Self-Propagating High-Temperature Synthesis (SHS)" Materials 12, no. 15: 2405. https://doi.org/10.3390/ma12152405
APA StyleYasenchuk, Y., Marchenko, E., Gunther, V., Radkevich, A., Kokorev, O., Gunther, S., Baigonakova, G., Hodorenko, V., Chekalkin, T., Kang, J.-h., Weiss, S., & Obrosov, A. (2019). Biocompatibility and Clinical Application of Porous TiNi Alloys Made by Self-Propagating High-Temperature Synthesis (SHS). Materials, 12(15), 2405. https://doi.org/10.3390/ma12152405







