Enhanced Oil Spill Remediation by Adsorption with Interlinked Multilayered Graphene
Abstract
:1. Introduction
2. Materials and Methods
2.1. Site Description
2.2. Adsorbent Materials
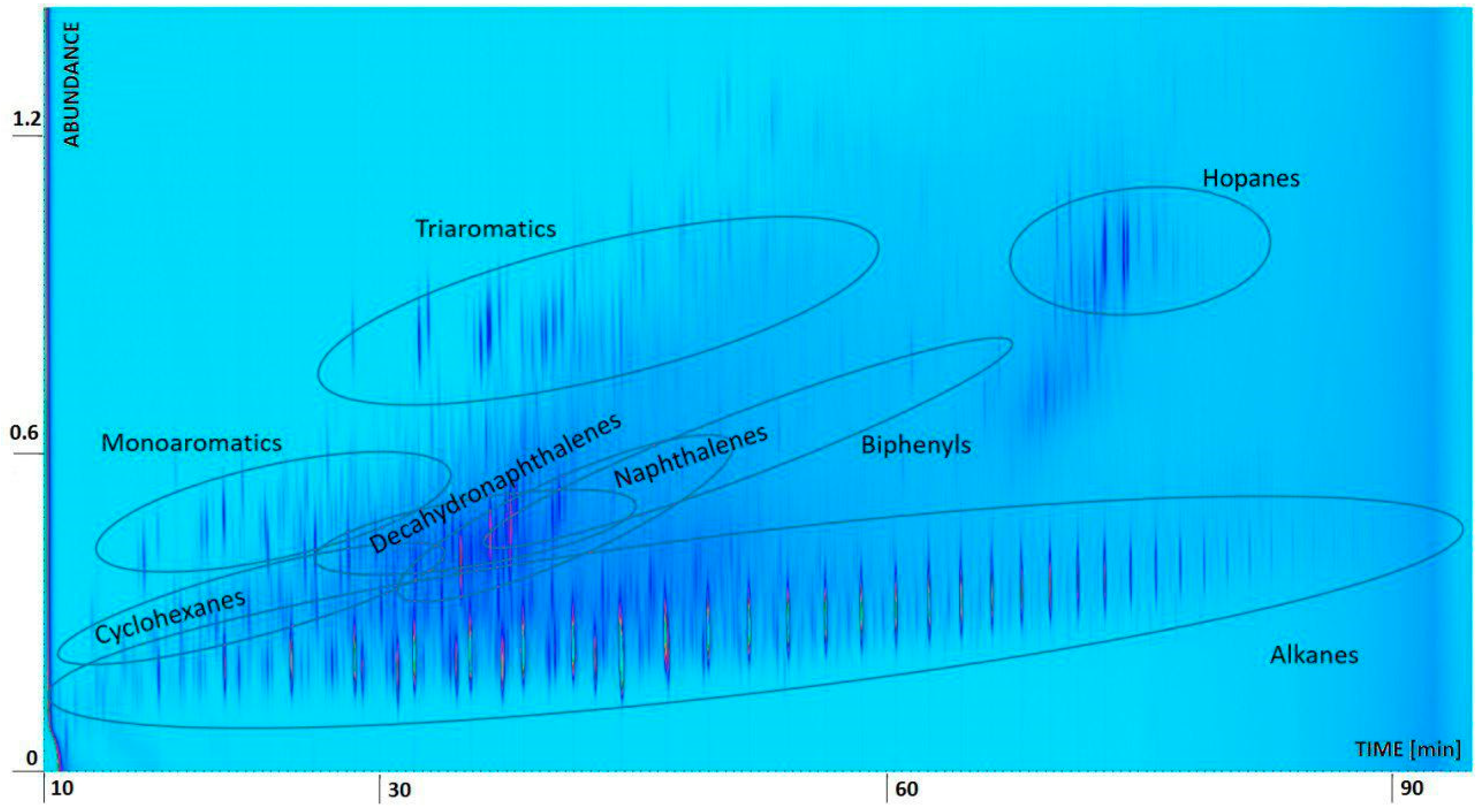
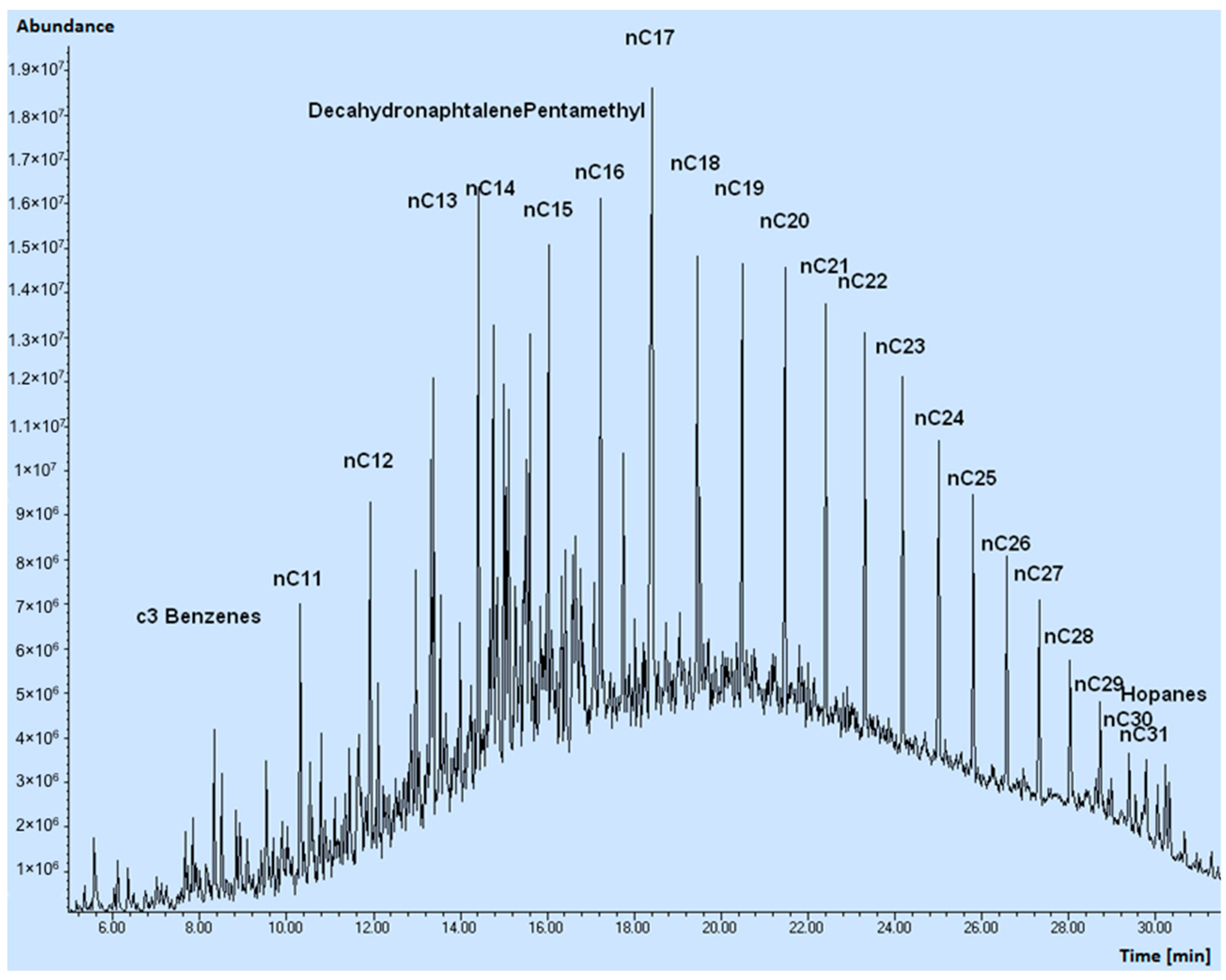
2.3. Oil Spill Composition
2.4. Experimental Campaign
3. Results and Discussion
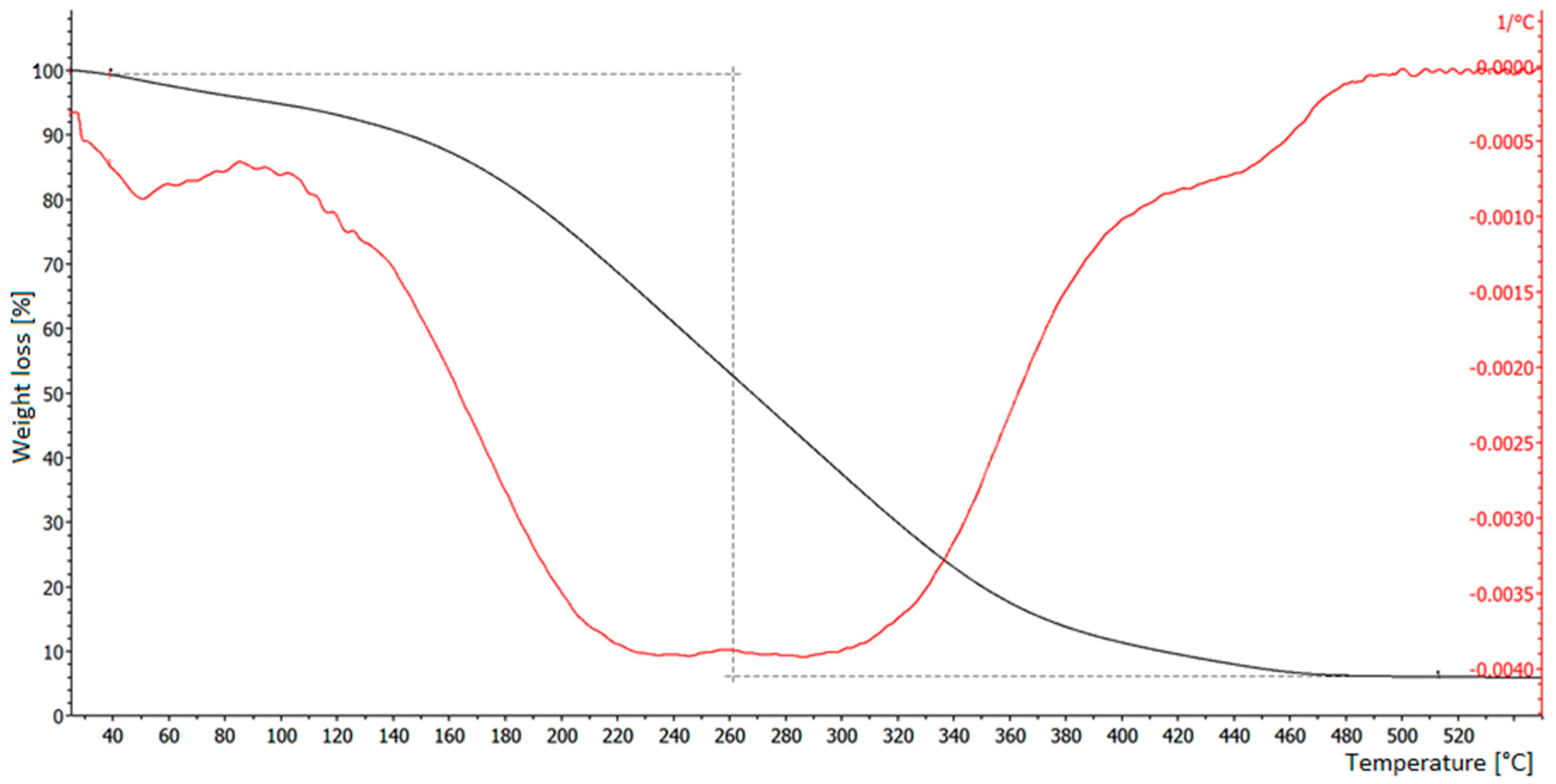
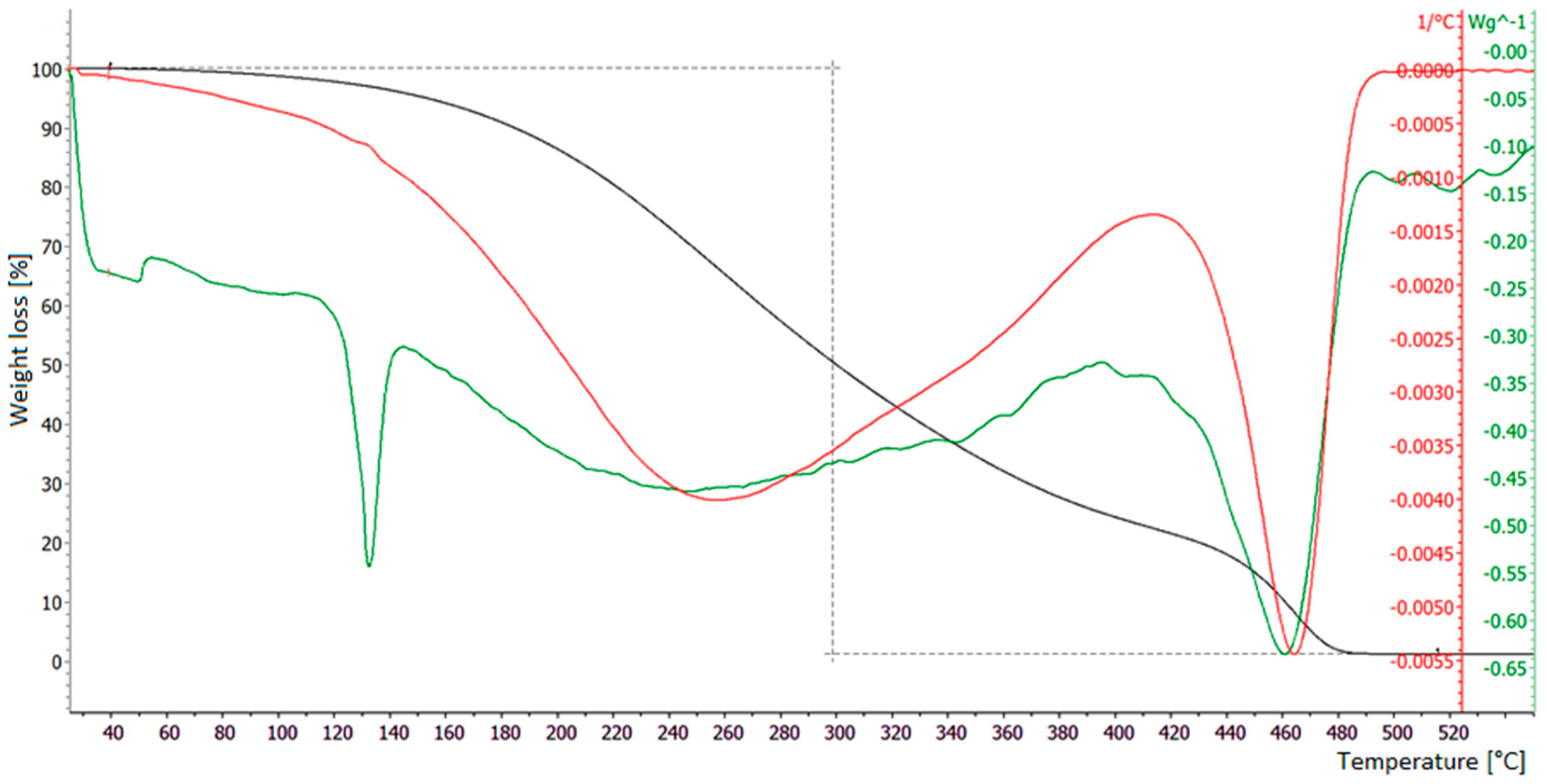
3.1. Thermogravimetric Analysis of Samples
3.2. TGA Tests on Grafysorber®
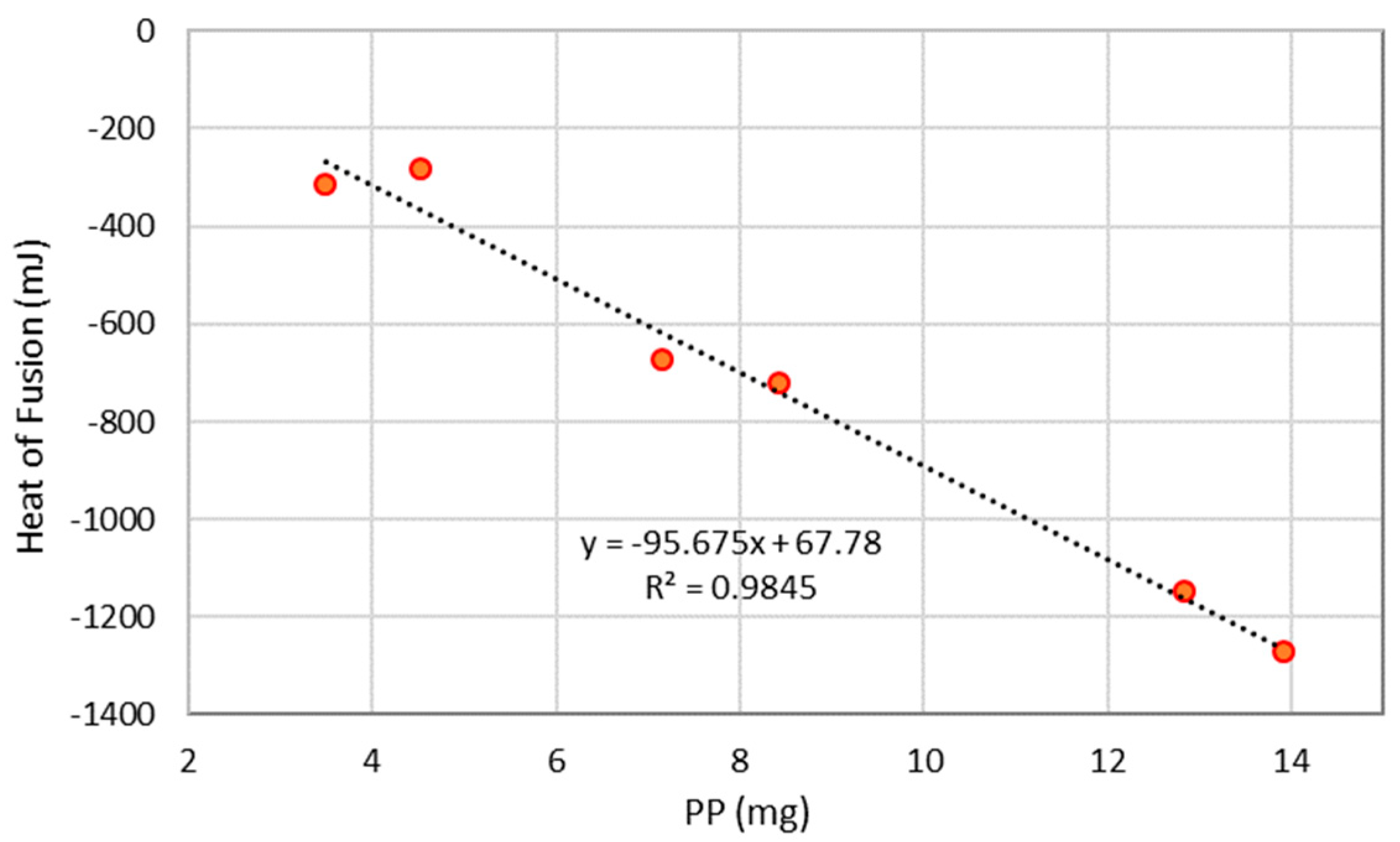
3.3. TGA Tests on Polypropylene
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Palazzi, E.; Currò, F.; Fabiano, B. Simplified modelling for risk assessment of hydrocarbon spills in port area. Process. Saf. Environ. Prot. 2004, 82, 412–420. [Google Scholar] [CrossRef]
- Barros, F.C.D.F.; Vasconcellos, L.C.G.; Carvalho, T.V.; Nascimento, R.F.D. Removal of petroleum spill in water by chitin and chitosan. Orbital: Electron. J. Chem. 2014, 6, 70–74. [Google Scholar]
- Vairo, T.; Del Giudice, T.; Quagliati, M.; Barbucci, A.; Fabiano, B. From land- to water-use-planning: A consequence based case-study related to cruise ship risk. Saf. Sci. 2017, 97, 120–133. [Google Scholar] [CrossRef]
- Vocciante, M.; Reverberi, A.P.; Dovì, V.G. Approximate solution of the inverse Richards’ problem. Appl. Math. Model. 2016, 40, 5364–5376. [Google Scholar] [CrossRef]
- Vocciante, M.; Reverberi, A.P.; Pietrelli, L.; Dovì, V.G. Improved Remediation Processes through Cost-effective Estimation of Soil Properties from Surface Measurements. J. Clean. Prod. 2017, 167, 680–686. [Google Scholar] [CrossRef]
- Vairo, T.; Magrì, S.; Qualgliati, M.; Reverberi, A.P.; Fabiano, B. An oil pipeline catastrophic failure: Accident scenario modelling and emergency response development. Chem. Eng. Trans. 2017, 57, 373–378. [Google Scholar]
- Bhardwaj, N.; Bhaskarwar, A.N. A review on sorbent devices for oil-spill control. Environ. Pollut. 2018, 243, 1758–1771. [Google Scholar] [CrossRef]
- Cutler, J.; Cleveland, C.J.; Saundry, P. Exxon Valdez Oil Spill. In Encyclopedia of Earth. 2010. Available online: https://editors.eol.org/eoearth/wiki/Exxon_Valdez_oil_spill (accessed on 16 May 2019).
- Al-Majed, A.A.; Adebayo, A.R.; Hossain, M.E. A sustainable approach to controlling oil spills. J. Environ. Manag. 2012, 113, 213–227. [Google Scholar] [CrossRef]
- Sethi, A.; Vyawahare, M.; Iyer, A. From Collision to Cover Up, the Inside Story of Chennai Oil Spill. 2017. Available online: https://www.hindustantimes.com/interactives/chennai-oil-spill-collision-cover-up/ (accessed on 16 May 2019).
- Vairo, T.; Magri, S.; De Gaetano, P.; Quagliati, M.; Fabiano, B. Multicomponent dispersion of hydrocarbons at sea: Source term evaluation and hydrodynamic simulation of the spill. Chem. Eng. Trans. 2018, 67, 61–66. [Google Scholar]
- Oebius, H.U. Physical properties and processes that influence the clean up of oil spills in the marine environment. Spill Sci. Technol. Bull. 1999, 5, 177–289. [Google Scholar] [CrossRef]
- Carmody, O.; Frost, R.; Xi, Y.; Kokot, S. Adsorption of hydrocarbons on organo-clays—Implications for oil spill remediation. J. Colloid Interface Sci. 2007, 305, 17–24. [Google Scholar] [CrossRef] [PubMed]
- Kyriakopoulos, G.; Doulia, D. Morphology of polymeric resins in adsorption of organic pesticides. Fresen. Environ. Bull. 2007, 16, 731–734. [Google Scholar]
- Vocciante, M.; D’Auris, A.D.F.; Finocchi, A.; Tagliabue, M.; Bellettato, M.; Ferrucci, A.; Reverberi, A.P.; Ferro, S. Adsorption of ammonium on clinoptilolite in presence of competing cations: Investigation on groundwater remediation. J. Clean. Prod. 2018, 198, 480–487. [Google Scholar] [CrossRef]
- Pietrelli, L.; Ippolito, N.M.; Ferro, S.; Dovì, V.G.; Vocciante, M. Removal of Mn and As from drinking water by red mud and pyrolusite. J. Environ. Manag. 2019, 237, 526–533. [Google Scholar] [CrossRef] [PubMed]
- Pietrelli, L.; Ferro, S.; Vocciante, M. Raw materials recovery from spent hydrochloric acid-based galvanizing wastewater. Chem. Eng. J. 2018, 341, 539–546. [Google Scholar] [CrossRef]
- Yati, I.; Aydin, G.O.; Sonmez, H.B. Cross-linked poly (tetrahydrofuran) as promising sorbent for organic solvent/oil spill. J. Hazard. Mater. 2016, 309, 210–218. [Google Scholar] [CrossRef] [PubMed]
- Toccafondi, C.; Dante, S.; Reverberi, A.P.; Salerno, M. Biomedical applications of anodic porous alumina. Curr. Nanosci. 2015, 11, 572–580. [Google Scholar] [CrossRef]
- Pham, V.H.; Dickerson, J.H. Superhydrophobic silanized melamine sponges as high efficiency oil absorbent materials. ACS Appl. Mater. Interfaces 2014, 6, 14181–14188. [Google Scholar] [CrossRef]
- Oribayo, O.; Feng, X.; Rempel, G.L.; Pan, Q. Synthesis of lignin-based polyurethane/graphene oxide foam and its application as an absorbent for oil spill clean-ups and recovery. Chem. Eng. J. 2017, 323, 191–202. [Google Scholar] [CrossRef]
- Feng, J.; Nguyen, S.T.; Fan, Z.; Duong, H.M. Advanced fabrication and oil absorption properties of super-hydrophobic recycled cellulose aerogels. Chem. Eng. J. 2015, 270, 168–175. [Google Scholar] [CrossRef]
- ITOPF. Use of Sorbent Materials in Oil Spill Response. 2014. Available online: http://www.itopf.com/knowledge-resources/documents-guides/document/tip-8-use-of-sorbent-materials-in-oil-spill-response/ (accessed on 11 May 2019).
- Fabiano, B.; Pistritto, F.; Reverberi, A.; Palazzi, E. Ethylene—Air mixtures under flowing conditions: A model-based approach to explosion conditions. Clean Technol. Environ. Policy 2015, 17, 1261–1270. [Google Scholar] [CrossRef]
- Solisio, C.; Reverberi, A.P.; Del Borghi, A.; Dovi’, V.G. Inverse estimation of temperature profiles in landfills using heat recovery fluids measurements. J. Appl. Math. 2012, 2012. [Google Scholar] [CrossRef]
- Toyoda, M.; Aizawa, J.; Inagaki, M. Sorption and recovery of heavy oil by using exfoliated graphite. Desalination 1998, 115, 199–201. [Google Scholar] [CrossRef]
- Toyoda, M.; Moriya, K.; Aizawa, J.; Konno, H.; Inagaki, M. Sorption and recovery of heavy oils by using exfoliated graphite Part 1: Maximum sorption capacity. Desalination 2000, 128, 205–211. [Google Scholar] [CrossRef]
- Inagaki, M.; Konno, H.; Toyoda, M.; Moriya, K.; Kihara, T. Sorption and recovery of heavy oils by using exfoliated graphite Part 2: Recovery of heavy oil and recycling of exfoliated graphite. Desalination 2000, 128, 213–218. [Google Scholar] [CrossRef]
- Inagaki, M.; Shibata, K.; Setou, S.; Toyoda, M.; Aizawa, J.; Kihara, T. Sorption and recovery of heavy oils by using exfoliated graphite Part 3: Trials for practical applications. Desalination 2000, 128, 219–222. [Google Scholar] [CrossRef]
- Toyoda, M.; Inagaki, M. Sorption and recovery of heavy oils by using exfoliated graphite. Spill Sci. Technol. Bull. 2003, 8, 467–474. [Google Scholar] [CrossRef]
- Yao, T.; Zhang, Y.; Xiao, Y.; Zhao, P.; Guo, L.; Yang, H.; Li, F. The effect of environmental factors on the adsorption of lubricating oil onto expanded graphite. J. Mol. Liq. 2016, 218, 611–614. [Google Scholar] [CrossRef]
- Pasila, A. A biological oil adsorption filter. Mar. Pollut. Bull. 2004, 49, 1006–1012. [Google Scholar] [CrossRef] [PubMed]
- Inagaki, M.; Toyoda, M.; Iwashita, N.; Nishi, Y.; Konno, H. Exfoliated graphite for spilled heavy oil recovery. Carbon Lett. 2001, 2, 1–8. [Google Scholar]
- Tryba, B.; Morawski, A.W.; Kaleńczuk, R.J.; Inagaki, M. Exfoliated graphite as a new sorbent for removal of engine oils from wastewater. Spill Sci. Technol. Bull. 2003, 8, 569–571. [Google Scholar] [CrossRef]
- Zheng, Y.P.; Wang, H.N.; Kang, F.Y.; Wang, L.N.; Inagaki, M. Sorption capacity of exfoliated graphite for oils-sorption in and among worm-like particles. Carbon 2004, 42, 2603–2607. [Google Scholar] [CrossRef]
- Shane, J.H.; Russell, R.J.; Bochman, R.A. Flexible Graphite Material of Expanded Particles Compressed Together. U.S. Patent 3,404,061, 10 January 1968. [Google Scholar]
- Ronald, A.G. Expandable Graphite and Method. U.S. Patent 64,006,612, 18 June 2002. [Google Scholar]
- Mercuri, R.A. Production of Nano-Structures. U.S. Patent 9,034,297, 19 May 2015. [Google Scholar]
- GEnIuS Project—An Innovative Graphene-Based Solution for Tackling Environmental Emergencies through the Adsorption of Oils and Hydrocarbons. Available online: http://www.pdc.minambiente.it/tcpdf/examples/pdf_progetto.php?nid=1652&lang=en (accessed on 27 June 2019).


| Temperature (°C) | Density (g cm−3) | Dynamic Viscosity (mPa·s) | Cinematic Viscosity (mm2 s−1) |
|---|---|---|---|
| 45 | 0.8795 | 9.42 | 10.71 |
| 25 | 0.8929 | 21.14 | 23.67 |
| 20 | 0.8969 | 72.14 | 80.43 |
| Adsorbent | Format | Size (cm) | Initial Weight (g) | Final Weight (g) | Adsorption (g/g) |
|---|---|---|---|---|---|
| Grafysorber® | Boom | 50 × 7.5 | 66 | 1500 | 22.73 |
| 50 × 7.5 | 67 | 1500 | 22.39 | ||
| Pillow | 50 × 50 | 106 | 2200 | 20.75 | |
| 50 × 50 | 102 | 1800 | 17.65 | ||
| Polypropylene | Boom | 50 × 7.5 | 185 | 1600 | 8.65 |
| 50 × 7.5 | 184 | 1500 | 8.15 | ||
| Pillow | 50 × 50 | 453 | 3300 | 7.28 | |
| 50 × 50 | 455 | 3300 | 7.25 |
| Test | Initial Weight (mg) | Weight Loss (mg) | Adsorbent Weight (mg) | Adsorption (mg/mg) |
|---|---|---|---|---|
| Boom_1_1 | 10.243 | −9.806 | 0.437 | 22.43 |
| Boom_1_2 | 25.575 | −24.264 | 1.311 | 18.51 |
| Boom_1_3 | 35.045 | −32.312 | 2.733 | 11.82 |
| Boom_1_4 | 6.842 | −6.426 | 0.415 | 15.45 |
| Boom_1_5 | 31.848 | −30.097 | 1.750 | 17.19 |
| Boom_1_6 | 15.959 | −14.982 | 0.977 | 15.34 |
| Test | Initial Weight (mg) | Weight Loss (mg) | Heat of Fusion (mJ) | TGA Final T (°C) | Δ Weight (mg) |
|---|---|---|---|---|---|
| PP 1 | 4.519 | −4.499 | −280.9 | 550 | 0.020 |
| PP 2 | 12.825 | −6.773 | −1146.0 | 400 | 6.052 |
| PP 3 | 7.151 | −7.144 | −672.7 | 550 | 0.007 |
| PP 4 | 8.418 | −8.388 | −720.9 | 550 | 0.030 |
| PP 5 | 13.911 | −13.895 | −1271.4 | 550 | 0.016 |
| PP 6 | 3.491 | −3.501 | −315.3 | 550 | −0.010 |
| Test | Initial Weight (mg) | Weight Loss (mg) | Heat of Fusion (mJ) | Adsorbent Weight * (mg) | Adsorption (mg/mg) |
|---|---|---|---|---|---|
| Boom_5_1 | 12.841 | −12.713 | −167.22 | 2.548 | 4.99 |
| Boom_5_2 | 20.966 | −20.797 | −236.01 | 3.256 | 6.39 |
| Boom_5_3 | 19.938 | −19.735 | −282.23 | 3.732 | 5.29 |
| Boom_5_4 | 20.751 | −20.499 | −327.26 | 4.195 | 4.89 |
| Boom_5_5 | 19.938 | −19.735 | −282.23 | 3.732 | 5.29 |
| Boom_5_6 | 11.659 | −7.345 | −101.26 | 1.870 | 3.93 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vocciante, M.; Finocchi, A.; De Folly D′Auris, A.; Conte, A.; Tonziello, J.; Pola, A.; Reverberi, A.P. Enhanced Oil Spill Remediation by Adsorption with Interlinked Multilayered Graphene. Materials 2019, 12, 2231. https://doi.org/10.3390/ma12142231
Vocciante M, Finocchi A, De Folly D′Auris A, Conte A, Tonziello J, Pola A, Reverberi AP. Enhanced Oil Spill Remediation by Adsorption with Interlinked Multilayered Graphene. Materials. 2019; 12(14):2231. https://doi.org/10.3390/ma12142231
Chicago/Turabian StyleVocciante, Marco, Antonio Finocchi, Alessandra De Folly D′Auris, Alessandro Conte, Jacopo Tonziello, Annalisa Pola, and Andrea Pietro Reverberi. 2019. "Enhanced Oil Spill Remediation by Adsorption with Interlinked Multilayered Graphene" Materials 12, no. 14: 2231. https://doi.org/10.3390/ma12142231
APA StyleVocciante, M., Finocchi, A., De Folly D′Auris, A., Conte, A., Tonziello, J., Pola, A., & Reverberi, A. P. (2019). Enhanced Oil Spill Remediation by Adsorption with Interlinked Multilayered Graphene. Materials, 12(14), 2231. https://doi.org/10.3390/ma12142231







