Development and Characterization of Composites Consisting of Calcium Phosphate Cements and Mesoporous Bioactive Glass for Extrusion-Based Fabrication
Abstract
1. Introduction
2. Materials and Methods
2.1. Synthesis and Characterization of MBG
2.2. Synthesis and Characterization of MBG-CPC Composites
2.2.1. Preparation of MBG-CPC Composites
2.2.2. Injectability and Extrusion Properties
2.2.3. Setting Time
2.3. Scaffold Fabrication by 3D Plotting
2.3.1. Scaffold Design, Plotting Procedure and Post-Processing
2.3.2. Porosity Measurement
2.3.3. Microscopy
2.3.4. Mechanical Characterization
2.4. Release of Calcium and Strontium Ions
2.5. Statistical Evaluation
3. Results
3.1. Preparation of Mesopouros Bioactive Glass (MBG) Particles
3.2. Storability of MBG-CPC Composite Pastes
3.3. Extrudability of MBG-CPC Composite Pastes as a Function of Their Composition
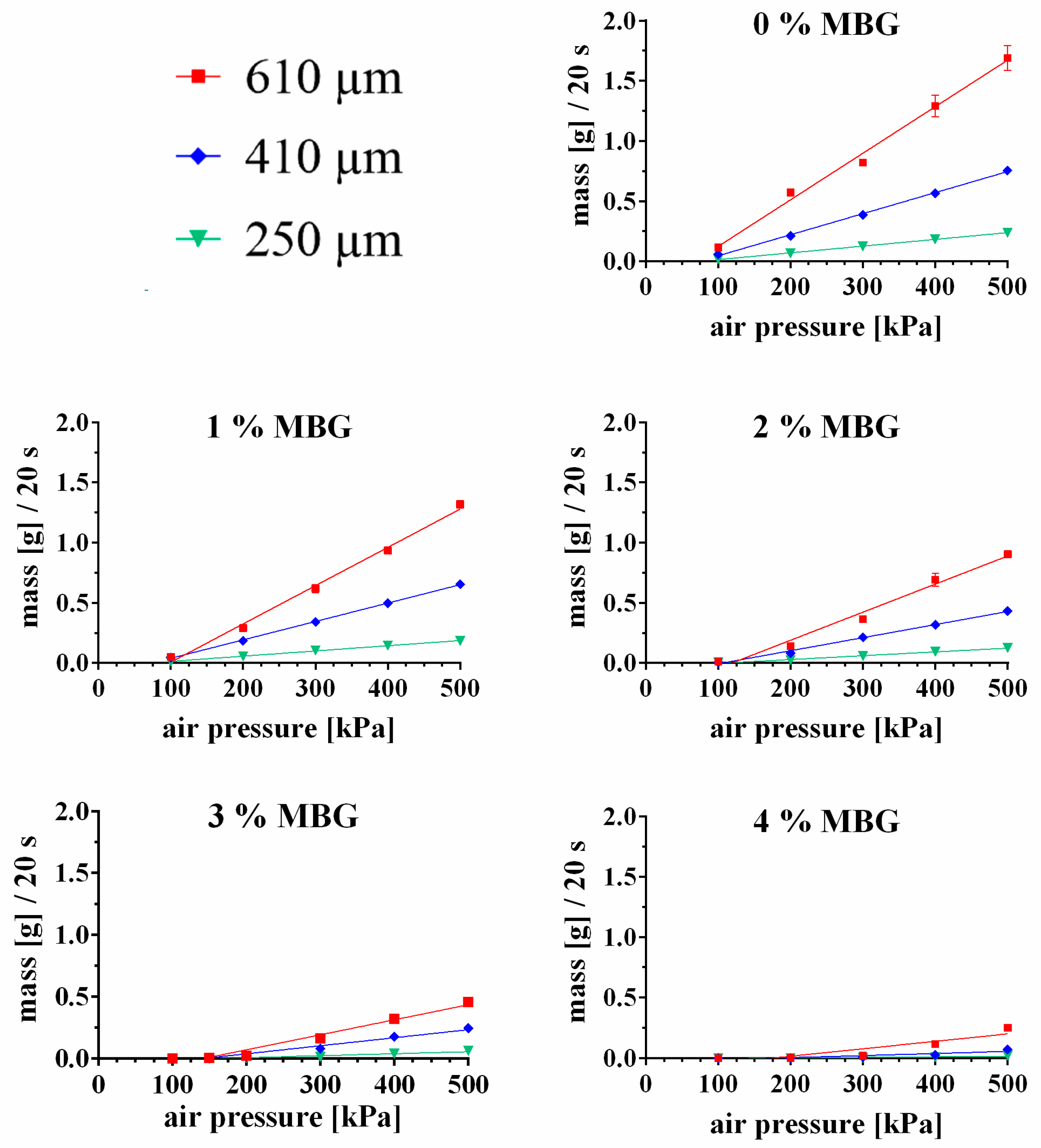
3.3.1. Influence of MBG Particle Concentration on Extrudability of the CPC Paste
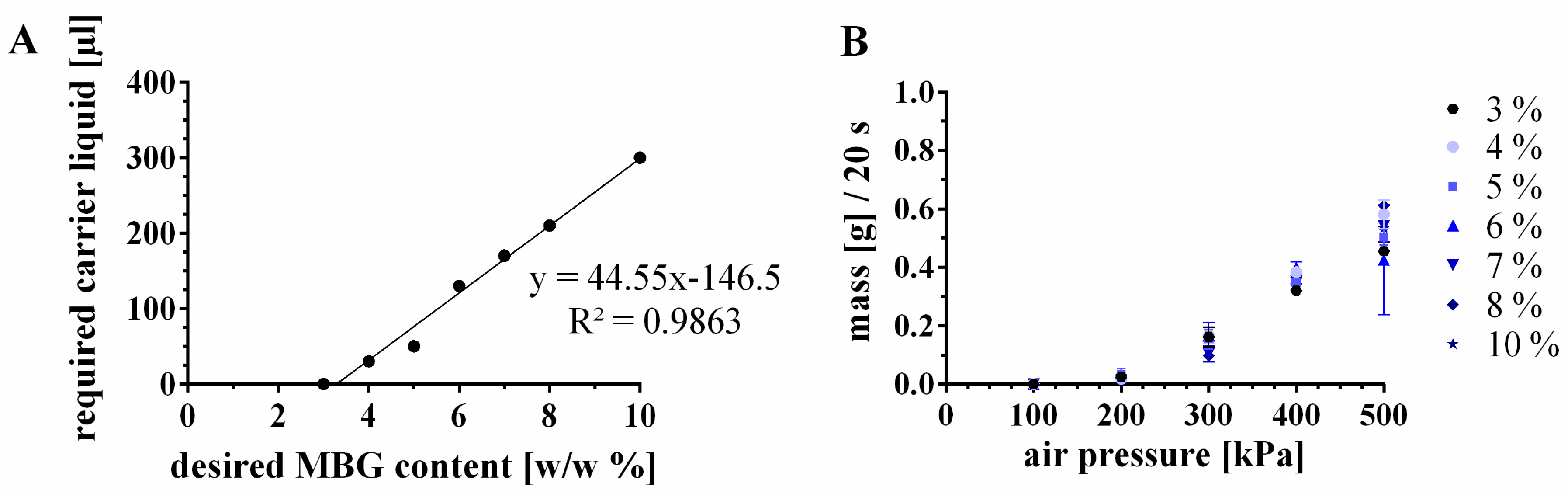
3.3.2. Incorporation of Higher Amounts of MBG by Modification of the CPC Paste
3.4. Three-Dimensional Plotting of MBG-CPC Composites in Mild Conditions
3.5. Characterization of Three-Dimensional Plotted MBG-CPC Scaffolds
3.5.1. Mechanical Properties
3.5.2. Microstructure
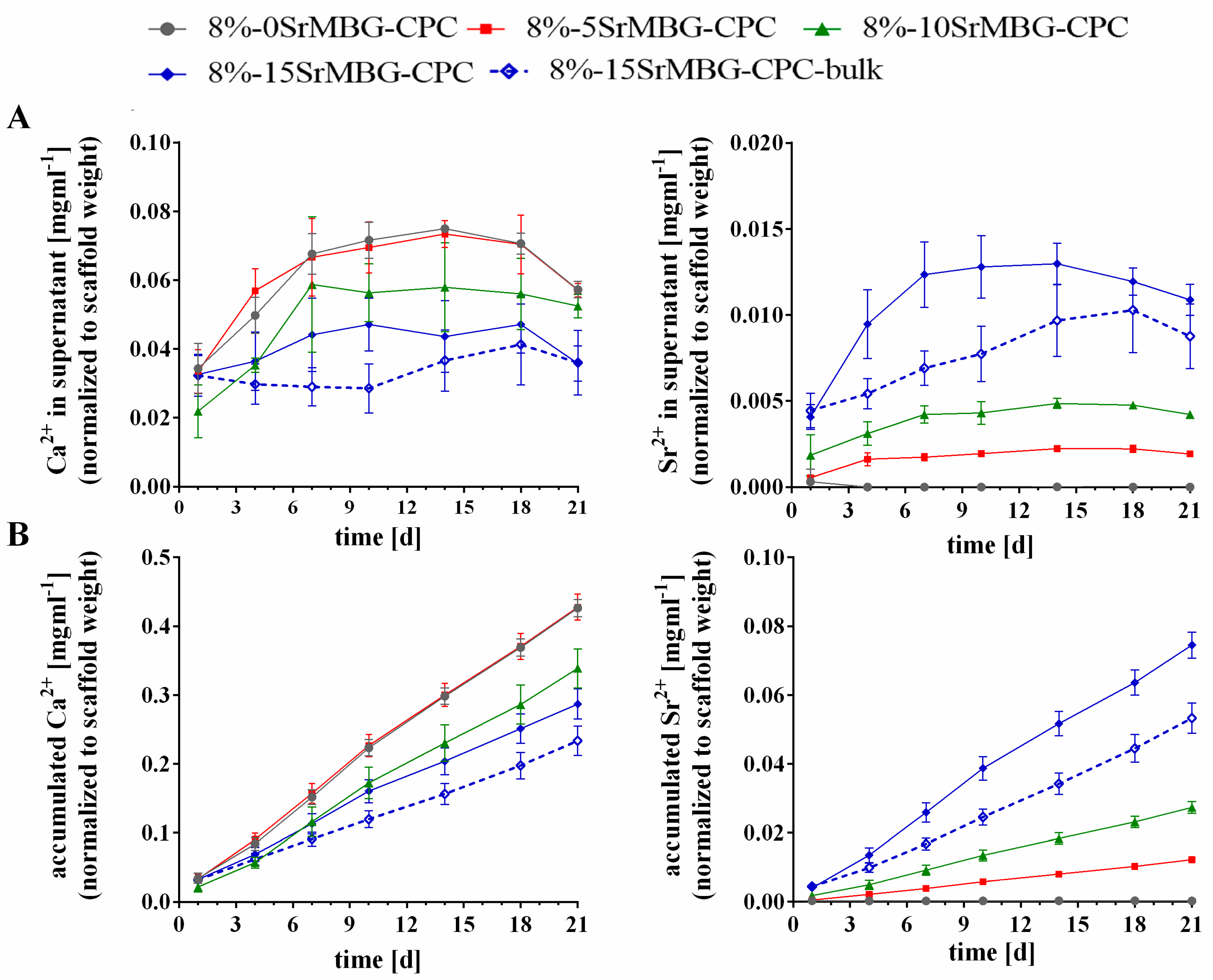
3.6. Ion Release from Sr-MBG-CPC Scaffolds
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Samavedi, S.; Whittington, A.R.; Goldstein, A.S. Calcium phosphate ceramics in bone tissue engineering: A review of properties and their influence on cell behavior. Acta Biomater. 2013, 9, 8037–8045. [Google Scholar] [CrossRef] [PubMed]
- Dorozhkin, S.V. Calcium Orthophosphates as Bioceramics: State of the Art. J. Funct. Biomater. 2010, 1, 22–107. [Google Scholar] [CrossRef] [PubMed]
- Habraken, W.; Habibovic, P.; Epple, M.; Bohner, M. Calcium phosphates in biomedical applications: materials for the future? Mater. Today 2016, 19, 69–87. [Google Scholar] [CrossRef]
- Xu, H.H.; Wang, P.; Wang, L.; Bao, C.; Chen, Q.; Weir, M.D.; Chow, L.C.; Zhao, L.; Zhou, X.; Reynolds, M.A. Calcium phosphate cements for bone engineering and their biological properties. Bone Res. 2017, 5, 17056. [Google Scholar] [CrossRef] [PubMed]
- Bohner, M.; Baroud, G. Injectability of calcium phosphate pastes. Biomaterials 2005, 26, 1553–1563. [Google Scholar] [CrossRef]
- Meininger, S.; Blum, C.; Schamel, M.; Barralet, J.E.; Ignatius, A.; Gbureck, U. Phytic acid as alternative setting retarder enhanced biological performance of dicalcium phosphate cement in vitro. Sci. Rep. 2017, 7, 558. [Google Scholar] [CrossRef]
- Bergmann, C.J.D.; Odekerken, J.C.E.; Welting, T.J.M.; Jungwirth, F.; Devine, D.; Bouré, L.; Zeiter, S.; van Rhijn, L.W.; Telle, R.; Fischer, H.; et al. Calcium phosphate based three-dimensional cold plotted bone scaffolds for critical size bone defects. BioMed Res. Int. 2014, 2014, 852610. [Google Scholar] [CrossRef]
- Li, C.; Gao, L.; Chen, F.; Liu, C. Fabrication of mesoporous calcium silicate/calcium phosphate cement scaffolds with high mechanical strength by freeform fabrication system with micro-droplet jetting. J. Mater. Sci. 2015, 50, 7182–7191. [Google Scholar] [CrossRef]
- Yu, L.; Li, Y.; Zhao, K.; Tang, Y.; Cheng, Z.; Chen, J.; Zang, Y.; Wu, J.; Kong, L.; Liu, S.; et al. A Novel Injectable Calcium Phosphate Cement-Bioactive Glass Composite for Bone Regeneration. PLoS ONE 2013, 8. [Google Scholar] [CrossRef]
- Montufar, E.B.; Traykova, T.; Schacht, E.; Ambrosio, L.; Santin, M.; Planell, J.A.; Ginebra, M.-P. Self-hardening calcium deficient hydroxyapatite/gelatine foams for bone regeneration. J. Mater. Sci. Mater. Med. 2010, 21, 863–869. [Google Scholar] [CrossRef]
- Maazouz, Y.; Montufar, E.B.; Malbert, J.; Espanol, M.; Ginebra, M.-P. Self-hardening and thermoresponsive alpha tricalcium phosphate/pluronic pastes. Acta Biomater. 2017, 49, 563–574. [Google Scholar] [CrossRef] [PubMed]
- Heinemann, S.; Rössler, S.; Lemm, M.; Ruhnow, M.; Nies, B. Properties of injectable ready-to-use calcium phosphate cement based on water-immiscible liquid. Acta Biomater. 2013, 9, 6199–6207. [Google Scholar] [CrossRef] [PubMed]
- Lode, A.; Meissner, K.; Luo, Y.; Sonntag, F.; Glorius, S.; Nies, B.; Vater, C.; Despang, F.; Hanke, T.; Gelinsky, M. Fabrication of porous scaffolds by three-dimensional plotting of a pasty calcium phosphate bone cement under mild conditions. J. Tissue Eng. Regen. Med. 2014, 8, 682–693. [Google Scholar] [CrossRef] [PubMed]
- Maazouz, Y.; Montufar, E.B.; Guillem-Marti, J.; Fleps, I.; Öhman, C.; Persson, C.; Ginebra, M. Robocasting of biomimetic hydroxyapatite scaffolds using self-setting inks. J. Mater. Chem. B 2014, 2, 5378–5386. [Google Scholar] [CrossRef]
- Barba, A.; Diez-Escudero, A.; Maazouz, Y.; Rappe, K.; Espanol, M.; Montufar, E.B.; Bonany, M.; Sadowska, J.M.; Guillem-Marti, J.; Öhman-Mägi, C.; et al. Osteoinduction by Foamed and 3D-Printed Calcium Phosphate Scaffolds: Effect of Nanostructure and Pore Architecture. ACS Appl. Mater. Interfaces 2017, 9, 41722–41736. [Google Scholar] [CrossRef] [PubMed]
- Akkineni, A.R.; Luo, Y.; Schumacher, M.; Nies, B.; Lode, A.; Gelinsky, M. 3D plotting of growth factor loaded calcium phosphate cement scaffolds. Acta Biomater. 2015, 27, 264–274. [Google Scholar] [CrossRef] [PubMed]
- Ahlfeld, T.; Akkineni, A.R.; Förster, Y.; Köhler, T.; Knaack, S.; Gelinsky, M.; Lode, A. Design and Fabrication of Complex Scaffolds for Bone Defect Healing: Combined 3D Plotting of a Calcium Phosphate Cement and a Growth Factor-Loaded Hydrogel. Ann. Biomed. Eng. 2017, 45, 224–236. [Google Scholar] [CrossRef] [PubMed]
- Ahlfeld, T.; Doberenz, F.; Kilian, D.; Vater, C.; Korn, P.; Lauer, G.; Lode, A. Michael Gelinsky Bioprinting of mineralized constructs utilizing multichannel plotting of a self-setting calcium phosphate cement and a cell-laden bioink. Biofabrication 2018, 10, 045002. [Google Scholar] [CrossRef]
- Schumacher, M.; Reither, L.; Thomas, J.; Kampschulte, M.; Gbureck, U.; Lode, A.; Gelinsky, M. Calcium phosphate bone cement/mesoporous bioactive glass composites for controlled growth factor delivery. Biomater. Sci. 2017, 5, 578–588. [Google Scholar] [CrossRef]
- Kauschke, V.; Schneider, M.; Jauch, A.; Schumacher, M.; Kampschulte, M.; Rohnke, M.; Henss, A.; Bamberg, C.; Trinkaus, K.; Gelinsky, M.; et al. Effects of a Pasty Bone Cement Containing Brain-Derived Neurotrophic Factor-Functionalized Mesoporous Bioactive Glass Particles on Metaphyseal Healing in a New Murine Osteoporotic Fracture Model. Int. J. Mol. Sci. 2018, 19, 3531. [Google Scholar] [CrossRef]
- Yan, X.; Yu, C.; Zhou, X.; Tang, J.; Zhao, D. Highly Ordered Mesoporous Bioactive Glasses with Superior In Vitro Bone-Forming Bioactivities. Angew. Chem. Int. Ed. 2004, 43, 5980–5984. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Fan, W.; Gelinsky, M.; Xiao, Y.; Simon, P.; Schulze, R.; Doert, T.; Luo, Y.; Cuniberti, G. Bioactive SrO–SiO2 glass with well-ordered mesopores: Characterization, physiochemistry and biological properties. Acta Biomater. 2011, 7, 1797–1806. [Google Scholar] [CrossRef] [PubMed]
- Lode, A.; Heiss, C.; Knapp, G.; Thomas, J.; Nies, B.; Gelinsky, M.; Schumacher, M. Strontium-modified premixed calcium phosphate cements for the therapy of osteoporotic bone defects. Acta Biomater. 2018, 65, 475–485. [Google Scholar] [CrossRef] [PubMed]
- Ooms, E.M.; Wolke, J.G.C.; van der Waerden, J.P.C.M.; Jansen, J.A. Trabecular bone response to injectable calcium phosphate (Ca-P) cement. J. Biomed. Mater. Res. 2002, 61, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Lewis, G. Injectable bone cements for use in vertebroplasty and kyphoplasty: State-of-the-art review. J. Biomed. Mater. Res. Part B Appl. Biomater. 2006, 76B, 456–468. [Google Scholar] [CrossRef] [PubMed]
- Guo, H.; Wei, J.; Yuan, Y.; Liu, C. Development of calcium silicate/calcium phosphate cement for bone regeneration. Biomed. Mater. 2007, 2, S153–S159. [Google Scholar] [CrossRef] [PubMed]
- Baranowski, A.; Klein, A.; Ritz, U.; Götz, H.; Mattyasovszky, S.G.; Rommens, P.M.; Hofmann, A. Evaluation of Bone Sialoprotein Coating of Three-Dimensional Printed Calcium Phosphate Scaffolds in a Calvarial Defect Model in Mice. Materials 2018, 11, 2336. [Google Scholar] [CrossRef] [PubMed]
- Ahlfeld, T.; Schuster, F.P.; Förster, Y.; Quade, M.; Akkineni, A.R.; Rentsch, C.; Rammelt, S.; Gelinsky, M.; Lode, A. 3D Plotted Biphasic Bone Scaffolds for Growth Factor Delivery: Biological Characterization In Vitro and In Vivo. Adv. Healthc. Mater. 2019, 8, 1801512. [Google Scholar] [CrossRef] [PubMed]
- Eqtesadi, S.; Motealleh, A.; Miranda, P.; Pajares, A.; Lemos, A.; Ferreira, J.M.F. Robocasting of 45S5 bioactive glass scaffolds for bone tissue engineering. J. Eur. Ceram. Soc. 2014, 34, 107–118. [Google Scholar] [CrossRef]
- Luo, Y.; Wu, C.; Lode, A.; Gelinsky, M. Hierarchical mesoporous bioactive glass/alginate composite scaffolds fabricated by three-dimensional plotting for bone tissue engineering. Biofabrication 2012, 5, 015005. [Google Scholar] [CrossRef]
- Zhang, J.; Zhao, S.; Zhu, Y.; Huang, Y.; Zhu, M.; Tao, C.; Zhang, C. Three-dimensional printing of strontium-containing mesoporous bioactive glass scaffolds for bone regeneration. Acta Biomater. 2014, 10, 2269–2281. [Google Scholar] [CrossRef] [PubMed]
- Leite, Á.J.; Sarker, B.; Zehnder, T.; Silva, R.; Mano, J.F.; Boccaccini, A.R. Bioplotting of a bioactive alginate dialdehyde-gelatin composite hydrogel containing bioactive glass nanoparticles. Biofabrication 2016, 8, 035005. [Google Scholar] [CrossRef] [PubMed]
- Ahlfeld, T.; Köhler, T.; Czichy, C.; Lode, A.; Gelinsky, M. A Methylcellulose Hydrogel as Support for 3D Plotting of Complex Shaped Calcium Phosphate Scaffolds. Gels 2018, 4, 68. [Google Scholar] [CrossRef] [PubMed]
- Wagner, A.-S.; Schumacher, M.; Rohnke, M.; Glenske, K.; Gelinsky, M.; Arnhold, S.; Mazurek, S.; Wenisch, S. Incorporation of silicon into strontium modified calcium phosphate bone cements promotes osteoclastogenesis of human peripheral mononuclear blood cells. Biomed. Mater. 2019, 14, 025004. [Google Scholar] [CrossRef] [PubMed]
- Bose, S.; Fielding, G.; Tarafder, S.; Bandyopadhyay, A. Understanding of dopant-induced osteogenesis and angiogenesis in calcium phosphate ceramics. Trends Biotechnol. 2013, 31, 594–605. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Chang, J. Mesoporous bioactive glasses: Structure characteristics, drug/growth factor delivery and bone regeneration application. Interface Focus 2012, 2, 292–306. [Google Scholar] [CrossRef] [PubMed]
- El-Rashidy, A.A.; Roether, J.A.; Harhaus, L.; Kneser, U.; Boccaccini, A.R. Regenerating bone with bioactive glass scaffolds: A review of in vivo studies in bone defect models. Acta Biomater. 2017, 62, 1–28. [Google Scholar] [CrossRef]
- Choudhary, S.; Halbout, P.; Alander, C.; Raisz, L.; Pilbeam, C. Strontium Ranelate Promotes Osteoblastic Differentiation and Mineralization of Murine Bone Marrow Stromal Cells: Involvement of Prostaglandins. J. Bone Miner. Res. 2007, 22, 1002–1010. [Google Scholar] [CrossRef]
- Canalis, E.; Hott, M.; Deloffre, P.; Tsouderos, Y.; Marie, P.J. The divalent strontium salt S12911 enhances bone cell replication and bone formation in vitro. Bone 1996, 18, 517–523. [Google Scholar] [CrossRef]
- Baron, R.; Tsouderos, Y. In vitro effects of S12911-2 on osteoclast function and bone marrow macrophage differentiation. Eur. J. Pharmacol. 2002, 450, 11–17. [Google Scholar] [CrossRef]
- Takahashi, N.; Sasaki, T.; Tsouderos, Y.; Suda, T. S 12911-2 Inhibits Osteoclastic Bone Resorption In Vitro. J. Bone Miner. Res. 2003, 18, 1082–1087. [Google Scholar] [CrossRef] [PubMed]
- Schumacher, M.; Henß, A.; Rohnke, M.; Gelinsky, M. A novel and easy-to-prepare strontium(II) modified calcium phosphate bone cement with enhanced mechanical properties. Acta Biomater. 2013, 9, 7536–7544. [Google Scholar] [CrossRef] [PubMed]
- Schumacher, M.; Wagner, A.S.; Kokesch-Himmelreich, J.; Bernhardt, A.; Rohnke, M.; Wenisch, S.; Gelinsky, M. Strontium substitution in apatitic CaP cements effectively attenuates osteoclastic resorption but does not inhibit osteoclastogenesis. Acta Biomater. 2016, 37, 184–194. [Google Scholar] [CrossRef] [PubMed]
- Izquierdo-Barba, I.; Arcos, D.; Sakamoto, Y.; Terasaki, O.; López-Noriega, A.; Vallet-Regí, M. High-Performance Mesoporous Bioceramics Mimicking Bone Mineralization. Chem. Mater. 2008, 20, 3191–3198. [Google Scholar] [CrossRef]
- Fredholm, Y.C.; Karpukhina, N.; Law, R.V.; Hill, R.G. Strontium containing bioactive glasses: Glass structure and physical properties. J. Non-Cryst. Solids 2010, 356, 2546–2551. [Google Scholar] [CrossRef]
- Fredholm, Y.C.; Karpukhina, N.; Brauer, D.S.; Jones, J.R.; Law, R.V.; Hill, R.G. Influence of strontium for calcium substitution in bioactive glasses on degradation, ion release and apatite formation. J. R. Soc. Interface 2012, 9, 880–889. [Google Scholar] [CrossRef] [PubMed]




| MBG | Molar ratio | Label | CPC | MBG | Additional Carrier Liquid | Composition | |||
|---|---|---|---|---|---|---|---|---|---|
| [wMBG/wCPC %] | [80 Si/X/5 P] | - | [g] | [g] | [µL] | [g] | [wcl/wCPC %] | Solid [w/w %] | Liquid [w/w %] |
| 0 | - | CPC | 10 | 0 | 0 | 0 | 0 | 86 | 14 |
| - | CPC-cl | 10 | 0 | 210 | 0.189 | 1.89 | 84.4047 | 15.5952 | |
| 1 | 15 Ca2+ | 1%-MBG-CPC | 10 | 0.1 | 0 | 0 | 0 | 86.1386 | 13.8613 |
| 2 | 15 Ca2+ | 2%-MBG-CPC | 10 | 0.2 | 0 | 0 | 0 | 86.2745 | 13.7254 |
| 3 | 15 Ca2+ | 3%-MBG-CPC | 10 | 0.3 | 0 | 0 | 0 | 86.4077 | 13.5922 |
| 4 | 15 Ca2+ | 4%-MBG-CPC-I | 10 | 0.4 | 0 | 0 | 0 | 86.5384 | 13.4615 |
| 15 Ca2+ | 4%-MBG-CPC | 10 | 0.4 | 30 | 0.027 | 0.27 | 86.3143 | 13.6856 | |
| 5 | 15 Ca2+ | 5%-MBG-CPC | 10 | 0.5 | 50 | 0.045 | 0.45 | 86.2968 | 13.7031 |
| 6 | 15 Ca2+ | 6%-MBG-CPC | 10 | 0.6 | 130 | 0.117 | 1.17 | 85.8449 | 14.1550 |
| 7 | 15 Ca2+ | 7%-MBG-CPC | 10 | 0.7 | 170 | 0.153 | 1.53 | 85.6905 | 14.3094 |
| 8 | 15 Ca2+ | 8%-MBG-CPC/ 8%-0SrMBG-CPC | 10 | 0.8 | 210 | 0.189 | 1.89 | 85.5400 | 14.4599 |
| 10 Ca2+, 5 Sr2+ | 8%-5SrMBG-CPC | 10 | 0.8 | 210 | 0.189 | 1.89 | 85.5400 | 14.4599 | |
| 5 Ca2+, 10 Sr2+ | 8%-10SrMBG-CPC | 10 | 0.8 | 210 | 0.189 | 1.89 | 85.5400 | 14.4599 | |
| 0 Ca2+, 15 Sr2+ | 8%-15SrMBG-CPC | 10 | 0.8 | 210 | 0.189 | 1.89 | 85.5400 | 14.4599 | |
| 10 | 15 Ca2+ | 10%-MBG-CPC | 10 | 1.0 | 300 | 0.27 | 2.7 | 85.1818 | 14.8181 |
| Label | Pressure [kPa] | Plotting Speed [mms−1] |
|---|---|---|
| CPC | 120 | 5.5 |
| CPC-cl | 50 | 10 |
| 8%-MBG-CPC | 350 | 10 |
| 8%-xSrMBG-CPC (x = 0, 5, 10, 15) | 325 | 10 |
| R2 | 610 µm | 410 µm | 250 µm |
|---|---|---|---|
| 0% MBG | 0.9931 | 0.9987 | 0.9994 |
| 1% MBG | 0.9942 | 0.9995 | 0.9988 |
| 2% MBG | 0.9807 | 0.9946 | 0.9914 |
| 3% MBG | 0.9664 | 0.9631 | 0.9136 |
| 4% MBG | 0.8163 | 0.7998 | 0.795 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Richter, R.F.; Ahlfeld, T.; Gelinsky, M.; Lode, A. Development and Characterization of Composites Consisting of Calcium Phosphate Cements and Mesoporous Bioactive Glass for Extrusion-Based Fabrication. Materials 2019, 12, 2022. https://doi.org/10.3390/ma12122022
Richter RF, Ahlfeld T, Gelinsky M, Lode A. Development and Characterization of Composites Consisting of Calcium Phosphate Cements and Mesoporous Bioactive Glass for Extrusion-Based Fabrication. Materials. 2019; 12(12):2022. https://doi.org/10.3390/ma12122022
Chicago/Turabian StyleRichter, Richard Frank, Tilman Ahlfeld, Michael Gelinsky, and Anja Lode. 2019. "Development and Characterization of Composites Consisting of Calcium Phosphate Cements and Mesoporous Bioactive Glass for Extrusion-Based Fabrication" Materials 12, no. 12: 2022. https://doi.org/10.3390/ma12122022
APA StyleRichter, R. F., Ahlfeld, T., Gelinsky, M., & Lode, A. (2019). Development and Characterization of Composites Consisting of Calcium Phosphate Cements and Mesoporous Bioactive Glass for Extrusion-Based Fabrication. Materials, 12(12), 2022. https://doi.org/10.3390/ma12122022






