CVD Synthesis of Monodisperse Graphene/Cu Microparticles with High Corrosion Resistance in Cu Etchant
Abstract
:1. Introduction
2. Experimental
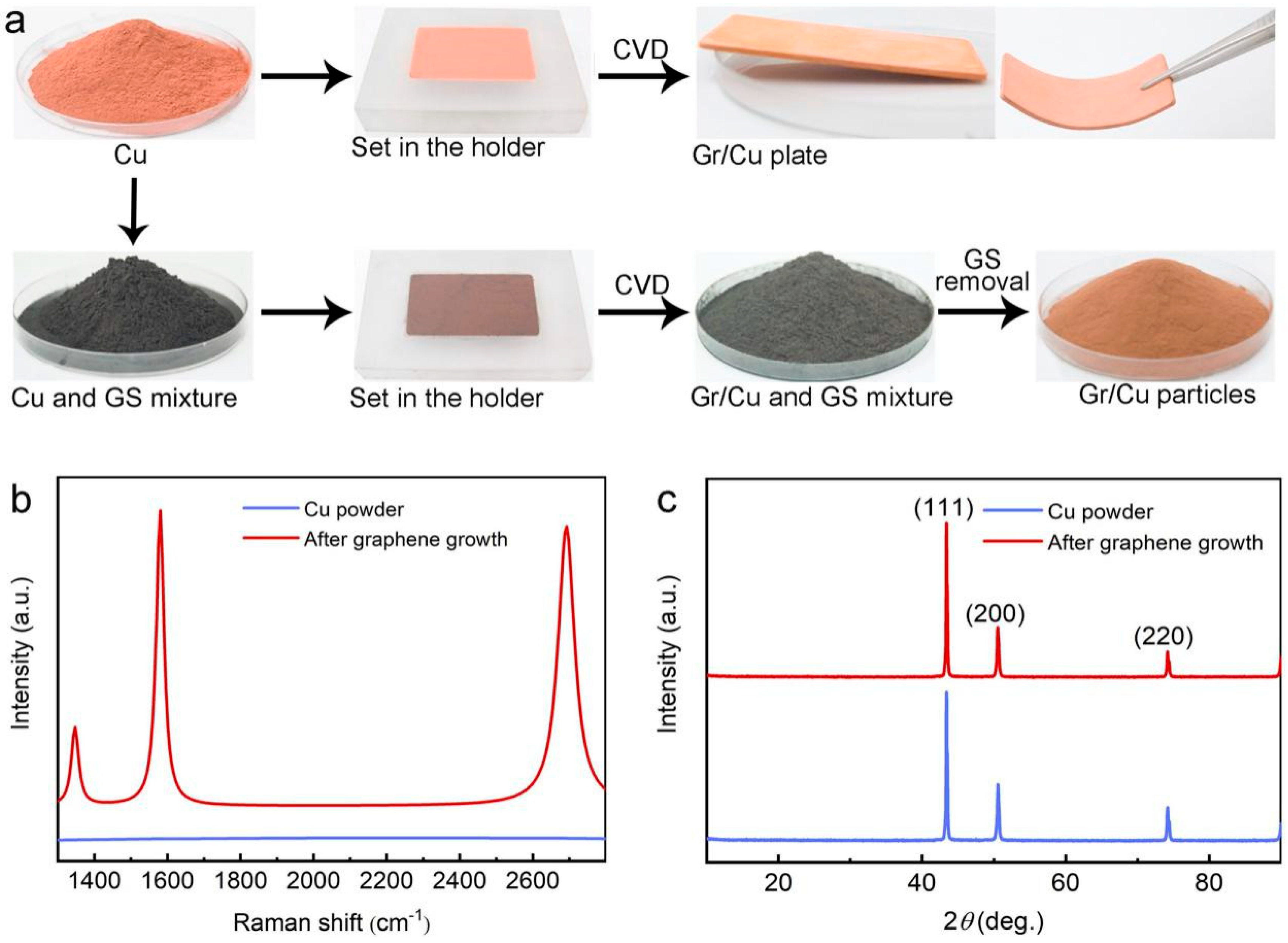
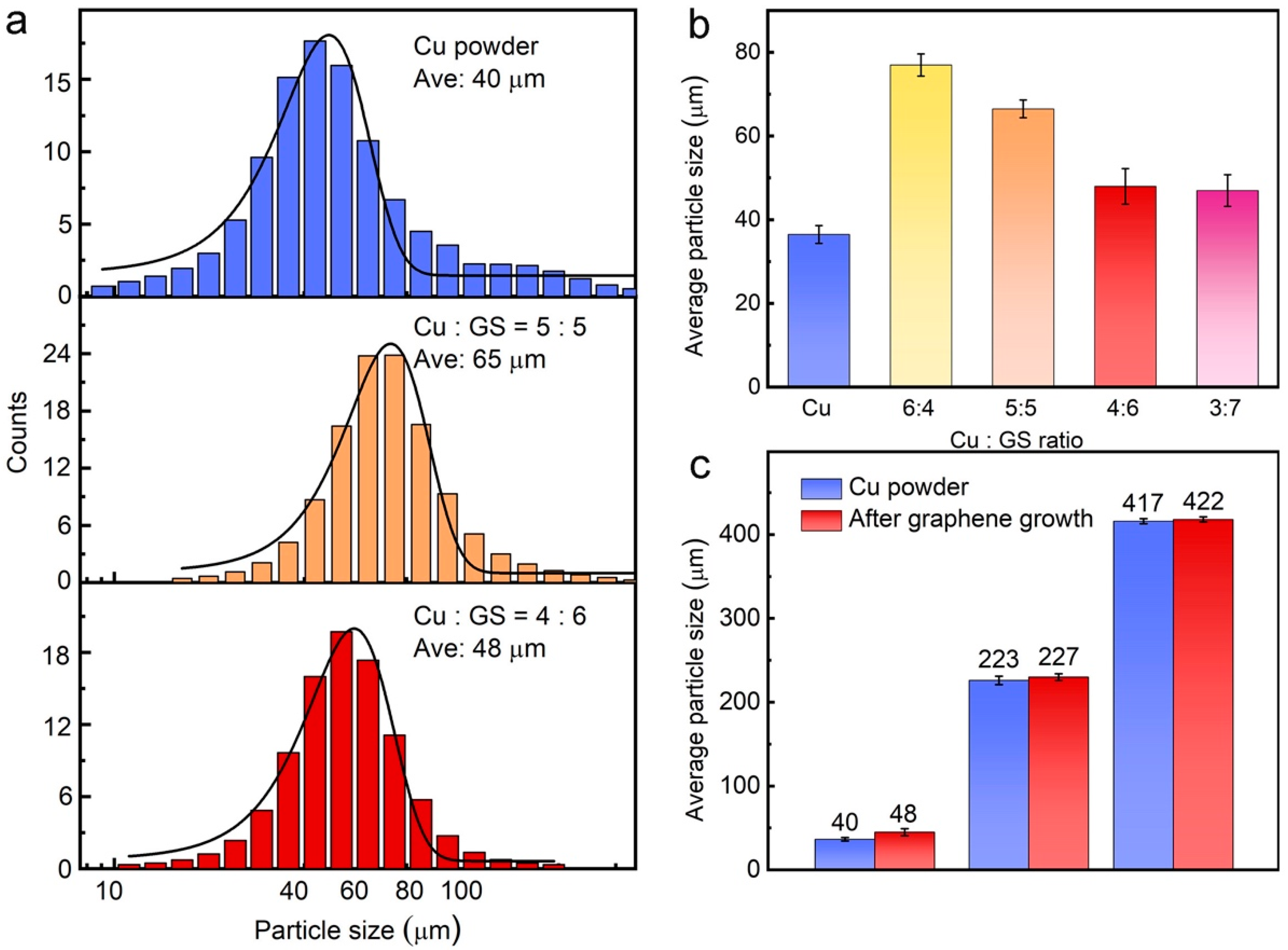
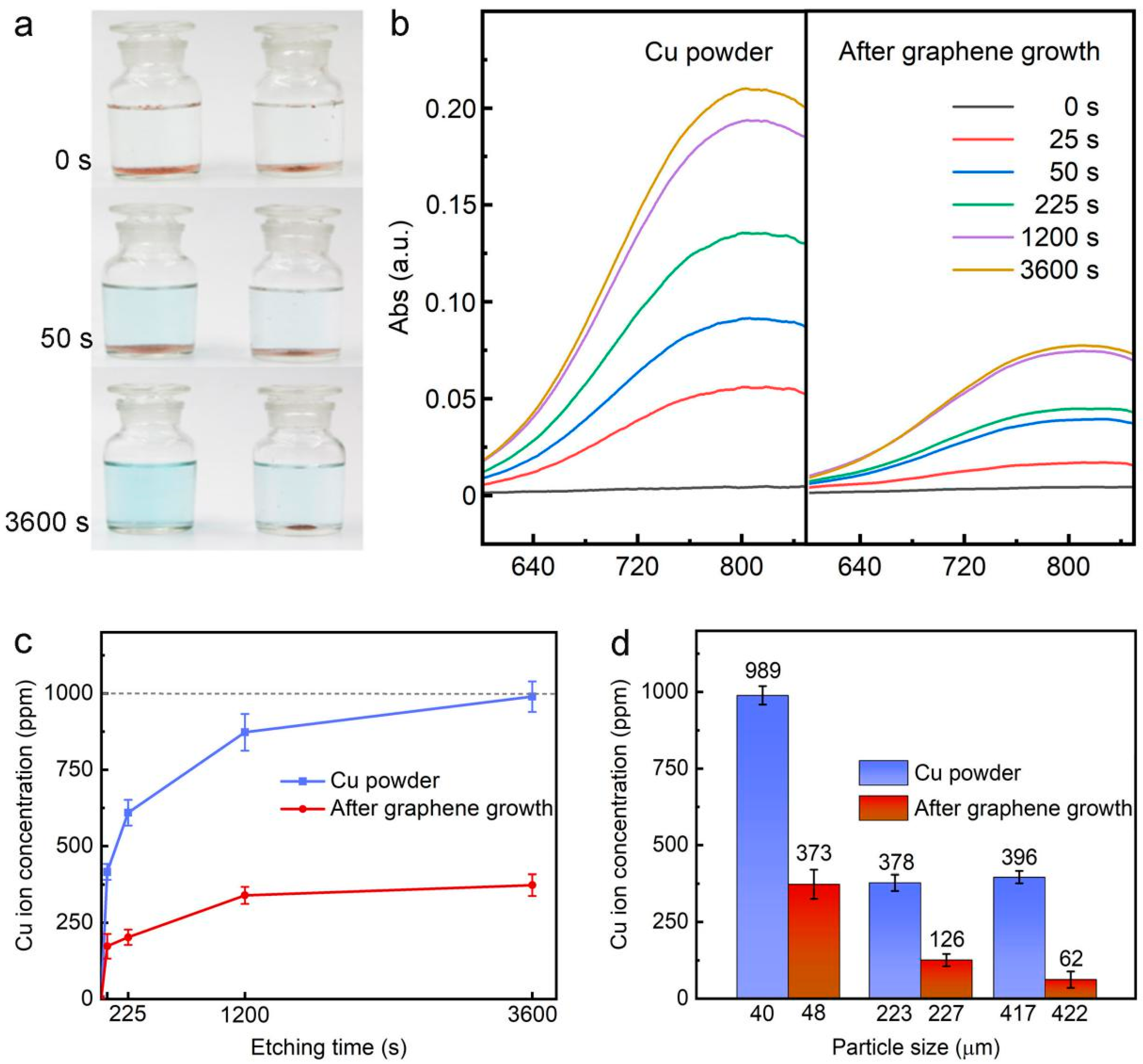
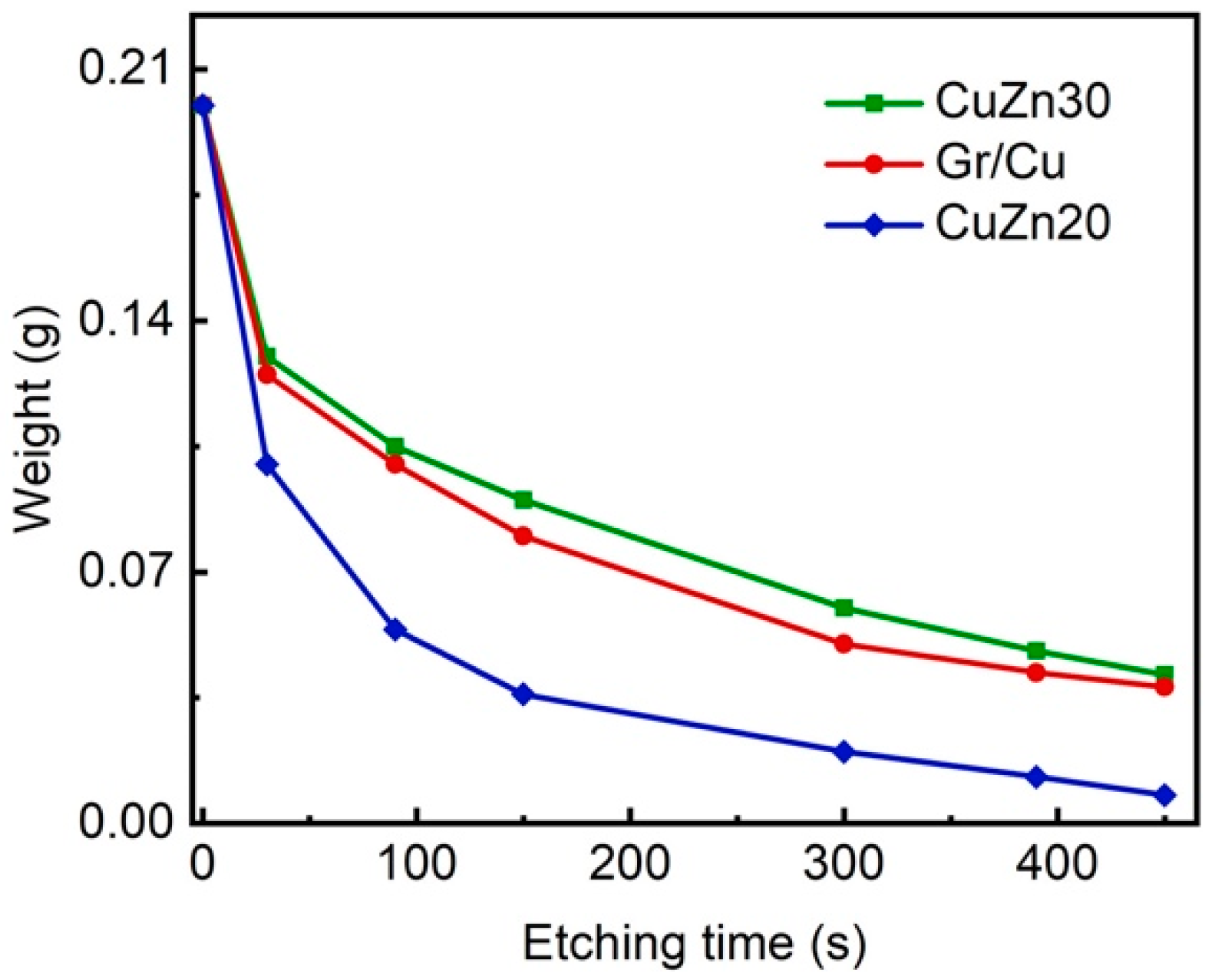
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Bares, J.A.; Argibay, N.; Mauntler, N.; Dudder, G.J.; Perry, S.S.; Bourne, G.R.; Sawyer, W.G. High current density copper-on-copper sliding electrical contacts at low sliding velocities. Wear 2009, 267, 417–424. [Google Scholar] [CrossRef]
- Chiba, H.; Ogushi, T.; Nakajima, H.; Ikeda, T. Heat transfer capacity of lotus-type porous copper heat sink. JSME Int. J. 2005, 47, 516–521. [Google Scholar] [CrossRef]
- Ding, T.; Chen, G.X.; Bu, J.; Zhang, W.H. Effect of temperature and arc discharge on friction and wear behaviours of carbon strip/copper contact wire in pantograph—Catenary systems. Wear 2011, 271, 1629–1636. [Google Scholar] [CrossRef]
- Lee, H.S.; Lee, H.J.; Kwon, H.C. New methodology for enhancing electrical conductivity and strength of copper alloy using combined structure. In Proceedings of the 2010 60th Electronic Components and Technology Conference (ECTC), Las Vegas, NV, USA, 1–4 June 2010; pp. 2031–2034. [Google Scholar]
- Abyzov, A.M.; Kidalov, S.V.; Shakhov, F.M. High thermal conductivity composites consisting of diamond filler with tungsten coating and copper (silver) matrix. J. Mater. Sci. 2011, 46, 1424–1438. [Google Scholar] [CrossRef]
- Bellakhal, N.; Dachraoui, M. Study of the benzotriazole efficiency as a corrosion inhibitor for copper in humid air plasma. Mater. Chem. Phys. 2004, 85, 366–369. [Google Scholar] [CrossRef]
- Singh, I.; Sabita, M.P.; Altekar, V.A. Silver tarnishing and its prevention—A review. Anti-Corros. Method Mater. 1983, 30, 4–8. [Google Scholar] [CrossRef]
- Yuan, S.J.; Pehkonen, S.O. Surface characterization and corrosion behavior of 70/30 Cu-Ni alloy in pristine and sulfide-containing simulated seawater. Corros. Sci. 2007, 49, 1276–1304. [Google Scholar] [CrossRef]
- Vojtěch, D.; Kubásek, J.; Serák, J.; Novák, P. Mechanical and corrosion properties of newly developed biodegradable Zn-based alloys for bone fixation. Acta Biomater. 2011, 7, 3515–3522. [Google Scholar] [CrossRef] [PubMed]
- Kahlert, V.; Streck, C. Metal Cap Layer with Enhanced Etch Resistivity for Copper-Based Metal Regions in Semiconductor Devices. U.S. Patent 8,432,035, 30 April 2013. [Google Scholar]
- Özyılmaz, A.T.; Tüken, T.; Yazıcı, B.; Erbil, M. The electrochemical synthesis and corrosion performance of polyaniline on copper. Prog. Org. Coat. 2005, 52, 92–97. [Google Scholar] [CrossRef]
- Guo, L.; Zhang, Z.; Sun, H.; Dai, D.; Cui, J.; Li, M.; Xu, Y.; Xu, M.; Du, Y.; Jiang, N. Direct formation of wafer-scale single-layer graphene films on the rough surface substrate by PECVD. Carbon 2018, 129, 456–461. [Google Scholar] [CrossRef]
- Novoselov, K.S.; Geim, A.K.; Morozov, S.V.; Jiang, D.; Zhang, Y.; Dubonos, S.V.; Grigorieva, I.V.; Firsov, A.A. Electric field effect in atomically thin carbon films. Science 2004, 306, 666–669. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Li, X.; Du, M.; Guo, Y.; Li, Y.; Li, H.; Yang, Y.; Alam, F.E.; Lin, C.T.; Fang, Y. Solid-phase coalescence of electrochemically exfoliated graphene flakes into a continuous film on copper. Chem. Mater. 2016, 28, 3360–3366. [Google Scholar] [CrossRef]
- Sun, H.; Li, X.; Li, Y.; Chen, G.; Liu, Z.; Alam, F.E.; Dai, D.; Li, L.; Tao, L.; Xu, J.B. High-quality monolithic graphene films via laterally stitched growth and structural repair of isolated flakes for transparent electronics. Chem. Mater. 2017, 29, 7808–7815. [Google Scholar] [CrossRef]
- Berry, V. Impermeability of graphene and its applications. Carbon 2013, 62, 1–10. [Google Scholar] [CrossRef]
- Chen, S.; Brown, L.; Levendorf, M.; Cai, W.; Ju, S.Y.; Edgeworth, J.; Li, X.; Magnuson, C.W.; Velamakanni, A.; Piner, R.D. Oxidation resistance of graphene-coated Cu and Cu/Ni alloy. ACS Nano 2011, 5, 1321–1327. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Yi, D.; Wang, Z.; Yu, J.; Zhang, Z.; Qiao, R.; Sun, Z.; Hu, Z.; Gao, P.; Peng, H. Greatly enhanced anticorrosion of Cu by commensurate graphene coating. Adv. Mater. 2018, 30, 1702944. [Google Scholar] [CrossRef] [PubMed]
- Parra, C.; Montero-Silva, F.; Gentil, D.; Campo, V.D.; Henríquez, R.; Häberle, P.; Garín, C.; Ramírez, C.; Fuentes, R. The many faces of graphene as protection barrier. performance under microbial corrosion and Ni allergy conditions. Materials 2017, 10, 1406. [Google Scholar] [CrossRef] [PubMed]
- Wu, T.; Liu, Z.; Chen, G.; Dai, D.; Sun, H.; Dai, W.; Jiang, N.; Jiang, Y.H.; Lin, C.T. A study of the growth-time effect on graphene layer number based on a Cu-Ni bilayer catalyst system. RSC Adv. 2016, 6, 23956–23960. [Google Scholar] [CrossRef]
- Xiao, Q.; Yi, X.; Jiang, B.; Qin, Z.; Hu, J.; Jiang, Y.; Liu, H.; Wang, B.; Yi, D. In-situ synthesis of graphene on surface of copper powder by rotary CVD and its application in fabrication of reinforced Cu-matrix composites. Adv. Mater. Sci. 2017. [Google Scholar] [CrossRef]
- Kalita, G.; Ayhan, M.E.; Sharma, S.; Shinde, S.M.; Ghimire, D.; Wakita, K.; Umeno, M.; Tanemura, M. Low temperature deposited graphene by surface wave plasma CVD as effective oxidation resistive barrier. Corro. Sci. 2014, 78, 183–187. [Google Scholar] [CrossRef]
- Lee, S.; Hong, J.; Koo, J.H.; Lee, H.; Lee, S.; Choi, T.; Jung, H.; Koo, B.; Park, J.; Kim, H. Synthesis of few-layered graphene nanoballs with copper cores using solid carbon source. ACS Appl. Mater Interfaces 2013, 5, 2432–2437. [Google Scholar] [CrossRef] [PubMed]
- Lee, E.; Lee, H.C.; Jo, S.B.; Lee, H.; Lee, N.S.; Park, C.G.; Lee, S.K.; Kim, H.H.; Bong, H.; Cho, K. Heterogeneous solid carbon source-assisted growth of high-quality graphene via CVD at low temperatures. Adv. Funct. Mater. 2016, 26, 562–568. [Google Scholar] [CrossRef]
- Ferrari, A.C.; Meyer, J.C.; Scardaci, V.; Casiraghi, C.; Lazzeri, M.; Mauri, F.; Piscanec, S.; Jiang, D.; Novoselov, K.S.; Roth, S. Raman spectrum of graphene and graphene layers. Phys. Rev. Lett. 2006, 97, 187401. [Google Scholar] [CrossRef] [PubMed]
- Ramakrishnan, S.; Jelmy, E.J.; Senthilkumar, R.; Rangarajan, M.; Kothurkar, N.K. One-step RF-CVD method for the synthesis of graphene decorated with metal and metal oxide nanoparticles. J. Nanosci. Nanotechnol. 2018, 18, 1089–1096. [Google Scholar] [CrossRef] [PubMed]
- Frank, O.; Mohr, M.; Maultzsch, J.; Thomsen, C.; Riaz, I.; Jalil, R.; Novoselov, K.S.; Tsoukleri, G.; Parthenios, J.; Papagelis, K. Raman 2D-band splitting in graphene: Theory and experiment. ACS Nano 2011, 5, 2231. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Zhang, X.; Liu, E.; He, C.; Han, Y.; Li, Q.; Nash, P.; Zhao, N. Fabrication of three-dimensional graphene/Cu composite by in-situ CVD and its strengthening mechanism. J. Alloys Compd. 2016, 688, 69–76. [Google Scholar] [CrossRef]
- Peissker, T. Production and properties of electrolytic copper powder. Int. J. Powder Metall. 1984, 20, 87. [Google Scholar]
- Kang, D.; Kwon, J.Y.; Cho, H.; Sim, J.H.; Hwang, H.S.; Kim, C.S.; Yong, J.K.; Ruoff, R.S.; Shin, H.S. Oxidation resistance of iron and copper foils coated with reduced graphene oxide multilayers. ACS Nano 2012, 6, 7763. [Google Scholar] [CrossRef] [PubMed]
- Ge, F.; Li, M.M.; Ye, H.; Zhao, B.X. Effective removal of heavy metal ions Cd, Zn, Pb, Cu from aqueous solution by polymer-modified magnetic nanoparticles. J. Hazard. Mater. 2003, 99, 366–372. [Google Scholar]
- Dinan, T.; Datta, M. The kinetics of copper etching in acidic ammonium persulfate solutions. High Rate Met. Dissol. Process. 1995, 10, 189–201. [Google Scholar]
- Zhang, L.; Liu, Y.; Wang, Y.; Xu, M.; Hu, X. UV–Vis spectroscopy combined with chemometric study on the interactions of three dietary flavonoids with copper ions. Food Chem. 2018, 263, 208–215. [Google Scholar] [CrossRef] [PubMed]
- Nizam, M.K. Synthesis of graphene flakes over recovered copper etched in ammonium persulfate solution. Sains Malays. 2017, 46, 1039–1045. [Google Scholar]
- He, B.; Han, P.; Lu, C.; Bai, X. Effect of soil particle size on the corrosion behavior of natural gas pipeline. Eng. Fail. Anal. 2015, 58, 19–30. [Google Scholar] [CrossRef]

© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, S.; Hou, B.; Dai, D.; Shu, S.; Wu, M.; Li, A.; Han, Y.; Zhu, Z.-x.; Chen, B.-a.; Ding, Y.; et al. CVD Synthesis of Monodisperse Graphene/Cu Microparticles with High Corrosion Resistance in Cu Etchant. Materials 2018, 11, 1459. https://doi.org/10.3390/ma11081459
Li S, Hou B, Dai D, Shu S, Wu M, Li A, Han Y, Zhu Z-x, Chen B-a, Ding Y, et al. CVD Synthesis of Monodisperse Graphene/Cu Microparticles with High Corrosion Resistance in Cu Etchant. Materials. 2018; 11(8):1459. https://doi.org/10.3390/ma11081459
Chicago/Turabian StyleLi, Shuangyi, Baosen Hou, Dan Dai, Shengcheng Shu, Mingliang Wu, Ao Li, Yu Han, Zhi-xiang Zhu, Bao-an Chen, Yi Ding, and et al. 2018. "CVD Synthesis of Monodisperse Graphene/Cu Microparticles with High Corrosion Resistance in Cu Etchant" Materials 11, no. 8: 1459. https://doi.org/10.3390/ma11081459
APA StyleLi, S., Hou, B., Dai, D., Shu, S., Wu, M., Li, A., Han, Y., Zhu, Z.-x., Chen, B.-a., Ding, Y., Zhang, Q., Wang, Q., Jiang, N., & Lin, C.-T. (2018). CVD Synthesis of Monodisperse Graphene/Cu Microparticles with High Corrosion Resistance in Cu Etchant. Materials, 11(8), 1459. https://doi.org/10.3390/ma11081459





