Preparation of a Highly Porous Carbon Material Based on Quinoa Husk and Its Application for Removal of Dyes by Adsorption
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemical and Materials
2.2. Preparation of PC–QH
2.3. Batch Equilibrium Studies
2.4. Kinetic and Equilibrium Models
2.5. Reusability of PC–QH
2.6. Characterization
3. Results
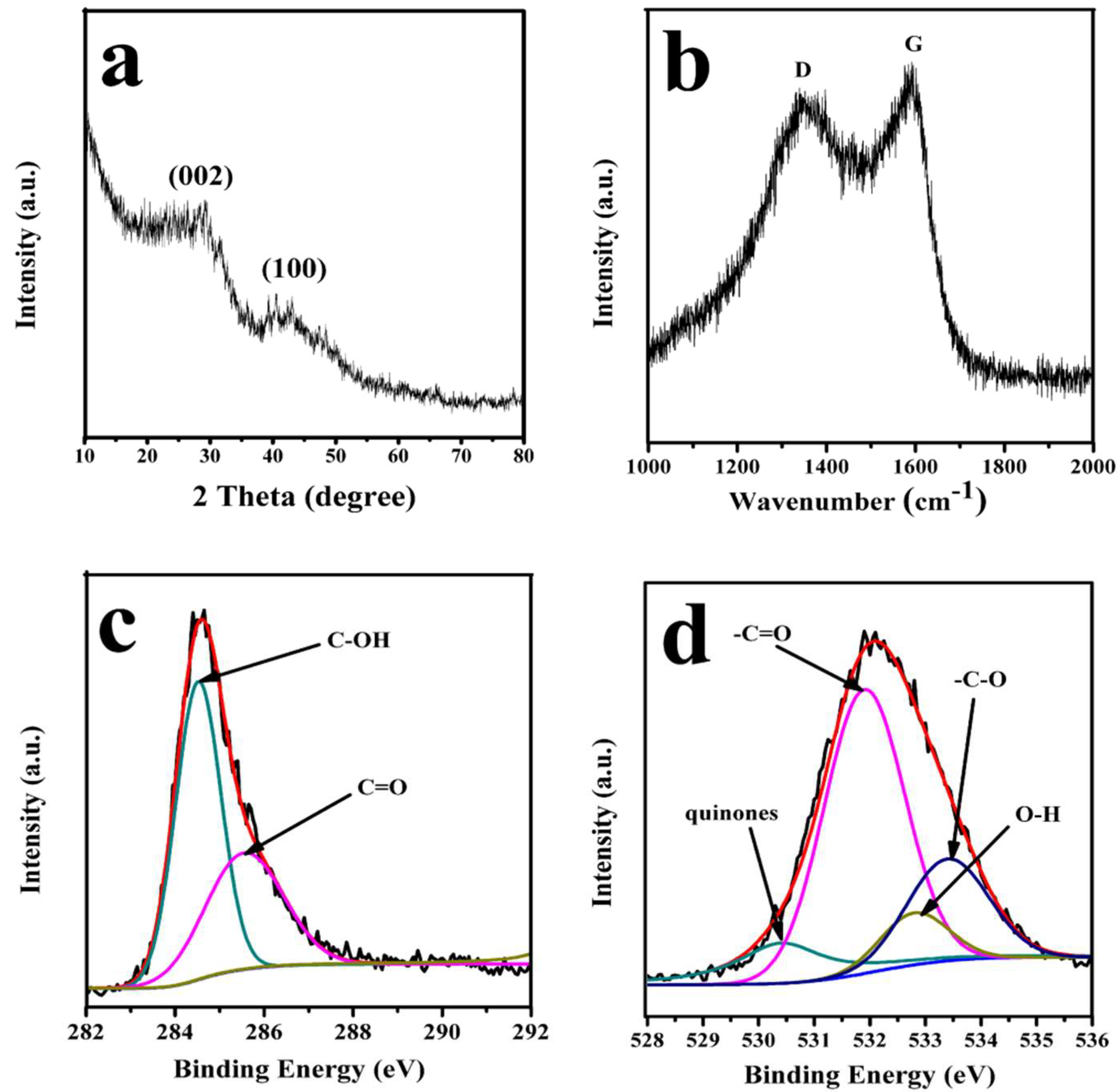
3.1. Characterization of PC–QH
3.2. Preparation of Activated Carbon
3.2.1. Effect of Activation Temperature
3.2.2. Effect of Activation Time
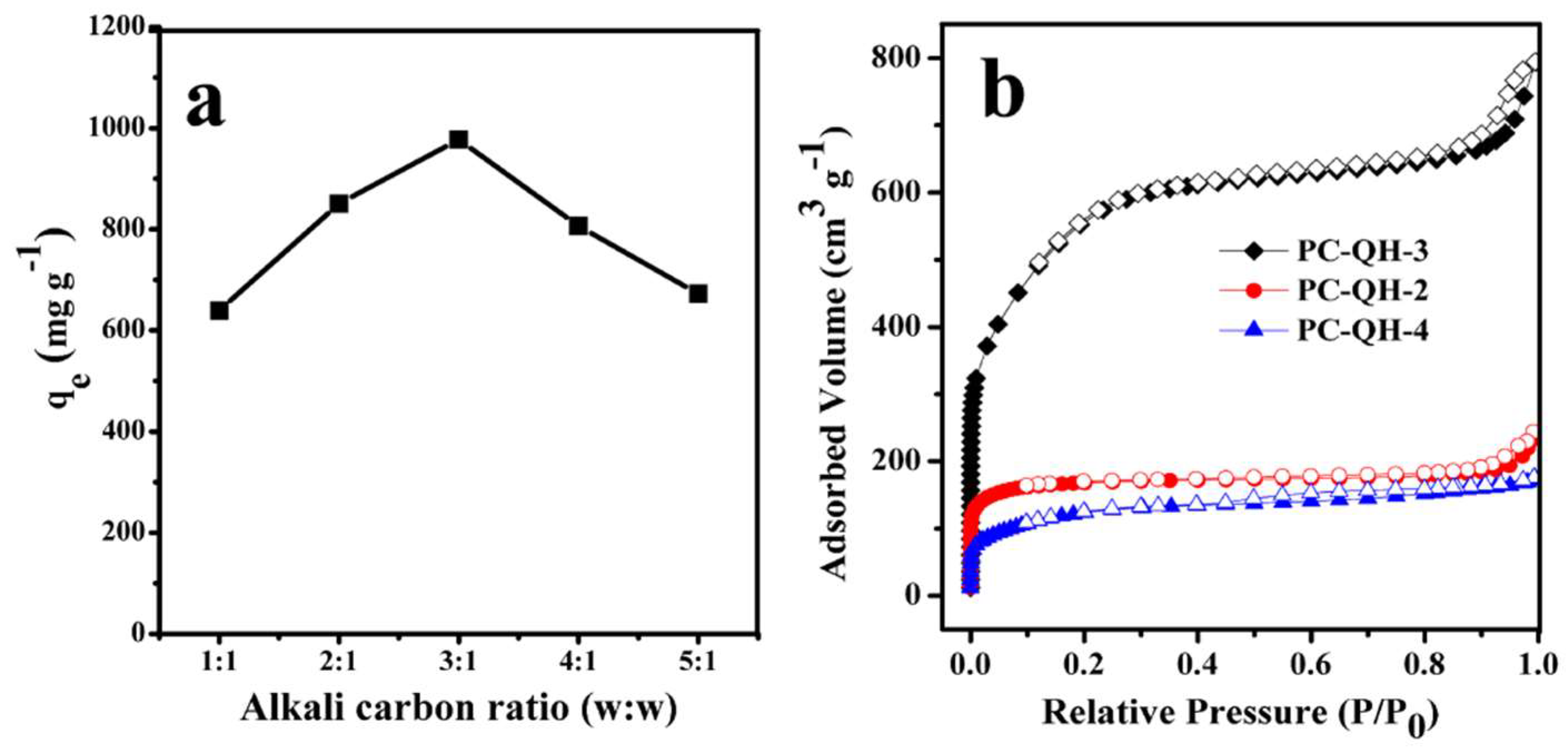
3.2.3. Effect of Alkali/Carbon Ratio
3.3. Adsorption Studies
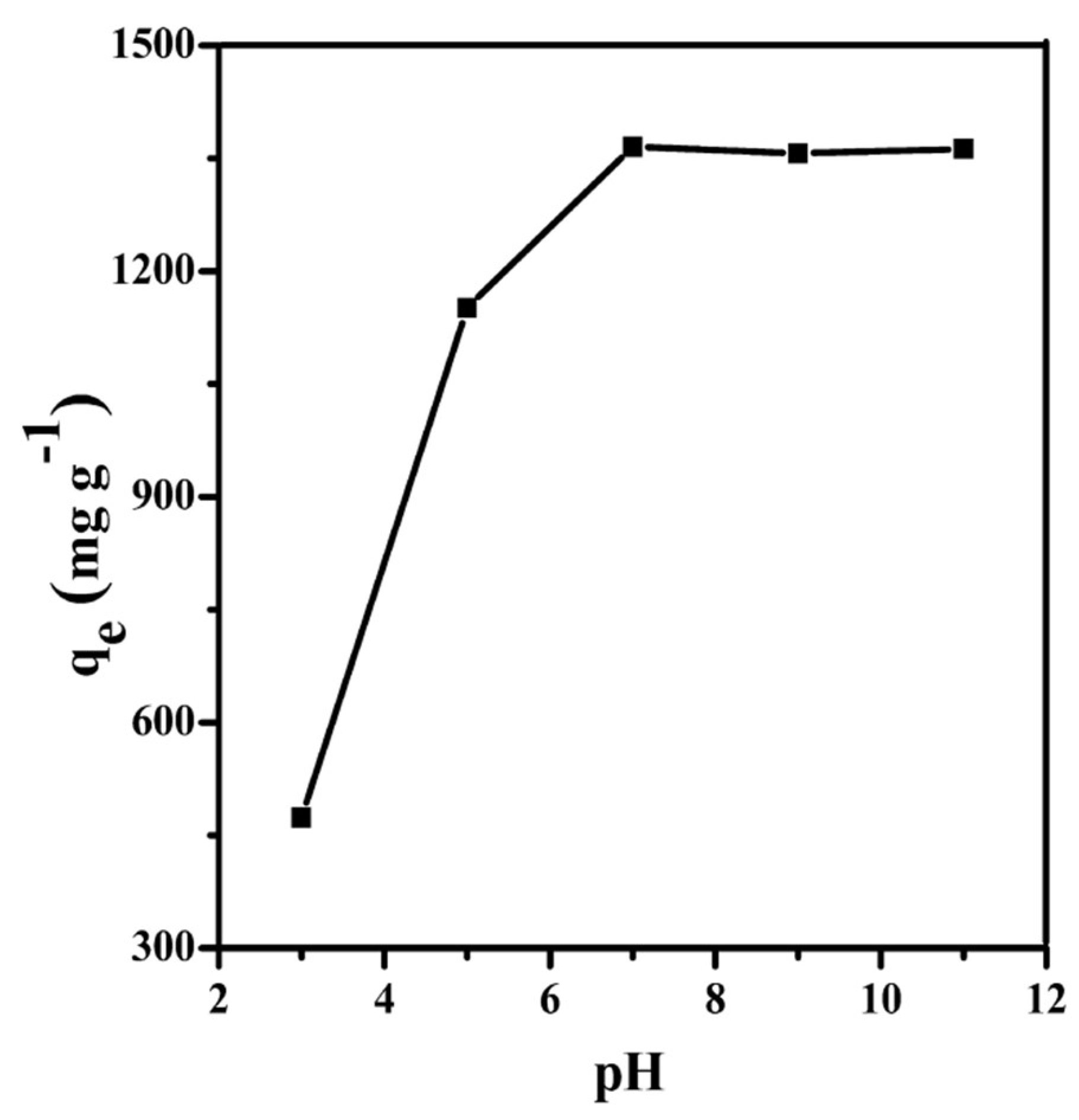
3.3.1. Effect of pH on Adsorption Capacity
3.3.2. Adsorption Kinetics
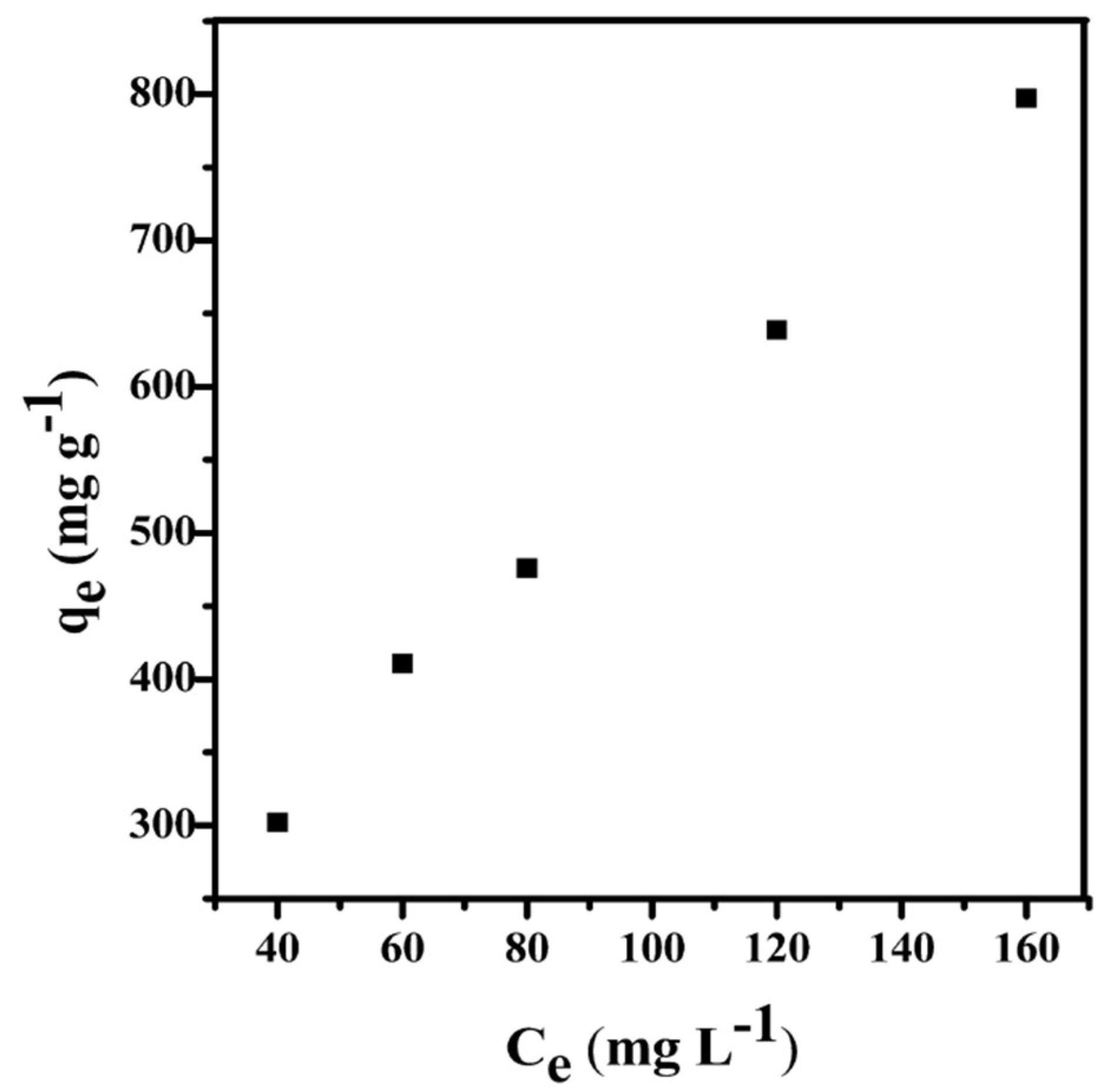
3.3.3. Adsorption Isotherms
3.4. Reusability of PC–QH
3.5. Adsorption Selectivity of PC–QH
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Chuenchom, L.; Kraehnert, R.; Smarsly, B.M. Recent progress in soft-templating of porous carbon materials. Soft Matter 2012, 8, 10801–10812. [Google Scholar] [CrossRef]
- Wang, J.; Senkovska, I.; Kaskel, S.; Liu, Q. Chemically activated fungi-based porous carbons for hydrogen storage. Carbon 2014, 75, 372–380. [Google Scholar] [CrossRef]
- Chen, S.; Wang, J.; Wu, Z.; Deng, Q.; Tu, W.; Dai, G.; Zeng, Z.; Deng, S. Enhanced Cr(VI) removal by polyethylenimine- and phosphoruscodoped hierarchical porous carbons. J. Colloid Interface Sci. 2018, 523, 110–120. [Google Scholar] [CrossRef] [PubMed]
- Dutta, S.; Bhaumik, A.; Wu, K.C.W. Hierarchically porous carbon derived from polymers and biomass: Effect of interconnected pores on energy applications. Energy Environ. Sci. 2014, 7, 3574–3592. [Google Scholar] [CrossRef]
- Widjaya, E.R.; Chen, G.; Bowtell, L.; Hill, C. Gasification of non-woody biomass: A literature review. Renew. Sustain. Energy Rev. 2018, 89, 184–193. [Google Scholar] [CrossRef]
- Wang, C.; Wu, D.; Wang, H.; Gao, Z.; Xu, F.; Jiang, K. Biomass derived nitrogen-doped hierarchical porous carbon sheets for supercapacitors with high performance. J. Colloid Interface Sci. 2018, 523, 133–143. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Qi, J.Q.; Sui, Y.W.; He, Y.Z.; Meng, Q.K.; Wei, F.X.; Jin, Y. An asymmetric supercapacitor based on activated porous carbon derived from walnut shells and NiCo2O4 nanoneedle arrays electrodes. J. Nanosci. Nanotechnol. 2018, 18, 5600–5608. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Ma, J.; Shi, M.; Xie, Y.; Feng, C. Biomass based iron and nitrogen co-doped 3D porous carbon as an efficient oxygen reduction catalyst. J. Colloid Interface Sci. 2018, 523, 144–150. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Zhu, J.; Liang, L.; Li, C.; Liu, C.; Liao, J.; Xing, W. Biomass-derived N-doped carbon and its application inelectrocatalysis. Appl. Catal. B Environ. 2014, 154–155, 177–182. [Google Scholar] [CrossRef]
- Chowdhury, S.; Mishra, R.; Saha, P.; Kushwaha, P. Adsorption thermodynamics; kinetics and isosteric heat of adsorption of malachite green onto chemically modified rice husk. Desalination 2011, 265, 159–168. [Google Scholar] [CrossRef]
- Danish, M.; Ahmad, T. A review on utilization of wood biomass as a sustainable precursor for activated carbon production and application. Renew. Sustain. Energy Rev. 2018, 87, 1–21. [Google Scholar] [CrossRef]
- Yagub, M.T.; Sen, T.K.; Afroze, S.; Ang, H.M. Dye and its removal from aqueous solution by adsorption: A review. Adv. Colloid Interface Sci. 2014, 209, 172–184. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Zhu, F. Quinoa starch: Structure; properties; and applications. Carbohydr. Polym. 2018, 181, 851–861. [Google Scholar] [CrossRef] [PubMed]
- Vilcacundo, R.; Miralles, B.; Carrillo, W.; Ledesma, B. In vitro chemopreventive properties of peptides released from quinoa (Chenopodium quinoa Willd.) protein under simulated gastrointestinal digestion. Food Res. Int. 2018, 105, 403–411. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Rockstraw, D.A. Activated carbons prepared from rice hull by one-step phosphoric acid activation. Microporous Mesoporous Mater. 2007, 100, 12–19. [Google Scholar] [CrossRef]
- Wang, W.; Liang, T.; Bai, H.; Dong, W.; Liu, X. All cellulose composites based on cellulose diacetate and nanofibrillated cellulose prepared by alkali treatment. Carbohydr. Polym. 2018, 179, 297–304. [Google Scholar] [CrossRef] [PubMed]
- Vann, K.L.; Thi, T.T.L. Activated carbon derived from rice husk by NaOH activation and its application in supercapacitor. Prog. Nat. Sci. 2014, 24, 191–198. [Google Scholar] [CrossRef]
- Kumar, T.S.M.; Rajini, N.; Reddy, K.O.; Rajulu, A.V.; Siengchin, S.; Ayrilmis, N. All-cellulose composite films with cellulose matrix and Napier grass cellulose fibril fillers. Int. J. Biol. Macromol. 2018, 112, 1310–1315. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, M.; Kanaguri, Y.; Smith, R.L., Jr. Hydrothermal separation of lignin from bark of Japanese cedar. J. Supercrit. Fluids 2018, 133, 696–703. [Google Scholar] [CrossRef]
- Du, J.; Li, C.; Zhao, Y.; Wang, H. Hemicellulose isolated from waste liquor of viscose fiber mill for preparation of polyacrylamide-hemicellulose hybrid films. Int. J. Biol. Macromol. 2018, 108, 1255–1260. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Zhang, W.; Lin, H.; Li, Y.; Lu, H.; Wang, Y. Hierarchical porous carbon based on the self-templating structure of rice husk for high-performance supercapacitors. RSC Adv. 2015, 5, 19294–19300. [Google Scholar] [CrossRef]
- Hussain, I.; Li, Y.; Qi, J.; Li, J.; Sun, X.; Shen, J.; Han, W.; Wang, L. Synthesis of magnetic yolk-shell mesoporous carbon architecture for the effective adsorption of sulfamethazine drug. Microporous Mesoporous Mater. 2018, 255, 110–118. [Google Scholar] [CrossRef]
- Janes, A.; Kurig, H.; Lust, E. Characterisation of activated nanoporous carbon for supercapacitor electrode materials. Carbon 2007, 45, 1226–1233. [Google Scholar] [CrossRef]
- Okuda, H.; Young, R.J.; Wolverson, D.; Tanaka, F.; Yamamoto, G.; Okabe, T. Investigating nanostructures in carbon fibres using raman spectroscopy. Carbon 2018, 130, 178–184. [Google Scholar] [CrossRef]
- Yang, D.; Velamakanni, A.; Bozoklu, G.; Park, S.; Stoller, M.; Piner, R.D.; Stankovich, S.; Jung, I.; Field, D.A.; Ventrice, C.A., Jr.; et al. Chemical analysis of graphene oxide films after heat and chemical treatments by X-ray photoelectron and Micro-Raman spectroscopy. Carbon 2009, 47, 145–152. [Google Scholar] [CrossRef]
- Darmstadt, H.; Roy, C.; Kaliaguine, S.; Choi, S.J.; Ryoo, R. Surface chemistry of ordered mesoporous carbons. Carbon 2002, 40, 2673–2683. [Google Scholar] [CrossRef]
- Arrigo, R.; Havecker, M.; Wrabetz, S.; Blume, R.; Lerch, M.; McGregor, J.; Parrott, E.P.; Zeitler, J.A.; Gladden, L.F.; Knop-Gericke, A.; et al. Tuning the acid/base properties of nanocarbons by functionalization via amination. J. Am. Chem. Soc. 2010, 132, 9616–9630. [Google Scholar] [CrossRef] [PubMed]
- Yin, C.; Tao, C.; Cai, F.; Song, C.; Gong, H.; Wang, J. Effects of activation temperature on the deoxygenation; specific surface area and supercapacitor performance of grapheme. Carbon 2016, 109, 558–565. [Google Scholar] [CrossRef]
- Xia, H.; Wang, K.; Yang, S.; Shi, Z.; Wang, H.; Wang, J. Formation of graphene flowers during high temperature activation of mesocarbon microbeads with KOH. Microporous Mesoporous Mater. 2016, 234, 384–391. [Google Scholar] [CrossRef]
- Pallares, J.; Gonzalez-Cencerrado, A.; Arauzo, I. Production and characterization of activated carbon from barley straw by physical activation with carbon dioxide and steam. Biomass Bioenergy 2018, 115, 64–73. [Google Scholar] [CrossRef]
- Lin, L.; Zhai, S.; Xiao, Z.; Song, Y.; An, Q.; Song, X. Dye adsorption of mesoporous activated carbons produced from NaOH-pretreated rice husks. Bioresour. Technol. 2013, 136, 437–443. [Google Scholar] [CrossRef] [PubMed]
- Foo, K.Y.; Hameed, B.H. Adsorption characteristics of industrial solid waste derived activated carbon prepared by microwave heating for methylene blue. Fuel Process. Technol. 2012, 99, 103–109. [Google Scholar] [CrossRef]
- Naseeruteen, F.; Hamid, N.S.A.; Suah, F.B.M.; Ngah, W.S.W.; Mehamod, F.S. Adsorption of malachite green from aqueous solution by using novel chitosan ionic liquid beads. Int. J. Biol. Macromol. 2018, 107, 1270–1277. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Zhao, P.; Lu, N.; Chen, H.; Zhang, C.; Hou, X. Facile fabrication of Fe3O4/MIL-101(Cr) for effective removal of acid red 1 and orange G from aqueous solution. Chem. Eng. J. 2016, 295, 403–413. [Google Scholar] [CrossRef]
- Ruan, W.; Hu, J.; Qi, J.; Hou, Y.; Cao, R.; Wei, X. Removal of crystal violet by using reduced-graphene-oxide supported bimetallic Fe/Ni nanoparticles (rGO/Fe/Ni): Application of artificial intelligence modeling for the optimization process. Materials 2018, 11, 865. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.D.; Tsai, F.C.; Ma, N.; Xia, Y.; Liu, H.L.; Zhan, X.Q.; Yu, X.Y.; Zeng, X.Z.; Jiang, T.; Shi, D.; et al. Adsorption behavior of high stable Zr-based MOFs for the removal of acid organic dye from water. Materials 2017, 10, 205. [Google Scholar] [CrossRef] [PubMed]
- Fan, Y.; Liu, H.; Zhang, Y.; Chen, Y. Adsorption of anionic MO or cationic MB from MO/MB mixture using polyacrylonitrile fiber hydrothermally treated with hyperbranched polyethylenimine. J. Hazard. Mater. 2015, 283, 321–328. [Google Scholar] [CrossRef] [PubMed]

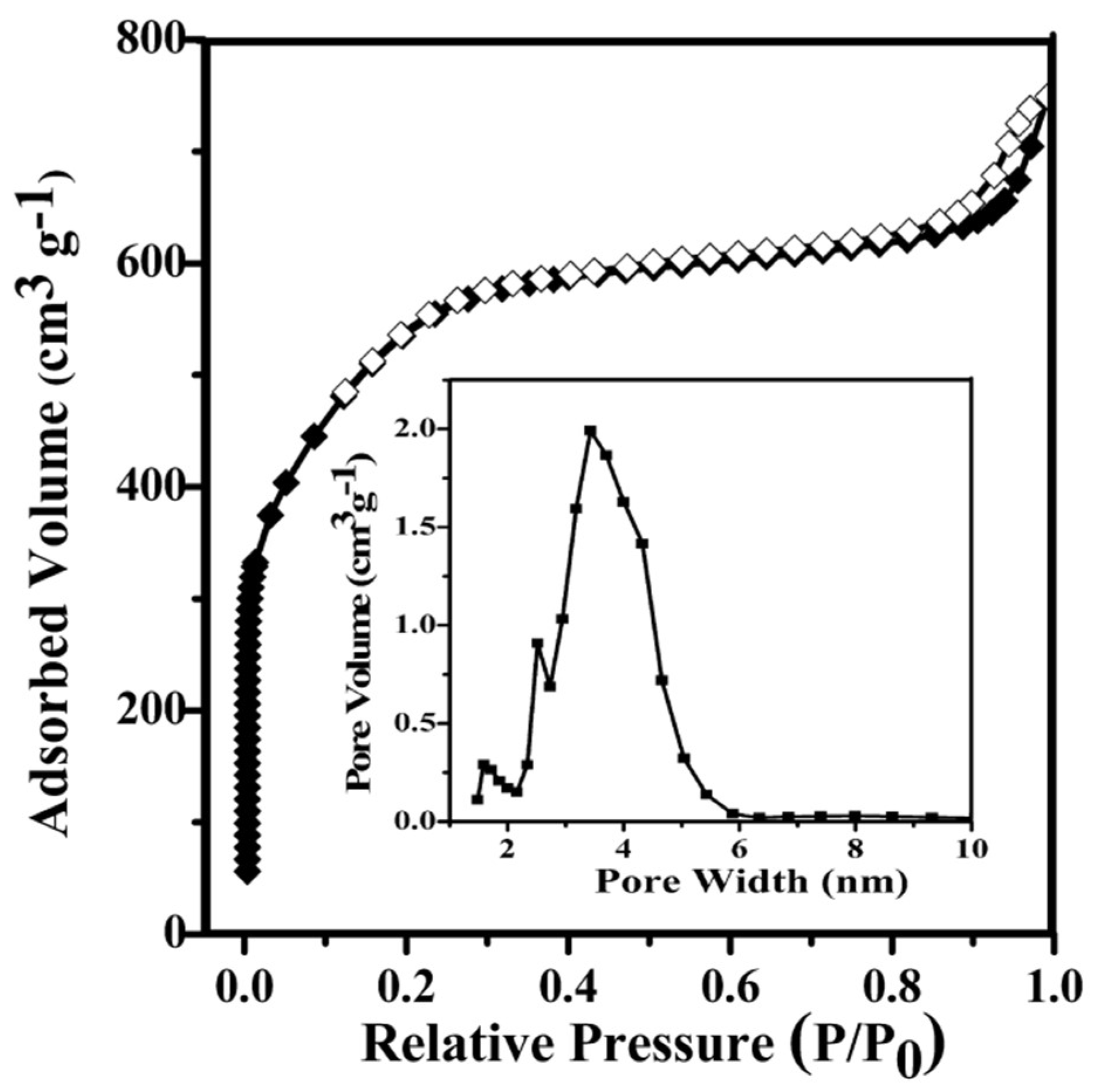



| Samples | Activiation Conditions | SBET 2 (m2 g−1) | Vmicro 3 (cm3 g−1) | Vtotal 4 (cm3 g−1) | Dpore 5 (nm) | ||
|---|---|---|---|---|---|---|---|
| T (°C) | t (min) | A/Cr 1 | |||||
| C–QH | --- | --- | --- | 0.42 | 0.0001 | 0.0026 | 24.78 |
| PC–QH | 650 | 90 | 3:1 | 1713 | 0.53 | 1.23 | 2.87 |
| PC–QH | 600 | 90 | 3:1 | 760 | 0.34 | 0.53 | 2.79 |
| PC–QH | 700 | 90 | 3:1 | 120 | 0.01 | 0.45 | 15.01 |
| PC–QH | 650 | 60 | 3:1 | 228 | 0.05 | 0.42 | 2.44 |
| PC–QH | 650 | 120 | 3:1 | 174 | 0.01 | 0.58 | 13.38 |
| PC–QH | 650 | 90 | 2:1 | 634 | 0.25 | 0.34 | 2.14 |
| PC–QH | 650 | 90 | 4:1 | 437 | 0.14 | 0.27 | 2.43 |
| C0 (mg L−1) | qe (mg g−1) | Pseudo-First-Order Kinetic | Pseudo-Second-Order Kinetic | Intra-Particle Kinetic | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| K1 (min−1) | Qe.cat (mg g−1) | R2 | K2 (g mg−1 min−1) | Qe.cat (mg g−1) | R2 | K3 (mg g−1 min−0.5) | Qe.cat (mg g−1) | R2 | ||
| 40 | 302.12 | 0.0398 | 129.51 | 0.9785 | 0.0008 | 303.03 | 0.9982 | 17.397 | 161.96 | 0.9725 |
| 60 | 410.66 | 0.0633 | 162.85 | 0.9864 | 0.0007 | 416.67 | 0.9998 | 22.733 | 245.10 | 0.8884 |
| 80 | 475.84 | 0.0405 | 167.19 | 0.9821 | 0.0008 | 500.01 | 0.9998 | 24.529 | 294.47 | 0.9156 |
| 120 | 638.73 | 0.0240 | 201.93 | 0.9512 | 0.0006 | 666.67 | 0.9987 | 26.375 | 438.08 | 0.9220 |
| 160 | 797.20 | 0.0580 | 302.41 | 0.7225 | 0.0005 | 833.33 | 0.9971 | 28.260 | 568.27 | 0.8759 |
| Langmuir | Freundlich | ||||
|---|---|---|---|---|---|
| Qm (mg g−1) | KL (L mg−1) | R2 | KF (mg g−1 (L mg−1)1/n) | n | R2 |
| 1666.67 | 0.0053 | 0.9549 | 23.9332 | 1.4533 | 0.9972 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, S.; Tang, S.; Sun, Y.; Wang, G.; Chen, H.; Yu, X.; Su, Y.; Chen, G. Preparation of a Highly Porous Carbon Material Based on Quinoa Husk and Its Application for Removal of Dyes by Adsorption. Materials 2018, 11, 1407. https://doi.org/10.3390/ma11081407
Chen S, Tang S, Sun Y, Wang G, Chen H, Yu X, Su Y, Chen G. Preparation of a Highly Porous Carbon Material Based on Quinoa Husk and Its Application for Removal of Dyes by Adsorption. Materials. 2018; 11(8):1407. https://doi.org/10.3390/ma11081407
Chicago/Turabian StyleChen, Siji, Shanshan Tang, Yang Sun, Gang Wang, Huan Chen, Xiaoxiao Yu, Yingjie Su, and Guang Chen. 2018. "Preparation of a Highly Porous Carbon Material Based on Quinoa Husk and Its Application for Removal of Dyes by Adsorption" Materials 11, no. 8: 1407. https://doi.org/10.3390/ma11081407
APA StyleChen, S., Tang, S., Sun, Y., Wang, G., Chen, H., Yu, X., Su, Y., & Chen, G. (2018). Preparation of a Highly Porous Carbon Material Based on Quinoa Husk and Its Application for Removal of Dyes by Adsorption. Materials, 11(8), 1407. https://doi.org/10.3390/ma11081407





