Durability Modeling Review of Thermal- and Environmental-Barrier-Coated Fiber-Reinforced Ceramic Matrix Composites Part I
Abstract
1. Introduction
2. TBC and EBC Physical Characteristics
3. TBC and EBC Processing Methods
3.1. Plasma-Sprayed Coatings
3.2. The Electron-Beam Physical Vapor Deposition (EB-PVD) Process
3.3. Sputter-Deposited Thermal Barrier Coating
3.4. Processing of Plasma-Spray Physical Vapor Deposition (PS-PVD) TBC Coating
3.5. Slurry Deposition Technique
3.6. Chemical Deposition Process (CVD)
4. Testing of EBCs and TBCs
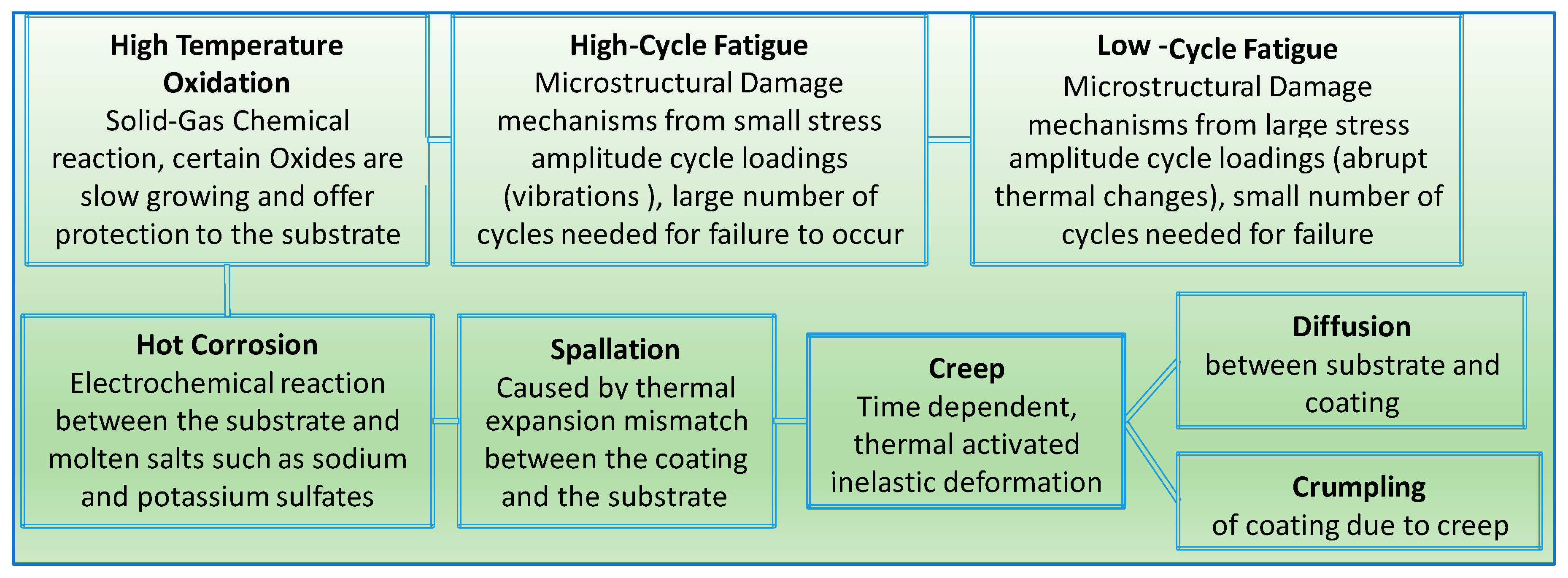
5. Failure Modes of TBCs and EBCs
- (1)
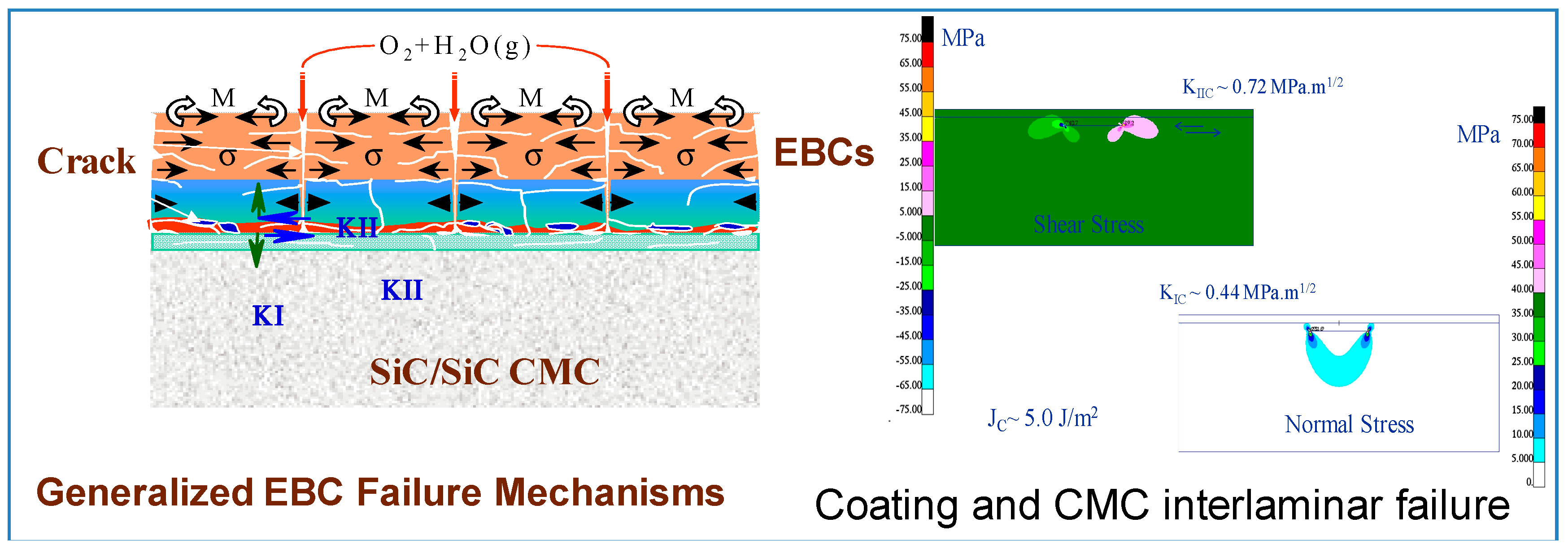
- Cracks spanning from the top coat through the thickness to the bond/intermediate coat interface and to the bond/substrate line with a formation and growth of horizontal crack alongside the interfaces. These cracks are linked and eventual spallation of the coating is imminent. Also, reactions at the interface and the formation of internal pores further accelerate the coating spallation. Figure 6 shows the inter-laminar failure of the coating and the stress contour due to combined thermal and mechanical load obtained from a finite element based analysis.
- (2)
- Moisture through the coating to the substrate/bond coat interface and oxygen diffusion followed by formation of pores and oxidation of the substrate, linkage of the pore and eventual spallation of the coating. This observation is noted when a mismatch of thermal expansion between the substrate and the coating co-exist. An example of this mechanism is shown in Figure 7, which shows pore formation at the substrate/coating interface leading to coating delamination.
6. Conclusions
- (1)
- The need to develop modeling methodologies to determine and assess the life of the EBC and its durability is very essential to its success. However, factors that affect or influence the EBC stability, mechanisms of degradation and failure modes are to be determined and included in the modeling process.
- (2)
- In order for the CMC to be successful in the use of making engine components, the coatings that are applied have to offer a reliant means of protection. This translates into the fact that the life of the coated CMC is highly dependent on the life of the coating and not the uncoated substrate life. It further means that if the coating fails under certain condition, then then the CMC life is considerably decreased.
- (3)
- Predicting the durability and the life of the EBC alone and the CMC after the coating failure is very crucial to introducing CMC engine components coated with EBC.
- (4)
- Finally, a detailed representation of TBC and EBC physical characteristics along with their processing methodologies, testing procedures/approach and failure modes has been summarized. The focus on their failure modes and in particular the EBC is an essential element of this review since it is quite complex and modeling such aspects of failure modes requires extensive analytical/experimental testing efforts to include the causing factors. However, this is outside the scope of this article.
Funding
Conflicts of Interest
Nomenclature
| EB-PVD | Electron-beam physical vapor deposition |
| TBC | Thermal barrier coating |
| EBC | Environmental barrier coating |
| CMC | Ceramic matrix composite |
| CTE | Thermal expansion |
| E | Young’s modulus |
| v | Poisson’s ratio |
| ST | Tensile strength |
| SC | Compressive strength |
| SS | Shear stress |
| PS-PVD | Plasma-spray-assisted physical vapor deposition |
| CVD | Chemical deposition process |
| PECVD | Chemical vapor deposition |
| LACVD | Laser-assisted chemical vapor deposition |
| BSAS | Barium strontium aluminosilicate |
| αc | Coefficient of thermal expansion of the coating |
| αsubstrate | Coefficients of thermal expansion of the substrate, respectively |
| Ec | Young’s modulus of the coating |
| νc | Possion’s ratio of the coating |
References
- Evans, A.G.; Mumm, D.R.; Hutchinson, J.W.; Meier, G.H.; Pettit, F.S. Mechanisms controlling the durability of thermal barrier coatings. Prog. Mater. Sci. 2001, 46, 505–553. [Google Scholar] [CrossRef]
- Clarke, D.R.; Oechsner, M.; Padture, N.P. Thermal-barrier coatings for more efficient gas-turbine engines. MRS Bull. 2012, 37, 891–898. [Google Scholar] [CrossRef]
- Bose, S. High Temperature Coatings; Elsevier: Amsterdam, The Netherlands, 2007. [Google Scholar]
- Jacobson, N.S.; Fox, D.S.; Smialek, J.L.; Opila, E.J.; Dellacorte, C.; Lee, K.N. ASM Handbook; Cramer, S.D., Covino, B.S., Jr., Eds.; ASM International: Materials Park, OH, USA, 2005; pp. 565–578. [Google Scholar]
- Padture, N.P.; Gell, M.; Jordan, E.H. Thermal barrier coatings for gas-turbine engine applications. Science 2002, 296, 280–284. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.N.; Fox, D.S.; Eldridge, J.I.; Zhu, D.; Robinson, R.C.; Bansal, N.P.; Miller, R.A. Upper temperature limit of environmental barrier coatings based on mullite and BSAS. J. Am. Ceram. Soc. 2003, 86, 1299–1306. [Google Scholar] [CrossRef]
- Lee, K.N. Current status of environmental barrier coatings for Si-based ceramics. Surf. Coat. Technol. 2000, 133–134, 1–7. [Google Scholar] [CrossRef]
- Richard, C.S.; Beranger, G.; Lu, J.; Flavenot, J.F. The influences of heat treatments and interdiffusion on the adhesion of plasma-sprayed NiCrAlY coatings. Surf. Coat. Technol. 1996, 82, 99–109. [Google Scholar] [CrossRef]
- National Materials Advisory Board Committee on Coatings for High Temperature Structural Materials. Coatings for High-Temperature Structural Materials: Trends and Opportunities; National Academy Press: Washington, DC, USA, 1996. [Google Scholar]
- Abdul-Aziz, A.; Bhatt, R.T.; Grady, J.E.; Zhu, D. Environmental barrier coating (EBC) durability modeling: An overview and preliminary analysis. Process. Prop. Adv. Ceram. Compos. IV 2012, 234, 313–323. [Google Scholar]
- Zhu, D.; Miller, R.A. Thermal conductivity and elastic modulus evolution of thermal barrier coatings under high heat flux conditions. J. Therm. Spray Technol. 2000, 9, 175–180. [Google Scholar] [CrossRef]
- Ramasamy, S.; Tewari, S.N.; Lee, K.N.; Bhatt, R.T.; Fox, D.S. Mullite–gadolinium silicate environmental barrier coatings for melt infiltrated SiC/SiC composites. Surf. Coat. Technol. 2011, 205, 3578–3581. [Google Scholar] [CrossRef]
- Jorgensen, P.J.; Wadsworth, M.E.; Cutler, I.B. Oxidation of silicon carbide. J. Am. Ceram. Soc. 1959, 42, 613–616. [Google Scholar] [CrossRef]
- Smialek, J.L.; Robinson, R.C.; Opila, E.J.; Fox, D.S.; Jacobson, N.S. SiC and Si3N4 recession due to SiO2 scale volatility under combustor conditions. Adv. Compos. Mater. 1999, 8, 33–45. [Google Scholar] [CrossRef]
- More, K.L.; Tortorelli, P.F.; Walker, L.R. Effects of High Water Vapor Pressures on Oxidation of SiC-Based Fiber-Reinforced Composites. Mater. Sci. Forum 2001, 369, 385–394. [Google Scholar] [CrossRef]
- More, K.L.; Tortorelli, P.F.; Walker, L.R.; Miriyala, N.; Price, J.R.; van Roode, M. High-temperature stability of SiC-based composites in high-water-vapor-pressure environments. J. Am. Ceram. Soc. 2003, 86, 1272–1281. [Google Scholar] [CrossRef]
- Miller, R.A. Thermal Barrier Coating Workshop; NASA Lewis Research Center: Cleveland, OH, USA, 1995; pp. 17–34. [Google Scholar]
- Auger, M.L.; Sarin, V.K. The development of CVD mullite coatings for high temperature corrosive applications. Surf. Coat. Technol. 1997, 94–95, 46–52. [Google Scholar] [CrossRef]
- Roode, M.V.; Ferber, M.K.; Richerson, D.W. Ceramic Gas Turbine Design and Test Experience: Progress in Ceramic Gas Turbine Development; ASME Press: New York, NY, USA, 2003. [Google Scholar]
- Schulz, U.; Leyens, C.; Fritscher, K.; Peters, M.; Saruhan-Brings, B.; Lavigne, O.; Caliez, M. Some recent trends in research and technology of advanced thermal barrier coatings. Aerosp. Sci. Technol. 2003, 7, 73–80. [Google Scholar] [CrossRef]
- Bhatt, R.T.; Choi, S.R.; Cosgriff, L.M.; Fox, D.S.; Lee, K.N. Impact resistance of environmental barrier coated SiC/SiC composites. Mater. Sci. Eng. A 2008, 476, 8–19. [Google Scholar] [CrossRef]
- Haynes, J.A.; Lance, M.J.; Cooley, K.M.; Ferber, M.K.; Lowden, R.A.; Stinton, D.P. CVD Mullite Coatings in High-Temperature, High-Pressure Air–H2O. J. Am. Ceram. Soc. 2000, 83, 657–659. [Google Scholar] [CrossRef]
- Rigney, D.V.; Viguie, R.; Wortman, D.J.; Skelly, D.W. PVD thermal barrier coating applications and process development for aircraft engines. J. Therm. Spray Technol. 1997, 6, 167–175. [Google Scholar] [CrossRef]
- Movchan, B.A.; Demchishin, A.V. The Physics of metals and metallography; Pergamon Press for Pergamon Institute: New York, NY, USA, 1969. [Google Scholar]
- Thornton, J.A. Influence of apparatus geometry and deposition conditions on the structure and topography of thick sputtered coatings. J. Vac. Sci. Technol. 1974, 11, 666. [Google Scholar] [CrossRef]
- Thornton, J.A. High rate thick film growth. Ann. Rev. Mater. Sci. 1977, 7, 239–260. [Google Scholar] [CrossRef]
- National Materials Advisory Board Commission and Engineering and Technical Systems; Materials and Process, Page 22, Table 3–4; National Research Council, National Academy Press: Washington, DC, USA, 1996.
- Kellermann, D.C.; Furukawa, T. A theory of strongly orthotropic continuum mechanics. In Proceedings of the 5th Australasian Congress on Applied Mechanics (ACAM 2007), Brisbane, Australia, 10–12 December 2007; pp. 588–593. [Google Scholar]
- Nguyen, P.; Harding, S.; Ho, S.Y. Experimental studies on slurry based thermal barrier coatings. Eng. Aust. 2007, 1, 545–550. [Google Scholar]
- Fèvre, M.; Finel, A.; Caudron, R.; Mévrel, R. Local order and thermal conductivity in yttria-stabilized zirconia. II. Numerical and experimental investigations of thermal conductivity. Phys. Rev. B 2005, 72, 104118. [Google Scholar] [CrossRef]
- Jiang, X.; Liu, C.; Lin, F. Overview on the development of nanostructured thermal barrier coatings. J. Mater. Sci. Technol. 2007, 23, 449–456. [Google Scholar]
- Robinson, R.C.; Smialek, J.L. SiC recession caused by SiO2 scale volatility under combustion conditions: Experimental results, empirical model and I. J. Am. Ceram. Soc. 1999, 82, 1817–1825. [Google Scholar] [CrossRef]
- American Society of Testing and Materials (ASTM) C28 Standards; ASTM: West Conshohocken, PA, USA, 2010; Volume 15.01.
- Soechting, F.O. Proceedings of the Thermal Barrier Coating Workshop, Cleveland, OH, USA, 27–29 March 1995; NASA Conference Publication 3312; NASA Lewis Research Center: Cleveland, OH, USA, 1995; pp. 1–15.
- Lee, K.N. Key Durability Issues with Mullite-Based Environmental Barrier Coatings for Si-Based Ceramics. Trans. ASME 2000, 122, 632–636. [Google Scholar] [CrossRef]
- Lee, K.N.; Fox, D.S.; Bansal, N.P. Rare Earth Environmental Barrier Coatings for SiC/SiIC Composites and Si3N4 Ceramics. J. Eur. Ceram. Soc. 2005, 25, 1705–1715. [Google Scholar] [CrossRef]
- Bose, S.; DeMasi-Marcin, J. Thermal barrier coating experience in gas turbine engines at Pratt & Whitney. J. Therm. Spray Technol. 1997, 6, 99–104. [Google Scholar]
- Brindley, W.J. Properties of plasma-sprayed bond coats. J. Therm. Spray Technol. 1997, 6, 85–90. [Google Scholar] [CrossRef]
- Wortman, D.J.; Duderstadt, E.C.; Nelson, W.A. ASME Paper 89-GT-134; ASME: New York, NY, USA, 1989. [Google Scholar]
- Petrus, G.J.; Ferguson, B.L. A software tool to design thermal barrier coatings: A technical note. J. Therm. Spray Technol. 1997, 6, 29–34. [Google Scholar] [CrossRef]
- Chang, G.C.; Phucharoen, W.A.; Miller, R.A. Current status of thermal barrier coatings—An overview. Surf. Coat. Technol. 1987, 30, 1–11. [Google Scholar]
- Lee, K.N.; Fox, D.S.; Robinson, R.C.; Bansal, N.P. Environmental Barrier Coatings for Silicon-Based Ceramics. High Temperature Ceramic Matrix Composites. In High Temperature Ceramic Matrix Composites; Krenkel, W., Naslain, R., Schneider, H., Eds.; Wiley-Vch: Weinheim, Germany, 2001; pp. 224–229. [Google Scholar]
- Zhu, D.M.; Bansal, N.P.; Miller, R.A. Thermal Conductivity and Stability of HfO2-Y2O3 and La2Zr2O7 Evaluated for 1650 °C. In Advances in Ceramic Matrix Composites IX; Bansal, N.P., Singh, J.P., Kriven, W.M., Schneider, H., Eds.; The American Ceramic Society: Westerville, OH, USA, 2003; Volume 153, pp. 331–343. [Google Scholar]
- Zhu, D.M.; Miller, R.A.; Fox, D.S. Thermal and Environmental Barrier Coating Development for Advanced Propulsion Engine Systems. In Proceedings of the 8th AIAA/ASME/ASCE/AHS/ASC Structures, Structural Dynamics, and Materials Conference, San Diego, CA, USA, 4–8 January 2008. [Google Scholar]
- Abdul-Aziz, A.; Wroblewski, A.C.; Bhatt, R.T.; Jaskowiak, M.H.; Gorican, D.; Rauser, R.W. Assessment of NDE methods for detecting cracks and damage in environmental barrier coated CMC tested under tension. In Proceedings of the SPIE 9436, Smart Sensor Phenomena, Technology, Networks, and Systems Integration 2015, San Diego, CA, USA, 9–10 March 2015. [Google Scholar]

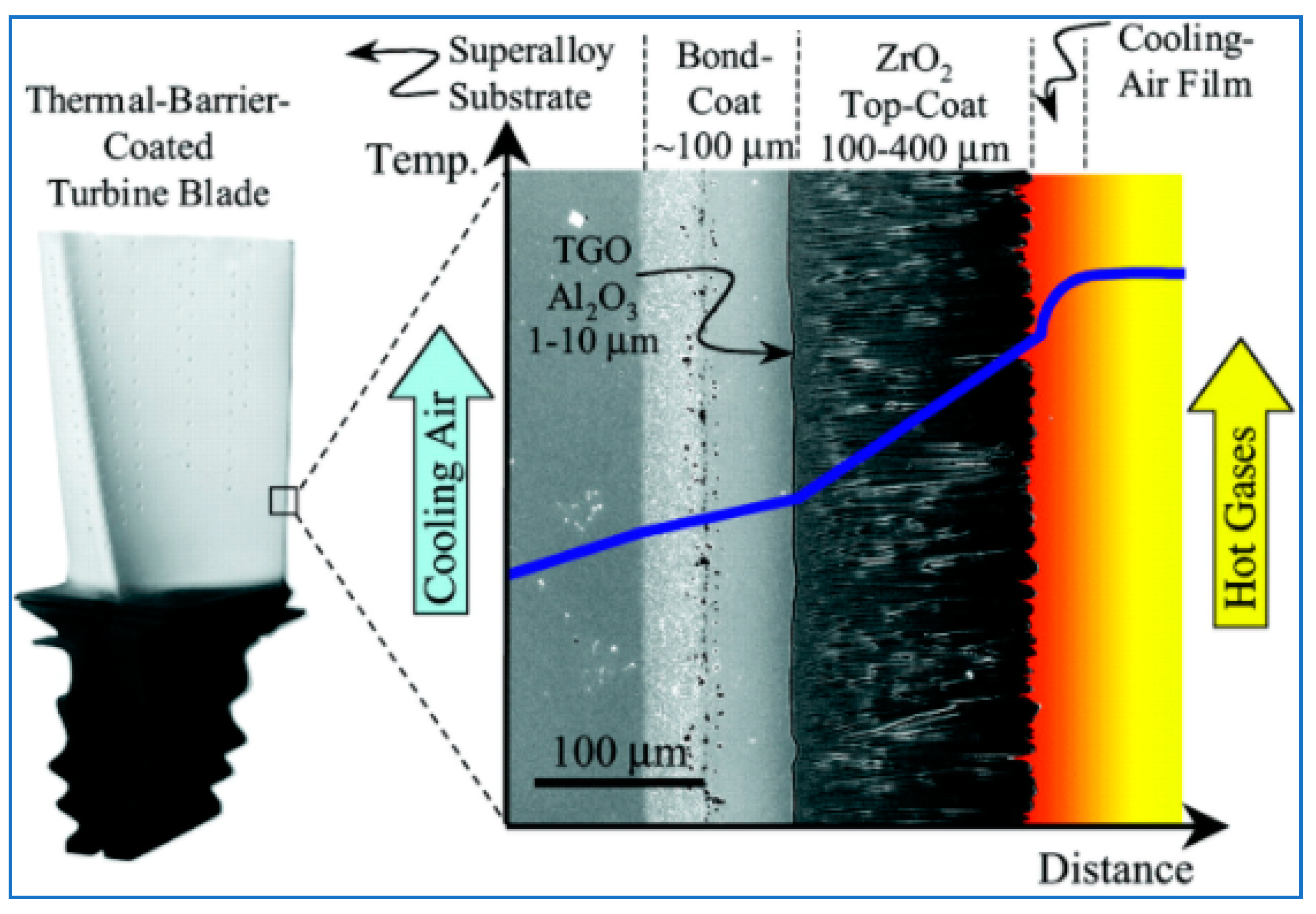
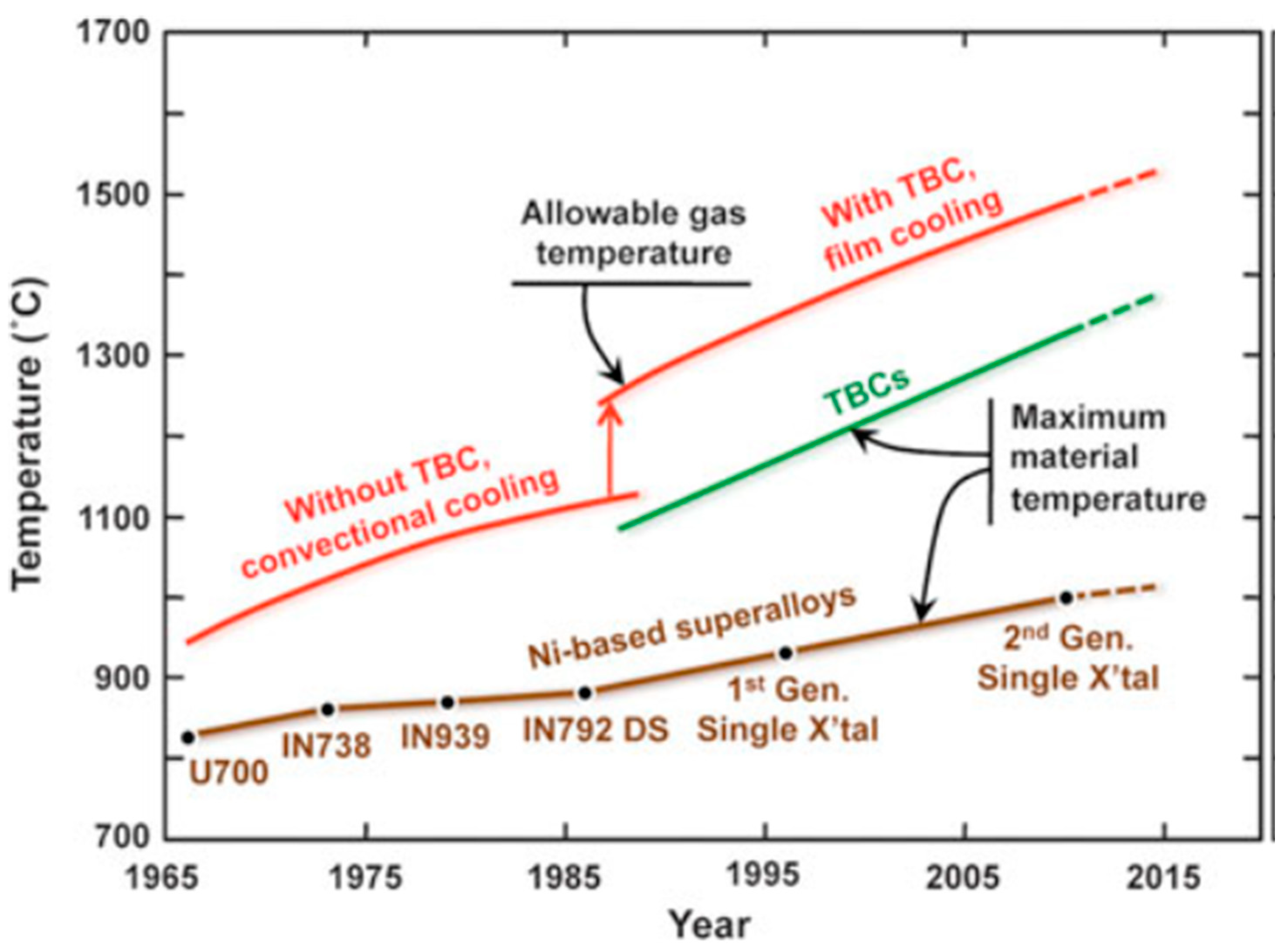


| Thermal Barrier Coating (TBC) | Environmental Barrier Coating (EBC) |
|---|---|
| Applied and used in hot gas engine environment on metal, their role is to protect the engine components in the hot gas path from the effects of the operating temperature. | Used on ceramic matrix composites (CMCs) or for any high temperature application where oxygen is present, it is to help decreasing oxidation-induced recession in silicon-based ceramic composites, and a need to lessen the operating temperature to weaken creep in oxide-based composites. |
| An aero TBC is a zirconia-yttria (or other zirconia based) ceramic on a metallic MCrAlY bond coat over a superalloy. MCrAlY coatings (where M = Co, Ni or Co/Ni) are widely applied to first and second stage turbine blades and nozzle guide vanes, where they may be used as corrosion resistant overlays or as bond-coats for use with thermal barrier coatings. | An EBC or EBC/TBC has a zirconia or hafnia TBC top coat for thermal insulation over a silicate-ceramic environmental coating to protect the substrate from water-vapor attack. |
| Because of temperature, a buildup of a layer called thermally grown oxide (TGO) is generated between the ceramic layer and the bond coat. This is due to the oxidation effect of the bond coat during oxidation and thermal shock. The TGO is to hinder the process of oxidation of the bond coat. | A silicon bond coat is between the EBC and the substrate, which is a CMC. EBC are for ceramic and CMC substrates. EBC role is to provide protection from environmental assault. |
| TBCs are for metallic substrates and provide thermal protection. The TBC top coat is in compression. | EBC top coat is in tension. Cracks are easy to form, but tension cracks in EBCs are not as damaging as compression cracks are in TBCs |
| Compared to EBCs, TBCs have better strain tolerance. | Both TBCs and EBCs require a bond coat on top of their substrate followed by a top coat on the surface. Bond coats may be multilayered. |
| The major trigger of failure in thermal barrier coating is the stresses, they generally initiated by bond coat oxidation, bond coat surface irregularities, yttria stabilized zirconia (YSZ) phase transformation, and YSZ sintering [5]. | Environmental barrier coating failure is generally triggered by chemical reactions not stresses. They further lead to degradation and spallation. Lead life-limiting reactions are water vapor volatility of the surface layer, chemical reactions between various EBC layers, including silica TGO, and the oxidation of silicon bond coat. Therefore, EBC design should take into account the latter effects and ensure that chemical reactions are limited to minimum [5]. |
| Typical thickness: 1–1.5 mil bond coat, 3–4 mil top coats. | Typical Thickness: 15 mil for combustor (rough coating), 5–10 mil for vane (smooth), 5 mil goal for blade (smooth). |
| Features | Evaporation | Sputtering Deposition | CVD | Electro Deposition | Thermal Spraying |
|---|---|---|---|---|---|
| Mechanism to produce deposition species | Thermal energy | Momentum transfer | Chemicals reaction | Solution | Flame or Plasma |
| Deposition rate | Moderate up to 750,000 A/min | Low | Moderate | Low to High | Very high |
| Deposition species | Atoms | Atoms/Ions | Atoms/ion | Ions | Droplets |
| Complex Shape | Poor line of sight | Good but non uniform | Good | Good | Poor resolution |
| Deposits in small, blind holes | Poor | Poor | Limited | Limited | Very Limited |
| Metal/alloy deposition | Yes | Yes | Yes | Yes | Yes |
| Refractory compounds and ceramics | Yes | Yes | Yes | Limited | Yes |
| Energy of deposits species | Low | Can be high | Can be high | Can be high | Can be high |
| Growth interface perturbation | Not normally | Yes | Yes | No | No |
| Substrate heating | Yes normally | Not generally | Yes | No | Not normally |
© 2018 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abdul-Aziz, A. Durability Modeling Review of Thermal- and Environmental-Barrier-Coated Fiber-Reinforced Ceramic Matrix Composites Part I. Materials 2018, 11, 1251. https://doi.org/10.3390/ma11071251
Abdul-Aziz A. Durability Modeling Review of Thermal- and Environmental-Barrier-Coated Fiber-Reinforced Ceramic Matrix Composites Part I. Materials. 2018; 11(7):1251. https://doi.org/10.3390/ma11071251
Chicago/Turabian StyleAbdul-Aziz, Ali. 2018. "Durability Modeling Review of Thermal- and Environmental-Barrier-Coated Fiber-Reinforced Ceramic Matrix Composites Part I" Materials 11, no. 7: 1251. https://doi.org/10.3390/ma11071251
APA StyleAbdul-Aziz, A. (2018). Durability Modeling Review of Thermal- and Environmental-Barrier-Coated Fiber-Reinforced Ceramic Matrix Composites Part I. Materials, 11(7), 1251. https://doi.org/10.3390/ma11071251





