Effect of PDLA and Amide Compounds as Mixed Nucleating Agents on Crystallization Behaviors of Poly (l-lactic Acid)
Abstract
1. Introduction
2. Materials and Methods
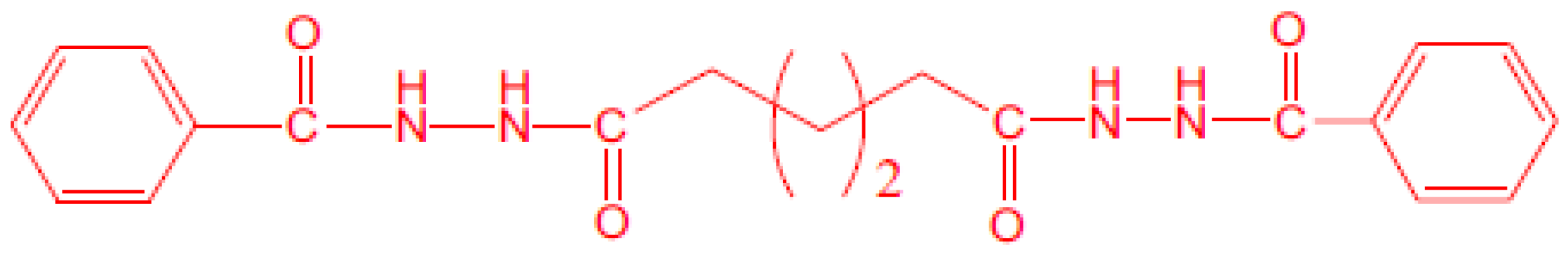
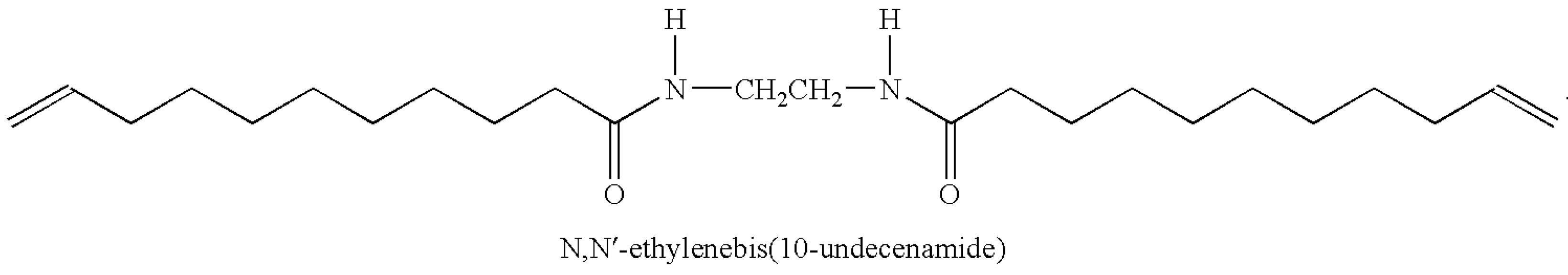
2.1. Materials
2.2. Sample Preparations
2.3. Characterization
2.3.1. Differential Scanning Calorimetry
2.3.2. WAXD Measurement
2.3.3. Rheological Measurement
3. Results
3.1. Crystal Behaviors of PLLA with PDLA
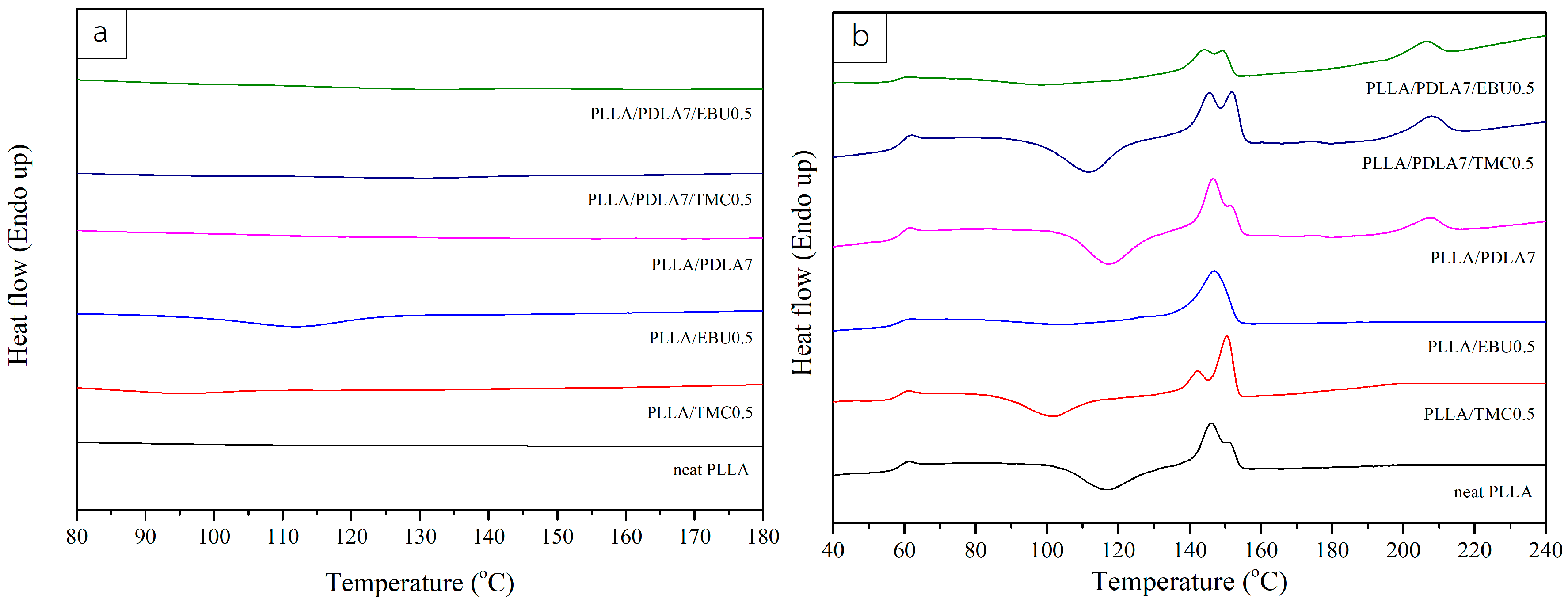
3.2. Thermal Characteristics of Nucleated PLLA
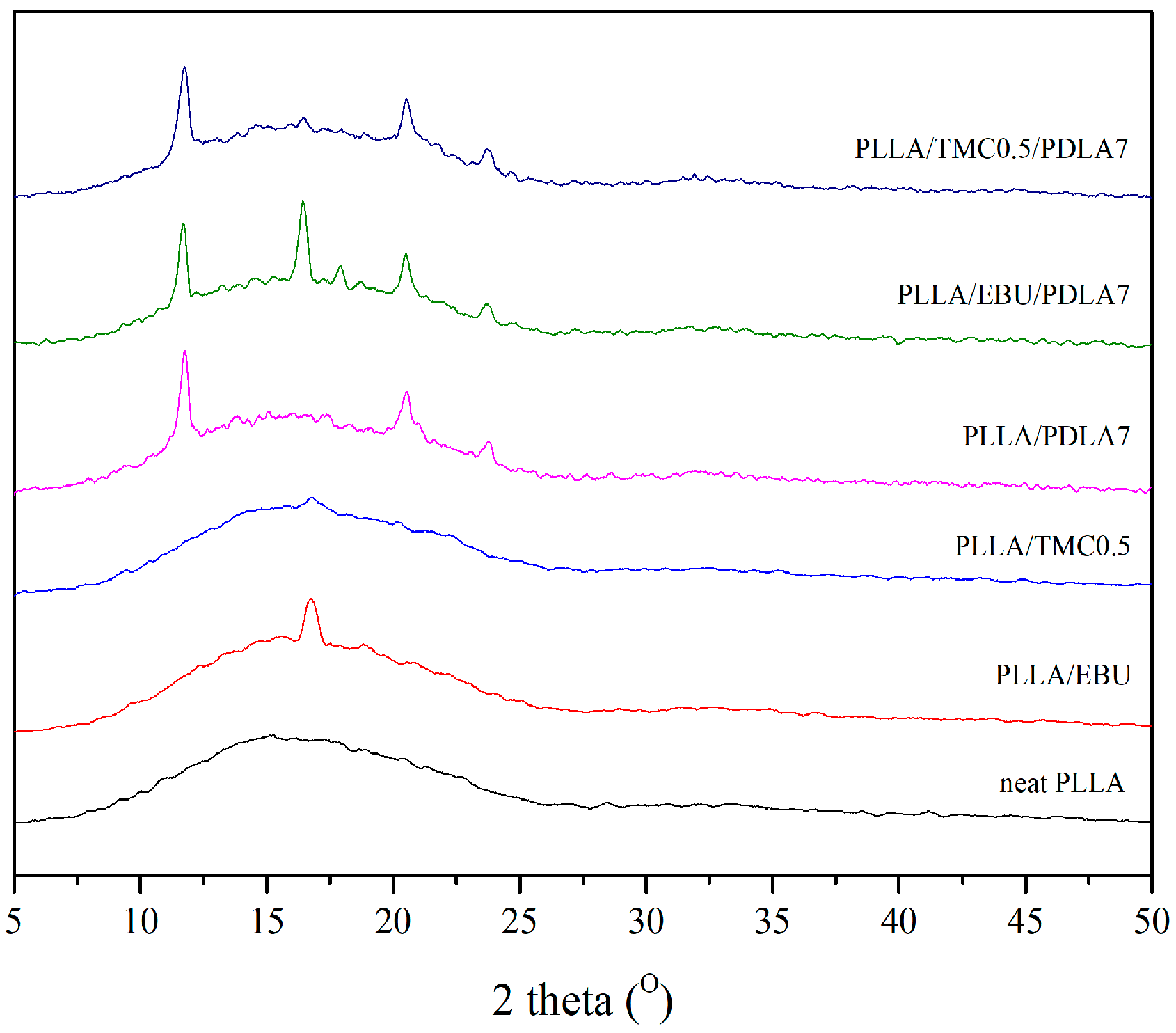
3.3. Crystal Structures of Nucleated PLLA
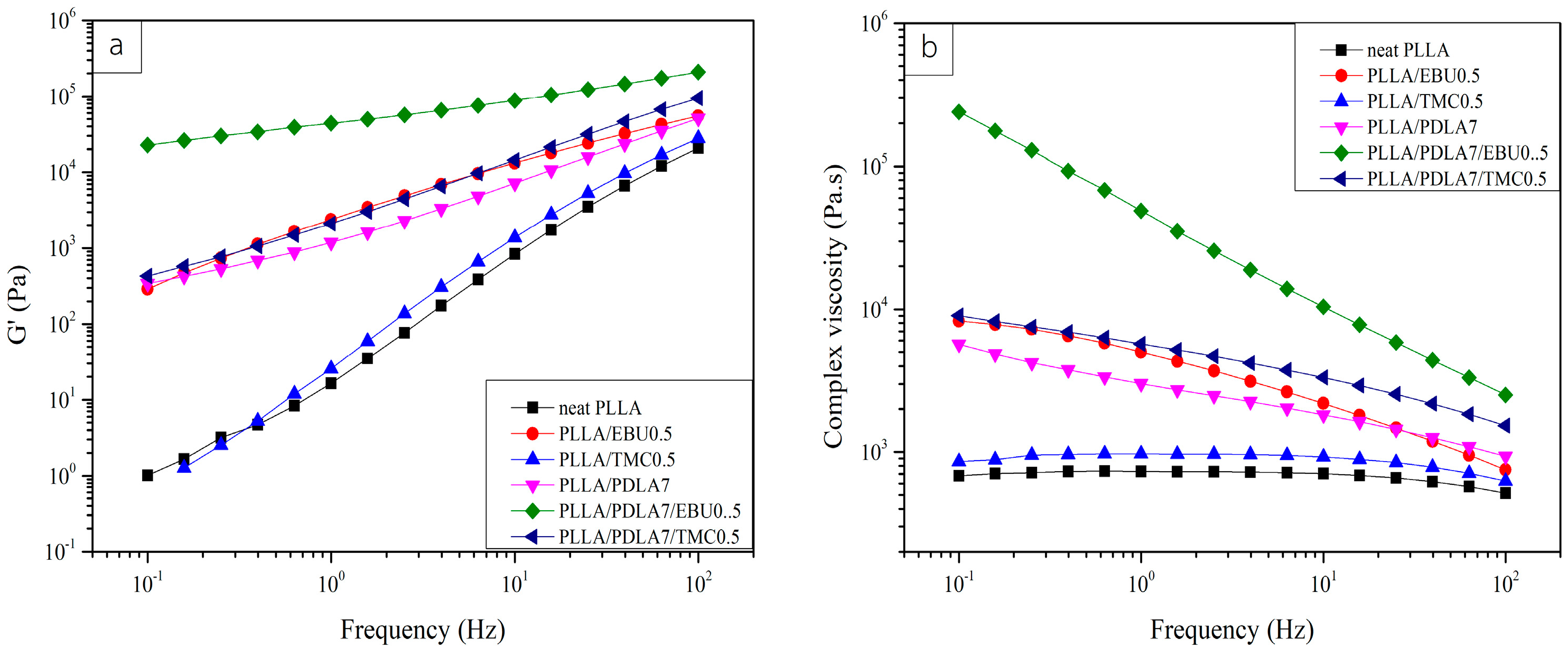
3.4. Rheological Properties
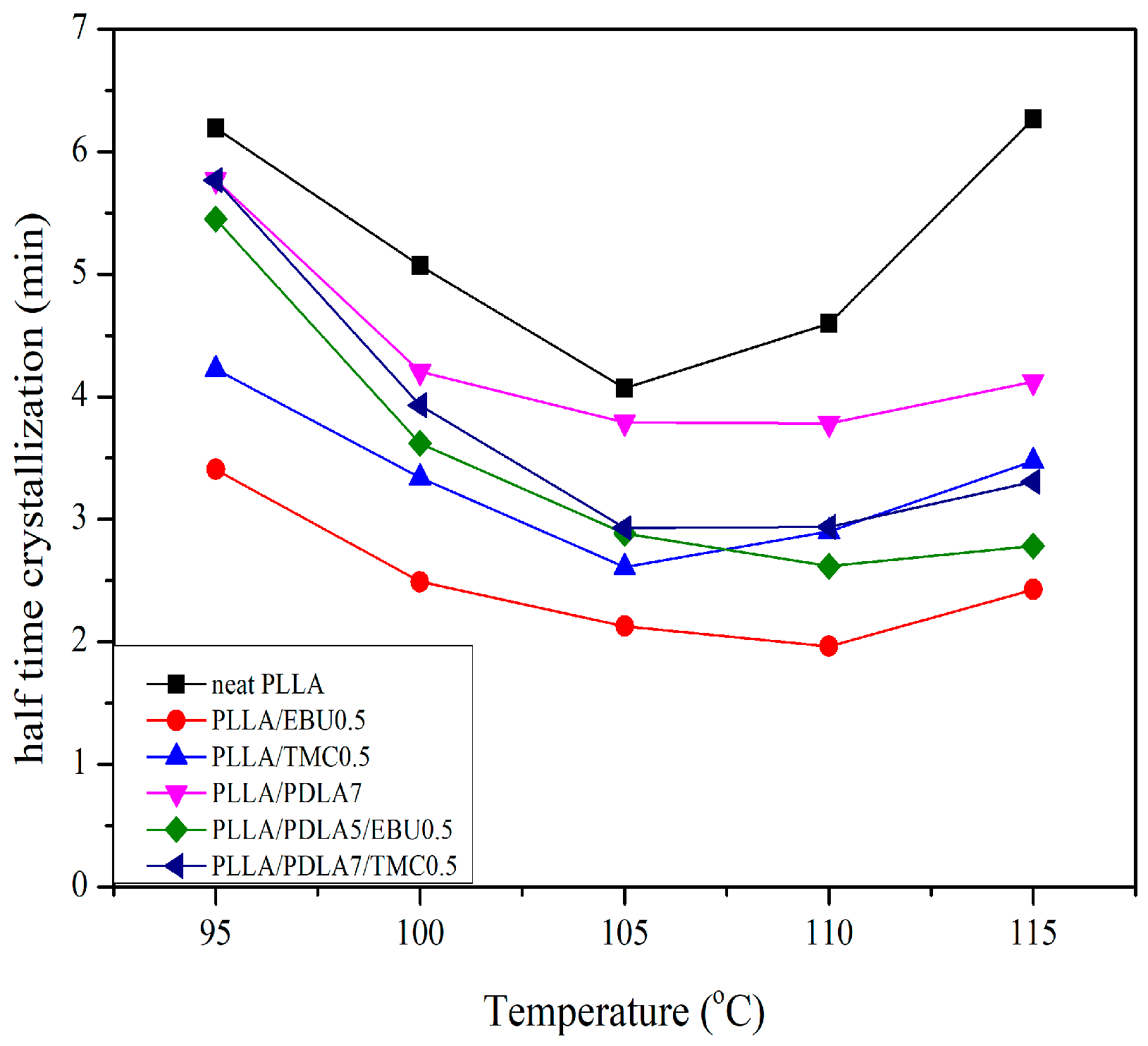
3.5. Isothermal Crystallization Behaviors
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Jiang, X.; Luo, Y. Chemical Structure of Poly(Lactic Acid); In Poly(Lactic Acid); John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2010; pp. 67–82. ISBN 9780470649848. [Google Scholar]
- Dorgan, J.R.; Lehermeier, H. Thermal and Rheological Properties of Commercial-Grade Poly(Lactic Acid)s. J. Polym. Environ. 2000, 8, 1–9. [Google Scholar] [CrossRef]
- Harris, A.M.; Lee, E.C. Improving mechanical performance of injection molded PLA by controlling crystallinity. J. Appl. Polym. Sci. 2008, 107, 2246–2255. [Google Scholar] [CrossRef]
- Zhang, X.; Meng, L. Effect of nucleating agents on the crystallization behavior and heat resistance of poly(l-lactide). J. Appl. Polym. Sci. 2016, 133. [Google Scholar] [CrossRef]
- Shakoor, A.; Thomas, N.L. Talc as a nucleating agent and reinforcing filler in poly(lactic acid) composites. Polym. Eng. Sci. 2014, 54, 64–70. [Google Scholar] [CrossRef]
- Nagarajan, V.; Mohanty, A.K. Crystallization behavior and morphology of polylactic acid (PLA) with aromatic sulfonate derivative. J. Appl. Polym. Sci. 2016, 133. [Google Scholar] [CrossRef]
- Wang, L.; Wang, Y.-N. Heat resistance, crystallization behavior, and mechanical properties of polylactide/nucleating agent composites. Mater. Des. 2015, 66, 7–15. [Google Scholar] [CrossRef]
- Xing, Q.; Zhang, X. Low-molecular weight aliphatic amides as nucleating agents for poly (L-lactic acid): Conformation variation induced crystallization enhancement. Polymer 2012, 53, 2306–2314. [Google Scholar] [CrossRef]
- Nanthananon, P.; Seadan, M. Enhanced Crystallization of Poly (Lactic Acid) through Reactive Aliphatic Bisamide. IOP Conf. Ser. Mater. Sci. Eng. 2015, 87, 012067. Available online: http://stacks.iop.org/1757-899X/87/i=1/a=012067 (accessed on 5 May 2018). [CrossRef]
- Tsuji, H. Poly(lactide) Stereocomplexes: Formation, Structure, Properties, Degradation, and Applications. Macromol. Biosci. 2005, 5, 569–597. [Google Scholar] [CrossRef] [PubMed]
- Wei, X.-F.; Bao, R.-Y. Stereocomplex Crystallite Network in Asymmetric PLLA/PDLA Blends: Formation, Structure, and Confining Effect on the Crystallization Rate of Homocrystallites. Macromolecules 2014, 47, 1439–1448. [Google Scholar] [CrossRef]
- Brennan, D.J.; White, J.E. Polyepoxy Compounds Having an Amide Linkage. U.S. Patent No. 7,300,986, 27 November 2007. [Google Scholar]
- Ahmed, J. Thermal Properties of Polylactides and Stereocomplex. In Glass Transition and Phase Transitions in Food and Biological Materials; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2017; pp. 261–279. ISBN 9781118935682. [Google Scholar]
- Song, P.; Wei, Z. Crystallization behavior and nucleation analysis of poly(l-lactic acid) with a multiamide nucleating agent. Polym. Eng. Sci. 2012, 52, 1058–1068. [Google Scholar] [CrossRef]

| Sample | Content of Substance | ||||
|---|---|---|---|---|---|
| PLLA (%) | PDLA (%) | TMC (%) | EBU (%) | Peroxide (phr) | |
| Neat PLLA | 100.0 | - | - | - | - |
| PLLA/EBU0.5 * | 99.5 | - | - | 0.5 | 0.1 |
| PLLA/TMC0.5 | 99.5 | - | 0.5 | - | - |
| PLLA/PDLA7 | 93.0 | 7.0 | - | - | - |
| PLLA/PDLA7/EBU0.5 * | 92.5 | 7.0 | - | 0.5 | 0.1 |
| PLLA/PDLA7/TMC0.5 | 92.5 | 7.0 | 0.5 | - | - |
| Sample Name | Tg (°C) | Tc (°C) | Tcc (°C) | Tm (°C) | Hm (J/g) | Hcc (J/g) | Xc (%) | Tsc (°C) | Hm,sc (J/g) |
|---|---|---|---|---|---|---|---|---|---|
| Neat PLLA | 58.7 | - | 117.1 | 146.1, 149.8 | 26.37 | 22.84 | 3.80 | - | - |
| PLLA/EBU0.5 * | 59.1 | 112.3 | 102.6 | 146.7 | 25.15 | 2.04 | 24.94 | - | - |
| PLLA/TMC0.5 | 58.4 | 97.5 | 102.0 | 142.2, 150.4 | 25.75 | 17.74 | 8.61 | - | - |
| PLLA/PDLA7 | 59.6 | - | 117.2 | 146.6, 150.7 | 25.82 | 19.45 | 7.35 | 207.0 | 6.20 |
| PLLA/EBU0.5/PDLA7 * | 58.9 | - | 98.5 | 144.0, 149.1 | 19.48 | 6.87 | 14.64 | 206.0 | 6.66 |
| PLLA/TMC0.5/PDLA7 | 59.8 | - | 111.8 | 145.6, 151.8 | 25.45 | 18.05 | 8.59 | 207.9 | 7.47 |
| Sample Name | Temp. (°C) | n | k (min−1) | t1/2 (min) |
|---|---|---|---|---|
| Neat PLLA | 95 | 2.5 | 0.0073 | 6.19 |
| 100 | 2.9 | 0.0063 | 5.07 | |
| 105 | 2.7 | 0.0157 | 4.07 | |
| 110 | 2.5 | 0.0153 | 4.60 | |
| 115 | 2.3 | 0.0102 | 6.26 | |
| PLLA/EBU0.5 * | 95 | 2.7 | 0.0253 | 3.41 |
| 100 | 2.8 | 0.0539 | 2.49 | |
| 105 | 3.0 | 0.0721 | 2.13 | |
| 110 | 3.1 | 0.0858 | 1.96 | |
| 115 | 3.0 | 0.0486 | 2.43 | |
| PLLA/TMC0.5 | 95 | 2.4 | 0.0218 | 4.22 |
| 100 | 2.8 | 0.0236 | 3.34 | |
| 105 | 2.6 | 0.0573 | 2.61 | |
| 110 | 3.2 | 0.0229 | 2.90 | |
| 115 | 3.8 | 0.0061 | 3.48 | |
| PLLA/PDLA7 | 95 | 2.6 | 0.0073 | 5.77 |
| 100 | 2.9 | 0.0108 | 4.20 | |
| 105 | 3.0 | 0.0127 | 3.79 | |
| 110 | 3.0 | 0.0128 | 3.79 | |
| 115 | 2.8 | 0.0131 | 4.12 | |
| PLLA/PDLA7/TMC0.5 | 95 | 2.3 | 0.0123 | 5.77 |
| 100 | 2.5 | 0.0226 | 3.93 | |
| 105 | 2.6 | 0.0425 | 2.93 | |
| 110 | 3.2 | 0.0220 | 2.94 | |
| 115 | 3.3 | 0.0134 | 3.31 | |
| PLLA/PDLA7/EBU0.5 * | 95 | 2.9 | 0.0051 | 5.45 |
| 100 | 3.1 | 0.0129 | 3.62 | |
| 105 | 3.0 | 0.0289 | 2.88 | |
| 110 | 3.1 | 0.0352 | 2.62 | |
| 115 | 3.2 | 0.0263 | 2.78 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khwanpipat, T.; Seadan, M.; Suttiruengwong, S. Effect of PDLA and Amide Compounds as Mixed Nucleating Agents on Crystallization Behaviors of Poly (l-lactic Acid). Materials 2018, 11, 1139. https://doi.org/10.3390/ma11071139
Khwanpipat T, Seadan M, Suttiruengwong S. Effect of PDLA and Amide Compounds as Mixed Nucleating Agents on Crystallization Behaviors of Poly (l-lactic Acid). Materials. 2018; 11(7):1139. https://doi.org/10.3390/ma11071139
Chicago/Turabian StyleKhwanpipat, Thanawat, Manus Seadan, and Supakij Suttiruengwong. 2018. "Effect of PDLA and Amide Compounds as Mixed Nucleating Agents on Crystallization Behaviors of Poly (l-lactic Acid)" Materials 11, no. 7: 1139. https://doi.org/10.3390/ma11071139
APA StyleKhwanpipat, T., Seadan, M., & Suttiruengwong, S. (2018). Effect of PDLA and Amide Compounds as Mixed Nucleating Agents on Crystallization Behaviors of Poly (l-lactic Acid). Materials, 11(7), 1139. https://doi.org/10.3390/ma11071139





