Scanning Kelvin Probe Microscopy: Challenges and Perspectives towards Increased Application on Biomaterials and Biological Samples
Abstract
1. Introduction
2. Considerations about SKPM Data
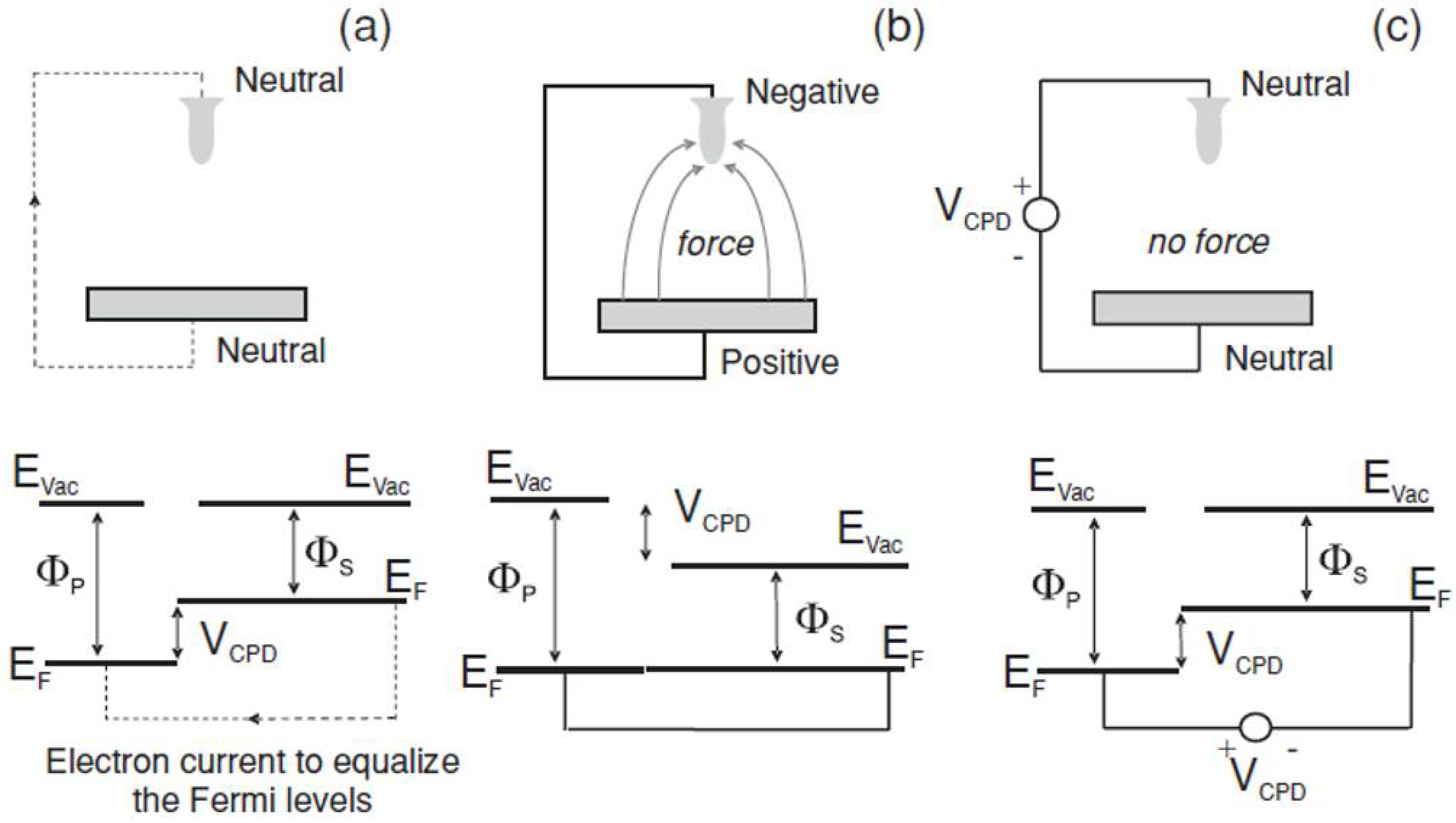
2.1. SKPM Image Contrast: Meaning and Limitations
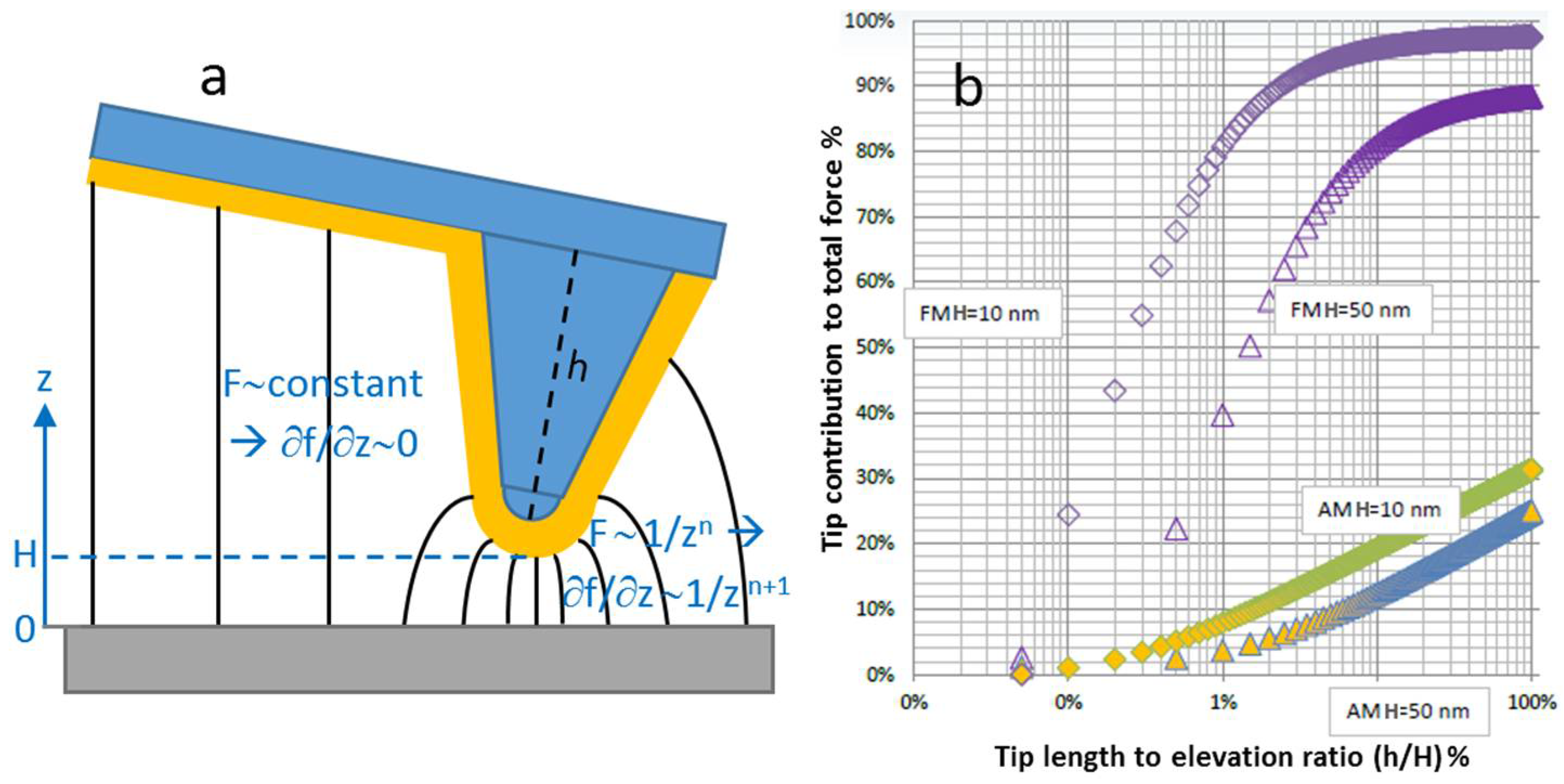
2.2. More Issues in SKPM Imaging: The Technique
3. SKPM Imaging of Biomaterials and Biological Samples
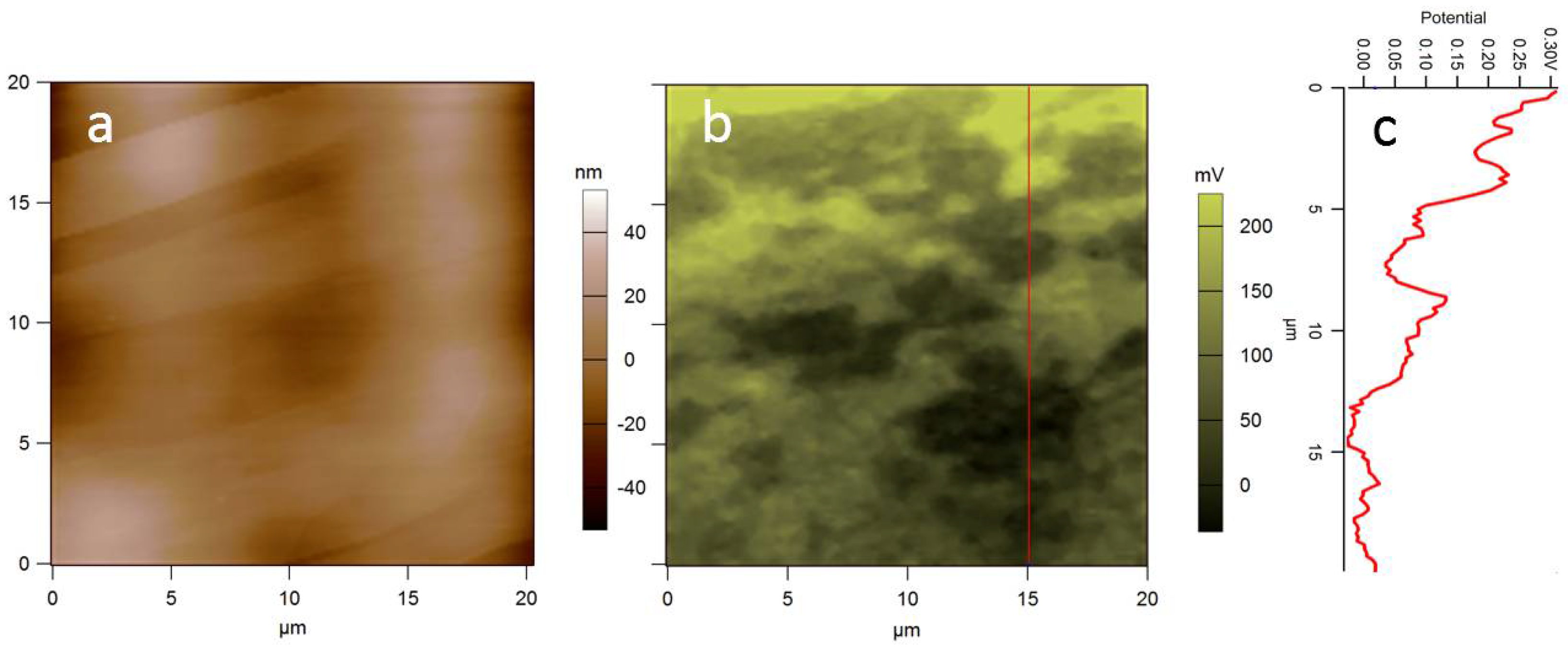
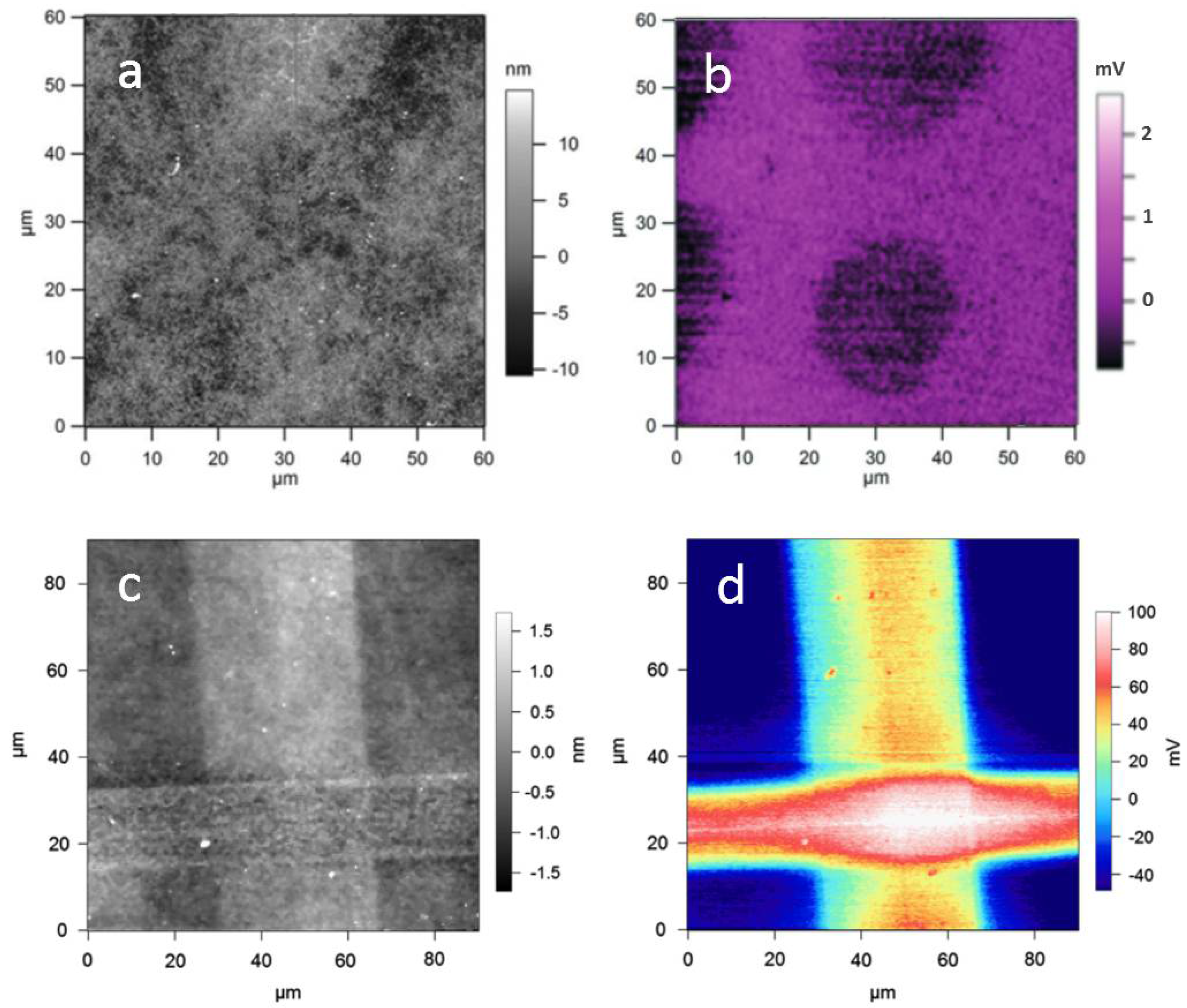
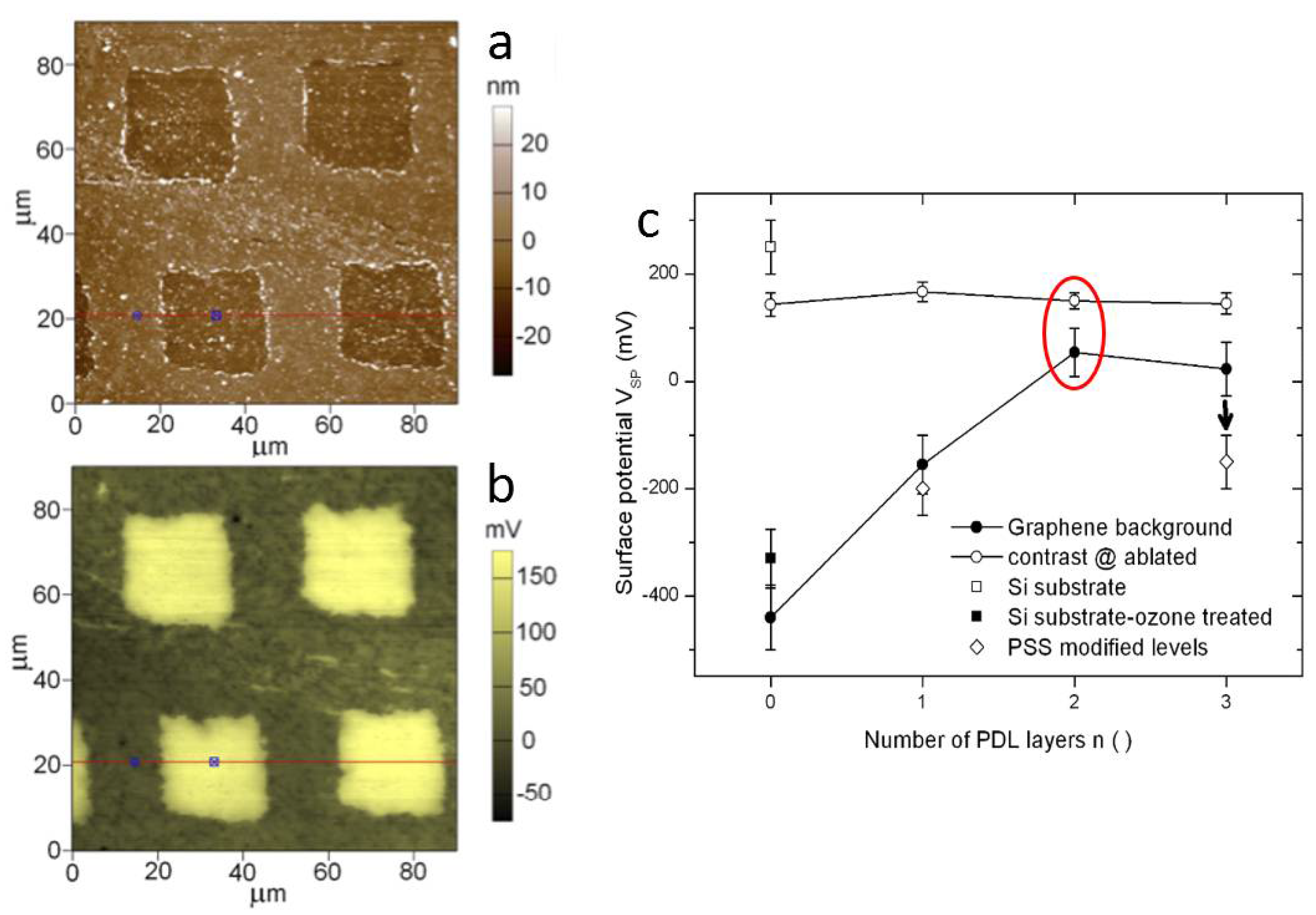
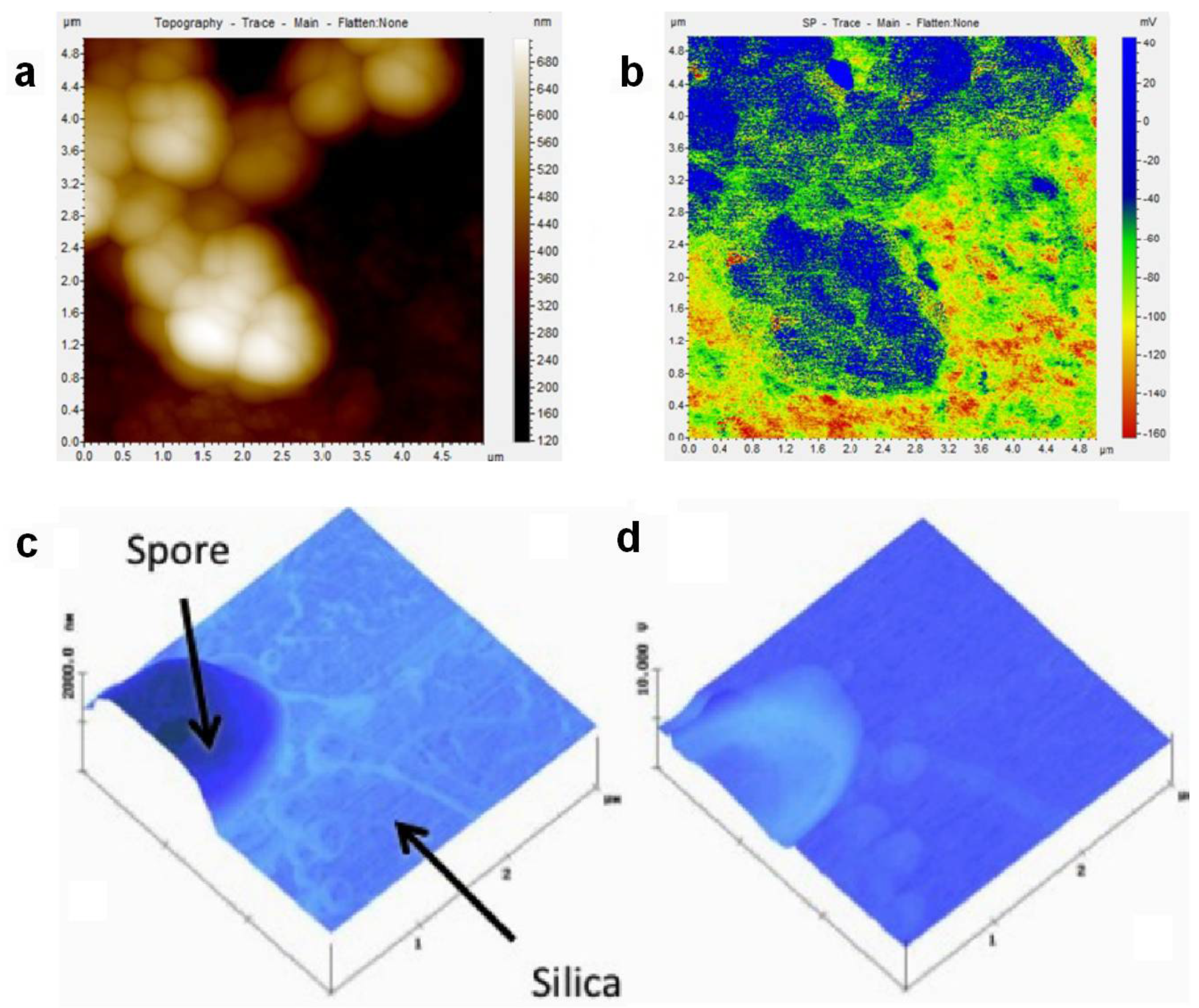
3.1. Additive Lithography-Manufactured Bio-Substrates
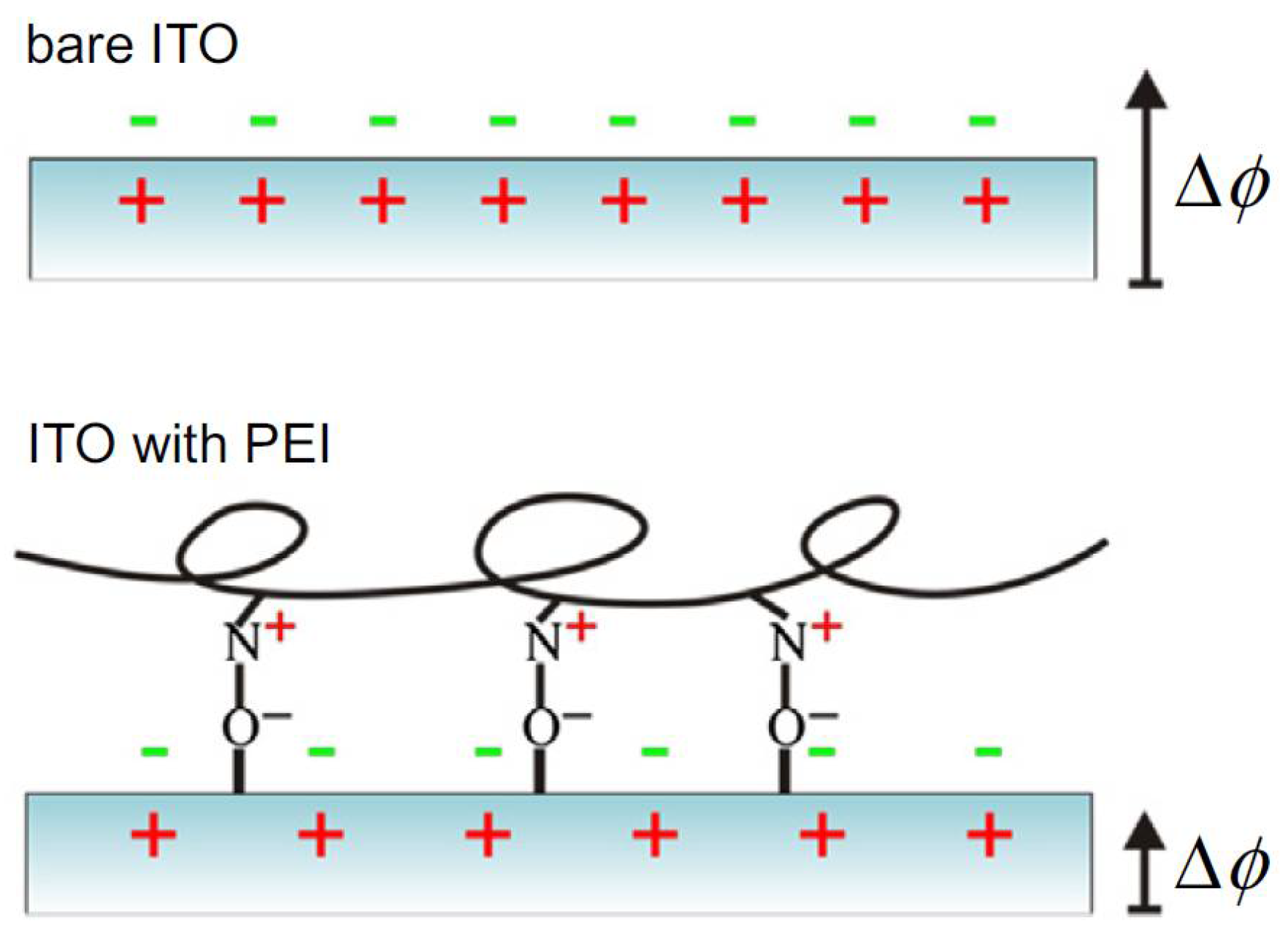
3.2. Subtractive Lithography-Manufactured Bio-Substrates
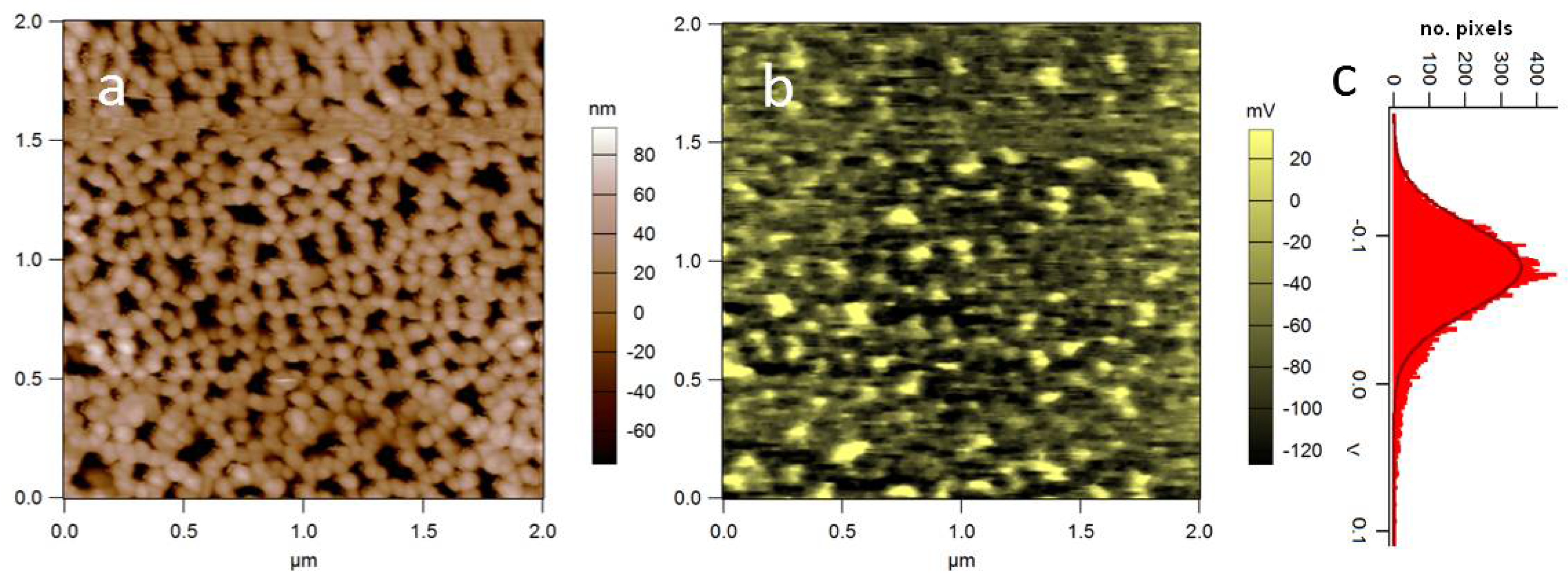
3.3. A Self-Organized Biosubstrate: Anodic Porous Alumina
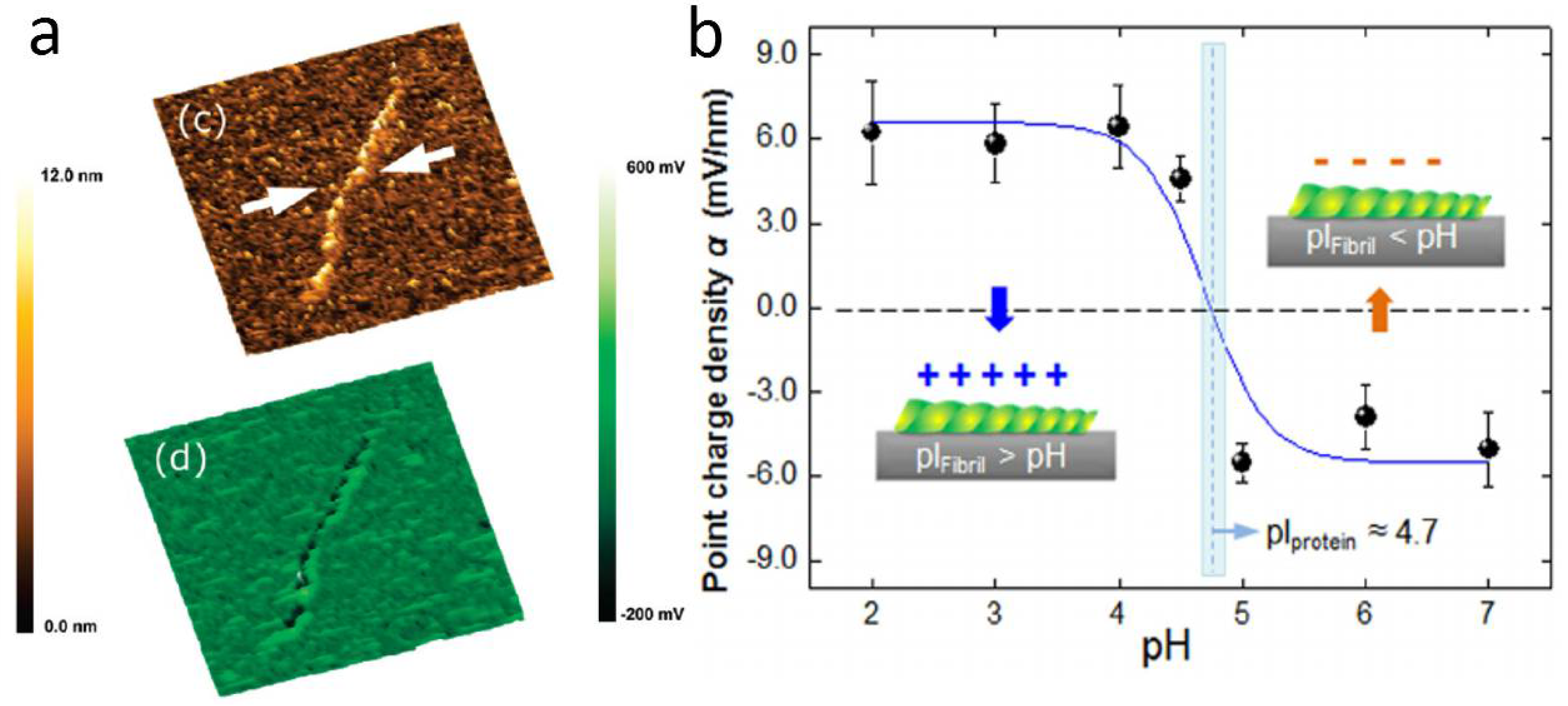
3.4. Single Biomolecular Constructs
3.5. SKPM of Living Cells
4. Conclusions and Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- Melitz, W.; Shen, J.; Kummel, A.C.; Lee, S. Kelvin probe force microscopy and its application. Surf. Sci. Rep. 2011, 66, 1–27. [Google Scholar] [CrossRef]
- Kalinin, S.V.; Gruverman, A. Scanning Probe Microscopy, Electrical and Electromechanical Phenomena at the Nanoscale; Springer: New York, NY, USA, 2007; Volume II, ISBN 9780387286679. [Google Scholar]
- Lanza, M. Conductive Atomic Force Microscopy; Wiley-VCH: Weinheim, Germany, 2017; ISBN 9783527340910. [Google Scholar]
- Davis, M.E.; Mccammon, J.A. Electrostatics in Biomolecular Structure and Dynamics. Chem. Rev. 1990, 90, 509–521. [Google Scholar] [CrossRef]
- Huang, Y.; Samorajski, J.; Kreimer, R.; Searson, P.C. The Influence of Electric Field and Confinement on Cell Motility. PLoS ONE 2013, 8, e59447. [Google Scholar] [CrossRef] [PubMed]
- Funk, R.H.W. Endogenous electric fields as guiding cue for cell migration. Front. Physiol. 2015, 6, 143. [Google Scholar] [CrossRef] [PubMed]
- Kelvin, L.V. Contact electricity of metals. Lond. Edinb. Dublin Philos. Mag. J. Sci. 1898, 46, 82–120. [Google Scholar] [CrossRef]
- Asylum-Research. Chapter 10: Scanning Kelvin Probe Microscopy. In Applications Guide—USER GUIDE 3, Ver.13A; Asylum Research: Goleta, CA, USA, 2013; pp. 107–114. [Google Scholar]
- Seo, H.; Goo, D.; Jung, G. How to Obtain Sample Potential Data for SKPM Measurement. Available online: https://www.parksystems.com/images/media/appnote/How%20to%20obtain%20sample%20potential%20data%20for%20SKPM%20measurement.pdf (accessed on 3 June 2018).
- Beerbom, M.M.; Lägel, B.; Cascio, A.J.; Doran, B.V.; Schlaf, R. Direct comparison of photoemission spectroscopy and in situ Kelvin probe work function measurements on indium tin oxide films. J. Electron. Spectrosc. Relat. Phenom. 2006, 152, 12–17. [Google Scholar] [CrossRef]
- Lorenzoni, M.; Brandi, F.; Dante, S.; Giugni, A.; Torre, B. Simple and effective graphene laser processing for neuron patterning application. Sci. Rep. 2013, 3, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Rumble, J.R. CRC Handbook of Chemistry and Physics, 95th ed.; CRC Press: Boca Raton, FL, USA, 2014; ISBN 1482208679. [Google Scholar]
- Abdellatif, M.H.; Ghosh, S.; Liakos, I.; Scarpellini, A.; Marras, S.; Diaspro, A.; Salerno, M. Effect of nanoscale size and medium on metal work function in oleylamine-capped gold nanocrystals. J. Phys. Chem. Solids 2016, 89, 7–14. [Google Scholar] [CrossRef]
- Ellison, D.J.; Lee, B.; Podzorov, V.; Frisbie, C.D. Surface potential mapping of SAM-functionalized organic semiconductors by Kelvin probe force microscopy. Adv. Mater. 2011, 23, 502–507. [Google Scholar] [CrossRef] [PubMed]
- Castellani, M.; Winkler, S.; Bröker, B.; Baumgarten, M.; Müllen, K.; Koch, N. Work function increase of transparent conductive electrodes by solution processed electron acceptor molecular monolayers. Appl. Phys. A Mater. Sci. Process. 2014, 114, 291–295. [Google Scholar] [CrossRef]
- Dawidczyk, T.J.; Martínez Hardigree, J.F.; Johns, G.L.; Ozgun, R.; Alley, O.; Andreou, A.G.; Markovic, N.; Katz, H.E. Visualizing and quantifying charge distributions correlated to threshold voltage shifts in lateral organic transistors. ACS Nano 2014, 8, 2714–2724. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, J.F.; Somoza, A.M.; Palacios-Lidón, E. Charge distribution from SKPM images. Phys. Chem. Chem. Phys. 2017, 19, 27299–27304. [Google Scholar] [CrossRef] [PubMed]
- Nonnenmacher, M.; O’Boyle, M.P.; Wickramasinghe, H.K. Kelvin probe force microscopy. Appl. Phys. Lett. 1991, 58, 2921–2923. [Google Scholar] [CrossRef]
- Kolchuzhin, V.A.; Sheremet, E.; Bhattacharya, K.; Rodriguez, R.D.; Paul, S.D.; Mehner, J.; Hietschold, M.; Zahn, D.R.T. Mechanical properties and applications of custom-built gold AFM cantilevers. Mechatronics 2016, 40, 281–286. [Google Scholar] [CrossRef]
- Charrier, D.S.H.; Kemerink, M.; Smalbrugge, B.E.; de Vries, T.; Janssen, R.A.J. Real versus measured surface potentials in scanning Kelvin probe microscopy. ACS Nano 2008, 2, 622–626. [Google Scholar] [CrossRef] [PubMed]
- Cherniavskaya, O.; Chen, L.; Weng, V.; Yuditsky, L.; Brus, L.E. Quantitative noncontact electrostatic force imaging of nanocrystal polarizability. J. Phys. Chem. B 2003, 107, 1525–1531. [Google Scholar] [CrossRef]
- Koley, G.; Spencer, M.G.; Bhangale, H.R. Cantilever effects on the measurement of electrostatic potentials by scanning Kelvin probe microscopy Cantilever effects on the measurement of electrostatic potentials by scanning Kelvin probe microscopy. Appl. Phys. Lett. 2011, 545, 1999–2002. [Google Scholar] [CrossRef]
- Elias, G.; Glatzel, T.; Meyer, E.; Schwarzman, A.; Boag, A.; Rosenwaks, Y. The role of the cantilever in Kelvin probe force microscopy measurements. Beilstein J. Nanotechnol. 2011, 2, 252–260. [Google Scholar] [CrossRef] [PubMed]
- Bruker Application Note #140: PeakForce Kelvin Probe Force Microscopy. Available online: https://bruker.com/fileadmin/user_upload/8-PDF-Docs/SurfaceAnalysis/AFM/ApplicationNotes/AN140-RevA1-PeakForce_KPFM-AppNote.pdf (accessed on 3 June 2018).
- Lee, W.; Jung, H.; Son, M.; Lee, H.; Kwak, T.J.; Lee, G.; Kim, C.H.; Lee, S.W.; Yoon, D.S. Characterization of the regrowth behavior of amyloid-like fragmented fibrils decomposed by ultrasonic treatment. RSC Adv. 2014, 4, 56561–56566. [Google Scholar] [CrossRef]
- Park, J.; Yang, J.; Lee, C.Y.; Na, S.; Lee, S.W.; Haam, S.; Huh, Y.-M.; Yoon, D.S.; Eom, K.; Kwon, T. Single-molecule recognition of biomolecular interaction via KPFM.pdf. ACS Nano 2011, 5, 6981–6990. [Google Scholar] [CrossRef] [PubMed]
- Park, J.; Bang, D.; Jang, K.; Haam, S.; Yang, J.; Na, S. The work function of doped polyaniline nanoparticles observed by Kelvin probe force microscopy. Nanotechnology 2012, 23, 365705. [Google Scholar] [CrossRef] [PubMed]
- Schedin, F.; Geim, A.K.; Morozov, S.V.; Hill, E.W.; Blake, P.; Katsnelson, M.I.; Novoselov, K.S. Detection of individual gas molecules adsorbed on graphene. Nat. Mater. 2007, 6, 652–655. [Google Scholar] [CrossRef] [PubMed]
- Ponomarenko, L.A.; Yang, R.; Mohiuddin, T.M.; Katsnelson, M.I.; Novoselov, K.S.; Morozov, S.V.; Zhukov, A.A.; Schedin, F.; Hill, E.W.; Geim, A.K. Effect of a high-κ environment on charge carrier mobility in graphene. Phys. Rev. Lett. 2009, 102, 100–103. [Google Scholar] [CrossRef] [PubMed]
- Saito, N.; Hayashi, K.; Sugimura, H.; Takai, O.; Nakagiri, N. Surface potential images of self-assembled monolayers patterned by organosilanes: Ab initio molecular orbital calculations. Surf. Interface Anal. 2002, 34, 601–605. [Google Scholar] [CrossRef]
- Fujihira, M. Kelvin probe force microscopy of molecular surfaces. Annu. Rev. Mater. Sci. 1999, 29, 353–380. [Google Scholar] [CrossRef]
- Lu, J.; Delamarche, E.; Eng, L.; Bennewitz, R.; Meyer, E.; Güntherodt, H. Kelvin probe force microscopy on surfaces: Investigation of the Surface potential of self-assembled monolayers on gold. Langmuir 1999, 15, 8184–8188. [Google Scholar] [CrossRef]
- Yan, L.; Song, Y.; Zhou, Y.; Song, B.; Li, Y. Effect of PEI cathode interlayer on work function and interface resistance of ITO electrode in the inverted polymer solar cells. Org. Electron. Phys. Mater. Appl. 2015, 17, 94–101. [Google Scholar] [CrossRef]
- Zhou, Y.; Fuentes-Hernandez, C.; Shim, J.; Meyer, J.; Giordano, A.J.; Li, H.; Winget, P.; Papadopoulos, T.; Cheun, H.; Kim, J.; et al. A universal method to produce low-work function electrodes for organic electronics. Science 2012, 336, 327–332. [Google Scholar] [CrossRef] [PubMed]
- Pelto, J.M.; Haimi, S.P.; Siljander, A.S.; Miettinen, S.S.; Tappura, K.M.; Higgins, M.J.; Wallace, G.G. Surface properties and interaction forces of biopolymer-doped conductive polypyrrole surfaces by atomic force microscopy. Langmuir 2013, 29, 6099–6108. [Google Scholar] [CrossRef] [PubMed]
- Martin, Y.; Abraham, D.W.; Wickramasinghe, H.K. High-resolution capacitance measurement and potentiometry by force microscopy. Appl. Phys. Lett. 1988, 52, 1103–1105. [Google Scholar] [CrossRef]
- Salerno, M.; Dante, S.; Patra, N.; Diaspro, A. AFM measurement of the stiffness of layers of agarose gel patterned with polylysine. Microsc. Res. Tech. 2010, 73, 982–990. [Google Scholar] [CrossRef] [PubMed]
- Petrelli, A.; Marconi, E.; Salerno, M.; De Pietri Tonelli, D.; Berdondini, L.; Dante, S. Nano-volume drop patterning for rapid on-chip neuronal connect-ability assays. Lab Chip 2013, 13, 4419. [Google Scholar] [CrossRef] [PubMed]
- Canale, C.; Petrelli, A.; Salerno, M.; Diaspro, A.; Dante, S. A new quantitative experimental approach to investigate single cell adhesion on multifunctional substrates. Biosens. Bioelectron. 2013, 48C, 172–179. [Google Scholar] [CrossRef] [PubMed]
- Feng, L.; Liu, Z. Graphene in biomedicine: Opportunities and challenges. Nanomedicine 2011, 6, 317–324. [Google Scholar] [CrossRef] [PubMed]
- Pinto, A.M.; Gonçalves, I.C.; Magalhães, F.D. Graphene-based materials biocompatibility: A review. Colloids Surf. B Biointerfaces 2013, 111, 188–202. [Google Scholar] [CrossRef] [PubMed]
- Kalbacova, M.; Broz, A.; Kong, J.; Kalbac, M. Graphene substrates promote adherence of human osteoblasts and mesenchymal stromal cells. Carbon 2010, 48, 4323–4329. [Google Scholar] [CrossRef]
- Aryaei, A.; Jayatissa, A.H.; Jayasuriya, A.C. The effect of graphene substrate on osteoblast cell adhesion and proliferation. J. Biomed. Mater. Res. Part A 2014, 102, 3282–3290. [Google Scholar] [CrossRef] [PubMed]
- Keshavan, S.; Oropesa-Nuñez, R.; Diaspro, A.; Canale, C.; Dante, S. Adhesion and migration of CHO cells on micropatterned single layer graphene. 2D Mater. 2017, 4. [Google Scholar] [CrossRef]
- Keshavan, S.; Naskar, S.; Diaspro, A.; Cancedda, L.; Dante, S. Developmental refinement of synaptic transmission on micropatterned single layer graphene. Acta Biomater. 2018, 65, 363–375. [Google Scholar] [CrossRef] [PubMed]
- Salerno, M.; El Merhie, A.; Diaspro, A.; Dante, S. Step-by-step Surface Potential Tuning of Patterned Graphene by Polyelectrolyte Coating. Thin Solid Films 2018. under review. [Google Scholar]
- Toccafondi, C.; Dante, S.; Reverberi, A.P.; Salerno, M. Biomedical applications of anodic porous alumina. Curr. Nanosci. 2015, 11, 572–580. [Google Scholar] [CrossRef]
- Grasso, V.; Lambertini, V.; Ghisellini, P.; Valerio, F.; Stura, E.; Perlo, P.; Nicolini, C. Nanostructuring of a porous alumina matrix for a biomolecular microarray. Nanotechnology 2006, 17, 795–798. [Google Scholar] [CrossRef]
- Toccafondi, C.; La Rocca, R.; Scarpellini, A.; Salerno, M.; Das, G.; Dante, S. Thin nanoporous alumina-based SERS platform for single cell sensing. Appl. Surf. Sci. 2015, 351. [Google Scholar] [CrossRef]
- Losic, D.; Editors, A.S. Nanoporous Alumina; Springer Series in Materials Science; Springer International Publishing: Basel, Switzerland, 2015; Volume 219, ISBN 978-3-319-20333-1. [Google Scholar]
- Poinern, G.E.J.; Shackleton, R.; Mamun, S.I.; Fawcett, D. Significance of novel bioinorganic anodic aluminum oxide nanoscaffolds for promoting cellular response. Nanotechnol. Sci. Appl. 2011, 4, 11–24. [Google Scholar] [CrossRef] [PubMed]
- Salerno, M.; Caneva-Soumetz, F.; Pastorino, L.; Patra, N.; Diaspro, A.; Ruggiero, C. Adhesion and proliferation of osteoblast-like cells on anodic porous alumina substrates with different morphology. IEEE Trans. Nanobiosci. 2013, 12, 106–111. [Google Scholar] [CrossRef] [PubMed]
- Brüggemann, D. Nanoporous aluminium oxide membranes as cell interfaces. J. Nanomater. 2013, 2013, 460870. [Google Scholar] [CrossRef]
- Thorat, S.; Diaspro, A.; Scarpellini, A.; Povia, M.; Salerno, M. Comparative study of loading of anodic porous alumina with silver nanoparticles using different methods. Materials 2013, 6, 206–216. [Google Scholar] [CrossRef] [PubMed]
- Kumeria, T.; Santos, A.; Losic, D. Nanoporous anodic alumina platforms: Engineered surface chemistry and structure for optical sensing applications. Sensors 2014, 14, 11878–11918. [Google Scholar] [CrossRef] [PubMed]
- Salerno, M.; Shayganpour, A.; Salis, B.; Dante, S. Surface-enhanced Raman scattering of self-assembled thiol monolayers and supported lipid membranes on thin anodic porous alumina. Beilstein J. Nanotechnol. 2017, 8, 74–81. [Google Scholar] [CrossRef] [PubMed]
- Das, G.; Patra, N.; Gopalakrishnan, A.; Zaccaria, R.P.; Toma, A.; Thorat, S.; Di Fabrizio, E.; Diaspro, A.; Salerno, M. Fabrication of large-area ordered and reproducible nanostructures for SERS biosensor application. Analyst 2012, 137, 1785–1792. [Google Scholar] [CrossRef] [PubMed]
- Toccafondi, C.; Zaccaria, R.P.; Dante, S.; Salerno, M. Fabrication of gold coated ultra thin anodic porous alumina substrates for augmented SERS. Materials 2016, 9, 403–414. [Google Scholar] [CrossRef] [PubMed]
- Sulka, G.D. Highly Ordered Anodic Porous Alumina; Wiley Online Library: Hoboken, NJ, USA, 2008; ISBN 9783527318766. [Google Scholar]
- Pedimonte, B.J.; Moest, T.; Luxbacher, T.; Von Wilmowsky, C.; Fey, T.; Schlegel, K.A.; Greil, P. Morphological zeta-potential variation of nanoporous anodic alumina layers and cell adherence. Acta Biomater. 2014, 10, 968–974. [Google Scholar] [CrossRef] [PubMed]
- Runge, J.M. The Metallurgy of Anodizing Aluminum—Connecting Science to Practice, 1st ed.; Springer International Publishing: Basel, Switzerland, 2018; ISBN 978-3-319-72175-0. [Google Scholar]
- Gao, P.; Cai, Y. Label-free detection of the aptamer binding on protein patterns using Kelvin probe force microscopy (KPFM). Anal. Bioanal. Chem. 2009, 394, 207–214. [Google Scholar] [CrossRef] [PubMed]
- Kwak, K.J.; Yoda, S.; Fujihira, M. Observation of stretched single DNA molecules by Kelvin probe force microscopy. Appl. Surf. Sci. 2003, 210, 73–78. [Google Scholar] [CrossRef]
- Leung, C.; Kinns, H.; Hoogenboom, B.W.; Howorka, S.; Mesquida, P. Imaging surface charges of individual biomolecules. Nano Lett. 2009, 9, 2769–2773. [Google Scholar] [CrossRef] [PubMed]
- Leung, C.; Maradan, D.; Kramer, A.; Howorka, S.; Mesquida, P.; Hoogenboom, B.W. Improved Kelvin probe force microscopy for imaging individual DNA molecules on insulating surfaces. Appl. Phys. Lett. 2010, 97, 20–22. [Google Scholar] [CrossRef]
- Lee, W.; Lee, H.; Woo Lee, S.; Sung Yoon, D.; Eom, K.; Kwon, T. Mapping the surface charge distribution of amyloid fibril. Appl. Phys. Lett. 2012, 101, 43703–43704. [Google Scholar] [CrossRef]
- Knapp, H.F.; Mesquida, P.; Stemmer, A. Imaging the surface potential of active purple membrane. Surf. Interface Anal. 2002, 33, 108–112. [Google Scholar] [CrossRef]
- Briones, X.G.; Urzúa, M.D.; Ríos, H.E.; Espinoza-Beltrán, F.J.; Dabirian, R.; Yazdani-Pedram, M. Thin films of amphiphilic polyelectrolytes. Soft materials characterized by Kelvin probe force microscopy. Polymer 2013, 54, 5733–5740. [Google Scholar] [CrossRef]
- Song, Y.; Yan, L.; Zhou, Y.; Song, B.; Li, Y. Lowering the Work Function of ITO by Covalent Surface Grafting of Aziridine: Application in Inverted Polymer Solar Cells. Adv. Mater. Interfaces 2015, 2, 1–6. [Google Scholar] [CrossRef]
- Guo, S.; Akhremitchev, B.B. Packing density and structural heterogeneity of insulin amyloid fibrils measured by AFM nanoindentation. Biomacromolecules 2006, 7, 1630–1636. [Google Scholar] [CrossRef] [PubMed]
- Choi, B.; Kim, T.; Lee, S.W.; Eom, K. Nanomechanical characterization of amyloid fibrils using single-molecule experiments and computational simulations. J. Nanomater. 2016, 2016. [Google Scholar] [CrossRef]
- Canale, C.; Oropesa-Nuñez, R.; Diaspro, A.; Dante, S. Amyloid and membrane complexity: The toxic interplay revealed by AFM. Semin. Cell Dev. Biol. 2018, 73, 82–94. [Google Scholar] [CrossRef] [PubMed]
- Drolle, E.; Gaikwad, R.M.; Leonenko, Z. Nanoscale electrostatic domains in cholesterol-laden lipid membranes create a target for amyloid binding. Biophys. J. 2012, 103, 27–29. [Google Scholar] [CrossRef] [PubMed]
- Ma, Q.; Wei, G.; Yang, X. Influence of Au nanoparticles on the aggregation of amyloid-β-(25–35) peptides. Nanoscale 2013, 5, 10397. [Google Scholar] [CrossRef] [PubMed]
- Sinensky, A.K.; Belcher, A.M. Label-free and high-resolution protein/DNA nanoarray analysis using Kelvin probe force microscopy. Nat. Nanotechnol. 2007, 2, 653–659. [Google Scholar] [CrossRef] [PubMed]
- Tsai, C.-C.; Hung, H.-H.; Liu, C.-P.; Chen, Y.-T.; Pan, C.-Y. Changes in plasma membrane surface potential of PC12 cells as measured by Kelvin probe force microscopy. PLoS ONE 2012, 7, e33849. [Google Scholar] [CrossRef] [PubMed]
- Birkenhauer, E.; Neethirajan, S. Surface Potential Measurement of Bacteria Using Kelvin Probe Force Microscopy. J. Vis. Exp. 2014. [Google Scholar] [CrossRef] [PubMed]
- Lee, I.; Chung, E.; Kweon, H.; Yiacoumi, S.; Tsouris, C. Scanning surface potential microscopy of spore adhesion on surfaces. Colloids Surf. B Biointerfaces 2012, 92, 271–276. [Google Scholar] [CrossRef] [PubMed]
- Howork, S. High-Resolution Imaging of the Electric Surface Potential of Biomolecular Structures. Available online: http://gtr.ukri.org/projects?ref=BB/E00959X/1 (accessed on 3 June 2018).
- Salerno, M. Coating of tips for electrochemical scanning tunneling microscopy by means of silicon, magnesium, and tungsten oxides. Rev. Sci. Instrum. 2010, 81, 93703. [Google Scholar] [CrossRef] [PubMed]
- Menozzi, C.; Gazzadi, G.C.; Alessandrini, A.; Facci, P. Focused ion beam-nanomachined probes for improved electric force microscopy. Ultramicroscopy 2005, 104, 220–225. [Google Scholar] [CrossRef] [PubMed]
- A Basic Guide to Particle Characterization. Available online: https://www.malvernpanalytical.com/en/learn/knowledge-center/whitepapers/WP120620BasicGuidePartChar (accessed on 3 June 2018).
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Salerno, M.; Dante, S. Scanning Kelvin Probe Microscopy: Challenges and Perspectives towards Increased Application on Biomaterials and Biological Samples. Materials 2018, 11, 951. https://doi.org/10.3390/ma11060951
Salerno M, Dante S. Scanning Kelvin Probe Microscopy: Challenges and Perspectives towards Increased Application on Biomaterials and Biological Samples. Materials. 2018; 11(6):951. https://doi.org/10.3390/ma11060951
Chicago/Turabian StyleSalerno, Marco, and Silvia Dante. 2018. "Scanning Kelvin Probe Microscopy: Challenges and Perspectives towards Increased Application on Biomaterials and Biological Samples" Materials 11, no. 6: 951. https://doi.org/10.3390/ma11060951
APA StyleSalerno, M., & Dante, S. (2018). Scanning Kelvin Probe Microscopy: Challenges and Perspectives towards Increased Application on Biomaterials and Biological Samples. Materials, 11(6), 951. https://doi.org/10.3390/ma11060951





