Characterization of Magnesium Silicate Hydrate (MSH) Gel Formed by Reacting MgO and Silica Fume
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Preparation
2.2. Sample Characterization
3. Results
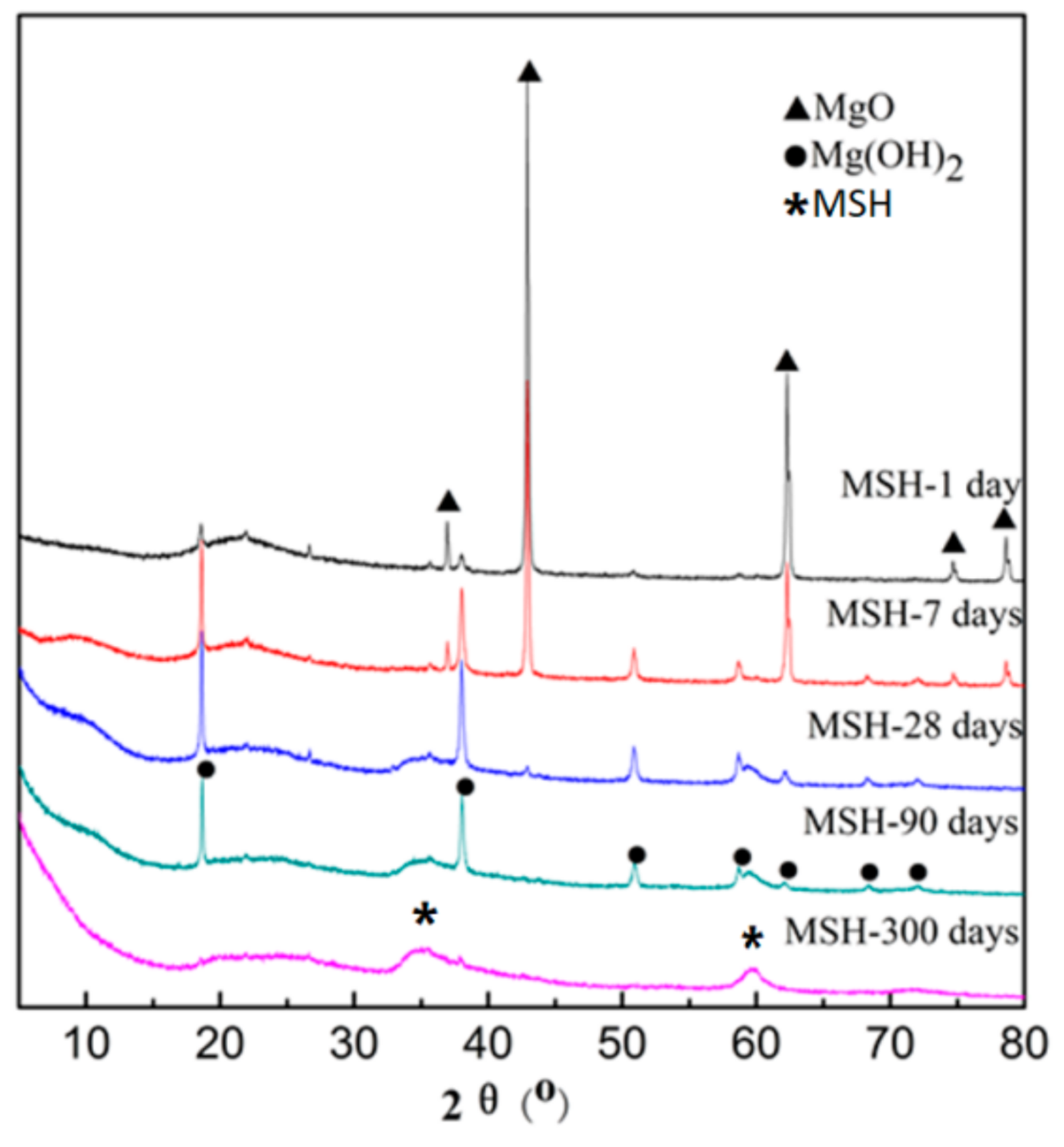
3.1. XRD Analysis
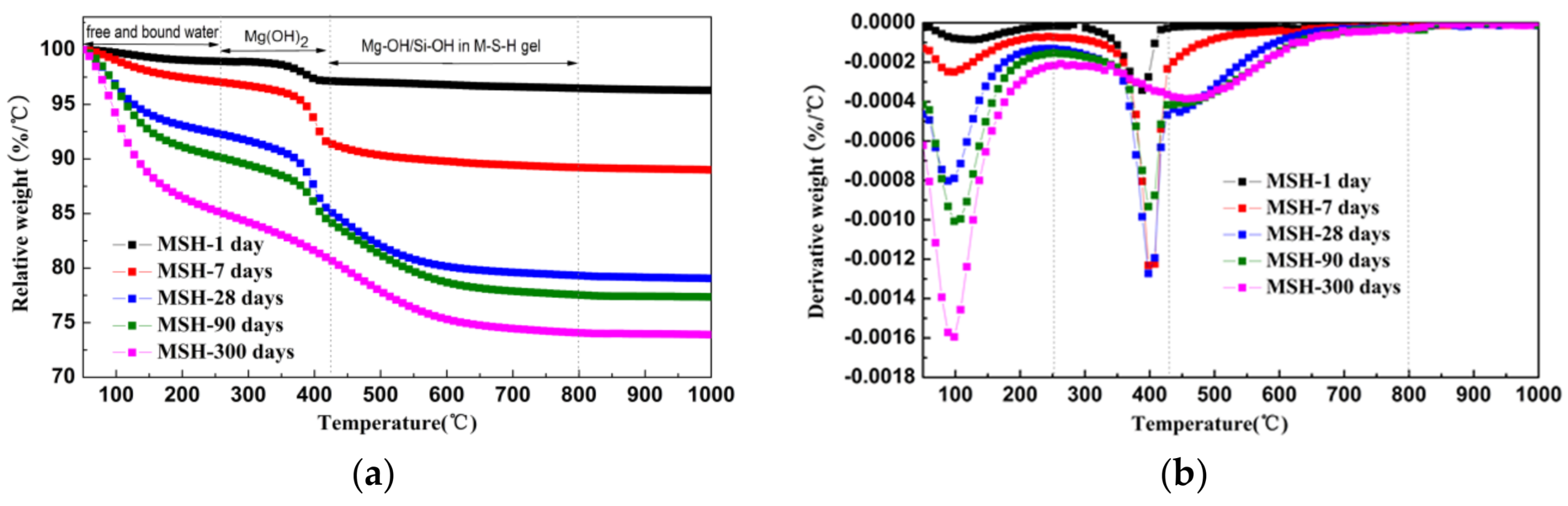
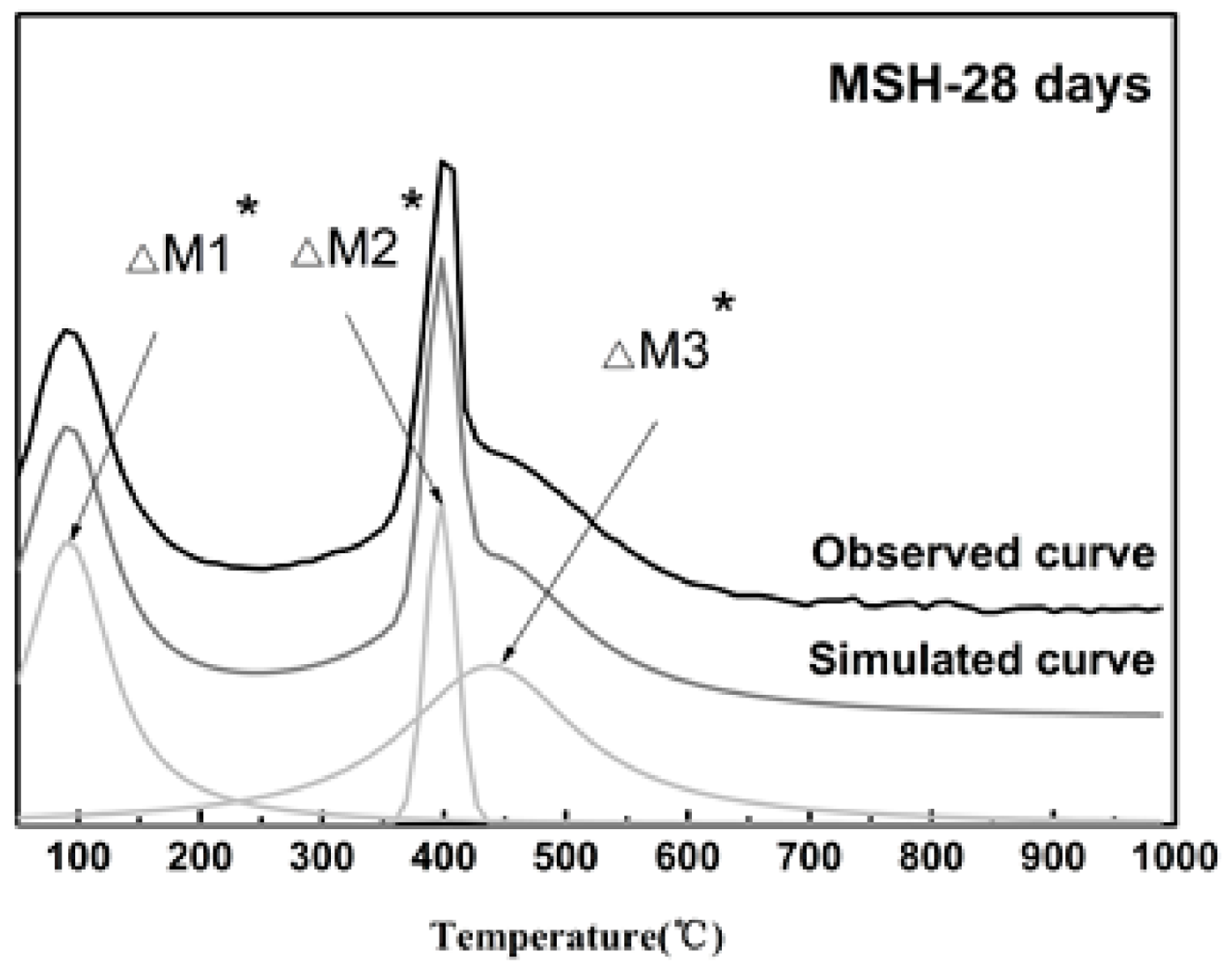
3.2. TG/DTG Analysis
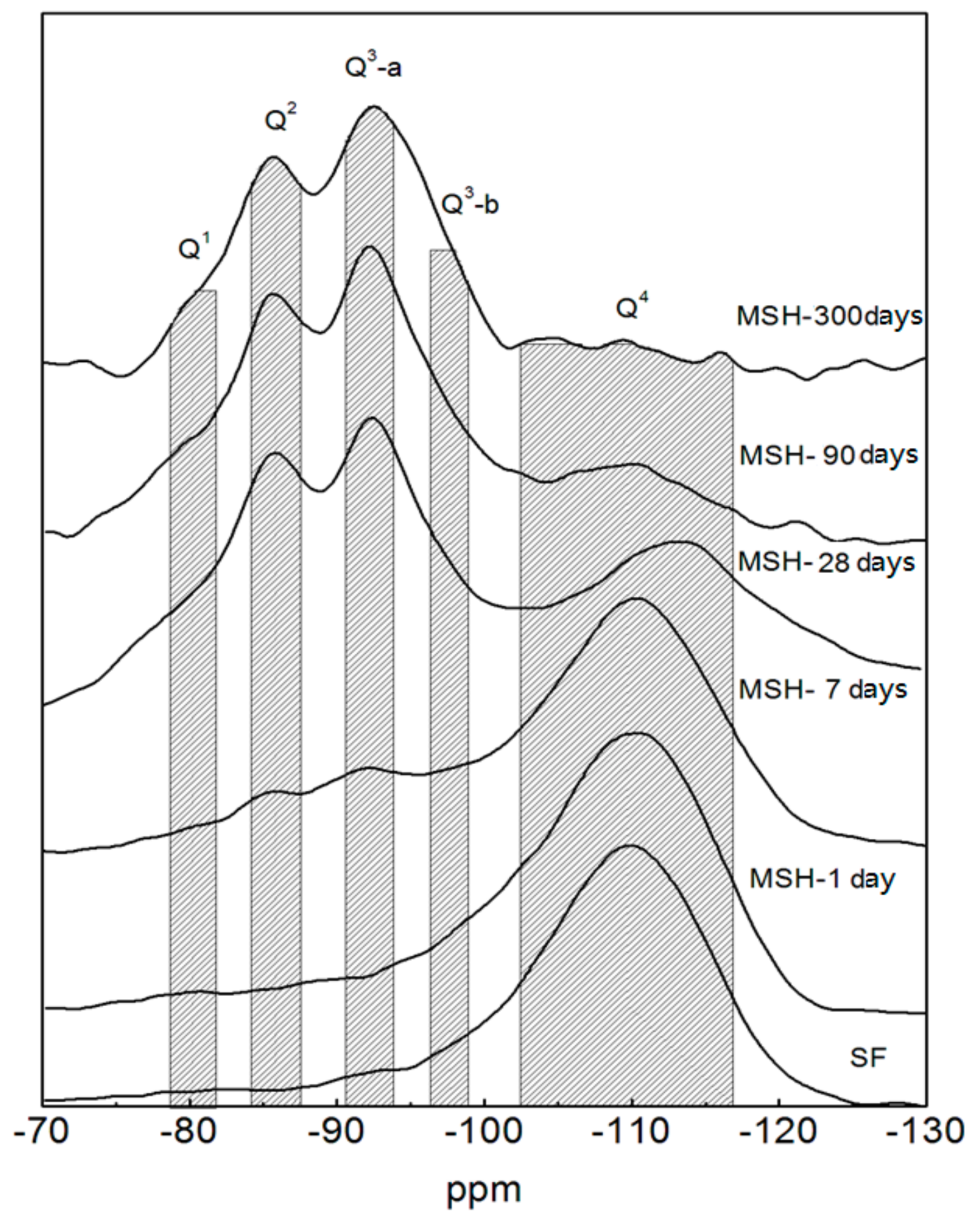
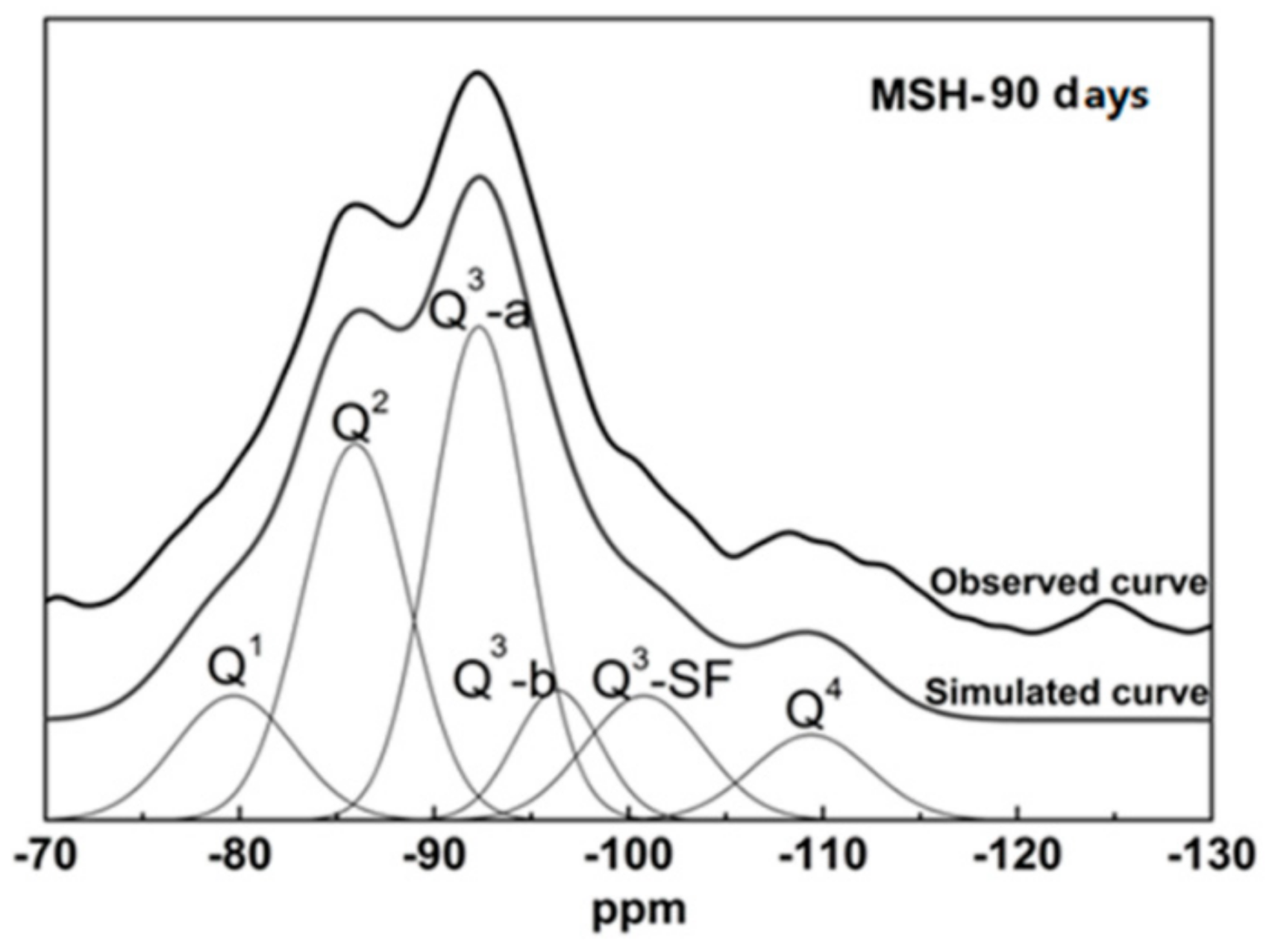
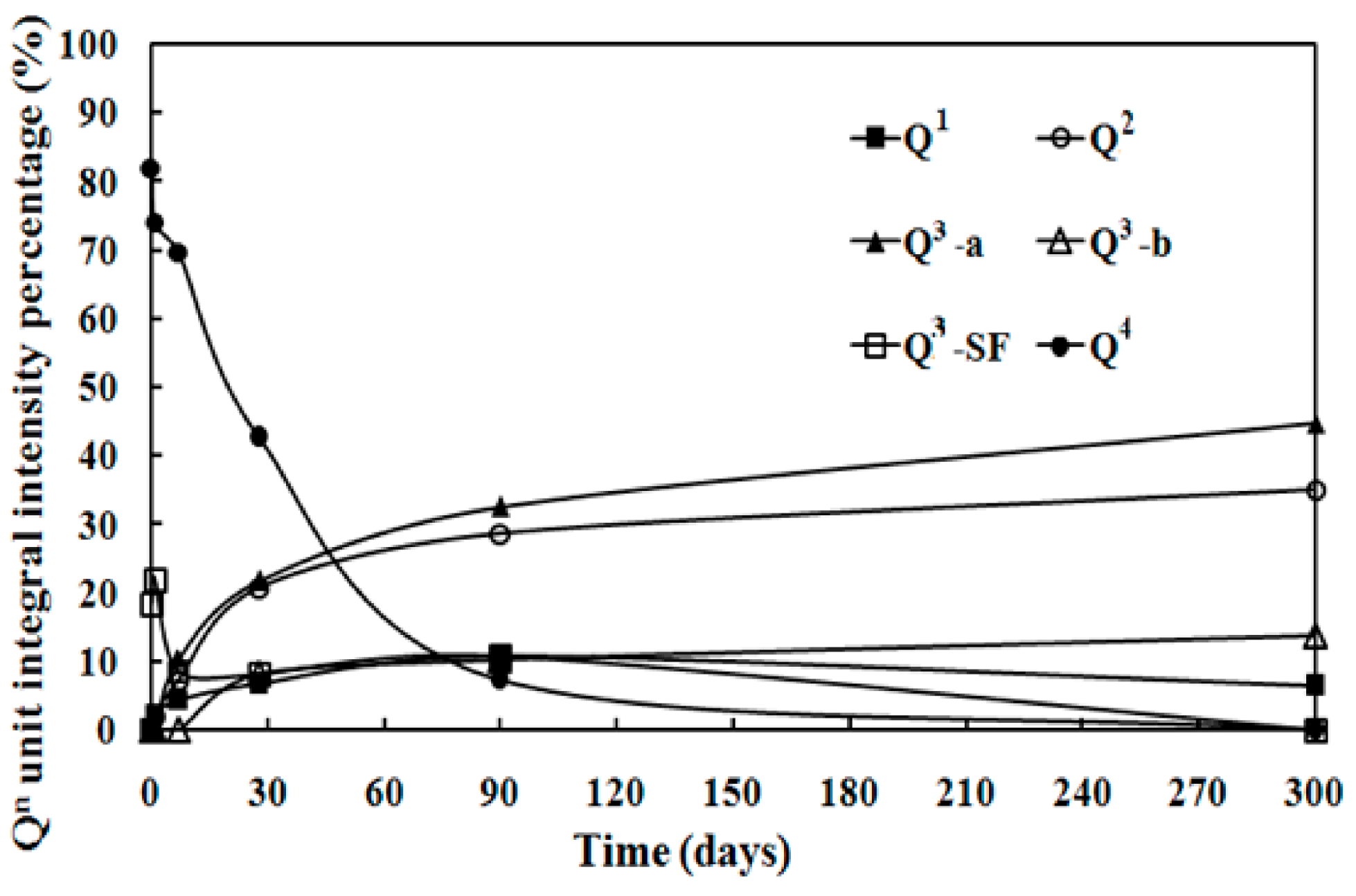
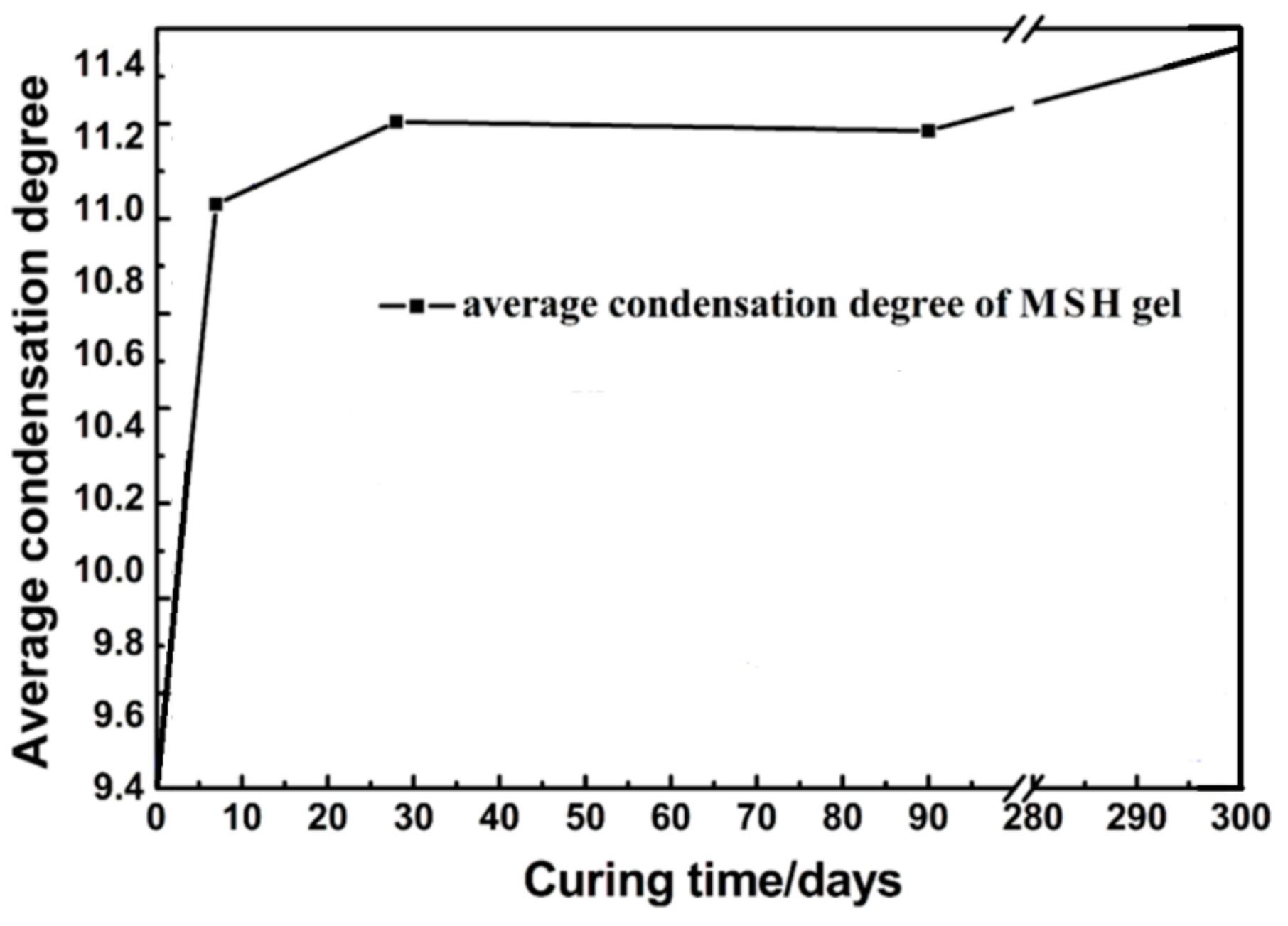
3.3. 29Si MAS NMR Spectra
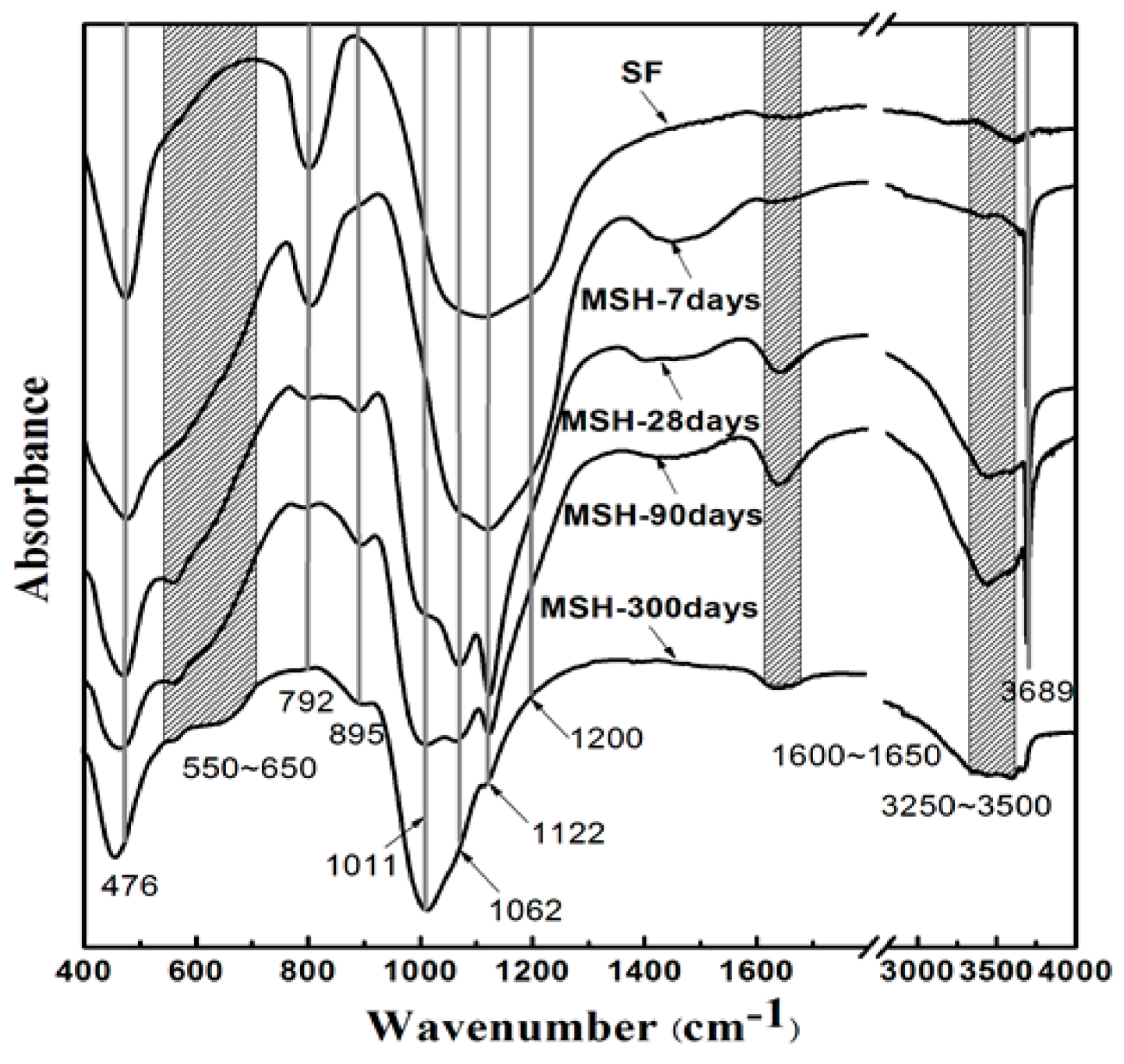
3.4. FT-IR Spectra
3.5. Transmission Electron Microscopy (TEM)
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gaucher, E.C.; Blanc, P. Cement/clay interactions—A review: Experiments, natural analogues and modeling. Waste Manag. 2006, 26, 776–788. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.T.; Vandeperre, L.J.; Cheeseman, C.R. Bottom-up design of a cement for nuclear waste encapsulation. In Ceramic Materials for Energy Applications; John Wiley & Sons, Inc.: New York, NY, USA, 2011; pp. 41–49. [Google Scholar] [CrossRef]
- Zhang, T.T.; Cheeseman, C.R.; Vandeperre, L.J. Characterization of corrosion of nuclear metal wastes encapsulated in magnesium silicate hydrate (MSH) cement. In Ceramic Materials for Energy Applications II; John Wiley & Sons, Inc.: New York, NY, USA, 2012; pp. 159–167. [Google Scholar] [CrossRef]
- Zhang, T.T.; Vandeperre, L.J.; Cheeseman, C.R. Magnesium silicate-hydrate cements for encapsulating problematic aluminium containing wastes. J. Sustain. Cem. Based Mater. 2012, 1, 34–45. [Google Scholar] [CrossRef]
- Berner, U.; Kulik, D.A.; Kosakowski, G. Geochemical impact of a low-pH cement liner on the near field of a repository for spent fuel and high-level radioactive waste. Phys. Chem. Earth 2013, 64, 46–56. [Google Scholar] [CrossRef]
- Walling, S.A.; Kinoshita, H.; Bernal, A.; Collier, N.C.; Provis, J.L. Structure and properties of binder gels formed in the system Mg(OH)2-SiO2-H2O for immobilization of Magnox sludge. Dalton Trans. 2015, 44, 8126–8137. [Google Scholar] [CrossRef] [PubMed]
- Walling, S.A. Conversion of Magnesium Bearing Radioactive Wastes into Cementitious Binders. Ph.D. Thesis, University of Sheffield, Sheffield, UK, 2016. [Google Scholar]
- Brew, D.R.M.; Glasser, F.P. Synthesis and characterization of magnesium silicate hydrate gels. Cem. Concr. Res. 2005, 35, 85–98. [Google Scholar] [CrossRef]
- Brew, D.R.M.; Glasser, F.P. The magnesia–silica gel phase in slag cements: Alkali (K, Cs) sorption potential of synthetic gels. Cem. Concr. Res. 2005, 35, 77–83. [Google Scholar] [CrossRef]
- Dauzeres, A.; Le Bescop, P.; Sardini, P.; Cau Dit Coumes, C. Physico-chemical investigation of clayey/cement-based materials interaction in the context of geological waste disposal: Experimental approach and results. Cem. Concr. Res. 2010, 40, 1327–1340. [Google Scholar] [CrossRef]
- Lothenbach, B.; Le, G.S.; Ben, M.H.; Figi, R.; Wieland, E. Hydration of a low-alkali CEM III/B-SiO2 cement (LAC). Cem. Concr. Res. 2012, 42, 410–423. [Google Scholar] [CrossRef]
- Pusch, R.; Zwahr, H.; Gerber, R.; Schomburg, J. Interaction of cement and smectitie clay theory and practice. Appl. Clay Sci. 2003, 23, 203–210. [Google Scholar] [CrossRef]
- Honty, M.; De, M.C.; Wang, L.; Madejová, J.; Czímerová, A.; Pentrák, M.; Stríček, I.; Van, M.G. The effect of high pH alkaline solutions on the mineral stability of the Boom Clay-Batch experiments at 60 °C. Appl. Geochem. 2010, 25, 825–840. [Google Scholar] [CrossRef]
- Savage, D.; Walker, C.; Arthur, R.; Rochelle, C.; Oda, C.; Takase, H. Alteration of bentonite by hyper-alkaline fluids: A review of the role of secondary minerals. Phys. Chem. Earth 2007, 32, 287–297. [Google Scholar] [CrossRef]
- Kosakowski, G.; Berner, U. The evolution of clay rock/cement interfaces in a cementitious repository for low-and intermediate level radioactive waste. Phys. Chem. Earth 2013, 64, 65–86. [Google Scholar] [CrossRef]
- Szczerba, J.; Prorok, R.; Śnieżek, E.; Madej, D.; Maślona, K. Influence of time and temperature on ageing and phases synthesis in the MgO-SiO2-H2O system. Thermochim. Acta 2013, 567, 57–64. [Google Scholar] [CrossRef]
- Chen, Y.; Wei, J. Hydrated Magnesium Silica System Gelling Material Coagulating and Hardening at Normal Temperature and Its Preparation Method. Chinese Patent CN1267374 C, 2 August 2006. [Google Scholar]
- Chen, Y.; Wei, J. Hydrated Magnesium Silica and Synthesis Method. Chinese Patent CN1315726 C, 16 May 2007. [Google Scholar]
- Lothenbach, B.; Nied, D.; L’Hôpital, E.; Achiedo, G.; Dauzères, A. Magnesium and calcium silicate hydrates. Cem. Concr. Res. 2015, 77, 60–68. [Google Scholar] [CrossRef]
- Chiang, W.S.; Ferraro, G.; Fratini, E.; Ridi, F.; Yeh, Y.Q.; Jeng, U.S.; Chen, S.H.; Baglioni, P. Multiscale structure of calcium-and magnesium-silicate-hydrate gels. J. Mater. Chem. A 2016, 2, 12991–12998. [Google Scholar] [CrossRef]
- Temuujin, J.; Okada, K.; MacKenzie, K.J.D. Formation of layered magnesium silicate during the aging of hydroxide−silica mixtures. J. Am. Ceram. Soc. 1998, 81, 754–756. [Google Scholar] [CrossRef]
- Temuujin, J.; Okada, K.; MacKenzie, K.J.D. Role of water in the mechanochemical reactions of MgO-SiO2 systems. J. Solid State Chem. 1998, 138, 169–177. [Google Scholar] [CrossRef]
- Barron, P.F.; Slade, P.; Frost, R.L. Solid-state silicon-29 spin-lattice relaxation in several 2:1 phyllosilicate minerals. J. Phys. Chem. 1985, 89, 3305–3310. [Google Scholar] [CrossRef]
- Hayashi, H.; Otsuka, H.R.; Imai, N. Infrared study of sepiolite and palygorskite on heating. Am. Mineral. 1969, 54, 1613–1624. [Google Scholar] [CrossRef]
- Shuali, U.; Bram, L.; Steinberg, M. Infrared study of the thermal-treatment of sepiolite and palygorskite saturated with organic amines. Thermochim. Acta 1989, 148, 445–456. [Google Scholar] [CrossRef]
- MacKenzie, K.J.D.; Meinhold, R.H. The thermal reaction of talc studied by 29Si and 25Mg MAS NMR. Thermochim. Acta 1994, 244, 195–203. [Google Scholar] [CrossRef]
- Mitsuda, T.; Taguchi, H. Formation of magnesium silicate hydrate and its crystallization to talc. Cem. Concr. Res. 1977, 7, 223–230. [Google Scholar] [CrossRef]
- Tosca, N.J.; Macdonald, F.A.; Strauss, J.V.; Johnston, D.T.; Knoll, A.H. Sedimentary talc in Neoproterozoic carbonate successions. Earth Planet. Sci. Lett. 2011, 306, 11–22. [Google Scholar] [CrossRef]
- Tonelli, M.; Martini, F.; Calucci, L.; Fratini, E.; Geppi, M.; Ridi, F.; Borsacchi, S.; Baglioni, P. Structural characterization of magnesium silicate hydrate: Towards the design of eco-sustainable cements. Dalton Trans. 2016, 45, 3294–3304. [Google Scholar] [CrossRef] [PubMed]
- Mackenzie, K.J.D.; Meinhold, R.H. Thermal-reactions of chrysotile revisited-a Si-29 and Mg-25 MAS NMR-study. Am. Mineral. 1994, 79, 43–50. [Google Scholar]
- Liao, J.; Senna, M. Thermal behavior of mechanically amorphized talc. Thermochim. Acta 1992, 197, 295–306. [Google Scholar] [CrossRef]
- Suquet, H. Effects of dry grinding and leaching on the crystal structure of chrysotile. Clays Clay Miner. 1989, 37, 439–445. [Google Scholar] [CrossRef]
- Nied, D.; Enemark-Rasmussen, K.; L’Hopital, E.; Skibsted, J.; Lothenbach, B. Properties of magnesium silicate hydrates (MSH). Cem. Concr. Res. 2016, 79, 323–332. [Google Scholar] [CrossRef]
- Roosza, C.; Grangeonb, S.; Blancb, P.; Montouilloutc, V.; Lothenbachd, B.; Henocqa, P.; Giffauta, E.; Vieillarde, P.; Gaboreau, S. Crystal structure of magnesium silicate hydrates (MSH): The relation with 2:1 Mg–Si phyllosilicates. Cem. Concr. Res. 2015, 73, 228–237. [Google Scholar] [CrossRef]
- Vidmer, A.; Sclauzero, G.; Pasquarello, A. Infrared spectra of jennite and tobermorite from first-principles. Cem. Concr. Res. 2014, 60, 11–23. [Google Scholar] [CrossRef]
- Sáez del Bosquea, I.F.; Martínez-Ramíreza, S.; Blanco-Varela, S. FTIR study of the effect of temperature and nanosilica on the nanostructure of C–S–H gel formed by hydrating tricalcium silicate. Constr. Build. Mater. 2014, 52, 314–323. [Google Scholar] [CrossRef]
- Andersen, M.D.; Jakobsen, H.J.; Skibsted, J. Characterization of white Portland cement hydration and the C–S–H structure in the presence of sodium aluminate by 27Al and 29Si MAS NMR spectroscopy. Cem. Concr. Res. 2004, 34, 857–868. [Google Scholar] [CrossRef]
- Rulmont, A.; Cahay, R.; Liegeoisduyckaerts, M.; Tarte, P. Vibrational spectroscopy of phosphates-general correlations between structure and spectra. Eur. J. Inorg. Chem. 1991, 28, 207–219. [Google Scholar]
- Gunde, M.K. Vibrational modes in amorphous silicon dioxide. Physica B 2000, 292, 286–295. [Google Scholar] [CrossRef]
- Hartman, J.S.; Millard, R.L. Gel synthesis of magnesium silicates: A 29Si magic angle spinning NMR study. Phys. Chem. Miner. 1990, 17, 1–8. [Google Scholar] [CrossRef]
- Li, Z.H.; Zhang, T.S.; Hu, T.S.J.; Tang, Y.; Niu, Y.F.; Wei, J.X.; Yu, Q.J. Characterization of reaction products and reaction process of MgO-SiO2-H2O system at room temperature. Constr. Build. Mater. 2014, 61, 252–259. [Google Scholar] [CrossRef]
- Li, J.X.; Yu, Q.J.; Wei, J.X.; Zhang, T.S. Structural characteristics and hydration kinetics of modified steel slag. Cem. Concr. Res. 2011, 3, 324–329. [Google Scholar] [CrossRef]
- Barron, P.F.; Frost, R.L. Solid state 29Si NMR examination of the 2:1 ribbon magnesium silicates, sepiolite and palygorskite. Am. Mineral. 1985, 70, 758–766. [Google Scholar]
- Maruyama, I.; Nishioka, Y.; Lgarashi, G.; Matsui, K. Microstructural and bulk property changes in hardened cement paste during the first drying process. Cem. Concr. Res. 2014, 58, 20–34. [Google Scholar] [CrossRef]
- Jia, Y.; Wang, B.M.; Zhang, T.T. A Comparative Study of Different Amorphous and Paracrystalline Silica by NMR and SEM/EDS. J. Wuhan Univ. Technol.-Mater. Sci. Ed. 2015, 30, 900–907. [Google Scholar] [CrossRef]
- Salomao, R.; Bittencourt, L.R.M.; Pandolfelli, V.C. A novel approach for magnesia hydration assessment in refractory castables. Ceram. Int. 2007, 33, 803–810. [Google Scholar] [CrossRef]
- Vella, E.; Buscarino, G.; Vaccaro, G.; Boscaino, R. Structural organization of silanol and silicon hydride groups in the amorphous silicon dioxide network. Eur. Phys. J. B 2011, 83, 47–52. [Google Scholar] [CrossRef]
- Jia, Y.; Wang, B.M.; Wu, Z.L.; Han, J.N.; Zhang, T.T.; Vandeperre, L.J.; Cheeseman, C.R. Role of sodium hexametaphosphate in MgO/SiO2 cement pastes. Cem. Concr. Res 2016, 89, 63–71. [Google Scholar] [CrossRef]
- Jin, F.; Al-Tabbaa, A. Characterization of different commercial reactive magnesia. ICE Adv. Cem. Res. 2014, 26, 101–113. [Google Scholar] [CrossRef]

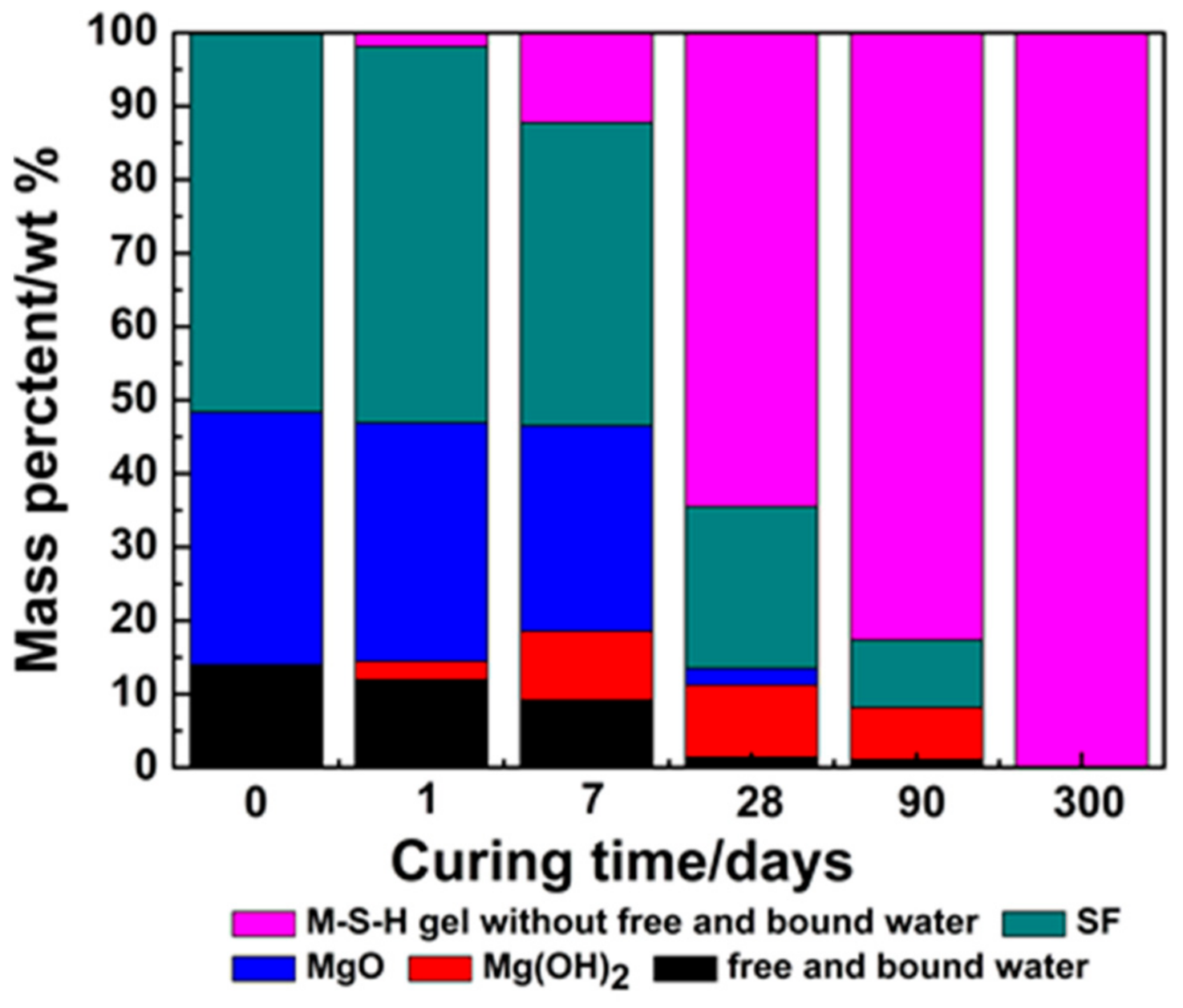
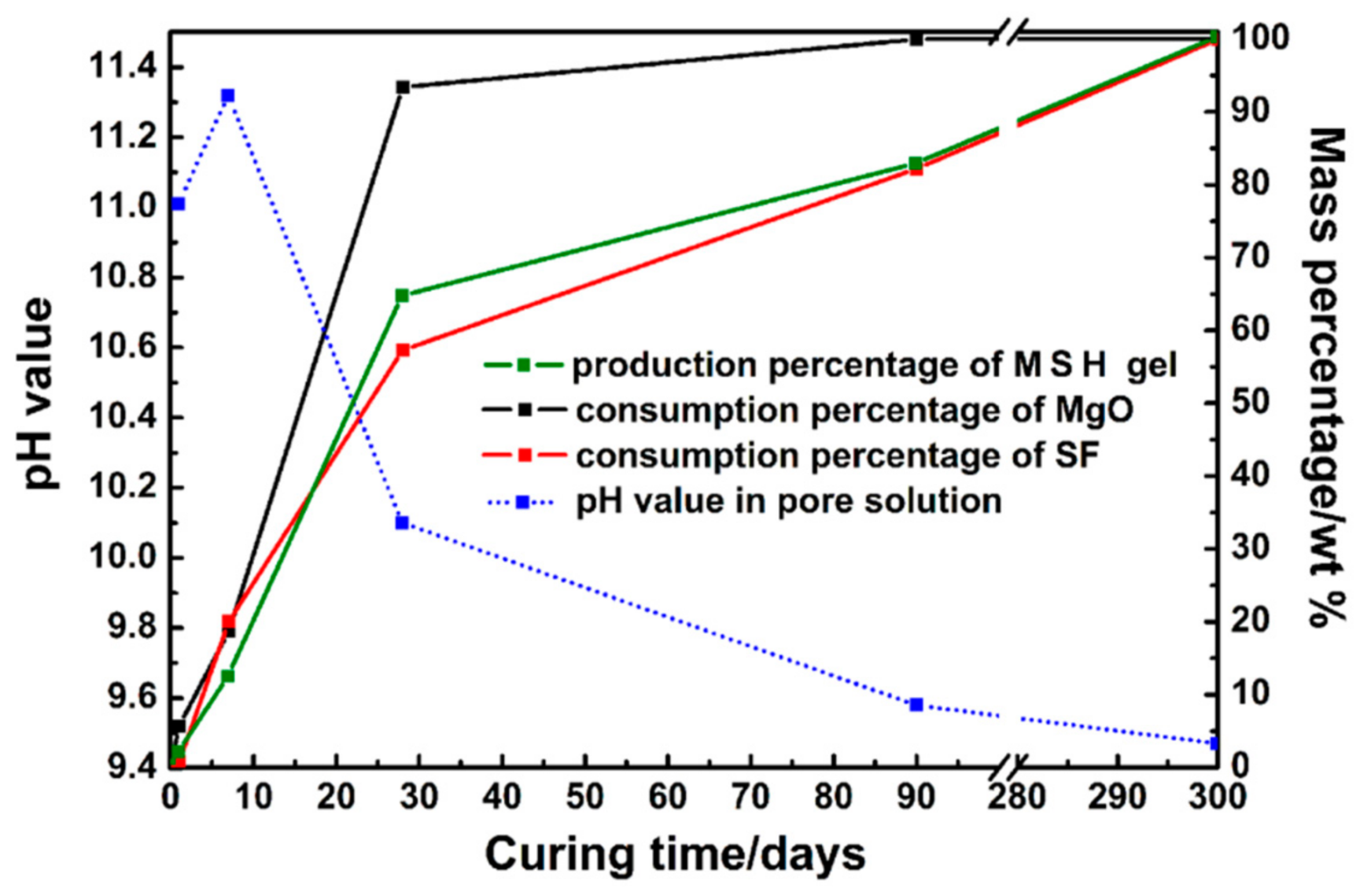
| Oxide (wt %) | MgO | SF |
|---|---|---|
| MgO | 97.2 | 1.18 |
| CaO | 0.80 | 0.68 |
| SiO2 | 0.35 | 93.71 |
| Fe2O3 | 0.15 | 0.24 |
| Al2O3 | 0.10 | 0.23 |
| Na2O | - | 0.35 |
| K2O | - | 1.74 |
| SO3 | 0.02 | 0.37 |
| P2O5 | - | 0.38 |
| Cl | 0.35 | - |
| Loss in ignition | 1.03 | 1.12 |
| Bulk Density, loose (g/cm3) | 0.35 | 0.28 |
| Mean Particle Size (μm) | 6.7 | 0.3 |
| BET surface area (m2/g) | 26 | 18 |
| Curing time (day) | 0 | 1 | 7 | 28 | 90 | 300 |
| Consumption percentage of MgO (%) | 0 | 7 | 18 | 92 | 100 | 100 |
| Sample ID | De-Convoluted Data (wt %) | ||
|---|---|---|---|
| ∆M1* | ∆M2* | ∆M3* | |
| MSH-1 day | 2 ± 1 | 1 ± 1 | 2 ± 1 |
| MSH-7 days | 2 ± 1 | 4 ± 1 | 4 ± 1 |
| MSH-28 days | 8 ± 2 | 3 ± 1 | 11 ± 2 |
| MSH-90 days | 10 ± 2 | 2 ± 1 | 11 ± 2 |
| MSH-300 days | 15 ± 2 | - | 11 ± 2 |
| Sample ID | Q1 | Q2 | Q3 | Q4 | ||||
|---|---|---|---|---|---|---|---|---|
| Center (ppm) | Area (%) | Center (ppm) | Area (%) | Center (ppm) | Area (%) | Center (ppm) | Area (%) | |
| SF | - | - | - | - | 101.0 (sh/Q3-SF) | 18 ± 3 | −110.2 (p) | 82 ± 6 |
| MSH-1 day | −80.6 (p) | 2 ± 1 | −88.9 (p) | 2 ± 1 | −99.1 (sh/Q3-SF) | 22 ± 3 | −110.3 (p) | 74 ± 5 |
| MSH-7 days | −80.7 (p) | 4 ± 1 | −85.5 (p) | 7 ± 1 | −92.1 (p/Q3-a) | 10 ± 1 | −110.3 (p) | 70 ± 5 |
| −98.9 (sh/Q3-SF) | 8.5 ± 1 | |||||||
| MSH-28 days | −79.7 (p) | 6.7 ± 1 | −85.9 (p) | 21 ± 2 | −92.6 (p/Q3-a) | 21.7 ± 3 | −112.9 (p) | 43 ± 3 |
| −97.2 (sh/Q3-b) | 8.1 ± 1 | |||||||
| MSH-90 days | −79.7 (p) | 11 ± 1 | −85.9 (p) | 29 ± 3 | −92.3 (p/Q3-a) | 33 ± 3 | −109.4 (p) | 7 ± 1 |
| −96.3 (sh/Q3-b) | 10 ± 1 | |||||||
| −100.8 (sh/Q3-SF) | 11 ± 1 | |||||||
| MSH-300 days | −80.5 (p) | 6 ± 1 | −85.7 (p) | 35 ± 3 | −92.6 (p/Q3-a) | 45 ± 3 | - | - |
| −97.2 (p/Q3-b) | 14 ± 1 | |||||||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, T.; Zou, J.; Wang, B.; Wu, Z.; Jia, Y.; Cheeseman, C.R. Characterization of Magnesium Silicate Hydrate (MSH) Gel Formed by Reacting MgO and Silica Fume. Materials 2018, 11, 909. https://doi.org/10.3390/ma11060909
Zhang T, Zou J, Wang B, Wu Z, Jia Y, Cheeseman CR. Characterization of Magnesium Silicate Hydrate (MSH) Gel Formed by Reacting MgO and Silica Fume. Materials. 2018; 11(6):909. https://doi.org/10.3390/ma11060909
Chicago/Turabian StyleZhang, Tingting, Jing Zou, Baomin Wang, Zhenlin Wu, Yuan Jia, and Christopher R. Cheeseman. 2018. "Characterization of Magnesium Silicate Hydrate (MSH) Gel Formed by Reacting MgO and Silica Fume" Materials 11, no. 6: 909. https://doi.org/10.3390/ma11060909
APA StyleZhang, T., Zou, J., Wang, B., Wu, Z., Jia, Y., & Cheeseman, C. R. (2018). Characterization of Magnesium Silicate Hydrate (MSH) Gel Formed by Reacting MgO and Silica Fume. Materials, 11(6), 909. https://doi.org/10.3390/ma11060909







