Sustainable Blended Cements—Influences of Packing Density on Cement Paste Chemical Efficiency
Abstract
1. Introduction
2. Materials and Methods
2.1. Powders
2.2. Fresh Cement Pastes
- (1)
- The heat of hydration was measured by isothermal calorimetry—TAM Air by TA instruments (New Castle, DE, USA) to study the heat flow from the moment the blended cement was mixed with water. All the samples were mixed with a constant water-to-cement ratio (0.4).
- (2)
- The workability of the fresh cement paste was determined based on normal consistency. Each cement powder—blended or original—was mixed with the amount of water needed to obtain a normal consistency according to EN-196-3 [24]. These pastes were then used to study the properties at the harden stage, as described in the following section.
2.3. Hardened Cement Pastes
- (1)
- The amount of the non-evaporable water in the hydrated pastes was determined in order to evaluate the degree of hydration (also at late ages, from 2 h to 28 days). For cement pastes, the degree of hydration may be determined as follows: the hydration of 1 g of anhydrous cement produces 0.23 g of non-evaporable water. The LOI (loss-on-ignition) non-evaporable water content is determined by the relative mass loss between 105 °C and 1000 °C, corrected for the LOI caused by decarbonation [25,26].
- (2)
- Quantitative phase analysis of X-ray diffractions (XRDs), using the Rietveld method, was applied to determine the mineral content of the cement pastes, including both the original cements and the cements mixed with limestone at the age of 28 days after casting. XRD analysis was performed using XRD—EMPYREAN X-Ray Diffractometer (CuKα radiation, 45 kV, 40 mA, PANalytical, Almelo, The Netherlands) in a scanning range of 7° to 53° in 2θ at an internal of 0.020°.
- (3)
- Thermogravimetry and differential thermogravimetry (TG/DTG) was used for the determination of the weight loss due to the dehydration of calcium hydroxide and the decarbonation of calcium carbonate in the hardened sample from 2 h to 28 days after casting, using a TGA—TA Instruments Q500 (New Castle, DE, USA). The samples were heated in the 20 °C to 1000 °C range at a constant rate of 10 °C/min in an atmosphere of N2.
3. Results and Discussion
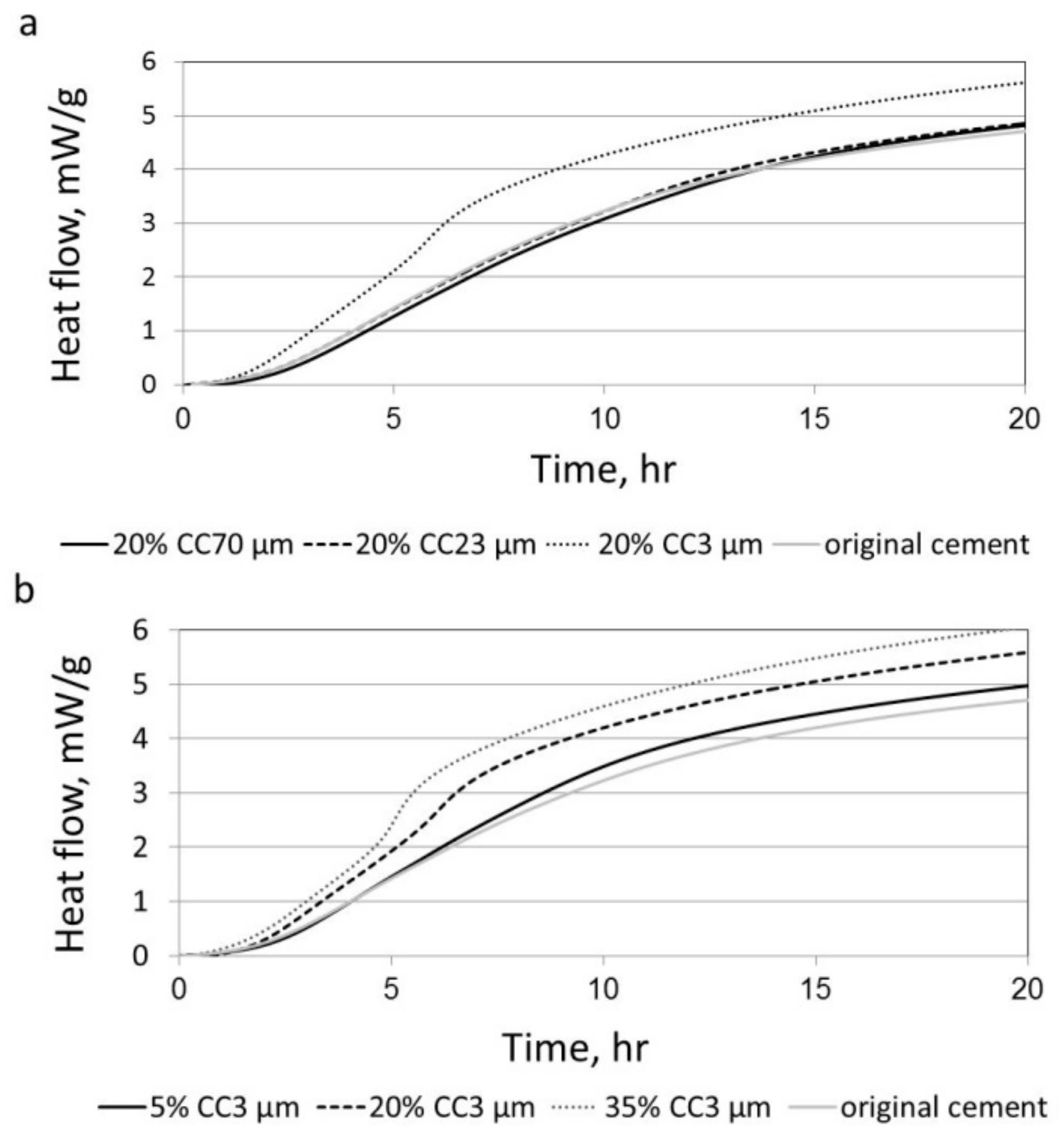
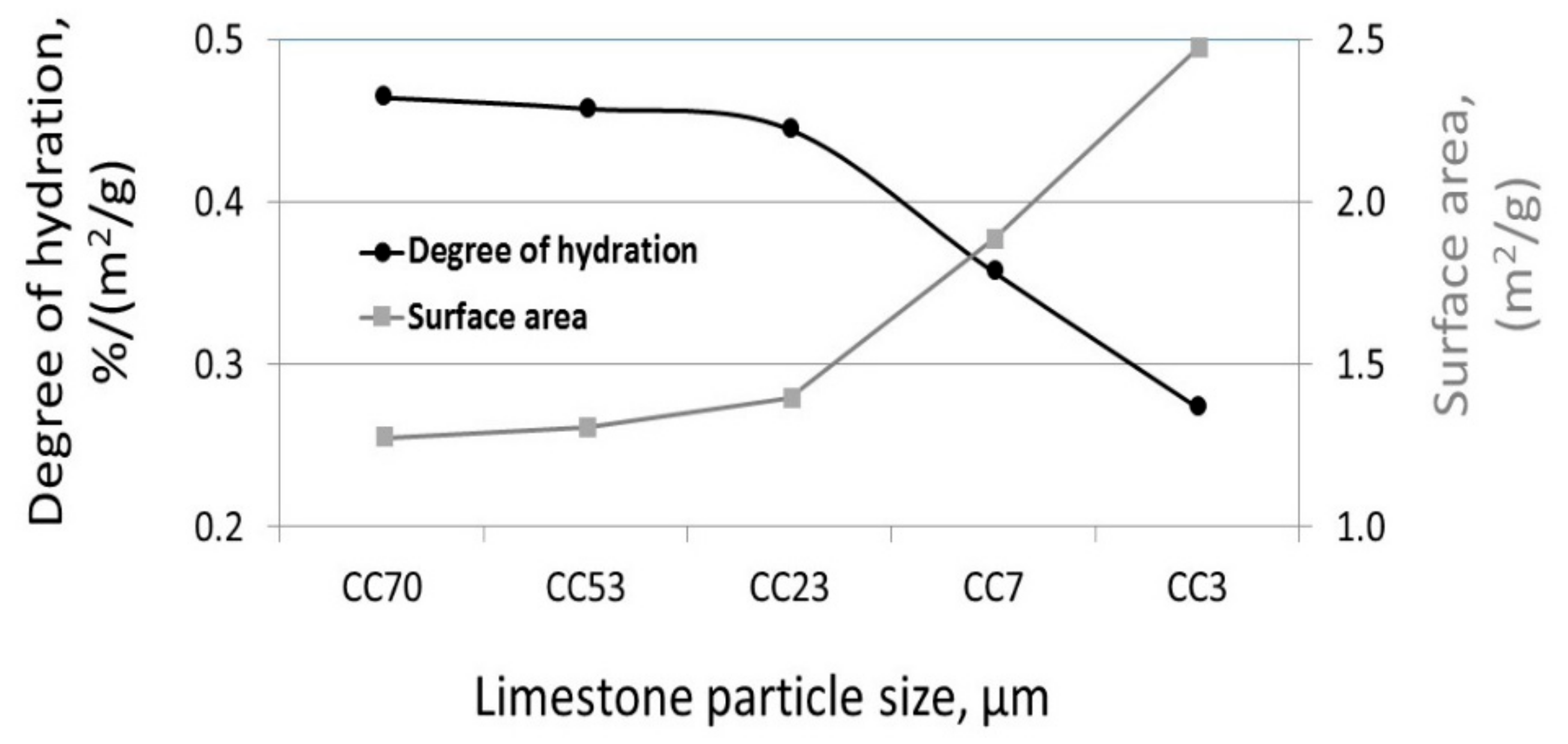
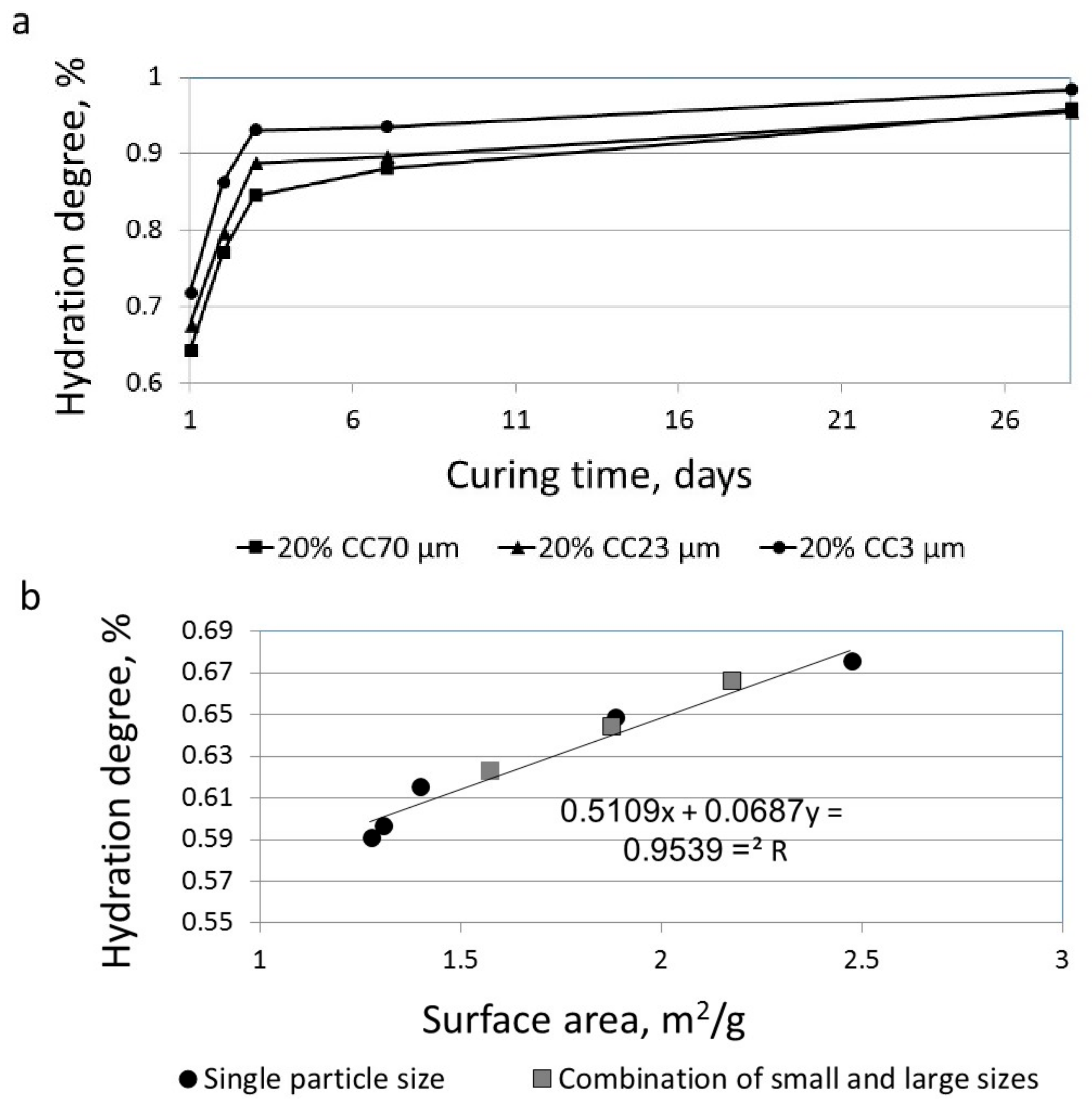
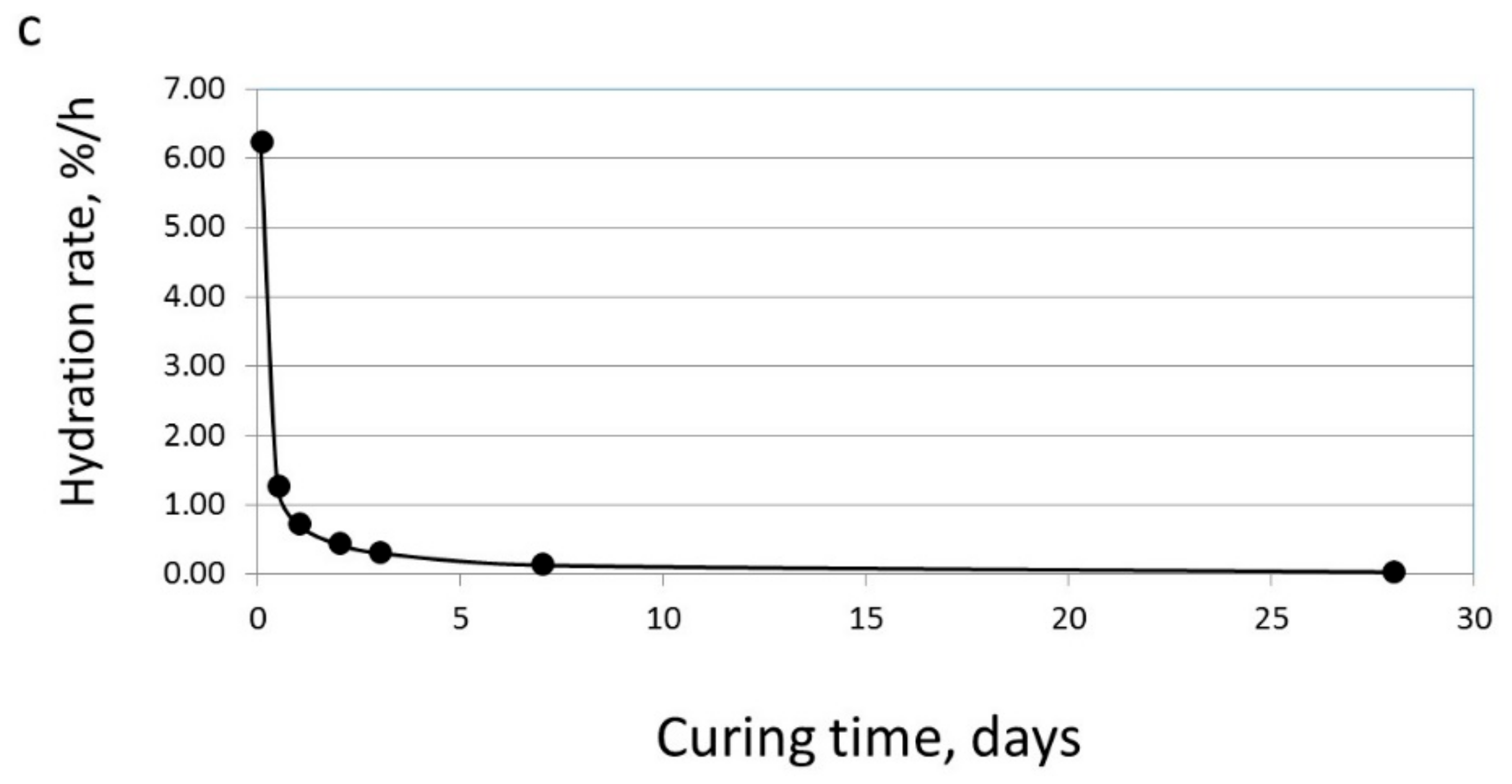
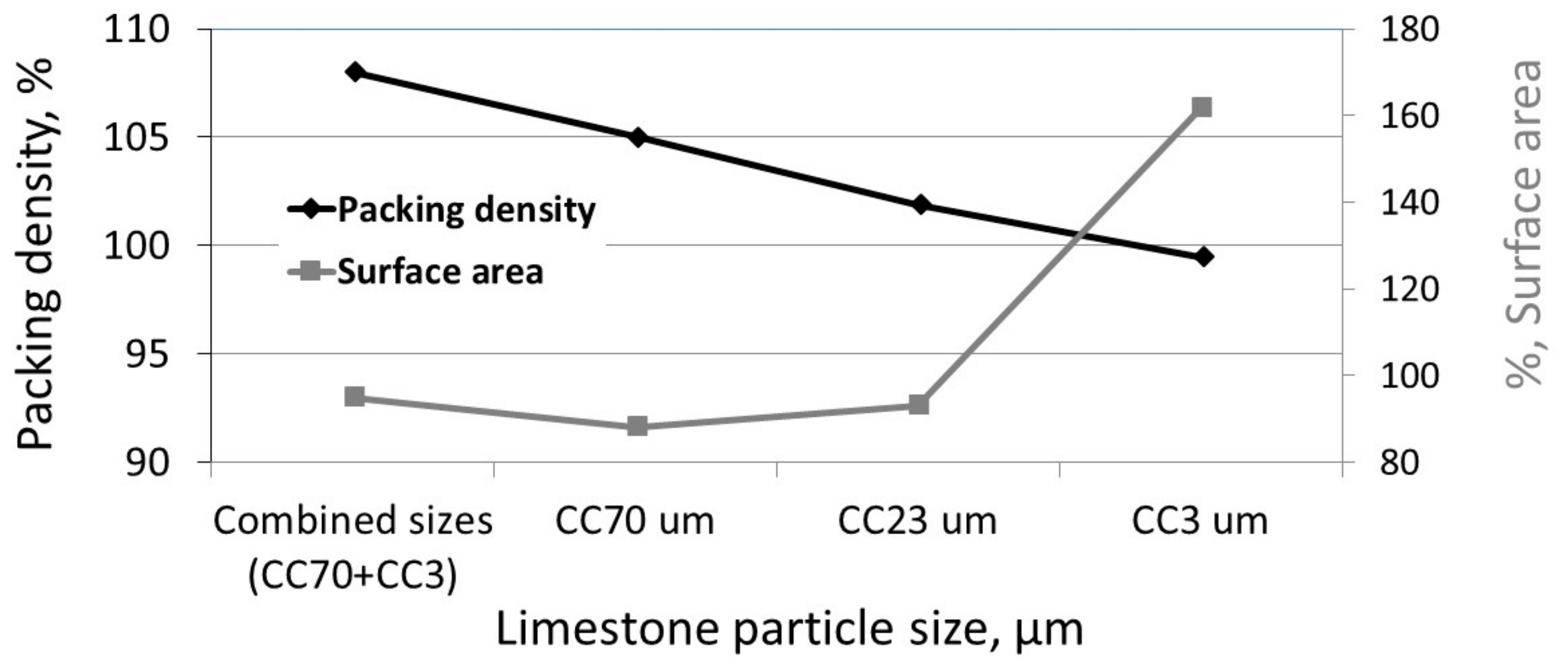
3.1. The Effect of Particle Size on Hydration Reaction
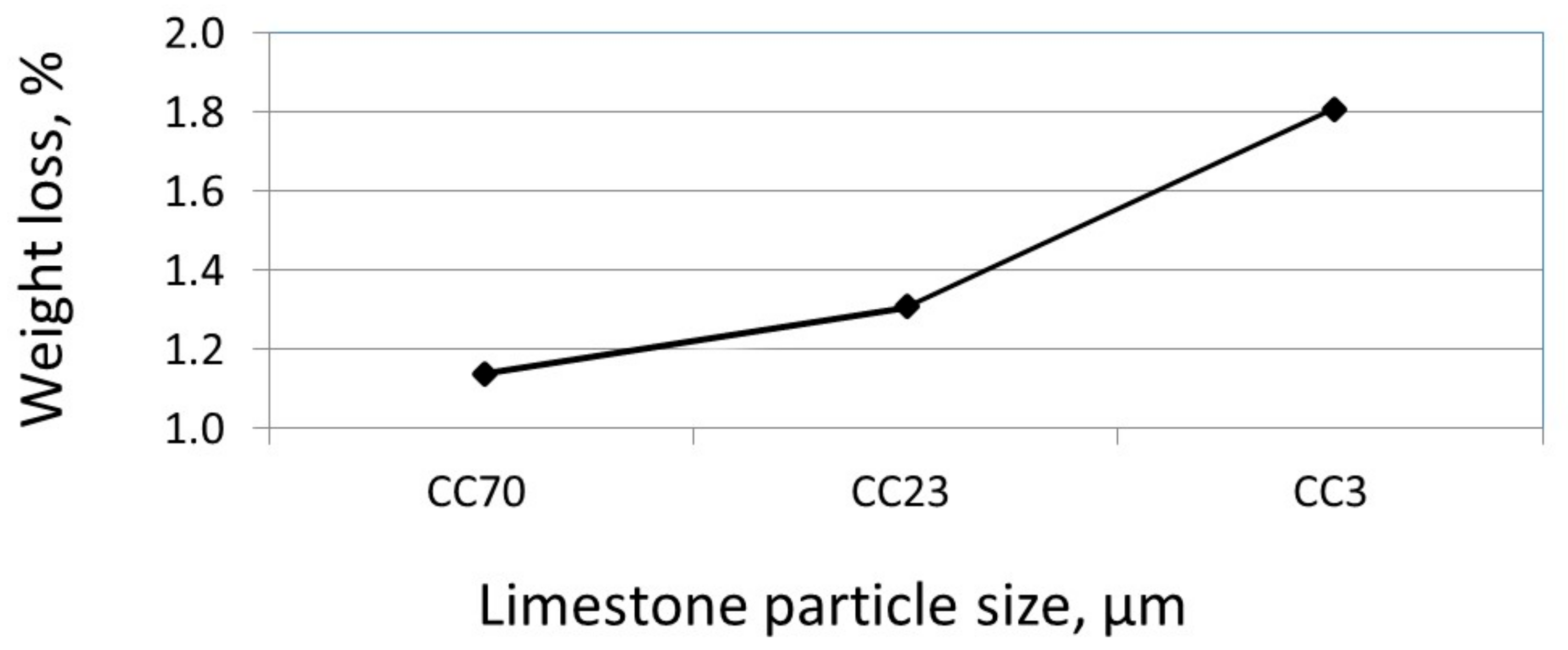
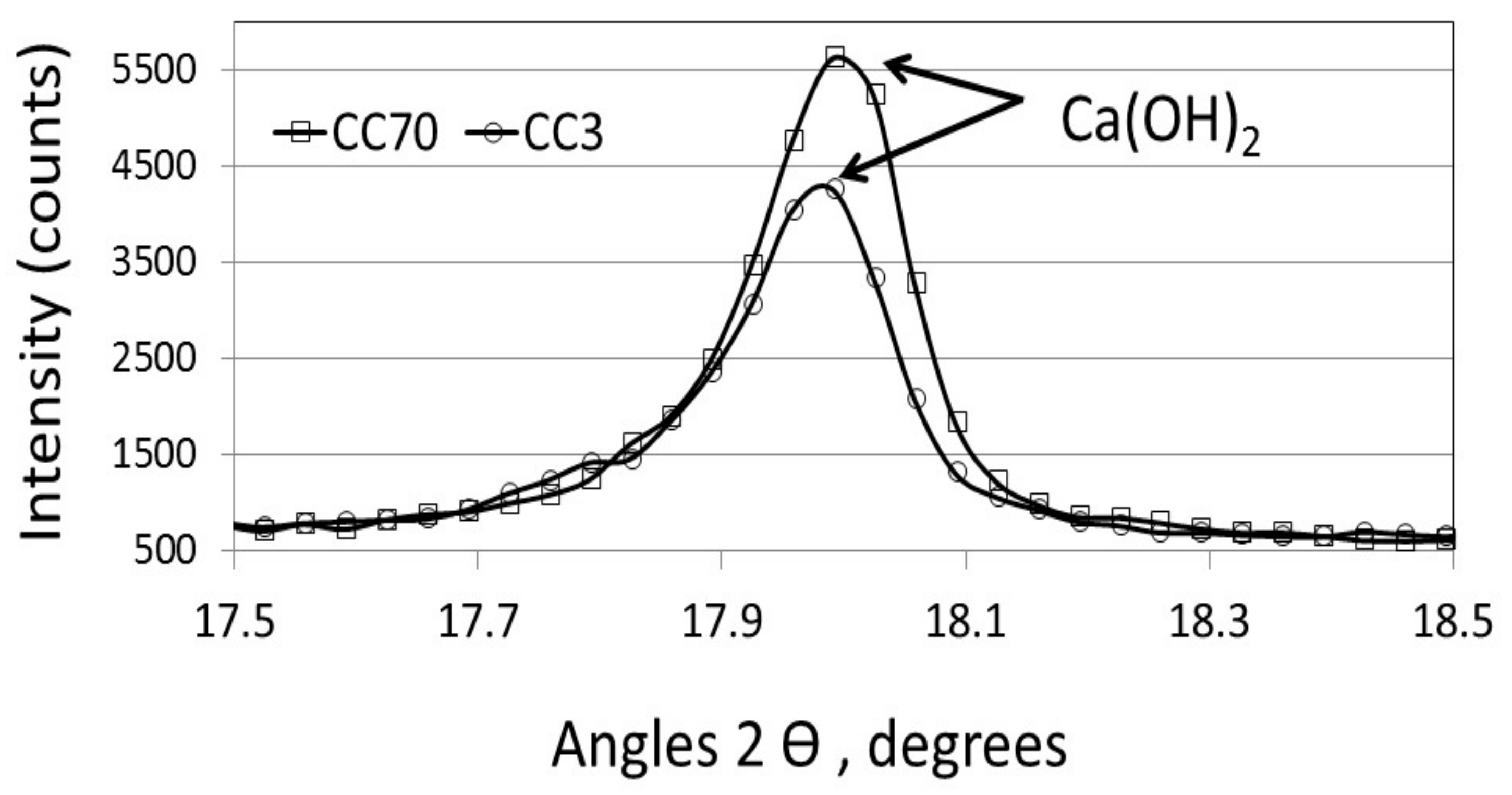
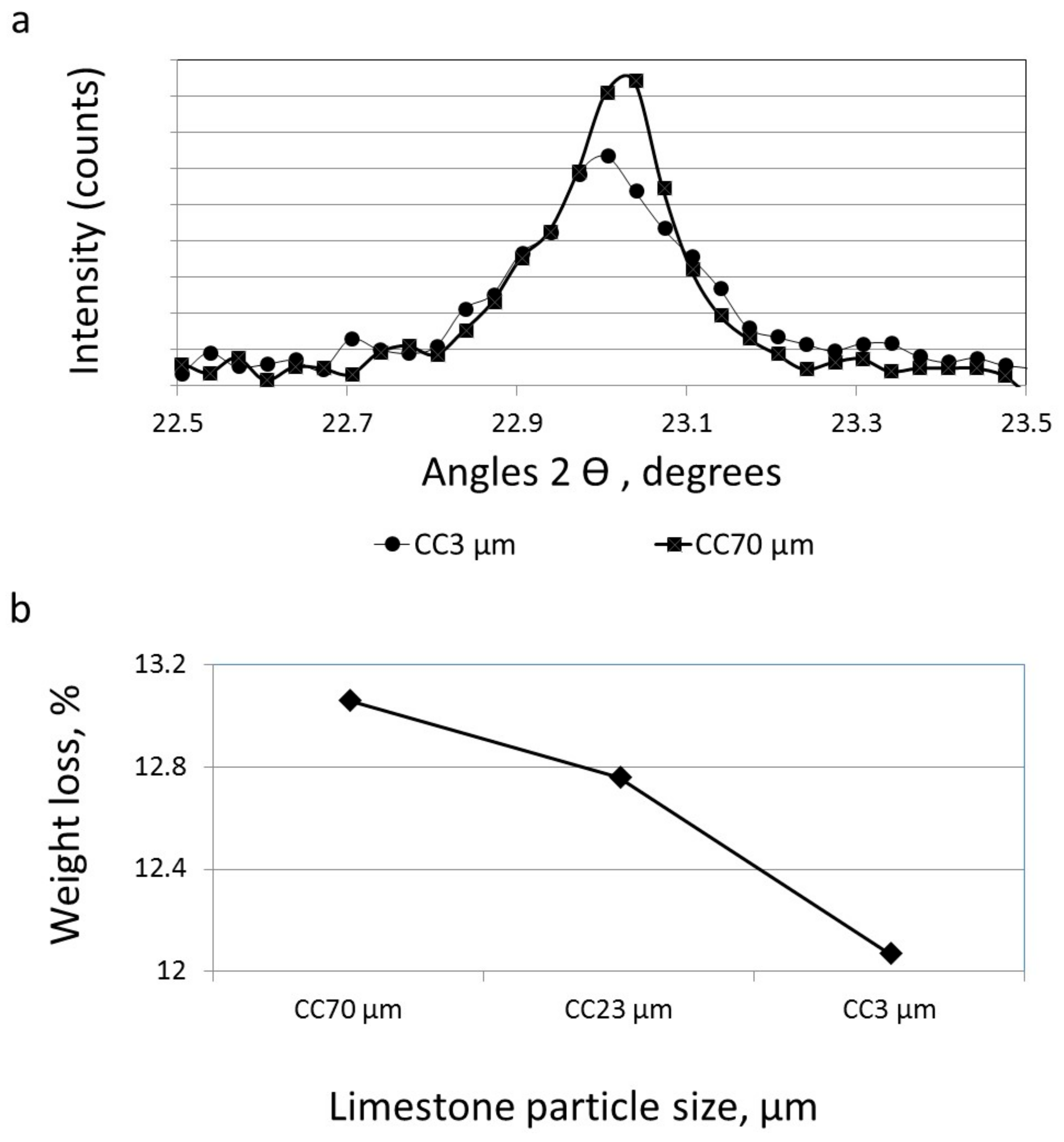
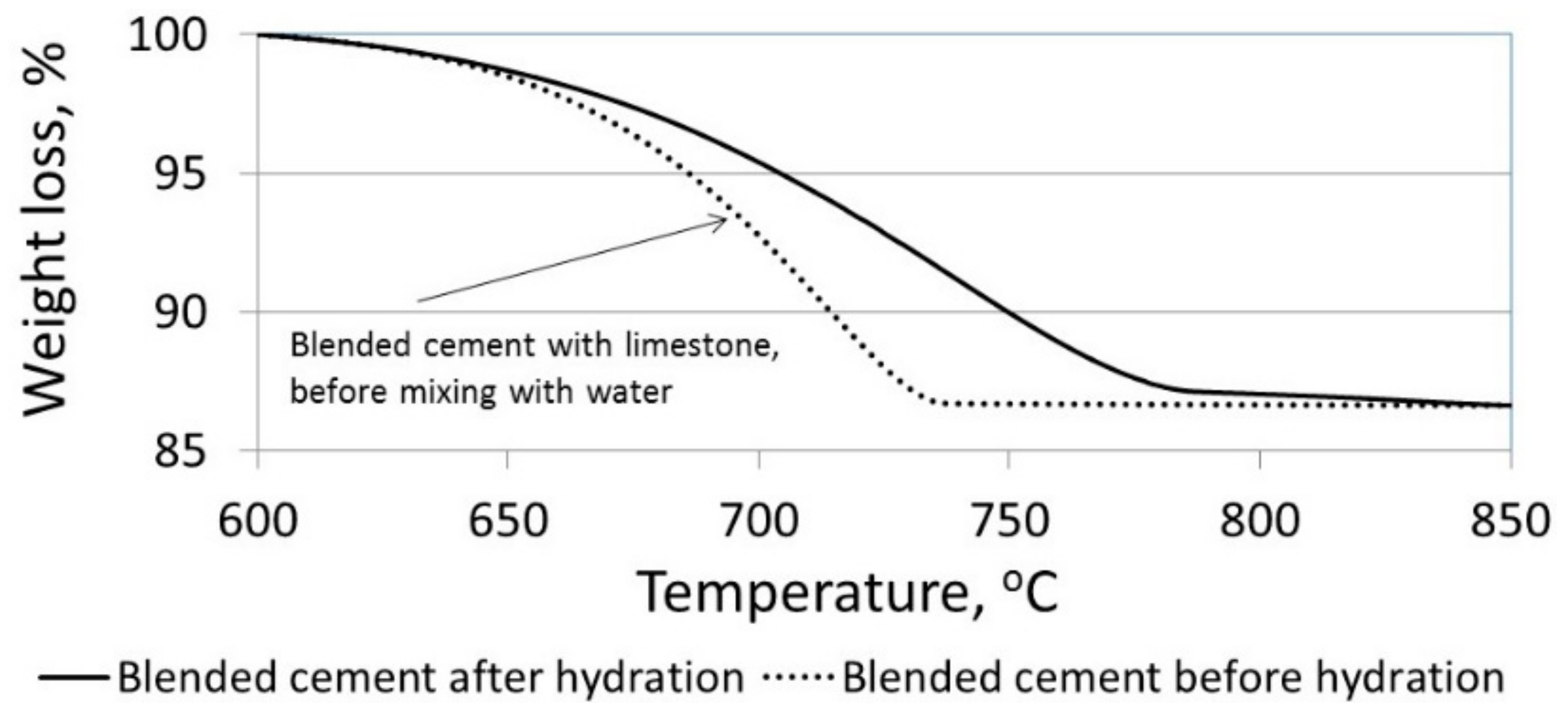
3.2. The Effect of Particle Size on the Amount of Ca(OH)2
- -
- Up to 300 °C: removal of water from hydrated products.
- -
- 400–500 °C: dehydration of calcium hydroxide.
- -
- 600–800 °C: decarbonation of calcium carbonate.
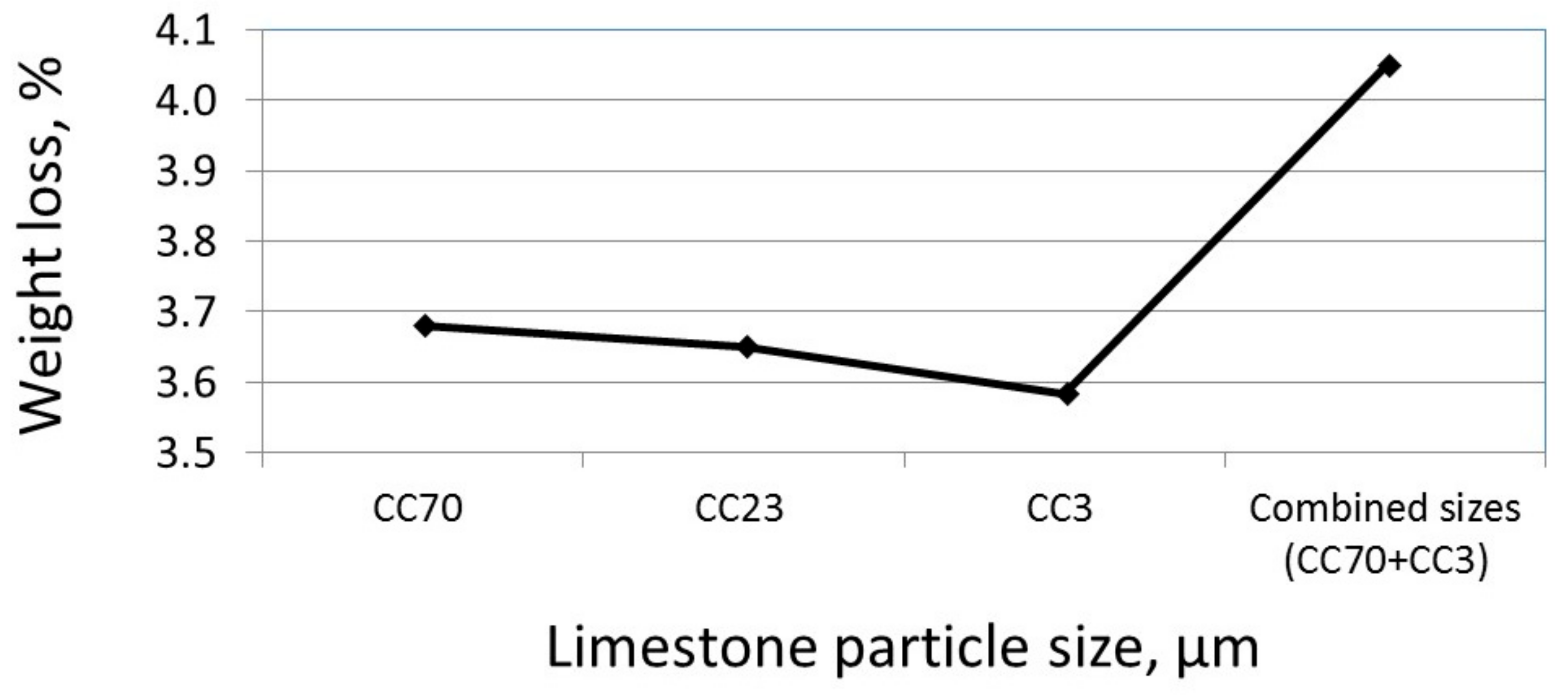
3.3. The Effect of Particle Size on the Reactivity of Limestone Powder
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Mayer, C. The greening of the concrete industry. Cem. Concr. Compos. 2009, 31, 601–605. [Google Scholar] [CrossRef]
- Malhotra, V.M. Role of supplementary cementing materials in reducing greenhouse gas emissions. In Concrete Technology for a Sustainable Development in the 21st Century; E&FN Spon: London, UK, 2000; pp. 35–226. [Google Scholar]
- McLellan, C.B.; Williams, P.R.; Janine, L.; Riessen, R.; Corder, D.G. Cost and carbon emissions for geopolymer pastes in comparison to ordinary portland cement. J. Clean. Prod. 2011, 19, 1080–1090. [Google Scholar] [CrossRef]
- Chindaprasirt, P.; Jaturapitakkul, C.; Sinsiri, T. Effect of fly ash fineness on microstructure of blended cement paste. Constr. Build. Mater. 2007, 21, 1534–1541. [Google Scholar] [CrossRef]
- Quanlin, N.; Feng, N.; Yang, J.; Zheng, X. Effect of superfine slag powder on cement properties. Cem. Concr. Res. 2002, 32, 615–621. [Google Scholar]
- Mija, H.; Hubler, A.; Jeffrey, J.; Thomas, B.; Hamlin, M.J. Influence of nucleation seeding on the hydration kinetics and compressive strength of alkali activated slag paste. Cem. Concr. Res. 2011, 41, 842–846. [Google Scholar]
- Rojas, M.F.; de Rojas, M.I.S. The effect of high curing temperature on the reaction kinetics in MK/lime and MK-blended cement matrices at 60 °C. Cem. Concr. Res. 2003, 33, 643–649. [Google Scholar] [CrossRef]
- Cement—Part 1: Compositions, Specifications and Conformity Criteria for Common Cements; CSN EN197-1 ed.2; Czech Office for Standards, Metrology and Testing: Praha, Czech Republic, 2011.
- Vuk, T.; Tinta, V.; Gabrovsek, R.; Kaucic, V. The effects of limestone addition, clinker type and fineness on properties of Portland cement. Cem. Concr. Res. 2001, 317, 135–139. [Google Scholar] [CrossRef]
- Henglin, L.; Fuping, J.; Cheng, Y.; Yong, L.; Long, Y.; Yang, J. Study on properties of blended finely ground limestone powder cement and compressive strength of mortar. Adv. Mater. Res. 2011, 168, 1449–1454. [Google Scholar]
- Sezera, G.I.; Çopuroglu, O.; Ramyar, K. Microstructure of 2- and 28-day cured Portland limestone cement pastes. Indian J. Eng. Mater. Sci. 2010, 17, 289–294. [Google Scholar]
- Kadri, E.H.; Aggoun, S.; de Schutterk, G.; Ezziane, K. Combined effect of chemical nature and fineness of mineral powders on Portland cement hydration. Mater. Struct. 2010, 43, 665–673. [Google Scholar] [CrossRef]
- Tsivolis, S.; Kakali, G.; Chaniotakis, E.; Souvaridou, A. A study on the hydration of Portland limestone cement by means of TG. J. Therm. Anal. 1988, 52, 863–870. [Google Scholar] [CrossRef]
- Knop, Y.; Peled, A.; Cohen, R. Influence of limestone particle size distributions and contents on blended cement properties. Constr. Build. Mater. 2014, 71, 26–34. [Google Scholar] [CrossRef]
- Kumar, A.; Oey, T.; Seohyum, K.; Thomas, D.; Badran, S.; Li, J.; Fernandes, F.; Neithalath, N.; Sant, G. Simple method to estimate the influence of limestone fillers on reaction and property evolution in cementitious materials. Cem. Concr. Comp. 2013, 42, 20–29. [Google Scholar] [CrossRef]
- Gallias, J.L.; Kara-Ali, R.; Bigas, J.P. The effect of fine mineral admixtures on water requirement of cement pastes. Cem. Concr. Res. 2000, 30, 1543–1549. [Google Scholar] [CrossRef]
- Wong, H.C.H.C.; Kwan, A.K.H. Packing density of cementitious materials: Part 1—Measurement using a wet packing method. Mater. Struct. 2008, 41, 689–701. [Google Scholar] [CrossRef]
- Knop, Y.; Peled, A. Setting behavior of blended cement with limestone: Influence of particle size and content. Mater. Struct. 2016, 49, 439–452. [Google Scholar] [CrossRef]
- Knop, Y.; Peled, A. Packing density modeling of blended cement with limestone having different particle sizes. Constr. Build. Mater. 2016, 102, 44–50. [Google Scholar] [CrossRef]
- Ramachandran, V. Thermal Analysis of cement components hydrated in the presence of calcium carbonate. Thermochim. Acta 1988, 127, 385–394. [Google Scholar] [CrossRef]
- Ingram, K.D.; Daugherty, K.E. A review of limestone addition on Portland cement and concrete. Cem. Concr. Comp. 1991, 13, 165–170. [Google Scholar] [CrossRef]
- Lothenbach, B.; Saout, L.G.; Gallucci, E.; Scrivener, K. Influence of limestone on the hydration of Portland cements. Cem. Concr. Res. 2008, 38, 848–860. [Google Scholar] [CrossRef]
- Kakali, G.; Tsivilis, S.; Aggeli, E.; Bati, M. Hydration products of C3A, C3S and Portland cement in the presence of CaCO3. Cem. Concr. Res. 2000, 30, 1073–1077. [Google Scholar] [CrossRef]
- Methods of Testing Cement—Part 3: Determination of Setting Times and Soundness; EN 196-3; British Standards Institution: London, UK, 2016.
- Feng, X.; Garboczia, E.J.; Bentz, D.P.; Stutzman, P.E.; Mason, T.O. Estimation of the degree of hydration of blended cement pastes by a scanning electron microscope point-counting procedure. Cem. Concr. Res. 2004, 34, 1787–1793. [Google Scholar] [CrossRef]
- Bentz, D.P. Modeling the influence of limestone filler on cement hydration using CEMHYD3D. Cem. Concr. Comp. 2006, 283, 124–129. [Google Scholar] [CrossRef]
- Hewlett, P.C. Lea’s Cemistry of Cement and Concrete, 4th ed.; Elsevier: Amsterdam, The Netherlands, 2004. [Google Scholar]
- Wang, X.Y.; Lee, H.S. A model predicting carbonation depth of concrete containing silica fume. Mater. Struct. 2009, 42, 691–704. [Google Scholar] [CrossRef]
- Talukdar, S.; Banthnia, N.; Grace, J.R. Carbonation in concrete infrastructure in the context of global climate change—Part 1: Experimental results and model development. Cem. Concr. Com. 2012, 34, 924–930. [Google Scholar] [CrossRef]

| Component | CaO | SiO2 | Al2O3 | Fe2O3 | MgO | TiO2 | K2O | Na2O | P2O5 | Mn2O3 | SO3 |
|---|---|---|---|---|---|---|---|---|---|---|---|
| % | 65.07 | 18.96 | 4.5 | 2.46 | 1.16 | 0.36 | 0.33 | 0.21 | 0.32 | 0.30 | 2.86 |
| Property | Unit | Cement CEM I | CC70 µm | CC53 µm | CC23 µm | CC7 µm | CC3 µm |
|---|---|---|---|---|---|---|---|
| Mean particle size | µm | 17.02 | 70.28 | 53.40 | 23.01 | 7.07 | 2.99 |
| Surface area (BET) | m2/gr | 1.53 | 0.23 | 0.39 | 0.85 | 3.29 | 6.22 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Knop, Y.; Peled, A. Sustainable Blended Cements—Influences of Packing Density on Cement Paste Chemical Efficiency. Materials 2018, 11, 625. https://doi.org/10.3390/ma11040625
Knop Y, Peled A. Sustainable Blended Cements—Influences of Packing Density on Cement Paste Chemical Efficiency. Materials. 2018; 11(4):625. https://doi.org/10.3390/ma11040625
Chicago/Turabian StyleKnop, Yaniv, and Alva Peled. 2018. "Sustainable Blended Cements—Influences of Packing Density on Cement Paste Chemical Efficiency" Materials 11, no. 4: 625. https://doi.org/10.3390/ma11040625
APA StyleKnop, Y., & Peled, A. (2018). Sustainable Blended Cements—Influences of Packing Density on Cement Paste Chemical Efficiency. Materials, 11(4), 625. https://doi.org/10.3390/ma11040625




