The Use of Municipal Solid Waste Incineration Ash in Various Building Materials: A Belgian Point of View
Abstract
1. Introduction
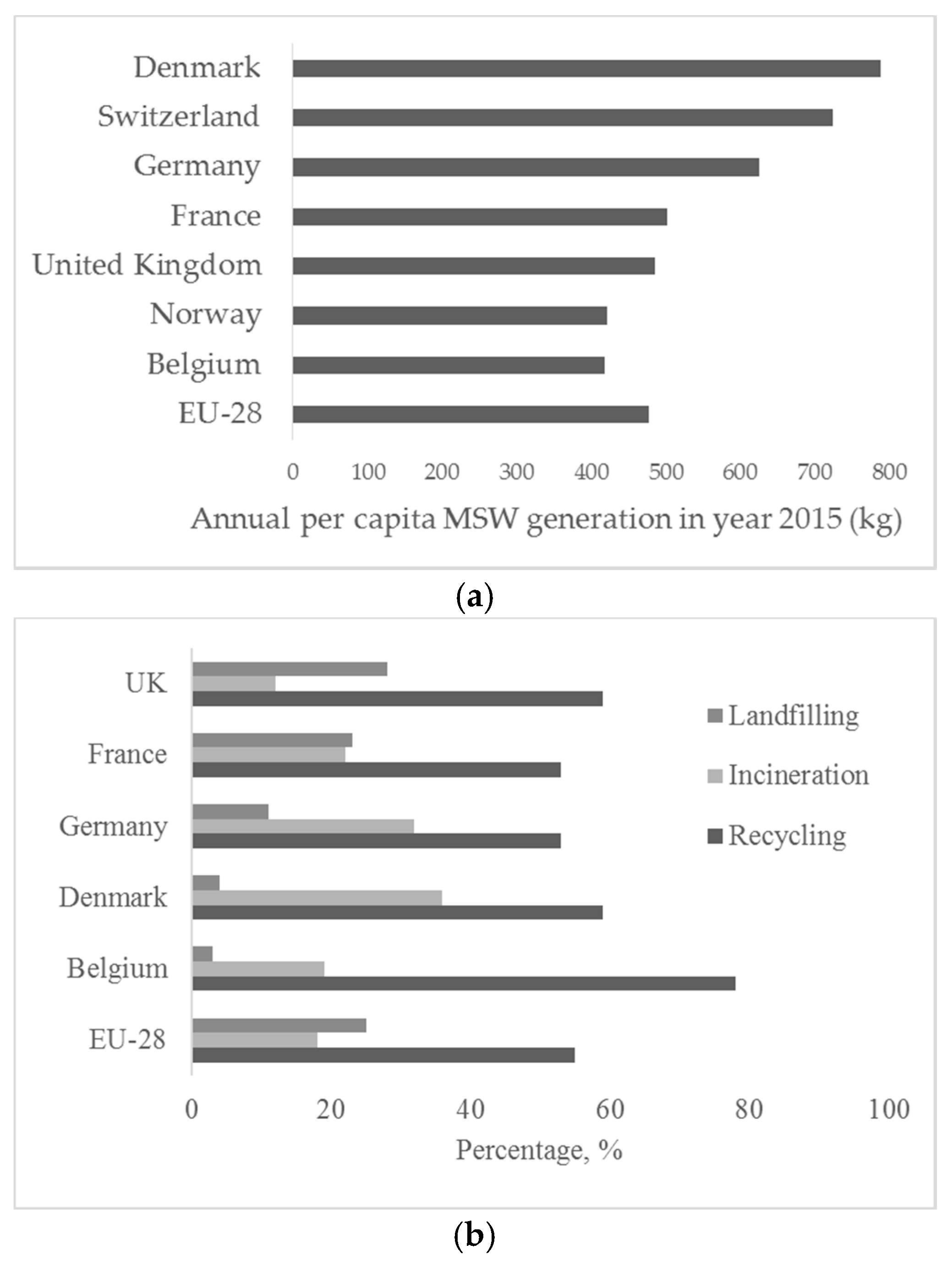
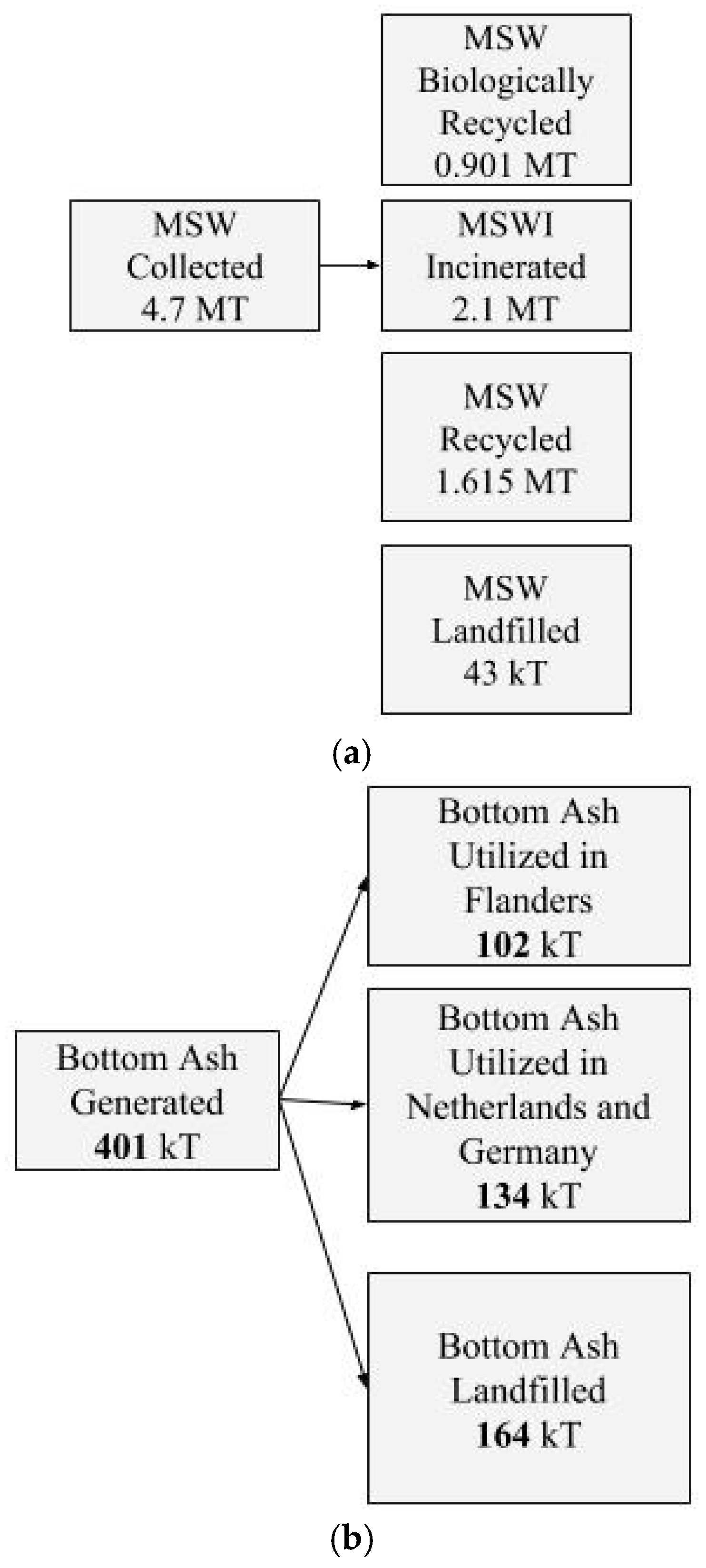
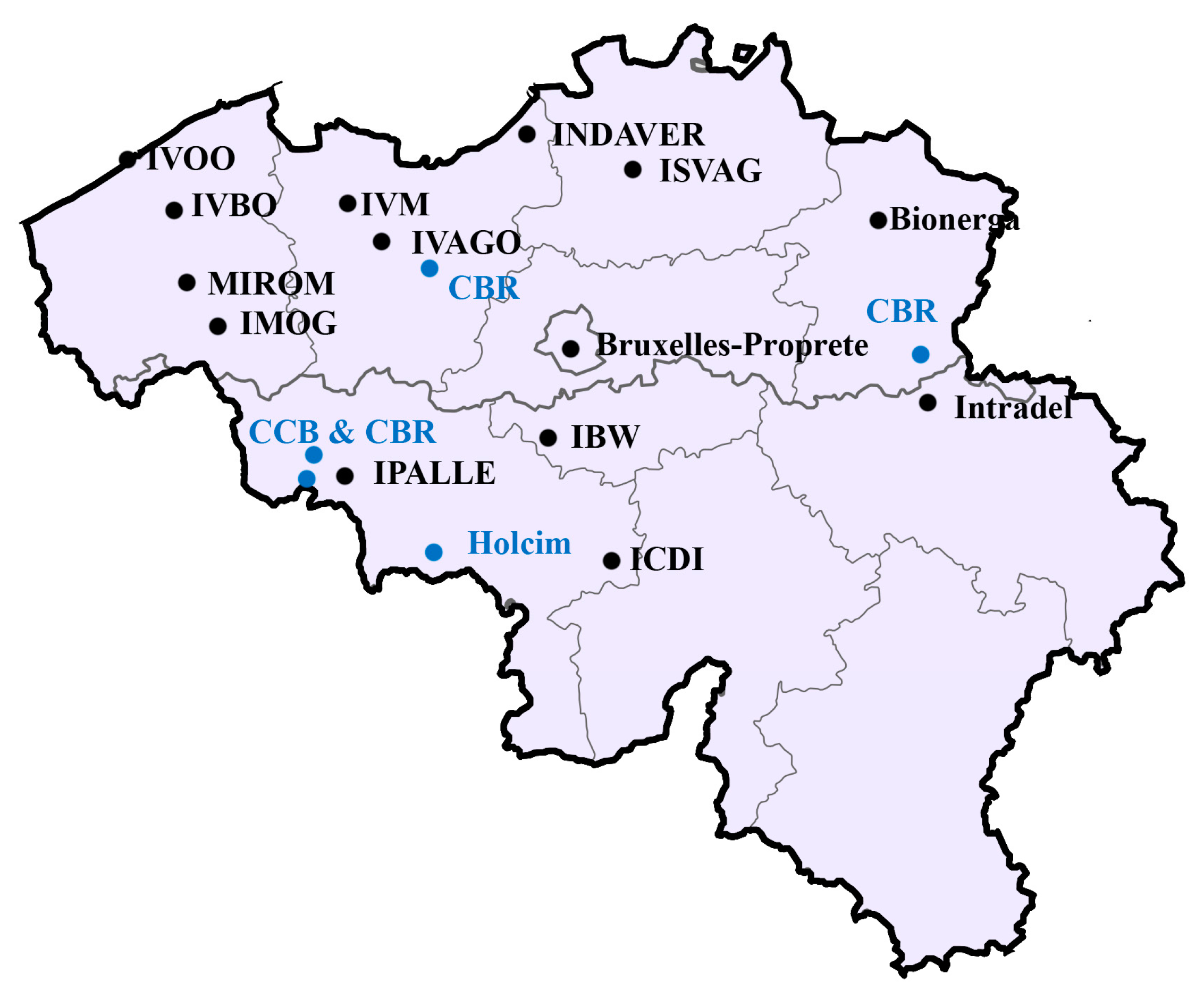
2. Solid Waste Management and Incineration Ash Recycling in Belgium
3. Types of Incinerators and Ashes in Belgium
4. Characteristics of Incineration Ashes
4.1. Physical Properties
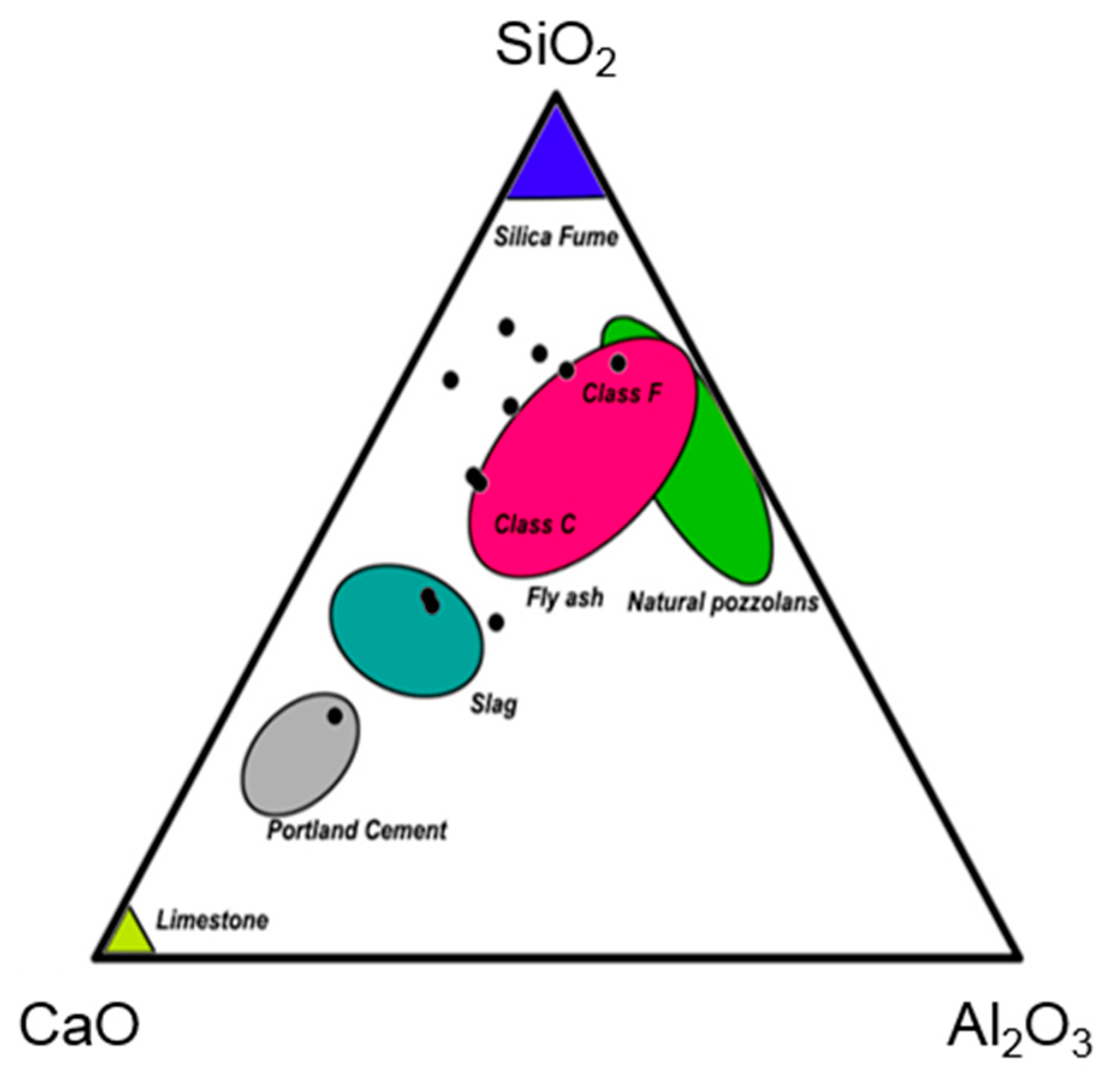
4.2. Chemical Properties
4.2.1. Chemical Analysis by XRF
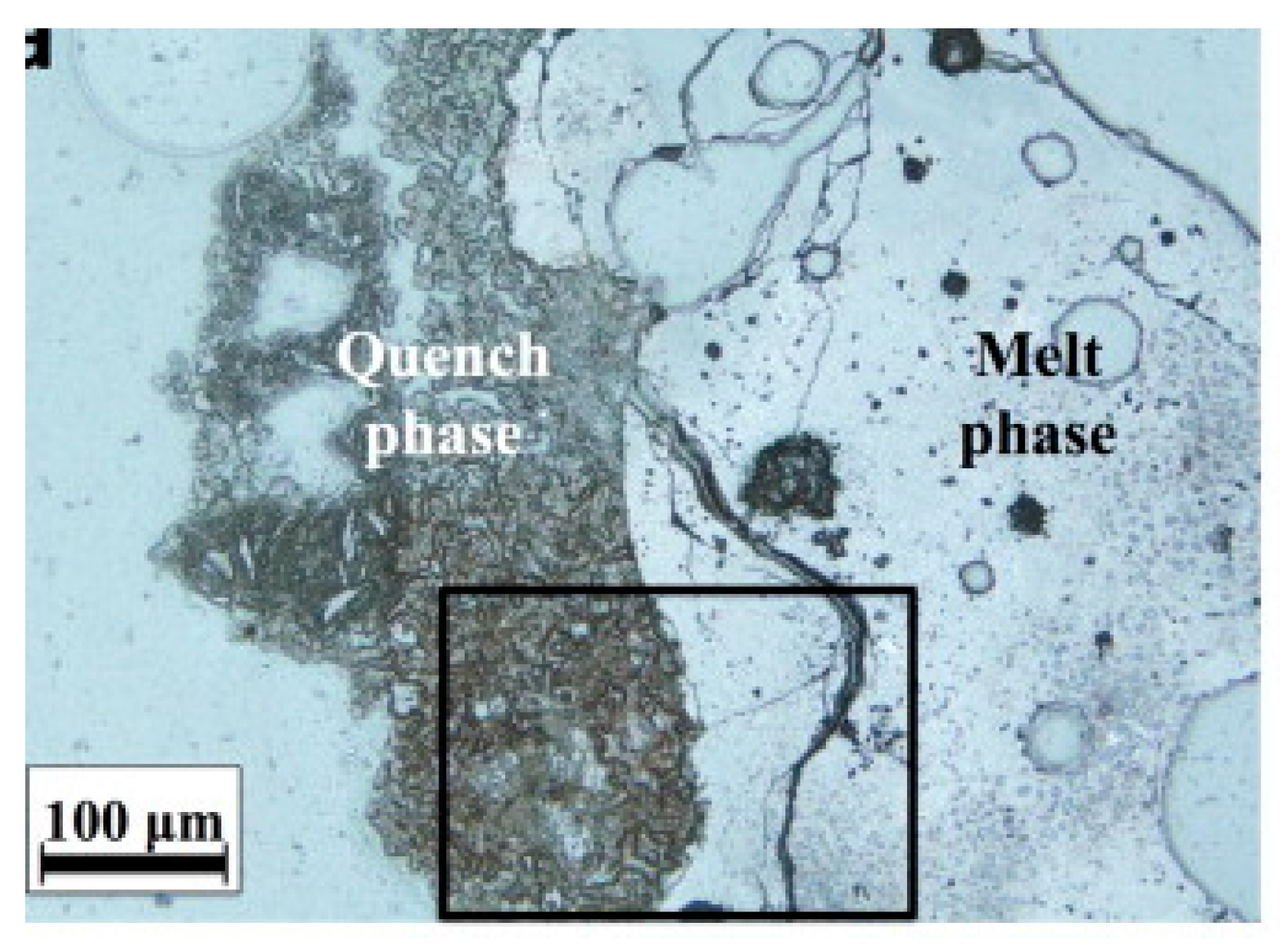
4.2.2. Mineralogical Composition
5. Beneficiation Needs for Use of MSWI Ash in Building Materials
5.1. Metallic Aluminum and Zinc
5.2. Salts
5.3. Heavy Metals
5.4. Persistent Organic Pollutants
- PCBs: 50 mg/kg
- PCDDs and PCDFs: 15 µg TEQ/kg
- Aldrin, chlordane, DDT, dieldrin, endrin, heptachlor, hexachlorobenzene (HCB), mirex and toxaphene: 50 mg/kg for each of these POPs.
5.5. Amorphous Silica Content
6. Bottom Ash Pre-Treatment Techniques
6.1. Pre-Treatment Techniques Used at the Industrial Scale
6.1.1. Magnetic Separation
6.1.2. Eddy Current Separation
6.1.3. Washing
6.1.4. Shaking Table
6.1.5. Jig Head Separation
6.1.6. Ageing
6.2. Pre-Treatment Techniques in the Research Stage
6.2.1. Washing with Alkali
6.2.2. Sulfide Rich Effluent
6.2.3. Wet Grinding
6.2.4. Carbonation
6.2.5. Phosphation
6.2.6. Cement/Other Binder Stabilization
6.2.7. Hydrothermal Treatment
6.2.8. Thermal Treatment
6.2.9. Electrodialytic Remediation
6.2.10. Revasol Process
7. Research Regarding Ash Pre-Treatment in Belgium
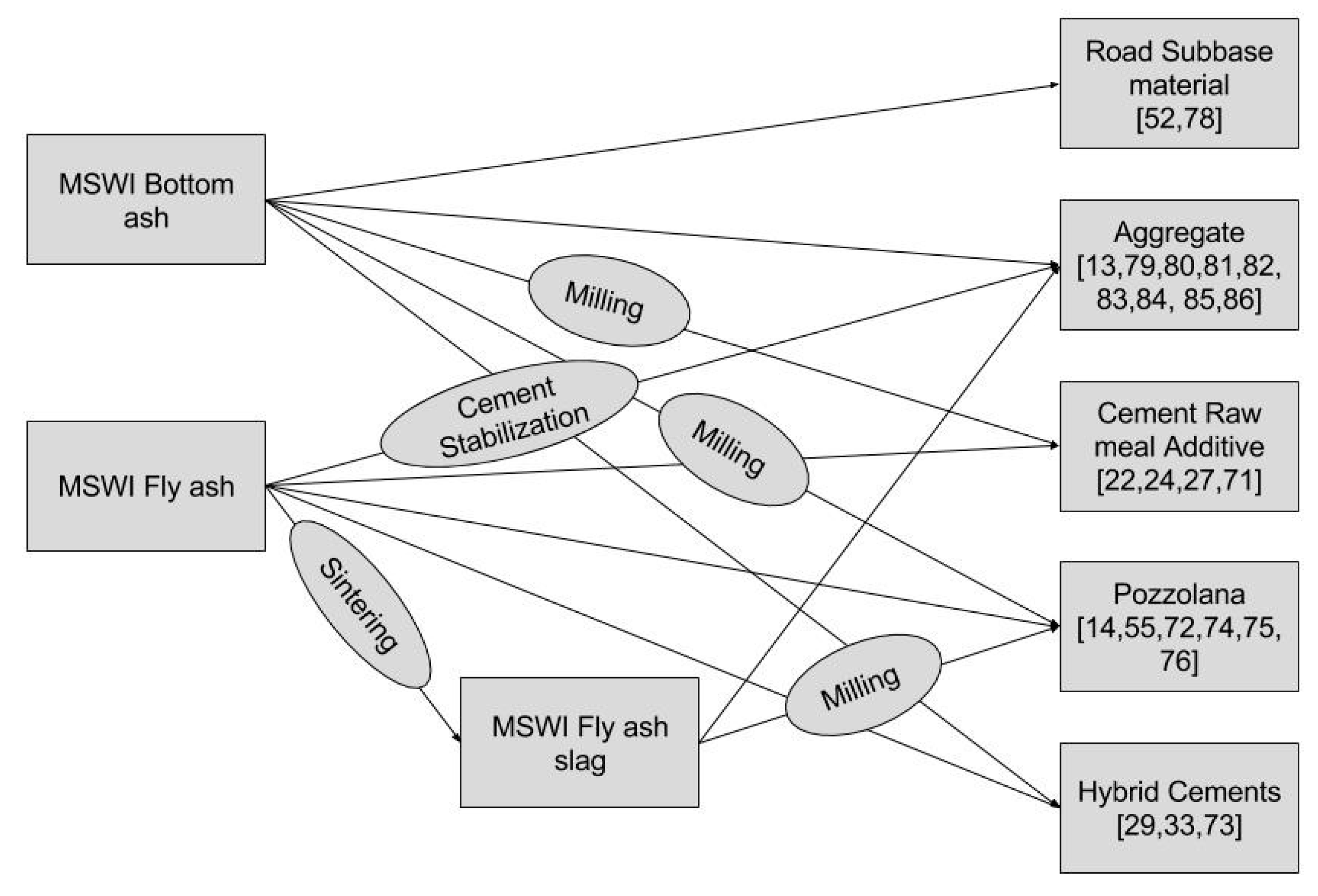
8. Utilization of MSWI Bottom Ash in Building Materials
8.1. Present Areas of Utilization
8.1.1. As a Road Construction Material and Landfill Stabilizer
8.1.2. As Cement Raw Meal Additive
8.1.3. MSWI Bottom Ash as Aggregate in Concrete
8.2. Potential Areas of Utilization
8.2.1. MSWI Ash as a Pozzolanic Addition in Cement
8.2.2. Autoclaved Aerated Concrete
8.2.3. Manufacture of Ceramics
9. Cement Industry and Utilization Potential of MSWI Ashes in Belgium
10. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- European Commision. Circular Economy. 2017. Available online: http://ec.europa.eu/environment/circular-economy/index_en.htm (accessed on 5 October 2017).
- European Environment Agency. Managing Municipal Solid Waste—A Review of Achievements in 32 European Countries; European Environment Agency: Copenhagen, Denmark, 2013. [Google Scholar]
- Hoornweg, D.; Bhada-Tata, P.; Joshi-Ghani, A. What a Waste: A Global Review of Solid Waste Management; Urban Development and Local Government Unit, World Bank: Washington, DC, USA, 2009. [Google Scholar]
- Belevi, H.; Moench, H. Factors determining the element behavior in municipal solid waste incinerators. 1. Field studies. Environ. Sci. Technol. 2000, 34, 2501–2506. [Google Scholar] [CrossRef]
- Cheng, H.; Hu, Y. Municipal solid waste (MSW) as a renewable source of energy: Current and future practices in China. Bioresour. Technol. 2010, 101, 3816–3824. [Google Scholar] [CrossRef] [PubMed]
- European Commission. Integrated Pollution Prevention and Control—Reference Document on the Best Available Techniques for Waste Incineration; European Commission: Brussels, Belgium, 2006. [Google Scholar]
- Bertolini, L.; Carsana, M.; Cassago, D.; Curzio, A.Q.; Collepardi, M. MSWI ashes as mineral additions in concrete. Cem. Concr. Res. 2004, 34, 1899–1906. [Google Scholar] [CrossRef]
- Sawell, S.; Chandler, A.; Eighmy, T.; Hartlén, J.; Hjelmar, O.; Kosson, D.; van der Sloot, H.; Vehlow, J. An international perspective on the characterisation and management of residues from MSW incinerators. Biomass Bioenergy 1995, 9, 377–386. [Google Scholar] [CrossRef]
- Csanyi, D.M.C. Environmental Hazards of Limestone Mining. Available online: http://education.seattlepi.com/environmental-hazards-limestone-mining-5608.html (accessed on 19 September 2016).
- Beretka, J.; de Vito, B.; Santoro, L.; Sherman, N.; Valenti, G.L. Utilisation of industrial wastes and by-products for the synthesis of special cements. Resour. Conserv. Recycl. 1993, 9, 179–190. [Google Scholar] [CrossRef]
- European Commision. EU Waste Legislation. 2016. Available online: http://ec.europa.eu/environment/waste/legislation/index.htm (accessed on 5 October 2017).
- Gentil, E.C. Municipal Waste Management in Belgium; European Environment Agency: Copenhagen, Denmark, 2013. [Google Scholar]
- Eurostat. Eurostat Data for Waste Management Indicators. Available online: http://ec.europa.eu/eurostat/statistics-explained/index.php/Waste_management_indicators (accessed on 5 October 2017).
- Wallon, G. Arrêté du Gouvernement Wallon Favorisant la Valorisation de Certains Déchets. 2001. Available online: http://environnement.wallonie.be/legis/dechets/decat024.htm (accessed on 5 October 2017).
- Vandecasteele, C.; Wauters, G.; Arickx, S.; Jaspers, M.; van Gerven, T. Integrated municipal solid waste treatment using a grate furnace incinerator: The Indaver case. Waste Manag. 2007, 27, 1366–1375. [Google Scholar] [CrossRef] [PubMed]
- Eurostat. Municipal Waste Statistics. European Commission, 2017. Available online: http://ec.europa.eu/eurostat/statistics-explained/index.php/Municipal_waste_statistics (accessed on 11 November 2017).
- Nielsen, P.; Kenis, C.; Vanassche, S.; Vrancken, K. Beste Beschikbare Technieken (BBT) Voor Behandeling van Bodemas van Huisvuilverbranding; Academia Press: New York, NY, USA, 2007. [Google Scholar]
- Leefmilieu, N.E.; MOVA; OVIT. Monitoringsysteem Duurzaam Oppervlaktedelfstoffenbeleid; De Vlaamse Overheid, Flanders Government: Brussels, Belgium, 2013. [Google Scholar]
- Born, J.P.; van Brecht, A. Recycling potentials of MSWI Bottom Ash. CEWEP 2014. Available online: http://www.cewep.eu/m_1318 (accessed on 5 October 2017).
- Valomac. Valoblock® : Des Mâchefers au Matériau de Construction. 2013. Available online: https://suezbelgium.be/fr/economie-circulaire/creer-des-matieres-premieres/valoblock (accessed on 5 October 2017).
- Niessen, W.R. Combustion and Incineration Processes; Marcel Dekker, Inc.: New York, NY, USA, 2002. [Google Scholar]
- Belgian Waste to Energy BW2E. Available online: http://www.bw2e.be/nl/page/leden (accessed on 5 October 2017).
- Belevi, H.; Langmeier, M. Factors determining the element behavior in municipal solid waste incinerators. 2. Laboratory experiments. Environ. Sci. Technol. 2000, 34, 2507–2512. [Google Scholar] [CrossRef]
- Wiles, C.C. Municipal solid waste combustion ash: State of the knowledge. J. Hazard. Mater. 1995, 3894, 20. [Google Scholar] [CrossRef]
- Müller, U.; Rübner, K. The microstructure of concrete made with municipal waste incinerator bottom ash as an aggregate component. Cem. Concr. Res. 2006, 36, 1434–1443. [Google Scholar] [CrossRef]
- Aubert, J.E.; Husson, B.; Sarramone, N. Utilization of municipal solid waste incineration (MSWI) fly ash in blended cement. Part 1: Processing and characterization of MSWI fly ash. J. Hazard. Mater. 2006, 136, 624–631. [Google Scholar] [CrossRef] [PubMed]
- Hyks, J.; Astrup, T. Influence of operational conditions, waste input and ageing on contaminant leaching from waste incinerator bottom ash: A full-scale study. Chemosphere 2009, 76, 1178–1184. [Google Scholar] [CrossRef] [PubMed]
- Polettini, A.; Pomi, R.; Carcani, G. The effect of Na and Ca salts on MSWI bottom ash activation for reuse as a pozzolanic admixture. Resour. Conserv. Recycl. 2005, 43, 403–418. [Google Scholar] [CrossRef]
- Alhassan, H.M.; Musa, A.; International, T.; Alhassan, H.M.; Tanko, A.M. Characterization of Solid Waste Incinerator Bottom Ash and the Potential for its Use. Int. J. Eng. Res. Appl. 2012, 2, 516–522. [Google Scholar]
- Silva, R.V.; de Brito, J.; Lynn, C.J.; Dhir, R.K. Use of municipal solid waste incineration bottom ashes in alkali-activated materials, ceramics and granular applications: A review. Waste Manag. 2017, 68, 207–220. [Google Scholar] [CrossRef] [PubMed]
- Izquierdo, M.; Querol, X.; Josa, A.; Vazquez, E.; López-Soler, A. Comparison between laboratory and field leachability of MSWI bottom ash as a road material. Sci. Total Environ. 2008, 389, 10–19. [Google Scholar] [CrossRef] [PubMed]
- Krammart, P.; Tangtermsirikul, S. Properties of cement made by partially replacing cement raw materials with municipal solid waste ashes and calcium carbide waste. Constr. Build. Mater. 2004, 18, 579–583. [Google Scholar] [CrossRef]
- Bethanis, S.; Sollars, C.J.; Cheeseman, C.R. Properties and Microstructure of Sintered Incinerator Bottom Ash. Ceram. Int. 2002, 28, 881–886. [Google Scholar] [CrossRef]
- Keppert, M.; Pavlík, Z.; Tydlitát, V.; Volfová, P.; Švarcová, S.; Šyc, M.; Černý, R. Properties of municipal solid waste incineration ashes with respect to their separation temperature. Waste Manag. Res. 2012, 30, 1041–1048. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Saffarzadeh, A.; Shimaoka, T.; Kawano, T. Existence of Cl in municipal solid waste incineration bottom ash and dechlorination effect of thermal treatment. J. Hazard. Mater. 2014, 267, 214–220. [Google Scholar] [CrossRef] [PubMed]
- Inkaew, K.; Saffarzadeh, A.; Shimaoka, T. Modeling the formation of the quench product in municipal solid waste incineration (MSWI) bottom ash. Waste Manag. 2015, 52, 159–168. [Google Scholar] [CrossRef] [PubMed]
- Qiao, X.C.; Tyrer, M.; Poon, C.S.; Cheeseman, C.R. Characterization of alkali-activated thermally treated incinerator bottom ash. Waste Manag. 2008, 28, 1955–1962. [Google Scholar] [CrossRef] [PubMed]
- Thipse, S.; Schoenitz, M.; Dreizin, E. Morphology and composition of the fly ash particles produced in incineration of municipal solid waste. Fuel Process. Technol. 2002, 75, 173–184. [Google Scholar] [CrossRef]
- Song, G.J.; Kim, K.H.; Seo, Y.C.; Kim, S.C. Characteristics of ashes from different locations at the MSW incinerator equipped with various air pollution control devices. Waste Manag. 2004, 24, 99–106. [Google Scholar] [CrossRef]
- Lothenbach, B.; Scrivener, K.; Hooton, R.D. Supplementary cementitious materials. Cem. Concr. Res. 2011, 41, 1244–1256. [Google Scholar] [CrossRef]
- Saikia, N.; Mertens, G.; van Balen, K.; Elsen, J.; van Gerven, T.; Vandecasteele, C. Pre-treatment of municipal solid waste incineration (MSWI) bottom ash for utilisation in cement mortar. Constr. Build. Mater. 2015, 96, 76–85. [Google Scholar] [CrossRef]
- Shih, P.H.; Chang, J.E.; Chiang, L.C. Replacement of raw mix in cement production by municipal solid waste incineration ash. Cem. Concr. Res. 2003, 33, 1831–1836. [Google Scholar] [CrossRef]
- Tang, P.; Florea, M.V.A.; Spiesz, P.; Brouwers, H.J. The investigation of the MSWI bottom ash fines (0–2 mm) as binder substitute after combined treatments. In Proceedings of the Eurasia Waste Management Symposium, Istanbul, Turkey, 28–30 April 2014; pp. 126–130. [Google Scholar]
- Kikuchi, R. Recycling of municipal solid waste for cement production: Pilot-scale test for transforming incineration ash of solid waste into cement clinker. Resour. Conserv. Recycl. 2001, 31, 137–147. [Google Scholar] [CrossRef]
- Qiao, X.C.; Tyrer, M.; Poon, C.S.; Cheeseman, C.R. Novel cementitious materials produced from incinerator bottom ash. Resour. Conserv. Recycl. 2008, 52, 496–510. [Google Scholar] [CrossRef]
- Song, Y.; Li, B.; Yang, E.; Liu, Y.; Ding, T. Feasibility study on utilization of municipal solid waste incineration bottom ash as aerating agent for the production of autoclaved aerated concrete. Cem. Concr. Compos. 2015, 56, 51–58. [Google Scholar] [CrossRef]
- Rambaldi, E.; Esposito, L.; Andreola, F.; Barbieri, L.; Lancellotti, I.; Vassura, I. The recycling of MSWI bottom ash in silicate based ceramic. Ceram. Int. 2010, 36, 2469–2476. [Google Scholar] [CrossRef]
- Piantone, P.; Bodénan, F.; Chatelet-Snidaro, L. Mineralogical study of secondary mineral phases from weathered MSWI bottom ash: Implications for the modelling and trapping of heavy metals. Appl. Geochem. 2004, 19, 1891–1904. [Google Scholar] [CrossRef]
- Bayuseno, A.P.; Schmahl, W.W. Understanding the chemical and mineralogical properties of the inorganic portion of MSWI bottom ash. Waste Manag. 2010, 30, 1509–1520. [Google Scholar] [CrossRef] [PubMed]
- Alba, N.; Gasso, S.; Lacorte, T.; Baldasano, J.M. Characterization of municipal solid waste incineration residues from facilities with different air pollution control systems. J. Air Waste Manag. Assoc. 1997, 47, 1170–1179. [Google Scholar] [CrossRef]
- Lin, K. The influence of municipal solid waste incinerator fly ash slag blended in cement pastes. Cem. Concr. Res. 2005, 35, 979–986. [Google Scholar] [CrossRef]
- Aubert, J.E.; Husson, B.; Vaquier, A. Use of municipal solid waste incineration fly ash in concrete. Cem. Concr. Res. 2004, 34, 957–963. [Google Scholar] [CrossRef]
- Violante, A.; Huang, P.M. Formation mechanism of aluminum hydroxide polymorphs. Clays Clay Miner. 1993, 41, 590–597. [Google Scholar] [CrossRef]
- Garcia-Lodeiro, I.; Carcelen-Taboada, V.; Fernández-Jiménez, A.; Palomo, A. Manufacture of hybrid cements with fly ash and bottom ash from a municipal solid waste incinerator. Constr. Build. Mater. 2016, 105, 218–226. [Google Scholar] [CrossRef]
- Pera, J.; Courtaz, J.; Ambroise, J.; Chababbet, M. Use of Incinerator Bottom Ash in Concrete. Cem. Concr. Res. 1997, 27, 1–5. [Google Scholar] [CrossRef]
- CUR. Bouwen Met Kennis Materiaal Voor Beton; CUR Bouw & Infra: Gouda, The Netherlands, 2012. [Google Scholar]
- Zhou, J.; Wu, S.; Pan, Y.; Zhang, L.; Cao, Z.; Zhang, X.; Yonemochi, S.; Hosono, S.; Wang, Y.; Oh, K.; et al. Enrichment of heavy metals in fine particles of municipal solid waste incinerator (MSWI) fly ash and associated health risk. Waste Manag. 2015, 43, 239–246. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.; Xu, Y.; Sun, J.; Cao, Z.; Zhou, J.; Pan, Y.; Qian, G. Inhibiting evaporation of heavy metal by controlling its chemical speciation in MSWI fly ash. Fuel 2015, 158, 764–769. [Google Scholar] [CrossRef]
- Bertolini, L.; Elsener, B.; Pedeferri, P.; Polder, R. Corrosion of Steel in Concrete; Wiley-VCH, Verlag GmbH & Co. KGaA: Weinheim, Germany, 2004. [Google Scholar]
- Geurts, R.; Spooren, J.; Quaghebeur, M.; Broos, K.; Kenis, C.; Debaene, L. Round robin testing of a percolation column leaching procedure. Waste Manag. 2016, 55, 31–37. [Google Scholar] [CrossRef] [PubMed]
- Van Gerven, T.; Geysen, D.; Stoffels, L.; Jaspers, M.; Wauters, G.; Vandecasteele, C. Management of incinerator residues in Flanders (Belgium) and in neighbouring countries. A comparison. Waste Manag. 2005, 25, 75–87. [Google Scholar] [CrossRef] [PubMed]
- OVAM. VLAREMA Decree of the Flemish Government to Establish the Flemish Regulations on the Sustainable Management of Material Circuits and Waste. Government, Flemish. Available online: https://navigator.emis.vito.be/mijn-navigator?woId=44707 (accessed on 5 October 2017).
- UGent; VITO; CRH; Indaver; Recmix. ASHCEM Novel Cements & Building Materials from Incineration Ashes—Project Document; SIM: Zwijnaarde, Belgium, 2014. [Google Scholar]
- Astrup, T.; Rosenblad, C.; Trapp, S.; Christensen, T.H. Chromium release from waste incineration air-pollution-control residues. Environ. Sci. Technol. 2005, 39, 3321–3329. [Google Scholar] [CrossRef] [PubMed]
- Van Caneghem, J.; Block, C.; Vandecasteele, C. Destruction and formation of dioxin-like PCBs in dedicated full scale waste incinerators. Chemosphere 2014, 94, 42–47. [Google Scholar] [CrossRef] [PubMed]
- Hutzinger, O.; Blumich, M.J.; Berg, M.V.D.; Olie, K. Sources and fate of PCDDs and PCDFs: An overview. Chemosphere 1985, 14, 581–600. [Google Scholar] [CrossRef]
- Zhan, M.; Chen, T.; Lin, X.; Fu, J.; Li, X.; Yan, J.; Buekens, A. Suppression of dioxins after the post-combustion zone of MSWIs. Waste Manag. 2016, 54, 153–161. [Google Scholar] [CrossRef] [PubMed]
- HRigo, G.; Chandler, A.J. Is there a strong dioxin: Chlorine link in commercial scale systems? Chemosphere 1998, 37, 2031–2046. [Google Scholar]
- Wu, S.; Zhou, J.; Pan, Y.; Zhang, J.; Zhang, L.; Ohtsuka, N.; Motegi, M.; Yonemochi, S.; Oh, K.; Hosono, S.; et al. Dioxin distribution characteristics and health risk assessment in different size particles of fly ash from MSWIs in China. Waste Manag. 2016, 50, 113–120. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Jin, Y.; Nie, Y. Investigation of accelerated and natural carbonation of MSWI fly ash with a high content of Ca. J. Hazard. Mater. 2010, 174, 334–343. [Google Scholar] [CrossRef] [PubMed]
- Van den Heede, P.; Ringoot, N.; Beirnaert, A.; van Brecht, A.; van den Brande, E.; de Schutter, G.; de Belie, N. Sustainable high quality recycling of aggregates from waste-to-energy, treated in a wet bottom ash processing installation, for use in concrete products. Materials 2016, 9, 9. [Google Scholar] [CrossRef] [PubMed]
- SBerkhout, P.M.; Oudenhoven, B.P.M.; Rem, P.C. Optimizing Non-Ferrous Metal Value from MSWI Bottom Ashes. J. Environ. Prot. (Irvine Calif.) 2011, 2, 564–570. [Google Scholar] [CrossRef]
- Shen, H.; Forssberg, E. An overview of recovery of metals from slags. Waste Manag. 2003, 23, 933–949. [Google Scholar] [CrossRef]
- Xia, Y.; He, P.; Shao, L.; Zhang, H. Metal distribution characteristic of MSWI bottom ash in view of metal recovery. J. Environ. Sci. 2016, 52, 1–12. [Google Scholar] [CrossRef] [PubMed]
- De Boom, A.; Degrez, M.; Hubaux, P.; Lucion, C. MSWI boiler fly ashes: Magnetic separation for material recovery. Waste Manag. 2011, 31, 1505–1513. [Google Scholar] [CrossRef] [PubMed]
- Biganzoli, L.; Ilyas, A.; van Praagh, M.; Persson, K.M.; Grosso, M. Aluminium recovery vs. hydrogen production as resource recovery options for fine MSWI bottom ash fraction. Waste Manag. 2013, 33, 1174–1181. [Google Scholar] [CrossRef] [PubMed]
- Mulder, E. Pre-treatment of MSWI fly ash for useful application. Waste Manag. 1996, 16, 181–184. [Google Scholar] [CrossRef]
- Chen, X.; Bi, Y.; Zhang, H.; Wang, J. Chlorides Removal and Control through Water-washing Process on MSWI Fly Ash. Procedia Environ. Sci. 2016, 31, 560–566. [Google Scholar] [CrossRef]
- Chimenos, J.M.; Fernandez, A.I.; Cervantes, A.; Miralles, L.; Fernandez, M.A.; Espiell, F. Optimizing the APC residue washing process to minimize the release of chloride and heavy metals. Waste Manag. 2005, 25, 686–693. [Google Scholar] [CrossRef] [PubMed]
- Hu, B.; Rem, P.C.; van de Winckel, T.P.M. Fine heavy non-ferrous and precious metals recovery in bottom ash treatment. ISWA Knowl. Database 2009, APESB2009, 1–8. [Google Scholar]
- Metso. Basics in Minerals Processing; Metso Corporation: Helsinki, Finland, 2015; p. 354. [Google Scholar]
- Grewal, I. Mineral Processing Introduction. Available online: http://met-solvelabs.com/library/articles/mineral-processing-introduction (accessed on 10 December 2017).
- Aubert, J.E.; Husson, B.; Sarramone, N. Utilization of municipal solid waste incineration (MSWI) fly ash in blended cement. Part 2. Mechanical strength of mortars and environmental impact. J. Hazard. Mater. 2007, 146, 12–19. [Google Scholar] [CrossRef] [PubMed]
- Sivula, L.; Väisänen, A.; Rintala, J. Stabilisation of MSWI bottom ash with sulphide-rich anaerobic effluent. Chemosphere 2008, 71, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Gao, X.; Li, T.; Cheng, S.; Yang, H.; Qiao, Y. Stabilization of heavy metals in fly ashes from municipal solid waste incineration via wet milling. Fuel 2018, 216, 153–159. [Google Scholar] [CrossRef]
- Chen, C.; Sun, C.; Gau, S.; Wu, C.; Chen, Y. The effects of the mechanical—Chemical stabilization process for municipal solid waste incinerator fly ash on the chemical reactions in cement paste. Waste Manag. 2013, 33, 858–865. [Google Scholar] [CrossRef] [PubMed]
- Arickx, S.; van Gerven, T.; Vandecasteele, C. Accelerated carbonation for treatment of MSWI bottom ash. J. Hazard. Mater. 2006, 137, 235–243. [Google Scholar] [CrossRef] [PubMed]
- Del Toro, M.A.; Calmano, W.; Ecke, H. Wet extraction of heavy metals and chloride from MSWI and straw combustion fly ashes. Waste Manag. 2009, 29, 2494–2499. [Google Scholar] [CrossRef] [PubMed]
- Bournonville, B.; Nzihou, A.; Sharrock, P.; Depelsenaire, G. Stabilisation of heavy metal containing dusts by reaction with phosphoric acid: Study of the reactivity of fly ash. J. Hazard. Mater. 2004, 116, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Nzihou, A.; Sharrock, P. Calcium phosphate stabilization of fly ash with chloride extraction. Waste Manag. 2002, 22, 235–239. [Google Scholar] [CrossRef]
- Cornelis, G.; van Gerven, T.; Vandecasteele, C. Antimony leaching from uncarbonated and carbonated MSWI bottom ash. J. Hazard. Mater. 2006, 137, 1284–1292. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.Y.; Tyrer, M.; Hills, C.D.; Yang, X.M.; Carey, P. Immobilisation of heavy metal in cement-based solidification/stabilisation: A review. Waste Manag. 2009, 29, 390–403. [Google Scholar] [CrossRef] [PubMed]
- Shi, H.; Kan, L. Leaching behavior of heavy metals from municipal solid wastes incineration (MSWI) fly ash used in concrete. J. Hazard. Mater. 2009, 164, 750–754. [Google Scholar] [CrossRef] [PubMed]
- Galiano, Y.L.; Pereira, C.F.; Vale, J. Stabilization/solidification of a municipal solid waste incineration residue using fly ash-based geopolymers. J. Hazard. Mater. 2011, 185, 373–381. [Google Scholar] [CrossRef] [PubMed]
- Bosio, A.; Zacco, A.; Borgese, L.; Rodella, N.; Colombi, P.; Benassi, L.; Depero, L.E.; Bontempi, E. A sustainable technology for Pb and Zn stabilization based on the use of only waste materials: A green chemistry approach to avoid chemicals and promote CO2 sequestration. Chem. Eng. J. 2014, 253, 377–384. [Google Scholar] [CrossRef]
- Rodella, N.; Bosio, A.; Dalipi, R.; Zacco, A.; Borgese, L.; Depero, L.E.; Bontempi, E. Waste silica sources as heavy metal stabilizers for municipal solid waste incineration fly ash. Arab. J. Chem. 2017, 10, S3676–S3681. [Google Scholar] [CrossRef]
- Jing, Z.; Matsuoka, N.; Jin, F.; Hashida, T.; Yamasaki, N. Municipal incineration bottom ash treatment using hydrothermal solidification. Waste Manag. 2007, 27, 287–293. [Google Scholar] [CrossRef] [PubMed]
- Jing, Z.; Ran, X.; Jin, F.; Ishida, E.H. Hydrothermal solidification of municipal solid waste incineration bottom ash with slag addition. Waste Manag. 2010, 30, 1521–1527. [Google Scholar] [CrossRef] [PubMed]
- Jing, Z.; Fan, X.; Zhou, L.; Fan, J.; Zhang, Y.; Pan, X.; Ishida, E.H. Hydrothermal solidification behavior of municipal solid waste incineration bottom ash without any additives. Waste Manag. 2003, 33, 1182–1189. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Zhang, P.; Chen, D.; Zhou, B.; Li, J.; Li, X. Hydrothermal treatment of municipal solid waste incineration fly ash for dioxin decomposition. J. Hazard. Mater. 2012, 207–208, 79–85. [Google Scholar] [CrossRef] [PubMed]
- Bayuseno, A.P.; Schmahl, W.W.; Müllejans, T. Hydrothermal processing of MSWI Fly Ash-towards new stable minerals and fixation of heavy metals. J. Hazard. Mater. 2009, 167, 250–259. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.; Itoh, H. Extraction of metals from municipal solid waste incinerator fly ash by hydrothermal process. J. Hazard. Mater. 2006, 136, 663–670. [Google Scholar] [CrossRef] [PubMed]
- Joseph, A.M.; van den Heede, P.; Matthys, S.; de Belie, N.; Snellings, R.; Van, A. Comparison of different beneficiation techniques to improve utilization potential of municipal solid waste incineration fly ash concrete. In Proceedings of the International Conference on Advances in Construction Materials and Systems, Chennai, India, 3–8 September 2017. [Google Scholar]
- Pedersen, A.J. Evaluation of assisting agents for electrodialytic removal of Cd, Pb, Zn, Cu and Cr from MSWI fly ash. J. Hazard. Mater. 2002, 95, 185–198. [Google Scholar] [CrossRef]
- Flanders, S. MARES, Strategic Initiative Materials in Flanders. 2017. Available online: http://www.sim-flanders.be/research-program/mares (accessed on 2 February 2017).
- Van Gerven, T.; van Keer, E.; Arickx, S.; Jaspers, M.; Wauters, G.; Vandecasteele, C. Carbonation of MSWI-bottom ash to decrease heavy metal leaching, in view of recycling. Waste Manag. 2005, 25, 291–300. [Google Scholar] [CrossRef] [PubMed]
- Lynn, C.J.; Ghataora, G.S.; OBE, R.K.D. Municipal incinerated bottom ash (MIBA) characteristics and potential for use in road pavements. Int. J. Pavement Res. Technol. 2017, 10, 185–201. [Google Scholar] [CrossRef]
- Colangelo, F.; Cioffi, R.; Montagnaro, F.; Santoro, L. Soluble salt removal from MSWI fly ash and its stabilization for safer disposal and recovery as road basement material. Waste Manag. 2012, 32, 1179–1185. [Google Scholar] [CrossRef] [PubMed]
- Kong, Q.; Yao, J.; Qiu, Z.; Shen, D. Effect of mass proportion of municipal solid waste incinerator bottom ash layer to municipal solid waste layer on the Cu and Zn discharge from landfill. BioMed Res. Int. 2016, 2016, 9687879. [Google Scholar] [CrossRef] [PubMed]
- Li, W.B.; Yao, J.; Malik, Z.; di Zhou, G.; Dong, M.; Shen, D.S. Impact of MSWI bottom ash codisposed with MSW on landfill stabilization with different operational modes. BioMed Res. Int. 2014, 2014, 167197. [Google Scholar] [CrossRef] [PubMed]
- Gineys, N.; Aouad, G.; Sorrentino, F.; Damidot, D. Effect of the clinker composition on the threshold limits for Cu, Sn or Zn. Cem. Concr. Res. 2012, 42, 1088–1093. [Google Scholar] [CrossRef]
- Shimoda, T.; Yokoyama, S. Eco-cement: A new portland cement to solve municipal solid waste and industrial waste problems. In Proceedings of the Creating with Concrete, University of Dundee, Dundee, UK, 6–10 September 1999; ICE Publishing: London, UK, May 1999. [Google Scholar]
- De Schepper, M. Completely Recyclable Concrete for a More Environment Friendly Construction. Ph.D. Dissertation, Ghent University, Ghent, Belgium, 1 July 2014. [Google Scholar]
- Wang, L.; Jin, Y.; Nie, Y.; Li, R. Recycling of municipal solid waste incineration fly ash for ordinary Portland cement production: A real-scale test. Resour. Conserv. Recycl. 2010, 54, 1428–1435. [Google Scholar] [CrossRef]
- Collivignarelli, C.; Sorlini, S. Reuse of municipal solid wastes incineration fly ashes in concrete mixtures. Waste Manag. 2002, 22, 909–912. [Google Scholar] [CrossRef]
- Chen, H.; Wang, S.; Tang, C. Reuse of incineration fly ashes and reaction ashes for manufacturing lightweight aggregate. Constr. Build. Mater. 2010, 24, 46–55. [Google Scholar] [CrossRef]
- Ferraris, M.; Salvo, M.; Ventrella, A.; Buzzi, L.; Veglia, M. Use of vitrified MSWI bottom ashes for concrete production. Waste Manag. 2009, 29, 1041–1047. [Google Scholar] [CrossRef] [PubMed]
- Cioffi, R.; Colangelo, F.; Montagnaro, F.; Santoro, L. Manufacture of artificial aggregate using MSWI bottom ash. Waste Manag. 2011, 31, 281–288. [Google Scholar] [CrossRef] [PubMed]
- Hwang, C.; Bui, L.A.; Lin, K.; Lo, C. Manufacture and performance of lightweight aggregate from municipal solid waste incinerator fly ash and reservoir sediment for self-consolidating lightweight concrete. Cem. Concr. Compos. 2012, 34, 1159–1166. [Google Scholar] [CrossRef]
- ASTM International. ASTM C 618—Standard Specification for Coal Fly Ash and Raw or Calcined Natural Pozzolan for Use; ASTM International: West Conshohocken, PA, USA, 2010. [Google Scholar]
- Bureau voor Normalisatie. NBN EN 450-1 Fly Ash for Concrete- Part 1: Definitions, Specifications and Conformity Criteria; Bureau voor Normalisatie: Brussels, Belgium, 2012. [Google Scholar]
- ASTM International. ASTM C 150—Standard Specification of Portland Cement.pdf; ASTM International: West Conshohocken, PA, USA, 2007. [Google Scholar]
- ASTM International. ASTM C 989 Standard Specification for Slag Cement for Use in Concrete and Mortars; ASTM International: West Conshohocken, PA, USA, 2004. [Google Scholar]
- Remond, S.; Pimienta, P.; Bentz, D.P. Effects of the incorporation of Municipal Solid Waste Incineration fly ash in cement pastes and mortars I. Experimental study. Cem. Concr. Res. 2002, 32, 303–311. [Google Scholar] [CrossRef]
- Gao, X.; Wang, W.; Ye, T.; Wang, F.; Lan, Y. Utilization of washed MSWI fly ash as partial cement substitute with the addition of dithiocarbamic chelate. J. Environ. Manage. 2008, 88, 293–299. [Google Scholar] [CrossRef] [PubMed]
- Lin, K.L.; Wang, K.S.; Lee, T.Y.; Tzeng, B.Y. The hydration characteristics of MSWI fly ash slag present in C3S. Cem. Concr. Res. 2003, 33, 957–964. [Google Scholar] [CrossRef]
- Lin, K.L.; Wang, K.S.; Tzeng, B.Y.; Wang, N.F.; Lin, C.Y. Effects of Al2O3 on the hydration activity of municipal solid waste incinerator fly ash slag. Cem. Concr. Res. 2004, 34, 587–592. [Google Scholar] [CrossRef]
- Wang, K.; Lin, K.; Huang, Z. Hydraulic activity of municipal solid waste incinerator fly-ash-slag-blended eco-cement. Cem. Concr. Res. 2001, 31, 97–103. [Google Scholar] [CrossRef]
- Lee, T.; Wang, W.; Shih, P.; Lin, K. Enhancement in early strengths of slag-cement mortars by adjusting basicity of the slag prepared from fl y-ash of MSWI. Cem. Concr. Res. 2009, 39, 651–658. [Google Scholar] [CrossRef]
- Lee, T.; Chang, C.; Rao, M.; Su, X. Modified MSWI ash-mix slag for use in cement concrete. Constr. Build. Mater. 2011, 25, 1513–1520. [Google Scholar] [CrossRef]
- Lee, T.; Rao, M. Recycling municipal incinerator fly- and scrubber-ash into fused slag for the substantial replacement of cement in cement-mortars. Waste Manag. 2009, 29, 1952–1959. [Google Scholar] [CrossRef] [PubMed]
- Lin, K.; Lin, D.; Wang, W.; Chang, C.; Lee, T. Pozzolanic reaction of a mortar made with cement and slag vitrified from a MSWI ash-mix and LED sludge. Constr. Build. Mater. 2014, 64, 277–287. [Google Scholar] [CrossRef]
- Boccaccini, A.R.; Kopf, M.; Stumpfe, W. Glass-ceramics from filter dusts from waste incinerators. Ceram. Int. 1995, 21, 231–235. [Google Scholar] [CrossRef]
- Beauchemin, L. Jaarverslag van de Belgische Cementnijverheid; Federatie van de Belgische cementnijverheid vzw: Brussels, Belgium, 2015. [Google Scholar]

| Name | Point of Collection |
|---|---|
| Grate ash | Ash collected from the grate |
| Grate siftings | Material collected from the hoppers underneath the grate |
| Bottom ash | Combined grate ash and grate siftings and sometimes heat recovery ash; it is mainly composed of bottle glass, metals, ceramics and organic residues [25] |
| Heat recovery system ash (HRA) | Ash collected from boiler, economizer and super heater |
| Fly ash | Raw particulate matter entrained in the flue gas stream prior to addition of scrubbing reagents. It is a type of Air Pollution Control residue |
| Air pollution control (APC) residue | All particulate material captured downstream of any reagent injection and prior to discharge of gases to stack; its reuse will be more difficult due to the significant presence of heavy metals and toxic compounds like Polychlorinated dibenzo-p-dioxins (PCDDs) and polychlorinated dibenzofurans (PCDFs) [26] |
| Combined ash | Mixture of bottom ash, grate siftings and APC residues |
| Sintered ash | Bottom ash or fly ash is sometimes sintered and solidified, thus reducing the leaching and facilitating utilization |
| Minerals Identified | Reference | Occurrence and Potential Use |
|---|---|---|
| Quartz (SiO2) | [26,33,34,35,49,51] | It acts as an inert filler when used in cement as SCM. Furthermore, it can have pozzolanic properties when very finely ground. It can be a source of silica when used as a cement raw material. |
| Calcite (CaCO3) | [33,34,35] | It can contribute carbonate to the system, leading to stabilization of ettringite and mono-carbonate/hemi-carbonate when used as an SCM, depending on the content of C3A. The rest of the calcite will act as a filler. Calcite is the commonly-used source of calcium, thus highly beneficial for clinker production. |
| Gehlenite (Ca2Al2SiO7) | [26,33,35,51] | Inert constituent in calcium aluminate cements, carbonatable. |
| Hematite (Fe2O3) | [26,33,34] | Largely inert, formed during incineration. |
| Magnetite | [25,49] | High temperature phase/inert. |
| Ettringite | [49] | Mainly formed by quenching of bottom ash, from the reaction between sulfates and reactive aluminates. |
| Hydrocalumite | [49] | Mainly formed by quenching of bottom ash. |
| Diopside (CaMgSi2O6) | [33] | Principal crystalline phase of sintered ash. |
| Clinoenstatite (MgSiO3) | [33] | Found in sintered ash/ceramics. |
| Wollastonite (CaSiO3) | [33] | Found in sintered ash/ceramics. |
| Ingersonite (γ-Ca2SiO4) | [50] | Reactive towards CO2. |
| Hedenbergite | [25] | Slag/ash component-inert. |
| Ferrohedenbergite | [25] | Slag/ash component-inert. |
| Feldspar | [25] | Common inert rock-forming mineral. |
| Melilite (Ca,Na)2(Al,Mg,Fe2+)[(Al,Si)SiO7] | [25,49] | Contains Mg; carbonatable. |
| Albite (NaAlSi3O8) | [33,34] | Found in sintered ash/ceramics. |
| Anorthite (CaAl2Si2O8) | [35,47,51] | Common inert rock-forming mineral. |
| Anhydrite (CaSO4) | [26,34,51] | Cement constituent, added to control setting. |
| Gypsum (CaSO4·2H2O) | [36,49] | Cement constituent, added to control setting. |
| Gismondine (CaAl2Si2O8·4H2O) | [50] | |
| Apatite(Ca5(PO4)3(OH,F,Cl)) | [26] | Fly ash treated by washing, phosphation and calcination to 750 °C. Bone fragments can also be a source of apatite in ash. |
| Whitlockite (β-Ca3(PO4)2) | [26] | Fly ash treated by washing, phosphation and calcination to 750 °C. |
| Titanite (CaTiSiO5) | [26] | Fly ash treated by washing, phosphation and calcination to 750 °C. |
| Perovskite (CaTiO3) | [26,52] | Inert. |
| Periclase | [25] | Carbonatable. |
| Aluminum in Ash (%) | Theoretical Volume of Hydrogen Produced in 1 m3 of Concrete (Assuming 25% Replacement of Cement by Ash and 450 kg/m3 of Cement Content) in m3; cf. Equation (2) at STP (Standard Temperature and Pressure) |
|---|---|
| 0.1 | 0.150259 |
| 1 | 1.502592 |
| Parameter | Flanders Criteria (VLAREMA) Total Concentration Limit mg/kg Dry Matter | Wallonia Criteria Total Concentration Limit mg/kg Dry Matter | Bottom Ash Total Typical Concentration mg/kg Dry Matter |
|---|---|---|---|
| Arsenic | 250 | 100 | 33 ± 17 |
| Cadmium | 10 | 8 | |
| Chromium | 1250 | 230 | 482 ± 73 |
| Copper | 375 | 210 | 4042 ± 888 |
| Mercury | 5 | 15 | 3 ± 2 |
| Lead | 1250 | 1150 | 1899 ± 396 |
| Nickel | 250 | 150 | 329 ± 69 |
| Zinc | 1250 | 680 | 5376 ± 782 |
| Obstacle | Pre-Treatment Technique | Advantages | Disadvantages |
|---|---|---|---|
| Metallic Aluminum and Zinc | Magnetic density separation | Versatile Simple | Initial cost, not for fines |
| Eddy current separation | Can detect through several layers Can provide an accurate separation | Initial cost, not for fines Susceptible to magnetic permeability changes | |
| Wet grinding | Consumes lower power per ton of product. Enables the use of wet screening or classification for close product control. Eliminates the dust problem. Enables the use of simple handling and transport methods such as pumps, pipes and launders. | Storage of wet slurries | |
| Washing with alkali | Simple | Cost of alkali | |
| Salts | Washing with water | Simple | Secondary pollution of water, unless the water in the slurry is used to make concrete from the material |
| Carbonation | CO2 from stack gas can be utilized and thus reduce the emission. Can be a method for carbon sequestration. | Not a very fast process, unless the CO2 concentration is very high, which in turn will require air tight enclosures | |
| Thermal treatment | Simple technology | Energy Consumption Cost | |
| Heavy metals | Washing with water | Simple | Secondary pollution of water |
| Treatment with sulfide rich effluent | Simple and can utilize another waste stream | Applicable for specific heavy metals | |
| Wet grinding | Simple No addition of additional chemicals | Storage of wet slurries | |
| Phosphation | Stabilizes heavy metals | Applicable only for specific applications | |
| Cement stabilization | Stabilizes heavy metals | Cost of cement | |
| Hydrothermal treatment | Capital cost | ||
| Electrodialytic remediation | Costly Energy Consumption |
| Categories | Cement Consumption in 2015 (MT) |
|---|---|
| Concrete products and fiber cement | 1 |
| Ready mix concrete | 2.767 |
| Delivery in construction site | 0.743 |
| Delivery in hardware shops | 0.381 |
| Total consumption | 4.891 |
| Import | 1.513 |
| Export | 1.384 |
| Cement Type | Strength Class | % Consumption |
|---|---|---|
| CEM I, II, V | 32.5 | 12 |
| 42.5 | 3 | |
| 52.5 | 26 | |
| CEM III | 32.5 | 8 |
| 42.5/52.5 | 51 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Joseph, A.M.; Snellings, R.; Van den Heede, P.; Matthys, S.; De Belie, N. The Use of Municipal Solid Waste Incineration Ash in Various Building Materials: A Belgian Point of View. Materials 2018, 11, 141. https://doi.org/10.3390/ma11010141
Joseph AM, Snellings R, Van den Heede P, Matthys S, De Belie N. The Use of Municipal Solid Waste Incineration Ash in Various Building Materials: A Belgian Point of View. Materials. 2018; 11(1):141. https://doi.org/10.3390/ma11010141
Chicago/Turabian StyleJoseph, Aneeta Mary, Ruben Snellings, Philip Van den Heede, Stijn Matthys, and Nele De Belie. 2018. "The Use of Municipal Solid Waste Incineration Ash in Various Building Materials: A Belgian Point of View" Materials 11, no. 1: 141. https://doi.org/10.3390/ma11010141
APA StyleJoseph, A. M., Snellings, R., Van den Heede, P., Matthys, S., & De Belie, N. (2018). The Use of Municipal Solid Waste Incineration Ash in Various Building Materials: A Belgian Point of View. Materials, 11(1), 141. https://doi.org/10.3390/ma11010141







