The Effect of Hydrogen Bonding in Enhancing the Ionic Affinities of Immobilized Monoprotic Phosphate Ligands
Abstract
:1. Introduction
2. Experimental
2.1. Synthesis
2.1.1. Synthesis of Diprotic Phosphonic Acid (DPA) Polymer
2.1.2. Synthesis of Phosphorylated Ethylene Glycol Monoprotic (pEG1M) Polymer
2.1.3. Synthesis of Phosphorylated Glycerol Monoprotic (pGlyM) Polymer
2.1.4. Synthesis of Phosphorylated Pentaerythritol Monoprotic (pPentaM)
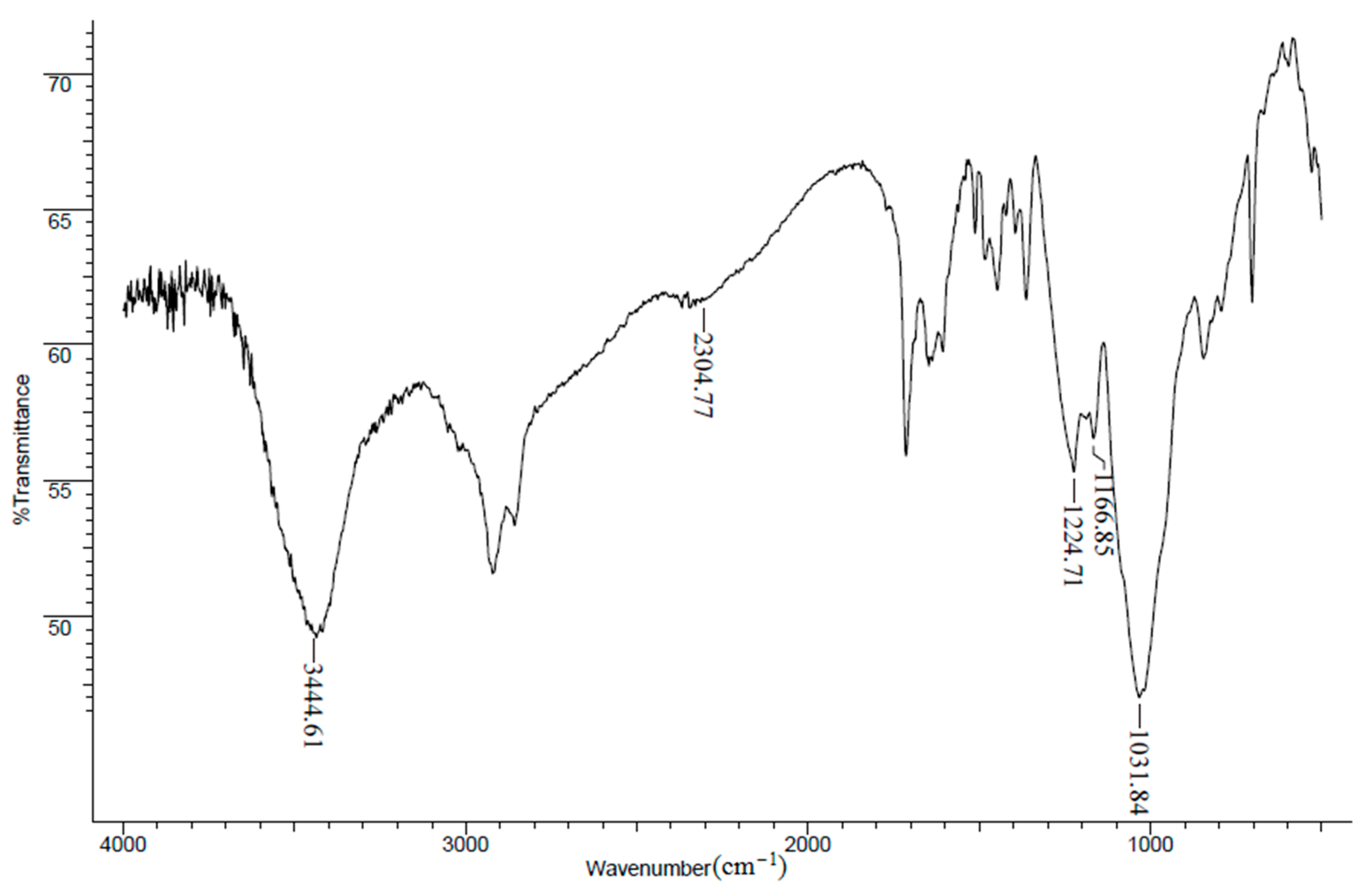
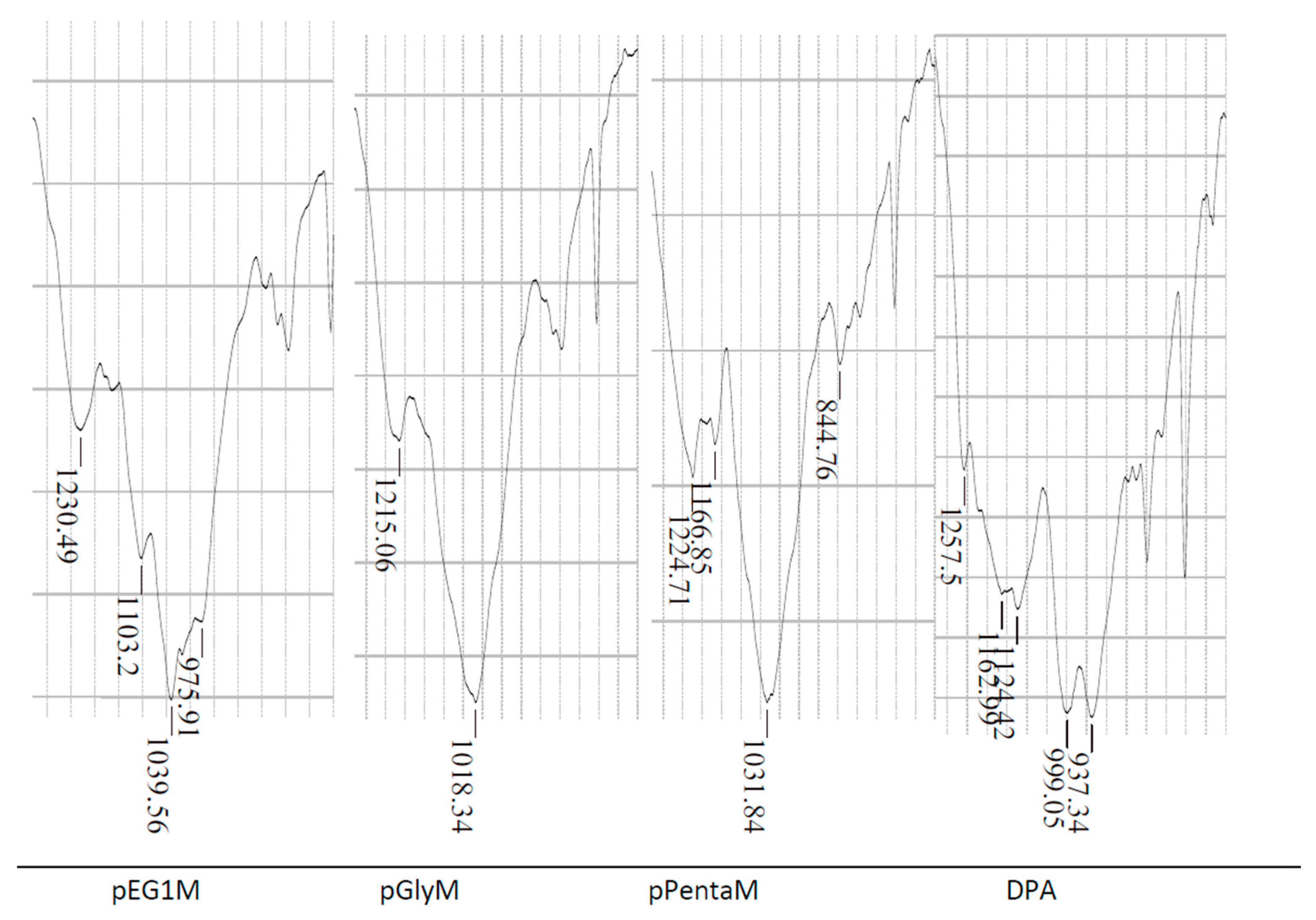
2.2. Characterization
2.3. Metal Ion Affinities
3. Results
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Zawierucha, I.; Kozlowski, C.; Malina, G. Immobilized materials for removal of toxic metal ions from surface/groundwaters and aqueous waste streams. Environ. Sci. Process. Impacts 2016, 18, 429–444. [Google Scholar] [CrossRef] [PubMed]
- Bruzzoniti, C.M.; Sarzanini, C.; Mentasti, E. Preconcentration of contaminants in water analysis. J. Chromatogr. A 2000, 902, 289–309. [Google Scholar] [CrossRef]
- Yam, V.W.W.; Lo, K.K.W. Recent advances in utilization of transition metal complexes and lanthanides as diagnostic tools. Coord. Chem. Rev. 1999, 184, 157–240. [Google Scholar] [CrossRef]
- Krea, M.; Khalaf, H. Liquid–liquid extraction of uranium and lanthanides from phosphoric acid using a synergistic DOPPA–TOPO mixture. Hydrometallurgy 2000, 58, 215–225. [Google Scholar] [CrossRef]
- Sessler, J.L.; Melfi, P.J.; Pantos, G.D. Uranium complexes of multidentate N-donor ligands. Coord. Chem. Rev. 2006, 250, 816–843. [Google Scholar] [CrossRef]
- Sasaki, T.; Kubo, S.; Kubota, T.; Takagi, I.; Moriyama, H. Complex formation of lanthanides(III) and actinides(III) with dicarboxylates containing soft donor groups. J. Alloys Compd. 2006, 408–412, 1283–1286. [Google Scholar] [CrossRef]
- Jensen, M.P.; Morss, L.R.; Beitz, J.V.; Ensor, D.D. Aqueous complexation of trivalent lanthanide and actinide cations by N,N,N′,N′-tetrakis(2-pyridylmethyl) ethylenediamine. J. Alloys Compd. 2000, 24, 137–141. [Google Scholar] [CrossRef]
- Atanassova, M.; Kurteva, V. Synergism as a phenomenon in solvent extraction of 4f-elements with calixarenes. RSC Adv. 2016, 6, 11303–11324. [Google Scholar] [CrossRef]
- Alexandratos, S.D. New polymer-supported ion-complexing agents: Design, preparation and metal ion affinities of immobilized ligands. J. Hazard. Mater. 2007, A139, 467–470. [Google Scholar] [CrossRef] [PubMed]
- Alexandratos, S.D.; Zhu, X. Bifunctional coordinating polymers: Auxiliary groups as a means of tuning the ionic affinity of immobilized phosphate ligands. Macromolecules 2005, 38, 5981–5986. [Google Scholar] [CrossRef]
- Alexandratos, S.D.; Zhu, X. The role of polarizability in determining metal ion affinities in polymer-supported reagents: Monoprotic phosphates and the effect of hydrogen bonding. New J. Chem. 2015, 39, 5366–5373. [Google Scholar] [CrossRef]
- Alexandratos, S.D.; Zhu, X. Polyols as scaffolds in the development of ion-selective polymer-supported reagents: The effect of auxiliary groups on the mechanism of metal ion complexation. Inorg. Chem. 2008, 47, 2831–2836. [Google Scholar] [CrossRef] [PubMed]
- Ober, R.D. Synthesis and Development of Selective Ion Exchange Resins for the Removal of Toxic Metal Ions from Water in the Environment. Ph.D. Thesis, University of Tennessee, Knoxville, TN, USA, March 1999. [Google Scholar]
- Marquardt, F.; Keul, H.; Moeller, M. Straightforward synthesis of phosphate functionalized linear polyglycidol. Eur. Polym. J. 2015, 69, 319–327. [Google Scholar] [CrossRef]
- Theisen, P.D.; Heathcock, C.H. Improved procedure for preparation of optically active 3-hydroxyglutarate monoesters and 3-hydroxy-5-oxoalkanoic acids. J. Organ. Chem. 1988, 53, 2374–2378. [Google Scholar] [CrossRef]
- Misono, M.; Ochiai, E.; Saito, Y.; Yoneda, Y. A new dual parameter scale for the strength of Lewis acids and bases with the evaluation of their softness. J. Inorg. Nucl. Chem. 1967, 29, 2685–2691. [Google Scholar] [CrossRef]
- Jyo, A.; Zhu, X. Metal ion selectivity of phosphoric acid resin in aqueous nitric acid media. In Chemistry for the Protection of the Environment; Pawlowski, L., Gonzales, M.A., Dudzinska, M.R., Lacy, W.J., Eds.; Plenum Press: New York, NY, USA, 1998; Volume 3, pp. 135–142. [Google Scholar]
- Yamabe, K.; Ihara, T.; Jyo, A. Metal ion selectivity of marcroreticular chelating cation exchange resins with phosphonic acid groups attached to phenyl groups of a styrene-divinylbenzene copolymer matrix. Sep. Sci. Technol. 2001, 36, 3511–3528. [Google Scholar]
- Spectral Database for Organic Compounds SDBS. Available online: http://riodb01.ibase.aist.go.jp/sdbs/ (accessed on 1 June 2017).
- Nagle, R.A.; Murthy, T.K.S. Synergistic effects in the extraction of uranium(VI) by di-4-octylphenyl phosphoric acid. Sep. Sci. Technol. 1979, 14, 499–514. [Google Scholar] [CrossRef]
- Oroujzadeh, N.; Gholivand, K.; Jamalabadi, N.R. New carbacylamidophosphates containing nicotinamide: Synthesis, crystallography and antibacterial activity. Polyhedron 2017, 122, 29–38. [Google Scholar] [CrossRef]
- Gramstad, T. Study of hydrogen bonding. X. The hydrogen-bonding ability of β-dimethyl-aminoethyl diethyl phosphate. Spectrochim. Acta 1963, 19, 1391–1392. [Google Scholar] [CrossRef]
- Herlinger, A.W.; Ferraro, J.R.; Chiarizia, R.; Horwitz, E.P. An investigation of P,P’-di(2-ethylhexyl) methanediphosphonic acid and some of its metal complexes. Polyhedron 1997, 16, 1843–1854. [Google Scholar] [CrossRef]
- Wu, J.; Hou, H.; Han, H.; Fan, Y. Highly selective ferric ion sorption and exchange by crystalline metal phosphonates constructed from tetraphosphonic acids. Inorg. Chem. 2007, 46, 7960–7970. [Google Scholar] [CrossRef] [PubMed]
- Alexandratos, S.D.; Zhu, X. Recovery of Uranium from Seawater: Modified Polyacrylonitrile Fibers as Selective Extractants; Technical Report for Nuclear Energy University Programs: Washington, DC, USA, March 2017. [Google Scholar]
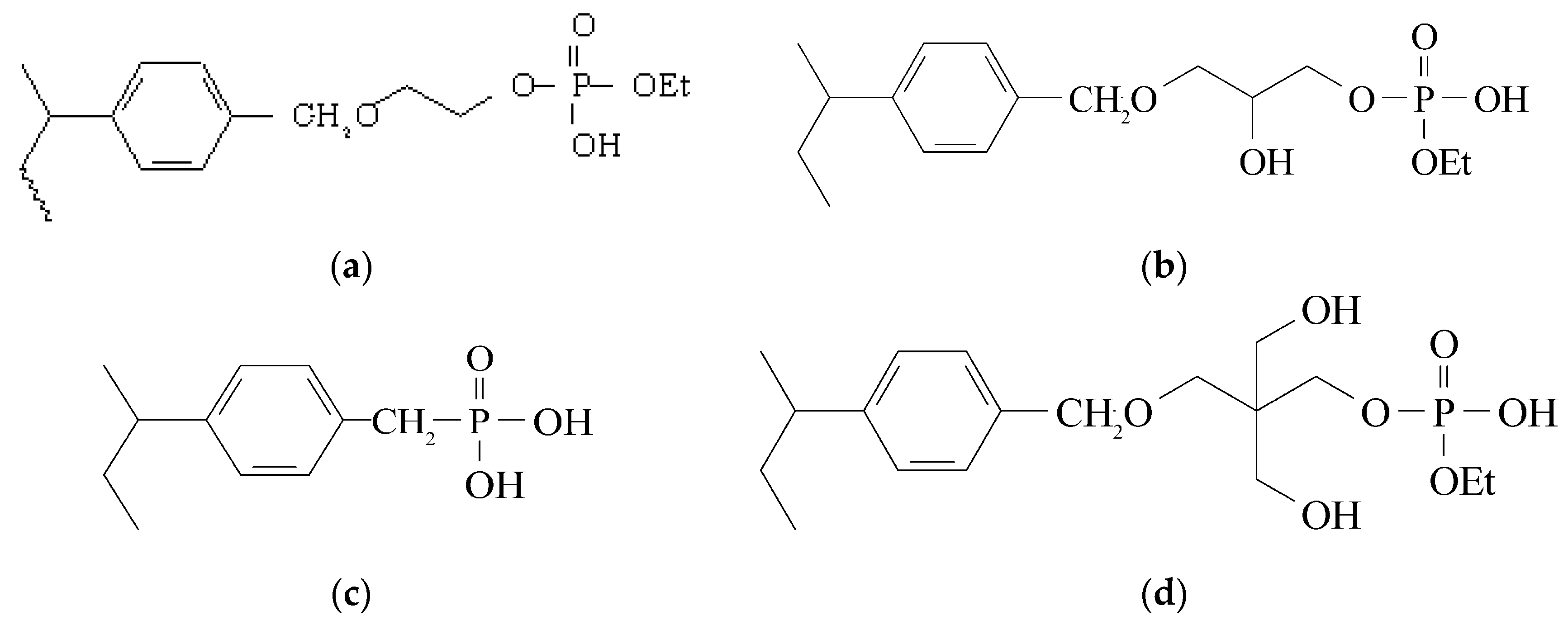
| Resin | Acid Capacity (mmol/g) | Phosphorus Capacity (mmol/g) | Acid/Phosphorus |
|---|---|---|---|
| DPA | 9.65 | 4.71 | 2.05 |
| pEG1M | 2.23 | 2.82 | 0.79 |
| pGlyM | 2.97 | 2.85 | 1.04 |
| pPentaM | 2.61 | 2.81 | 0.93 |
| pEG1M | ||||
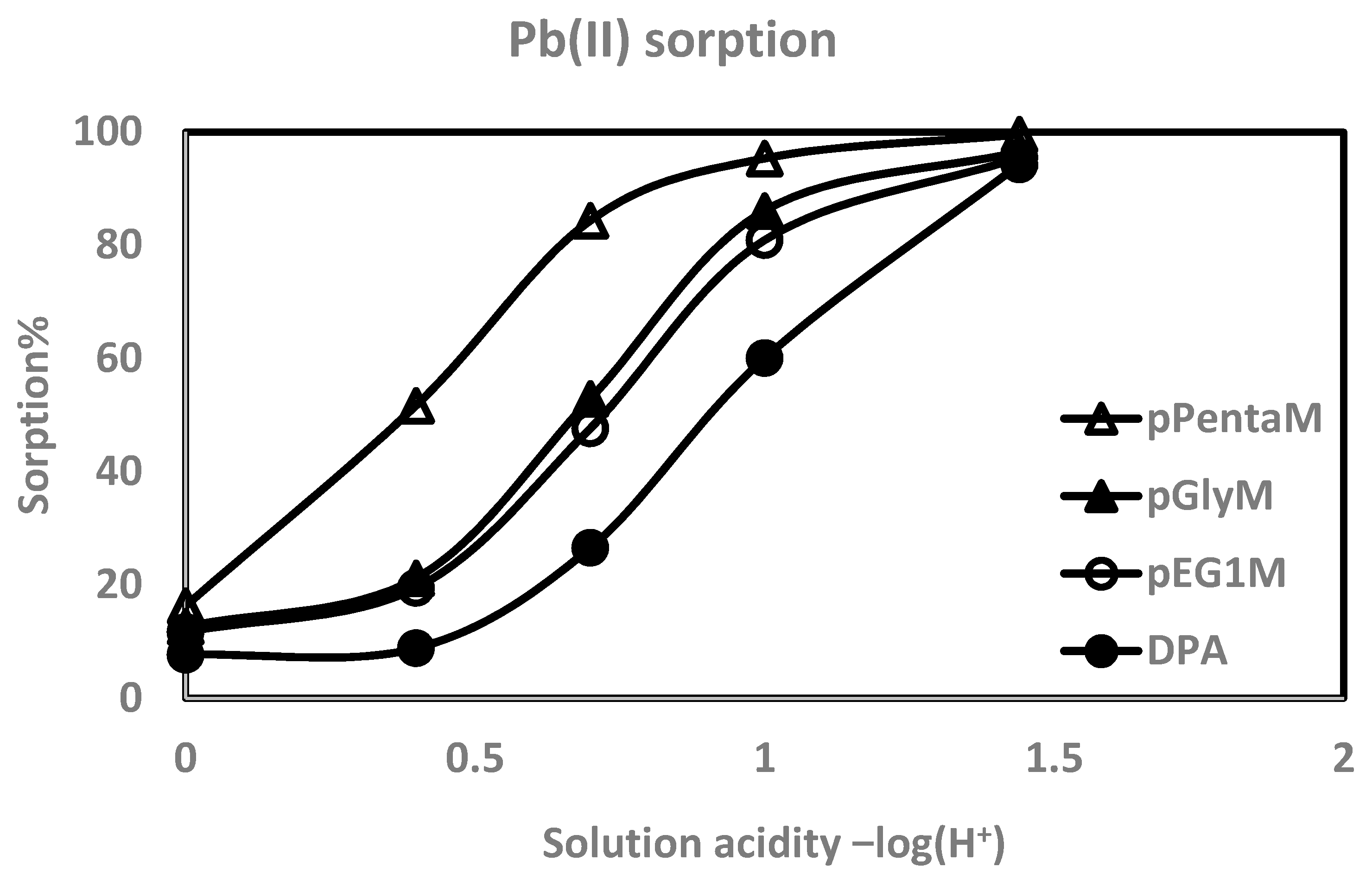
| pH | %Pb(II) abs (log D) * | %Cd(II) abs (log D) | %Cu(II) abs (log D) | %Zn(II) abs (log D) |
| 1.41 | 95.5% (2.77) | 83.1% (2.15) | 82.7% (2.14) | 62.2% (1.68) |
| 1.00 | 80.9 (2.07) | 54.7 (1.54) | 53.6 (1.51) | 23.6 (0.949) |
| 0.699 | 47.7 (1.40) | 36.6 (1.22) | 24.6 (0.976) | 10.8 (0.549) |
| 0.398 | 19.5 (0.841) | 11.8 (0.587) | 8.15 (0.398) | 0.990 (−0.545) |
| 0.00 | 11.7 (0.567) | 8.95 (0.448) | 4.60 (0.145) | - |
| pGlyM | ||||
| pH | %Pb(II) abs (log D) | %Cd(II) abs (log D) | %Cu(II) abs (log D) | %Zn(II) abs (log D) |
| 1.41 | 96.5 (2.91) | 93.2 (2.60) | 88.5 (2.35) | 80.7 (2.08) |
| 1.00 | 86.1 (2.25) | 75.4 (1.96) | 68.0 (1.79) | 51.8 (1.49) |
| 0.699 | 52.9 (1.53) | 43.4 (1.35) | 32.1 (1.15) | 17.3 (0.775) |
| 0.398 | 21.2 (0.893) | 17.2 (0.786) | 14.4 (0.683) | 9.27 (0.483) |
| 0.00 | 12.7 (0.638) | 8.14 (0.418) | 7.56 (0.389) | - |
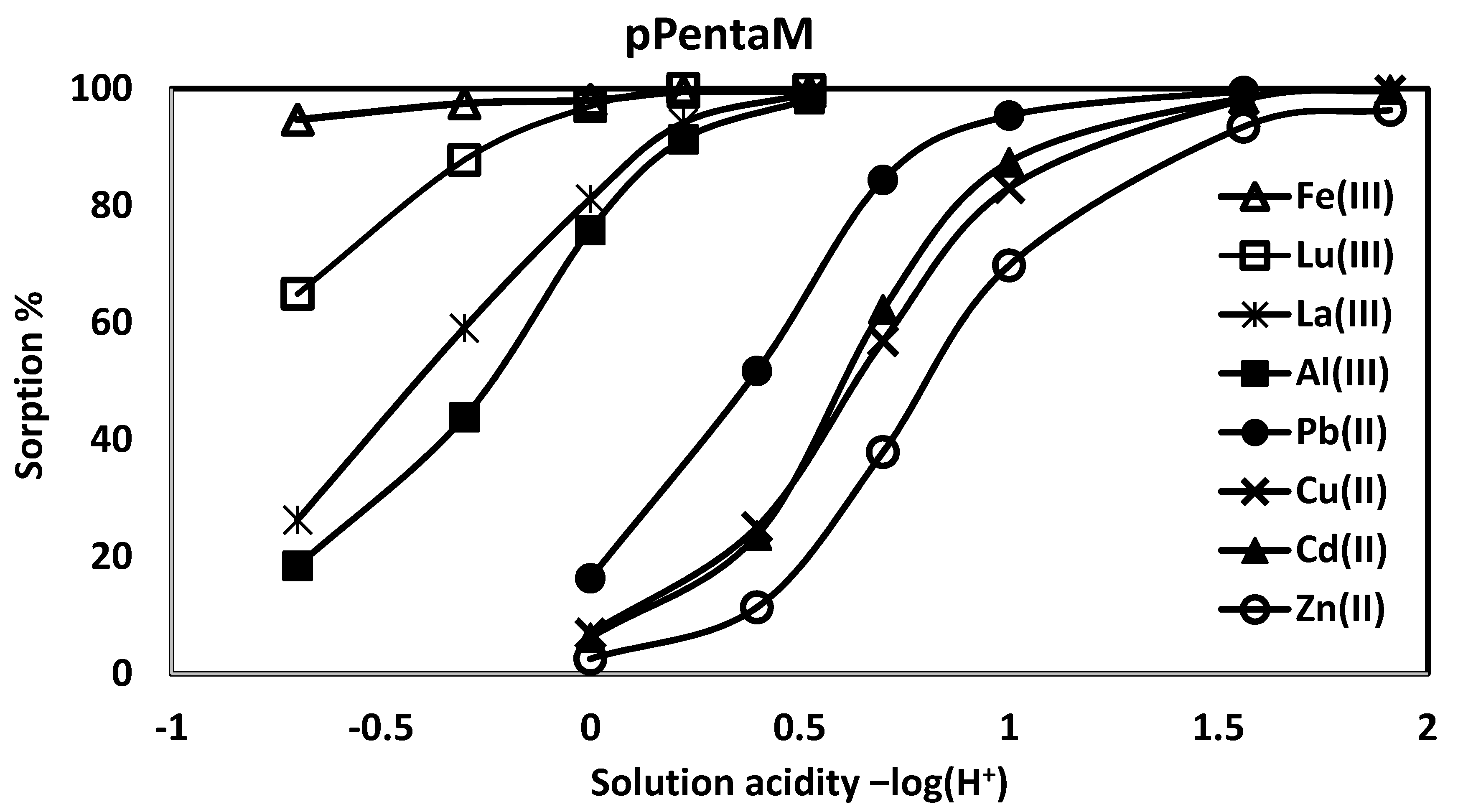
| pPentaM | ||||
| pH | %Pb(II) abs (log D) | %Cd(II) abs (log D) | %Cu(II) abs(log D) | %Zn(II) abs (log D) |
| 1.56 | 99.6 (3.84) | 98.4 (3.26) | 98.1 (3.17) | 93.5 (2.63) |
| 1.00 | 95.4 (2.79) | 87.4 (2.30) | 83.0 (2.16) | 69.8 (1.82) |
| 0.699 | 84.4 (2.21) | 62.3 (1.68) | 56.9 (1.60) | 37.9 (1.24) |
| 0.398 | 51.7 (1.51) | 23.7 (0.955) | 25.1 (1.01) | 11.4 (0.562) |
| 0.00 | 16.3 (0.762) | 6.18 (0.292) | 6.87 (0.332) | 2.55 (−0.112) |
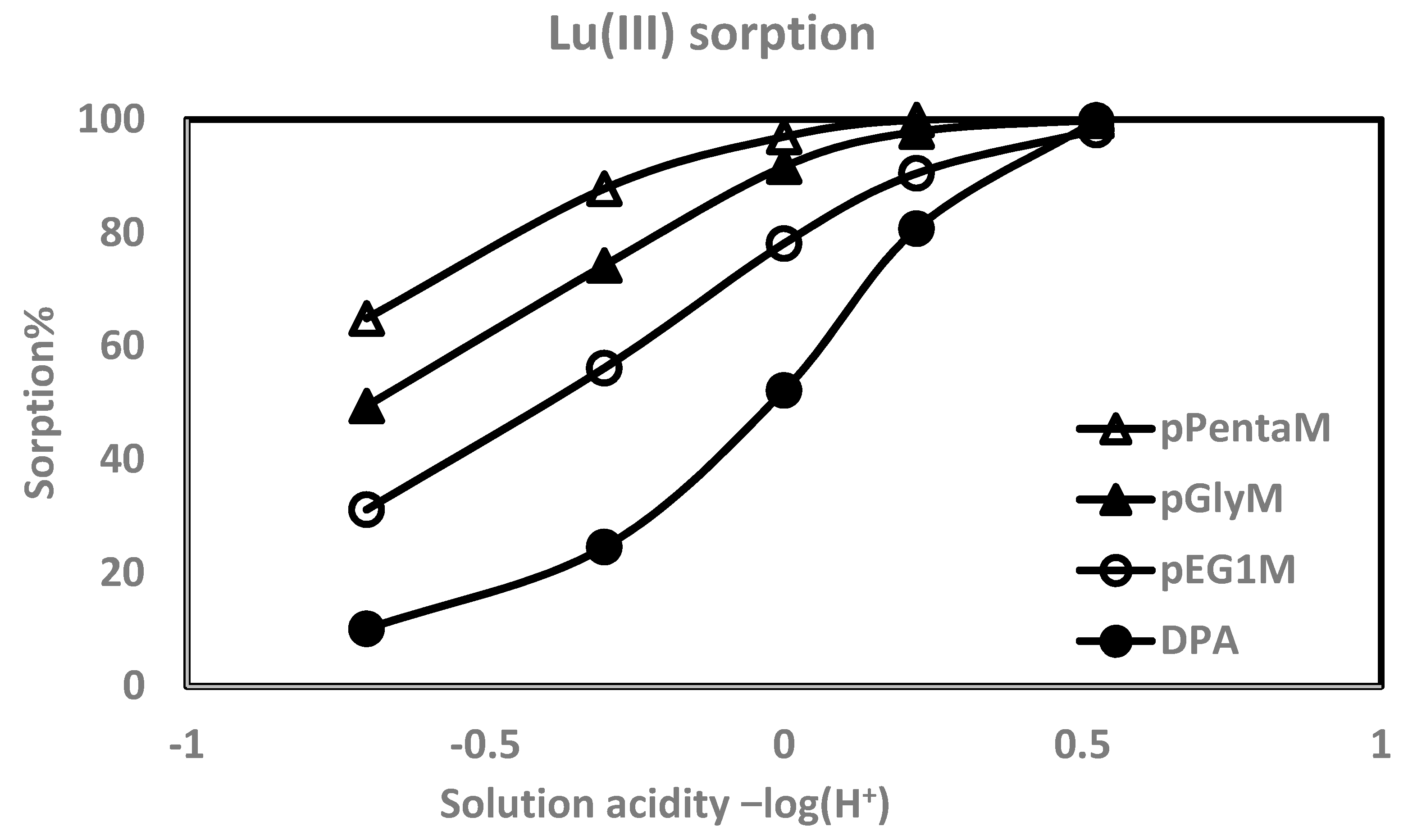
| pEG1M | ||||
| pH | %Fe(III) abs (log D) | %Al(III) abs (log D) | %La(III) abs (log D) | %Lu(III) abs (log D) |
| 0.523 | 91.0 (2.46) | 85.5 (2.22) | 98.7 (3.33) | 98.4 (3.17) |
| 0.222 | 89.4 (2.37) | 69.0 (1.79) | 88.6 (2.34) | 90.5 (2.43) |
| 0.00 | 87.6 (2.32) | 38.8 (1.27) | 78.6 (1.99) | 78.1 (2.00) |
| −0.301 | 80.1 (2.07) | 14.9 (0.692) | 46.7 (1.38) | 56.1 (1.56) |
| −0.699 | 60.3 (1.62) | 5.09 (0.183) | 16.4 (0.708) | 31.2 (1.10) |
| pGlyM | ||||
| pH | %Fe(III) abs (log D) | %Al(III) abs (log D) | %La(III) abs (log D) | %Lu(III) abs (log D) |
| 0.523 | 99.1 (3.50) | 98.6 (3.31) | 99.6 (3.85) | 99.9 (4.72) |
| 0.222 | 98.7 (3.36) | 92.6 (2.56) | 95.3 (2.77) | 97.9 (3.13) |
| 0.00 | 98.5 (3.27) | 76.0 (1.96) | 90.1 (2.46) | 91.7 (2.52) |
| −0.301 | 95.3 (1.77) | 36.4 (1.23) | 59.7 (1.67) | 74.3 (1.93) |
| −0.699 | 89.3 (2.39) | 15.5 (0.733) | 23.7 (0.972) | 49.5 (1.47) |
| pPentaM | ||||
| pH | %Fe(III) abs (log D) | %Al(III) abs (log D) | %La(III) abs (log D) | %Lu(III) abs (log D) |
| 0.523 | 99.4 (3.71) | 98.2 (3.21) | 99.1 (3.53) | 99.8 (4.11) |
| 0.222 | 99.4 (3.71) | 91.3 (2.49) | 94.2 (2.67) | 99.9 (3.63) |
| 0.00 | 98.0 (3.15) | 75.8 (1.97) | 81.2 (2.12) | 97.0 (2.96) |
| −0.301 | 97.5 (3.04) | 43.8 (1.37) | 59.1 (1.63) | 87.8 (2.31) |
| −0.699 | 94.7 (2.72) | 18.4 (0.821) | 26.3 (1.00) | 64.9 (1.75) |
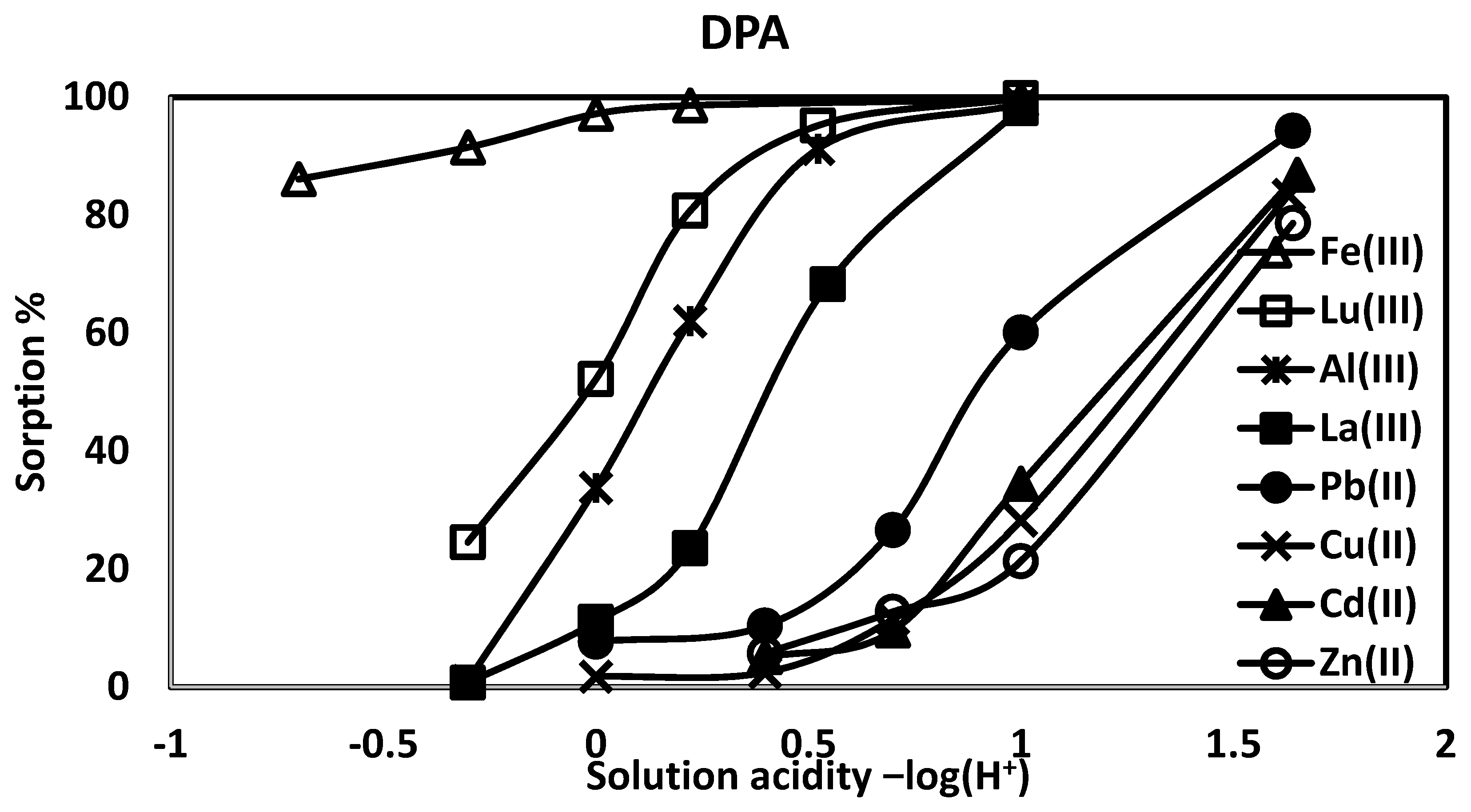
| pH | %Pb(II) abs (log D) | %Cd(II) abs (log D) | %Cu(II) abs (log D) | %Zn abs (II) (log D) |
| 1.64 | 94.3 (2.84) | 86.8 (2.44) | 83.4 (2.35) | 78.6 (2.17) |
| 1.00 | 60.1 (1.77) | 34.5 (1.35) | 28.2 (1.25) | 21.4 (1.09) |
| 0.699 | 26.6 (1.16) | 9.58 (0.642) | 11.5 (0.74) | 12.8 (0.508) |
| 0.398 | 10.4 (0.595) | 5.05 (0.355) | 2.48 (0.0658) | 5.71 (0.412) |
| 0.00 | 7.71 (0.535) | - | 1.79 (−0.116) | - |
| pH | %Fe(III) abs (log D) | %Al(III) abs (log D) | %La(III) abs (log D) | %Lu abs (III) (log D) |
| 1.00 | 99.6 (3.99) | 98.8 (3.55) | 98.1 (3.34) | 99.8 (4.34) |
| 0.523 | - | 91.2 (2.64) | 68.3 (1.95) | 95.2 (2.95) |
| 0.222 | 98.6 (3.49) | 62.0 (1.86) | 23.5 (1.12) | 80.7 (2.27) |
| 0.00 | 97.2 (3.19) | 33.8 (1.36) | 11.1 (0.724) | 52.2 (1.68) |
| −0.301 | 91.5 (2.67) | 1.05 (0.271) | 0.74 (−0.48) | 24.5 (1.17) |
| −0.699 | 86.1 (2.41) | - | - | - |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alexandratos, S.D.; Zhu, X. The Effect of Hydrogen Bonding in Enhancing the Ionic Affinities of Immobilized Monoprotic Phosphate Ligands. Materials 2017, 10, 968. https://doi.org/10.3390/ma10080968
Alexandratos SD, Zhu X. The Effect of Hydrogen Bonding in Enhancing the Ionic Affinities of Immobilized Monoprotic Phosphate Ligands. Materials. 2017; 10(8):968. https://doi.org/10.3390/ma10080968
Chicago/Turabian StyleAlexandratos, Spiro D., and Xiaoping Zhu. 2017. "The Effect of Hydrogen Bonding in Enhancing the Ionic Affinities of Immobilized Monoprotic Phosphate Ligands" Materials 10, no. 8: 968. https://doi.org/10.3390/ma10080968
APA StyleAlexandratos, S. D., & Zhu, X. (2017). The Effect of Hydrogen Bonding in Enhancing the Ionic Affinities of Immobilized Monoprotic Phosphate Ligands. Materials, 10(8), 968. https://doi.org/10.3390/ma10080968




