Effects of Bi Addition on the Microstructure and Mechanical Properties of Nanocrystalline Ag Coatings
Abstract
1. Introduction
2. Materials and Methods
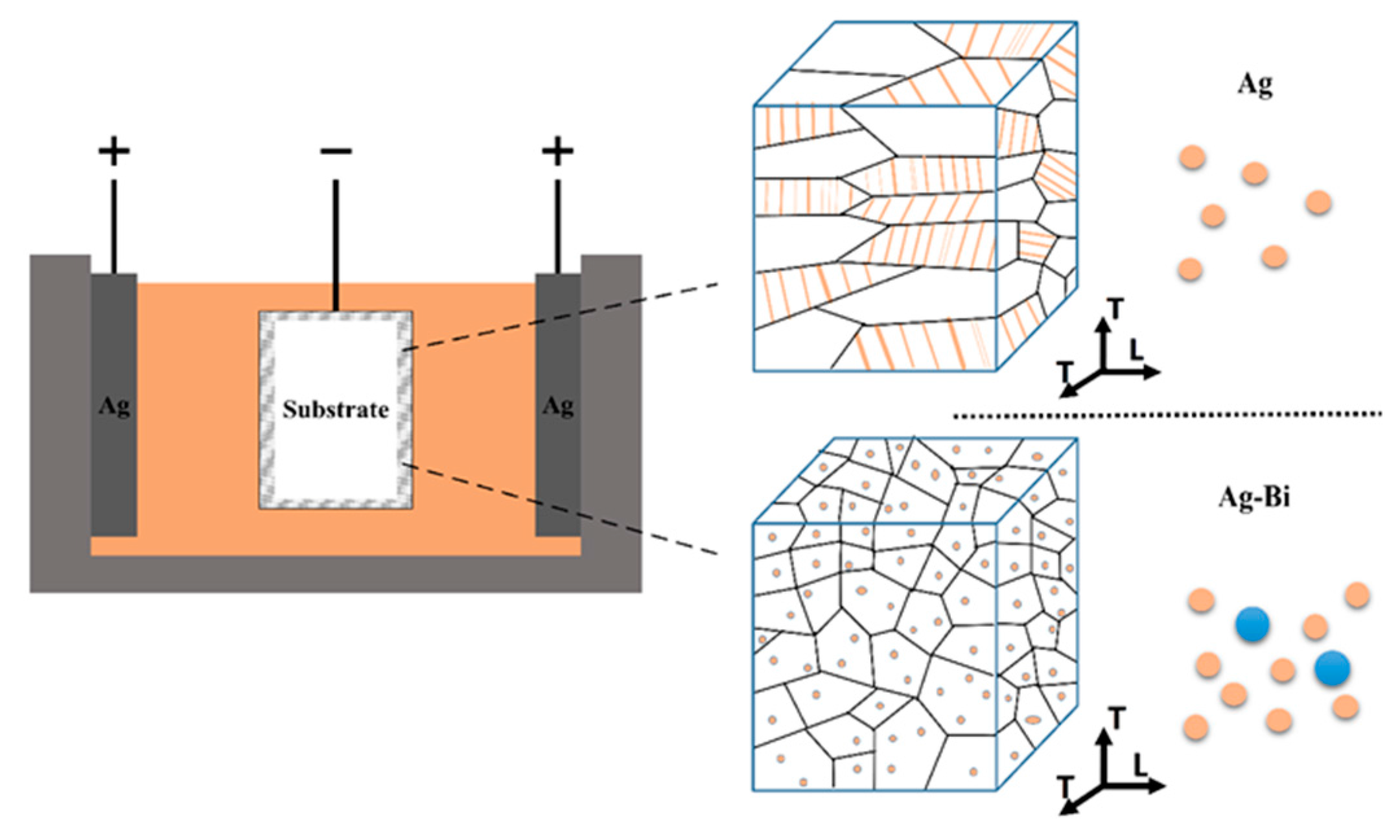
2.1. Sample Preparation and Characterization
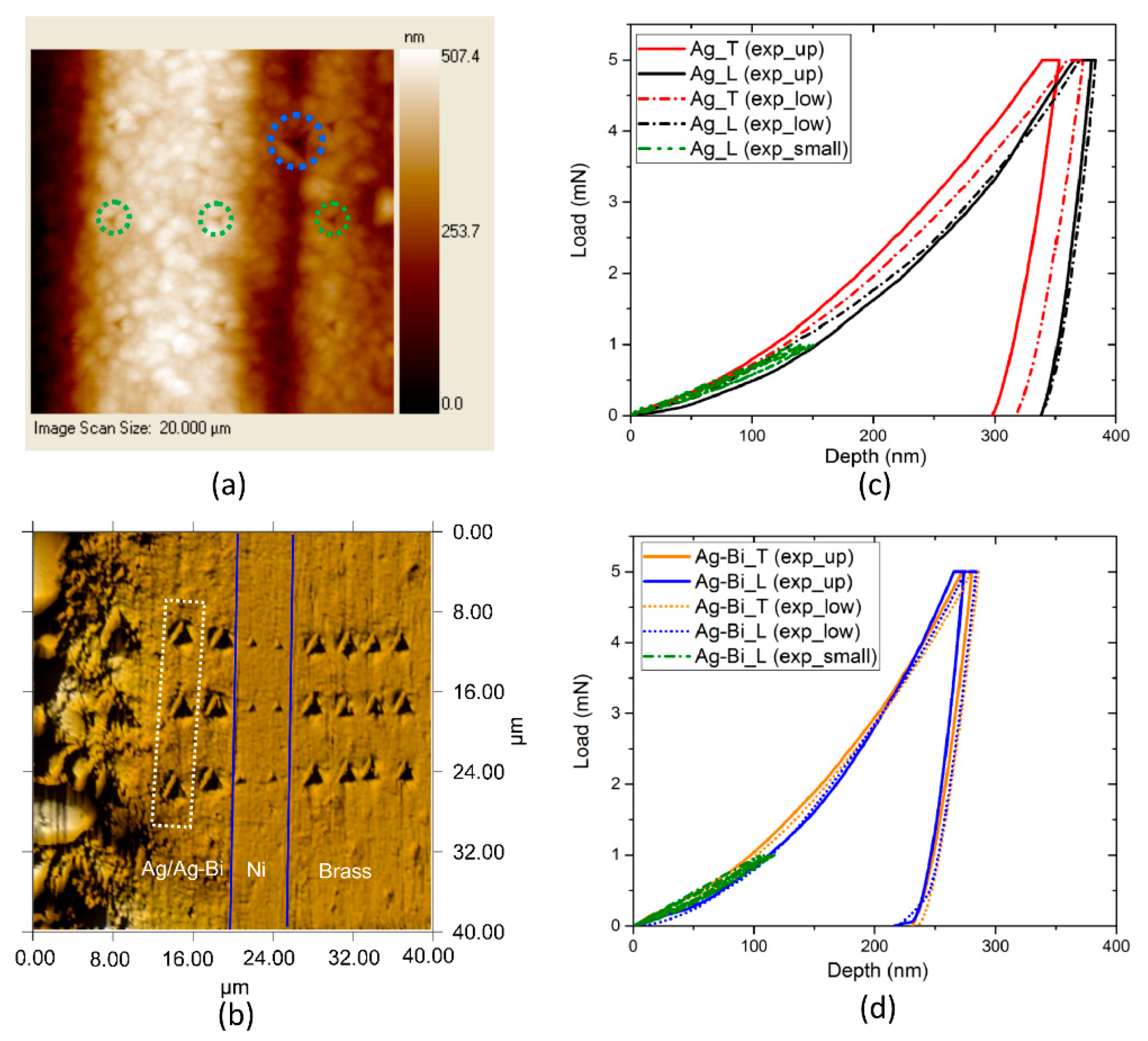
2.2. Nanoindentation
3. Results
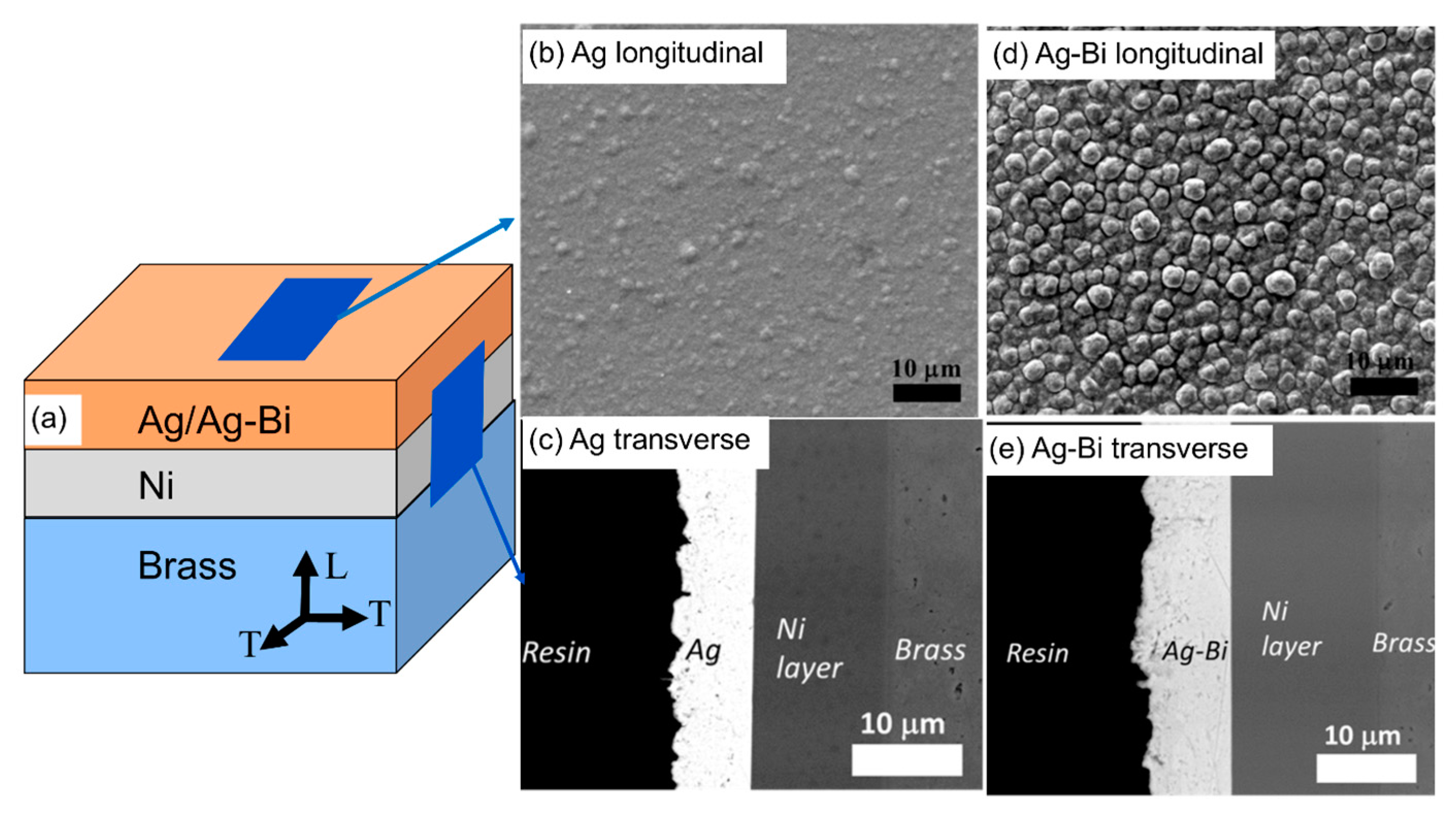
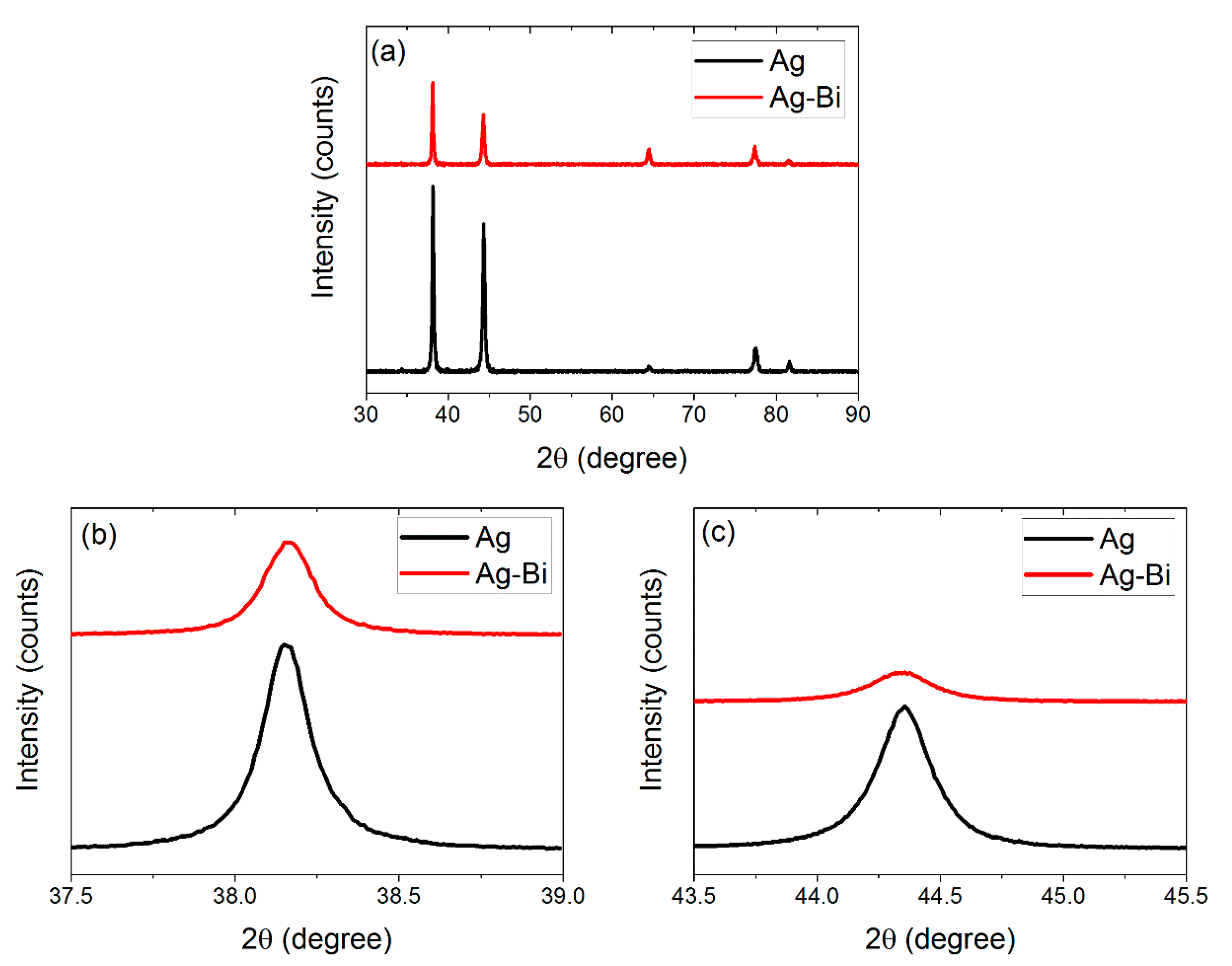
3.1. Microstructure of the Ag and Ag-Bi Coatings
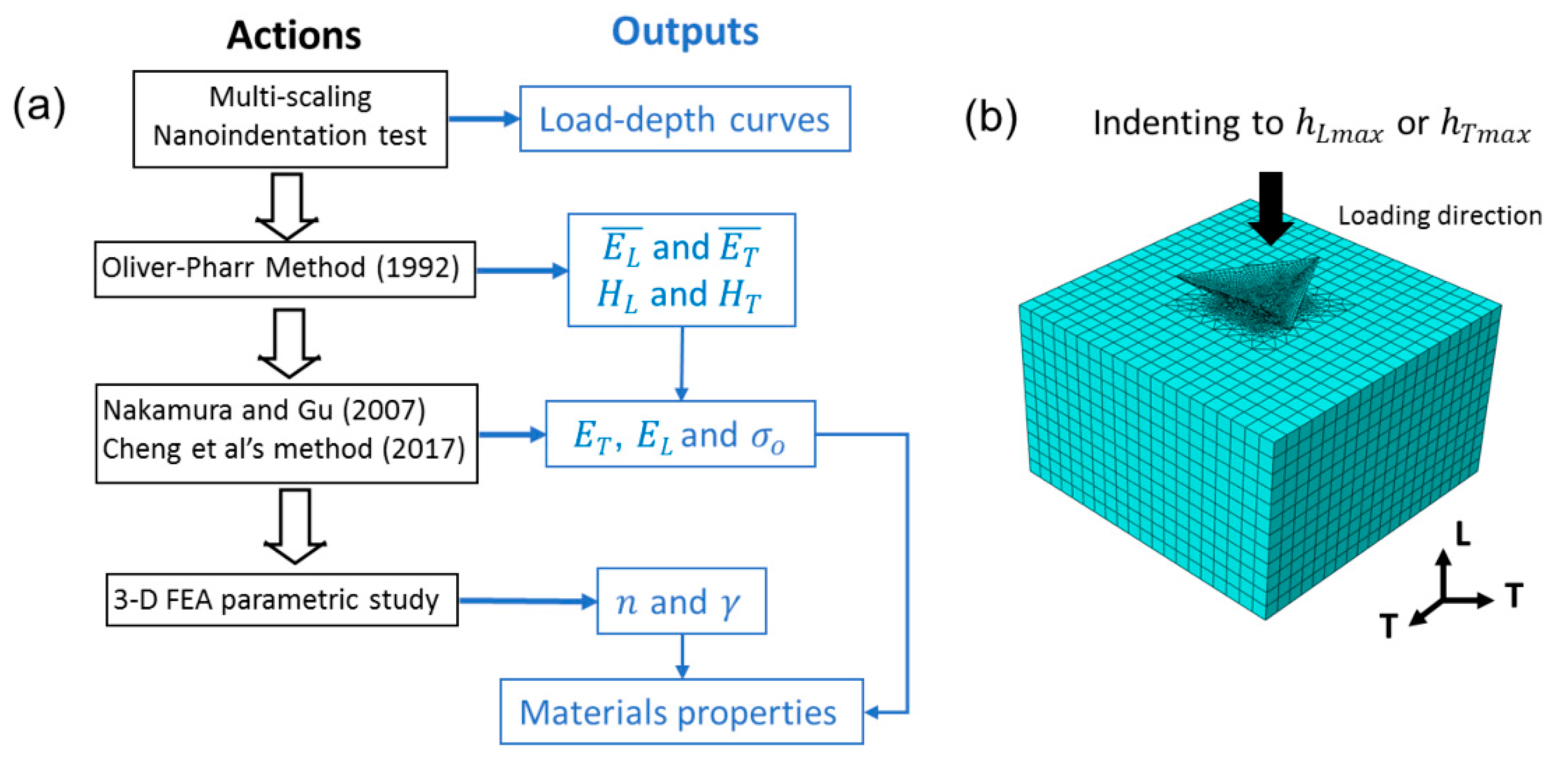
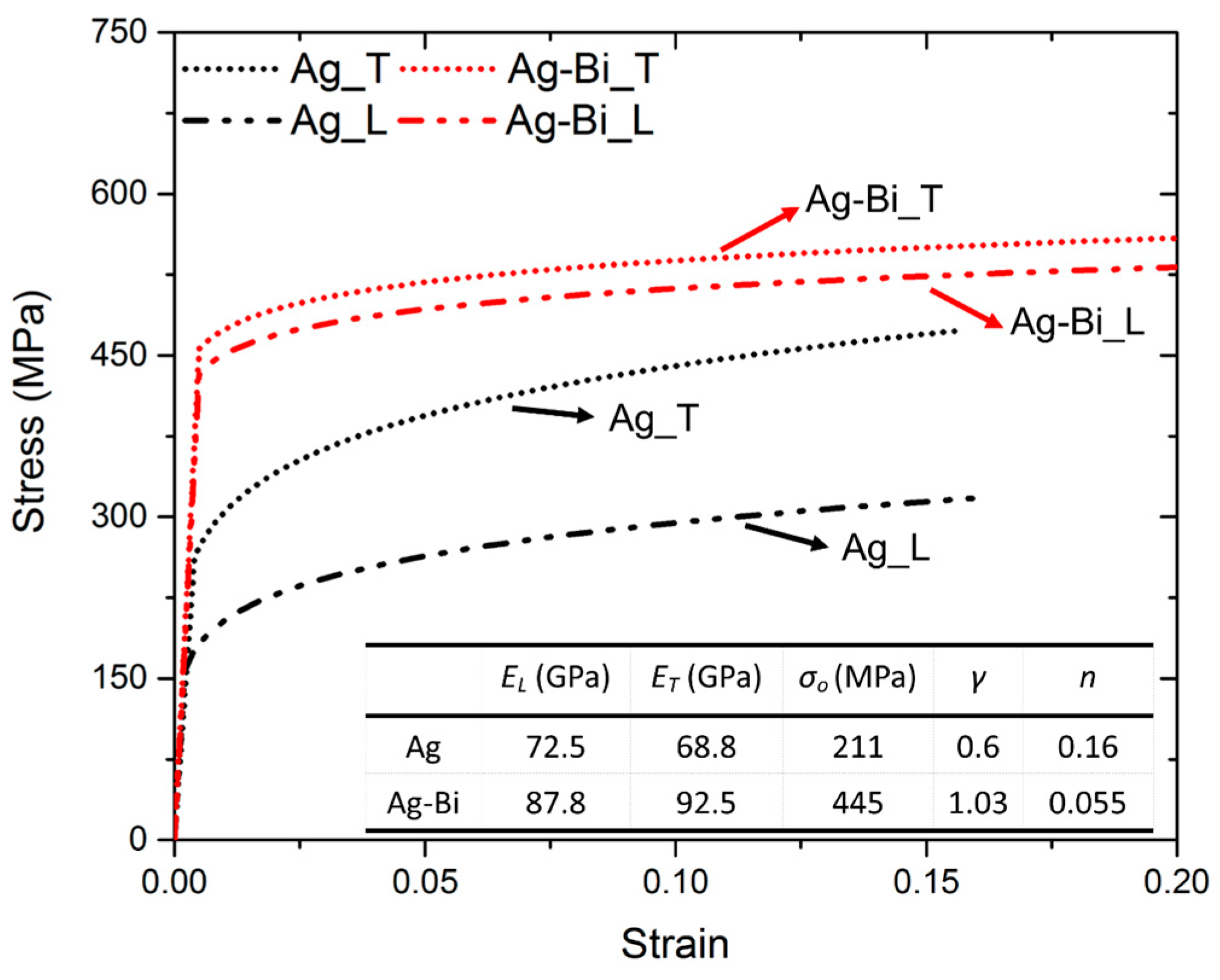
3.2. Nanoindentation and Inverse Calculation with Three-Dimensional FEA
- (1)
- With the Oliver-Pharr (OP) method [47], the nominal elastic modulus was calculated as = and for the two directions, respectively. The hardness values in the two directions were calculated as = and .
- (2)
- Since the difference between and is ~5%, which is quite similar to the previous observation of the Ag coating, and were replaced with and respectively. Meanwhile, with the previous yield strength-hardness relationship at 5 mN [48], and were calculated as 480 and 410 MPa, respectively. Regarding that the actual yield strength in the two directions ( + ) could be approximated by the sum of the nominal yield strength ( + ) [25,27], was calculated as 445 MPa.
- (3)
- Finally, a 3-D finite element indentation model was developed within the commercial finite element package, ABAQUS, to simulate the indentation process of the Ag-Bi coating in order to inversely determine and . A parametric FEA-based indentation study was carried out to calculate the maximum indentation loads in the two directions to compare with the experimental measurements as target values. As mentioned earlier, the indentation depth was ~300 nm, and the strong indentation size effect (ISE) could be expected considering the thin nature of the Ag-Bi coating. Thus, we first removed the ISE based on the characteristic length ( = 450 nm [49,50]) for the electrodeposited Ag-Bi coating. It should be noted that the target load values for the simulation are or rather than 5 mN [51]. With one set of and values, the Berkovich indenter penetrates the Ag-Bi coating to the same depth as , and the corresponding in the L direction was calculated. Next, the input materials properties were rotated by 90° around the T direction, the indenter penetrated the material to , and the corresponding was calculated in the T direction. Thus, different sets of vs. values can be obtained with corresponding and values. The most plausible combination of and n can be determined by choosing the corresponding set closest to the target experimentally measured the data point.
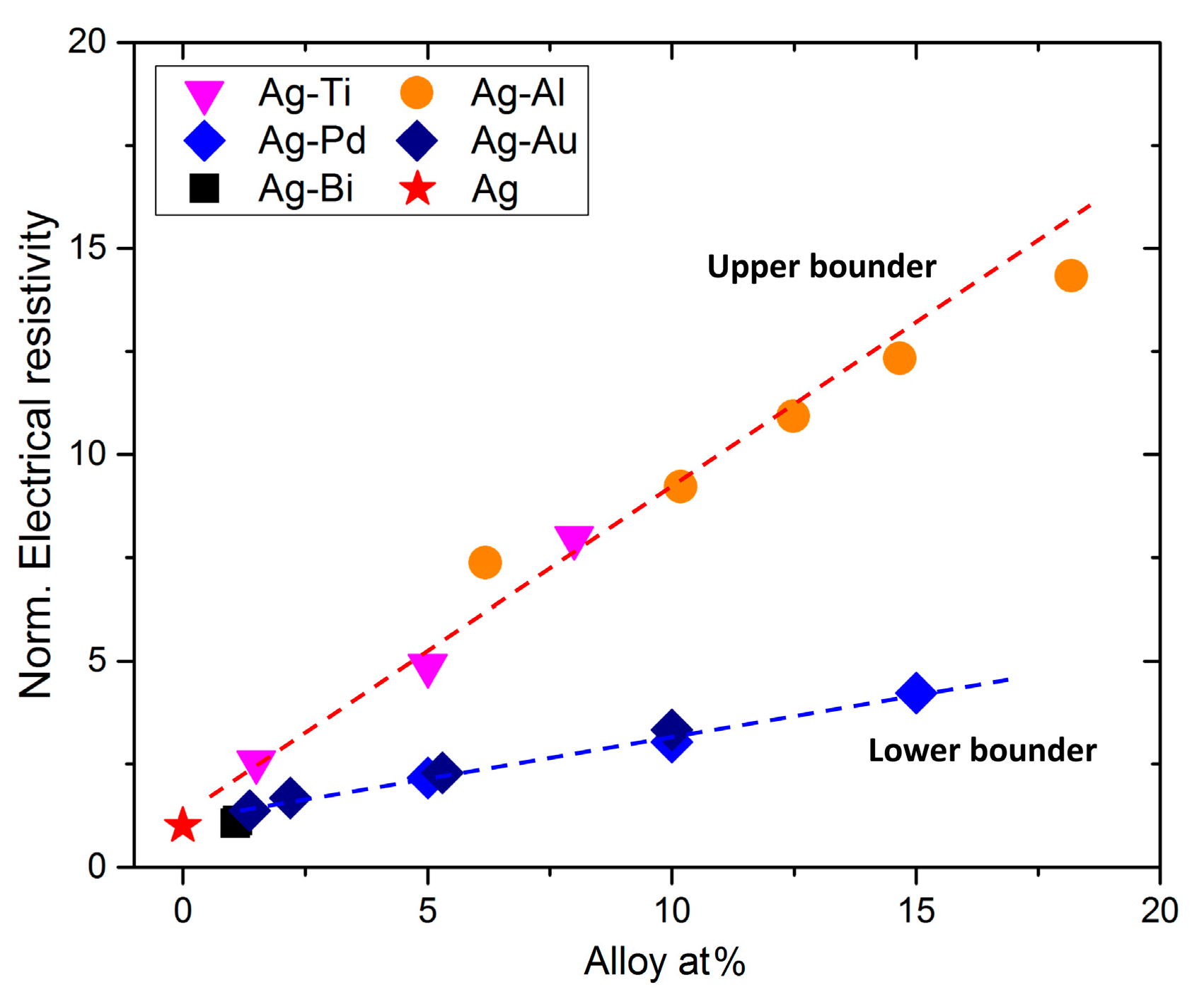
3.3. Electrical Resistivity of Ag and Ag-Bi Coatings
4. Discussion
4.1. Microstructure of the Ag and Ag-Bi Coatings
4.1.1. Significant Improvement of Flow Stress
4.1.2. Significant Decrease of the Hardening Exponent (n)
4.1.3. Slight Improvement of Elastic Modulus
4.1.4. Significant Reduced Plastic Anisotropy (γ)
4.2. Effects of Bi Addition on the Electrical Resistivity
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Antler, M. Electrical effects of fretting connector contact materials: A review. Wear 1985, 106, 5–33. [Google Scholar] [CrossRef]
- Findik, F.; Uzun, H. Microstructure, hardness and electrical properties of silver-based refractory contact materials. Mater. Des. 2003, 24, 489–492. [Google Scholar] [CrossRef]
- Krastev, I.; Valkova, T.; Zielonka, A. Structure and properties of electrodeposited silver–bismuth alloys. J. Appl. Electrochem. 2004, 34, 79–85. [Google Scholar] [CrossRef]
- Schlesinger, M.; Paunovic, M. Modern Electroplating, 5th ed.; John Wiley & Sons: Hoboken, NJ, USA, 2011. [Google Scholar]
- Krastev, I.; Valkova, T.; Zielonka, A. Effect of electrolysis conditions on the deposition of silver-bismuth alloys. J. Appl. Electrochem. 2003, 33, 1199–1204. [Google Scholar] [CrossRef]
- Yu, B.; Leung, K.M.; Guo, Q.; Lau, W.M.; Yang, J. Synthesis of Ag-TiO2 composite nano thin film for antimicrobial application. Nanotechnology 2011, 22, 115603. [Google Scholar] [CrossRef] [PubMed]
- Gay, P.-A.; Bercot, P.; Pagetti, J. Electrodeposition and characterisation of Ag–ZrO2 electroplated coatings. Surf. Coat. Technol. 2001, 140, 147–154. [Google Scholar] [CrossRef]
- Pasricha, R.; Gupta, S.; Srivastava, A.K. A facile and novel synthesis of Ag–graphene-based nanocomposites. Small 2009, 5, 2253–2259. [Google Scholar] [CrossRef] [PubMed]
- Mao, F.; Taher, M.; Kryshtal, O.; Kruk, A.; Czyrska-Filemonowicz, A.; Ottosson, M.; Andersson, A.M.; Wiklund, U.; Jansson, U. Combinatorial study of gradient Ag–Al thin films: Microstructure, phase formation, mechanical and electrical properties. ACS Appl. Mater. Interfaces 2016, 8, 30635–30643. [Google Scholar] [CrossRef] [PubMed]
- Butler, W.H.; Stocks, G.M. Calculated electrical-conductivity and thermopower of silver-palladium alloys. Phys. Rev. B 1984, 29, 4217–4223. [Google Scholar] [CrossRef]
- Chithra, S.; Lele, S.; Chattopadhyay, K. Structure evolution and phase change in Ag–5.1 at. % Bi alloy during mechanical alloying. Acta Mater. 2011, 59, 2009–2019. [Google Scholar] [CrossRef]
- Alford, T.; Adams, D.; Laursen, T.; Manfred Ullrich, B. Encapsulation of Ag films on SiO2 by ti reactions using Ag–Ti alloy/bilayer structures and an NH3 ambient. Appl. Phys. Lett. 1996, 68, 3251–3253. [Google Scholar] [CrossRef]
- Crisp, R.; Rungis, J. Thermoelectric power and thermal conductivity in the silver-gold alloy system from 3–300° k. Philos. Mag. 1970, 22, 217–236. [Google Scholar] [CrossRef]
- Tay, S.L. Electrodeposition Coatings by Ionic Co-Discharge Technique. Ph.D. Thesis, University of Auckland, Auckland, New Zealand, 2016. [Google Scholar]
- Kumar, M.; Deka, S. Multiply twinned AgNi alloy nanoparticles as highly active catalyst for multiple reduction and degradation reactions. ACS Appl. Mater. Interfaces 2014, 6, 16071–16081. [Google Scholar] [CrossRef] [PubMed]
- Adamik, M.; Barna, P.B.; Tomov, I. Correlation between texture and average grain size in polycrystalline Ag thin films. Thin Solid Films 2000, 359, 33–38. [Google Scholar] [CrossRef]
- Zhong, S.; Koch, T.; Wang, M.; Scherer, T.; Walheim, S.; Hahn, H.; Schimmel, T. Nanoscale twinned copper nanowire formation by direct electrodeposition. Small 2009, 5, 2265–2270. [Google Scholar] [CrossRef] [PubMed]
- Kumar, K.; Suresh, S.; Chisholm, M.; Horton, J.; Wang, P. Deformation of electrodeposited nanocrystalline nickel. Acta Mater. 2003, 51, 387–405. [Google Scholar] [CrossRef]
- Navinšek, B.; Panjan, P.; Milošev, I. PVD coatings as an environmentally clean alternative to electroplating and electroless processes. Surf. Coat. Technol. 1999, 116, 476–487. [Google Scholar] [CrossRef]
- Wang, Y.; Ju, Y.; Wei, S.; Lu, W.; Yan, B.; Gao, W. Mechanical properties and microstructure of Au–Ni–TiO2 nano-composite coatings. Mater. Charact. 2015, 102, 189–194. [Google Scholar] [CrossRef]
- Strehle, S.; Bartha, J.; Wetzig, K. Electrical properties of electroplated Cu (Ag) thin films. Thin Solid Films 2009, 517, 3320–3325. [Google Scholar] [CrossRef]
- Elliott, R.P.; Shunk, F.A. The Ag-Bi (silver-bismuth) system. Bull. Alloy Phase Diagr. 1980, 1, 62–64. [Google Scholar] [CrossRef]
- Krastev, I.; Valkova, T.; Zielonka, A. Internal stress in multilayer silver–bismuth coatings. J. Appl. Electrochem. 2005, 35, 539–544. [Google Scholar] [CrossRef]
- Manzano, C.V.; Abad, B.; Rojo, M.M.; Koh, Y.R.; Hodson, S.L.; Martinez, A.M.L.; Xu, X.; Shakouri, A.; Sands, T.D.; Borca-Tasciuc, T. Anisotropic effects on the thermoelectric properties of highly oriented electrodeposited Bi2Te3 films. Sci. Rep. 2016, 6, 19129. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, T.; Gu, Y. Identification of elastic–plastic anisotropic parameters using instrumented indentation and inverse analysis. Mech. Mater. 2007, 39, 340–356. [Google Scholar] [CrossRef]
- Pei, Y.T.; Song, G.M.; Sloof, W.G.; De Hosson, J.T.M. A methodology to determine anisotropy effects in non-cubic coatings. Surf. Coat. Technol. 2007, 201, 6911–6916. [Google Scholar] [CrossRef]
- Cheng, G.; Sun, X.; Wang, Y.; Tay, S.L.; Gao, W. Nanoindentation study of electrodeposited Ag thin coating: An inverse calculation of anisotropic elastic-plastic properties. Surf. Coat. Technol. 2017, 310, 43–50. [Google Scholar] [CrossRef]
- Xiang, Y.; Tsui, T.Y.; Vlassak, J.J. The mechanical properties of freestanding electroplated Cu thin films. J. Mater. Res. 2006, 21, 1607–1618. [Google Scholar] [CrossRef]
- Huang, H.; Spaepen, F. Tensile testing of free-standing Cu, Ag and Al thin films and Ag/Cu multilayers. Acta Mater. 2000, 48, 3261–3269. [Google Scholar] [CrossRef]
- You, Z.S.; Lu, L.; Lu, K. Tensile behavior of columnar grained Cu with preferentially oriented nanoscale twins. Acta Mater. 2011, 59, 6927–6937. [Google Scholar] [CrossRef]
- Baker, S.P.; Nix, W.D. Mechanical properties of compositionally modulated Au-Ni thin films: Nanoindentation and microcantilever deflection experiments. J. Mater. Res. 1994, 9, 3131–3145. [Google Scholar] [CrossRef]
- Fang, W.; Wickert, J. Determining mean and gradient residual stresses in thin films using micromachined cantilevers. J. Micromech. Microeng. 1996, 6, 301. [Google Scholar] [CrossRef]
- Nili, H.; Cheng, G.; Venkatesh, T.A.; Sriram, S.; Bhaskaran, M. Correlation between nanomechanical and piezoelectric properties of thin films: An experimental and finite element study. Mater. Lett. 2013, 90, 148–151. [Google Scholar] [CrossRef]
- Su, F.-H.; Huang, P. Microstructure and tribological property of nanocrystalline Co–W alloy coating produced by dual-pulse electrodeposition. Mater. Chem. Phys. 2012, 134, 350–359. [Google Scholar] [CrossRef]
- Suresh, S.; Nieh, T.G.; Choi, B.W. Nano-indentation of copper thin films on silicon substrates. Scr. Mater. 1999, 41, 951–957. [Google Scholar] [CrossRef]
- Cheng, G.; Choi, K.S.; Hu, X.; Sun, X. Determining individual phase properties in a multi-phase Q&P steel using multi-scale indentation tests. Mater. Sci. Eng. A 2016, 652, 384–395. [Google Scholar] [CrossRef]
- Cheng, G.; Sriram, S.; Bhaskaran, M.; Venkatesh, T.A. Nanoindentation response of piezoelectric nano-islands. Appl. Phys. Lett. 2014, 105, 122902. [Google Scholar] [CrossRef]
- Williams, J.J.; Walters, J.L.; Wang, M.Y.; Chawla, N.; Rohatgi, A. Extracting constitutive stress–strain behavior of microscopic phases by micropillar compression. JOM 2013, 65, 226–233. [Google Scholar] [CrossRef]
- Buzzi, S.; Dietiker, M.; Kunze, K.; Spolenak, R.; Löffler, J.F. Deformation behavior of silver submicrometer-pillars prepared by nanoimprinting. Philos. Mag. 2009, 89, 869–884. [Google Scholar] [CrossRef]
- Stewart, J.L.; Jiang, L.; Williams, J.J.; Chawla, N. Prediction of bulk tensile behavior of dual phase stainless steels using constituent behavior from micropillar compression experiments. Mater. Sci. Eng. A 2012, 534, 220–227. [Google Scholar] [CrossRef]
- Bautista, K. Four-Point Probe Operation; SP2004-TF-005 Erik Jonsson School of Engineering, The University of Texas: Dallas, TX, USA, 2003; Available online: http://www.utdallas.edu/~gpp052000/Docs/4PointProbe_Manual.pdf (accessed on 30 April 2015).
- Chen, J.; Bull, S. On the relationship between plastic zone radius and maximum depth during nanoindentation. Surf. Coat. Technol. 2006, 201, 4289–4293. [Google Scholar] [CrossRef]
- Fischer-Cripps, A.C. Introduction to Contact Mechanics, 2nd ed.; Springer: New York, NY, USA, 2007. [Google Scholar]
- Yeh, J.-W.; Chang, S.-Y.; Hong, Y.-D.; Chen, S.-K.; Lin, S.-J. Anomalous decrease in X-ray diffraction intensities of Cu–Ni–Al–Co–Cr–Fe–Si alloy systems with multi-principal elements. Mater. Chem. Phys. 2007, 103, 41–46. [Google Scholar] [CrossRef]
- Liu, L.; Wang, J.; Gong, S.; Mao, S. High resolution transmission electron microscope observation of zero-strain deformation twinning mechanisms in Ag. Phys. Rev. Lett 2011, 106, 175504. [Google Scholar] [CrossRef] [PubMed]
- Kibey, S.; Liu, J.; Johnson, D.; Sehitoglu, H. Predicting twinning stress in FCC metals: Linking twin-energy pathways to twin nucleation. Acta Mater. 2007, 55, 6843–6851. [Google Scholar] [CrossRef]
- Oliver, W.C.; Pharr, G.M. An improved technique for determining hardness and elastic modulus using load and displacement sensing indentation experiments. J. Mater. Res. 1992, 7, 1564–1583. [Google Scholar] [CrossRef]
- Elmustafa, A.A.; Stone, D.S. Indentation size effect in polycrystalline FCC metals. Acta Mater. 2002, 50, 3641–3650. [Google Scholar] [CrossRef]
- Ma, Q.; Clarke, D.R. Size dependent hardness of silver single crystals. J. Mater. Res. 1995, 10, 853–863. [Google Scholar] [CrossRef]
- Stegall, D.E.; Mamun, M.A.; Crawford, B.; Elmustafa, A. Indentation size effect in FCC metals: An examination of experimental techniques and the bilinear behavior. J. Mater. Res. 2012, 27, 1543–1552. [Google Scholar] [CrossRef]
- Cheng, G.; Barker, E.I.; Stephens, E.V.; Choi, K.S.; Sun, X. Quantifying grain level stress-strain behavior for AM40 via instrumented microindentation. MRS Adv. 2016, 1, 761–772. [Google Scholar] [CrossRef]
- Gurrappa, I.; Binder, L. Electrodeposition of nanostructured coatings and their characterization—A review. Sci. Technol. Adv. Mater. 2008, 9, 043001. [Google Scholar] [CrossRef] [PubMed]
- Askari, H.; Zbib, H.M.; Sun, X. Multiscale modeling of inclusions and precipitation hardening in metal matrix composites: Application to advanced high-strength steels. J. Nanomech. Micromech. 2012, 3, 24–33. [Google Scholar] [CrossRef]
- Rhee, M.; Hirth, J.; Zbib, H. A superdislocation model for the strengthening of metal matrix composites and the initiation and propagation of shear bands. Acta Mater. 1994, 42, 2645–2655. [Google Scholar] [CrossRef]
- Liang, X.; Chen, J.; Mora, M.T.; Urdaneta, J.F.; Zeng, Q. Effect of precipitation on the hardness of ternary metallic glass. Adv. Mater. Phys. Chem. 2017, 7, 255–262. [Google Scholar] [CrossRef][Green Version]
- Hansen, N. Hall–petch relation and boundary strengthening. Scr. Mater. 2004, 51, 801–806. [Google Scholar] [CrossRef]
- Schuh, C.A.; Nieh, T.G.; Yamasaki, T. Hall-petch breakdown manifested in abrasive wear resistance of nanocrystalline nickel. Scr. Mater. 2002, 46, 735–740. [Google Scholar] [CrossRef]
- Ma, E.; Wang, Y.; Lu, Q.; Sui, M.; Lu, L.; Lu, K. Strain hardening and large tensile elongation in ultrahigh-strength nano-twinned copper. Appl. Phys. Lett. 2004, 85, 4932–4934. [Google Scholar] [CrossRef]
- Hu, X.; Jain, M.; Wilkinson, D.; Mishra, R. Microstructure-based finite element analysis of strain localization behavior in AA5754 aluminum sheet. Acta Mater. 2008, 56, 3187–3201. [Google Scholar] [CrossRef]
- Cheng, G.; Choi, K.S.; Hu, X.; Sun, X. Predicting deformation limits of dual-phase steels under complex loading paths. JOM 2017, 69, 1046–1051. [Google Scholar] [CrossRef]
- Cheng, G.; Zhang, F.; Ruimi, A.; Field, D.P.; Sun, X. Quantifying the effects of tempering on individual phase properties of DP980 steel with nanoindentation. Mater. Sci. Eng. A 2016, 667, 240–249. [Google Scholar] [CrossRef]
- Archard, J. Contact and rubbing of flat surfaces. J. Appl. Phys. 1953, 24, 981–988. [Google Scholar] [CrossRef]



| Bath Composition and Plating Parameters | Quantity |
|---|---|
| Ag Deposition | |
| Silver metal | 30 g/L |
| KCN free | 120 g/L |
| KOH | 10 g/L |
| Temperature | 30 °C |
| Current density | 10 mA/cm2 |
| Agitation speed | 200 rpm |
| Plating time | 30 min |
| Ag-Bi Deposition | |
| Silver metal | 30 g/L |
| KCN free | 120 g/L |
| KOH | 10.35 g/L |
| Bi(NO3)3·5H2O | 0.2425 g/L |
| Tartaric acid | 0.075 g/L |
| Temperature | 30 °C |
| Current density | 10 mA/cm2 |
| Agitation speed | 200 rpm |
| Plating time | 30 min |
| (a) Symbol Descriptions | |
| and | Hardness in the L and T directions |
| and | Nominal elastic modulus in the L and T directions |
| and | Maximum indentation load in the L and T directions |
| and | Maximum indentation depth in the L and T directions |
| Indentation size effect (ISE) characteristic length | |
| The ratio of yield strength | |
| and | Maximum indentation load with modification in the L and T directions |
| and | Maximum indentation load from FEA in the L and T directions |
| and | Shear modulus in the L and T directions |
| Reference strength | |
| and | Isotropic plastic strain in the L and T directions |
| n | Strain hardening exponent |
| and | Nominal yield strength in the L and T directions |
| and | Nominal strain hardening exponent in the L and T directions |
| (b) Constitutive equations | |
| Elastic | Plastic |
for for | |
| Assumptions for inverse calculation | |
+ | |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Y.; Cheng, G.; Tay, S.L.; Guo, Y.; Sun, X.; Gao, W. Effects of Bi Addition on the Microstructure and Mechanical Properties of Nanocrystalline Ag Coatings. Materials 2017, 10, 932. https://doi.org/10.3390/ma10080932
Wang Y, Cheng G, Tay SL, Guo Y, Sun X, Gao W. Effects of Bi Addition on the Microstructure and Mechanical Properties of Nanocrystalline Ag Coatings. Materials. 2017; 10(8):932. https://doi.org/10.3390/ma10080932
Chicago/Turabian StyleWang, Yuxin, Guang Cheng, See Leng Tay, Yunxia Guo, Xin Sun, and Wei Gao. 2017. "Effects of Bi Addition on the Microstructure and Mechanical Properties of Nanocrystalline Ag Coatings" Materials 10, no. 8: 932. https://doi.org/10.3390/ma10080932
APA StyleWang, Y., Cheng, G., Tay, S. L., Guo, Y., Sun, X., & Gao, W. (2017). Effects of Bi Addition on the Microstructure and Mechanical Properties of Nanocrystalline Ag Coatings. Materials, 10(8), 932. https://doi.org/10.3390/ma10080932






