Decreasing Bio-Degradation Rate of the Hydrothermal-Synthesizing Coated Mg Alloy via Pre-Solid-Solution Treatment
Abstract
1. Introduction
2. Experimental
2.1. Materials and Hydrothermal Synthesizing Processing
2.2. Microstructure Characterization
2.3. Corrosion Tests
3. Results and Discussion
3.1. Microstructure Observation of the Substrate Alloys
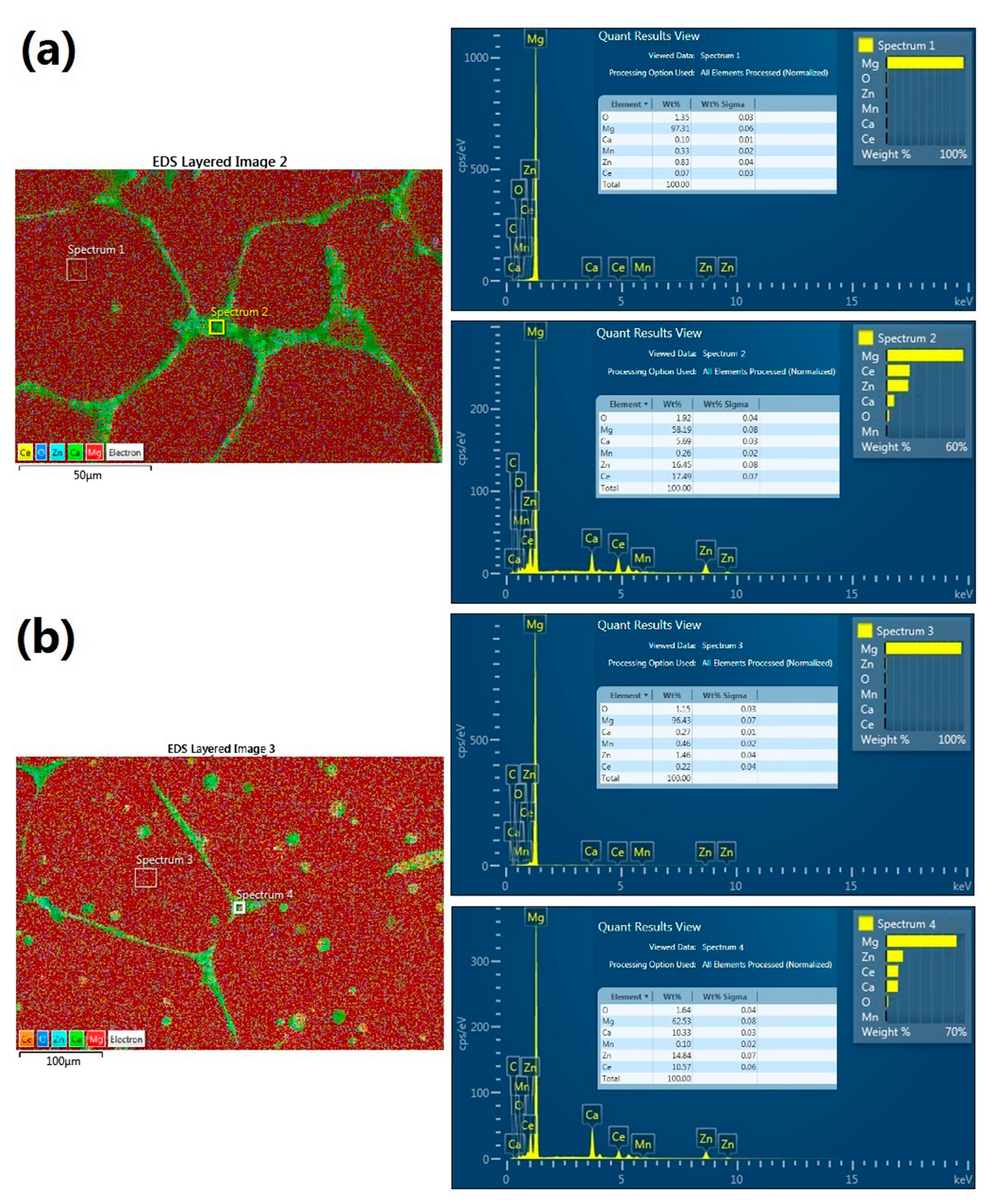
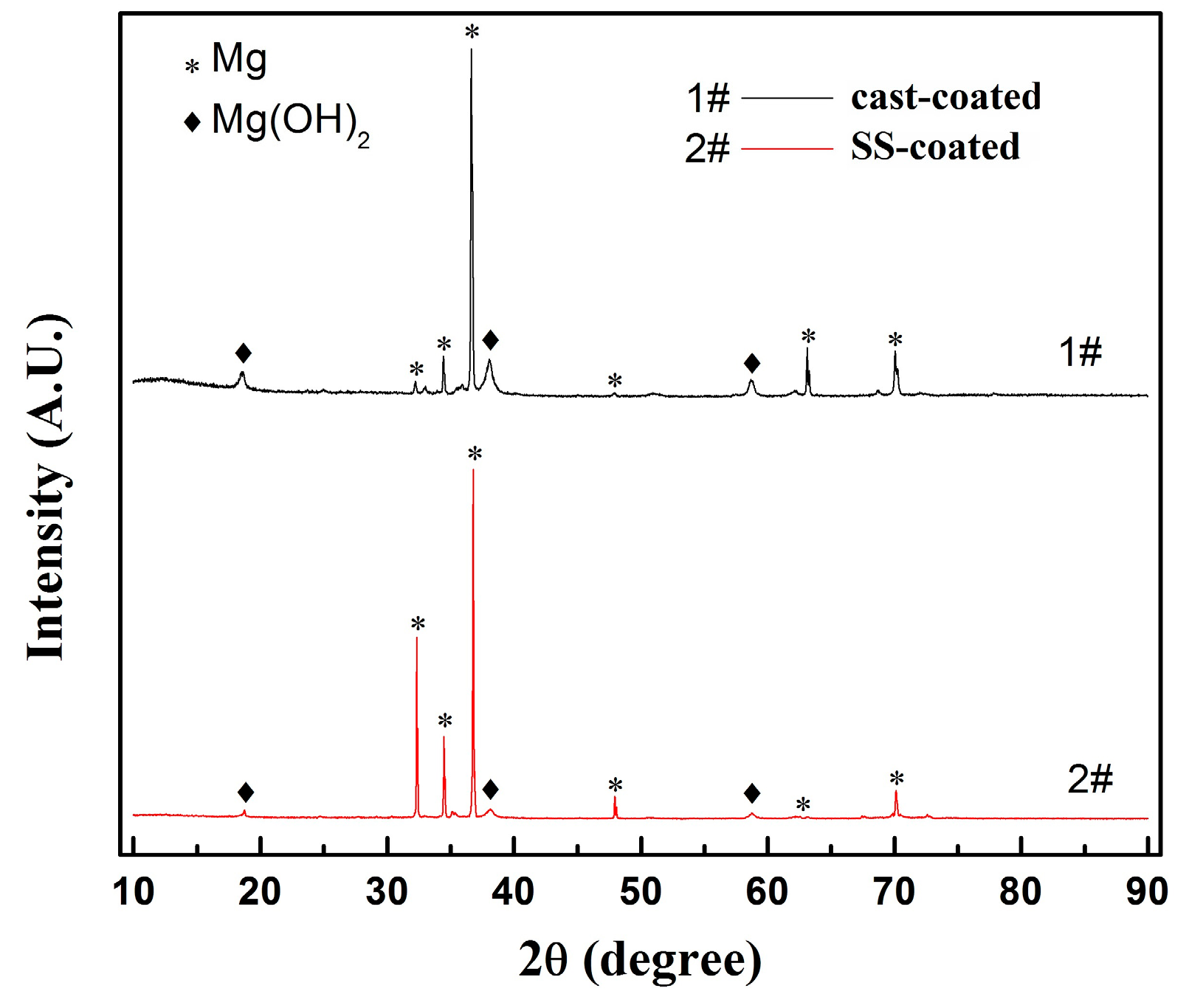
3.2. Microstructure Observation of the Coatings
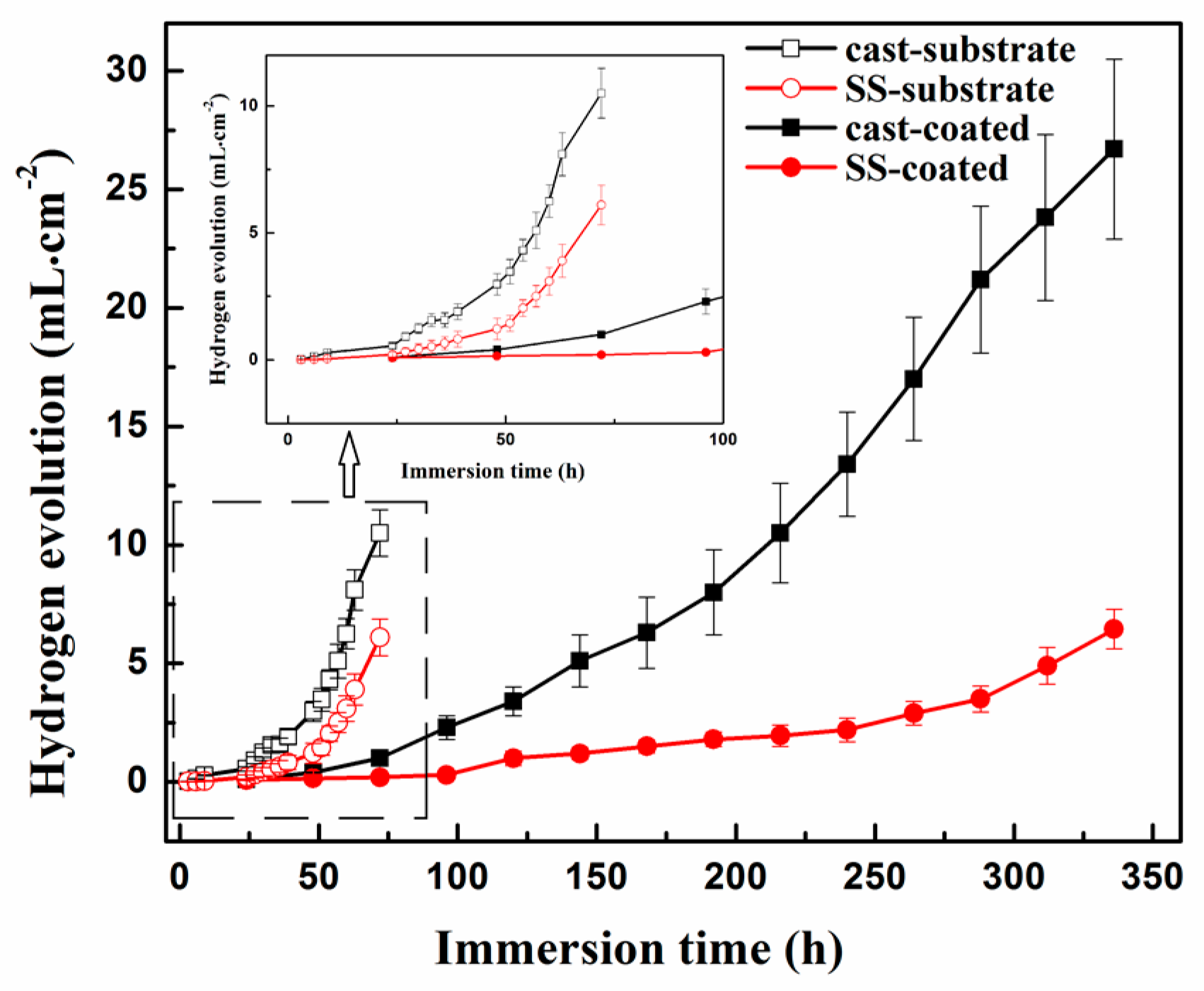
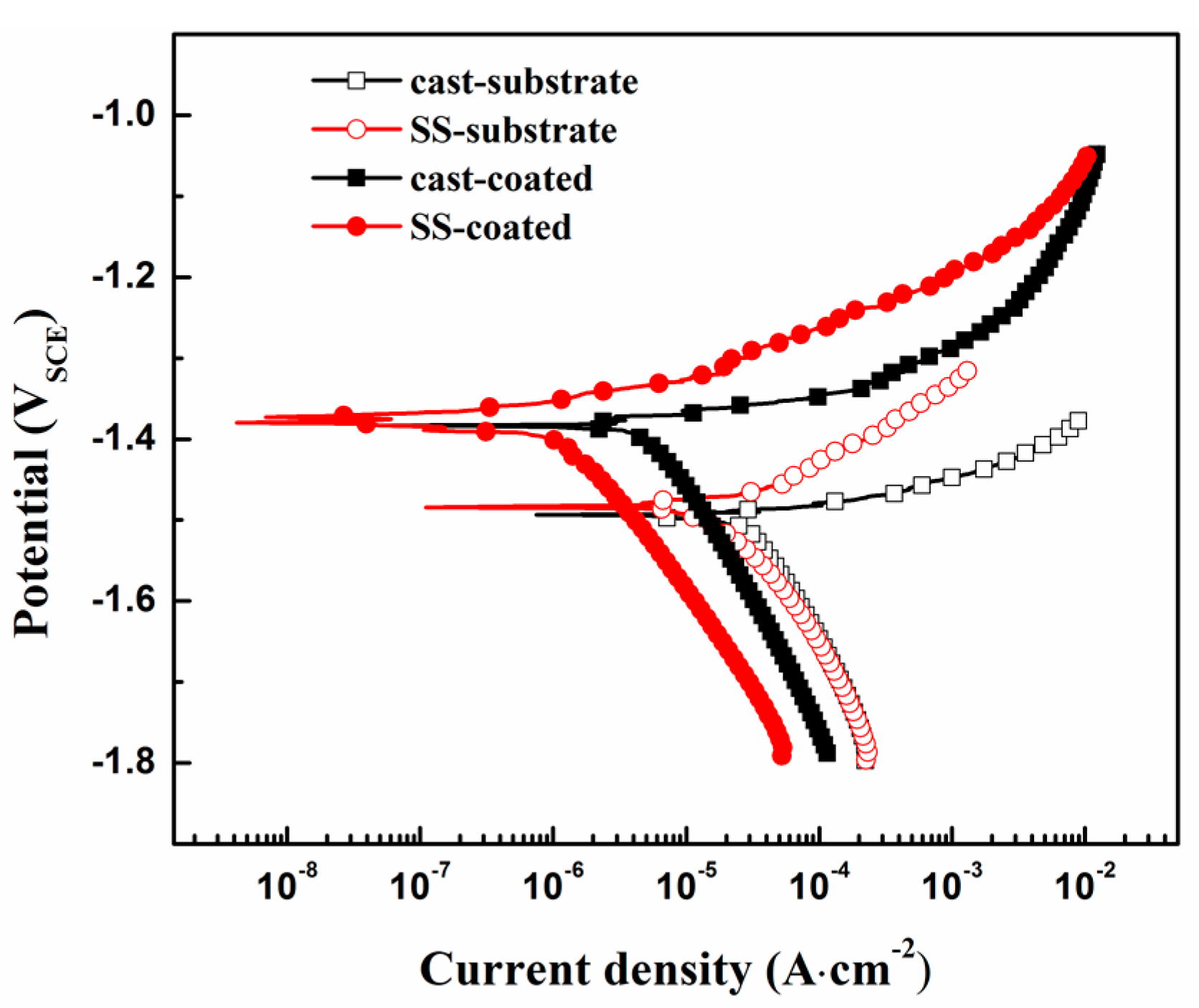
3.3. In-Vitro Degradation Behavior
3.4. Positive Effect of Pre-SS Treatment on the Coating and the Substrate
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Witte, F. The history of biodegradable magnesium implants: A review. Acta Biomater. 2010, 6, 1680–1692. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, S.; Curtin, J.; Duffy, B.; Jaiswal, S. Biodegradable magnesium alloys for orthopaedic applications: A review on corrosion, biocompatibility and surface modifications. Mater. Sci. Eng. 2016, 68, 948–963. [Google Scholar] [CrossRef] [PubMed]
- Nene, S.S.; Kashyap, B.P.; Prabhu, N.; Estrin, Y.; Al-Samman, T. Microstructure refinement and its effect on specific strength and bio-corrosion resistance in ultralight Mg-4Li-1Ca (LC41) alloy by hot rolling. J. Alloys Compd. 2014, 615, 501–506. [Google Scholar] [CrossRef]
- Staiger, M.P.; Pietak, A.M.; Huadmai, J.; Dias, G. Magnesium and its alloys as orthopedic biomaterials: A review. Biomaterials 2006, 27, 1728–1734. [Google Scholar] [CrossRef] [PubMed]
- Witte, F.; Hort, N.; Vogt, C.; Cohen, S.; Kainer, K.U.; Willumeit, R.; Feyerabend, F. Degradable biomaterials based on magnesium corrosion. Curr. Opin. Solid State Mater. Sci. 2008, 12, 63–72. [Google Scholar] [CrossRef]
- Yang, L.; Huang, Y.; Feyerabend, F.; Willumeit, R.; Mendis, C.; Kainer, K.U.; Hort, N. Microstructure, mechanical and corrosion properties of Mg-Dy-Gd-Zr alloys for medical applications. Acta Biomater. 2013, 9, 8499–8508. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, A.H.M.; Luthringer, B.J.C.; Feyerabend, F.; Willumeit, R. Mg and Mg alloys: How comparable are in vitro and in vivo corrosion rates? A review. Acta Biomater. 2015, 13, 16–31. [Google Scholar] [CrossRef] [PubMed]
- Hofstetter, J.; Martinelli, E.; Pogatscher, S.; Schmutz, P.; Povoden-Karadeniz, E.; Weinberg, A.M.; Uggowitzer, P.J.; Löffler, J.F. Influence of trace impurities on the in vitro and in vivo degradation of biodegradable Mg-5Zn-0.3Ca alloys. Acta Biomater. 2015, 23, 347–353. [Google Scholar] [CrossRef] [PubMed]
- Jamesh, M.I.; Wu, G.; Zhao, Y.; McKenzie, D.R.; Bilek, M.M.M.; Chu, P.K. Electrochemical corrosion behavior of biodegradable Mg-Y-RE and Mg-Zn-Zr alloys in Ringer’s solution and simulated body fluid. Corros. Sci. 2015, 91, 160–184. [Google Scholar] [CrossRef]
- Ascencio, M.; Pekguleryuz, M.; Omanovic, S. An investigation of the corrosion mechanisms of WE43 Mg alloy in a modified simulated body fluid solution: The influence of immersion time. Corros. Sci. 2014, 87, 489–503. [Google Scholar] [CrossRef]
- Zeng, R.C.; Qi, W.C.; Cui, H.Z.; Zhang, F.; Li, S.Q.; Han, E.H. In vitro corrosion of as-extruded Mg-Ca alloys—The influence of Ca concentration. Corros. Sci. 2015, 96, 23–31. [Google Scholar] [CrossRef]
- Zander, D.; Zumdick, N.A. Influence of Ca and Zn on the microstructure and corrosion of biodegradable Mg-Ca-Zn alloys. Corros. Sci. 2015, 93, 222–233. [Google Scholar] [CrossRef]
- Tok, H.Y.; Hamzah, E.; Bakhsheshi-Rad, H.R. The role of bismuth on the microstructure and corrosion behavior of ternary Mg–1.2Ca–xBi alloys for biomedical applications. J. Alloys Compd. 2015, 640, 335–346. [Google Scholar] [CrossRef]
- Liu, L.; Yuan, F.L.; Zhao, M.C.; Gao, C.D.; Feng, P.; Yang, Y.W.; Yang, S.; Shuai, C.J. Rare Earth Element Yttrium Modified Mg-Al-Zn Alloy: Microstructure, Degradation Properties and Hardness. Materials 2017, 10, 477. [Google Scholar] [CrossRef]
- Zhang, W.; Li, M.; Chen, Q.; Hu, W.; Zhang, W.; Xin, W. Effects of Sr and Sn on microstructure and corrosion resistance of Mg-Zr-Ca magnesium alloy for biomedical applications. Mater. Des. 2012, 39, 379–383. [Google Scholar] [CrossRef]
- Tsai, M.H.; Chen, M.S.; Lin, L.H.; Lin, M.H.; Wu, C.Z.; Ou, K.L.; Yu, C.H. Effect of heat treatment on the microstructures and damping properties of biomedical Mg-Zr alloy. J. Alloys Compd. 2011, 509, 813–819. [Google Scholar] [CrossRef]
- Lin, D.J.; Hung, F.Y.; Lui, T.S.; Yeh, M.L. Heat treatment mechanism and biodegradable characteristics of ZAX1330 Mg alloy. Mater. Sci. Eng. 2015, 51, 300–308. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Yuan, G.; Mao, L.; Niu, J.; Fu, P.; Ding, W. Effects of extrusion and heat treatment on the mechanical properties and biocorrosion behaviors of a Mg-Nd-Zn-Zr alloy. J. Mech. Behav. Biomed. Mater. 2012, 7, 77–86. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.Z.; Zhu, S.J.; Wang, L.G.; Guo, R.M.; Yue, G.C.; Guan, S.K. Microstructures and degradation mechanism in simulated body fluid of biomedical Mg-Zn-Ca alloy processed by high pressure torsion. Mater. Des. 2016, 96, 54–62. [Google Scholar] [CrossRef]
- Ge, Q.; Dellasega, D.; Demir, A.G.; Vedani, M. The processing of ultrafine-grained Mg tubes for biodegradable stents. Acta Biomater. 2013, 9, 8604–8610. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Kang, Z.; Wang, F. Mechanical properties and biocorrosion resistance of the Mg-Gd-Nd-Zn-Zr alloy processed by equal channel angular pressing. Mater. Sci. Eng. 2016, 68, 194–197. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Li, Y.D.; Li, Y.X.; Wu, Y.H.; Zheng, Y.F.; Han, Y. Effect of surface mechanical attrition treatment on biodegradable Mg–1Ca alloy. Mater. Sci. Eng. 2014, 35, 314–321. [Google Scholar] [CrossRef] [PubMed]
- Razavi, M.; Fathi, M.; Savabi, O.; Vashaee, D.; Tayebi, L. In vitro study of nanostructured diopside coating on Mg alloy orthopedic implants. Mater. Sci. Eng. 2014, 41, 168–177. [Google Scholar] [CrossRef] [PubMed]
- Gu, X.N.; Li, N.; Zhou, W.R.; Zheng, Y.F.; Zhao, X.; Cai, Q.Z.; Ruan, L. Corrosion resistance and surface biocompatibility of a microarc oxidation coating on a Mg-Ca alloy. Acta Biomater. 2011, 7, 1880–1889. [Google Scholar] [CrossRef] [PubMed]
- Gupta, R.K.; Mensah-Darkwa, K.; Kumar, D. Corrosion Protective Conversion Coatings on Magnesium Disks Using a Hydrothermal Technique. J. Mater. Sci. Technol. 2014, 30, 47–53. [Google Scholar] [CrossRef]
- Lee, H.P.; Lin, D.J.; Yeh, M.L. Phenolic Modified Ceramic Coating on Biodegradable Mg Alloy: The Improved Corrosion Resistance and Osteoblast-Like Cell Activity. Materials 2017, 10, 696. [Google Scholar] [CrossRef]
- Zhu, Y.; Zhao, Q.; Zhang, Y.H.; Wu, G. Hydrothermal synthesis of protective coating on magnesium alloy using de-ionized water. Surf. Coat. Technol. 2012, 206, 2961–2966. [Google Scholar] [CrossRef]
- Zhang, F.; Ma, A.; Song, D.; Jiang, J.; Lu, F.; Zhang, L.; Yang, D.; Chen, J. Improving in-vitro biocorrosion resistance of Mg-Zn-Mn-Ca alloy in Hank’s solution through addition of cerium. J. Rare Earths 2015, 33, 93–101. [Google Scholar] [CrossRef]
- Guo, G.; Song, D.; Jiang, J.; Ma, A.; Zhang, L.; Li, C. Effect of Synthesizing Temperature on Microstructure and Electrochemical Property of the Hydrothermal Conversion Coating on Mg-2Zn-0.5Mn-Ca-Ce Alloy. Metals 2016, 6, 44. [Google Scholar] [CrossRef]
- Song, D.; Guo, G.G.; Jiang, J.H.; Zhang, L.W.; Ma, A.B.; Ma, X.L.; Chen, J.Q.; Cheng, Z.J. Hydrothermal synthesis and corrosion behavior of the protective coating on Mg–2Zn–Mn–Ca–Ce alloy. Prog. Nat. Sci. Mater. Int. 2016, 26, 590–599. [Google Scholar] [CrossRef]
- Song, G.L.; Xu, Z. Crystal orientation and electrochemical corrosion of polycrystalline Mg. Corros. Sci. 2012, 63, 100–112. [Google Scholar] [CrossRef]
- Song, G.L.; Atrens, A. Corrosion Mechanisms of Magnesium Alloys. Adv. Eng. Mater. 1999, 1, 11–33. [Google Scholar] [CrossRef]
- Bakhsheshi-Rad, H.; Abdul-Kadir, M.; Idris, M.; Farahany, S. Relationship between the corrosion behavior and the thermal characteristics and microstructure of Mg-0.5Ca-xZn alloys. Corros. Sci. 2012, 64, 184–197. [Google Scholar] [CrossRef]
- Zhang, Y.F.; Blawert, C.; Tang, S.W.; Hu, J.; Mohedano, M.; Zheludkevich, M.L.; Kainer, K.U. Influence of surface pre-treatment on the deposition and corrosion properties of hydrophobic coatings on a magnesium alloy. Corros. Sci. 2016, 112, 483–494. [Google Scholar] [CrossRef]
- Mosiałek, M.; Mordarski, G.; Nowak, P.; Simka, W.; Nawrat, G.; Hanke, M.; Socha, R.P.; Michalska, J. Phosphate—Permanganate conversion coatings on the AZ81 magnesium alloy: SEM, EIS and XPS studies. Surf. Coat. Technol. 2011, 206, 51–62. [Google Scholar] [CrossRef]
- Zhang, X.; Zhang, K.; Li, X.; Deng, X.; Li, Y.; Ma, M.; Shi, Y. Effect of solid-solution treatment on corrosion and electrochemical behaviors of Mg-15Y alloy in 3.5 wt % NaCl solution. J. Rare Earths 2012, 30, 1158–1167. [Google Scholar] [CrossRef]
- Miao, Y.; Hongzhe, J.; Yaohui, L.; Yulai, S. Influence of solid-solution treatment on corrosion performance of cast AM50 magnesium alloy. In Proceedings of the 2011 International Conference on Mechatronic Science, Electric Engineering and Computer (MEC), Jilin, China, 19–22 August 2011; pp. 425–429. [Google Scholar]
- Li, J.; Jiang, Q.; Sun, H.; Li, Y. Effect of heat treatment on corrosion behavior of AZ63 magnesium alloy in 3.5 wt % sodium chloride solution. Corros. Sci. 2016, 111, 288–301. [Google Scholar] [CrossRef]








| Zn | Mn | Ca | Ce | Mg |
|---|---|---|---|---|
| 2.00 | 0.50 | 1.02 | 1.35 | balance |
| Solution | Chemical Composition (mmol·L−1) | |||||||
|---|---|---|---|---|---|---|---|---|
| NaCl | CaCl2 | MgSO4 | KCl | KH2PO4 | Na2HPO4 | D-Glucose | NaHCO3 | |
| Hanks’ | 137 | 1.261 | 0.814 | 5.33 | 0.44 | 0.338 | 5.56 | 4.17 |
| Samples | Ecorr (V) | Icorr ( A·cm−2) | βa (V·dec−1) | βc (V·dec−1) | Rp (ohm·cm2) |
|---|---|---|---|---|---|
| Cast-coated | −1.38 | 4.56 × 10−6 | 0.041 | 0.242 | 3343 |
| SS-coated | −1.37 | 1.27 × 10−6 | 0.060 | 0.233 | 16,334 |
| Cast-substrate | −1.49 | 2.71 × 10−5 | 0.038 | 0.256 | 531 |
| SS-substrate | −1.48 | 1.64 × 10−5 | 0.058 | 0.227 | 1225 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Song, D.; Li, C.; Zhang, L.; Ma, X.; Guo, G.; Zhang, F.; Jiang, J.; Ma, A. Decreasing Bio-Degradation Rate of the Hydrothermal-Synthesizing Coated Mg Alloy via Pre-Solid-Solution Treatment. Materials 2017, 10, 858. https://doi.org/10.3390/ma10080858
Song D, Li C, Zhang L, Ma X, Guo G, Zhang F, Jiang J, Ma A. Decreasing Bio-Degradation Rate of the Hydrothermal-Synthesizing Coated Mg Alloy via Pre-Solid-Solution Treatment. Materials. 2017; 10(8):858. https://doi.org/10.3390/ma10080858
Chicago/Turabian StyleSong, Dan, Cheng Li, Liwen Zhang, Xiaolong Ma, Guanghui Guo, Fan Zhang, Jinghua Jiang, and Aibin Ma. 2017. "Decreasing Bio-Degradation Rate of the Hydrothermal-Synthesizing Coated Mg Alloy via Pre-Solid-Solution Treatment" Materials 10, no. 8: 858. https://doi.org/10.3390/ma10080858
APA StyleSong, D., Li, C., Zhang, L., Ma, X., Guo, G., Zhang, F., Jiang, J., & Ma, A. (2017). Decreasing Bio-Degradation Rate of the Hydrothermal-Synthesizing Coated Mg Alloy via Pre-Solid-Solution Treatment. Materials, 10(8), 858. https://doi.org/10.3390/ma10080858





