Structure and Photoluminescence Properties of Sm3+ Ion-Doped YInGe2O7 Phosphor
Abstract
:1. Introduction
2. Experimental Procedure
2.1. Preparation of Samples
2.2. Characterization
3. Results and Discussion
3.1. Structure
3.2. Microstructures
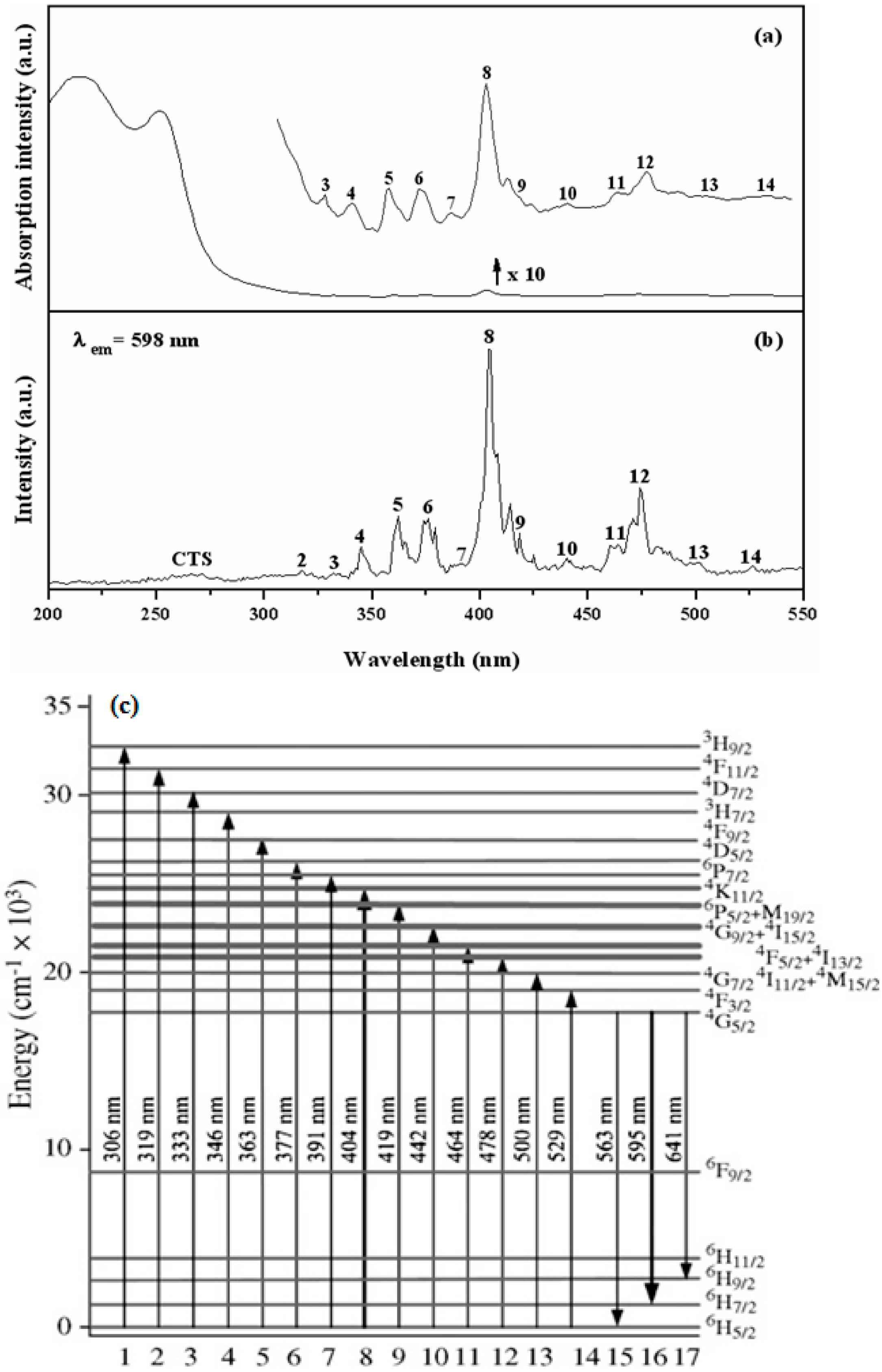
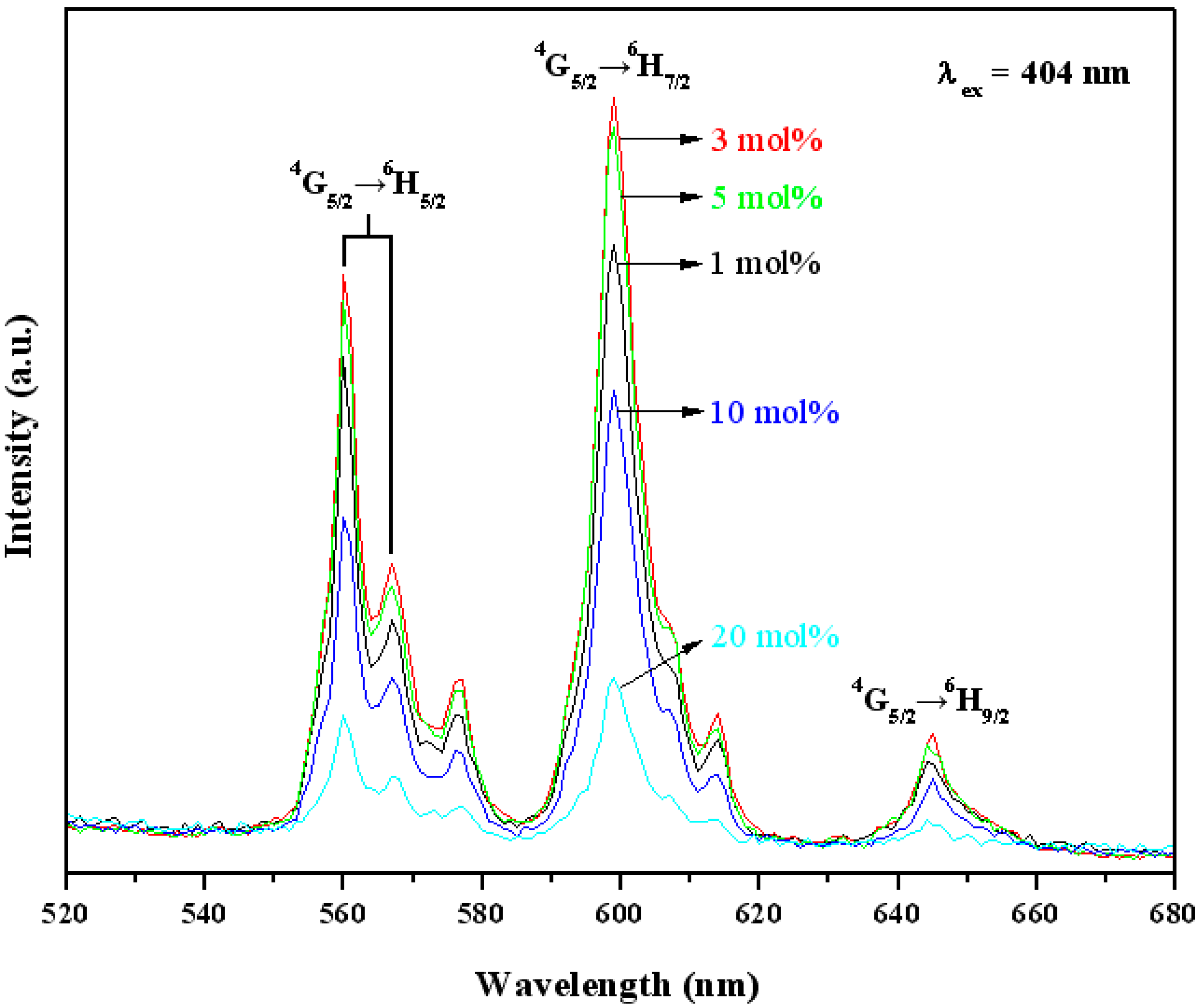
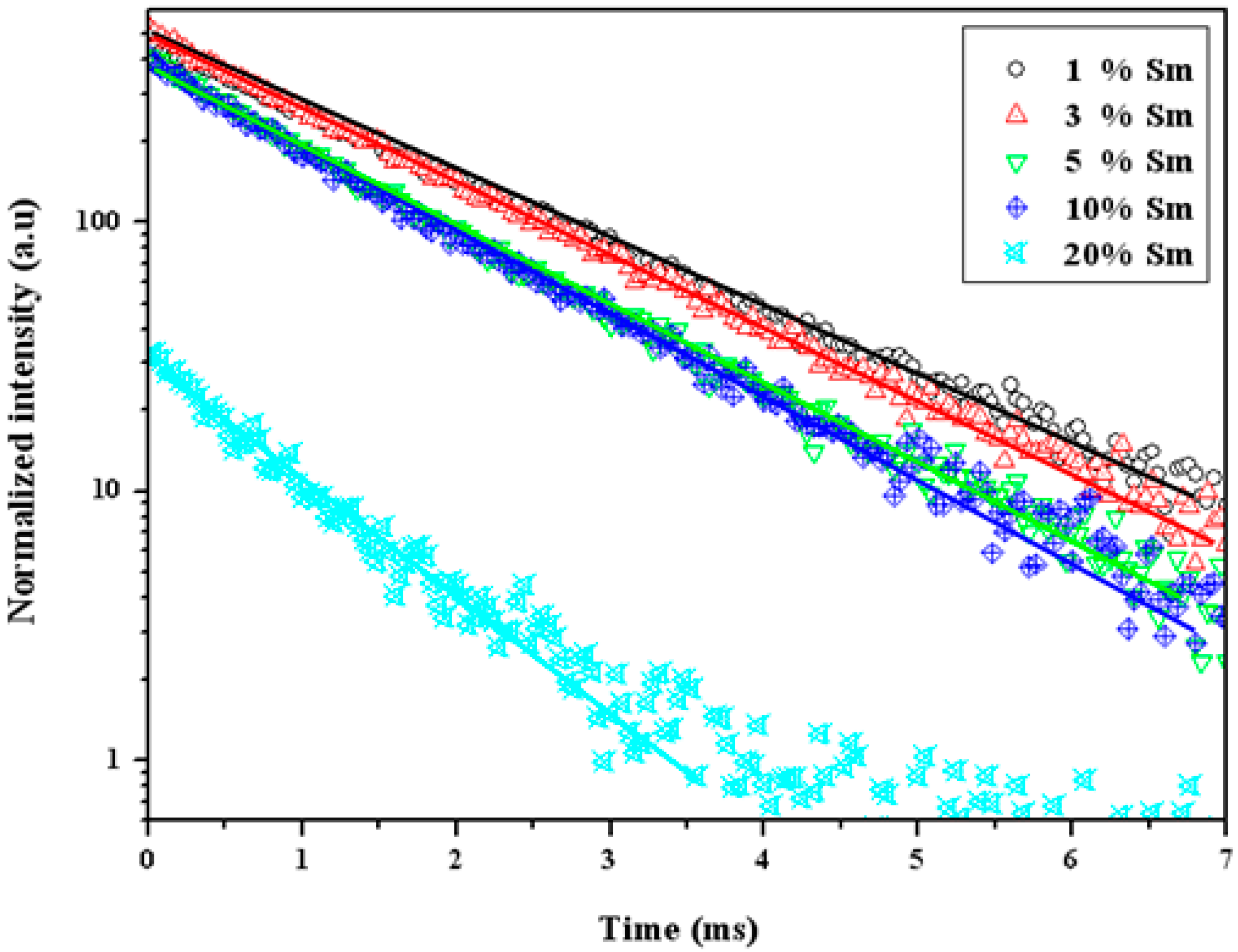
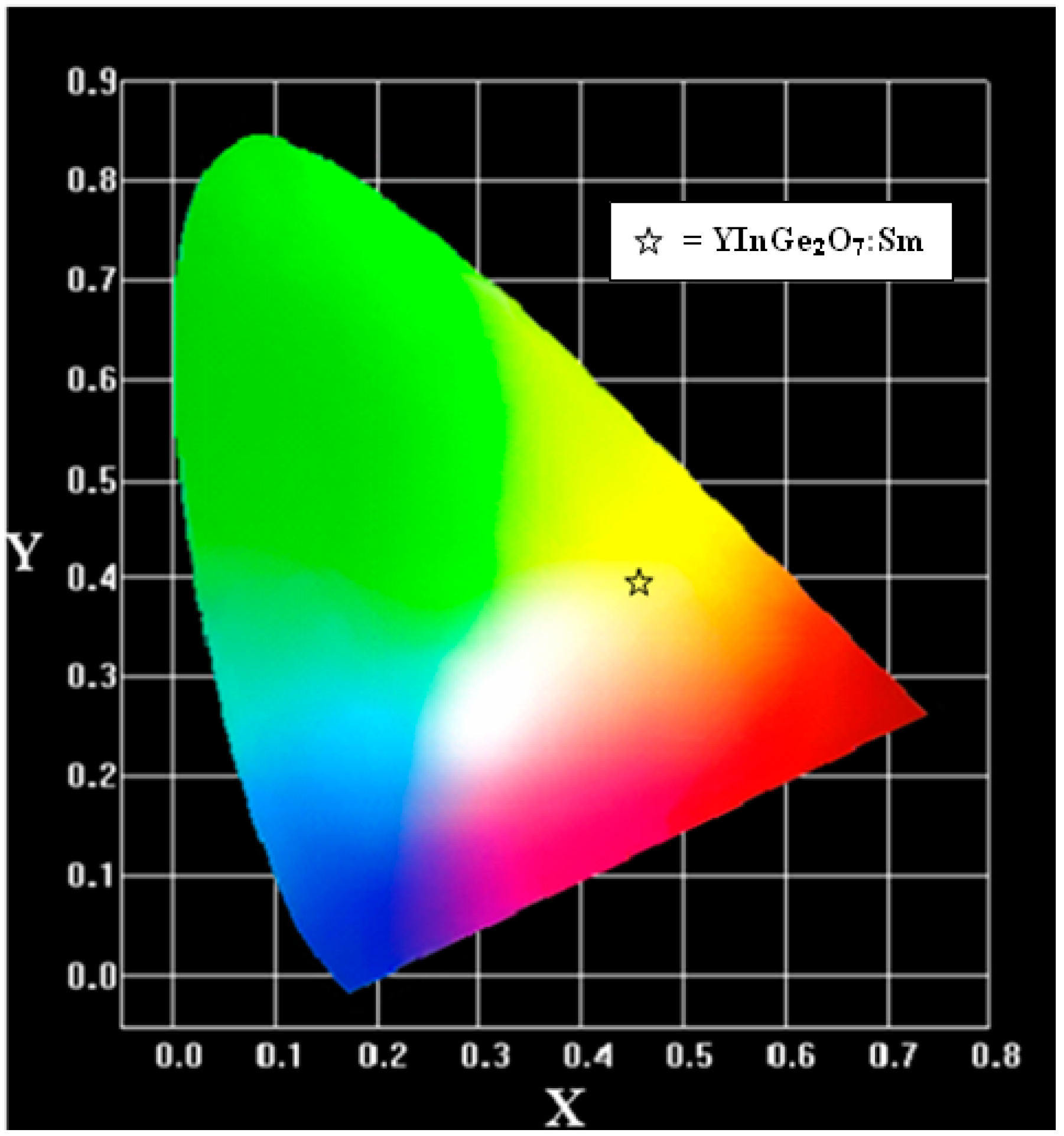
3.3. Optical Properties
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Hisamune, T. Technical trend of phosphors for plasma display panels. In Proceedings of the Ninth International Display Workshops, Hiroshima, Japan, 4–6 December 2002; p. 685. [Google Scholar]
- Yoshida, H.; Yoshimatsu, R.; Ogasawara, K. Luminescence properties of YAl3(BO3)4:Gd3+ phosphors substituted with Sc3+ ions. J. Lumin. 2007, 122–123, 488. [Google Scholar] [CrossRef]
- Song, M.S.; Park, Y.K.; Yun, J.J.; Hwang, Y.H.; Kang, B.K. Distributed circuit model for cold cathode fluorescent lamps in back-light unit of liquid crystal display. Displays 2010, 31, 104. [Google Scholar] [CrossRef]
- Tsai, M.T.; Chang, Y.S.; Chou, Y.H.; Tsai, K.M. Photoluminescence of Titanium-doped Zinc Spinel Blue-emitting Nanophosphors. J. Solid State Chem. 2014, 214, 86–90. [Google Scholar] [CrossRef]
- Kubota, S.; Hara, H.; Yamane, H.; Shimada, M. Luminescent property of Eu3+ in a new compound, (Sr0.99La1.01)Zn0.99O3.495. J. Electrochem. Soc. 2002, 149, 68. [Google Scholar] [CrossRef]
- Kim, K.Y.; Jung, H.K.; Park, H.D.; Kim, D. High luminance of new green emitting phosphor, Mg2SnO4:Mn. J. Lumin. 2002, 99, 169. [Google Scholar] [CrossRef]
- Zhang, Q.Y.; Pita, K.; Ye, W.; Que, W.X. Influence of annealing atmosphere and temperature on photoluminescence of Tb3+ or Eu3+-activated zinc silicate thin film phosphors via sol–gel method. Chem. Phys. Lett. 2002, 351, 163–170. [Google Scholar] [CrossRef]
- Juarez-Arellano, E.A.; Bucio, L.; Ruvalcaba, J.L.; Moreno-Tovar, R.; Garcia-Robledo, J.F.; Orozco, E. The crystal structure of InYGe2O7 germanate. Z. Kristallogr. 2002, 217, 201. [Google Scholar] [CrossRef]
- Chang, Y.S.; Lin, H.J.; Chai, Y.L.; Li, Y.C. Preparation and luminescent properties of europium-activated YInGe2O7 phosphors. J. Alloys Compd. 2008, 460, 421–425. [Google Scholar] [CrossRef]
- Chang, Y.S.; Lin, H.J.; Li, Y.C.; Chai, Y.L.; Tsai, Y.Y. Synthesis and luminescent properties of Tb3+-activated yttrium indium germanate phosphor. J. Solid State Chem. 2007, 180, 3076–3081. [Google Scholar] [CrossRef]
- Lin, H.J.; Chang, Y.S. Blue-Emitting Phosphor of YInGe2O7 Doped with Tm3+ Ions. Electrochem. Solid State Lett. 2007, 10, J79–J82. [Google Scholar] [CrossRef]
- Dai, P.L.; Tsai, B.S.; Tsai, Y.Y.; Chen, H.L.; Fang, T.H.; Liao, K.H.; Chang, Y.S. Synthesis and luminescence properties of YInGe2O7 phosphors activated by dysprosium ions. Opt. Mater. 2009, 32, 392–397. [Google Scholar] [CrossRef]
- Liao, J.S.; Liu, L.B.; You, H.Y.; Huang, H.P.; You, W.X. Hydrothermal preparation and luminescence property of MWO4:Sm3+ (M = Ca, Sr, Ba) red phosphors. Optik 2012, 123, 901–905. [Google Scholar] [CrossRef]
- Huang, Y.L.; Kai, W.F.; Cao, Y.G.; Jang, K.W.; Lee, H.S.; Kim, I.G.; Cho, E.J. Spectroscopic and structural studies of Sm2+ doped orthophosphate KSrPO4 crystal. J. Appl. Phys. 2008, 103, 053501. [Google Scholar] [CrossRef]
- Wang, Q.; Ci, Z.; Zhu, G.; Xin, S.; Wei, Z.; Que, M.; Wang, Y. Multicolor bright Ln3+ (Ln = Eu, Dy, Sm) activated tungstate phosphor for multifunctional applications. Opt. Mater. Express 2014, 4, 142–154. [Google Scholar] [CrossRef]
- Liu, Z.Q.; Stevens-Kalceff, M.A.; Wang, X.L.; Riesen, H. Effects of Postannealing on the Photoluminescence Properties of Coprecipitated Nanocrystalline BaFCl:Sm3+. J. Phys. Chem. A. 2013, 117, 1930–1934. [Google Scholar] [CrossRef] [PubMed]
- Shionoya, S.; Yen, W.M. Phosphor Handbook; CRC Press: Boca Raton, FL, USA, 1998. [Google Scholar]
- Blasse, G.; Brixner, L.H. X-ray excited luminescence of oxides doped with D10 ions. Mater. Chem. Phys. 1991, 28, 275–279. [Google Scholar] [CrossRef]
- Blasse, G. Do metal ions with d10 configuration luminesce? Chem. Phys. Lett. 1990, 175, 237–241. [Google Scholar] [CrossRef]
- Poulios, D.P.; Spoonhower, J.P.; Bigelow, N.P. Influence of oxygen deficiencies and hydrogen-loading on defect luminescence in irradiated Ge-doped silica glasses. J. Lumin. 2003, 101, 23–33. [Google Scholar] [CrossRef]
- Yang, L.Z.; Yu, X.B.; Yang, S.P.; Zhou, C.L.; Zhou, P.G.; Gao, W.J.; Ye, P. Preparation and luminescence properties of LED conversion novelphosphors SrZnO2:Sm. Mater. Lett. 2008, 62, 907–910. [Google Scholar] [CrossRef]
- Lei, B.; Liu, Y.; Tang, G.; Ye, Z.; Shi, C. Spectra and long-lasting properties of Sm3+-doped yttrium oxysulfide phosphor. Mater. Chem. Phys. 2004, 87, 227–232. [Google Scholar] [CrossRef]
- Zhang, H.; Fu, X.; Niu, S.; Xin, Q. Synthesis and luminescent properties of nanosized YVO4:Ln (Ln = Sm, Dy). J. Alloys Compd. 2008, 457, 61–65. [Google Scholar] [CrossRef]
- Li, X.; Yang, Z.; Guan, L.; Guo, Q.; Liu, C.; Li, P. Fabrication and luminescence properties of red emitting phosphor Y2O2S:Sm3+ for white LED by combustion method. J. Alloys Compd. 2008, 464, 565–568. [Google Scholar] [CrossRef]
- Zhou, Y.; Lin, J.; Wang, S. Energy transfer and up conversion luminescence properties of Y2O3:Sm and Gd2O3:Sm phosphors. J. Solid State Chem. 2003, 171, 391–395. [Google Scholar] [CrossRef]
- Moix, J.M.; Ma, J.; Cao, J.S. Exact evaluation of the energy transfer rates and steady state absorption and emission spectra in the b850 photosynthetic complexes of LH2. J. Chem. Phys. 2015, 142, 094108. [Google Scholar] [CrossRef] [PubMed]



© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shih, H.-R.; Chang, Y.-S. Structure and Photoluminescence Properties of Sm3+ Ion-Doped YInGe2O7 Phosphor. Materials 2017, 10, 779. https://doi.org/10.3390/ma10070779
Shih H-R, Chang Y-S. Structure and Photoluminescence Properties of Sm3+ Ion-Doped YInGe2O7 Phosphor. Materials. 2017; 10(7):779. https://doi.org/10.3390/ma10070779
Chicago/Turabian StyleShih, Hung-Rung, and Yee-Shin Chang. 2017. "Structure and Photoluminescence Properties of Sm3+ Ion-Doped YInGe2O7 Phosphor" Materials 10, no. 7: 779. https://doi.org/10.3390/ma10070779
APA StyleShih, H.-R., & Chang, Y.-S. (2017). Structure and Photoluminescence Properties of Sm3+ Ion-Doped YInGe2O7 Phosphor. Materials, 10(7), 779. https://doi.org/10.3390/ma10070779



