Coaxial MoS2@Carbon Hybrid Fibers: A Low-Cost Anode Material for High-Performance Li-Ion Batteries
Abstract
:1. Introduction
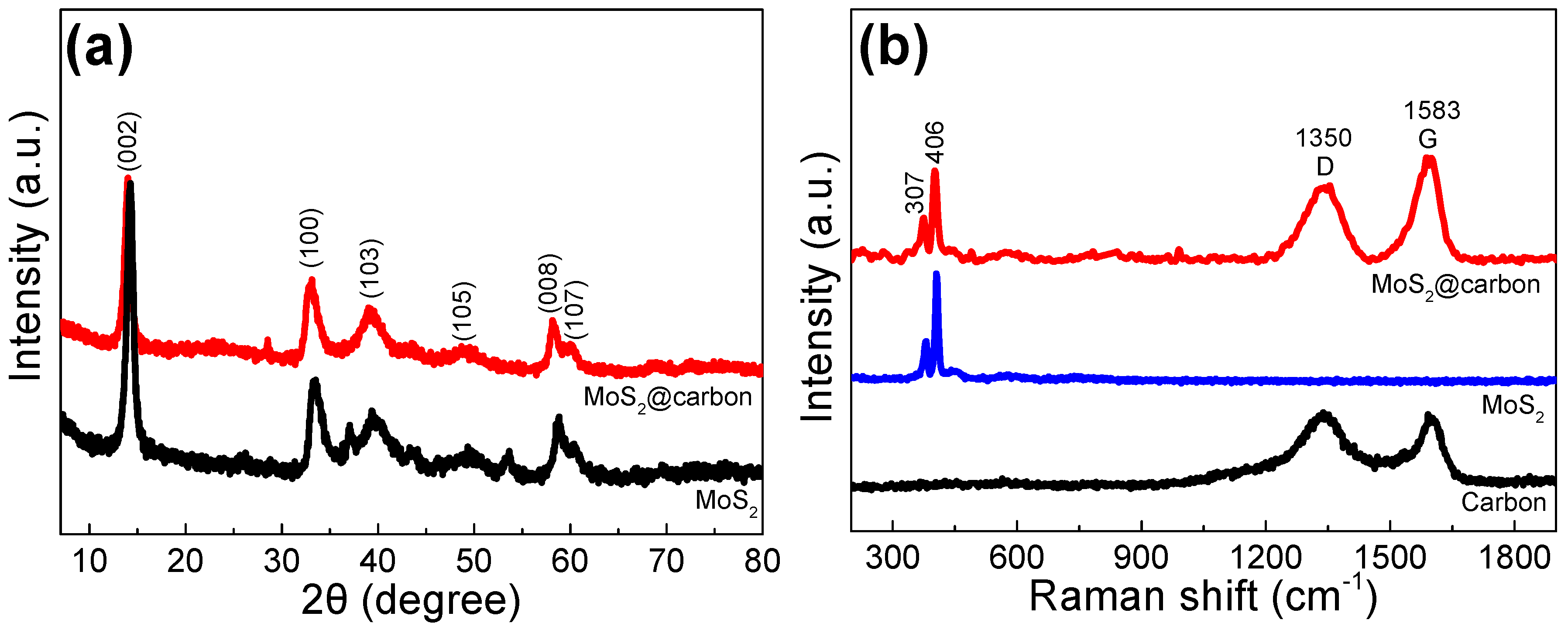
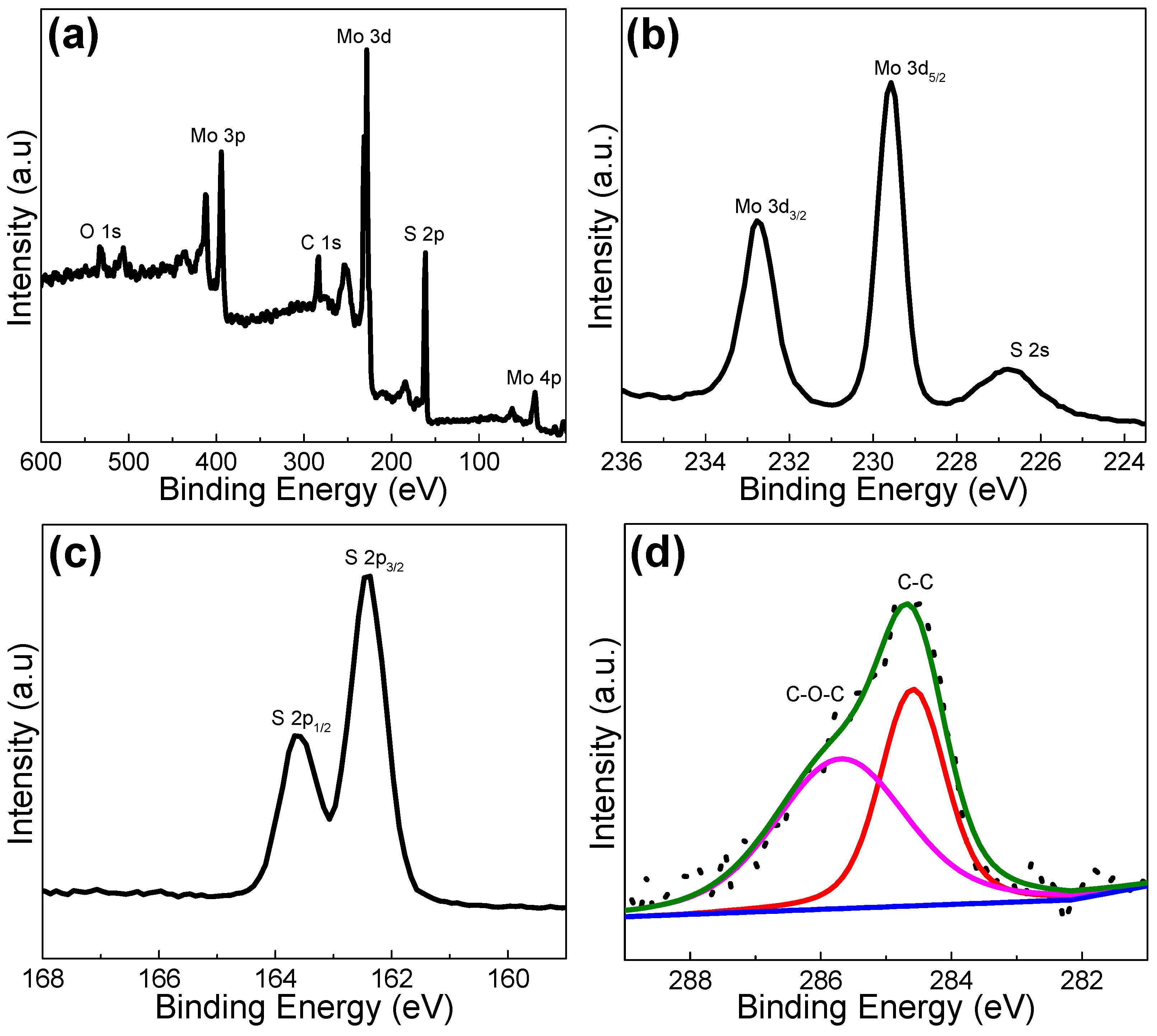
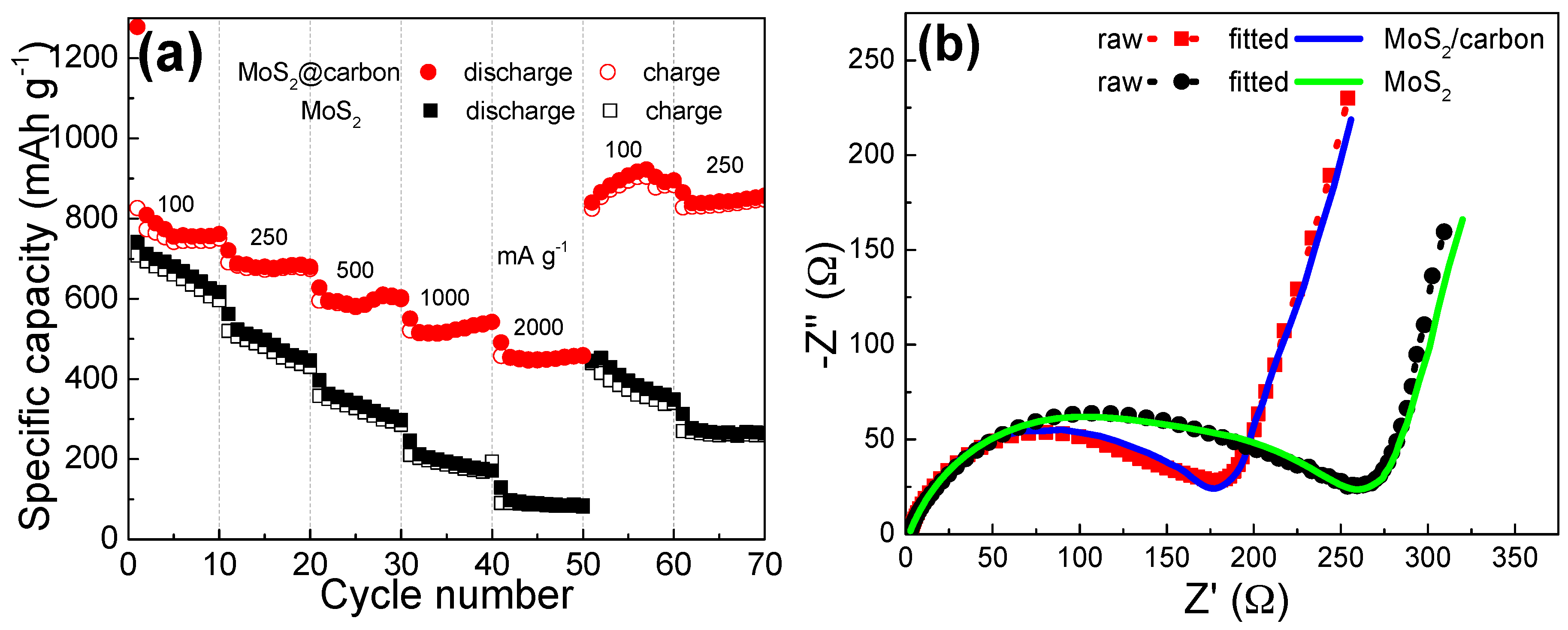
2. Results and Discussion
3. Materials and Methods
3.1. Synthesis of the MoS2@Carbon Composites
3.2. Material Characterization
3.3. Electrochemical Measurements
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Nitta, N.; Wu, F.; Lee, J.T.; Yushin, G. Li-ion battery materials: Present and future. Mater. Today 2015, 18, 252–264. [Google Scholar] [CrossRef]
- Wang, J.-G.; Jin, D.; Zhou, R.; Li, X.; Liu, X.R.; Shen, C.; Xie, K.; Li, B.; Kang, F.; Wei, B. Highly flexible graphene/Mn3O4 nanocomposite membrane as advanced anodes for Li-ion batteries. ACS Nano 2016, 10, 6227–6234. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.-G.; Jin, D.; Liu, H.; Zhang, C.; Zhou, R.; Shen, C.; Xie, K.; Wei, B. All-manganese-based Li-ion batteries with high rate capability and ultralong cycle life. Nano Energy 2016, 22, 524–532. [Google Scholar] [CrossRef]
- Reddy, M.V.; Subba Rao, G.V.; Chowdari, B.V. Metal oxides and oxysalts as anode materials for Li ion batteries. Chem. Rev. 2013, 113, 5364–5457. [Google Scholar] [CrossRef] [PubMed]
- Xu, B.; Qian, D.; Wang, Z.; Meng, Y.S. Recent progress in cathode materials research for advanced lithium ion batteries. Mat. Sci. Eng. R 2012, 73, 51–65. [Google Scholar] [CrossRef]
- Wang, J.; Hou, X.; Zhang, M.; Li, Y.; Wu, Y.; Liu, X.; Hu, S. 3-aminopropyltriethoxysilane-assisted Si@SiO2/CNTs hybrid microspheres as superior anode materials for Li-ion batteries. Silicon 2016, 9, 97–104. [Google Scholar] [CrossRef]
- Sun, Y.-H.; Liu, S.; Zhou, F.-C.; Nan, J.-M. Electrochemical performance and structure evolution of core-shell nano-ring α-Fe2O3@carbon anodes for lithium-ion batteries. Appl. Surf. Sci. 2016, 390, 175–184. [Google Scholar] [CrossRef]
- Wang, J.-G.; Zhang, C.; Jin, D.; Xie, K.; Wei, B. Synthesis of ultralong MnO/C coaxial nanowires as freestanding anodes for high-performance lithium ion batteries. J. Mater. Chem. A 2015, 3, 13699–13705. [Google Scholar] [CrossRef]
- Ramakrishna Matte, H.S.S.; Gomathi, A.; Manna, A.K.; Late, D.J.; Datta, R.; Pati, S.K.; Rao, C.N.R. MoS2 and WS2 analogues of graphene. Angew. Chem. Int. Ed. 2010, 122, 4153–4156. [Google Scholar] [CrossRef]
- Bhandavat, R.; David, L.; Singh, G. Synthesis of surface-functionalized WS2 nanosheets and performance as li-ion battery anodes. J. Phys. Chem. Lett. 2012, 3, 1523–1530. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Zou, R.; Xia, W.; Ma, J.; Qiu, B.; Mahmood, A.; Zhao, R.; Yang, Y.; Xia, D.; Xu, Q. Facile synthesis of ultrasmall CoS2 nanoparticles within thin N-doped porous carbon shell for high performance lithium-ion batteries. Small 2015, 11, 2511–2517. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.; Yang, J.F.; Lou, X.W. Formation of CoS2 nanobubble hollow prisms for highly reversible lithium storage. Angew. Chem. Int. Ed. 2016, 55, 13422–13426. [Google Scholar] [CrossRef] [PubMed]
- Tan, C.; Zhang, H. Two-dimensional transition metal dichalcogenide nanosheet-based composites. Chem. Soc. Rev. 2015, 44, 2713–2731. [Google Scholar] [CrossRef] [PubMed]
- Jin, B.; Zhou, X.; Huang, L.; Licklederer, M.; Yang, M.; Schmuki, P. Aligned MoOx/MoS2 core-shell nanotubular structures with a high density of reactive sites based on self-ordered anodic molybdenum oxide nanotubes. Angew. Chem. Int. Ed. 2016, 55, 12252–12256. [Google Scholar] [CrossRef] [PubMed]
- Hu, S.; Chen, W.; Zhou, J.; Yin, F.; Uchaker, E.; Zhang, Q.; Cao, G. Preparation of carbon coated MoS2 flower-like nanostructure with self-assembled nanosheets as high-performance lithium-ion battery anodes. J. Mater. Chem. A 2014, 2, 7862–7872. [Google Scholar] [CrossRef]
- Stephenson, T.; Li, Z.; Olsen, B.; Mitlin, D. Lithium ion battery applications of molybdenum disulfide (MoS2) nanocomposites. Energy Environ. Sci. 2014, 7, 209–231. [Google Scholar] [CrossRef]
- Xiong, F.; Cai, Z.; Qu, L.; Zhang, P.; Yuan, Z.; Asare, O.K.; Xu, W.; Lin, C.; Mai, L. Three-dimensional crumpled reduced graphene oxide/MoS2 nanoflowers: A stable anode for lithium-ion batteries. ACS Appl. Mater. Inter. 2015, 7, 12625–12630. [Google Scholar] [CrossRef] [PubMed]
- Xie, D.; Wang, D.H.; Tang, W.J.; Xia, X.H.; Zhang, Y.J.; Wang, X.L.; Gu, C.D.; Tu, J.P. Binder-free network-enabled MoS2-ppy-rGO ternary electrode for high capacity and excellent stability of lithium storage. J. Power Sources 2016, 307, 510–518. [Google Scholar] [CrossRef]
- Jeong, J.M.; Lee, K.G.; Chang, S.J.; Kim, J.W.; Han, Y.K.; Lee, S.J.; Choi, B.G. Ultrathin sandwich-like MoS2@N-doped carbon nanosheets for anodes of lithium ion batteries. Nanoscale 2015, 7, 324–329. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; He, X.; Hanlon, D.; Harvey, A.; Khan, U.; Li, Y.; Coleman, J.N. Electrical, mechanical, and capacity percolation leads to high-performance MoS2/nanotube composite lithium ion battery electrodes. ACS Nano 2016, 10, 5980–5990. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Zhao, Y.; Jiao, L.; Chen, J. A graphene-like MoS2/graphene nanocomposite as a high performance anode for lithium ion batteries. J. Mater. Chem. A 2014, 2, 13109. [Google Scholar] [CrossRef]
- Li, H.; Yu, K.; Fu, H.; Guo, B.; Lei, X.; Zhu, Z. MoS2/graphene hybrid nanoflowers with enhanced electrochemical performances as anode for lithium-ion batteries. J. Phys. Chem. C 2015, 119, 7959–7968. [Google Scholar] [CrossRef]
- Zhu, C.; Mu, X.; van Aken, P.A.; Yu, Y.; Maier, J. Single-layered ultrasmall nanoplates of MoS2 embedded in carbon nanofibers with excellent electrochemical performance for lithium and sodium storage. Angew. Chem. Int. Ed. 2014, 53, 2152–2156. [Google Scholar] [CrossRef] [PubMed]
- Kong, J.; Zhao, C.; Wei, Y.; Lu, X. MoS2 nanosheets hosted in polydopamine-derived mesoporous carbon nanofibers as lithium-ion battery anodes: Enhanced MoS2 capacity utilization and underlying mechanism. ACS Appl. Mater. Inter. 2015, 7, 24279–24287. [Google Scholar] [CrossRef] [PubMed]
- Bian, X.; Zhu, J.; Liao, L.; Scanlon, M.D.; Ge, P.; Ji, C.; Girault, H.H.; Liu, B. Nanocomposite of MoS2 on ordered mesoporous carbon nanospheres: A highly active catalyst for electrochemical hydrogen evolution. Electrochem. Commu. 2012, 22, 128–132. [Google Scholar] [CrossRef]
- Xie, X.; Ao, Z.; Su, D.; Zhang, J.; Wang, G. MoS2/graphene composite anodes with enhanced performance for sodium-ion batteries: The role of the two-dimensional heterointerface. Adv. Func. Mater. 2015, 25, 1393–1403. [Google Scholar] [CrossRef]
- Qu, Q.; Qian, F.; Yang, S.; Gao, T.; Liu, W.; Shao, J.; Zheng, H. Layer-by-layer polyelectrolyte assisted growth of 2D ultrathin MoS2 nanosheets on various 1D carbons for superior Li-storage. ACS Appl. Mater. Int. 2016, 8, 1398–1405. [Google Scholar] [CrossRef] [PubMed]
- Xie, X.; Makaryan, T.; Zhao, M.; van Aken, K.L.; Gogotsi, Y.; Wang, G. MoS2 nanosheets vertically aligned on carbon paper: A freestanding electrode for highly reversible sodium-ion batteries. Adv. Energy Mater. 2016, 6, 1502161. [Google Scholar] [CrossRef]
- Li, Y.; Wang, H.; Xie, L.; Liang, Y.; Hong, G.; Dai, H. MoS2 nanoparticles grown on graphene: An advanced catalyst for the hydrogen evolution reaction. J. Am. Chem. Soc. 2011, 133, 7296–7299. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Qin, J.; Zhang, X.; Shi, C.; Liu, E.; Li, J.; Zhao, N.; He, C. 2D space-confined synthesis of few-layer MoS2 anchored on carbon nanosheet for lithium-ion battery anode. ACS Nano 2015, 9, 3837–3848. [Google Scholar] [CrossRef] [PubMed]
- Shan, T.; Xin, S.; You, Y.; Cong, H.; Yu, S. Arumugam Manthiram. Combining nitrogen-doped graphene sheets and MoS2: A unique film–foam–film structure for enhanced lithium storage. Angew. Chem. Int. Ed. 2016, 55, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.-G.; Kang, F.; Wei, B. Engineering of MnO2-based nanocomposites for high performance supercapacitors. Prog. Mater. Sci. 2015, 74, 51–124. [Google Scholar] [CrossRef]
- Zhu, H.; Lyu, F.; Du, M.; Zhang, M.; Wang, Q.; Yao, J.; Guo, B. Design of two-dimensional ultrathin MoS2 nanoplates fabricated within one-dimensional carbon nanofibers with thermosensitive morphology: High-performance electrocatalysts for the hydrogen evolution reaction. ACS Appl. Mater. Int. 2014, 6, 22126–22137. [Google Scholar] [CrossRef] [PubMed]
- Ramkumar, M.; Minakshi, M. A biopolymer gel-decorated cobalt molybdate nanowafer: Effective graft polymer cross-linked with an organic acid for better energy storage. New J. Chem. 2016, 44, 2863–2877. [Google Scholar] [CrossRef]
- Barmi, M.J.; Minakshi, M. Tuning the Redox Properties of the Nanostructured CoMoO4 Electrode: Effects of Surfactant Content and Synthesis Temperature. ChemPlusChem 2016, 81, 964–977. [Google Scholar] [CrossRef]
- Ramkumar, M.; Minakshi, M. Fabrication of ultrathin CoMoO4 nanosheets modified with chitosan and their improved performance in energy storage device. Dalton Trans. 2015, 44, 6158–6168. [Google Scholar] [CrossRef] [PubMed]
- Fang, Y.; Lv, Y.; Gong, F.; Elzatahry, A.A.; Zheng, G.; Zhao, D. Synthesis of 2D-mesoporous-carbon/MoS2 heterostructures with well-defined interfaces for high-performance lithium-ion batteries. Adv. Mater. 2016, 28, 9385–9390. [Google Scholar] [CrossRef] [PubMed]
- Jiang, L.; Lin, B.; Li, X.; Song, X.; Xia, H.; Li, L.; Zeng, H. Monolayer MoS2-graphene hybrid aerogels with controllable porosity for lithium-ion batteries with high reversible capacity. ACS Appl. Mater. Int. 2016, 8, 2680–2687. [Google Scholar] [CrossRef]
- Zhou, F.; Xin, S.; Liang, H.W.; Song, L.T.; Yu, S.H. Carbon nanofibers decorated with molybdenum disulfide nanosheets: Synergistic lithium storage and enhanced electrochemical performance. Angew. Chem. Int. Ed. 2014, 53, 11552–11556. [Google Scholar] [CrossRef] [PubMed]
- Zuo, X.; Chang, K.; Zhao, J.; Xie, Z.; Tang, H.; Li, B.; Chang, Z. Bubble-template-assisted synthesis of hollow fullerene-like MoS2 nanocages as a lithium ion battery anode material. J. Mater. Chem. A 2016, 4, 51–58. [Google Scholar] [CrossRef]
- Teng, Y.; Zhao, H.; Zhang, Z.; Li, Z.; Xia, Q.; Zhang, Y.; Zhao, L.; Du, X.; Du, Z.; Lv, P.; Świerczek, K. MoS2 Nanosheets vertically grown on graphene sheets for lithium-ion battery anodes. ACS Nano 2016, 10, 8526–8535. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Fan, Z.; Yu, X.; Ding, S.; Yu, D.; Lou, X. A nanosheets-on-channel architecture constructed from MoS2 and CMK-3 for high-capacity and long-cycle-life lithium storage. Adv. Energy Mater. 2014, 4, 1400902. [Google Scholar] [CrossRef]
- Chang, K.; Chen, W. l-cysteine-assisted synthesis of layered MoS2/graphene composites with excellent electrochemical performances for lithium ion batteries. ACS Nano 2011, 5, 4720–4728. [Google Scholar] [CrossRef] [PubMed]
- Ji, L.; Lin, Z.; Alcoutlabi, M.; Zhang, X. Recent developments in nanostructured anode materials for rechargeable lithium-ion batteries. Energy Environ. Sci. 2011, 4, 2682–2699. [Google Scholar]
- Agubra, V.A.; Zuniga, L.; Garza, D.D.; Gallegos, L.; Pokhrel, M.; Alcoutlabi, M. Forcespinning: A new method for the mass production of Sn/C composite nanofiber anodes for lithium ion batteries. Solid State Ion. 2016, 286, 72–82. [Google Scholar] [CrossRef]
- Agubra, V.A.; Zuniga, L.; Flores, D.; Villareal, J.; Alcoutlabi, M. Composite nanofibers as advanced materials for Li-ion, Li-O2 and Li-S batteries. Electrochim. Acta 2016, 192, 529–550. [Google Scholar] [CrossRef]
- Li, J.-C.; Hou, P.X.; Zhao, S.Y.; Liu, C.; Tang, D.M.; Cheng, M.; Zhang, F.; Cheng, H.M. A 3D bi-functional porous N-doped carbon microtube sponge electrocatalyst for oxygen reduction and oxygen evolution reactions. Energy Environ. Sci. 2016, 9, 3079–3084. [Google Scholar] [CrossRef]


© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhou, R.; Wang, J.-G.; Liu, H.; Liu, H.; Jin, D.; Liu, X.; Shen, C.; Xie, K.; Wei, B. Coaxial MoS2@Carbon Hybrid Fibers: A Low-Cost Anode Material for High-Performance Li-Ion Batteries. Materials 2017, 10, 174. https://doi.org/10.3390/ma10020174
Zhou R, Wang J-G, Liu H, Liu H, Jin D, Liu X, Shen C, Xie K, Wei B. Coaxial MoS2@Carbon Hybrid Fibers: A Low-Cost Anode Material for High-Performance Li-Ion Batteries. Materials. 2017; 10(2):174. https://doi.org/10.3390/ma10020174
Chicago/Turabian StyleZhou, Rui, Jian-Gan Wang, Hongzhen Liu, Huanyan Liu, Dandan Jin, Xingrui Liu, Chao Shen, Keyu Xie, and Bingqing Wei. 2017. "Coaxial MoS2@Carbon Hybrid Fibers: A Low-Cost Anode Material for High-Performance Li-Ion Batteries" Materials 10, no. 2: 174. https://doi.org/10.3390/ma10020174
APA StyleZhou, R., Wang, J.-G., Liu, H., Liu, H., Jin, D., Liu, X., Shen, C., Xie, K., & Wei, B. (2017). Coaxial MoS2@Carbon Hybrid Fibers: A Low-Cost Anode Material for High-Performance Li-Ion Batteries. Materials, 10(2), 174. https://doi.org/10.3390/ma10020174







