Study of Thermal Electrical Modified Etching for Glass and Its Application in Structure Etching
Abstract
:1. Introduction
2. Materials and Methods
3. Results and Discussion
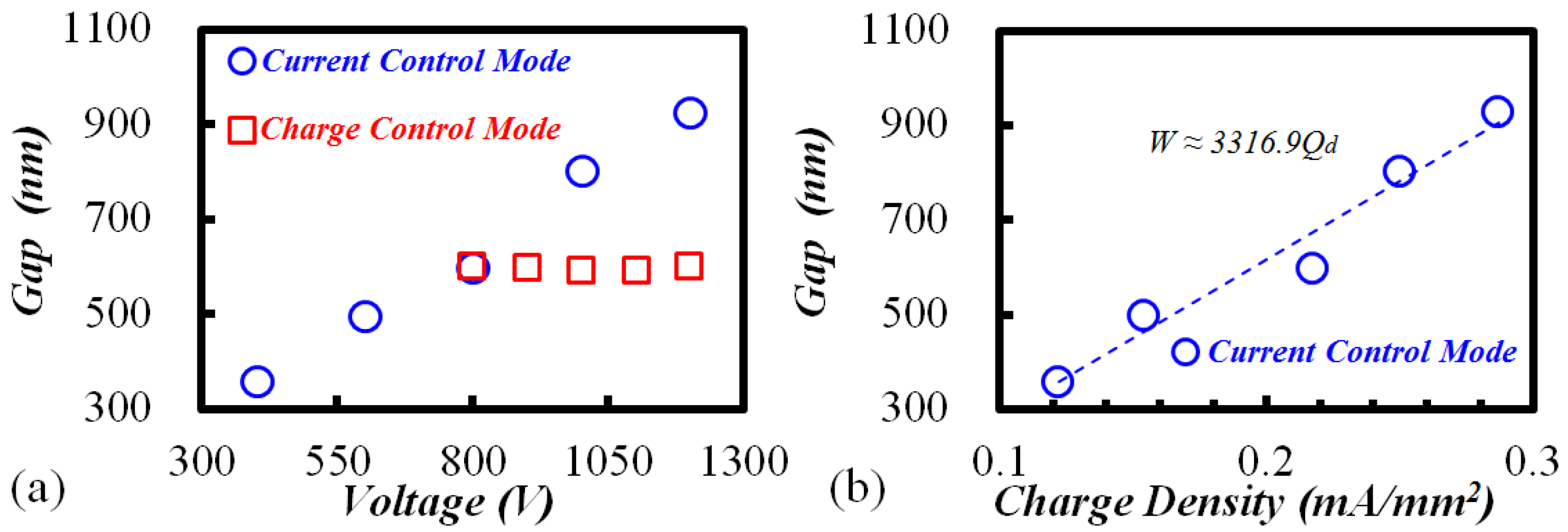
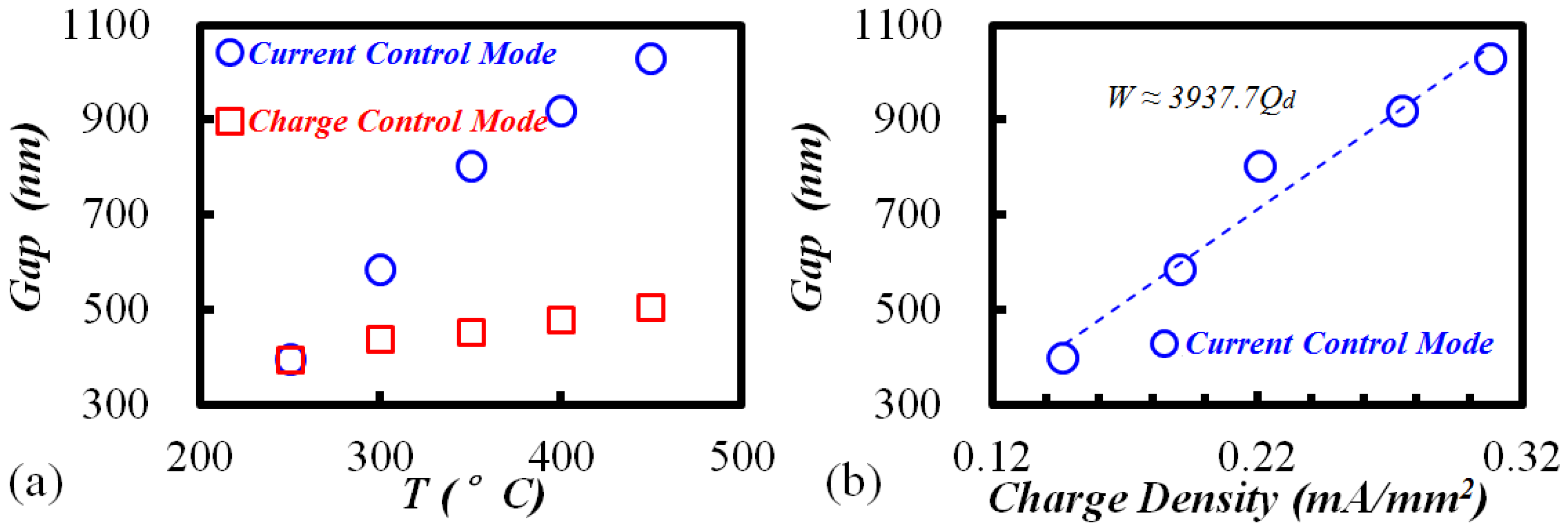
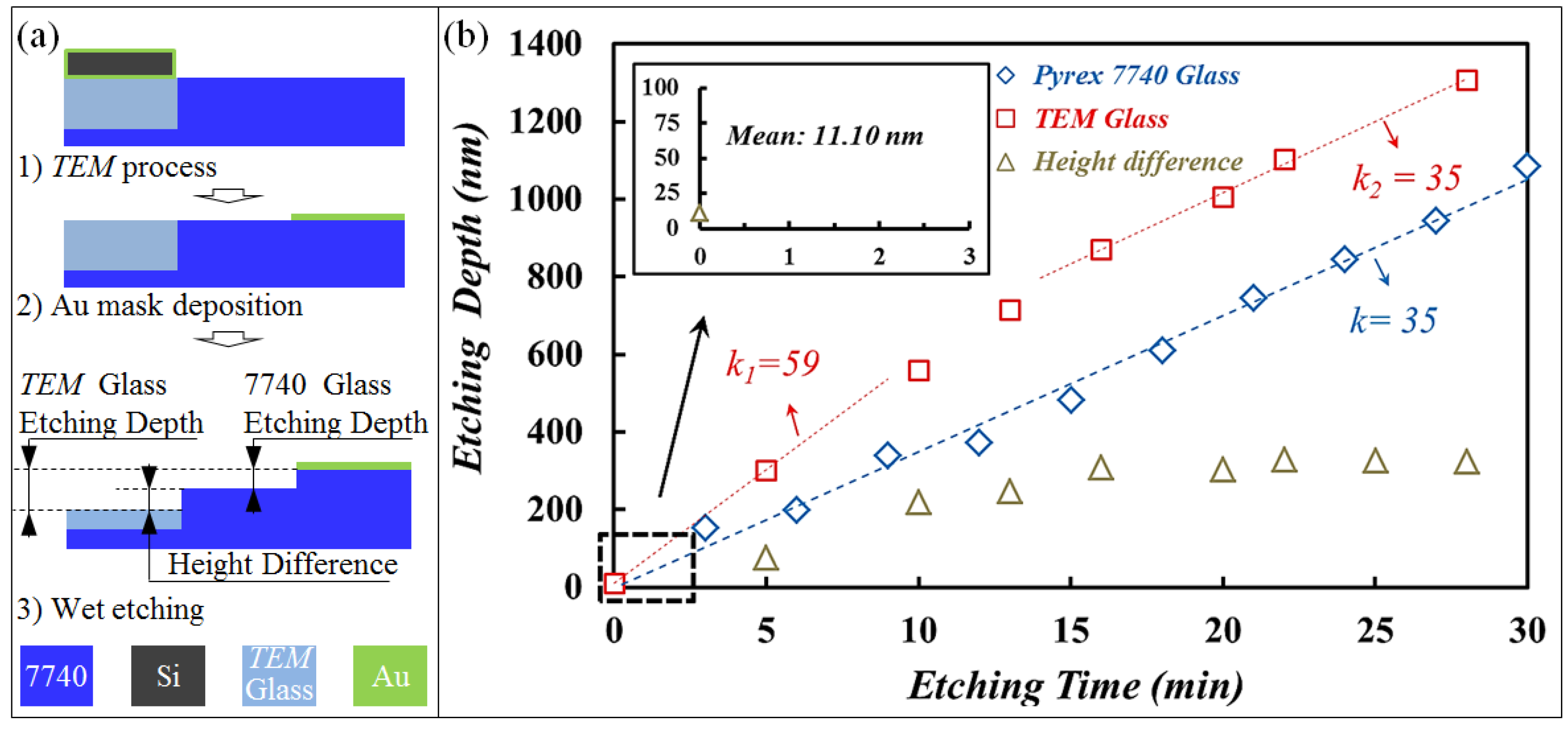
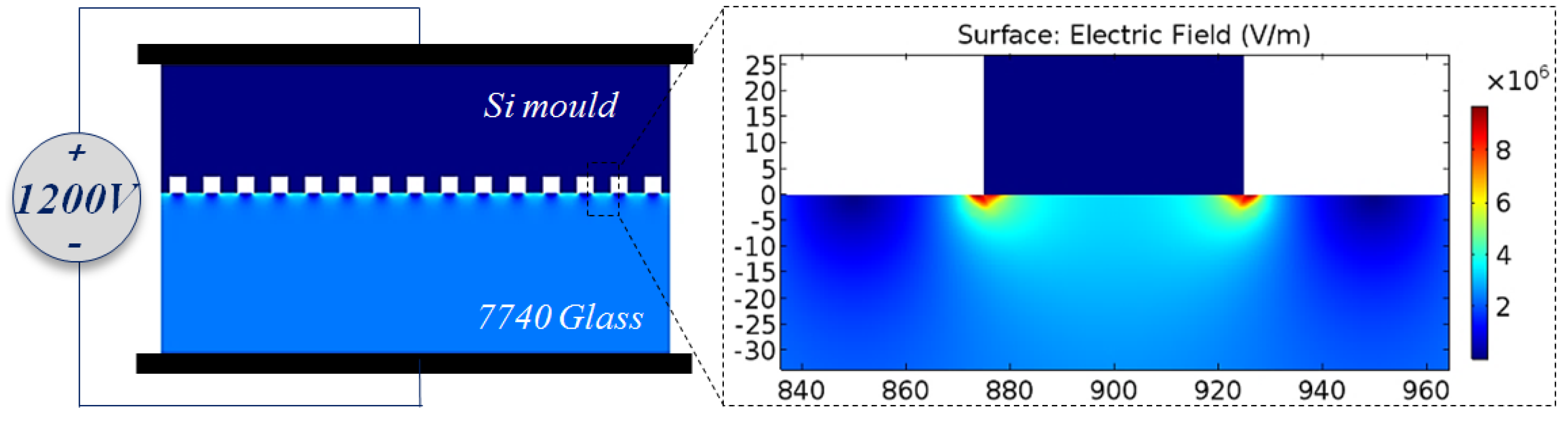
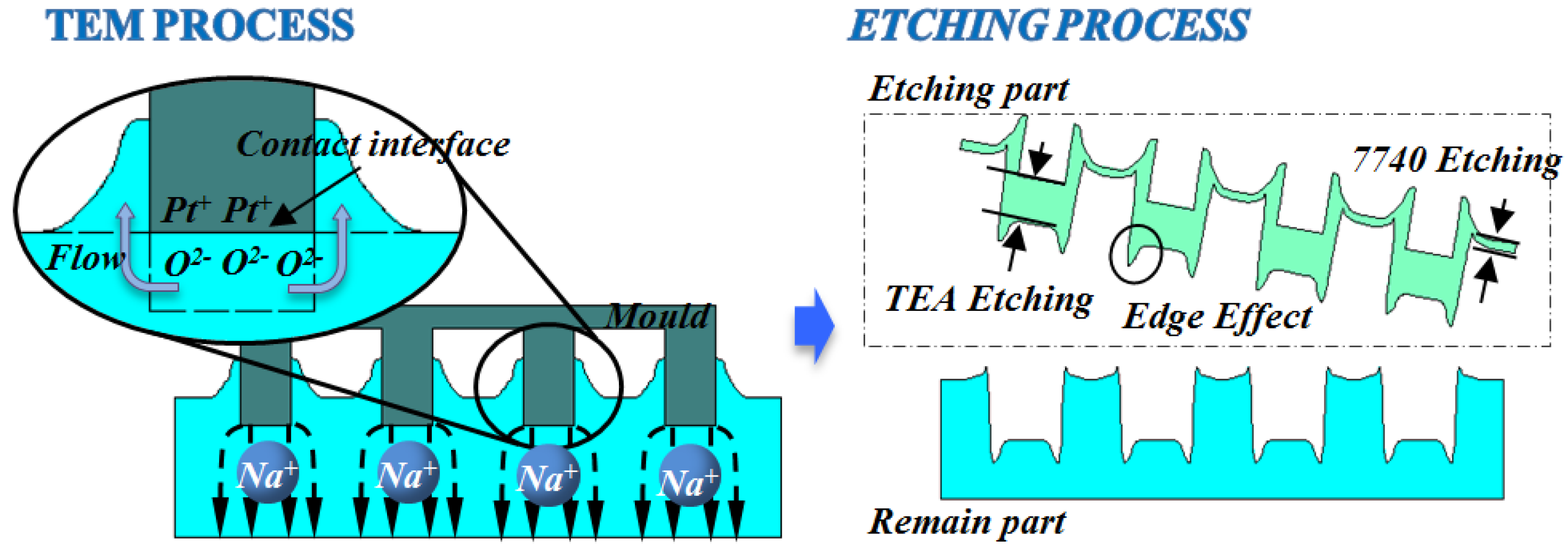
3.1. Phenomenon of TEM Etching in Anodic Bonding
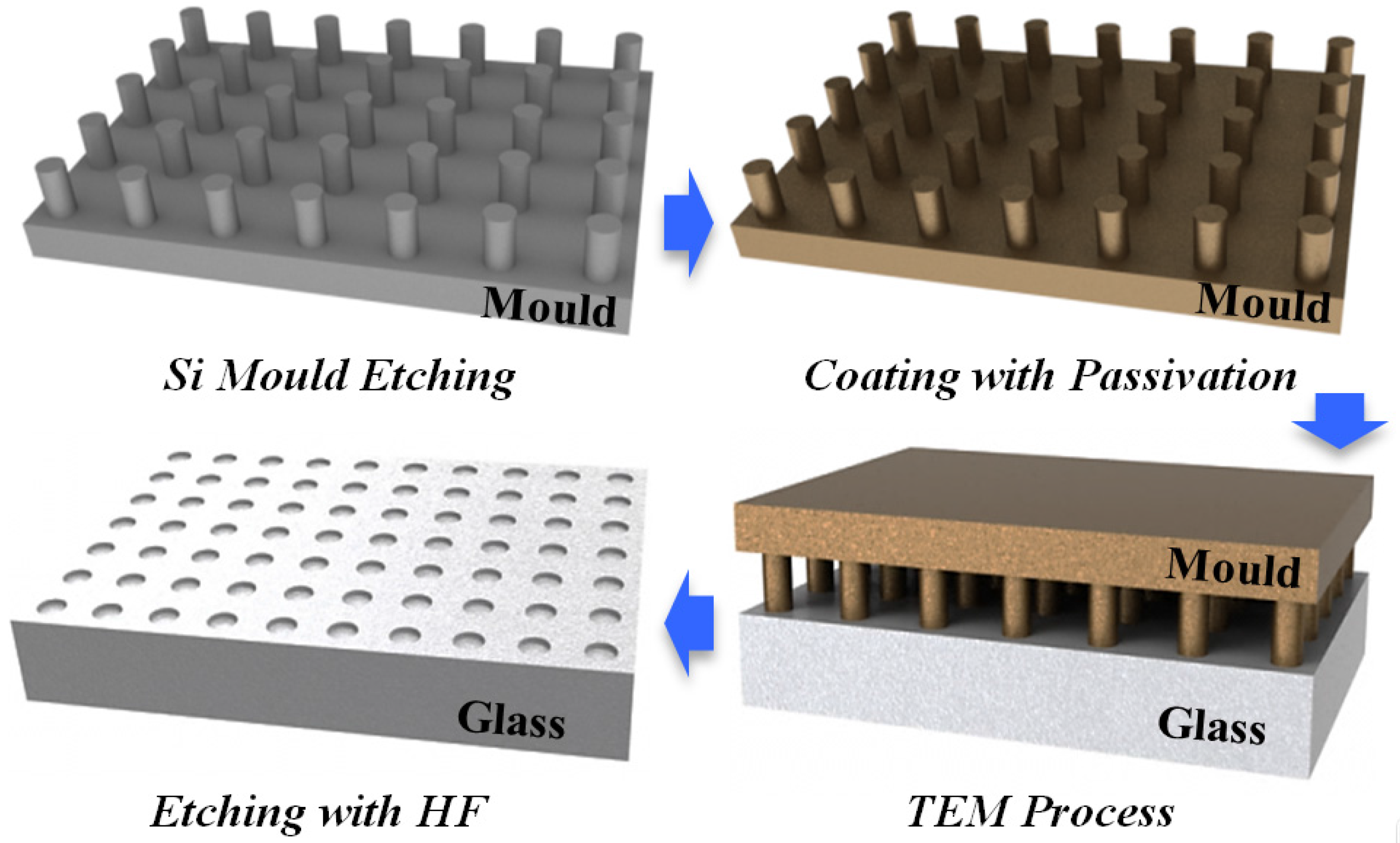
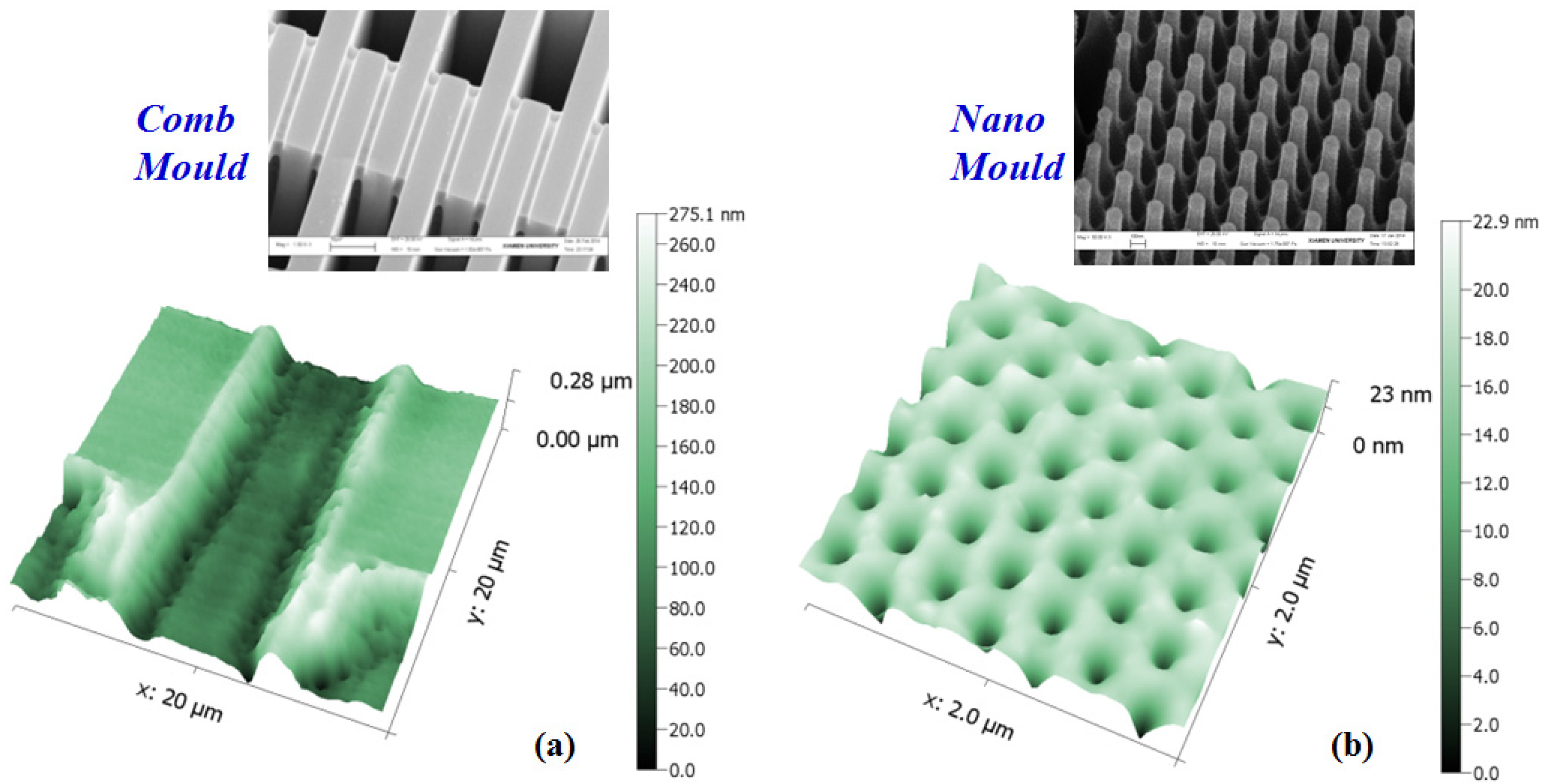
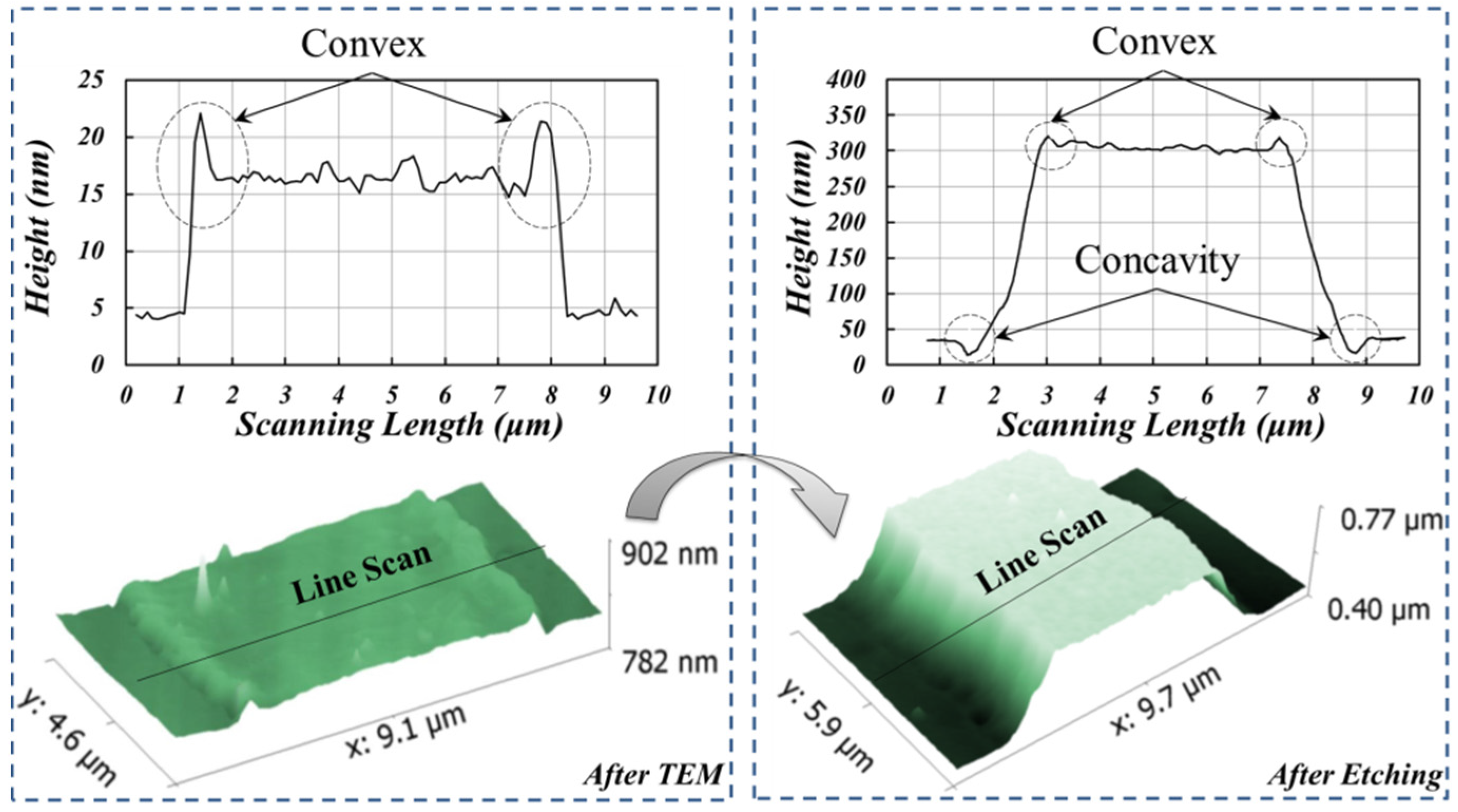
3.2. A Novel Method for Micro/Nano Structure Micromachining by TEM Etching
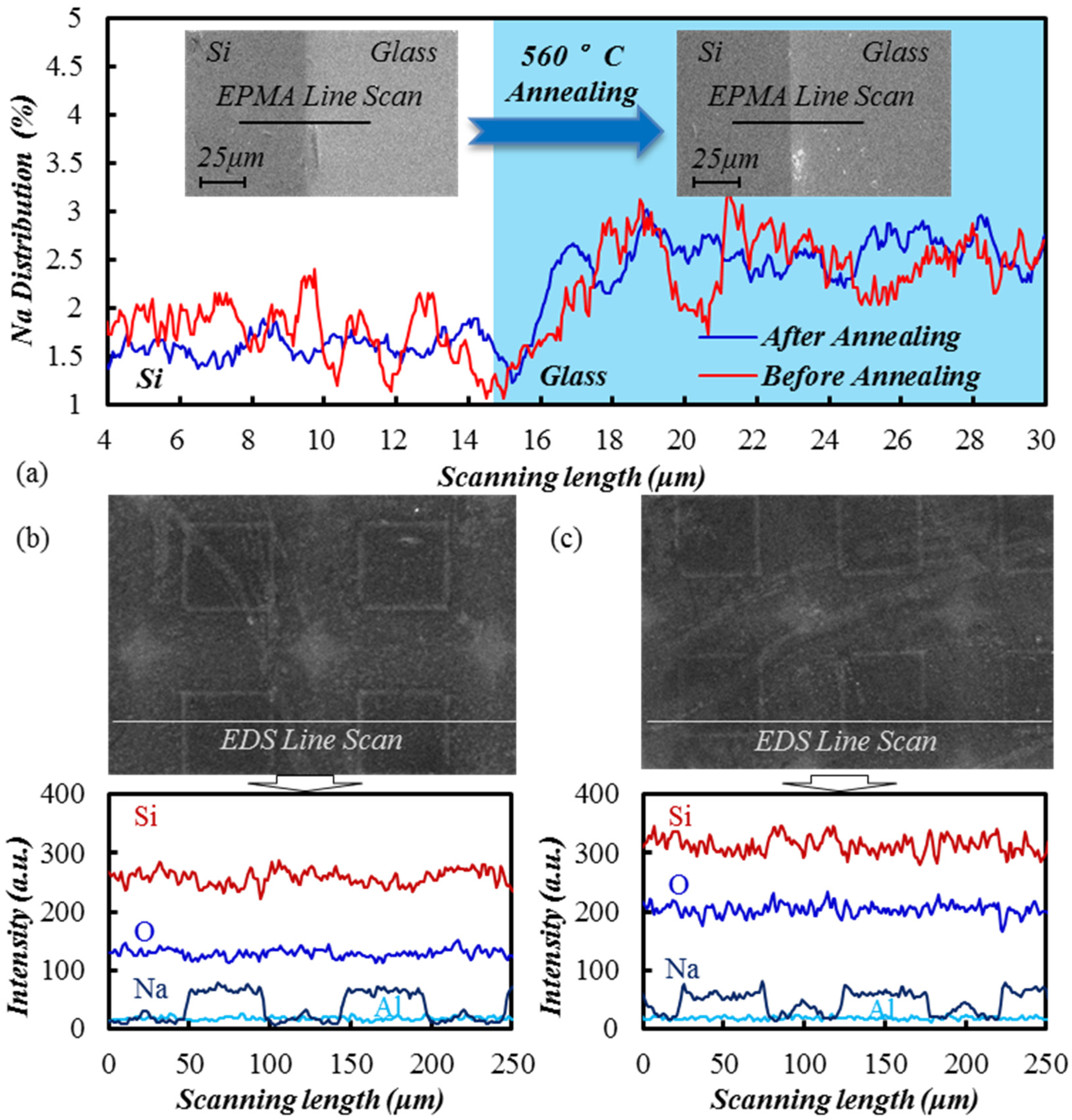
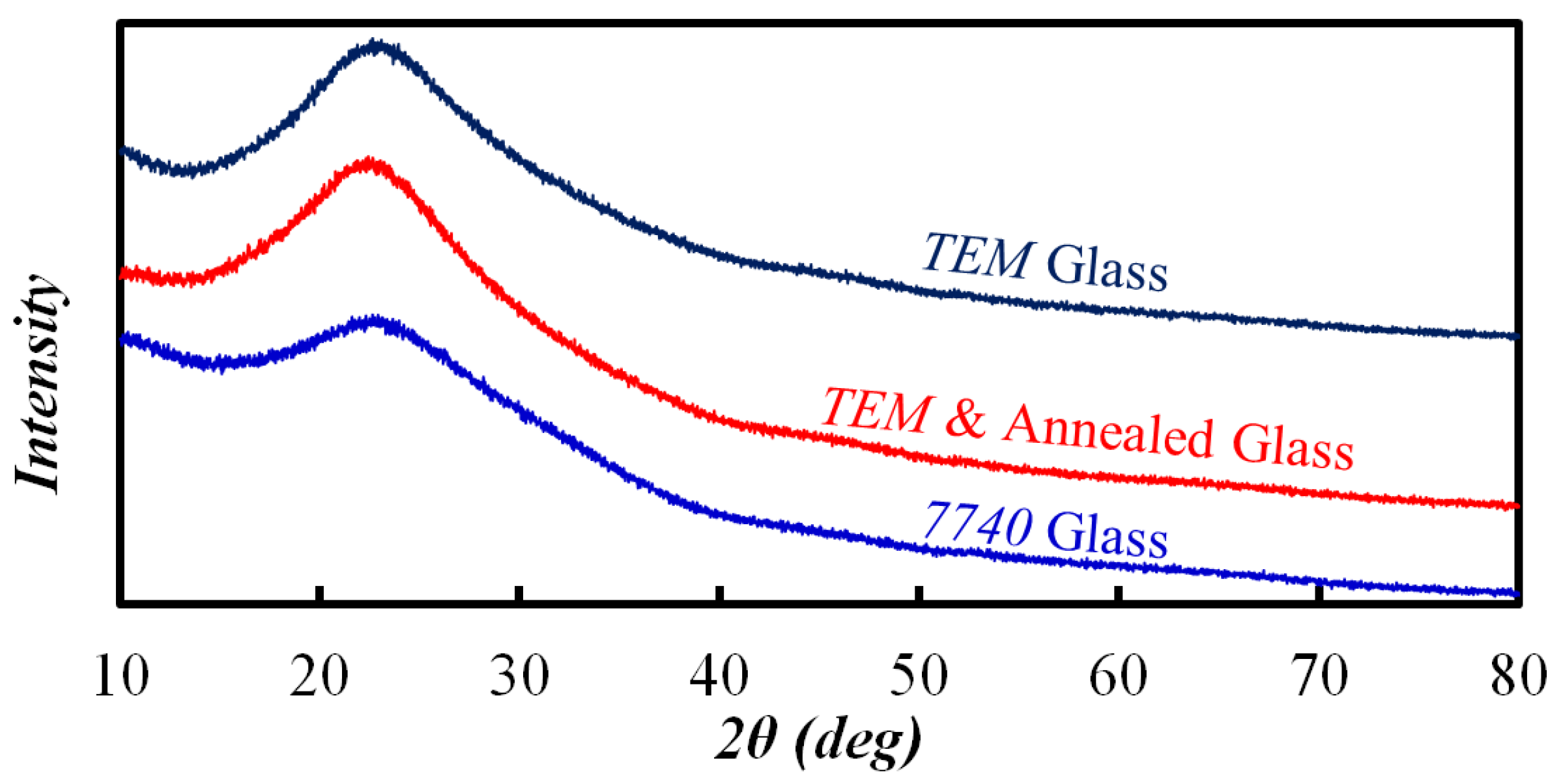
3.3. TEM Etching Mechanism Discussion
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- De Silva, C.W.; Khoshnoud, F.; Li, M.; Halgamuge, S.K. Mechatronics: Fundamentals and Applications; CRC Press: Boca Raton, FL, USA, 2015; pp. 221–252. [Google Scholar]
- Jiang, L.; Whitten, W.B.; Pau, S. A planar ion trapping microdevice with integrated waveguides for optical detection. Opt. Express 2011, 19, 3037–3043. [Google Scholar] [CrossRef] [PubMed]
- Sakhuja, M.; Son, J.; Yang, H.; Bhatia, C.S.; Danner, A.J. Outdoor performance and durability testing of antireflecting and self-cleaning glass for photovoltaic applications. Sol. Energy 2014, 110, 231–238. [Google Scholar] [CrossRef]
- Serrano, D.E.; Zaman, M.F.; Rahafrooz, A.; Hrudey, P.; Lipka, R.; Younkin, D.; Nagpal, S.; Jafri, I.; Ayazi, F. Substrate-decoupled, bulk-acoustic wave gyroscopes: Design and evaluation of next-generation environmentally robust devices. Microsyst. Nanoeng. 2016, 2. [Google Scholar] [CrossRef]
- Mourzina, Y.; Steffen, A.; Offenhäusser, A. The evaporated metal masks for chemical glass etching for biomems. Microsyst. Technol. 2005, 11, 135–140. [Google Scholar] [CrossRef]
- Ryu, H.; Kim, P.K.; Lim, G. Advanced glass etching method exhibiting the controllable etch stop using metal etchant. J. Micromech. Microeng. 2012, 22, 125010. [Google Scholar] [CrossRef]
- Li, X.; Abe, T.; Liu, Y.; Esashi, M. Fabrication of high-density electrical feed-throughs by deep-reactive-ion etching of pyrex glass. J. Microelectromech. Syst. 2002, 11, 625–630. [Google Scholar] [CrossRef]
- Laouamri, H.; Giljean, S.; Arnold, G.; Kolli, M.; Bouaouadja, N.; Tuilier, M.-H. Roughness influence on the optical properties and scratch behavior of acrylic coating deposited on sandblasted glass. Prog. Org. Coat. 2016, 101, 400–406. [Google Scholar] [CrossRef]
- Chung, C.; Lin, S.; Wang, H.; Tan, T.; Tu, K.; Lung, H. Fabrication and simulation of glass micromachining using CO2 laser processing with pdms protection. Appl. Phys. A 2013, 113, 501–507. [Google Scholar] [CrossRef]
- Suh, J.; Cytrynowicz, D.; Gerner, F.M.; Henderson, H.T. A mems bubble pump for an electronic cooling device. J. Micromech. Microeng. 2010, 20, 125025. [Google Scholar] [CrossRef]
- Takagi, H.; Miyazawa, S.-I.; Takahashi, M.; Maeda, R. Electrostatic imprint process for glass. Appl. Phys. Express 2008, 1, 024003. [Google Scholar] [CrossRef]
- Iliescu, C.; Jing, J.; Tay, F.E.; Miao, J.; Sun, T. Characterization of masking layers for deep wet etching of glass in an improved HF/HCl solution. Surf. Coat. Technol. 2005, 198, 314–318. [Google Scholar] [CrossRef]
- Sarapulova, O.; Kyrychok, T.; Sherstiuk, V.; Orlov, A. Modern Printing Technologies in Micro-and Nanoelectronics. In Proceedings of the 2013 IEEE XXXIII International Scientific Conference Electronics and Nanotechnology (ELNANO), Kiev, Ukraine, 16–19 April 2013; IEEE Computer Society: Washington, DC, USA, 2013; pp. 151–155. [Google Scholar]
- Chou, S.Y.; Krauss, P.R.; Zhang, W.; Guo, L.; Zhuang, L. Sub-10 nm imprint lithography and applications. J. Vac. Sci. Technol. B 1997, 15, 2897–2904. [Google Scholar] [CrossRef]
- Razi-ul, M.H.; Wise, K.D. A glass-in-silicon reflow process for three-dimensional microsystems. J. Microelectromech. Syst. 2013, 22, 1470–1477. [Google Scholar]
- Mekaru, H.; Okuyama, C.; Tsuchida, T.; Yasui, M.; Kitadani, T.; Yamashita, M.; Uegaki, J.-I.; Takahashi, M. Development of Ni-P-Plated Inconel Alloy Mold for Imprinting on Pyrex Glass. Jpn. J. Appl. Phys. 2009, 48, 06FH06. [Google Scholar] [CrossRef]
- Saha, B.; Toh, W.Q.; Liu, E.; Tor, S.B.; Hardt, D.E.; Lee, J. A review on the importance of surface coating of micro/nano-mold in micro/nano-molding processes. J. Micromech. Microeng. 2015, 26, 013002. [Google Scholar] [CrossRef]
- Knowles, K.; van Helvoort, A. Anodic bonding. Int. Mater. Rev. 2006, 51, 273–311. [Google Scholar] [CrossRef]
- Anthony, T.R. Anodic bonding of imperfect surfaces. J. Appl. Phys. 1983, 54, 2419–2428. [Google Scholar] [CrossRef]
- Schift, H.; Heyderman, L.; der Maur, M.A.; Gobrecht, J. Pattern formation in hot embossing of thin polymer films. Nanotechnology 2001, 12, 173. [Google Scholar] [CrossRef]
- Lani, S.; Bosseboeuf, A.; Belier, B.; Clerc, C.; Gousset, C.; Aubert, J. Gold metallizations for eutectic bonding of silicon wafers. Microsyst. Technol. 2006, 12, 1021–1025. [Google Scholar] [CrossRef]
- Vijaykumar, T.; Kulkarni, G. Local anodic oxidation patterning of Au deposited Si surfaces. J. Nanosci. Nanotechnol. 2009, 9, 5351–5354. [Google Scholar] [CrossRef] [PubMed]
- Tilli, M.; Motooka, T.; Airaksinen, V.-M.; Franssila, S.; Paulasto-Krockel, M.; Lindroos, V. Handbook of Silicon Based Mems Materials and Technologies; William Andrew: San Diego, CA, USA, 2015; pp. 125–129. [Google Scholar]
- Williams, K.R.; Gupta, K.; Wasilik, M. Etch rates for micromachining processing-Part II. J. Microelectromech. Syst. 2003, 12, 761–778. [Google Scholar] [CrossRef]
- Ciccotti, M. Stress-corrosion mechanisms in silicate glasses. J. Phys. D Appl. Phys. 2009, 42, 214006. [Google Scholar] [CrossRef]
- Tang, J.; Cai, C.; Ming, X.; Yu, X.; Zhao, S.; Tu, S.-T.; Liu, H. Morphology and stress at silicon-glass interface in anodic bonding. Appl. Surf. Sci. 2016, 387, 139–148. [Google Scholar] [CrossRef]
- Sadaba, I.; Fox, C.H.; McWilliam, S. An investigation of residual stress effects due to the anodic bonding of glass and silicon in mems fabrication. In Applied Mechanics and Materials; Patrick, S.K., Ed.; Trans Tech Publications: Aedermannsdorf, Switzerland, 2006. [Google Scholar]
- Brunkov, P.; Melekhin, V.; Goncharov, V.; Lipovskii, A.; Petrov, M. Submicron-resolved relief formation in poled glasses and glass-metal nanocomposites. Tech. Phys. Lett. 2008, 34, 1030–1033. [Google Scholar] [CrossRef]
- Cozma, A.; Puers, B. Characterization of the electrostatic bonding of silicon and pyrex glass. J. Micromech. Microeng. 1995, 5, 98. [Google Scholar] [CrossRef]
- Inzinga, R.; Lin, T.-W.; Yadav, M.; Johnson, H.; Horn, G. Characterization and control of residual stress and curvature in anodically bonded devices and substrates with etched features. Exp. Mech. 2012, 52, 637–648. [Google Scholar] [CrossRef]






| Method | Mask | Patterning | Etching | Pros. | Cons. |
|---|---|---|---|---|---|
| Wet etching | Photoresist/Metal | Photolitho-graphy | Chemical etching | Easy for etching; High surface quality | Pinhole and crack |
| Dry etching | Photoresist/Metal | Photolitho-graphy | Reactive ion etching | Vertical sidewall | Low selectivity; Surface damage |
| Sandblast | Photoresist | Photolitho-graphy | Sandblast | Deep etching | Surface damage |
| Laser | / | Direct-writing | Laser | Deep etching | Time-consuming; Surface damage |
| Drilling | / | Direct-writing | Drilling | Deep etching | Surface damage |
| Imprint | Mould | Replicating | Hot emboss | Fast for patterning; High-accuracy | Hard to demould; Mould damage |
| Sample | Original | 7740 Etching | TEM Etching |
|---|---|---|---|
| RMS | 1.41 nm | 1.10 nm | 1.30 nm |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhan, Z.; Li, W.; Yu, L.; Wang, L.; Sun, D. Study of Thermal Electrical Modified Etching for Glass and Its Application in Structure Etching. Materials 2017, 10, 158. https://doi.org/10.3390/ma10020158
Zhan Z, Li W, Yu L, Wang L, Sun D. Study of Thermal Electrical Modified Etching for Glass and Its Application in Structure Etching. Materials. 2017; 10(2):158. https://doi.org/10.3390/ma10020158
Chicago/Turabian StyleZhan, Zhan, Wei Li, Lingke Yu, Lingyun Wang, and Daoheng Sun. 2017. "Study of Thermal Electrical Modified Etching for Glass and Its Application in Structure Etching" Materials 10, no. 2: 158. https://doi.org/10.3390/ma10020158
APA StyleZhan, Z., Li, W., Yu, L., Wang, L., & Sun, D. (2017). Study of Thermal Electrical Modified Etching for Glass and Its Application in Structure Etching. Materials, 10(2), 158. https://doi.org/10.3390/ma10020158




