Doped Calcium Silicate Ceramics: A New Class of Candidates for Synthetic Bone Substitutes
Abstract
:1. Introduction
- Ability to maintain in vivo mechanical stability at the defect site and withstand physiological loads.
- Radiopacity for easy implant monitoring using non-invasive methods such as X-ray and micro-computed tomography (µ-CT).
- Bioactivity to promote implant integration with host bone, as well as induce bone formation inside and surrounding the implant.
- Ability to be manufactured into macroporous scaffolds with high porosity and interconnectivity to promote bone ingrowth and vascularisation.
- Ability to degrade at a controlled rate that matches the rate of new bone formation.
- Ability to allow easy handling and sterilisation.
- Constitutes a crystalline material (hence excluding silicate-based bioactive glasses and glass-ceramics).
- Constitutes a monophasic material with a single identifiable crystalline phase.
- Contains a dopant which is (1) an element incorporated for ionic substitution of calcium; (2) a metal oxide incorporated into the xCaO–ySiO2 structure; or (3) a combination of both strategies.
- Has been tested for biocompatibility or bioactivity through at least one in vitro or in vivo experiment.
2. Synthesis of DCSCs
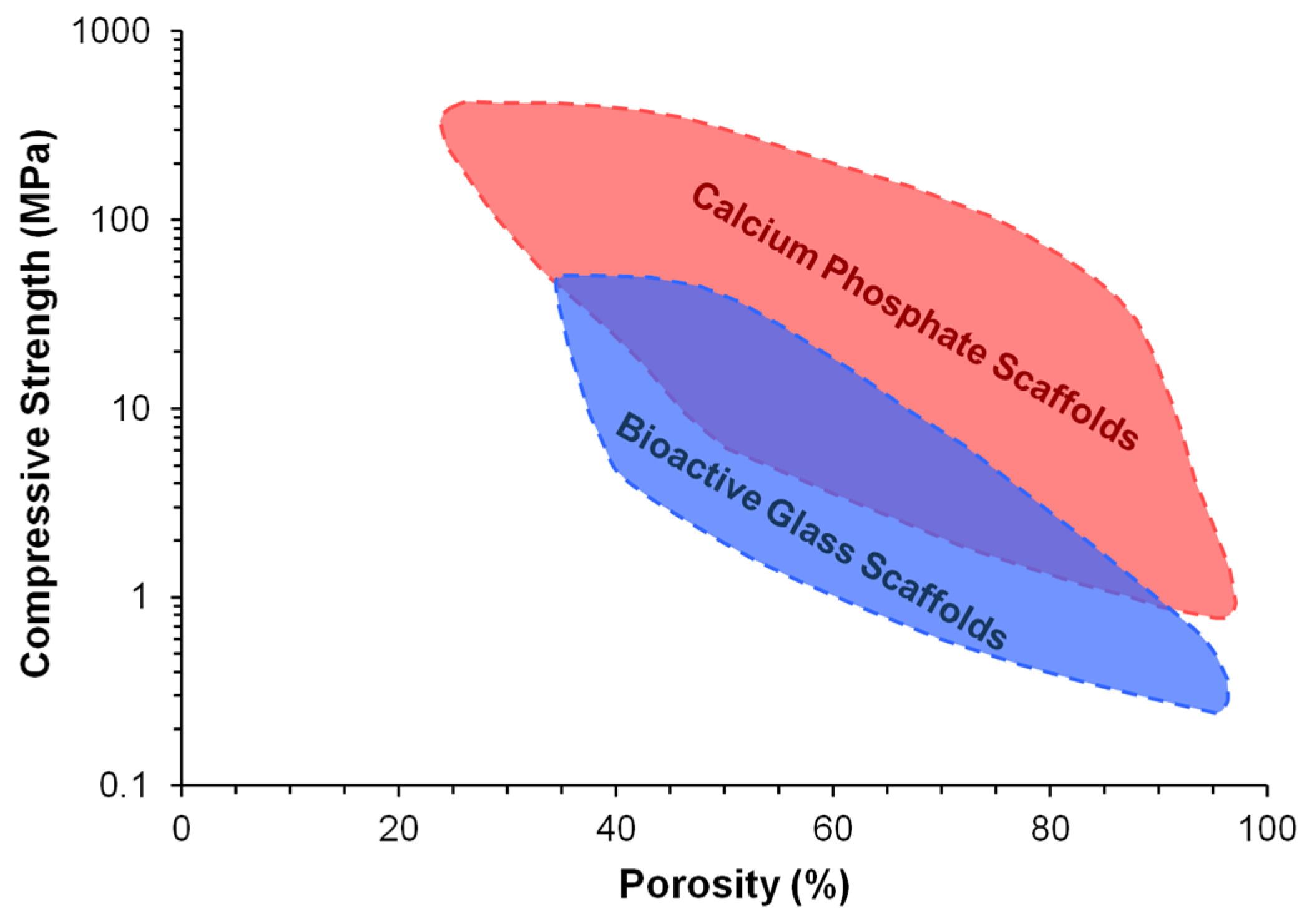
3. Mechanical Properties of Solid and Porous DCSCs
Influence of Fabrication Method on the Mechanical Properties of DCSC Scaffolds
4. Degradation and Ion Release Characteristics of DCSCs
5. Radiopacity of DCSCs
6. In Vitro Cell Interactions with DCSCs
7. In Vivo Performance of DCSCs
8. Development of DCSCs for Broader Clinical Applications
8.1. DCSC-Inorganic Composites
8.2. Polymer-DCSC Composites
8.3. Coating of DCSC Scaffolds
8.4. DCSC-Coated Metal Implants
9. Conclusions and Future Perspectives
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Giannoudis, P.V.; Dinopoulos, H.; Tsiridis, E. Bone substitutes: An update. Injury 2005, 36, S20–S27. [Google Scholar] [CrossRef]
- Fernyhough, J.C.; Schimandle, J.J.; Weigel, M.C.; Edwards, C.C.; Levine, A.M. Chronic donor site pain complicating bone graft harvesting from the posterior iliac crest for spinal fusion. Spine 1992, 17, 1474–1480. [Google Scholar] [CrossRef] [PubMed]
- Seiler, J.; Johnson, J. Iliac crest autogenous bone grafting: Donor site complications. J. South Orthop. Assoc. 1999, 9, 91–97. [Google Scholar]
- Zimmermann, G.; Moghaddam, A. Allograft bone matrix versus synthetic bone graft substitutes. Injury 2011, 42, S16–S21. [Google Scholar] [CrossRef] [PubMed]
- Greenwald, A.S.; Boden, S.D.; Goldberg, V.M.; Khan, Y.; Laurencin, C.T.; Rosier, R.N. Bone-graft substitutes: Facts, fictions, and applications. J. Bone Joint Surg. Am. 2001, 83, S98–S103. [Google Scholar] [CrossRef]
- Nguyen, H.; Morgan, D.A.; Forwood, M.R. Sterilization of allograft bone: Effects of gamma irradiation on allograft biology and biomechanics. Cell Tissue Bank. 2007, 8, 93–105. [Google Scholar] [CrossRef] [PubMed]
- Salgado, A.J.; Coutinho, O.P.; Reis, R.L. Bone tissue engineering: State of the art and future trends. Macromol. Biosci. 2004, 4, 743–765. [Google Scholar] [CrossRef] [PubMed]
- Burg, K.J.; Porter, S.; Kellam, J.F. Biomaterial developments for bone tissue engineering. Biomaterials 2000, 21, 2347–2359. [Google Scholar] [CrossRef]
- Dorozhkin, S.V.; Epple, M. Biological and medical significance of calcium phosphates. Angew. Chem. Int. Ed. 2002, 41, 3130–3146. [Google Scholar] [CrossRef]
- Hench, L.L.; Polak, J.M. Third-generation biomedical materials. Science 2002, 295, 1014–1017. [Google Scholar] [CrossRef] [PubMed]
- LeGeros, R.Z. Properties of osteoconductive biomaterials: Calcium phosphates. Clin. Orthop. Relat. Res. 2002, 395, 81–98. [Google Scholar] [CrossRef]
- Samavedi, S.; Whittington, A.R.; Goldstein, A.S. Calcium phosphate ceramics in bone tissue engineering: A review of properties and their influence on cell behavior. Acta Biomater. 2013, 9, 8037–8045. [Google Scholar] [CrossRef] [PubMed]
- Wagoner Johnson, A.J.; Herschler, B.A. A review of the mechanical behavior of CaP and CaP/polymer composites for applications in bone replacement and repair. Acta Biomater. 2011, 7, 16–30. [Google Scholar] [CrossRef] [PubMed]
- Yoshikawa, H.; Myoui, A. Bone tissue engineering with porous hydroxyapatite ceramics. J. Artif. Organs 2005, 8, 131–136. [Google Scholar] [CrossRef] [PubMed]
- Albrektsson, T.; Johansson, C. Osteoinduction, osteoconduction and osseointegration. Eur. Spine J. 2001, 10, S96–S101. [Google Scholar] [PubMed]
- Li, J.J.; Kaplan, D.L.; Zreiqat, H. Scaffold-based regeneration of skeletal tissues to meet clinical challenges. J. Mater. Chem. B 2014, 2, 7272–7306. [Google Scholar] [CrossRef]
- Fu, Q.; Saiz, E.; Rahaman, M.N.; Tomsia, A.P. Bioactive glass scaffolds for bone tissue engineering: State of the art and future perspectives. Mater. Sci. Eng. C Mater. Biol. Appl. 2011, 31, 1245–1256. [Google Scholar] [CrossRef] [PubMed]
- LeGeros, R.Z. Calcium phosphate-based osteoinductive materials. Chem. Rev. 2008, 108, 4742–4753. [Google Scholar] [CrossRef] [PubMed]
- Jones, J.R. Review of bioactive glass: From Hench to hybrids. Acta Biomater. 2013, 9, 4457–4486. [Google Scholar] [CrossRef] [PubMed]
- De Aza, P.; Guitian, F.; De Aza, S. Bioactivity of wollastonite ceramics: In vitro evaluation. Scr. Metall. Mater. 1994, 31, 1001–1005. [Google Scholar] [CrossRef]
- De Aza, P.; Luklinska, Z.; Martinez, A.; Anseau, M.; Guitian, F.; De Aza, S. Morphological and structural study of pseudowollastonite implants in bone. Microscopy 2000, 197, 60–67. [Google Scholar] [CrossRef]
- Ni, S.; Chang, J.; Chou, L. A novel bioactive porous CaSiO3 scaffold for bone tissue engineering. J. Biomed. Mater. Res. A 2006, 76, 196–205. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.; Lin, K.; Wang, Z.; Chang, J.; Wang, L.; Lu, J.; Ning, C. Reconstruction of calvarial defect of rabbits using porous calcium silicate bioactive ceramics. Biomaterials 2008, 29, 2588–2596. [Google Scholar] [CrossRef] [PubMed]
- Iimori, Y.; Kameshima, Y.; Okada, K.; Hayashi, S. Comparative study of apatite formation on CaSiO3 ceramics in simulated body fluids with different carbonate concentrations. J. Mater. Sci.: Mater. Med. 2005, 16, 73–79. [Google Scholar] [CrossRef] [PubMed]
- Zreiqat, H.; Ramaswamy, Y.; Wu, C.; Paschalidis, A.; Lu, Z.; James, B.; Birke, O.; McDonald, M.; Little, D.; Dunstan, C.R. The incorporation of strontium and zinc into a calcium–silicon ceramic for bone tissue engineering. Biomaterials 2010, 31, 3175–3184. [Google Scholar] [CrossRef] [PubMed]
- Ni, S.; Chang, J. In vitro degradation, bioactivity, and cytocompatibility of calcium silicate, dimagnesium silicate, and tricalcium phosphate bioceramics. J. Biomater. Appl. 2009, 24, 139–158. [Google Scholar] [CrossRef] [PubMed]
- Lin, K.; Zhai, W.; Ni, S.; Chang, J.; Zeng, Y.; Qian, W. Study of the mechanical property and in vitro biocompatibility of CaSiO3 ceramics. Ceram. Int. 2005, 31, 323–326. [Google Scholar] [CrossRef]
- Wang, G.; Lu, Z.; Dwarte, D.; Zreiqat, H. Porous scaffolds with tailored reactivity modulate in vitro osteoblast responses. Mater. Sci. Eng. C Mater. Biol. Appl. 2012, 32, 1818–1826. [Google Scholar] [CrossRef]
- Wu, C.; Ramaswamy, Y.; Boughton, P.; Zreiqat, H. Improvement of mechanical and biological properties of porous CaSiO3 scaffolds by poly(d,l-lactic acid) modification. Acta Biomater. 2008, 4, 343–353. [Google Scholar] [CrossRef] [PubMed]
- Lin, K.; Xia, L.; Li, H.; Jiang, X.; Pan, H.; Xu, Y.; Lu, W.W.; Zhang, Z.; Chang, J. Enhanced osteoporotic bone regeneration by strontium-substituted calcium silicate bioactive ceramics. Biomaterials 2013, 34, 10028–10042. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Ramaswamy, Y.; Kwik, D.; Zreiqat, H. The effect of strontium incorporation into CaSiO3 ceramics on their physical and biological properties. Biomaterials 2007, 28, 3171–3181. [Google Scholar] [CrossRef] [PubMed]
- Kong, N.; Lin, K.; Li, H.; Chang, J. Synergy effects of copper and silicon ions on stimulation of vascularization by copper-doped calcium silicate. J. Mater. Chem. B 2014, 2, 1100–1110. [Google Scholar] [CrossRef]
- Ramaswamy, Y.; Wu, C.; Van Hummel, A.; Combes, V.; Grau, G.; Zreiqat, H. The responses of osteoblasts, osteoclasts and endothelial cells to zirconium modified calcium-silicate-based ceramic. Biomaterials 2008, 29, 4392–4402. [Google Scholar] [CrossRef] [PubMed]
- Roohani-Esfahani, S.-I.; No, Y.J.; Lu, Z.; Ng, P.Y.; Chen, Y.; Shi, J.; Pavlos, N.J.; Zreiqat, H. A bioceramic with enhanced osteogenic properties to regulate the function of osteoblastic and osteocalastic cells for bone tissue regeneration. Biomed. Mater. 2016, 11, 035018. [Google Scholar] [CrossRef] [PubMed]
- Tian, T.; Wu, C.; Chang, J. Preparation and in vitro osteogenic, angiogenic and antibacterial properties of cuprorivaite (CaCuSi4O10, Cup) bioceramics. RSC Adv. 2016, 6, 45840–45849. [Google Scholar] [CrossRef]
- Wu, C.; Chang, J. Degradation, bioactivity, and cytocompatibility of diopside, akermanite, and bredigite ceramics. J. Biomed. Mater. Res. B 2007, 83B, 153–160. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Chang, J.; Ni, S.; Wang, J. In vitro bioactivity of akermanite ceramics. J. Biomed. Mater. Res. A 2006, 76, 73–80. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Chang, J.; Wang, J.; Ni, S.; Zhai, W. Preparation and characteristics of a calcium magnesium silicate (bredigite) bioactive ceramic. Biomaterials 2005, 26, 2925–2931. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Chang, J.; Zhai, W. A novel hardystonite bioceramic: Preparation and characteristics. Ceram. Int. 2005, 31, 27–31. [Google Scholar] [CrossRef]
- Wu, C.; Ramaswamy, Y.; Soeparto, A.; Zreiqat, H. Incorporation of titanium into calcium silicate improved their chemical stability and biological properties. J. Biomed. Mater. Res. A 2008, 86, 402–410. [Google Scholar] [CrossRef] [PubMed]
- Schumacher, T.C.; Aminian, A.; Volkmann, E.; Lührs, H.; Zimnik, D.; Pede, D.; Wosniok, W.; Treccani, L.; Rezwan, K. Synthesis and mechanical evaluation of Sr-doped calcium-zirconium-silicate (baghdadite) and its impact on osteoblast cell proliferation and ALP activity. Biomed. Mater. 2015, 10, 055013. [Google Scholar] [CrossRef] [PubMed]
- Tian, T.; Han, Y.; Ma, B.; Wu, C.; Chang, J. Novel Co-akermanite (Ca2CoSi2O7) bioceramics with the activity to stimulate osteogenesis and angiogenesis. J. Mater. Chem. B 2015, 3, 6773–6782. [Google Scholar] [CrossRef]
- Staiger, M.P.; Pietak, A.M.; Huadmai, J.; Dias, G. Magnesium and its alloys as orthopedic biomaterials: A review. Biomaterials 2006, 27, 1728–1734. [Google Scholar] [CrossRef] [PubMed]
- Pemmer, B.; Roschger, A.; Wastl, A.; Hofstaetter, J.; Wobrauschek, P.; Simon, R.; Thaler, H.; Roschger, P.; Klaushofer, K.; Streli, C. Spatial distribution of the trace elements zinc, strontium and lead in human bone tissue. Bone 2013, 57, 184–193. [Google Scholar] [CrossRef] [PubMed]
- Hoppe, A.; Güldal, N.S.; Boccaccini, A.R. A review of the biological response to ionic dissolution products from bioactive glasses and glass-ceramics. Biomaterials 2011, 32, 2757–2774. [Google Scholar] [CrossRef] [PubMed]
- Geetha, M.; Singh, A.; Asokamani, R.; Gogia, A. Ti based biomaterials, the ultimate choice for orthopaedic implants—A review. Prog. Mater. Sci 2009, 54, 397–425. [Google Scholar] [CrossRef]
- Al-Amleh, B.; Lyons, K.; Swain, M. Clinical trials in zirconia: A systematic review. J. Oral Rehabil. 2010, 37, 641–652. [Google Scholar] [CrossRef] [PubMed]
- Sedel, L. Evolution of alumina-on-alumina implants: A review. Clin. Orthop. Relat. Res. 2000, 379, 48–54. [Google Scholar] [CrossRef]
- Harle, J.; Kim, H.-W.; Mordan, N.; Knowles, J.C.; Salih, V. Initial responses of human osteoblasts to sol–gel modified titanium with hydroxyapatite and titania composition. Acta Biomater. 2006, 2, 547–556. [Google Scholar] [CrossRef] [PubMed]
- Vrouwenvelder, W.; Groot, C.; De Groot, K. Better histology and biochemistry for osteoblasts cultured on titanium-doped bioactive glass: Bioglass 45S5 compared with iron-, titanium-, fluorine-and boron-containing bioactive glasses. Biomaterials 1994, 15, 97–106. [Google Scholar] [CrossRef]
- Schumacher, T.C.; Volkmann, E.; Yilmaz, R.; Wolf, A.; Treccani, L.; Rezwan, K. Mechanical evaluation of calcium-zirconium-silicate (baghdadite) obtained by a direct solid-state synthesis route. J. Mech. Behav. Biomed. Mater. 2014, 34, 294–301. [Google Scholar] [CrossRef] [PubMed]
- Nonami, T.; Tsutsumi, S. Study of diopside ceramics for biomaterials. J. Mater. Sci.: Mater. Med. 1999, 10, 475–479. [Google Scholar]
- Bandyopadhyay, A.; Bernard, S.; Xue, W.; Bose, S. Calcium phosphate-based resorbable ceramics: Influence of MgO, ZnO, and SiO2 dopants. J. Am. Ceram. Soc. 2006, 89, 2675–2688. [Google Scholar] [CrossRef]
- Fielding, G.A.; Bandyopadhyay, A.; Bose, S. Effects of silica and zinc oxide doping on mechanical and biological properties of 3D printed tricalcium phosphate tissue engineering scaffolds. Dent. Mater. 2012, 28, 113–122. [Google Scholar] [CrossRef] [PubMed]
- Seeley, Z.; Bandyopadhyay, A.; Bose, S. Influence of TiO2 and Ag2O addition on tricalcium phosphate ceramics. J. Biomed. Mater. Res. A 2007, 82, 113–121. [Google Scholar] [CrossRef] [PubMed]
- Xue, W.; Dahlquist, K.; Banerjee, A.; Bandyopadhyay, A.; Bose, S. Synthesis and characterization of tricalcium phosphate with Zn and Mg based dopants. J. Mater. Sci.: Mater. Med. 2008, 19, 2669–2677. [Google Scholar] [CrossRef] [PubMed]
- Cho, J.H.; Park, I.K.; Kim, H.G.; Chung, H.T. Sintering behavior of cadmium-doped Pb (Ni1/3Nb2/3) O3–PbZrO3–PbTiO3 ceramics. J. Am. Ceram. Soc. 1997, 80, 1523–1534. [Google Scholar] [CrossRef]
- Naga, S.; Hassan, A.; Awaad, M.; Bondioli, F. Influence of Ta2O5 doping on the microstructure, physical and mechanical properties of a-alumina ceramics. J. Ceram. Sci. Technol. 2013, 4, 187–192. [Google Scholar]
- Shirazi, S.F.S.; Gharehkhani, S.; Metselaar, H.S.C.; Nasiri-Tabrizi, B.; Yarmand, H.; Ahmadi, M.; Osman, N.A.A. Ion size, loading, and charge determine the mechanical properties, surface apatite, and cell growth of silver and tantalum doped calcium silicate. RSC Adv. 2016, 6, 190–200. [Google Scholar] [CrossRef]
- Hutmacher, D.W. Scaffolds in tissue engineering bone and cartilage. Biomaterials 2000, 21, 2529–2543. [Google Scholar] [CrossRef]
- Rezwan, K.; Chen, Q.Z.; Blaker, J.J.; Boccaccini, A.R. Biodegradable and bioactive porous polymer/inorganic composite scaffolds for bone tissue engineering. Biomaterials 2006, 27, 3413–3431. [Google Scholar] [CrossRef] [PubMed]
- Sadeghpour, S.; Amirjani, A.; Hafezi, M.; Zamanian, A. Fabrication of a novel nanostructured calcium zirconium silicate scaffolds prepared by a freeze-casting method for bone tissue engineering. Ceram. Int. 2014, 40, 16107–16114. [Google Scholar] [CrossRef]
- Han, Z.; Feng, P.; Gao, C.; Shen, Y.; Shuai, C.; Peng, S. Microstructure, mechanical properties and in vitro bioactivity of akermanite scaffolds fabricated by laser sintering. Biomed. Mater. Eng. 2014, 24, 2073–2080. [Google Scholar] [PubMed]
- Liu, A.; Sun, M.; Yang, X.; Ma, C.; Liu, Y.; Yang, X.; Yan, S.; Gou, Z. Three-dimensional printing akermanite porous scaffolds for load-bearing bone defect repair: An investigation of osteogenic capability and mechanical evolution. J. Biomater. Appl. 2016, 31, 650–660. [Google Scholar] [CrossRef] [PubMed]
- Zocca, A.; Franchin, G.; Elsayed, H.; Gioffredi, E.; Bernardo, E.; Colombo, P. Direct ink writing of a preceramic polymer and fillers to produce hardystonite (Ca2ZnSi2O7) bioceramic scaffolds. J. Am. Ceram. Soc. 2016, 99, 1960–1967. [Google Scholar] [CrossRef]
- Roohani-Esfahani, S.-I.; Newman, P.; Zreiqat, H. Design and fabrication of 3D printed scaffolds with a mechanical strength comparable to cortical bone to repair large bone defects. Sci. Rep. 2016, 6, 19468. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Luo, Y.; Cuniberti, G.; Xiao, Y.; Gelinsky, M. Three-dimensional printing of hierarchical and tough mesoporous bioactive glass scaffolds with a controllable pore architecture, excellent mechanical strength and mineralization ability. Acta Biomater. 2011, 7, 2644–2650. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Chang, J. A novel akermanite bioceramic: Preparation and characteristics. J. Biomater. Appl. 2006, 21, 119–129. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Chang, J.; Zhai, W.; Ni, S.; Wang, J. Porous akermanite scaffolds for bone tissue engineering: Preparation, characterization, and in vitro studies. J. Biomed. Mater. Res. B 2006, 78, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Ramaswamy, Y.; Zreiqat, H. Porous diopside (CaMgSi2O6) scaffold: A promising bioactive material for bone tissue engineering. Acta Biomater. 2010, 6, 2237–2245. [Google Scholar] [CrossRef] [PubMed]
- Roohani-Esfahani, S.; Dunstan, C.; Davies, B.; Pearce, S.; Williams, R.; Zreiqat, H. Repairing a critical-sized bone defect with highly porous modified and unmodified baghdadite scaffolds. Acta Biomater. 2012, 8, 4162–4172. [Google Scholar] [CrossRef] [PubMed]
- Hench, L.L. Bioceramics. J. Am. Ceram. Soc. 1998, 81, 1705–1728. [Google Scholar] [CrossRef]
- Chen, Q.Z.; Thompson, I.D.; Boccaccini, A.R. 45S5 Bioglass®-derived glass–ceramic scaffolds for bone tissue engineering. Biomaterials 2006, 27, 2414–2425. [Google Scholar] [CrossRef] [PubMed]
- Akao, M.; Miura, N.; Aoki, H. Fracture toughness of sintered hydroxyapatite and β-tricalcium phosphate. Yogyo Kyokaishi 1984, 92, 672–674. [Google Scholar] [CrossRef]
- Lu, J.; Descamps, M.; Dejou, J.; Koubi, G.; Hardouin, P.; Lemaitre, J.; Proust, J.P. The biodegradation mechanism of calcium phosphate biomaterials in bone. J. Biomed. Mater. Res. 2002, 63, 408–412. [Google Scholar] [CrossRef] [PubMed]
- Wenisch, S.; Stahl, J.P.; Horas, U.; Heiss, C.; Kilian, O.; Trinkaus, K.; Hild, A.; Schnettler, R. In vivo mechanisms of hydroxyapatite ceramic degradation by osteoclasts: Fine structural microscopy. J. Biomed. Mater. Res. A 2003, 67, 713–718. [Google Scholar] [CrossRef] [PubMed]
- Casey, W.H.; Ludwig, C. The mechanism of dissolution of oxide minerals. Nature 1996, 381, 506–509. [Google Scholar] [CrossRef]
- Crundwell, F. The mechanism of dissolution of minerals in acidic and alkaline solutions: Part II Application of a new theory to silicates, aluminosilicates and quartz. Hydrometallurgy 2014, 149, 265–275. [Google Scholar] [CrossRef]
- Kokubo, T.; Takadama, H. How useful is SBF in predicting in vivo bone bioactivity? Biomaterials 2006, 27, 2907–2915. [Google Scholar] [CrossRef] [PubMed]
- Bohner, M.; Lemaitre, J. Can bioactivity be tested in vitro with SBF solution? Biomaterials 2009, 30, 2175–2179. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Ramaswamy, Y.; Chang, J.; Woods, J.; Chen, Y.; Zreiqat, H. The effect of Zn contents on phase composition, chemical stability and cellular bioactivity in Zn–Ca–Si system ceramics. J. Biomed. Mater. Res. B 2008, 87, 346–353. [Google Scholar] [CrossRef] [PubMed]
- Botelho, C.; Lopes, M.; Gibson, I.; Best, S.; Santos, J. Structural analysis of Si-substituted hydroxyapatite: Zeta potential and X-ray photoelectron spectroscopy. J. Mater. Sci.: Mater. Med. 2002, 13, 1123–1127. [Google Scholar]
- Oliveira, J.; Correia, R.; Fernandes, M. Effects of Si speciation on the in vitro bioactivity of glasses. Biomaterials 2002, 23, 371–379. [Google Scholar] [CrossRef]
- Ni, S.; Chang, J.; Chou, L.; Zhai, W. Comparison of osteoblast-like cell responses to calcium silicate and tricalcium phosphate ceramics in vitro. J. Biomed. Mater. Res. B 2007, 80, 174–183. [Google Scholar] [CrossRef] [PubMed]
- Xia, L.; Zhang, Z.; Chen, L.; Zhang, W.; Zeng, D.; Zhang, X.; Chang, J.; Jiang, X. Proliferation and osteogenic differentiation of human periodontal ligament cells on akermanite and β-TCP bioceramics. Eur. Cells Mater. 2011, 22, e82. [Google Scholar]
- No, Y.J.; Roohani-Esfahani, S.-I.; Lu, Z.; Schaer, T.; Zreiqat, H. Injectable radiopaque and bioactive polycaprolactone-ceramic composites for orthopedic augmentation. J. Biomed. Mater. Res. B 2015, 103, 1465–1477. [Google Scholar] [CrossRef] [PubMed]
- Hubbell, J.H.; Seltzer, S.M. Tables of X-ray Mass Attenuation Coefficients and Mass Energy-Absorption Coefficients 1 keV to 20 MeV for Elements Z= 1 to 92 and 48 Additional Substances of Dosimetric Interest; National Institute of Standards and Technology: Gaithersburg, MD, USA, 1995.
- Ginebra, M.P.; Albuixech, L.; Fernández-Barragán, E.; Aparicio, C.; Gil, F.J.; San Román, J.; Vázquez, B.; Planell, J.A. Mechanical performance of acrylic bone cements containing different radiopacifying agents. Biomaterials 2002, 23, 1873–1882. [Google Scholar] [CrossRef]
- Hoekstra, J.W.M.; van den Beucken, J.J.; Leeuwenburgh, S.C.; Meijer, G.J.; Jansen, J.A. Tantalumpentoxide as a radiopacifier in injectable calcium phosphate cements for bone substitution. Tissue Eng. Part C Methods 2011, 17, 907–913. [Google Scholar] [CrossRef] [PubMed]
- Maniglia-Ferreira, C.; Gurgel-Filho, E.D.; de Araújo Silva, J.B., Jr.; de Paula, R.C.M.; de Andrade Feitosa, J.P.; de Sousa-Filho, F.J. Chemical composition and thermal behavior of five brands of thermoplasticized gutta-percha. Eur. J. Dent. 2013, 7, 201. [Google Scholar] [CrossRef] [PubMed]
- Bose, S.; Fielding, G.; Tarafder, S.; Bandyopadhyay, A. Understanding of dopant-induced osteogenesis and angiogenesis in calcium phosphate ceramics. Trends Biotechnol. 2013, 31, 594–605. [Google Scholar] [CrossRef] [PubMed]
- Hoppe, A.; Mouriño, V.; Boccaccini, A.R. Therapeutic inorganic ions in bioactive glasses to enhance bone formation and beyond. Biomater. Sci. 2013, 1, 254–256. [Google Scholar] [CrossRef]
- Mouriño, V.; Cattalini, J.P.; Boccaccini, A.R. Metallic ions as therapeutic agents in tissue engineering scaffolds: An overview of their biological applications and strategies for new developments. J. R. Soc. Interface 2012, 9, 401–419. [Google Scholar] [CrossRef] [PubMed]
- Gu, H.; Guo, F.; Zhou, X.; Gong, L.; Zhang, Y.; Zhai, W.; Chen, L.; Cen, L.; Yin, S.; Chang, J. The stimulation of osteogenic differentiation of human adipose-derived stem cells by ionic products from akermanite dissolution via activation of the ERK pathway. Biomaterials 2011, 32, 7023–7033. [Google Scholar] [CrossRef] [PubMed]
- Liu, N.; Fan, W.; Wu, C.; Fan, B. The interactions of Mg2+/Zn2+-containing silicate materials with stem cells and bacteria. Mater. Lett. 2013, 112, 105–108. [Google Scholar] [CrossRef]
- Zhou, Y.; Wu, C.; Zhang, X.; Han, P.; Xiao, Y. The ionic products from bredigite bioceramics induced cementogenic differentiation of periodontal ligament cells via activation of the Wnt/β-catenin signalling pathway. J. Mater. Chem. B 2013, 1, 3380–3389. [Google Scholar] [CrossRef]
- Dong, X.; Li, H.; Zhou, Y.; Ou, L.; Cao, J.; Chang, J. The stimulation of osteogenic differentiation of embryoid bodies from human induced pluripotent stem cells by akermanite bioceramics. J. Mater. Chem. B 2016, 4, 2369–2376. [Google Scholar] [CrossRef]
- Huang, Y.; Jin, X.; Zhang, X.; Sun, H.; Tu, J.; Tang, T.; Chang, J.; Dai, K. In vitro and in vivo evaluation of akermanite bioceramics for bone regeneration. Biomaterials 2009, 30, 5041–5048. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Wu, C.; Dai, K.; Chang, J.; Tang, T. Proliferation and osteoblastic differentiation of human bone marrow-derived stromal cells on akermanite-bioactive ceramics. Biomaterials 2006, 27, 5651–5657. [Google Scholar] [CrossRef] [PubMed]
- Xia, L.; Yin, Z.; Mao, L.; Wang, X.; Liu, J.; Jiang, X.; Zhang, Z.; Lin, K.; Chang, J.; Fang, B. Akermanite bioceramics promote osteogenesis, angiogenesis and suppress osteoclastogenesis for osteoporotic bone regeneration. Sci. Rep. 2016, 6, 22005. [Google Scholar] [CrossRef] [PubMed]
- Zhai, W.; Lu, H.; Wu, C.; Chen, L.; Lin, X.; Naoki, K.; Chen, G.; Chang, J. Stimulatory effects of the ionic products from Ca-Mg-Si bioceramics on both osteogenesis and angiogenesis in vitro. Acta Biomater. 2013, 9, 8004–8014. [Google Scholar] [CrossRef] [PubMed]
- Lu, H.; Kawazoe, N.; Tateishi, T.; Chen, G.; Jin, X.; Chang, J. In vitro proliferation and osteogenic differentiation of human bone marrow-derived mesenchymal stem cells cultured with hardystonite (Ca2ZnSi2O7) and β-TCP ceramics. J. Biomater. Appl. 2010, 25, 39–56. [Google Scholar] [PubMed]
- Lu, Z.; Wang, G.; Roohani-Esfahani, I.; Dunstan, C.R.; Zreiqat, H. Baghdadite ceramics modulate the cross talk between human adipose stem cells and osteoblasts for bone regeneration. Tissue Eng. Part A 2013, 20, 992–1002. [Google Scholar] [CrossRef] [PubMed]
- Coulombe, J.; Faure, H.; Robin, B.; Ruat, M. In vitro effects of strontium ranelate on the extracellular calcium-sensing receptor. Biochem. Biophys. Res. Commun. 2004, 323, 1184–1190. [Google Scholar] [CrossRef] [PubMed]
- Marie, P.J. The calcium-sensing receptor in bone cells: A potential therapeutic target in osteoporosis. Bone 2010, 46, 571–576. [Google Scholar] [CrossRef] [PubMed]
- Han, P.; Wu, C.; Xiao, Y. The effect of silicate ions on proliferation, osteogenic differentiation and cell signalling pathways (WNT and SHH) of bone marrow stromal cells. Biomater. Sci. 2013, 1, 379–392. [Google Scholar] [CrossRef]
- Zreiqat, H.; Valenzuela, S.M.; Nissan, B.B.; Roest, R.; Knabe, C.; Radlanski, R.J.; Renz, H.; Evans, P.J. The effect of surface chemistry modification of titanium alloy on signalling pathways in human osteoblasts. Biomaterials 2005, 26, 7579–7586. [Google Scholar] [CrossRef] [PubMed]
- Graney, P.L.; Roohani-Esfahani, S.-I.; Zreiqat, H.; Spiller, K.L. In vitro response of macrophages to ceramic scaffolds used for bone regeneration. J. R. Soc. Interface 2016, 13, 20160346. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Han, P.; Jaiprakash, A.; Wu, C.; Xiao, Y. A stimulatory effect of Ca3ZrSi2O9 bioceramics on cementogenic/osteogenic differentiation of periodontal ligament cells. J. Mater. Chem. B 2014, 2, 1415–1423. [Google Scholar] [CrossRef]
- Li, J.J.; Roohani-Esfahani, S.-I.; Dunstan, C.R.; Quach, T.; Steck, R.; Saifzadeh, S.; Pivonka, P.; Zreiqat, H. Efficacy of novel synthetic bone substitutes in the reconstruction of large segmental bone defects in sheep tibiae. Biomed. Mater. 2016, 11, 015016. [Google Scholar] [CrossRef] [PubMed]
- Luo, T.; Wu, C.; Zhang, Y. The in vivo osteogenesis of Mg or Zr-modified silicate-based bioceramic spheres. J. Biomed. Mater. Res. A 2012, 100, 2269–2277. [Google Scholar] [CrossRef] [PubMed]
- Gheisari, H.; Karamian, E.; Abdellahi, M. A novel hydroxyapatite–hardystonite nanocomposite ceramic. Ceram. Int. 2015, 41, 5967–5975. [Google Scholar] [CrossRef]
- Roohani-Esfahani, S.; Dunstan, C.; Li, J.; Lu, Z.; Davies, B.; Pearce, S.; Field, J.; Williams, R.; Zreiqat, H. Unique microstructural design of ceramic scaffolds for bone regeneration under load. Acta Biomater. 2013, 9, 7014–7024. [Google Scholar] [CrossRef] [PubMed]
- Roohani-Esfahani, S.-I.; Chen, Y.; Shi, J.; Zreiqat, H. Fabrication and characterization of a new, strong and bioactive ceramic scaffold for bone regeneration. Mater. Lett. 2013, 107, 378–381. [Google Scholar] [CrossRef]
- Li, J.J.; Roohani-Esfahani, S.-I.; Kim, K.; Kaplan, D.L.; Zreiqat, H. Silk coating on a bioactive ceramic scaffold for bone regeneration: Effective enhancement of mechanical and in vitro osteogenic properties towards load-bearing applications. J. Tissue Eng. Regen. Med. 2015. [Google Scholar] [CrossRef] [PubMed]
- Shuai, C.; Liu, T.; Gao, C.; Feng, P.; Peng, S. Mechanical reinforcement of diopside bone scaffolds with carbon nanotubes. Int. J. Mol. Sci. 2014, 15, 19319–19329. [Google Scholar] [CrossRef] [PubMed]
- Shuai, C.; Han, Z.; Feng, P.; Gao, C.; Xiao, T.; Peng, S. Akermanite scaffolds reinforced with boron nitride nanosheets in bone tissue engineering. J. Mater. Sci.: Mater. Med. 2015, 26, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Feng, P.; Gao, C.; Shuai, C.; Peng, S. Toughening and strengthening mechanisms of porous akermanite scaffolds reinforced with nano-titania. RSC Adv. 2015, 5, 3498–3507. [Google Scholar] [CrossRef]
- Zhang, M.; Liu, C.; Zhang, X.; Pan, S.; Xu, Y. Al2O3/diopside ceramic composites and their behaviour in simulated body fluid. Ceram. Int. 2010, 36, 2505–2509. [Google Scholar] [CrossRef]
- Zhang, M.; Liu, C.; Sun, J.; Zhang, X. Hydroxyapatite/diopside ceramic composites and their behaviour in simulated body fluid. Ceram. Int. 2011, 37, 2025–2029. [Google Scholar] [CrossRef]
- Zanetti, A.; McCandless, G.; Chan, J.; Gimble, J.; Hayes, D. Characterization of novel akermanite:poly-ϵ-caprolactone scaffolds for human adipose-derived stem cells bone tissue engineering. J. Tissue Eng. Regen. Med. 2015, 9, 389–404. [Google Scholar] [CrossRef] [PubMed]
- Jaiswal, A.K.; Chhabra, H.; Kadam, S.S.; Londhe, K.; Soni, V.P.; Bellare, J.R. Hardystonite improves biocompatibility and strength of electrospun polycaprolactone nanofibers over hydroxyapatite: A comparative study. Mater. Sci. Eng. C Mater. Biol. Appl. 2013, 33, 2926–2936. [Google Scholar] [CrossRef] [PubMed]
- Feng, P.; Guo, X.; Gao, C.; Gao, D.; Xiao, T.; Shuai, X.; Shuai, C.; Peng, S. Diopside modified porous polyglycolide scaffolds with improved properties. RSC Adv. 2015, 5, 54822–54829. [Google Scholar] [CrossRef]
- Liu, G.; Wu, C.; Fan, W.; Miao, X.; Sin, D.C.; Crawford, R.; Xiao, Y. The effects of bioactive akermanite on physiochemical, drug-delivery, and biological properties of poly (lactide-co-glycolide) beads. J. Biomed. Mater. Res. B 2011, 96, 360–368. [Google Scholar] [CrossRef] [PubMed]
- Ghorbanian, L.; Emadi, R.; Razavi, S.M.; Shin, H.; Teimouri, A. Fabrication and characterization of novel diopside/silk fibroin nanocomposite scaffolds for potential application in maxillofacial bone regeneration. Int. J. Biol. Macromol. 2013, 58, 275–280. [Google Scholar] [CrossRef] [PubMed]
- Kumar, J.P.; Lakshmi, L.; Jyothsna, V.; Balaji, D.; Saravanan, S.; Moorthi, A.; Selvamurugan, N. Synthesis and characterization of diopside particles and their suitability along with chitosan matrix for bone tissue engineering in vitro and in vivo. J. Biomed. Nanotechnol. 2014, 10, 970–981. [Google Scholar] [CrossRef] [PubMed]
- Zare-Harofteh, A.; Saber-Samandari, S.; Saber-Samandari, S. The effective role of akermanite on the apatite-forming ability of gelatin scaffold as a bone graft substitute. Ceram. Int. 2016, 42, 17781–17791. [Google Scholar] [CrossRef]
- Patrício, T.; Domingos, M.; Gloria, A.; Bártolo, P. Characterisation of PCL and PCL/PLA scaffolds for tissue engineering. Procedia CIRP 2013, 5, 110–114. [Google Scholar] [CrossRef]
- Sultana, N.; Wang, M. PHBV/PLLA-based composite scaffolds fabricated using an emulsion freezing/freeze-drying technique for bone tissue engineering: Surface modification and in vitro biological evaluation. Biofabrication 2012, 4, 015003. [Google Scholar] [CrossRef] [PubMed]
- Ba Linh, N.T.; Min, Y.K.; Lee, B.-T. Hybrid hydroxyapatite nanoparticles-loaded PCL/GE blend fibers for bone tissue engineering. J. Biomater. Sci. Polym. Ed. 2013, 24, 520–538. [Google Scholar] [CrossRef] [PubMed]
- Giordano, C.; Albani, D.; Gloria, A.; Tunesi, M.; Rodilossi, S.; Russo, T.; Forloni, G.; Ambrosio, L.; Cigada, A. Nanocomposites for neurodegenerative diseases: Hydrogel-nanoparticle combinations for a challenging drug delivery. Int. J. Artif. Organs 2011, 34, 1115–1127. [Google Scholar] [CrossRef] [PubMed]
- No, Y.J.; Roohani-Esfahani, S.-I.; Zreiqat, H. Nanomaterials: The next step in injectable bone cements. Nanomedicine 2014, 9, 1745–1764. [Google Scholar] [CrossRef] [PubMed]
- Gomoll, A.H.; Fitz, W.; Scott, R.D.; Thornhill, T.S.; Bellare, A. Nanoparticulate fillers improve the mechanical strength of bone cement. Acta Orthop. 2008, 79, 421–427. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.; Bhattacharyya, D.; Hunter, P.J. Experimental characterisation and object-oriented finite element modelling of polypropylene/organoclay nanocomposites. Compos. Sci. Technol. 2008, 68, 2864–2875. [Google Scholar] [CrossRef]
- Silani, M.; Ziaei-Rad, S.; Talebi, H.; Rabczuk, T. A semi-concurrent multiscale approach for modeling damage in nanocomposites. Theor. Appl. Fract. Mec. 2014, 74, 30–38. [Google Scholar] [CrossRef]
- Yunos, D.M.; Bretcanu, O.; Boccaccini, A.R. Polymer-bioceramic composites for tissue engineering scaffolds. J. Mater. Sci. 2008, 43, 4433–4442. [Google Scholar] [CrossRef]
- Velard, F.; Braux, J.; Amedee, J.; Laquerriere, P. Inflammatory cell response to calcium phosphate biomaterial particles: An overview. Acta Biomater. 2013, 9, 4956–4963. [Google Scholar] [CrossRef] [PubMed]
- Roohani-Esfahani, S.I.; Nouri-Khorasani, S.; Lu, Z.; Appleyard, R.; Zreiqat, H. The influence hydroxyapatite nanoparticle shape and size on the properties of biphasic calcium phosphate scaffolds coated with hydroxyapatite-PCL composites. Biomaterials 2010, 31, 5498–5509. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Zhai, D.; Wu, C.; Chang, J. Poly(d, l-lactic)-reinforced akermanite bioceramic scaffolds: Preparation and characterization. Ceram. Int. 2014, 40, 12765–12775. [Google Scholar] [CrossRef]
- Wang, G.; Moya, S.; Lu, Z.; Gregurec, D.; Zreiqat, H. Enhancing orthopedic implant bioactivity: Refining the nanotopography. Nanomedicine 2015, 10, 1327–1341. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Wang, G.; Liu, Y.; Zhao, X.; Zou, D.; Zhu, C.; Jin, Y.; Huang, Q.; Sun, J.; Liu, X.; et al. The synergistic effect of hierarchical micro/nano-topography and bioactive ions for enhanced osseointegration. Biomaterials 2013, 34, 3184–3195. [Google Scholar] [CrossRef] [PubMed]
- Mohseni, E.; Zalnezhad, E.; Bushroa, A.R. Comparative investigation on the adhesion of hydroxyapatite coating on Ti–6Al–4V implant: A review paper. Int. J. Adhes. Adhes. 2014, 48, 238–257. [Google Scholar] [CrossRef]
- Yi, D.; Wu, C.; Ma, X.; Ji, H.; Zheng, X.; Chang, J. Preparation and in vitro evaluation of plasma-sprayed bioactive akermanite coatings. Biomed. Mater. 2012, 7, 065004. [Google Scholar] [CrossRef] [PubMed]
- Xue, W.; Liu, X.; Zheng, X.; Ding, C. Plasma-sprayed diopside coatings for biomedical applications. Surf. Coat. Technol. 2004, 185, 340–345. [Google Scholar] [CrossRef]
- Wu, C.; Ramaswamy, Y.; Liu, X.; Wang, G.; Zreiqat, H. Plasma-sprayed CaTiSiO5 ceramic coating on Ti-6Al-4V with excellent bonding strength, stability and cellular bioactivity. J. R. Soc. Interface 2009, 6, 159–168. [Google Scholar] [CrossRef] [PubMed]
- Liang, Y.; Xie, Y.; Ji, H.; Huang, L.; Zheng, X. Excellent stability of plasma-sprayed bioactive Ca3ZrSi2O9 ceramic coating on Ti-6Al-4V. Appl. Surf. Sci. 2010, 256, 4677–4681. [Google Scholar] [CrossRef]
- Liu, X.; Ding, C. Characterization of plasma sprayed wollastonite powder and coatings. Surf. Coat. Technol. 2002, 153, 173–177. [Google Scholar] [CrossRef]
- Liu, X.; Tao, S.; Ding, C. Bioactivity of plasma sprayed dicalcium silicate coatings. Biomaterials 2002, 23, 963–968. [Google Scholar] [CrossRef]
- Liu, X.; Morra, M.; Carpi, A.; Li, B. Bioactive calcium silicate ceramics and coatings. Biomed. Pharmacother. 2008, 62, 526–529. [Google Scholar] [CrossRef] [PubMed]
- Bakhsheshi-Rad, H.R.; Hamzah, E.; Kasiri-Asgarani, M.; Jabbarzare, S.; Daroonparvar, M.; Najafinezhad, A. Fabrication, degradation behavior and cytotoxicity of nanostructured hardystonite and titania/hardystonite coatings on Mg alloys. Vacuum 2016, 129, 9–12. [Google Scholar] [CrossRef]
- Razavi, M.; Fathi, M.; Savabi, O.; Beni, B.H.; Razavi, S.M.; Vashaee, D.; Tayebi, L. Coating of biodegradable magnesium alloy bone implants using nanostructured diopside (CaMgSi2O6). Appl. Surf. Sci. 2014, 288, 130–137. [Google Scholar] [CrossRef]
- Razavi, M.; Fathi, M.; Savabi, O.; Vashaee, D.; Tayebi, L. In vitro study of nanostructured diopside coating on Mg alloy orthopedic implants. Mater. Sci. Eng. C Mater. Biol. Appl. 2014, 41, 168–177. [Google Scholar] [CrossRef] [PubMed]
- Razavi, M.; Fathi, M.; Savabi, O.; Razavi, S.M.; Heidari, F.; Manshaei, M.; Vashaee, D.; Tayebi, L. In vivo study of nanostructured diopside (CaMgSi2O6) coating on magnesium alloy as biodegradable orthopedic implants. Appl. Surf. Sci. 2014, 313, 60–66. [Google Scholar] [CrossRef]
- Razavi, M.; Fathi, M.; Savabi, O.; Razavi, S.M.; Beni, B.H.; Vashaee, D.; Tayebi, L. Controlling the degradation rate of bioactive magnesium implants by electrophoretic deposition of akermanite coating. Ceram. Int. 2014, 40, 3865–3872. [Google Scholar] [CrossRef]
- Razavi, M.; Fathi, M.; Savabi, O.; Hashemi Beni, B.; Vashaee, D.; Tayebi, L. Surface microstructure and in vitro analysis of nanostructured akermanite (Ca2MgSi2O7) coating on biodegradable magnesium alloy for biomedical applications. Colloids Surf. B 2014, 117, 432–440. [Google Scholar] [CrossRef] [PubMed]
- Razavi, M.; Fathi, M.; Savabi, O.; Vashaee, D.; Tayebi, L. In vivo study of nanostructured akermanite/PEO coating on biodegradable magnesium alloy for biomedical applications. J. Biomed. Mater. Res. A 2015, 103, 1798–1808. [Google Scholar] [CrossRef] [PubMed]


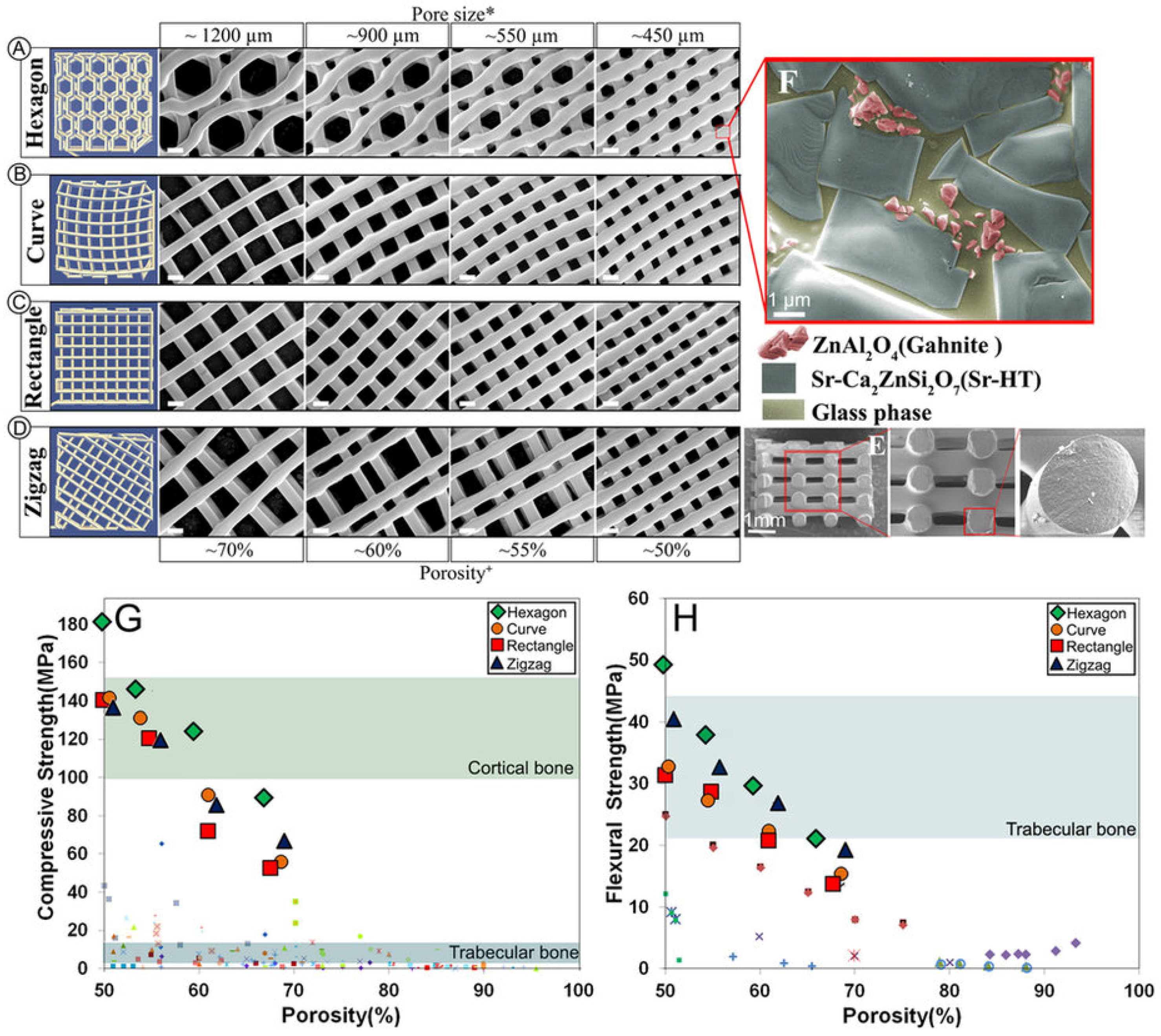

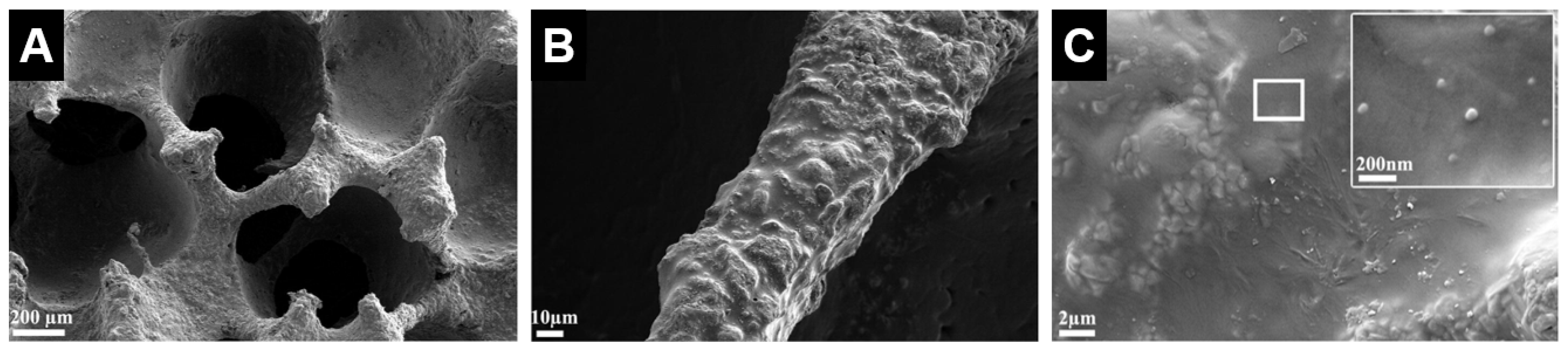
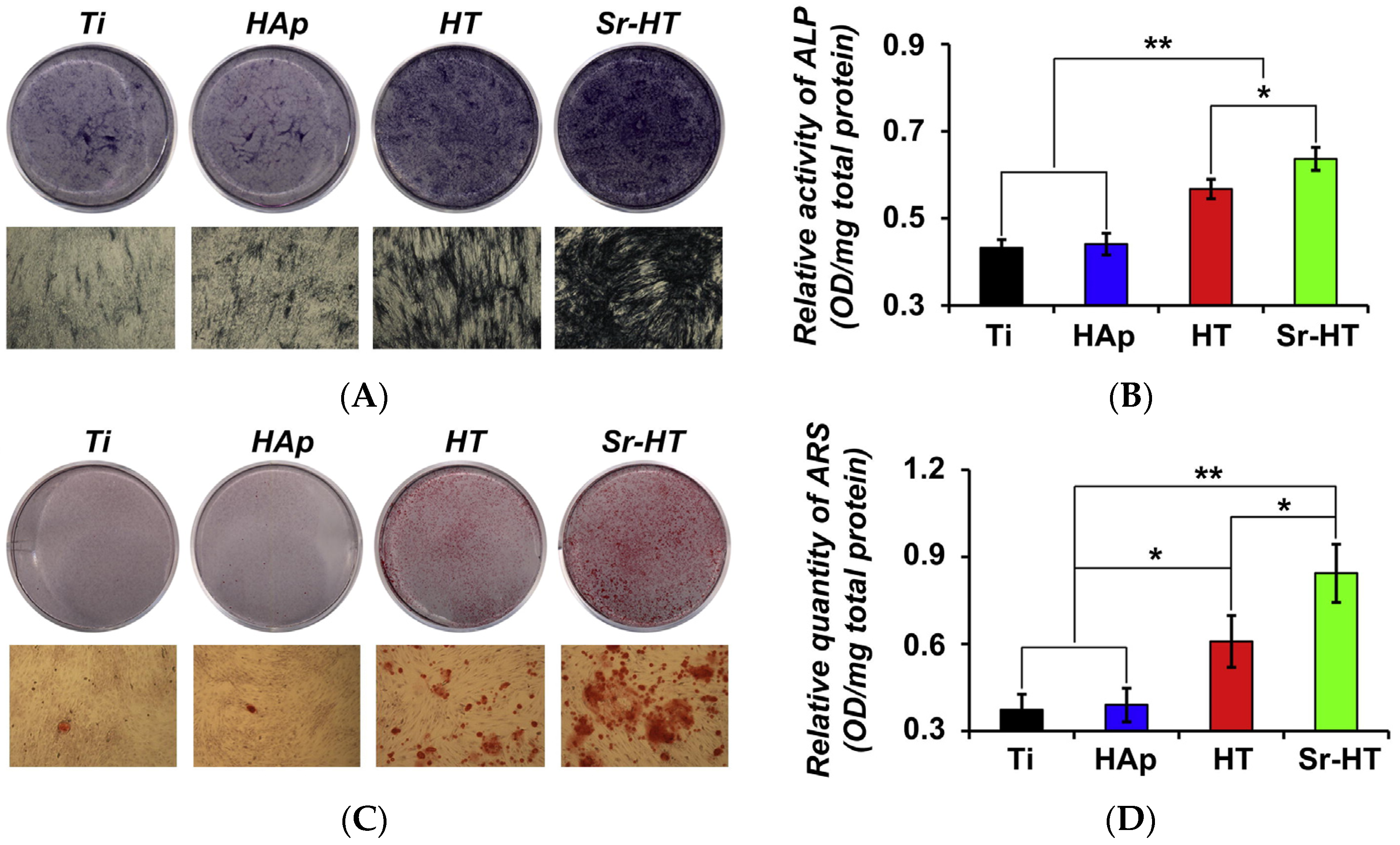
| Ceramic | Stoichiometric Formula | Fabrication Method and Heat Treatment | Ref. |
|---|---|---|---|
| α-calcium silicate (α-CS) (pseudowollastonite) | CaO–SiO2 | Chemical precipitation, sintered at 1250 °C for 3 h | [27] |
| β-calcium silicate (β-CS) | CaO–SiO2 | Chemical precipitation, sintered at 1100 °C for 3 h Chemical precipitation, sintered at 1090 °C for 2 h | [27] [30] |
| Sr-α-CaSiO3 (Sr-α-CS) | xSrO–(1 − x)CaO–SiO2; x = 0.01~0.10 | Chemical precipitation, sintered at 1250 °C for 3 h | [31] |
| Sr-β-CaSiO3 (Sr-β-CS) | xSrO–(1 − x)CaO–SiO2; x = 0.10 | Chemical precipitation, sintered at 1090 °C for 2 h | [30] |
| Cu-β-CaSiO3 (Cu-CS) | xCuO–(1 − x)CaO–SiO2; x = 0.025 | Chemical precipitation, calcined at 900 °C for 2 h | [32] |
| Akermanite (AK) | 2CaO–MgO–2SiO2 | Sol-gel, sintered at 1370 °C for 6 h | [37] |
| Co-Akermanite (Co-AK) | 2CaO–CoO–2SiO2 | Sol-gel, sintered at 1200 °C for 3 h | [42] |
| Diopside (DS) | CaO–MgO–2SiO2 | Co-precipitation, sintered at 1300 °C for 2 h | [36] |
| Bredigite (BD) | 7CaO–4SiO2–MgO | Sol-gel, sintered at 1350 °C for 8 h | [38] |
| Hardystonite (HT) | 2CaO–ZnO–2SiO2 | Sol-gel, sintered at 1350 °C for 5 h Sol-gel, sintered at 1250 °C for 3 h | [39] [25] |
| Sr-hardystonite (Sr-HT) | xSrO–(2 − x)CaO–ZnO–2SiO2; x = 0.10 | Sol-gel, sintered at 1250 °C for 3 h | [25] |
| Sphene (Sph) | CaO–TiO2–SiO2 | Sol-gel, sintered at 1280 °C, time not reported | [40] |
| Baghdadite (Bag) | 3CaO–ZrO2–2SiO2 | Sol-gel, sintered at 1400 °C for 3 h Solid-state sintering at 1400 °C for 3 h | [33] [51] |
| Sr-Bag (Sr-Bag) | xSrO–(3 − x)CaO–ZrO2–2SiO2, x = 0.1, 0.75 | Solid-state sintering at 1400 °C for 3 h | [41] |
| Cuprorivaite (Cup) | CaO–CuO–4SiO2 | Sol-gel, calcined at 1000 °C | [35] |
| Gehlenite (GLN) | 2CaO–Al2O3–SiO2 | Solid-state sintering at 1400 °C for 3 h | [34] |
| Ceramic | Porosity (%) | Young’s Modulus (GPa) | Mechanical Strength (MPa) | Fracture Toughness (MPa·m1/2) | Ref. |
|---|---|---|---|---|---|
| α-calcium silicate | 15.5 | NR | 39.7B | NR | [27] |
| 82.2PSST | ~0.012 | 0.3C | NR | [29] | |
| ~89PSST | NR | 0.03C | NR | [28] | |
| β-calcium silicate | 18.6 | NR | 65.9B | NR | [27] |
| Sr-α-CaSiO3 | No mechanical property evaluation | ||||
| Sr-β-CaSiO3 | No mechanical property evaluation | ||||
| Cu-β-CaSiO3 | No mechanical property evaluation | ||||
| Akermanite | 10.4 | 42 | 176.2B | 1.83 | [68] |
| 63.5PSST 81.7PSST 90.3PSST | NR | 1.13C 0.79C 0.53C | NR | [69] | |
| 57.9SLS | NR | 5.9C | 1.72 | [63] | |
| 53DIW | ~0.5 | 71C | NR | [64] | |
| Co-akermanite | No mechanical property evaluation | ||||
| Diopside | NR (dense) | 170 | 300B | 3.5 | [52] |
| 75PSST 82PSST | 0.07 0.01 | 1.4C 0.5C | NR | [70] | |
| Bredigite | 5.8 ~90PSST | 43 NR | 156B 0.233C | 1.57 NR | [38] |
| Hardystonite | 17.4 | NR | 136.4B | 1.24 | [39] |
| 77.5PSST | NR | 1.99 ± 0.45C | NR | [25] | |
| ~89PSST | NR | 0.06 | NR | [28] | |
| 74DIW | NR | 1.6 ± 0.3C | NR | [65] | |
| Sr-hardystonite | 78PSST | NR | 2.16 ± 0.52C | NR | [25] |
| Sphene | No mechanical property evaluation | ||||
| Baghdadite | 0.5 | 120 | 98B | 1.3 | [51] |
| 2.8 | NR | 168B | 1.2 | [41] | |
| ~88PSST | ~0.0153 | ~0.27C | NR | [71] | |
| Sr-Baghdadite | 3.4 | NR | 162B | 1.3 | [41] |
| Cuprorivaite | No mechanical property evaluation | ||||
| Gehlenite | 0.3 | 112 | 162B; 403C | 2.7 | [34] |
| Bioglass 45S5 | Dense | 35 | 42 | NR | [72] |
| Bioglass 45S5-derived scaffold | 86–94 | NR | 0.3–1.2B; 0.05–0.45C | NR | [73] |
| Hydroxyapatite (HA) | NR (dense) | 47 | 110B | 1.1 | [52] |
| <0.8 | 80–110 | 100–160B; 500C | 1.0 | [72] | |
| 2.2–7.0 | 87–97 | 84–113B | 0.69–0.96 | [74] | |
| β-Tricalcium phosphate (β-TCP) | <0.3 | 33–90 | 140–154B; 460–687C | NR | [72] |
| 0.6~1.4 | 87–95 | 118–133B | 1.14–1.30 | [74] | |
| Biphasic calcium phosphate (BCP) | ~88PSST | 0.0105 | 0.12C | NR | [71] |
| Cortical bone | 5–13 | 12–18 | 50–150B; 130–180C | 2–12 | [7,13,17] |
| Cancellous bone | 30–90 | 0.1–0.5 | 10–20B; 4–12C | 0.1–0.8 | [7,13,17] |
| Ceramic | Morphology and Concentration | Surrounding Aqueous Media | Weight Loss after 7 Days, (α-CS Value) | pH of Media after 7 Days, (α-CS Value) | Apatite Formation in SBF | Total ion Release in Media after 7 Days unless otherwise stated, (α-CS Value) | Ref. |
|---|---|---|---|---|---|---|---|
| β-calcium silicate | Solid disks, ratio of disk to media not reported | CM | NR | NR | Yes | Ca: ~160 ppm, (~120 ppm) | [84] |
| Si: ~90 ppm, (~80 ppm) | |||||||
| Sr-α-CaSiO3 | Solid disks, at 0.1 cm2/mL | SBF | 5% at 2.5 mol Sr, (7%) | 8.3, (8.4) | Yes | Ca: ~260 ppm, (~310 ppm) | [31] |
| Si: ~65 ppm, (~98 ppm) | |||||||
| Sr: ~2.6 ppm | |||||||
| 7% at 10 mol Sr, (7%) | 8.0, (8.4) | Yes | Ca: ~260 ppm, (~310 ppm) | [31] | |||
| Si: ~85 ppm, (~85 ppm) | |||||||
| Sr: ~7.9 ppm | |||||||
| Sr-β-CaSiO3 | No degradation evaluation of sintered disks/scaffolds | ||||||
| Cu-β-CaSiO3 | No degradation evaluation of sintered disks/scaffolds | ||||||
| Akermanite | Solid disks, at 0.1 cm2/mL | SBF | NR | 7.3 | Yes | Ca: ~240 ppm | [37] |
| Si: ~62 ppm | |||||||
| Mg: ~121 ppm | |||||||
| Solid disks, at 0.15 mm3/mL | Tris-HCl | 2.50% | NR | Yes | NR | [36] | |
| Solid disks in 48-well plate | CM | NR | NR | Ca: ~95 ppm | [36] | ||
| Si: ~26 ppm | |||||||
| Mg: ~30 ppm | |||||||
| Solid disks, 10 mm diameter in 1 mL solution | CM | NR | NR | Ca: ~100 ppm | [85] | ||
| Si: ~100 ppm | |||||||
| Mg: ~195 ppm | |||||||
| Porous scaffolds at 5 mg/mL | Ringer’s solution | 7% | NR | Yes | Cannot deduce concentration as volume of samples was not reported | [69] | |
| Co-akermanite | No degradation evaluation of sintered disks/scaffolds | ||||||
| Diopside | Solid disks, at 0.15 mm3/mL | Tris-HCl | 0.50% | NR | Yes | NR | [36] |
| Solid disks in 48-well plate | CM | NR | NR | Ca: ~87 ppm | [36] | ||
| Si: ~70 ppm | |||||||
| Mg: ~20 ppm | |||||||
| Porous scaffolds at 5 mg/mL | SBF | 1.00% | 7.5 | Yes | Si: ~150 ppm | [70] | |
| Bredigite | Solid disks, at 0.15 mm3/mL | Tris-HCl | 5% | NR | Yes | NR | [36] |
| Solid disks in 48-well plate | CM | NR | NR | Ca: ~70 ppm | [36] | ||
| Si: ~32 ppm | |||||||
| Mg: ~20 ppm | |||||||
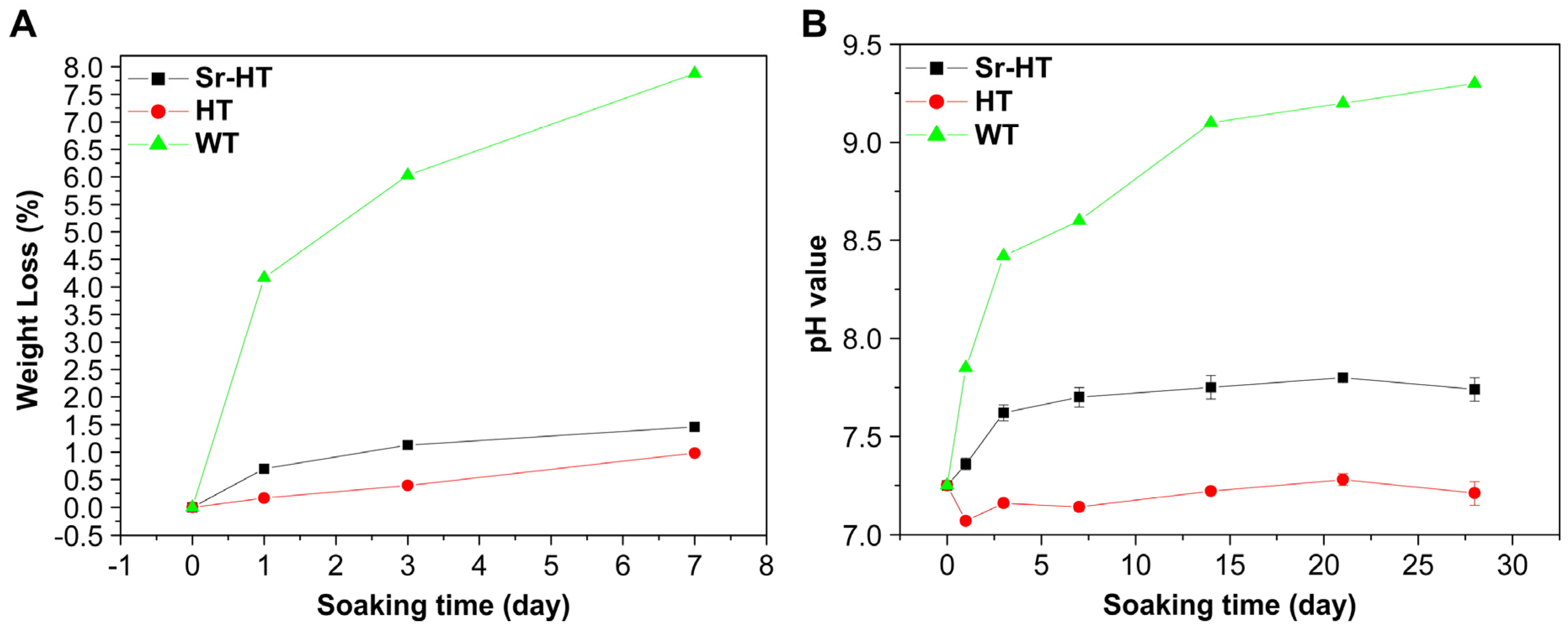
| Hardystonite | Solid disks, at 0.1 cm2/mL | SBF | NR | 7.5 | No | Ca: ~100 ppm14 days, (~600 ppm) | [81] |
| Si: ~33 ppm14 days, (~75 ppm) | |||||||
| Zn: ~0.4 ppm14 days | |||||||
| Porous scaffolds at 5 mg/mL | SBF | 0.7%, (8%) | 7.2, (8.6) | No | Ca: ~16 ppm, (340 ppm) | [25] | |
| Si: ~6 ppm, (98 ppm) | |||||||
| Zn: ~0.004 ppm | |||||||
| Porous scaffolds (7 × 7 × 7 mm3) in 15 mL | Tris-HCl | ~3%, (~11%) | 7.5, (8.2) | NR | Ca: 22 ppm, (144 ppm) | [28] | |
| Si: 5 ppm, (19 ppm) | |||||||
| Zn: 1 ppm | |||||||
| Sr-hardystonite | Porous scaffolds at 5 mg/mL | SBF | 1.2%, (8%) | 7.7, (8.6) | Yes | Ca: ~40 ppm, (340 ppm) | [25] |
| Si: ~11 ppm, (98 ppm) | |||||||
| Zn: ~0.0005 ppm | |||||||
| Sr: ~0.6 ppm | |||||||
| Sphene | Solid disks, at 0.1 cm2/mL | SBF | ~0%, (7%) | ~7.7, (~8.4) | No | Ca: ~20 ppm, (~310 ppm) | [40] |
| Si: 0 ppm, (~98 ppm) | |||||||
| Ti: 0 ppm | |||||||
| Baghdadite | Solid disks, ratio of disk to media not reported | CM | NR | 7.5, (8.1) | Yes | Ca: ~370 ppm, (~384 ppm) | [33] |
| Si: ~44 ppm, (~49 ppm) | |||||||
| Zr: 0 ppm | |||||||
| Porous scaffolds, 150 mg/L | SBF | 9% | 8 | Yes | Ca: ~200 ppm | [71] | |
| Si: ~32 ppm | |||||||
| Zr: 0.0005 ppm | |||||||
| Sr-Baghdadite | No degradation evaluation of sintered disks/scaffolds | ||||||
| Cuprorivaite | No degradation evaluation of sintered disks/scaffolds | ||||||
| Gehlenite | Solid disks, at 0.1 mm2/mL | SBF | ~0% | ~7.4 | No | Ca: ~45 ppm9 days, SBF | [34] |
| Tris-HCl | ~1% | ~7.4 | Si: ~5 ppm9 days, SBF | ||||
| Citric acid | ~7% | ~4 | Al: ~10 ppm9 days, SBF | ||||
| Ceramic | XMAC at 20 keV (Dense Material) |
|---|---|
| Cortical bone | 4.00 |
| Bioglass 45S5 | 4.09 |
| Diopside | 4.27 |
| Gehlenite | 5.31 |
| Akermanite | 5.36 |
| α-, β-CaSiO3 | 5.94 |
| Hydroxyapatite | 6.38 |
| Tricalcium phosphate | 6.49 |
| Bredigite | 6.62 |
| Sphene | 7.53 |
| Cu-β-CaSiO3 (2.5 mol % substitution of Ca) | 9.26 |
| Cuprorivaite | 9.54 |
| Sr-α-, β- CaSiO3 (10 mol % substitution of Ca) | 9.90 |
| Co-akermanite | 9.91 |
| Hardystonite | 12.96 |
| Sr-hardystonite (5 mol % substitution of Ca) | 13.61 |
| Baghdadite | 20.76 |
| Sr-Baghdadite (25 mol % substitution of Ca) | 21.74 |
| Ceramic | Cell Type | Ceramic Morphology | Main Findings | Ref. |
|---|---|---|---|---|
| Sr-α-CaSiO3 (Sr-α-CS) | Human bone-derived cells | Powder ionic extract | Sr ions in Sr-α-CS extract enhanced cell proliferation at lower Ca and Si concentrations, compared to α-CS extracts with no Sr | [31] |
| Sr-β-CaSiO3 (Sr-β-CS) | Ovariectomised rat bone marrow-derived stem cells | Powder ionic extract | Enhanced cell proliferation, ALP activity, and osteogenic gene expression (Runx2, BSP, OC, VEGF, OPG/RANKL ratio) in Sr-β-CS extract (6.25 mg/mL) compared to β-CS extract | [30] |
| Human umbilical vein endothelial cells | Powder ionic extract | Enhanced cell proliferation, angiogenic gene expression (VEGF, KDR), and in vitro angiogenesis in Sr-β-CS extract (3.1~12.5 mg/mL) compared to β-CS extract | [30] | |
| Cu-β-CaSiO3 (Cu-β-CS) | Human umbilical vein endothelial cells | Powder ionic extract | No difference in cell proliferation between β-CS and Cu-β-CS extracts; enhanced angiogenic gene expression (VEGF, KDR, HIF-1α) and in vitro angiogenesis in Sr-β-CS extract (3.1~12.5 mg/mL) compared to β-CS extract | [32] |
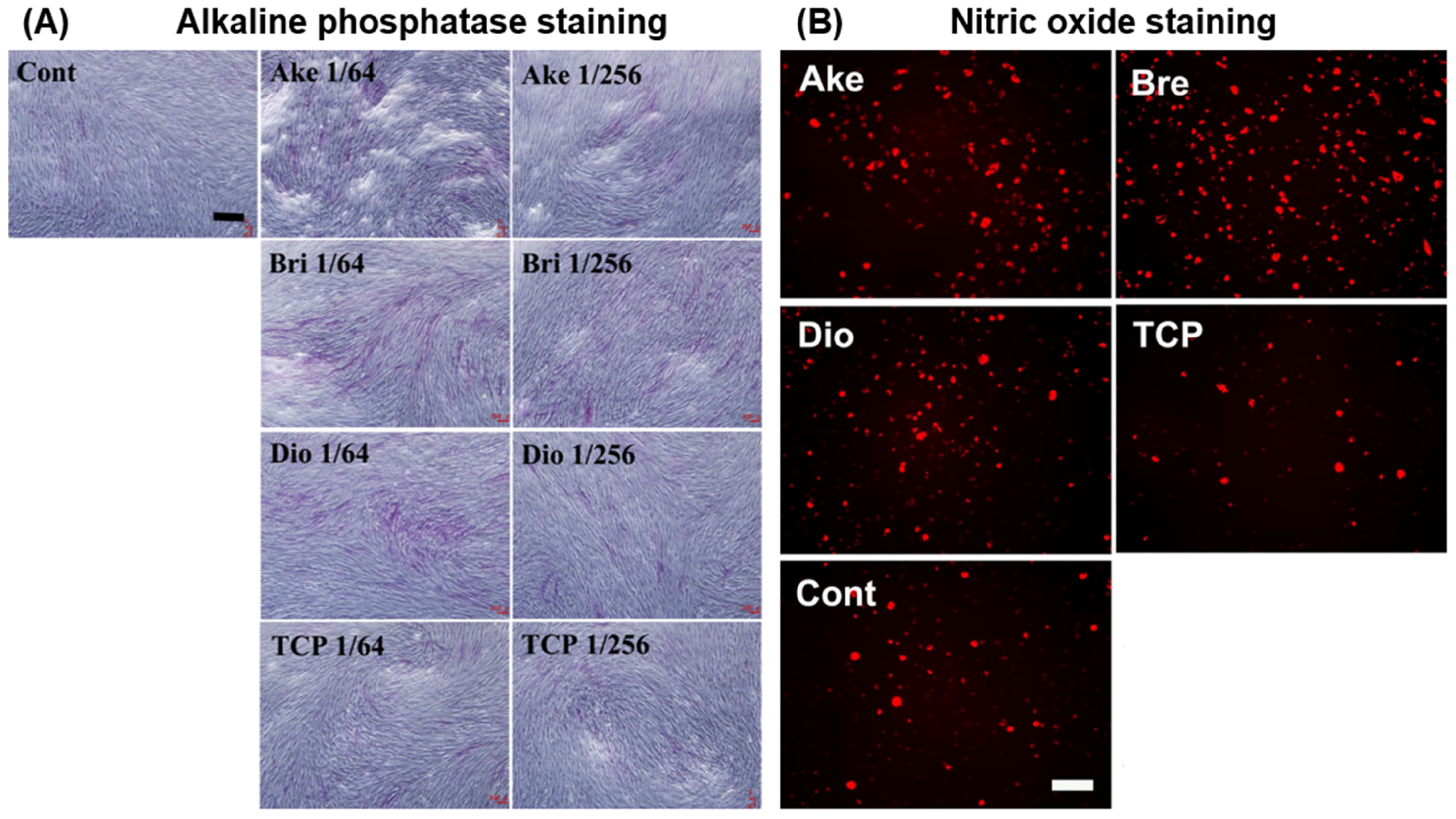
| Akermanite (AK) | Human bone marrow-derived stromal cells | Powder ionic extract | Enhanced proliferation, ALP activity, and osteogenic gene expression (OC, OPN) in AK extract (0.78 mg/mL) compared to β-TCP control | [98] |
| Human bone marrow-derived stromal cells | Direct seeding on dense ceramic disks | Enhanced proliferation, ALP activity, and osteogenic gene expression (ALP, BSP, OPN) on AK disk compared to β-TCP control | [99] | |
| Calf bone marrow stromal cells | Direct seeding on porous scaffold | Cells attached on AK scaffold; no significant difference in cell proliferation and ALP activity on AK scaffold compared to tissue culture plastic | [69] | |
| Human periodontal ligament cells | Direct seeding on dense ceramic disks | Enhanced attachment, proliferation, and osteogenic gene expression (OPN, DMP-1, OC) on AK disk compared to β-TCP control | [85] | |
| Human adipose-derived stem cells | Powder ionic extract | Slight inhibition of proliferation at high AK extract concentrations (25~100 mg/mL) compared to no AK extract control; significantly enhanced ALP activity, mineralisation, and OCN synthesis of cells in AK extract (25~50 mg/mL) compared to no extract control; enhanced osteogenic gene expression (Cbfα1, ALP, OCN), but reduced Col1 expression compared to no extract control; ERK pathway implicated in stimulation of osteogenic differentiation | [94] | |
| Human induced pluripotent stem cells | Powder ionic extract | AK extracts had no cytotoxic effects or effects on cell stemness; enhanced ALP activity, mineralisation, and osteogenic gene expression (ALP, BMP-2, Col1, OCN, Runx2) compared to culture medium without AK extract, with optimal extract concentration at 1.56 mg/mL | [97] | |
| Rat bone marrow-derived stem cells | Powder ionic extract | Enhanced proliferation, ALP activity, osteogenic (Runx2, BMP-2, BSP, OPN, OC, OPG/RANKL) and angiogenic (VEGF, ANG-1) gene expression, and inhibited TNF-α expression of cells in AK extract (12.5 mg/mL) compared to β-TCP control; activated ERK, P38, AKT and STAT3 pathways | [100] | |
| Rat bone marrow macrophages | Powder ionic extract | Inhibited mature osteoclast formation and osteoclastogenesis (TRAP, cathepsin K, NFATcl) compared to β-TCP control | [100] | |
| Human bone marrow-derived mesenchymal stem cells | Powder ionic extract | Enhanced cell proliferation (at 0.78–3.1 mg/mL), ALP activity, and osteogenic gene expression (OPN, Col1) compared to β-TCP extract | [101] | |
| Human aortic endothelial cells | Powder ionic extract | Enhanced cell proliferation, nitric oxide synthesis, angiogenic gene expression (eNOs, KDR, FGFR1, ACVRL1), and in vitro angiogenesis in AK extract (3.1~12.5 mg/mL) compared to β-TCP extract and ceramic-free control | [101] | |
| Co-akermanite (Co-AK) | Mouse osteoblast-like cells (MC3T3-E1) | Powder ionic extract | Inhibited cell proliferation in Co-AK extract (6.25–200 mg/mL); enhanced ALP activity in Co-AK extract of 0.78 mg/mL compared to β-CS | [42] |
| Human umbilical vein endothelial cells | Powder ionic extract | Inhibited cell proliferation in Co-AK extract (50–200 mg/mL); enhanced angiogenic gene expression (VEGF, eNOs) and in vitro angiogenesis in Co-AK extract of 0.78 mg/mL compared to β-CS | [42] | |
| Diopside (DS) | Human periodontal ligament cells and human bone marrow-derived mesenchymal stem cells | Powder ionic extract | Enhanced proliferation of hPDLCs at 100–200 mg/mL compared to β-TCP and hardystonite; enhanced OCN expression of hBMSCs at 50 mg/mL | [95] |
| Human bone marrow derived-mesenchymal stem cells | Powder ionic extract | Enhanced cell proliferation (at 1.6 mg/mL), ALP activity, and osteogenic gene expression (OPN) compared to β-TCP extract | [101] | |
| Human aortic endothelial cells | Powder ionic extract | No significant difference in cell proliferation, nitric oxide synthesis, angiogenic gene expression (eNOs, KDR, FGFR1, ACVRL1), and in vitro angiogenesis compared to β-TCP extract and ceramic-free control | [101] | |
| Bredigite (BD) | Human bone marrow-derived mesenchymal stem cells | Powder ionic extract | Enhanced cell proliferation (at 0.39–3.1 mg/mL), ALP activity, and osteogenic gene expression (OPN, Col1) compared to β-TCP extract | [101] |
| Human aortic endothelial cells | Powder ionic extract | Enhanced cell proliferation, nitric oxide synthesis, angiogenic gene expression (eNOs, KDR, FGFR1, ACVRL1), and in vitro angiogenesis in BD extract (3.1~12.5 mg/mL) compared to β-TCP extract and ceramic-free control | [101] | |
| Human periodontal ligament cells | Powder ionic extract | Enhanced cell proliferation at 6.25–25 mg/mL compared to tissue culture plastic; enhanced ALP activity and osteogenic gene expression (ALP, OC, OPN, BSP, CAP, CEMP1) at 50 mg/mL compared to tissue culture plastic; shown to activate Wnt/β-catenin signalling pathway | [96] | |
| Hardystonite (HT) | Human osteoblast-like cells | Direct seeding on dense ceramic disks | Cells adhered; significantly enhanced cell proliferation and ALP activity of cells on HT disks compared to α-CS | [81] |
| Human bone marrow derived mesenchymal stem cells | Direct seeding on dense ceramic disks; indirect co-culture of cells and ceramic disk | Enhanced proliferation in indirect culture compared to β-TCP and tissue culture plastic, while proliferation rate was lower for direct seeding; higher ALP activity on HT compared to β-TCP; significantly higher osteogenic expression (Col1, ALP, OPN, BSP, OC) compared to β-TCP for direct seeding | [102] | |
| Human periodontal ligament cells and human bone marrow-derived mesenchymal stem cells | Powder ionic extract | Enhanced ALP expression of hBMSCs at 12.5 mg/mL compared to diopside and β-TCP; enhanced antibacterial effect against E. faecalis compared to β-TCP, comparable antibacterial effect with calcium hydroxide | [95] | |
| Primary human osteoblasts | Direct seeding on porous ceramic scaffolds | Enhanced cell attachment and BSP gene expression for cells seeded on HT compared to calcium silicate, while all other osteogenic genes tested (Runx2, OPN, OC, Col1, ALP) showed insignificant difference or reduced expression compared to calcium silicate | [28] | |
| Primary human osteoblasts | Direct seeding on porous ceramic scaffolds | Enhanced cell proliferation and ALP activity on HT scaffolds compared to β-TCP, and enhanced OPN gene expression compared to tissue culture plastic | [25] | |
| Sr-hardystonite (Sr-HT) | Primary human osteoblasts | Direct seeding on porous ceramic scaffolds | Enhanced osteogenic gene expression (OC, BSP, OPN, Runx2) on Sr-HT scaffolds compared to hardystonite scaffolds and tissue culture plastic | [25] |
| Sphene (Sph) | Primary human bone-derived cells | Direct seeding on dense ceramic disks | Cells adhered; significantly enhanced cell proliferation and ALP activity of cells on hardystonite disks compared to α-CS | [40] |
| Baghdadite (Bag) | Primary human osteoblasts | Direct seeding on dense ceramic disks | Enhanced proliferation, ALP activity, and osteogenic expression (Col1, ALP, BSP, OC, RANKL, OPG) on Bag disks compared to α-CS | [33] |
| Primary human monocytes | Direct seeding on dense ceramic disks | Bag disks supported osteoclast differentiation from monocytes as opposed to α-CS | [33] | |
| Human dermal microvascular endothelial cells | Direct seeding on dense ceramic disks | Bag disks supported endothelial cell attachment and enhanced expression of VE-cadherin as opposed to α-CS | [33] | |
| Primary human ostoblasts; adipose-derived stem cells | Direct seeding on dense ceramic disks; indirect co-culture | Bag disks showed enhanced osteogenic expression in HOBs (Runx2, BSP, OPN, OC) and ASCs (Runx2, OPN); Bag shown to modulate cross-talk between HOBs and ASCs via BMP-2 pathway | [103] | |
| Unactivated macrophages derived from primary human monocytes | Direct seeding on porous scaffold; indirect co-culture | Bag disks promoted upregulation of genes related to pro-remodelling M2c phenotype | [108] | |
| Human periodontal ligament cells | Direct seeding on dense ceramic disks; powdered extract | Enhanced ALP activity, upregulated cementogenic and osteogenic gene expression, and upregulated Wnt/β-catenin pathway-related genes compared to β-TCP for both direct and indirect culture methods | [109] | |
| Human osteoblasts | Direct seeding on dense ceramic disks | Enhanced attachment, proliferation, and ALP expression of cells on Bag disks compared to α-CS | [41] | |
| Sr-Baghdadite (Sr-Bag) | Human osteoblasts | Direct seeding on dense ceramic disks | Enhanced attachment, proliferation, and ALP expression of cells on Sr-baghdadite disks compared to α-CS, with optimal ALP expression at 0.7 mol % Sr substitution of calcium | [41] |
| Cuprorivaite (Cup) | Mouse osteoblast-like cells (MC3T3-E1) | Powder ionic extract | Cytotoxic at 25–200 mg/mL; inhibited ALP activity of cells cultured in 0.195–0.78 mg/mL Cup extract compared to β-CS | [35] |
| Human umbilical vein endothelial cells | Powder ionic extract | Cytotoxic at 25–200 mg/mL; enhanced in vitro angiogenesis and VEGF expression of cells cultured in 0.39–0.78 mg/mL Cup extract compared to β-CS extract and copper extract; has antibacterial effects against E. coli | [35] | |
| Gehlenite (GLN) | Primary human osteoblasts | Direct seeding on dense ceramic disks | Enhanced cell attachment, proliferation, and osteogenic gene expression (Runx2, OPN, BSP, OC) on GLN disks compared to biphasic calcium phosphate disks | [34] |
| Mouse bone marrow macrophages | Direct seeding on dense ceramic disks | Promoted formation of TRAP-positive osteoclasts, and enhanced osteoclast attachment and polarisation | [34] |
| Ceramic | Implant Morphology | Animal Model | Implantation Period | Main Findings | Ref. |
|---|---|---|---|---|---|
| Sr-β-CaSiO3 (Sr-β-CS) | Porous scaffolds | Ovariectomised rat calvarial defects | 4 weeks | µ-CT analysis showed higher bone mineral density, trabecular thickness, and bone volume/total volume ratio for Sr-β-CS compared to β-CS; histomorphometric analysis showed higher new bone area, blood vessel area, and faster in vivo degradation for Sr-β-CS compared to β-CS | [30] |
| Akermanite (AK) | Porous scaffolds | Rabbit femoral defects | 8 and 16 weeks | Fluorescence labelling showed no significant difference in mineral apposition rate of new bone formation between AK and β-TCP scaffolds; histomorphometric analysis showed slightly higher new bone formation, and faster in vivo degradation of AK scaffolds compared to β-TCP | [98] |
| Porous scaffolds | Ovariectomised rat calvarial defects | 2, 4, 6 and 8 weeks | µ-CT analysis showed higher trabecular thickness and bone volume/total volume ratio in AK scaffolds compared to β-TCP; polychrome sequential fluorescent labelling showed enhanced new bone growth and mineral apposition in AK scaffolds compared to β-TCP; histomorphometric assay showed higher new bone area and blood vessel area in AK scaffolds compared to β-TCP | [100] | |
| Diopside (DP) | Dense specimens | Rabbit jaw bone defects | 12 weeks | Direct, gradient bonding between native bone and DP implant | [52] |
| Dense spheres (1–1.5 mm diameter) | Rat femoral defects | 2 and 4 weeks | Histological analysis showed new bone growth which formed tissue bridges with DP spheres, slightly higher bone regeneration score compared to β-TCP, and evidence of dynamic endochondral ossification; quantitative analysis on histology sections showed higher Col1 expression and similar OPN expression compared to β-TCP | [111] | |
| Hardystonite (HT) | Porous scaffolds | Rat tibial defects | 3 and 6 weeks | HT scaffolds showed new bone formation inside scaffold pores in both the external cortex and internal medullary cavity, in comparison to only external cortex for β-TCP control at both 3 and 6 weeks; limited in vivo resorption and limited ALP activity compared to β-TCP | [25] |
| Sr-hardystonite (Sr-HT) | Porous scaffolds | Rat tibial defects | 3 and 6 weeks | Sr-HT scaffolds showed new bone formation inside scaffold pores in both the external cortex and internal medullary cavity, in comparison to only external cortex for β-TCP control at both 3 and 6 weeks; limited in vivo resorption but extensive ALP activity compared to hardystonite and β-TCP | [25] |
| Baghdadite (Bag) | Dense 1–1.5 mm diameter spheres | Rat femoral defects | 2 and 4 weeks | Histological analysis showed new bone growth which formed tissue bridges with Bag spheres, significantly higher bone regeneration score compared to β-TCP, and evidence of dynamic endochondral ossification with increased amount of regularly arranged woven bone compared to diopside and β-TCP; significantly higher Col1 expression and OPN expression compared to diopside and β-TCP scaffolds | [111] |
| Porous scaffolds | Rabbit radial segmental defects | 12 weeks | Radiographic analysis showed enhanced defect bridging for Bag scaffolds compared to BCP scaffold; histological analysis showed enhanced bone ingrowth into pores of Bag scaffold compared to mostly peripheral bone growth for BCP scaffold; histomorphometric analysis showed increased new bone formation in Bag scaffolds (3.0 ± 3.1 mm2) compared to BCP (1.3 ± 1.0 mm2) at the scaffold midpoint; observed evidence of osteoclast-mediated resorption | [71] | |
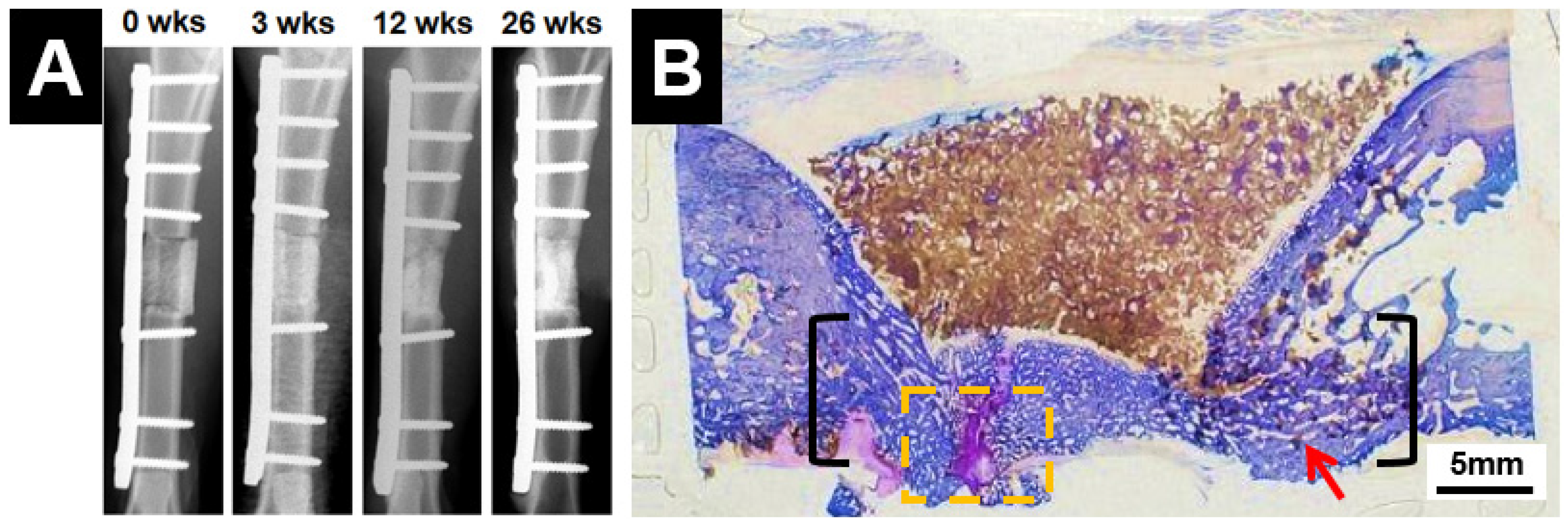
| Porous scaffolds | Sheep tibial segmental defects | Up to 26 weeks | Radiographic analysis showed clinical union at the bone-scaffold interface in all samples after 26 weeks; biomechanical analysis showed that torsional strength of the implant and associated bone reached ~10% of contralateral intact tibia; histological analysis showed average 80% bridging of the defect length in all samples, as well as new bone growth inside the scaffold pores | [110] |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
No, Y.J.; Li, J.J.; Zreiqat, H. Doped Calcium Silicate Ceramics: A New Class of Candidates for Synthetic Bone Substitutes. Materials 2017, 10, 153. https://doi.org/10.3390/ma10020153
No YJ, Li JJ, Zreiqat H. Doped Calcium Silicate Ceramics: A New Class of Candidates for Synthetic Bone Substitutes. Materials. 2017; 10(2):153. https://doi.org/10.3390/ma10020153
Chicago/Turabian StyleNo, Young Jung, Jiao Jiao Li, and Hala Zreiqat. 2017. "Doped Calcium Silicate Ceramics: A New Class of Candidates for Synthetic Bone Substitutes" Materials 10, no. 2: 153. https://doi.org/10.3390/ma10020153
APA StyleNo, Y. J., Li, J. J., & Zreiqat, H. (2017). Doped Calcium Silicate Ceramics: A New Class of Candidates for Synthetic Bone Substitutes. Materials, 10(2), 153. https://doi.org/10.3390/ma10020153








