Intratubular Biomineralization in a Root Canal Filled with Calcium-Enriched Material over 8 Years
Abstract
:1. Introduction
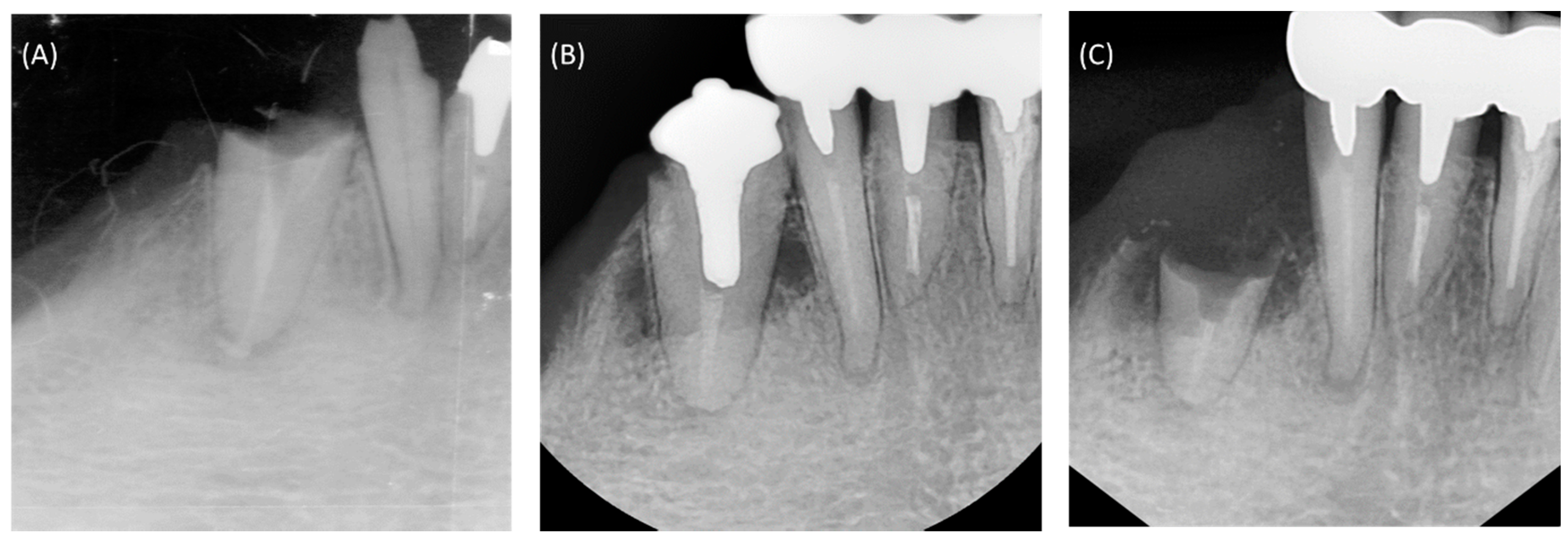
2. Clinical Presentation and Treatment
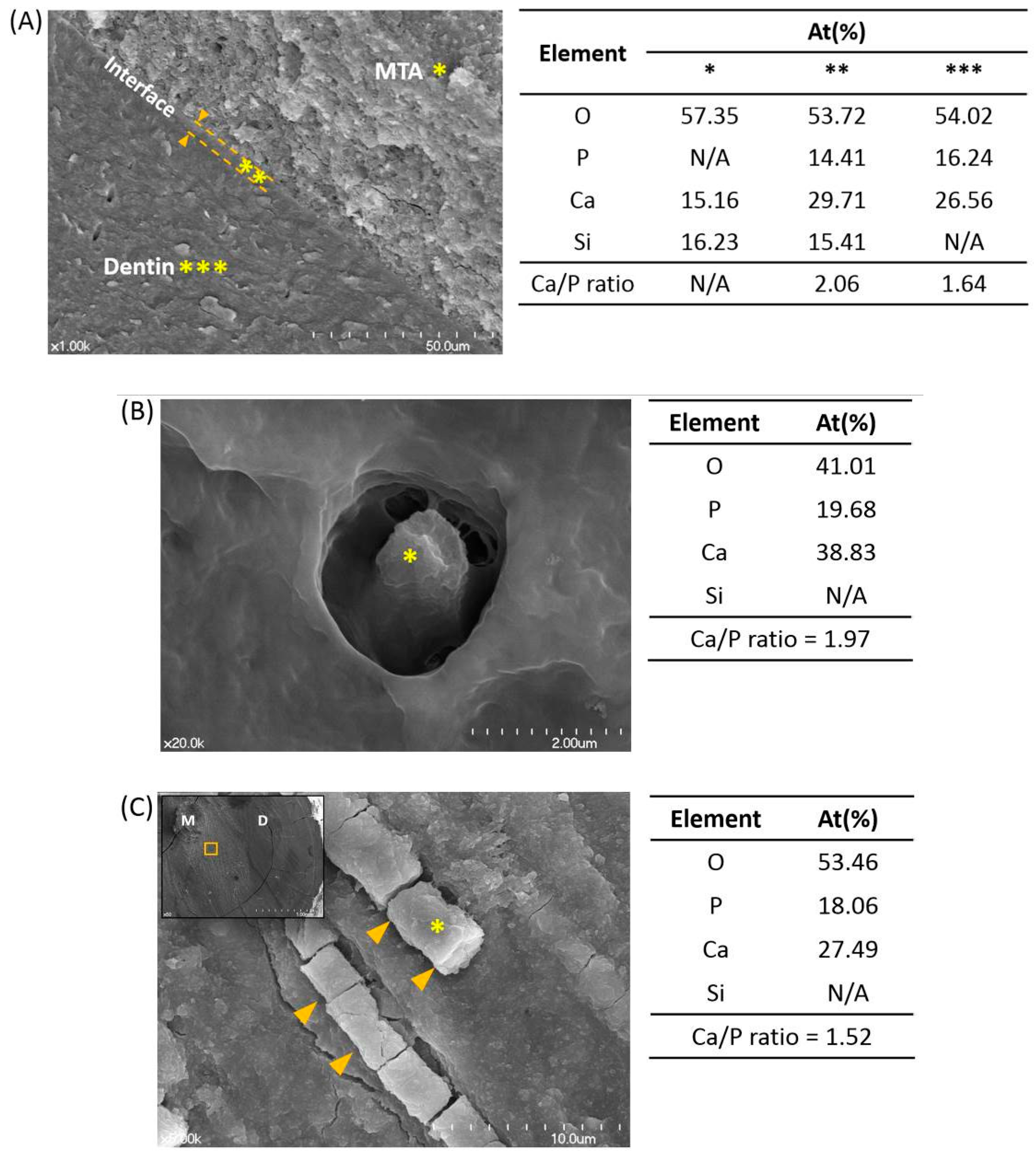
3. Laboratory Examination and Analysis
4. Results and Discussion
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Gandolfi, M.G.; Iezzi, G.; Piattelli, A.; Prati, C.; Scarano, A. Osteoinductive potential and bone-bonding ability of ProRoot MTA, MTA Plus and Biodentine in rabbit intramedullary model: Microchemical characterization and histological analysis. Dent. Mater. 2017, 33, e221–e238. [Google Scholar] [CrossRef] [PubMed]
- Boutsioukis, C.; Noula, G.; Lambrianidis, T. Ex vivo study of the efficiency of two techniques for the removal of mineral trioxide aggregate used as a root canal filling material. J. Endod. 2008, 34, 1239–1242. [Google Scholar] [CrossRef] [PubMed]
- Dreger, L.A.; Felippe, W.T.; Reyes-Carmona, J.F.; Felippe, G.S.; Bortoluzzi, E.A.; Felippe, M.C. Mineral trioxide aggregate and Portland cement promote biomineralization in vivo. J. Endod. 2012, 38, 324–329. [Google Scholar] [CrossRef] [PubMed]
- Reyes-Carmona, J.F.; Felippe, M.S.; Felippe, W.T. A phosphate-buffered saline intracanal dressing improves the biomineralization ability of mineral trioxide aggregate apical plugs. J. Endod. 2010, 36, 1648–1652. [Google Scholar] [CrossRef] [PubMed]
- Reyes-Carmona, J.F.; Felippe, M.S.; Felippe, W.T. The biomineralization ability of mineral trioxide aggregate and Portland cement on dentin enhances the push-out strength. J. Endod. 2010, 36, 286–291. [Google Scholar] [CrossRef] [PubMed]
- Reyes-Carmona, J.F.; Felippe, M.S.; Felippe, W.T. Biomineralization ability and interaction of mineral trioxide aggregate and white portland cement with dentin in a phosphate-containing fluid. J. Endod. 2009, 35, 731–736. [Google Scholar] [CrossRef] [PubMed]
- Yoo, Y.J.; Baek, S.H.; Kum, K.Y.; Shon, W.J.; Woo, K.M.; Lee, W. Dynamic intratubular biomineralization following root canal obturation with pozzolan-based mineral trioxide aggregate sealer cement. Scanning 2016, 38, 50–56. [Google Scholar] [CrossRef] [PubMed]
- Yoo, J.S.; Chang, S.W.; Oh, S.R.; Perinpanayagam, H.; Lim, S.M.; Yoo, Y.J.; Oh, Y.R.; Woo, S.B.; Han, S.H.; Zhu, Q.; et al. Bacterial entombment by intratubular mineralization following orthograde mineral trioxide aggregate obturation: A scanning electron microscopy study. Int. J. Oral Sci. 2014, 6, 227–232. [Google Scholar] [CrossRef] [PubMed]
- Oh, S.; Perinpanayagam, H.; Kum, D.J.W.; Lim, S.M.; Yoo, Y.J.; Chang, S.W.; Lee, W.; Baek, S.H.; Zhu, Q.; Kum, K.Y. Evaluation of three obturation techniques in the apical third of mandibular first molar mesial root canals using micro-computed tomography. J. Dent. Sci. 2016, 11, 95–102. [Google Scholar] [CrossRef]
- Chang, S.W.; Baek, S.H.; Yang, H.C.; Seo, D.G.; Hong, S.T.; Han, S.H.; Lee, Y.; Gu, Y.; Kwon, H.B.; Lee, W.; et al. Heavy metal analysis of ortho MTA and ProRoot MTA. J. Endod. 2011, 37, 1673–1676. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.Y.; Kim, K.J.; Yi, Y.A.; Seo, D.G. Quantitative microleakage analysis of root canal filling materials in single-rooted canals. Scanning 2015, 37, 237–245. [Google Scholar] [CrossRef] [PubMed]
- Atmeh, A.R.; Chong, E.Z.; Richard, G.; Festy, F.; Watson, T.F. Dentin-cement interfacial interaction: Calcium silicates and polyalkenoates. J. Dent. Res. 2012, 91, 454–459. [Google Scholar] [CrossRef] [PubMed]
- Camilleri, J.; Cutajar, A.; Mallia, B. Hydration characteristics of zirconium oxide replaced Portland cement for use as a root-end filling material. Dent. Mater. 2011, 27, 845–854. [Google Scholar] [CrossRef] [PubMed]
- Weng, J.; Liu, Q.; Wolke, J.G.; Zhang, X.; de Groot, K. Formation and characteristics of the apatite layer on plasma-sprayed hydroxyapatite coatings in simulated body fluid. Biomaterials 1997, 18, 1027–1035. [Google Scholar] [CrossRef]
- Sundqvist, G.; Figdor, D. Endodontic treatment of apical periodontitis. In Essential Endodontiology; Orstavik, D., Pitt Ford, T.R., Eds.; Oxford Blackwell Press: Oxford, UK, 1988; pp. 242–277. [Google Scholar]
- Figdor, D.; Davies, J.K.; Sundqvist, G. Starvation survival, growth and recovery of Enterococcus faecalis in human serum. Oral Microbiol. Immunol. 2003, 18, 234–239. [Google Scholar] [CrossRef] [PubMed]
- Gandolfi, M.G.; Taddei, P.; Modena, E.; Siboni, F.; Prati, C. Biointeractivity-related versus chemi/physisorption-related apatite precursor-forming ability of current root end filling materials. J. Biomed. Mater. Res. B Appl. Biomater. 2013, 101, 1107–1123. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.Y.; Oh, S.F.; Teng, N.C.; Kung, C.M.; Tsai, H.L.; Kuo-Tien, C.; Chu, K.L.; Ou, K.L. Phase transformation on bone cement: Monocalcium phosphate monohydrate into calcium-deficient hydroxyapatite during setting. Ceram. Int. 2013, 39, 2451–2455. [Google Scholar] [CrossRef]
- Seux, D.; Couble, M.L.; Hartmann, D.J.; Gauthier, J.P.; Magloire, H. Odontoblast-like cytodifferentiation of human dental pulp cells in vitro in the presence of a calcium hydroxide-containing cement. Arch. Oral Biol. 1991, 36, 117–128. [Google Scholar] [CrossRef]
- Holland, R.; de Souza, V.; Nery, M.J.; Estrada-Bernabé, P.F.; Filho, J.A.; Junior, E.D.; Murata, S.S. Calcium salts deposition in rat connective tissue after the implantation of calcium hydroxide-containing sealers. J. Endod. 2002, 28, 173–176. [Google Scholar] [CrossRef] [PubMed]
- Fridland, M.; Rosado, R. Mineral trioxide aggregate (MTA) solubility and porosity with different water-to-powder ratios. J. Endod. 2003, 29, 814–817. [Google Scholar] [CrossRef] [PubMed]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yoo, Y.-J.; Lee, Y.S.; Yoo, J.S.; Perinpanayagam, H.; Yoo, C.S.; Kang, H.S.; Oh, S.; Chang, S.W.; Kum, K.-Y. Intratubular Biomineralization in a Root Canal Filled with Calcium-Enriched Material over 8 Years. Materials 2017, 10, 1388. https://doi.org/10.3390/ma10121388
Yoo Y-J, Lee YS, Yoo JS, Perinpanayagam H, Yoo CS, Kang HS, Oh S, Chang SW, Kum K-Y. Intratubular Biomineralization in a Root Canal Filled with Calcium-Enriched Material over 8 Years. Materials. 2017; 10(12):1388. https://doi.org/10.3390/ma10121388
Chicago/Turabian StyleYoo, Yeon-Jee, Yoo Sang Lee, Jun Sang Yoo, Hiran Perinpanayagam, Chang Seon Yoo, Hyen Sug Kang, Soram Oh, Seok Woo Chang, and Kee-Yeon Kum. 2017. "Intratubular Biomineralization in a Root Canal Filled with Calcium-Enriched Material over 8 Years" Materials 10, no. 12: 1388. https://doi.org/10.3390/ma10121388
APA StyleYoo, Y.-J., Lee, Y. S., Yoo, J. S., Perinpanayagam, H., Yoo, C. S., Kang, H. S., Oh, S., Chang, S. W., & Kum, K.-Y. (2017). Intratubular Biomineralization in a Root Canal Filled with Calcium-Enriched Material over 8 Years. Materials, 10(12), 1388. https://doi.org/10.3390/ma10121388





