Aminated Polyethylene Terephthalate (PET) Nanofibers for the Selective Removal of Pb(II) from Polluted Water
Abstract
1. Introduction
2. Materials and Methods
2.1. Reactants and Solvents
2.2. Electrospinning Process
2.3. Aminolysis of PET Nanofiber Mats
2.4. Characterization
2.4.1. Scanning Electron Microscopy
2.4.2. Elemental Analysis
2.4.3. Kaiser Test
2.4.4. Attenuated Total Reflectance–Fourier Transform Infrared Spectrometry (ATR–FTIR)
2.4.5. Inductively Coupled Plasma Mass Spectroscopy (ICP-MS)
2.5. Adsorption Behavior in Batch Mode
2.6. Metal Adsorption in Continuous Mode
3. Results and Discussion
3.1. Electrospun PET Nanofibers
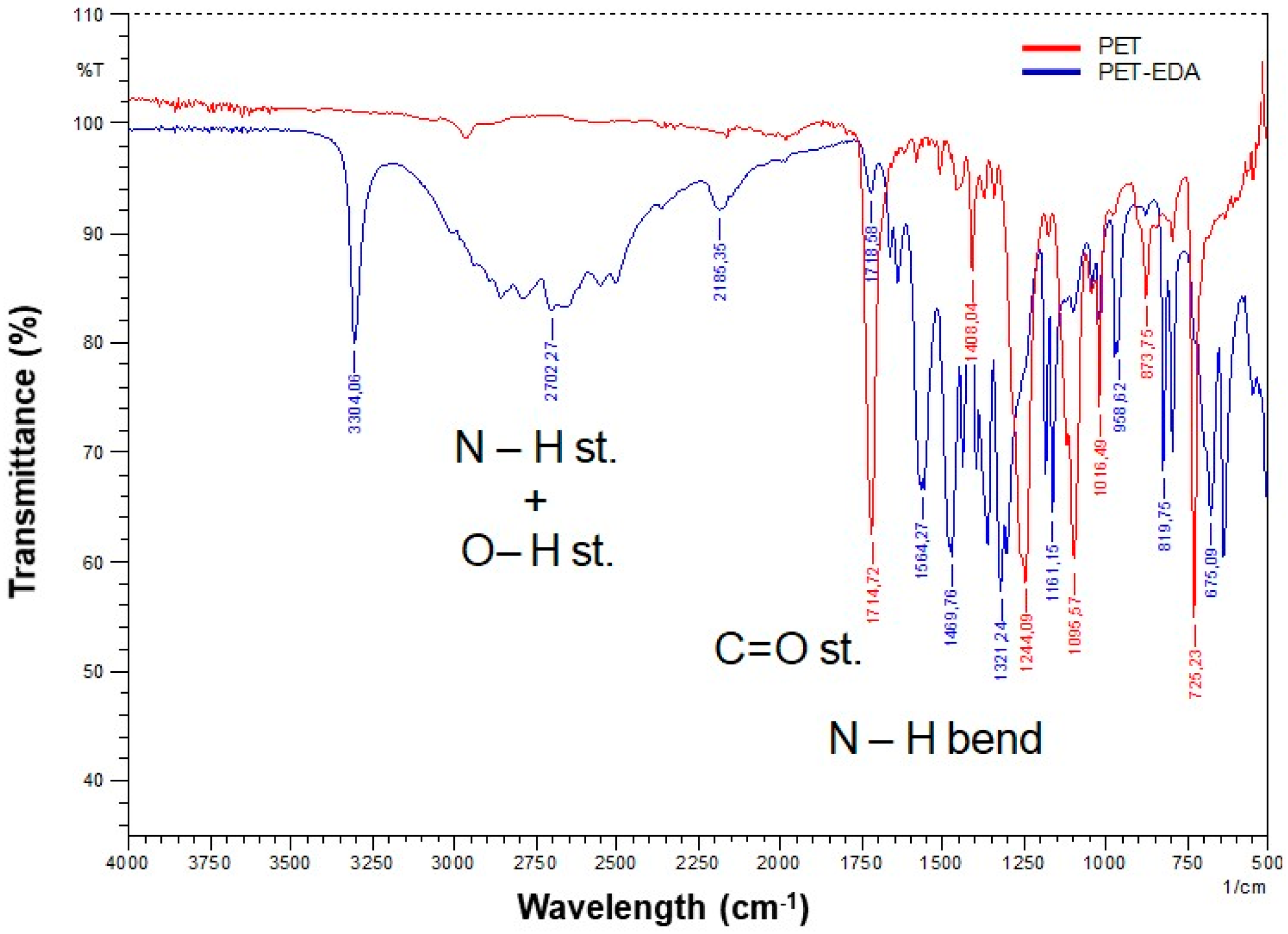
3.2. Aminated PET Nanofiber Mats
3.3. Adsorption Experiments in Batch Mode
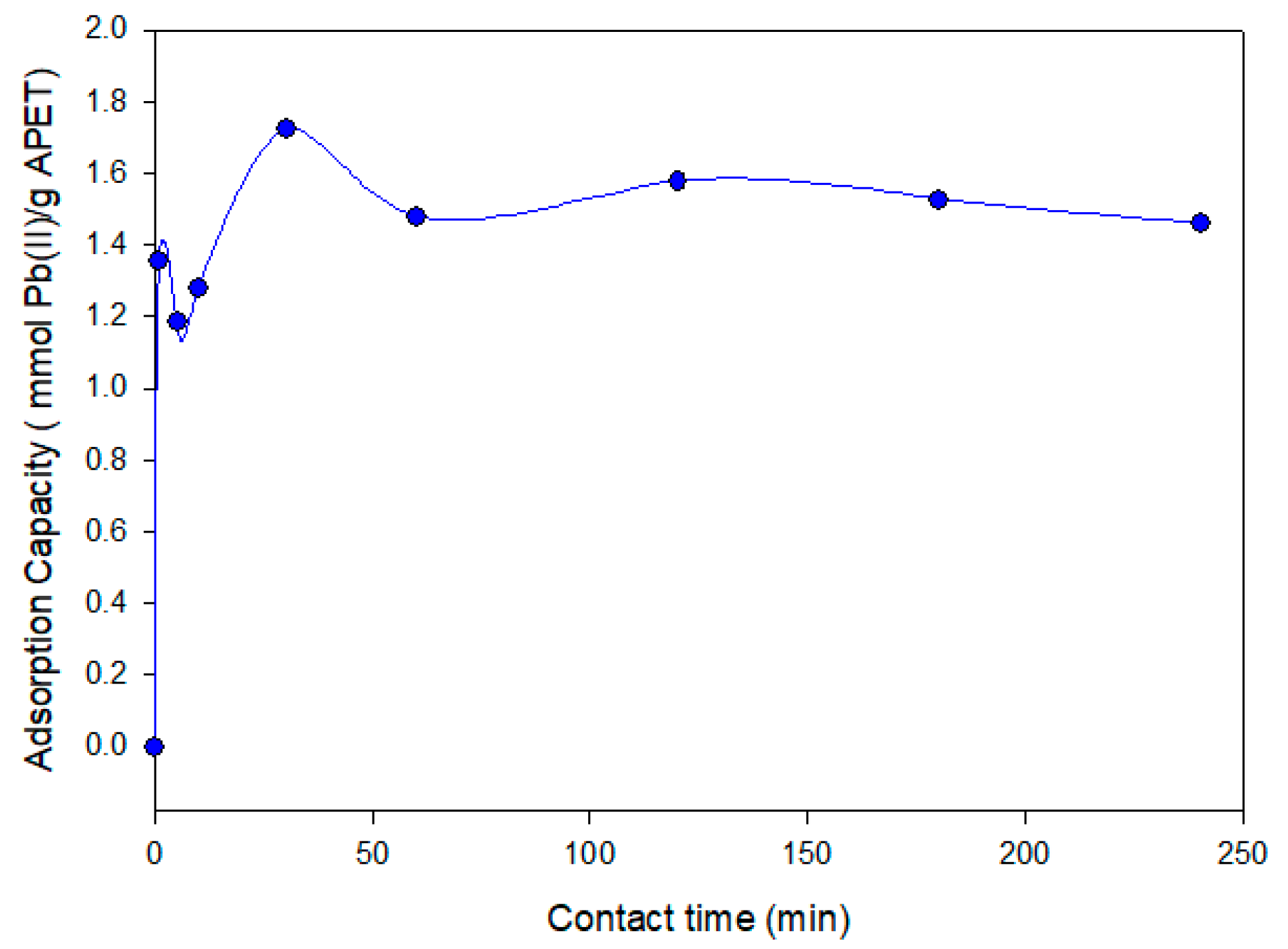
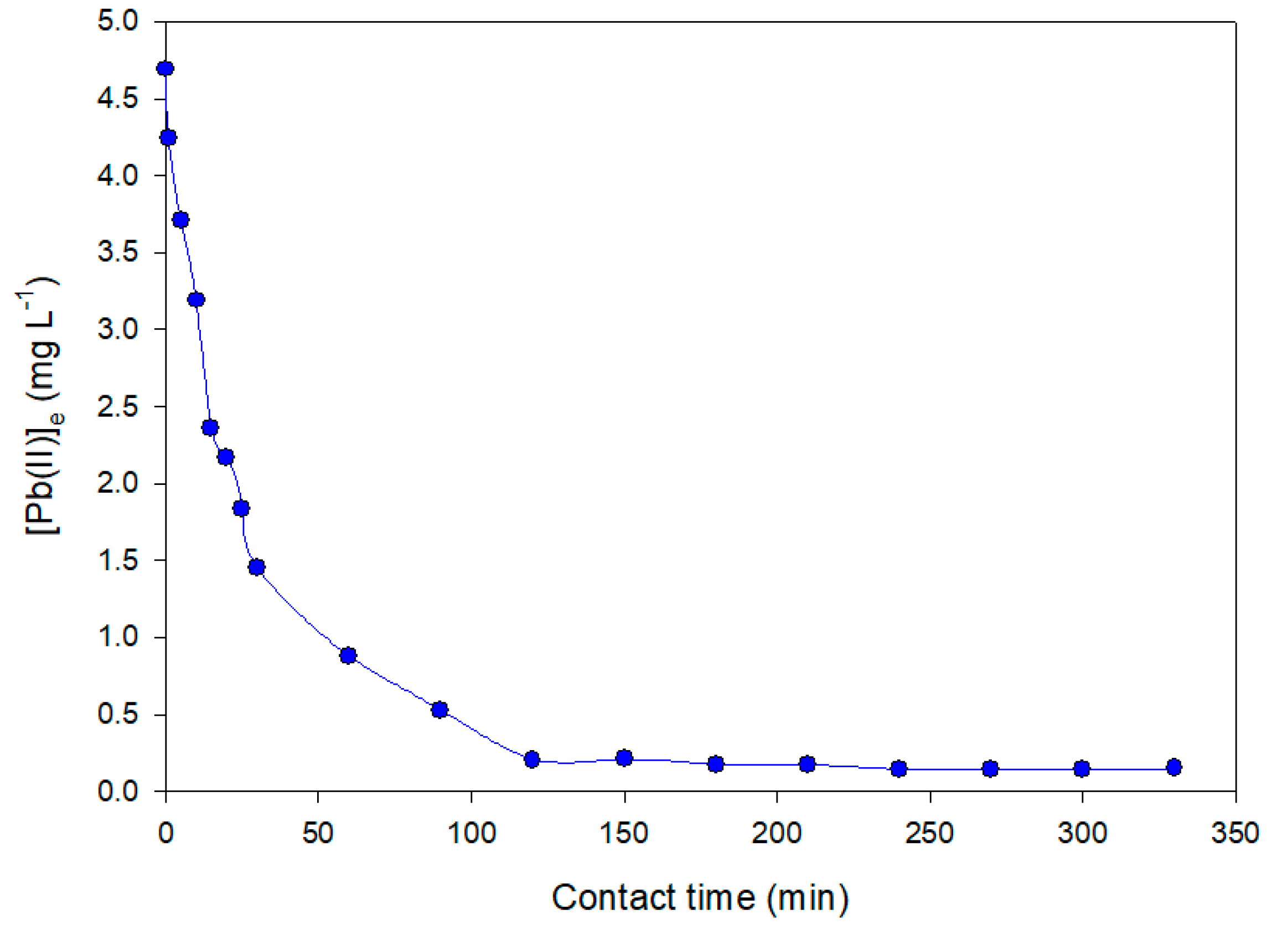
3.3.1. Effect of Contact Time in the Adsorption Process
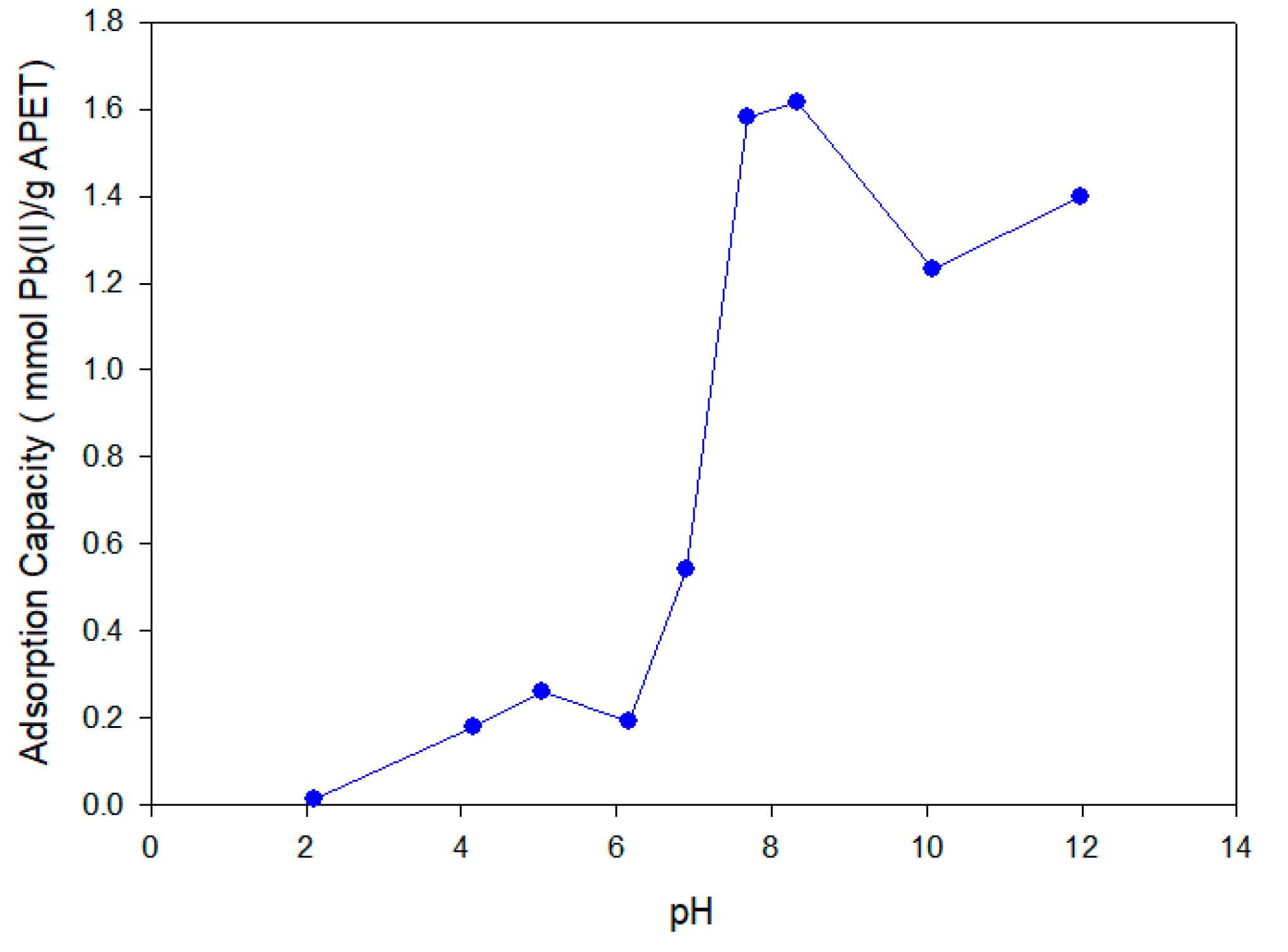
3.3.2. Effect of pH on the Adsorption Process
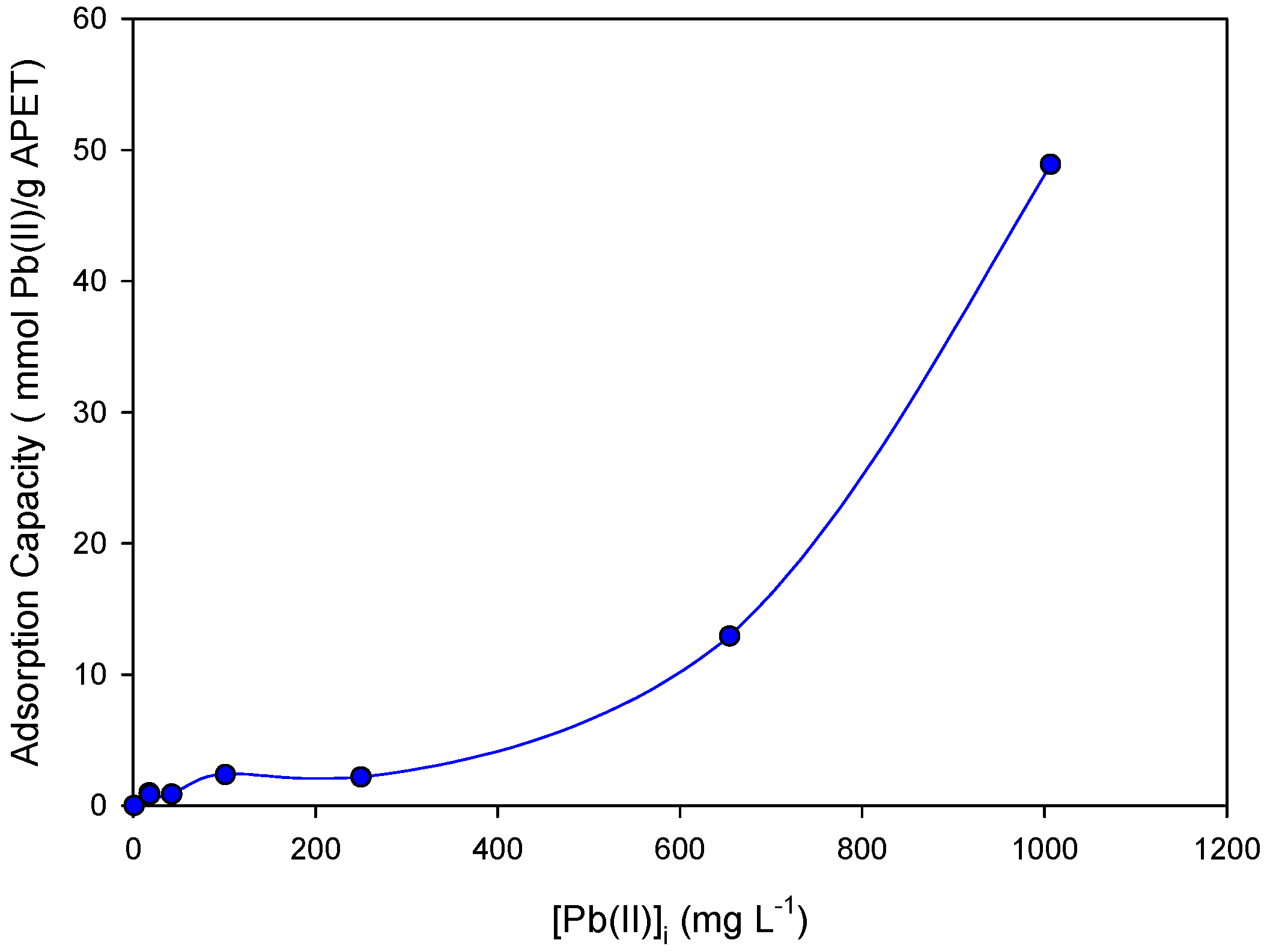
3.3.3. Effect of Metal Concentration on the Adsorption Process
3.3.4. Selectivity towards Interfering Metal Ions
3.4. Adsorption Experiments in Continuous Mode
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Hassaan, M.A.; El Nemr, A.; Madkour, F.F. Environmental assessment of heavy metal pollution and human health risk. Am. J. Water Sci. Eng. 2016, 2, 14–19. [Google Scholar]
- Gunatilake, S.K. Methods of removing heavy metals from industrial wastewater. J. Multidiscip. Eng. Sci. Stud. 2015, 1, 12–18. [Google Scholar]
- Singh, N.; Gupta, S.K. Adsorption of heavy metals: A review. Int J. Innov. Res. Sci. Eng. Technol. 2014, 5, 2267–2281. [Google Scholar]
- De Gisi, S.; Lofrano, G.; Grassi, M.; Notarnicola, M. Characteristics and adsorption capacities of low-cost sorbents for wastewater treatment: A review. Sustain. Mater. Technol. 2016, 9, 10–40. [Google Scholar] [CrossRef]
- Lee, S.S.; Bai, H.; Liu, Z.; Sun, D.D. Novel-structured electrospun TiO2/CuO composite nanofibers for high efficient photocatalytic cogeneration of clean water and energy from dye wastewater. Water Res. 2013, 47, 4059–4073. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Li, Y.; Li Puma, G.; Wang, C.; Wang, P.; Zhang, W.; Wang, Q. Mechanism and experimental study on the photocatalytic performance of Ag/AgCl @ chiral TiO2 nanofibers photocatalyst: The impact of wastewater components. J. Hazard. Mater. 2015, 285, 277–284. [Google Scholar] [CrossRef] [PubMed]
- Ren, T.; He, P.; Niu, W.; Wu, Y.; Ai, L.; Gou, X. Synthesis of α-Fe2O3 nanofibers for applications in removal and recovery of Cr (VI) from wastewater. Environ. Sci. Pollut. Res. 2013, 20, 155–162. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Liu, C.; Chen, S.; Shen, W.; Luo, X.; Guo, L. Controlled synthesis of recyclable, porous FMO/C@TiO2 core-shell nanofibers with high adsorption and photocatalysis properties for the efficient treatment of dye wastewater. ChemPlusChem 2016, 81, 282–291. [Google Scholar] [CrossRef]
- Talebzadeh, F.; Zandipak, R.; Sobhanardakani, S. CeO2 nanoparticles supported on CuFe2O4 nanofibers as novel adsorbent for removal of Pb(II), Ni(II), and V(V) ions from petrochemical wastewater. Desalin. Water Treat. 2016, 57, 28363–28377. [Google Scholar] [CrossRef]
- Mondal, K. Recent advances in the synthesis of metal oxide nanofibers and their environmental remediation applications. Inventions 2017, 2, 1–29. [Google Scholar] [CrossRef]
- Vadukumpully, S.; Basheer, C.; Jeng, C.S.; Valiyaveettil, S. Carbon nanofibers extracted from soot as a sorbent for the determination of aromatic amines from wastewater effluent samples. J. Chromatogr. A 2011, 1218, 3581–3587. [Google Scholar] [CrossRef] [PubMed]
- Qi, F.; Qian, L.; Liu, J.; Li, X.; Lu, L.; Xu, Q. A high-throughput nanofibers mat-based micro-solid phase extraction for the determination of cationic dyes in wastewater. J. Chromatogr. A 2016, 1460, 24–32. [Google Scholar] [CrossRef] [PubMed]
- Cai, Z.; Song, X.; Zhang, Q.; Zhai, T. Electrospun polyindole nanofibers as a nano-adsorbent for heavy metal ions adsorption for wastewater treatment. Fibers Polym. 2017, 18, 502–513. [Google Scholar] [CrossRef]
- Chen, L.F.; Liang, H.W.; Lu, Y.; Cui, C.H.; Yu, S.H. Synthesis of an attapulgite Clay@Carbon nanocomposite adsorbent by a hydrothermal carbonization process and their application in the removal of toxic metal ions from water. Langmuir 2011, 27, 8998–9004. [Google Scholar] [CrossRef] [PubMed]
- Deng, J.-H.; Zhang, X.-R.; Zeng, G.-M.; Gong, J.-L.; Niu, Q.-Y.; Liang, J. Simultaneous removal of Cd(II) and ionic dyes from aqueous solution using magnetic graphene oxide nanocomposite as an adsorbent. Chem. Eng. J. 2013, 226, 189–200. [Google Scholar] [CrossRef]
- Badruddoza, A.Z.; Shawon, Z.B.; Tay, W.J.; Hidajat, K.; Uddin, M.S. Fe3O4/cyclodextrin polymer nanocomposites for selective heavy metals removal from industrial wastewater. Carbohydr. Polym. 2013, 91, 322–332. [Google Scholar] [CrossRef] [PubMed]
- Jiang, L.; Liu, P.; Zhao, S. Magnetic ATP/FA/Poly(AA-co-AM) ternary nanocomposite microgel as selective adsorbent for removal of heavy metals from wastewater. Colloids Surf. A Physicochem. Eng. Asp. 2015, 470, 31–38. [Google Scholar] [CrossRef]
- Tran, H.V.; Tran, L.D.; Nguyen, T.N. Preparation of chitosan/magnetite composite beads and their application for removal of Pb(II) and Ni(II) from aqueous solution. Mater. Sci. Eng. C 2010, 30, 304–310. [Google Scholar] [CrossRef]
- Kharissova, O.V.; Dias, H.V.R.; Kharisov, B.I. Magnetic adsorbents based on micro- and nano-structured materials. RSC Adv. 2015, 5, 6695–6719. [Google Scholar] [CrossRef]
- Morillo, D.; Faccini, M.; Amantia, D.; Pérez, G.; García, M.A.; Valiente, M.; Aubouy, L. Superparamagnetic iron oxide nanoparticle-loaded polyacrylonitrile nanofibers with enhanced arsenate removal performance. Environ. Sci. Nano 2016, 3, 1165–1173. [Google Scholar] [CrossRef]
- Gupta, A.K.; Deva, D.; Sharma, A.; Nishith, V. Fe-grown carbon nanofibers for removal of arsenic (V) in wastewater. Ind. Eng. Chem. Res. 2010, 49, 7074–7084. [Google Scholar]
- Li, S.; Yue, X.; Jing, Y.; Bai, S.; Dai, Z. Fabrication of zonal thiol-functionalized silica nanofibers for removal of heavy metal ions from wastewater. Colloids Surf. A Physicochem. Eng. Asp. 2011, 380, 229–233. [Google Scholar] [CrossRef]
- Faccini, M.; Shabafrooz, V.; Borja, G.; Vazquez-Campos, S.; Amantia, D. High flux and bendable carbon nanofiber membranes for the filtration of nanoparticles from aqueous solutions. Procedia Eng. 2012, 44, 1788–1789. [Google Scholar] [CrossRef]
- Sehaqui, H.; Liu, P.; Pfenninger, N.; Zimmermann, T.; Perez de Larraya, U.; Mathew, A.P.; Tingaut, P. Enhancing adsorption of heavy metal ions onto biobased nanofibers from waste pulp residues for application in wastewater treatment. Cellulose 2014, 21, 2831–2844. [Google Scholar] [CrossRef]
- Faccini, M.; Borja, G.; Boerrigter, M.; Martín, D.M.; Crespiera, S.M.; Vázquez-Campos, S.; Aubouy, L.; Amantia, D. Electrospun carbon nanofiber membranes for filtration of nanoparticles from water. J. Nanomater. 2015, 2015, 2. [Google Scholar] [CrossRef]
- Li, S.; Jia, Z.; Li, Z.; Li, Y.; Zhu, R. Synthesis and characterization of mesoporous carbon nanofibers and its adsorption for dye in wastewater. Adv. Powder Technol. 2016, 27, 591–598. [Google Scholar] [CrossRef]
- Angammana, C.J.; Jayaram, S.H. Fundamentals of electrospinning and processing technologies. Part. Sci. Technol. 2015, 34, 72–82. [Google Scholar] [CrossRef]
- Lu, W.; Sun, J.; Jiang, X. Recent advances in electrospinning technology and biomedical applications of electrospun fibers. J. Mater. Chem. B 2014, 2, 2369–2380. [Google Scholar] [CrossRef]
- Luo, C.J.; Stoyanov, S.D.; Stride, E.; Pelan, E.; Edirisinghe, M. Electrospinning versus fibre production methods: From specifics to technological convergence. Chem Soc. Rev. 2012, 41, 4708–4735. [Google Scholar] [CrossRef] [PubMed]
- Dastbaz, A.; Keshtkar, A.R. Adsorption of Th4+, U6+, Cd2+, and Ni2+ from aqueous solution by a novel modified polyacrylonitrile composite nanofiber adsorbent prepared by electrospinning. Appl. Surf. Sci. 2014, 293, 336–344. [Google Scholar] [CrossRef]
- Celebioglu, A.; Demirci, S.; Uyar, T. Cyclodextrin-grafted electrospun cellulose acetate nanofibers via “click” reaction for removal of phenanthrene. Appl. Surf. Sci. 2014, 305, 581–588. [Google Scholar] [CrossRef]
- Si, Y.; Ren, T.; Li, Y.; Ding, B.; Yu, J. Fabrication of magnetic polybenzoxazine-based carbon nanofibers with Fe3O4 inclusions with a hierarchical porous structure for water treatment. Carbon 2012, 50, 5176–5185. [Google Scholar] [CrossRef]
- Matsumoto, H.; Tanioka, A. Functionality in electrospun nanofibrous membranes based on fiber’s size, surface area, and molecular orientation. Membranes 2011, 1, 249–264. [Google Scholar] [CrossRef] [PubMed]
- Feng, C.; Khulbe, K.C.; Matsuura, T.; Tabe, S.; Ismail, A.F. Preparation and characterization of electro-spun nanofiber membranes and their possible applications in water treatment. Sep. Purif. Technol. 2013, 102, 118–135. [Google Scholar] [CrossRef]
- Sousa, A.F.; Vilela, C.; Matos, M.; Freire, C.S.R.; Silvestre, A.J.D.; Coelho, J.F.J. Polyethylene Terephthalate: Copolyesters, Composites, and Renewable Alternatives; Elsevier: Amsterdam, The Netherlands, 2015; pp. 113–141. [Google Scholar]
- Coskun, R.; Soykan, C. Lead (II) adsorption from aqueous solution by poly(ethylene terephthalate)-g-acrylamide fibers. J. Polym. Res. 2005, 13, 1–8. [Google Scholar] [CrossRef]
- Temoçin, Z.; Yiğitoğlu, M. Studies on selective uptake behavior of Hg(II) and Pb(II) by functionalized poly(ethylene terephthalate) fiber with 4-vinyl pyridine/2-hydroxyethylmethacrylate. Water Air Soil Pollut. 2009, 210, 463–472. [Google Scholar] [CrossRef]
- Ahmad Panahi, H.; Abdouss, M.; Ghiabi, F.; Moniri, E.; Mousavi Shoushtari, A. Modification and characterization of poly (ethylene terephthalate)-grafted-acrylic acid/acryl amide fiber for removal of lead from human plasma and environmental samples. J. Appl. Polym. Sci. 2012, 124, 5236–5246. [Google Scholar] [CrossRef]
- Niu, Y.; Ying, D.; Li, K.; Wang, Y.; Jia, J. Adsorption of heavy-metal ions from aqueous solution onto chitosan-modified polyethylene terephthalate (PET). Res. Chem. Intermediat. 2017, 43, 4213–4225. [Google Scholar] [CrossRef]
- Meng, Y.; Wang, J.N.; Xu, L.; Li, A.M. Fast removal of Pb2+ from water using new chelating fiber modified with acylamino and amino groups. Chin. Chem. Lett. 2012, 23, 496–499. [Google Scholar] [CrossRef]
- Kaiser, E.; Colescott, R.L.; Bossinger, C.D.; Cook, P.I. Color test for detection in the solid-phase of free terminal synthesis amino of peptides groups. Anal. Biochem. 1970, 34, 595–598. [Google Scholar] [CrossRef]
- Cengiz, F.; Jirsak, O. The effect of salt on the roller electrospinning of polyurethane nanofibers. Fibers Polym. 2009, 10, 177–184. [Google Scholar] [CrossRef]
- Zong, X.; Kim, K.; Fang, D.; Ran, S.; Hsiao, B.S.; Chu, B. Structure and process relationship of electrospun bioabsorbable nanofiber membranes. Polymer 2002, 43, 4403–4412. [Google Scholar] [CrossRef]
- Zhang, C.; Yuan, X.; Wu, L.; Han, Y.; Sheng, J. Study on morphology of electrospun poly (vinyl alcohol) mats. Eur. Polym. J. 2005, 41, 423–432. [Google Scholar] [CrossRef]
- Zeronian, S.H.; Collins, M.J. Surface modification of polyester by alkaline treatments. Text. Prog. 1989, 20, 1–26. [Google Scholar] [CrossRef]
- Kardas, I.; Lipp-Symonowicz, B.; Sztajnowski, S. Comparison of the effect of PET fibres’ surface modification using enzymes and chemical substances with respect to changes in mechanical properties. Fibres Text. East. Eur. 2009, 17, 93–97. [Google Scholar]
- Perez Holmberg, J. Competitive Adsorption and Displacement Behaviour of Heavy Metals on Peat. Master’s Thesis, Chalmers University of Technology, Göteborg, Sweden, 2006. [Google Scholar]

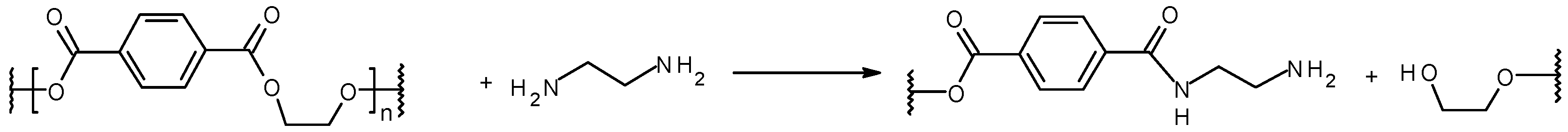

| Sample | PET Concentration (wt %) | TBAB Concentration (wt %) | Feed Rate (mL/h) | Voltage (kv) | Distance (mm) |
|---|---|---|---|---|---|
| PET-1 | 20 | 0.1 | 1.2 | 25 | 220 |
| PET-2 | 15 | 0.1 | 1.0 | 28 | 180 |
| PET- 3 | 15 | 0.2 | 0.5 | 30 | 150 |
| Sample | PET (wt %) | TBAB (wt %) | Viscosity (cP) | Conductivity (µS/cm) | Fiber Diameter (nm) |
|---|---|---|---|---|---|
| PET-1 | 20 | 0.1 | 298.5 | 10.7 | 3000 ± 500 |
| PET-2 | 15 | 0.1 | 170.4 | 46.8 | 724 ± 125 |
| PET-3 | 15 | 0.2 | 174.5 | 126.2 | 488 ± 61 |
| Sample | EDA (wt %) | Time (min) | % N | % C | % H | % O | mg EDA/g |
|---|---|---|---|---|---|---|---|
| PET Blank | — | — | 0.45 | 61.17 | 2.95 | 35.43 | — |
| APET-1 | 10 | 60 | 0.92 | 60.84 | 3.25 | 34.99 | 0.02 |
| APET-2 | 15 | 5.51 | 56.21 | 3.23 | 35.05 | 0.12 | |
| APET-3 | 5 | 60 | 0.90 | 60.68 | 3.35 | 35.08 | 0.02 |
| APET-4 | 15 | 6.73 | 56.28 | 4.53 | 32.46 | 0.14 | |
| APET-5 | 2.5 | 60 | 0.86 | 61.63 | 2.73 | 34.78 | 0.02 |
| APET-6 | 15 | 4.97 | 57.69 | 4.34 | 33.00 | 0.11 |
| Pb(II)–Cd(II)/Ni(II)/Cu(II) Molar Ratio | q (Pb(II); mmol/g) | q (Ni(II); mmol/g) | q (Cu(II); mmol/g) | q (Cd(II); mmol/g) | Decrease of q (Pb(II); %) |
|---|---|---|---|---|---|
| 1:0 | 1.78 | — | — | — | — |
| 1:1 | 1.17 | 0.11 | 0.45 | 0.28 | 34.27 |
| 1:2 | 0.74 | 8.26 | 0.65 | 1.62 | 58.43 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Morillo Martín, D.; Magdi Ahmed, M.; Rodríguez, M.; García, M.A.; Faccini, M. Aminated Polyethylene Terephthalate (PET) Nanofibers for the Selective Removal of Pb(II) from Polluted Water. Materials 2017, 10, 1352. https://doi.org/10.3390/ma10121352
Morillo Martín D, Magdi Ahmed M, Rodríguez M, García MA, Faccini M. Aminated Polyethylene Terephthalate (PET) Nanofibers for the Selective Removal of Pb(II) from Polluted Water. Materials. 2017; 10(12):1352. https://doi.org/10.3390/ma10121352
Chicago/Turabian StyleMorillo Martín, Diego, Mohamed Magdi Ahmed, Mónica Rodríguez, María A. García, and Mirko Faccini. 2017. "Aminated Polyethylene Terephthalate (PET) Nanofibers for the Selective Removal of Pb(II) from Polluted Water" Materials 10, no. 12: 1352. https://doi.org/10.3390/ma10121352
APA StyleMorillo Martín, D., Magdi Ahmed, M., Rodríguez, M., García, M. A., & Faccini, M. (2017). Aminated Polyethylene Terephthalate (PET) Nanofibers for the Selective Removal of Pb(II) from Polluted Water. Materials, 10(12), 1352. https://doi.org/10.3390/ma10121352





