Influence of KMnO4 Concentrationon Infrared Emissivity of Coatings Formed on TC4 Alloys by Micro-Arc Oxidation
Abstract
:1. Introduction
2. Results and Discussion
2.1. Surface Morphology
2.2. Phase Composition
2.3. Infrared Emissivity
2.4. The Reaction Mechanism
- a
- Cathodic reaction:2H+ + 2e−→H2↑
- b
- Anodic reactions:4OH− − 4e−→O2↑ + 2H2OTi + 4OH− − 4e−→TiO2 + 2H2O2MnO4−→MnO42− + MnO2 + O2↑2SiO32− − 4e−→2SiO2 + O2↑2PO3− + 2OH−→P2O74− + H2O
3. Materials and Methods
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Li, Z.; Zheng, F.; Gong, H.Q.; Hu, P.F.; Song, S.L.; Zhen, Q. Study on ZrSiO4–aluminosilicate glass coating with high infrared emissivity and anti-oxidation properties. Compet. Comm. 2017, 4, 16–19. [Google Scholar] [CrossRef]
- Cheng, X.D.; Duan, W.; Chen, W.; Ye, W.P.; Mao, F.; Ye, F.; Zhang, Q. Infrared Radiation Coatings Fabricated by Plasma Spray. J. Therm. Spray Technol. 2009, 18, 448–450. [Google Scholar] [CrossRef]
- Guo, J.C.; Guo, X.W.; Xu, W.B.; Zhang, Z.C.; Dong, C.; Peng, L.M.; Ding, W.J. A Zn-Ni coating with both high electrical conductivity and infrared emissivity prepared by hydrogen evolution method. Appl. Surf. Sci. 2017, 402, 92–98. [Google Scholar] [CrossRef]
- Brodu, E.; Balat-Pichelin, M.; Sans, J.L.; Freeman, M.D.; Kasper, J.C. Efficiency and behavior of textured high emissivity metallic coatings at high temperature. Mater. Des. 2015, 83, 85–94. [Google Scholar] [CrossRef]
- Hong, Z.L.; Ouyang, J.H.; Liu, Z.G.; Wang, Y.M. Microstructure, thermal shock resistance and thermal emissivity of plasma sprayed LaMAl11O19 (M = Mg, Fe) coatings for metallic thermal protection systems. Appl. Surf. Sci. 2013, 271, 52–59. [Google Scholar] [CrossRef]
- Huang, Z.B.; Zhou, W.C.; Tang, X.F.; Zhu, D.M.; Luo, F. Effects of substrate roughness on infrared-emissivity characteristics of Au films deposited on Ni alloy. Thin Solid Films 2011, 519, 3100–3106. [Google Scholar] [CrossRef]
- Zhen, Y.L.; Li, L.Y.; Deng, T.G.; Ding, Y.B.; Pan, L.; Zhao, J.P.; Li, Y. Magnetron sputtering SiC films on nickel photonic crystals with high emissivity for high temperature applications. Appl. Surf. Sci. 2012, 259, 811–815. [Google Scholar] [CrossRef]
- Huang, J.P.; Li, Y.B.; Song, G.P.; Zhang, X.J.; Sun, Y.; He, X.D.; Du, S.Y. Highly enhanced infrared spectral emissivity of porous CeO2 coating. Mater.Lett. 2012, 85, 57–60. [Google Scholar] [CrossRef]
- Sah, S.P.; Tsuji, E.; Aoki, Y.; Habazaki, H. Cathodic pulse breakdown of anodic films on aluminium in alkaline silicate electrolyte–understanding the role of cathodic half-cycle in AC plasma electrolytic oxidation. Corros. Sci. 2012, 55, 90–96. [Google Scholar] [CrossRef]
- Tang, H.; Xin, T.Z.; Sun, Q.; Yi, H.G.; Jiang, Z.H.; Wang, F.P. Influence of FeSO4 concentration on thermal emissivity of coatings formed on titanium alloy by micro-arc oxidation. Appl. Surf. Sci. 2011, 257, 10839–10844. [Google Scholar] [CrossRef]
- Xu, Y.J.; Yao, Z.P.; Jia, F.Z.; Wang, Y.L.; Jiang, Z.H.; Bu, H.T. Preparation of PEO ceramic coating on Ti alloy and its high temperature oxidation resistance. Curr. Appl. Phys. 2010, 10, 698–702. [Google Scholar] [CrossRef]
- Li, Q.B.; Yang, W.B.; Liu, C.C.; Wang, D.A.; Liang, J. Correlations between the growth mechanism and properties of micro-arc oxidation coatings on titanium alloy: Effects of electrolytes. Surf. Coat. Technol. 2017, 316, 162–170. [Google Scholar] [CrossRef]
- Li, Q.B.; Liang, J.; Liu, B.X.; Peng, Z.J.; Wang, Q. Effects of cathodic voltages on structure and wear resistance of plasma electrolytic oxidation coatings formed on aluminium alloy. Appl. Surf. Sci. 2014, 297, 176–181. [Google Scholar] [CrossRef]
- Ma, H.J.; Li, D.L.; Liu, C.; Huang, Z.Q.; He, D.L.; Yan, Q.; Liu, P.; Nash, P.; Shen, D.J. An investigation of (NaPO3)6 effects and mechanisms during micro-arc oxidation of AZ31 magnesium alloy. Surf. Coat. Technol. 2015, 266, 151–159. [Google Scholar] [CrossRef]
- Li, J.; Cai, H.; Xue, X.N.; Jiang, B.L. The outward–inward growth behavior of microarc oxidation coatings in phosphate and silicate solution. Mater. Lett. 2010, 64, 2102–2104. [Google Scholar] [CrossRef]
- Zhang, R.F.; Qiao, L.P.; Qu, B.; Zhang, S.F.; Chang, W.H.; Xiang, J.H. Biocompatibility of micro-arc oxidation coatings developed on Ti6Al4V alloy in a solution containing organic phosphate. Mater. Lett. 2015, 153, 77–80. [Google Scholar] [CrossRef]
- Shokouhfar, M.; Dehghanian, C.; Montazeri, M.; Baradaran, A. Preparation of ceramic coating on Ti substrate by plasma electrolytic oxidation in different electrolytes and evaluation of its corrosion resistance: Part II. Appl. Surf. Sci. 2012, 258, 2416–2423. [Google Scholar] [CrossRef]
- Shokouhfar, M.; Allahkaram, S.R. Formation mechanism and surface characterization of ceramic composite coatings on pure titanium prepared by micro-arc oxidation in electrolytes containing nanoparticles. Surf. Coat. Technol. 2016, 291, 396–405. [Google Scholar] [CrossRef]
- Chu, H.J.; Liang, C.J.; Chen, C.H.; He, J.L. Optical emission spectroscopic determination of the optimum regions for micro-arc oxidation of titanium. Surf. Coat. Technol. 2017, 325, 166–173. [Google Scholar] [CrossRef]
- Si, P.Z.; Li, D.; Lee, J.W.; Choi, C.J.; Zhang, Z.D.; Geng, D.Y.; Bruck, E. Unconventional exchange bias in oxide-coated manganese nanoparticles. Appl. Phys. Lett. 2005, 87, 1413–1415. [Google Scholar] [CrossRef]
- Shi, C.J.; Daun, K.J.; Wells, M.A. Spectral emissivity characteristics of the Usibor® 1500P steel during austenitization in argon and air atmospheres. Int. J. Heat Mass Transf. 2015, 91, 818–828. [Google Scholar] [CrossRef]
- Raman, R.; Thakur, A. Emissivity of homogeneous thin films. Thin Solid Films 1982, 87, 101–112. [Google Scholar] [CrossRef]
- Wu, Y.B.; Ma, X.F.; Zhang, H.Z.; Zhou, Y. A new high emissivity coating on Ni-Based superalloysubstrate. Rare Met. Mater. Eng. 2016, 45, 588–592. [Google Scholar] [CrossRef]
- Lan, L.C.; Zhao, W.R.; Deng, L.L.; Fang, X.B.; Zhong, J.M. Preparation and properties of MnO2-Cr2O3-NiO system infrared radiant materials. J. Synth. Cryst. 2013, 42, 2369–2374. [Google Scholar] [CrossRef]
- Cheng, X.D.; Min, J.; Zhu, Z.Q.; Ye, W.P. Preparation of high emissivity NiCr2O4 powders with a spinel structure by spray drying. Int. J. Min. Metall. Mater. 2012, 19, 173–178. [Google Scholar] [CrossRef]
- Chen, X.B.; Liu, L.; Yu, P.Y.; Mao, S.S. Increasing solar absorption for photocatalysis with black hydrogenated titanium dioxide nanocrystals. Science 2011, 331, 746–750. [Google Scholar] [CrossRef] [PubMed]
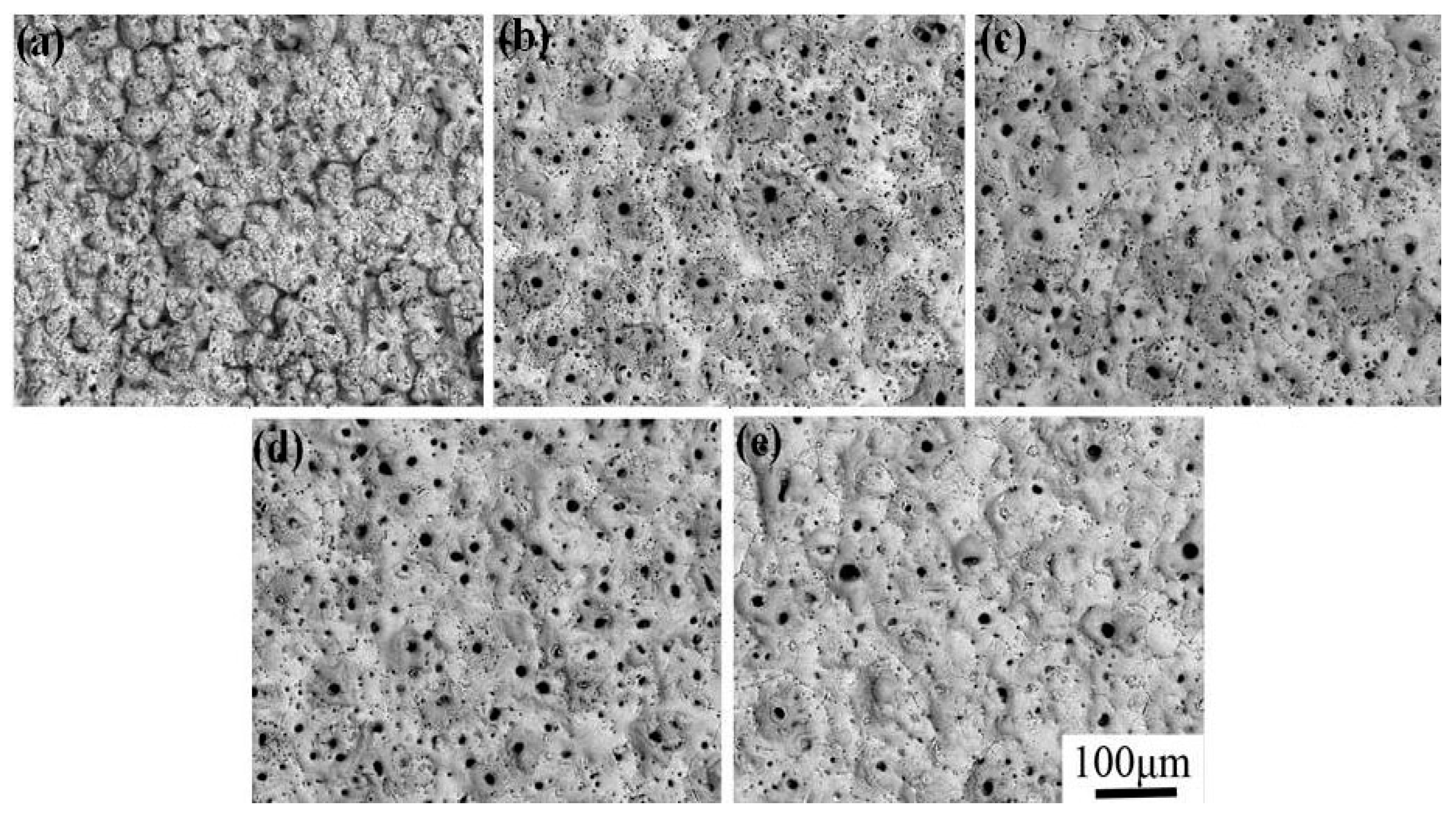

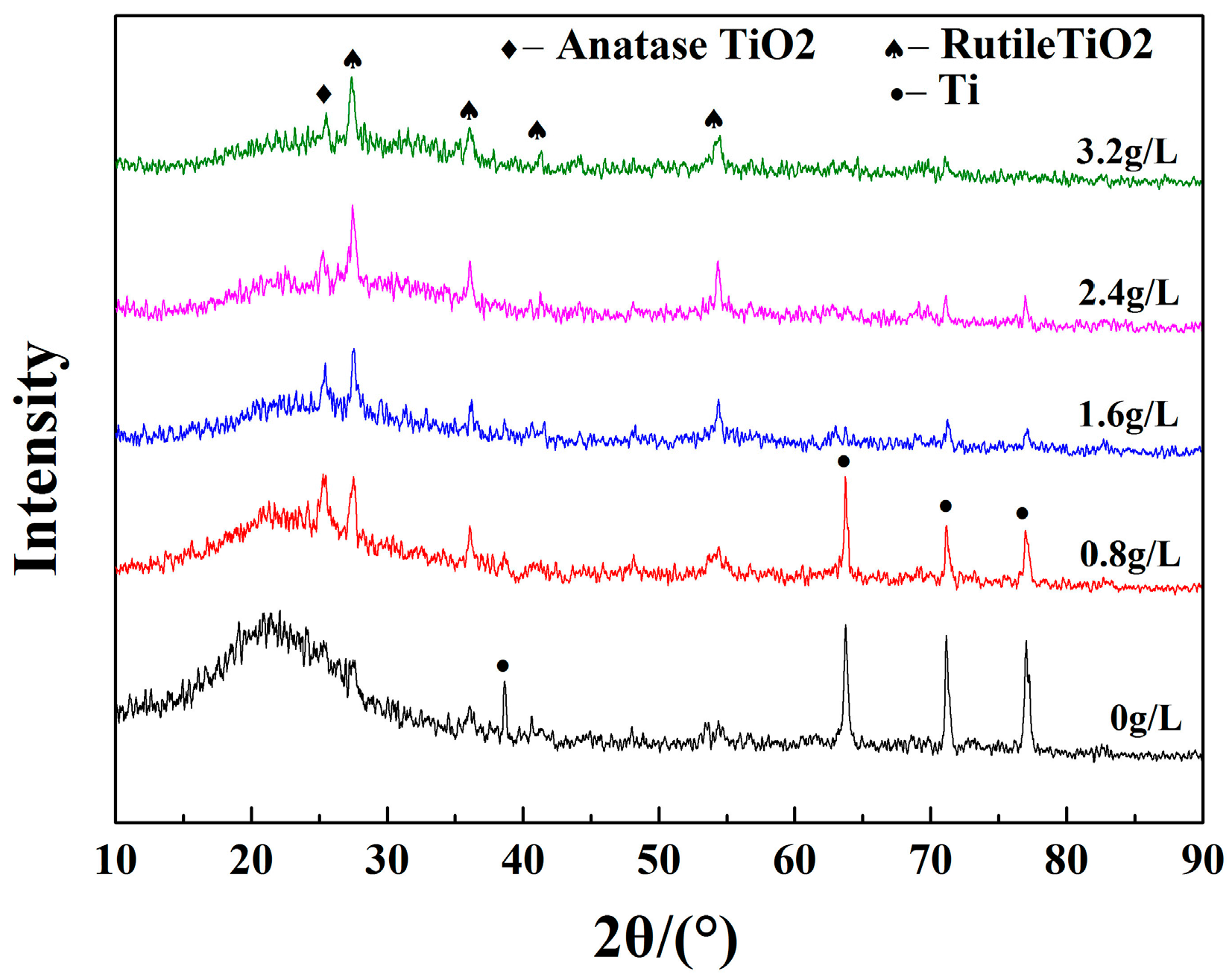


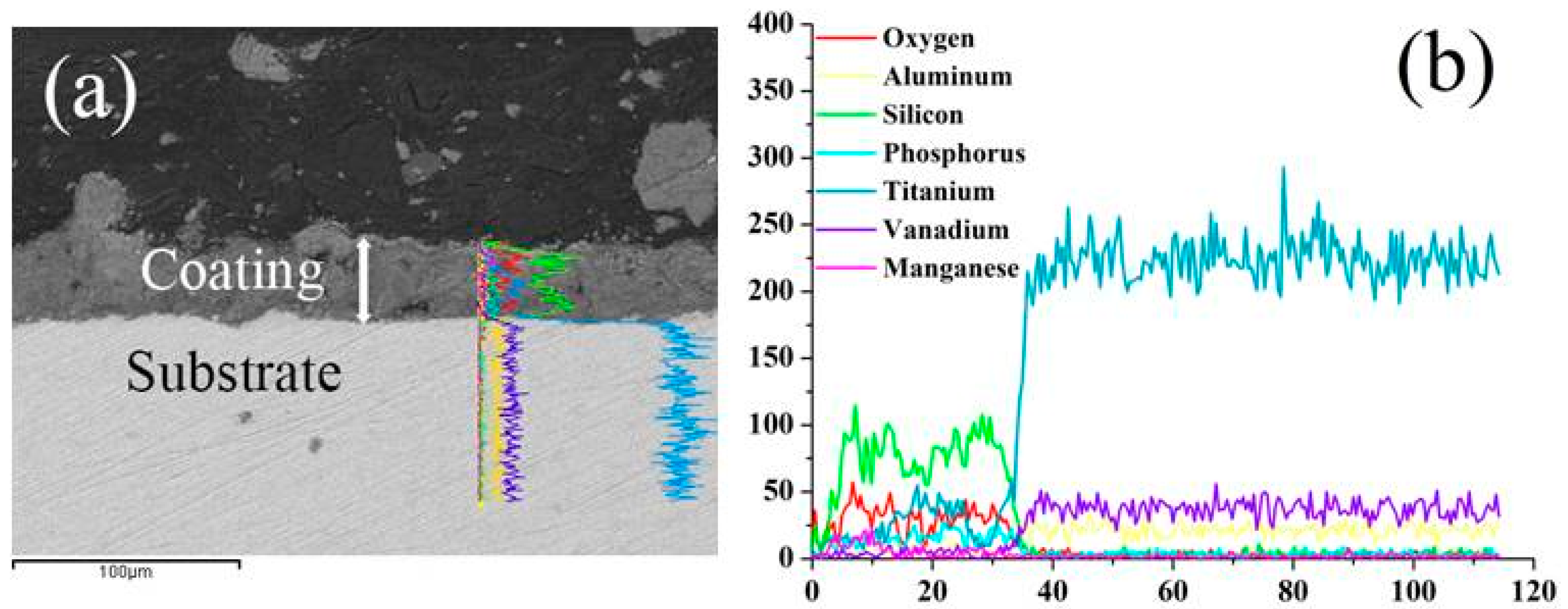
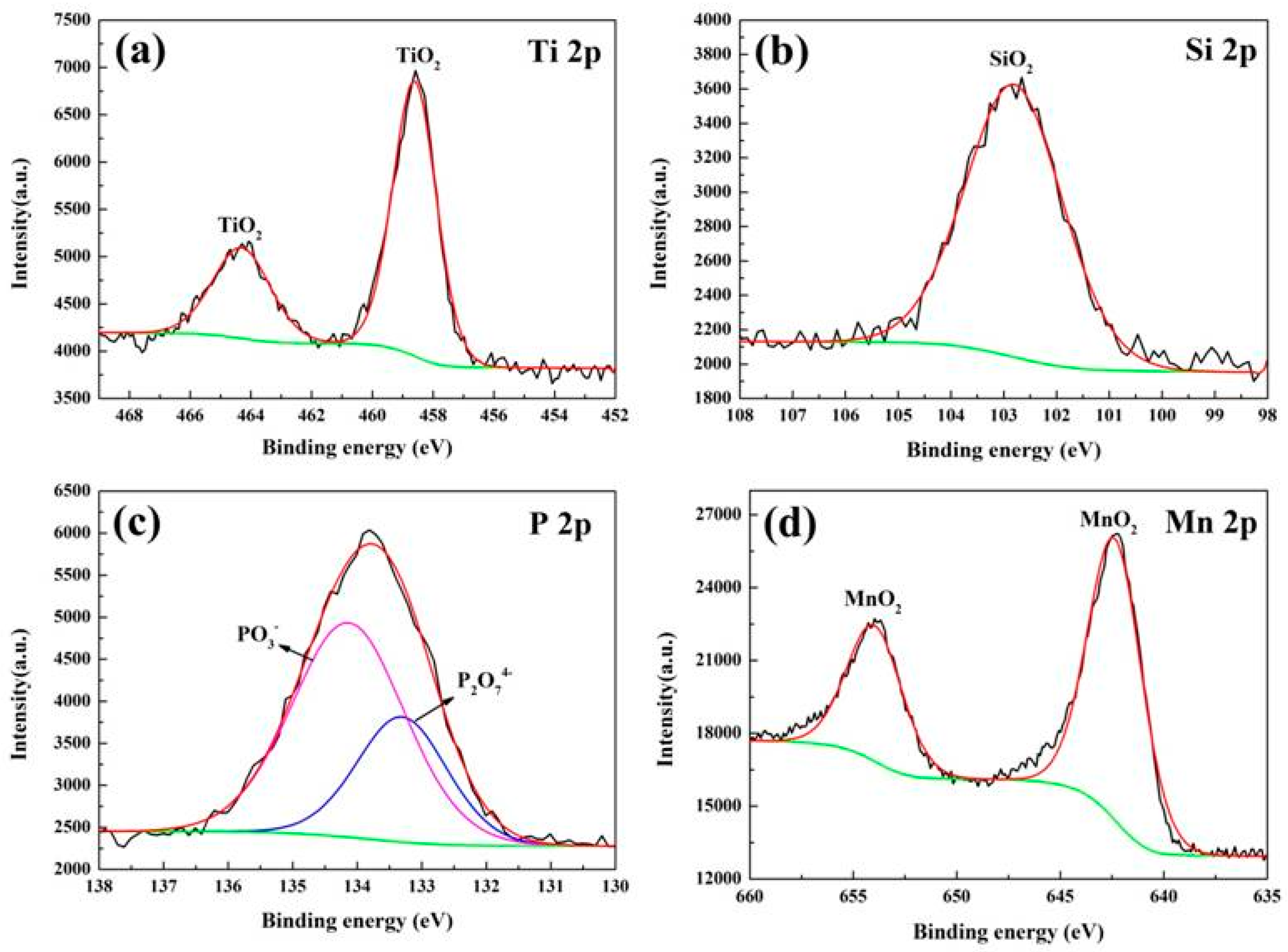

| KMnO4 Concentration (g/L) | 0 | 0.8 | 1.6 | 2.4 | 3.2 |
|---|---|---|---|---|---|
| Thickness (μm) | 35 | 33 | 40 | 41 | 46 |
| standard deviation | 0.415 | 0.621 | 0.543 | 0.572 | 0.608 |
| Roughness (μm) | 6.07 | 2.66 | 2.92 | 2.96 | 3.80 |
| standard deviation | 0.711 | 0.459 | 0.674 | 0.703 | 0.569 |
| KMnO4 Concetration (g/L) | Element Content of MAO Coatings (at.%) | ||||||
|---|---|---|---|---|---|---|---|
| O | Na | Al | Si | P | Ti | Mn | |
| 0 | 71.75 | 1.59 | 0.34 | 21.48 | 1.84 | 3.01 | / |
| 0.8 | 70.54 | 2.28 | 0.65 | 13.28 | 4.45 | 4.64 | 4.16 |
| 1.6 | 68.92 | 3.31 | 0.54 | 11.98 | 4.79 | 4.06 | 6.41 |
| 2.4 | 67.80 | 2.96 | 0.49 | 11.42 | 4.55 | 4.34 | 8.44 |
| 3.2 | 67.14 | 3.26 | 0.62 | 11.14 | 4.14 | 4.48 | 9.32 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gao, G.; Li, Y.; Li, C.; Hu, D.; Li, Z.; Xi, Z. Influence of KMnO4 Concentrationon Infrared Emissivity of Coatings Formed on TC4 Alloys by Micro-Arc Oxidation. Materials 2017, 10, 1301. https://doi.org/10.3390/ma10111301
Gao G, Li Y, Li C, Hu D, Li Z, Xi Z. Influence of KMnO4 Concentrationon Infrared Emissivity of Coatings Formed on TC4 Alloys by Micro-Arc Oxidation. Materials. 2017; 10(11):1301. https://doi.org/10.3390/ma10111301
Chicago/Turabian StyleGao, Guangrui, Ying Li, Chaozhong Li, Dan Hu, Zhengxian Li, and Zhengping Xi. 2017. "Influence of KMnO4 Concentrationon Infrared Emissivity of Coatings Formed on TC4 Alloys by Micro-Arc Oxidation" Materials 10, no. 11: 1301. https://doi.org/10.3390/ma10111301
APA StyleGao, G., Li, Y., Li, C., Hu, D., Li, Z., & Xi, Z. (2017). Influence of KMnO4 Concentrationon Infrared Emissivity of Coatings Formed on TC4 Alloys by Micro-Arc Oxidation. Materials, 10(11), 1301. https://doi.org/10.3390/ma10111301





