Reduced Graphene Oxide on Nickel Foam for Supercapacitor Electrodes
Abstract
:1. Introduction
2. Results and Discussion
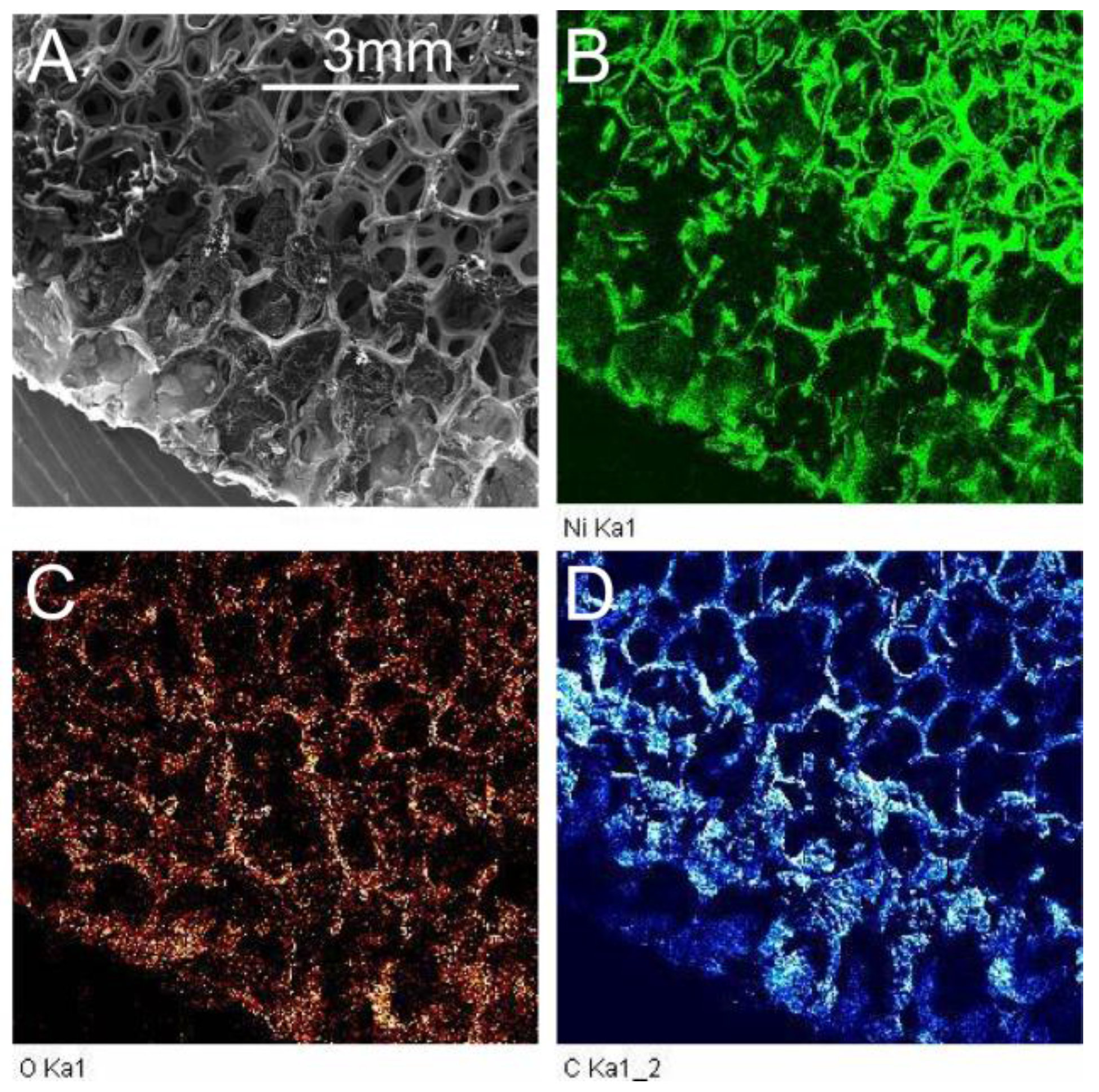
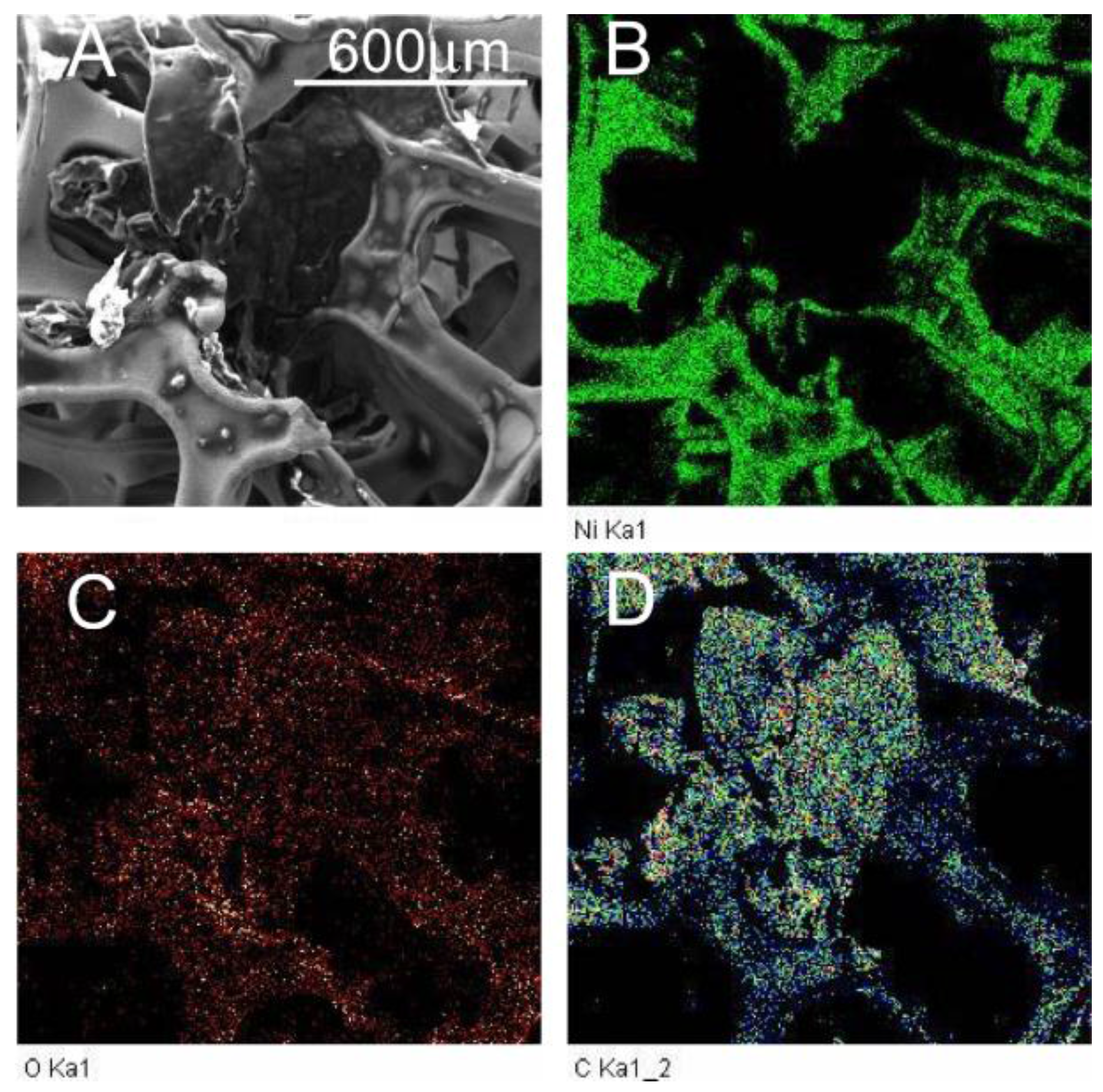
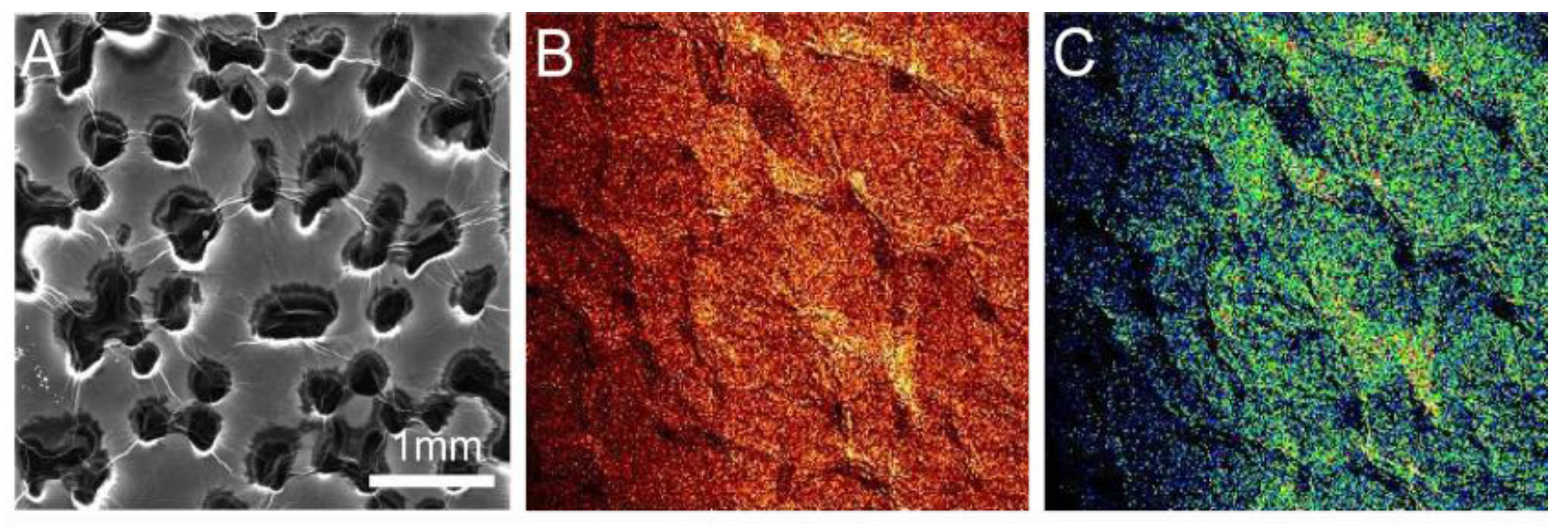
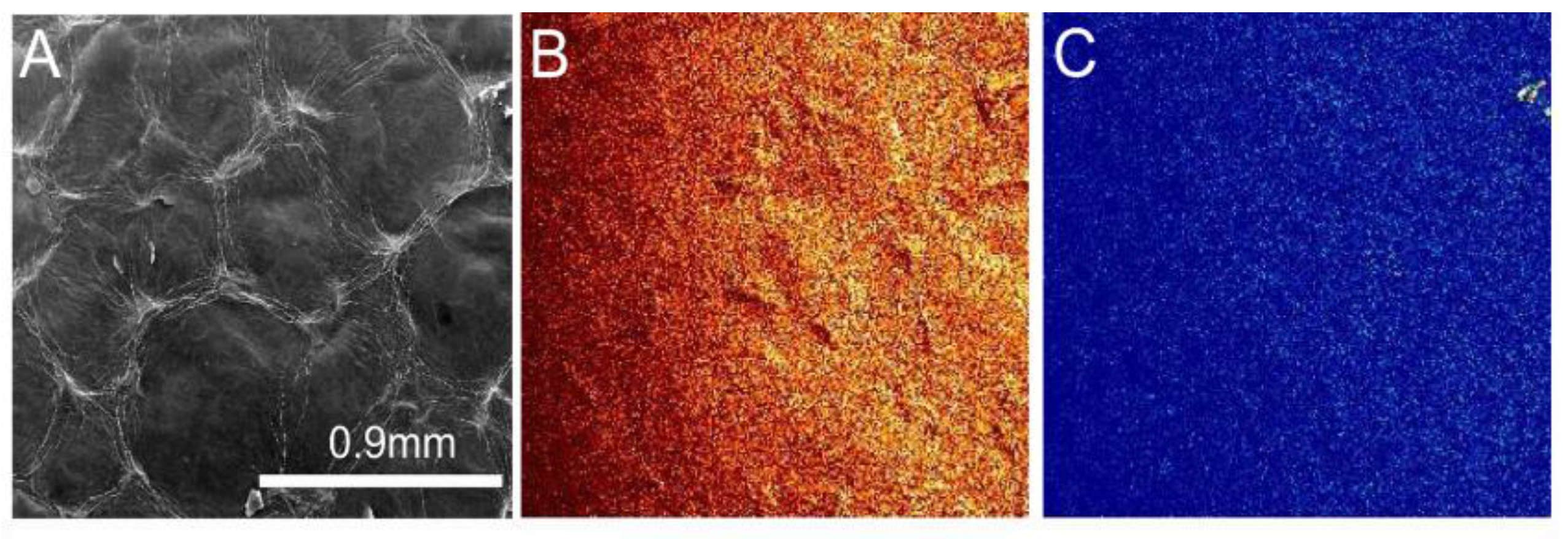
2.1. Environmental Scanning Electron Microscopy (ESEM) Results
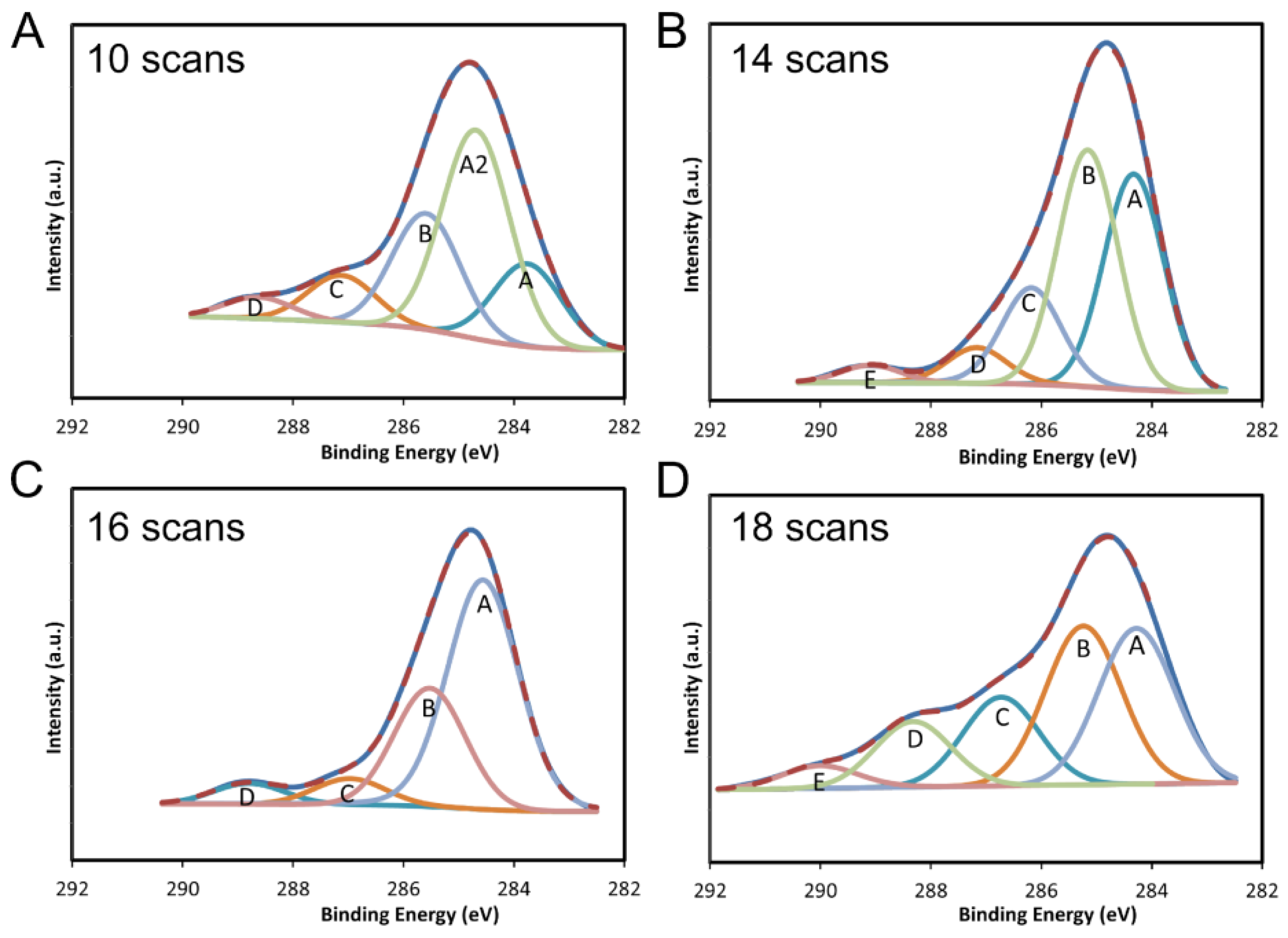
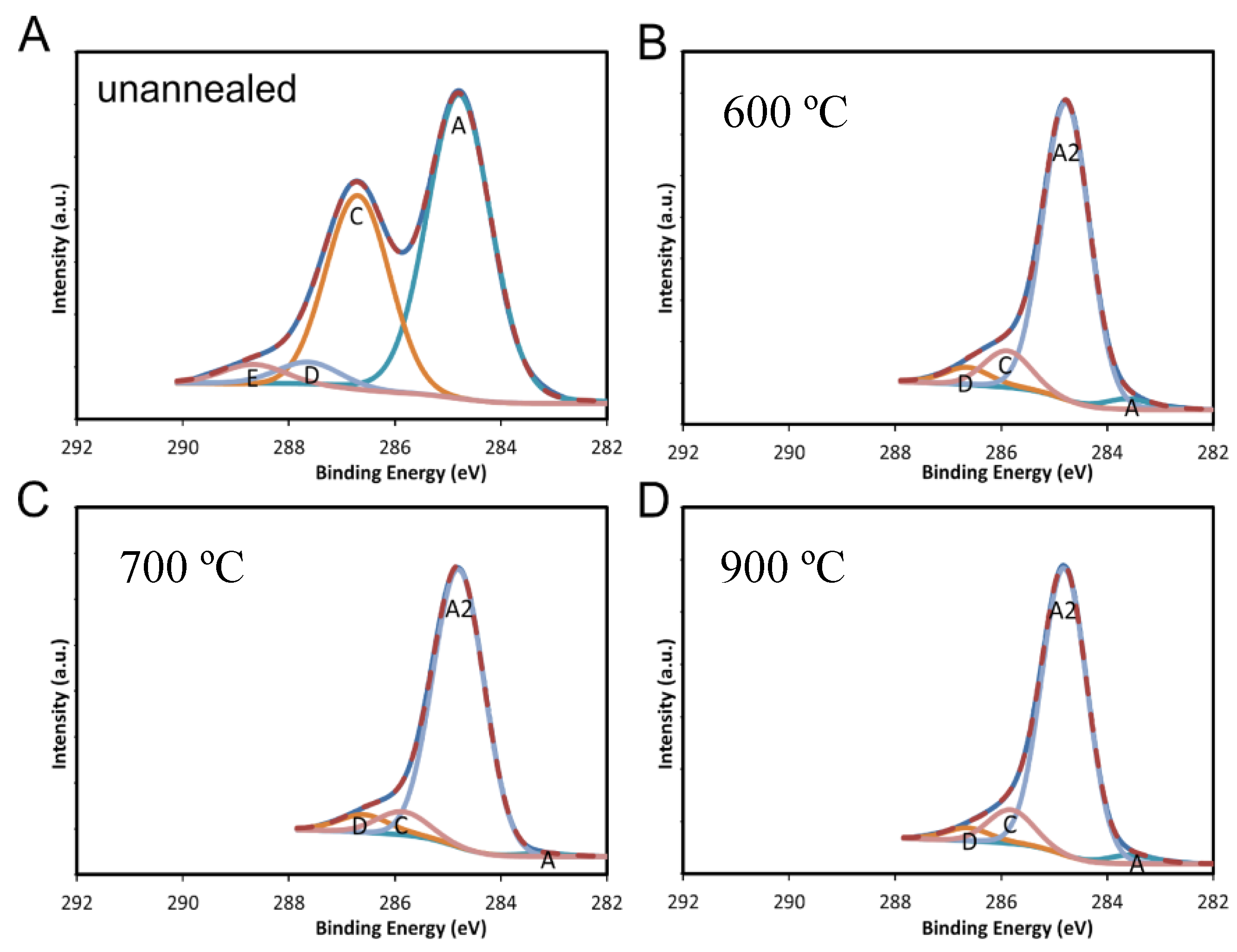
2.2. X-ray Photoelectron Spectroscopy (XPS) Results
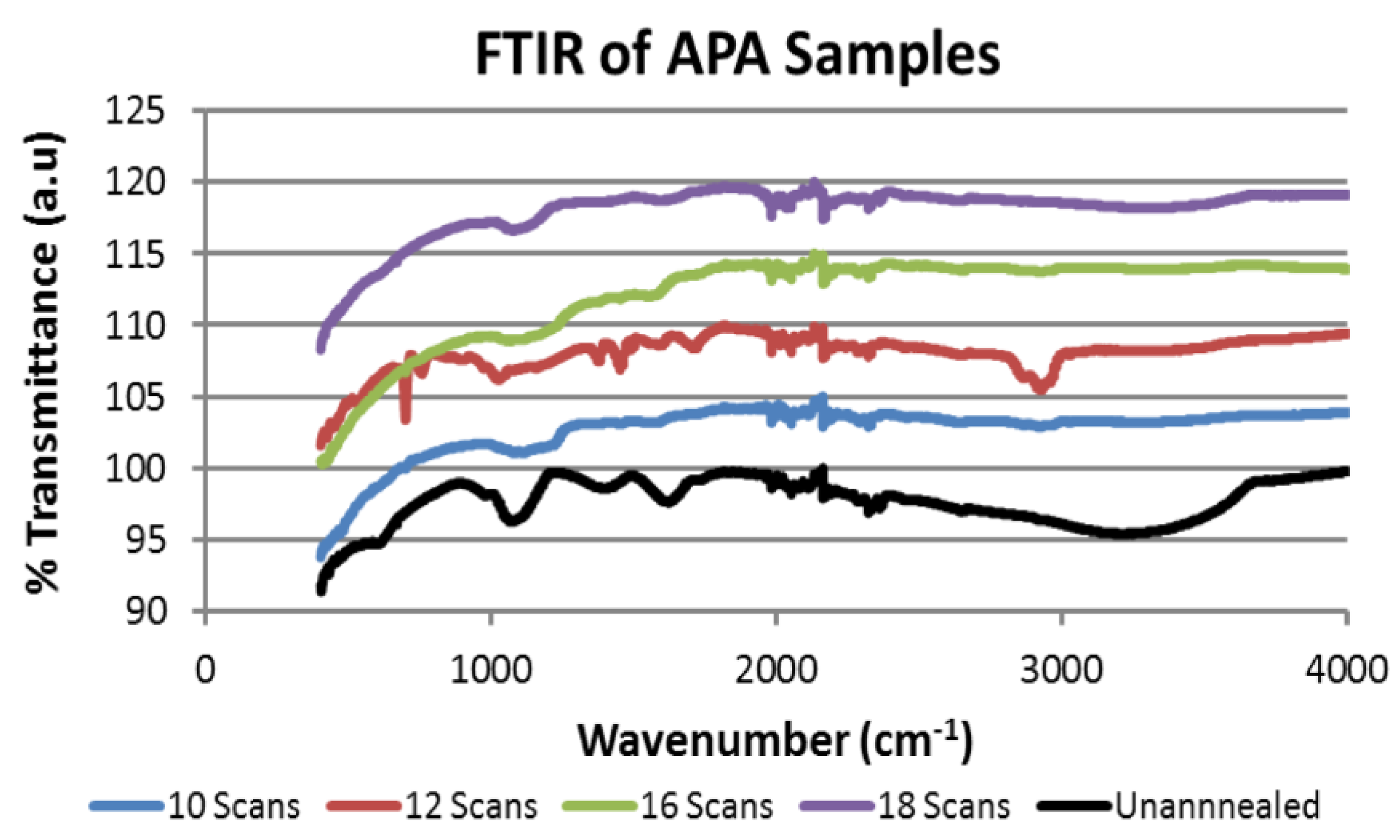
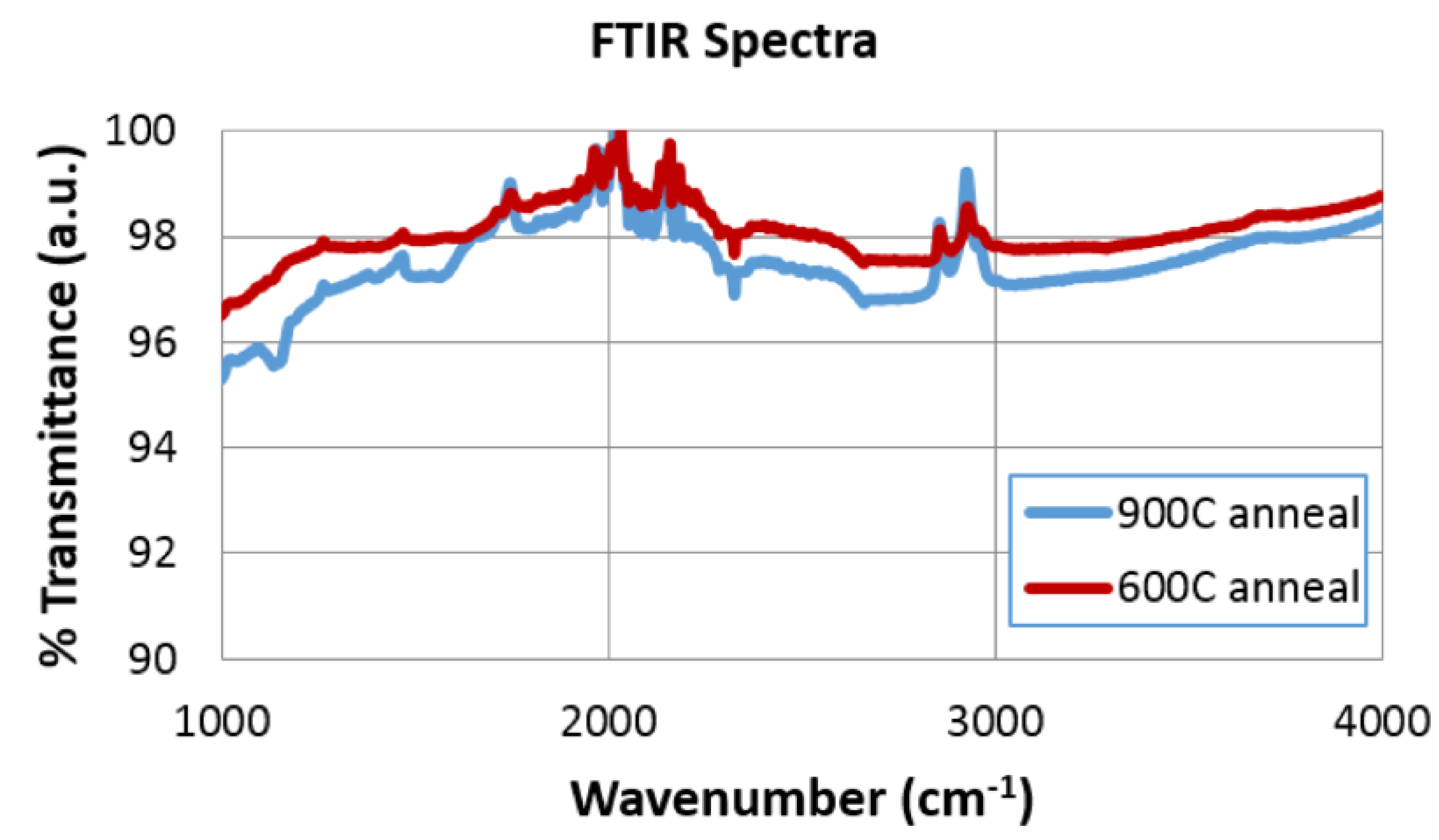
2.3. Fourier Transform Infrared Spectroscopy (FTIR) Results
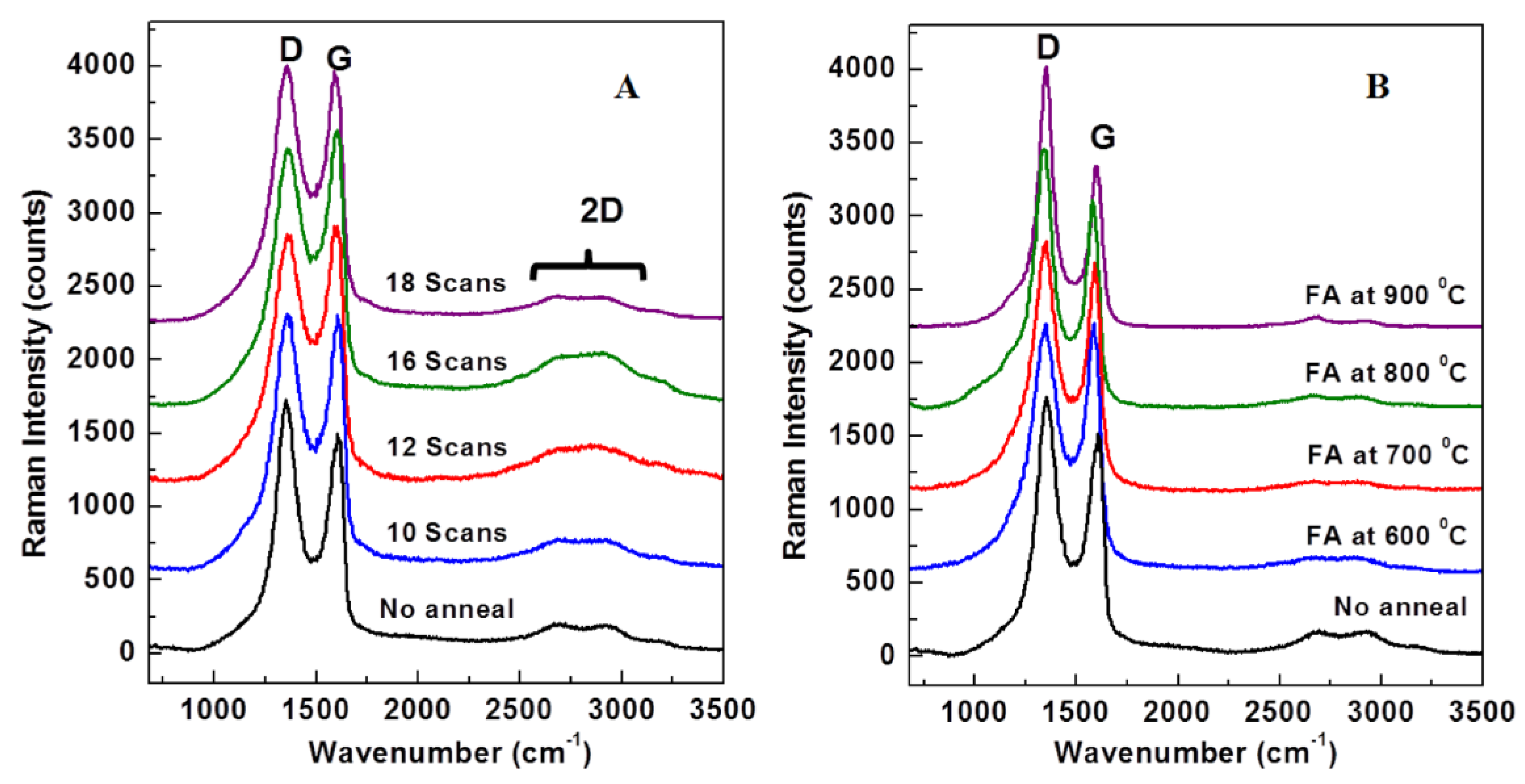
2.4. Raman Spectroscopy Results from APA Samples
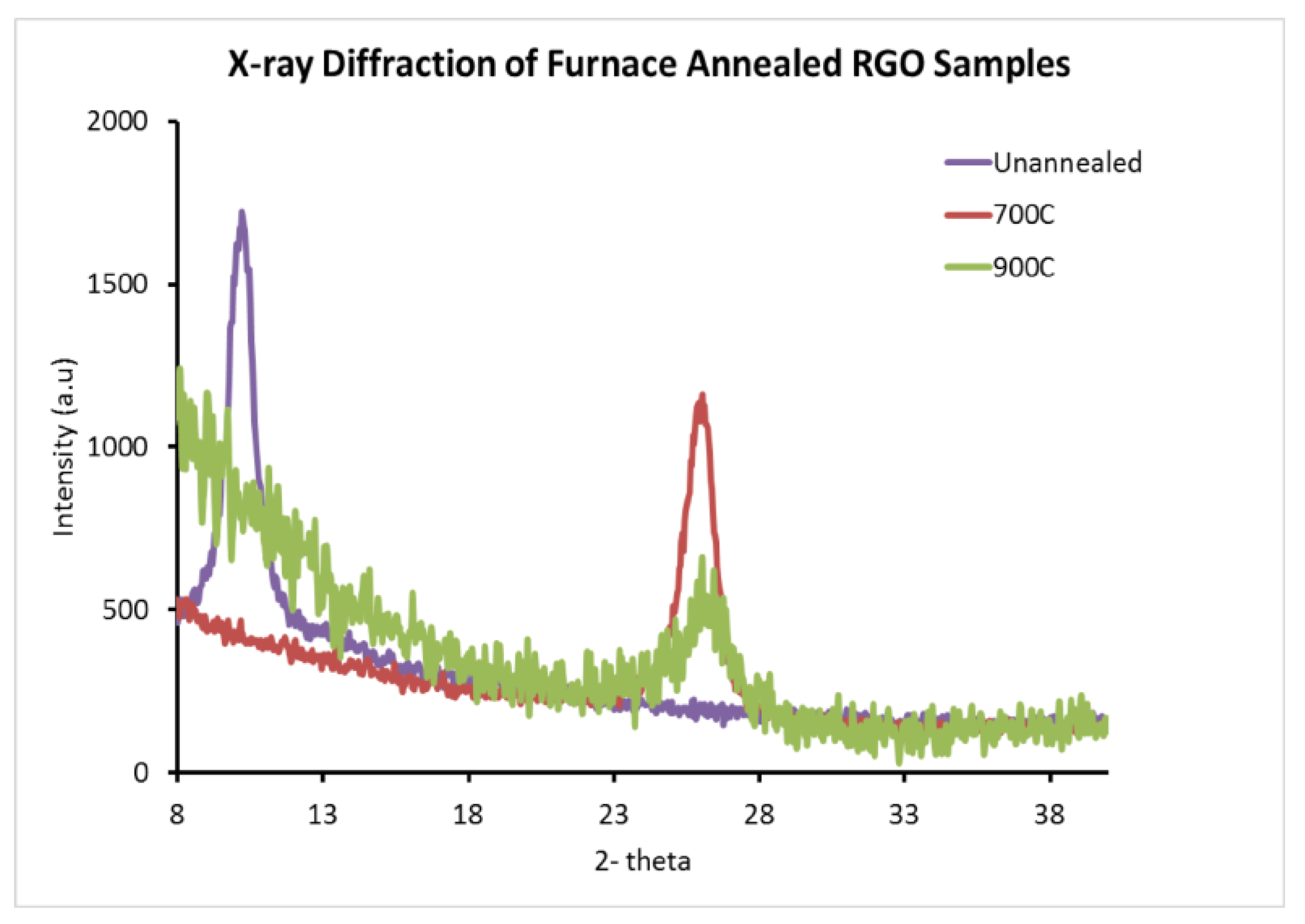
2.5. X-ray Diffraction (XRD) Results
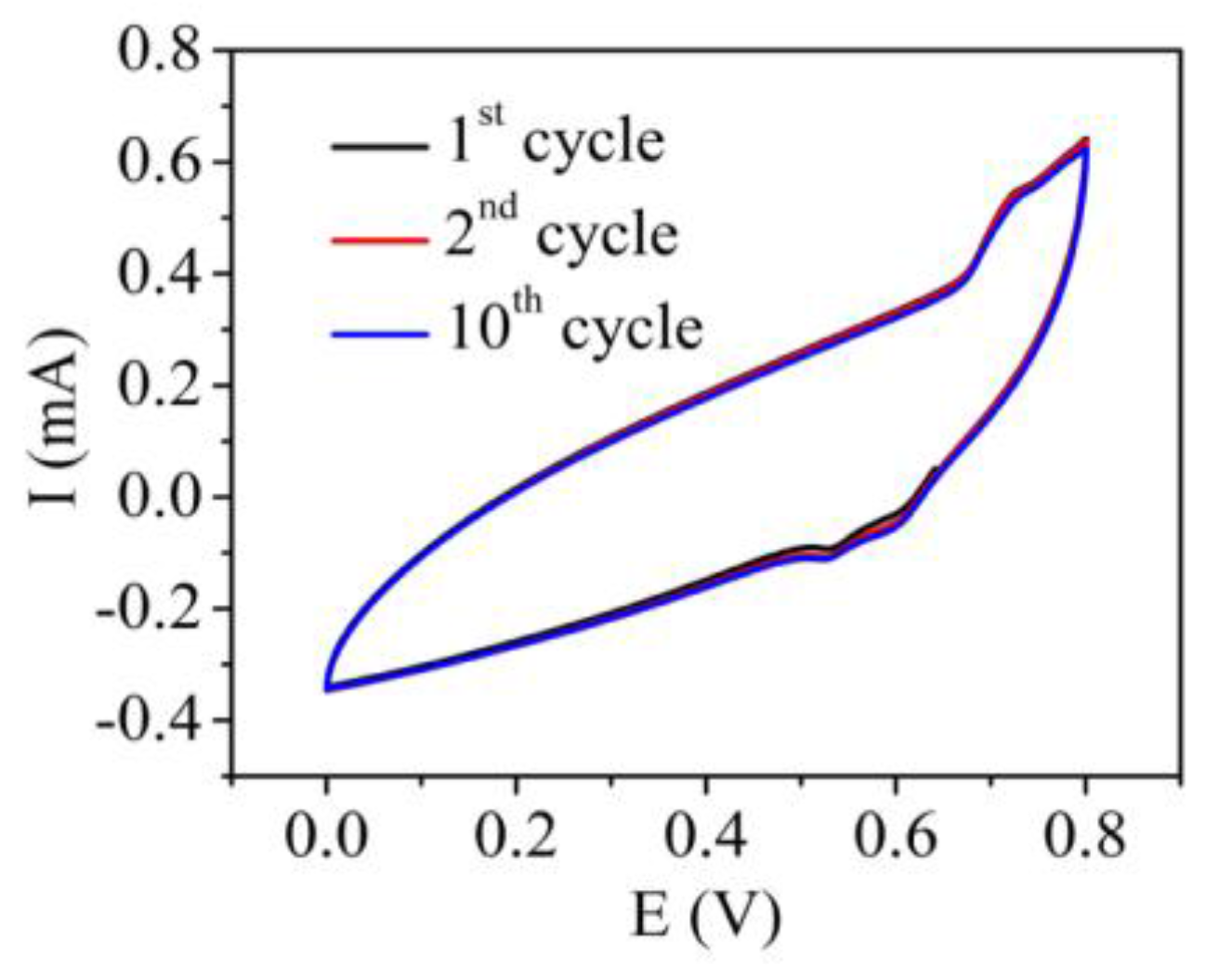
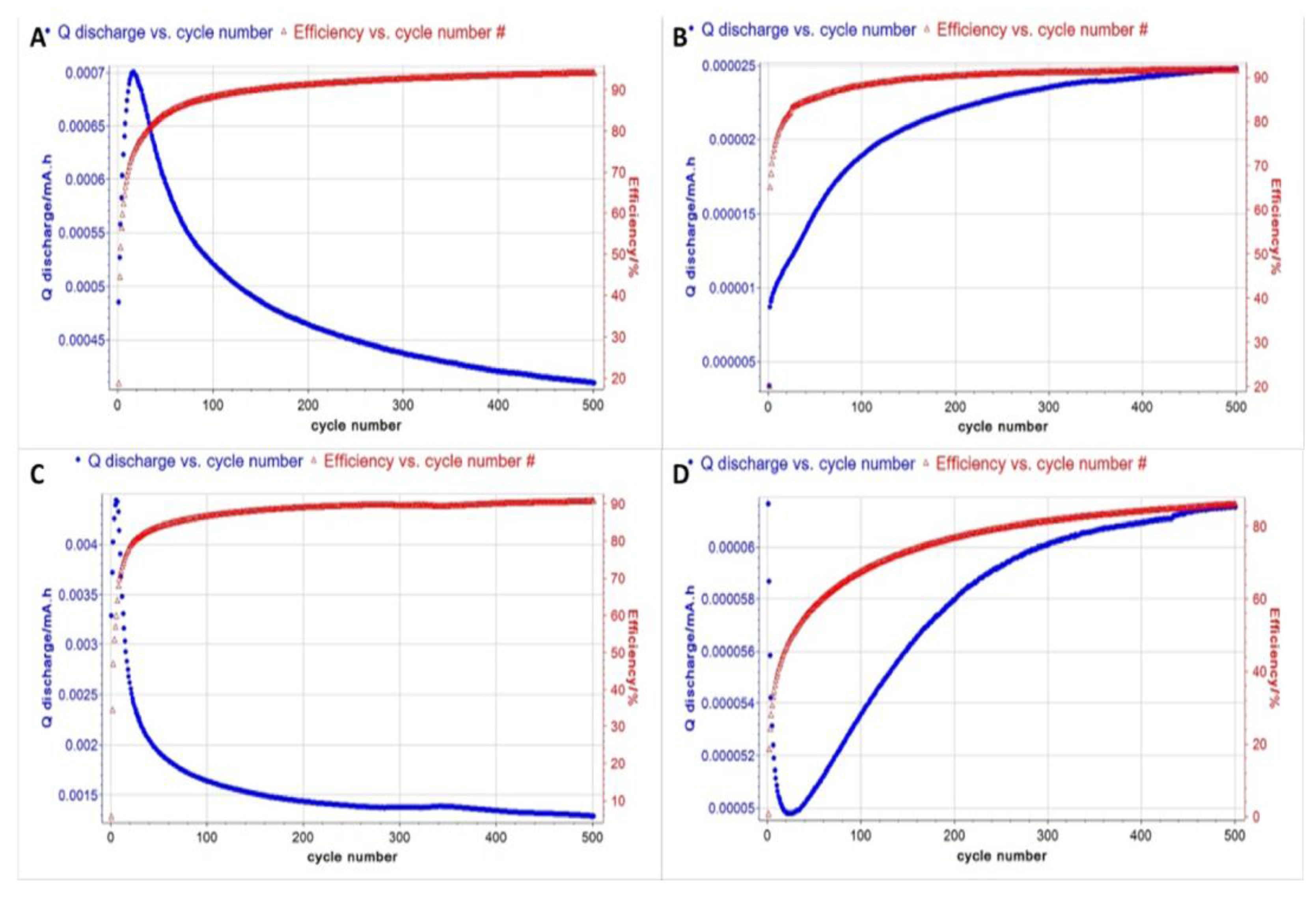
2.6. Capacitance Measurements
3. Materials and Methods
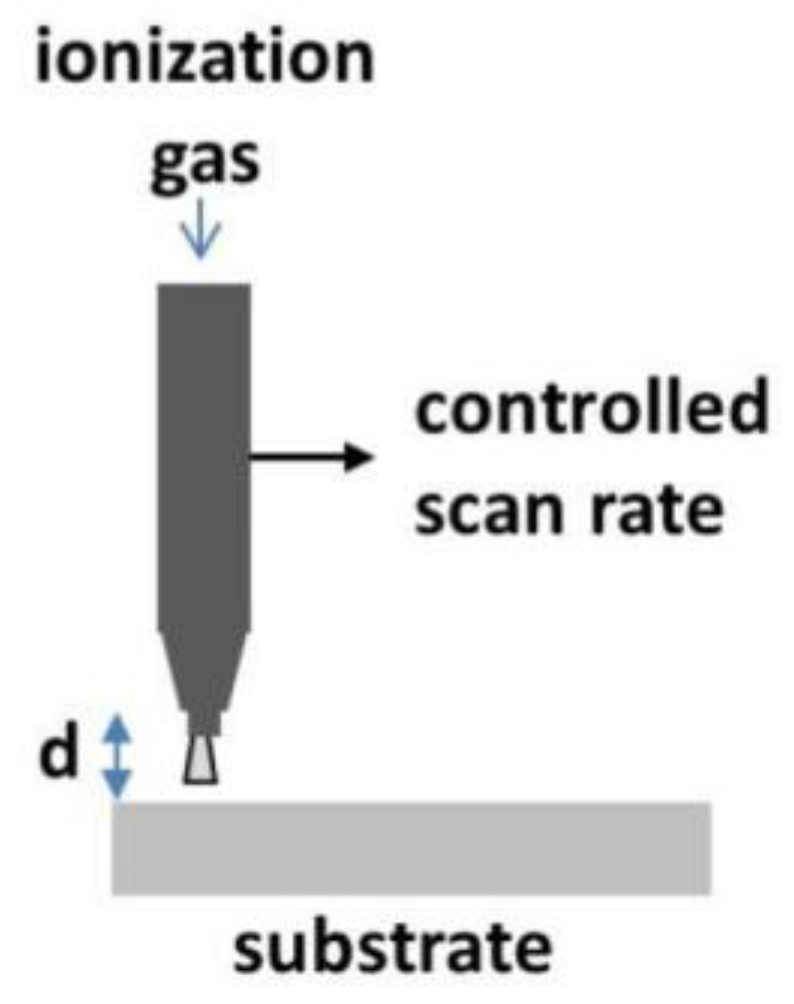
3.1. Atmospheric Plasma Annealing
3.2. Furnace Annealing
3.3. Sample Characterization
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Conte, M.; Genovese, A.; Ortenzi, F.; Vellucci, F. Hybrid battery-supercapacitor storage for an electric forklift: A life-cycle cost assessment. J. Appl. Electrochem. 2014, 44, 523–532. [Google Scholar] [CrossRef]
- Hong, S.Y.; Kim, Y.; Park, Y.; Choi, A.; Choi, N.-S.; Lee, K.T. Charge carriers in rechargeable batteries: Na ions vs. Li ions. Energy Environ. Sci. 2013, 6, 2067–2081. [Google Scholar] [CrossRef]
- Zhang, J.; Jiang, J.; Li, H.; Zhao, X.S. A high-performance asymmetric supercapacitor fabricated with graphene-based electrodes. Energy Environ. Sci. 2011, 4, 4009–4015. [Google Scholar] [CrossRef]
- Zhang, S.; Xiong, R.; Zhou, X. Comparison of the topologies for a hybrid energy-storage system of electric vehicles via a novel optimization method. Sci. China Technol. Sci. 2015, 58, 1173–1185. [Google Scholar] [CrossRef]
- Simon, P.; Gogotsi, Y. Materials for electrochemical capacitors. Nat. Mater. 2008, 7, 845–854. [Google Scholar] [CrossRef] [PubMed]
- López-Naranjo, E.J.; González-Ortiz, L.J.; Apátiga, L.M.; Rivera-Muñoz, E.M.; Manzano-Ramírez, A. Transparent Electrodes: A Review of the Use of Carbon-Based Nanomaterials. J. Nanomater. 2016, 2016. [Google Scholar] [CrossRef]
- Tung, V.C.; Chen, L.-M.; Allen, M.J.; Wassei, J.K.; Nelson, K.; Kaner, R.B.; Yang, Y. Low-temperature solution processing of graphene-carbon nanotube hybrid materials for high-performance transparent conductors. Nano Lett. 2009, 9, 1949–1955. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.; Dai, L. Carbon nanomaterials for high-performance supercapacitors. Mater. Today 2013, 16, 272–280. [Google Scholar] [CrossRef]
- Signorelli, R.; Ku, D.C.; Kassakian, J.G.; Schindall, J.E. Electrochemical double-layer capacitors using carbon nanotube electrode structures. Proc. IEEE 2009, 97, 1837–1847. [Google Scholar] [CrossRef]
- Pan, H.; Li, J.; Feng, Y.P. Carbon nanotubes for supercapacitor. Nanoscale Res. Lett. 2010, 5, 654–668. [Google Scholar] [CrossRef] [PubMed]
- Yi, M.; Shen, Z. A review on mechanical exfoliation for the scalable production of graphene. J. Mater. Chem. A 2015, 3, 11700–11715. [Google Scholar] [CrossRef]
- Zhao, L.; Rim, K.T.; Zhou, H.; He, R.; Heinz, T.F.; Pinczuk, A.; Flynn, G.W.; Pasupathy, A.N. Influence of copper crystal surface on the CVD growth of large area monolayer graphene. Solid State Commun. 2011, 151, 509–513. [Google Scholar] [CrossRef]
- Mohan, V.B.; Brown, R.; Jayaraman, K.; Bhattacharyya, D. Characterisation of reduced graphene oxide: Effects of reduction variables on electrical conductivity. Mater. Sci. Eng. B Solid-State Mater. Adv. Technol. 2015, 193, 49–60. [Google Scholar] [CrossRef]
- Chen, Y.; Fu, K.; Zhu, S.; Luo, W.; Wang, Y.; Li, Y.; Hitz, E.; Yao, Y.; Dai, J.; Wan, J.; et al. Reduced graphene oxide films with ultrahigh conductivity as Li-ion battery current collectors. Nano Lett. 2016, 16, 3616–3623. [Google Scholar] [CrossRef] [PubMed]
- Gao, J. Free-standing reduced graphene oxide paper with high electrical conductivity. J. Electron. Mater. 2016, 45, 1290–1295. [Google Scholar] [CrossRef]
- Jin, S.; Gao, Q.; Zeng, X.; Zhang, R.; Liu, K.; Shao, X.; Jin, M. Effects of reduction methods on the structure and thermal conductivity of free-standing reduced graphene oxide films. Diam. Relat. Mater. 2015, 58, 54–61. [Google Scholar] [CrossRef]
- Stoller, M.D.; Park, S.; Yanwu, Z.; An, J.; Ruoff, R.S. Graphene-Based ultracapacitors. Nano Lett. 2008, 8, 3498–3502. [Google Scholar] [CrossRef] [PubMed]
- El-Kady, M.F.; Strong, V.; Dubin, S.; Kaner, R.B. Laser Scribing of High-Performance and Flexible Graphene-Based Electrochemical Capacitors. Science 2012, 335, 1326–1330. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.W.; Mattevi, C.; Chhowalla, M.; Sankaran, R.M. Plasma-assisted reduction of graphene oxide at low temperature and atmospheric pressure for flexible conductor applications. J. Phys. Chem. Lett. 2012, 3, 772–777. [Google Scholar] [CrossRef] [PubMed]
- Zheng, C.; Zhang, J.; Zhang, Q.; You, B.; Chen, G. Three dimensional Ni foam-supported graphene oxide for binder-free pseudocapacitor. Electrochim. Acta 2015, 152, 216–221. [Google Scholar] [CrossRef]
- Luan, V.H.; Tien, H.N.; Hoa, L.T.; Hien, N.T.M.; Oh, E.-S.; Chung, J.; Kim, E.J.; Choi, W.M.; Kong, B.-S.; Hur, S.H. Synthesis of a highly conductive and large surface area graphene oxide hydrogel and its use in a supercapacitor. J. Mater. Chem. A 2013, 1, 208–211. [Google Scholar] [CrossRef]
- Cao, X.; Shi, Y.; Shi, W.; Lu, G.; Huang, X.; Yan, Q.; Zhang, Q.; Zhang, H. Preparation of novel 3D graphene networks for supercapacitor applications. Small 2011, 7, 3163–3168. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.; Velamakanni, A.; Bozoklu, G.; Park, S.; Stoller, M.; Piner, R.D.; Stankovich, S.; Jung, I.; Field, D.A.; Ventrice, C.A.; et al. Chemical analysis of graphene oxide films after heat and chemical treatments by X-ray photoelectron and Micro-Raman spectroscopy. Carbon N. Y. 2009, 47, 145–152. [Google Scholar] [CrossRef]
- Yamada, Y.; Yasuda, H.; Murota, K.; Nakamura, M.; Sodesawa, T.; Sato, S. Analysis of heat-treated graphite oxide by X-ray photoelectron spectroscopy. J. Mater. Sci. 2013, 48, 8171–8198. [Google Scholar] [CrossRef]
- Cuong, T.V.; Pham, V.H.; Tran, Q.T.; Hahn, S.H.; Chung, J.S.; Shin, E.W.; Kim, E.J. Photoluminescence and Raman studies of graphene thin films prepared by reduction of graphene oxide. Mater. Lett. 2010, 64, 399–401. [Google Scholar] [CrossRef]
- Blanton, T.; Majumdar, D. Characterization of X-ray Irradiated graphene oxide coatings using X-ray diffraction, X-ray photoelectron spectroscopy, and atomic force microscopy. CPDS-Int. Centre Diffr. Data 2013, 28, 116–122. [Google Scholar] [CrossRef]
- Chen, J.H.; Li, W.Z.; Wang, D.Z.; Yang, S.X.; Wen, J.G.; Ren, Z.F. Electrochemical characterization of carbon nanotubes as electrode in electrochemical double-layer capacitors. Carbon N. Y. 2002, 40, 1193–1197. [Google Scholar] [CrossRef]
- An, K.H.; Kim, W.S.; Park, Y.S.; Moon, J.M.; Bae, D.J.; Lim, S.C.; Lee, Y.S.; Lee, Y.H. Electrochemical properties of high-power supercapacitors using single-walled carbon nanotube electrodes. Adv. Funtional Mater. 2001, 11, 387–392. [Google Scholar] [CrossRef]
- Sugimoto, W.; Iwata, H.; Yasunaga, Y.; Murakami, Y.; Takasu, Y. Preparation of ruthenic acid nanosheets and utilization of its interlayer surface for electrochemical energy storage. Angew. Chemie Int. Ed. 2003, 42, 4092–4096. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.-Q.; Zhao, D.-D.; Tang, P.-Y.; Wang, Y.-M.; Xu, C.-L.; Li, H.-L. MnO2/graphene/nickel foam composite as high performance supercapacitor electrode via a facile electrochemical deposition strategy. Mater. Lett. 2012, 76, 127–130. [Google Scholar] [CrossRef]
- De Giglio, E.; Cometa, S.; Satriano, C.; Sabbatini, L.; Zambonin, P.G. Electrosynthesis of hydrogel films on metal substrates for the development of coatings with tunable drug delivery performances. J. Biomed. Mater. Res. A 2009, 88, 1048–1057. [Google Scholar] [CrossRef] [PubMed]
- Du, D.; Li, P.; Ouyang, J. Nitrogen-doped reduced graphene oxide prepared by simultaneous thermal reduction and nitrogen doping of graphene oxide in air and its application as an electrocatalyst. ACS Appl. Mater. Interfaces 2015, 7, 26952–26958. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Wang, H.; Robinson, J.T.; Sanchez, H.; Diankov, G.; Dai, H. Simultaneous nitrogen doping and reduction of graphene oxide. J. Am. Chem. Soc. 2009, 131, 15939–15944. [Google Scholar] [CrossRef] [PubMed]


| APA Samples | Furnace Annealed (FA) Samples—Trial 1,2,3 | |||
|---|---|---|---|---|
| # Scans | Average C:O | Annealing Temperature | Average C:O | Standard Deviation of C:O |
| 10 | 0.9 | Not Annealed | 1.04 | 0.12 |
| 12 | 2.8 | 600 °C | 14.11 | 2.81 |
| 14 | 1.4 | 700 °C | 16.03 | 2.10 |
| 16 | 9 | 800 °C | 22.17 | 5.77 |
| 18 | 13.9 | 900 °C | 35.06 | 9.58 |
| Attribute | Binding Energy (eV) | % | Attribute | Binding Energy (eV) | % |
|---|---|---|---|---|---|
| 10 scans | 12 scans | ||||
| A: C–C | 283.98 | 17.38 | A: C–C | 284.34 | 35.85 |
| A2: C–C | 284.87 | 43.82 | B: C=C | 285.35 | 28.86 |
| B: C=C | 285.74 | 24.4 | C: C–O–C | 286.67 | 17.61 |
| C: C–O–C | 287.15 | 9.88 | D: C=O | 287.95 | 10.56 |
| D: C=O | 288.64 | 4.52 | E: O–C=O | 289.12 | 7.13 |
| 14 scans | 16 scans | ||||
| A: C–C | 284.35 | 35.42 | A: C–C | 284.56 | 59.24 |
| B: C=C | 285.16 | 38.92 | B: C=C | 285.53 | 29.17 |
| C: C–O–C | 286.18 | 16.62 | C: C–O–C | 286.97 | 6.45 |
| D: C=O | 287.7 | 6.15 | D: C=O | 288.77 | 5.14 |
| E: O–C=O | 289.09 | 2.88 | - | - | - |
| 18 scans | 20 scans | ||||
| A: C–C | 284.28 | 31.61 | A: C–C | 284.62 | 60.76 |
| B: C=C | 285.24 | 32.22 | B: C=C | 285.22 | 32.79 |
| C: C–O–C | 286.73 | 18.01 | C: C–O–C | 286.64 | 6.45 |
| D: C=O | 288.33 | 13.32 | - | - | - |
| E: O–C=O | 290.01 | 4.84 | - | - | - |
| Temperature (°C) | Avg. % °C | Uncertainty % | Avg. % O | Uncertainty % | Avg C/O | Uncertainty % |
|---|---|---|---|---|---|---|
| Unannealed | 69.6 | 1.2 | 29.3 | 0.8 | 2.4 | 0.1 |
| 600 | 93.2 | 0.2 | 6.3 | 0.3 | 14.8 | 0.7 |
| 700 | 90.4 | 1.9 | 8.8 | 1.7 | 10.3 | 2.3 |
| 800 | 85.6 | 1.0 | 13.1 | 1.0 | 6.5 | 0.6 |
| 900 | 84.9 | 0.9 | 13.1 | 0.8 | 6.5 | 0.5 |
| Temperature (°C) | Capacitance (F/g) |
|---|---|
| Unannealed | 0.45 |
| 600 | 33 |
| 700 | 2.8 |
| 800 | 1.9 |
| 900 | 0.6 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ramabadran, U.; Ryan, G.; Zhou, X.; Farhat, S.; Manciu, F.; Tong, Y.; Ayler, R.; Garner, G. Reduced Graphene Oxide on Nickel Foam for Supercapacitor Electrodes. Materials 2017, 10, 1295. https://doi.org/10.3390/ma10111295
Ramabadran U, Ryan G, Zhou X, Farhat S, Manciu F, Tong Y, Ayler R, Garner G. Reduced Graphene Oxide on Nickel Foam for Supercapacitor Electrodes. Materials. 2017; 10(11):1295. https://doi.org/10.3390/ma10111295
Chicago/Turabian StyleRamabadran, Uma, Gillian Ryan, Xuan Zhou, Susan Farhat, Felicia Manciu, Yigang Tong, Ryan Ayler, and Graham Garner. 2017. "Reduced Graphene Oxide on Nickel Foam for Supercapacitor Electrodes" Materials 10, no. 11: 1295. https://doi.org/10.3390/ma10111295
APA StyleRamabadran, U., Ryan, G., Zhou, X., Farhat, S., Manciu, F., Tong, Y., Ayler, R., & Garner, G. (2017). Reduced Graphene Oxide on Nickel Foam for Supercapacitor Electrodes. Materials, 10(11), 1295. https://doi.org/10.3390/ma10111295




