1. Introduction
AC-LED is a new type of device that can be directly driven by the alternating current of city power. It has experienced rapid development and is widely applied in indoor and outdoor lighting due to its long life, low price, high efficiency, etc. [
1,
2,
3,
4].However, the vital defect of the AC-LED is the flickering effect, with a time gap of 5–20 ms [
4,
5,
6], resulting in visual fatigue and health hazards. There have been several solutions to reduce flashing in AC-LED, such as using a converter-free AC-LED driver, increasing drive frequency etc. However, these solutions suffer from low power efficiency or large size. Fortunately, the use of long afterglow phosphors is considered to be a promising method to compensate for the time gap and reduce the flickering effect. Long afterglow phosphors, such as SrSi
2O
2N
2:Eu
2+, Mn
2+ [
2], SrAl
2O
4:Eu
2+, Ce
3+, Li
+ [
5], and SrAl
2O
4:Eu
2+, and RE
3+ (RE = Y, Dy) have been studied and applied to AC-LEDs [
4,
7].
SrAl
2O
4:Eu
2+ is one of green phosphors for UVLEDs. When it is co-doped with RE
3+ (RE = Dy, Y, Nd and Ho), SrAl
2O
4:Eu
2+, Re
3+ can store electrons to achieve long and persistent luminescence. However, the afterglow time of SrAl
2O4:Eu
2+, Dy
3+ is too long to fulfill the requirement of AC-LED. In our previous research, we shortened the average lifetime of SrAl
2O
4:Eu
2+, Dy
3+ phosphors from 845.86 ms to 428.83 ms by changing the amount of SrCl
2 flux. Moreover, MAl
2O
4:Eu
2+, Dy
3+ (M = Ca
2+, Sr
2+, Ba
2+) phosphors with different cations also have different defect levels and afterglow times [
7,
8,
9,
10,
11,
12,
13]. Therefore, the substitution of Sr
2+ by Ca
2+ or Ba
2+ ions in SrAl
2O
4:Eu
2+, Dy
3+ phosphors is expected to tune the afterglow time. Furthermore, the emission and excitation spectra of Eu
2+ ions can also be modified by the different radius of Ca
2+, Sr
2+ and Ba
2+ ions and crystal structures [
13,
14,
15,
16].
This paper aims to improve the emission color and the average lifetime of SrAl2O4:Eu2+, Dy3+ phosphors by partially substituting Sr2+ with Ca2+ or Ba2+ ions. The effect and correlation of Ca2+ or Ba2+ ions substitution on the obtained phases, optical properties, and decay times of SrAl2O4:Eu2+, Dy3+ phosphors are systematically investigated. The electroluminescent properties of the as-fabricated LEDs based on UV-chips and MAl2O4:Eu2+, Dy3+ (M = Ca2+, Sr2+, Ba2+) phosphors are also investigated.
2. Experimental Procedure
Sr0.90−xCaxAl2O4:0.05Eu2+, 0.05Dy3+ and Sr0.90−yBayAl2O4:0.05Eu2+, 0.05Dy3+ samples (x or y = 0.00, 0.15, 0.30, 0.45, 0.60, 0.75, 0.90) were prepared by a high temperature solid-state reaction method. The raw materials CaCO3 (99.99%), SrCO3 (99.99%), BaCO3 (99.99%), Al2O3 (99.99%), Eu2O3 (99.99%), Dy2O3 (99.99%) and H3BO3 (99.99%) were weighted stoichiometrically. The original materials were mixed into an agate mortar and placed into a small alumina crucible. Then, the mixtures were put into a tubular furnace and heated to 900 °C for 2 h. Being reground, they were heated to 1400 °C for 4 h under a reductive atmosphere from burning activated carbon. Finally, the samples were cooled to room temperature, resulting in Sr0.9−xCaxAl2O4:0.05Eu2+, 0.05Dy3+, and Sr0.9−yBayAl2O4:0.05Eu2+, 0.05Dy3+ compounds.
The structure of Sr0.90−xCaxAl2O4:0.05Eu2+, 0.05Dy3+, and Sr0.90−yBayAl2O4:0.05Eu2+, 0.05Dy3+ phosphors were explored by X-ray powder diffraction (XRD) using a Diffractometer (X′ Pert PRO, Panalytical, Almelo, Netherlands) with Cu Kα radiation at 40 kV and 20 mA. The data were collected in the 2θ range of 10° to 80°. The photoluminescence (PL) spectra and decay curves were performed by a spectrofluorometer (F-4600, Hitachi, Tokyo, Japan) equipped with a 150W Xe lamp as the light source.
The phosphors were pre-coated on near-UV Ga(In)N chips with 365 and 395 nm emission, respectively. The emission spectra and parameters of the as-fabricated LEDs were measured by the LED spectrophotocolorimeter (PMS 50, Everfine Co., Ltd., Hangzhou, China) with an integrating sphere of 50 cm diameter. The normal forward-bias current was 20 mA. All measurements were conducted at room temperature.
3. Results and Discussion
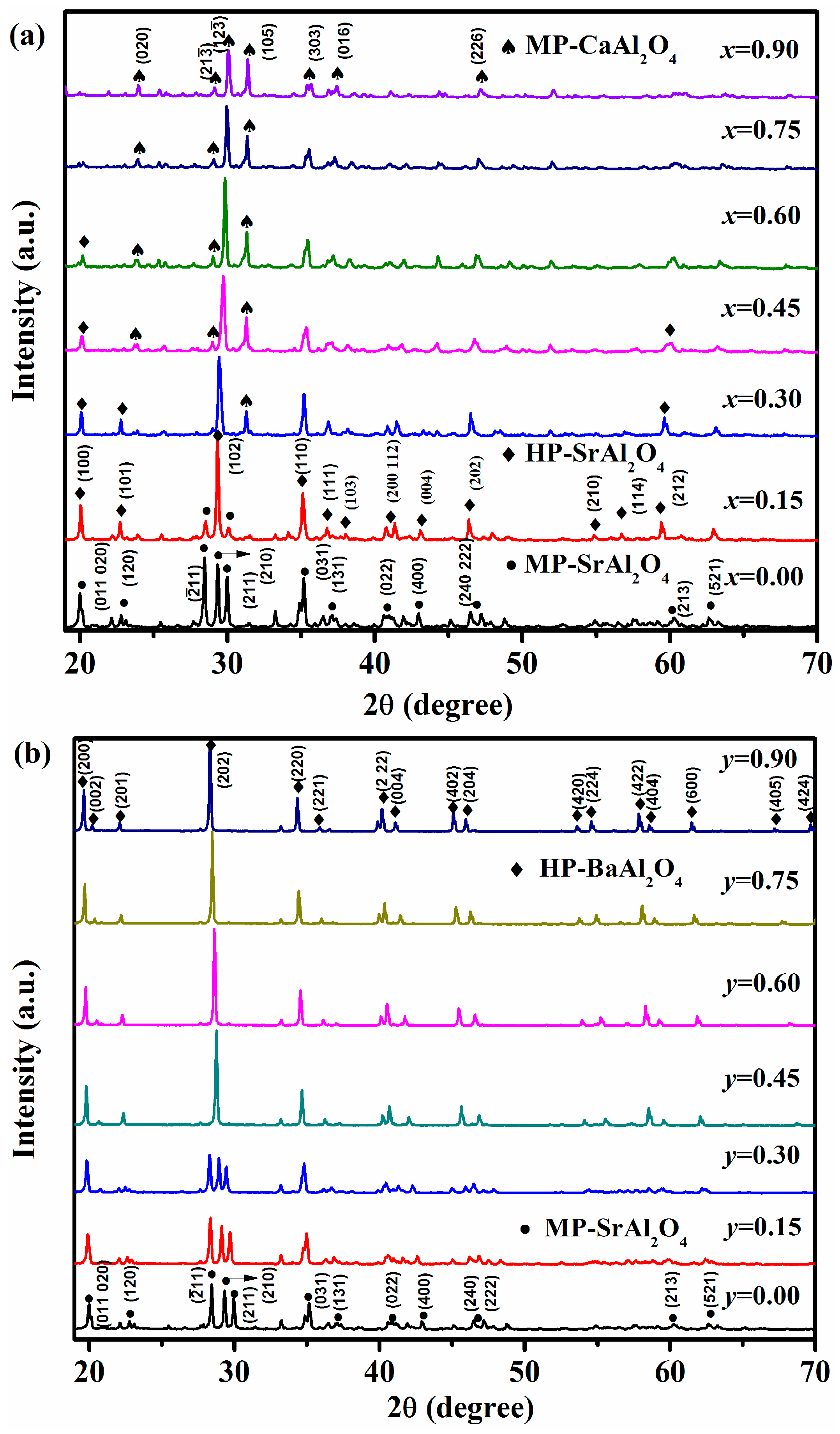
3.1. X-ray Diffraction Analysis
XRD patterns of Sr
0.90−xCa
xAl
2O
4:0.05Eu
2+, 0.05Dy
3+ (
x = 0.00–0.90) phosphors with various
x values are depicted in
Figure 1a. The patterns are compared with the JCPDS standard cards of possible phases and marked with different symbols. For
x(Ca
2+) = 0.00, the diffraction peaks are well matched with the monoclinic SrAl
2O
4 (MP-SrAl
2O
4, JCPDS card No. 74-0794), and minor content of the raw material—Al
2O
3—is observed due to the incomplete reaction, which is explained by our previous work [
4]. For
x(Ca
2+) = 0.15, the dominant phase is the hexagonal SrAl
2O
4 (HP-SrAl
2O
4, JCPDS card No. 31-1336). Thus, the different phase formation from the monoclinic to the hexagonal phase happens in Sr
0.90−xCa
xAl
2O
4:Eu
2+, Dy
3+ phosphors when the substitution of Sr
2+ by Ca
2+ increases from 0.00 to 0.15. The hexagonal phase remains when
x(Ca
2+) = 0.30. Simultaneously, the MP-SrAl
2O
4 disappears and minor content of monoclinic CaAl
2O
4 phase emerges. The monoclinic CaAl
2O
4 phase (MP-CaAl
2O
4, CPDS card No.53-0191) gradually increases with further elevating concentration of Ca
2+ from 0.45 to 0.75, accompanying with the decrease of HP-SrAl
2O
4. For
x(Ca
2+) = 0.90, the dominant phase isMP-CaAl
2O
4. In general, as
x(Ca
2+) varies from 0.00 to 0.90, and the obtained phases can be varied as follows: MP-SrAl
2O
4 → HP-SrAl
2O
4 → MP-Cal
2O
4. Furthermore, it can be concluded that two different phase always coexist due to the cations substitution except for the samples with
x(Ca
2+) = 0.00 and 0.90. The phase change is different with that of other report under different synthesized condition [
13].
Unlike the substitution of Sr
2+ by Ca
2+ in SrAl
2O
4:Eu
2+, Dy
3+ phosphors, there is only the variation from MP-SrAl
2O
4 to hexagonal BaAl
2O
4 phase (HP-BaAl
2O
4, JCPDS card No. 17-0306) during the substitution of Sr
2+ by Ba
2+. The coexistence of two phases cannot be observed from the XRD data, as displayed in
Figure 2b. Specifically, when
y(Ba
2+) = 0.00–0.30, the crystal structure remains the MP-SrAl
2O
4. However, as
y(Ba
2+) = 0.45, the XRD patterns are consistent with the standard HP-BaAl
2O
4. It remains unchanged while
y varies from 0.45 to 0.90, which stems from the small radius of Sr
2+ compared to that of Ba
2+ and the effect of chemical pressure of Ba
2+ on the crystal structure. Therefore, the Sr
0.90−yBa
yAl
2O
4:0.05Eu
2+, 0.05Dy
3+ compounds can be regarded as the hexagonal BaAl
2O
4phase replaced by Sr
2+ ions as
y(Ba
2+) = 0.45–0.90. The different phase formation from MP-SrAl
2O
4 to HP-BaAl
2O
4 can be obtained when Sr
2+ is replaced by Ba
2+ in SrAl
2O
4:Eu
2+, Dy
3+ phosphors.
Another finding is that the peaks of the Sr
0.90−xCa
xAl
2O
4:0.05Eu
2+, 0.05Dy
3+ shift toward larger 2
θ with Ca
2+ concentration increased (
x = 0.45–0.90), while the peaks of the Sr
0.90−yBa
yAl
2O
4:0.05Eu
2+, 0.05Dy
3+ shift to smaller 2
θ when Ba
2+ concentration increased (
y = 0.00–0.90). Both of these shifts can be explained by the variation of crystal lattice due to the different ionic radius of Ca
2+, Sr
2+, and Ba
2+ ions of 0.114 nm, 0.123 nm and 0.134 nm, respectively [
13,
17]. Therefore, when Sr
2+ is replaced by Ca
2+, the crystal lattice of SrAl
2O
4 phase will shrink. On the contrary, it will expand when Sr
2+ is substituted by Ba
2+. Therefore, the difference in the ionic radius leads to the shifts of the diffraction peaks.
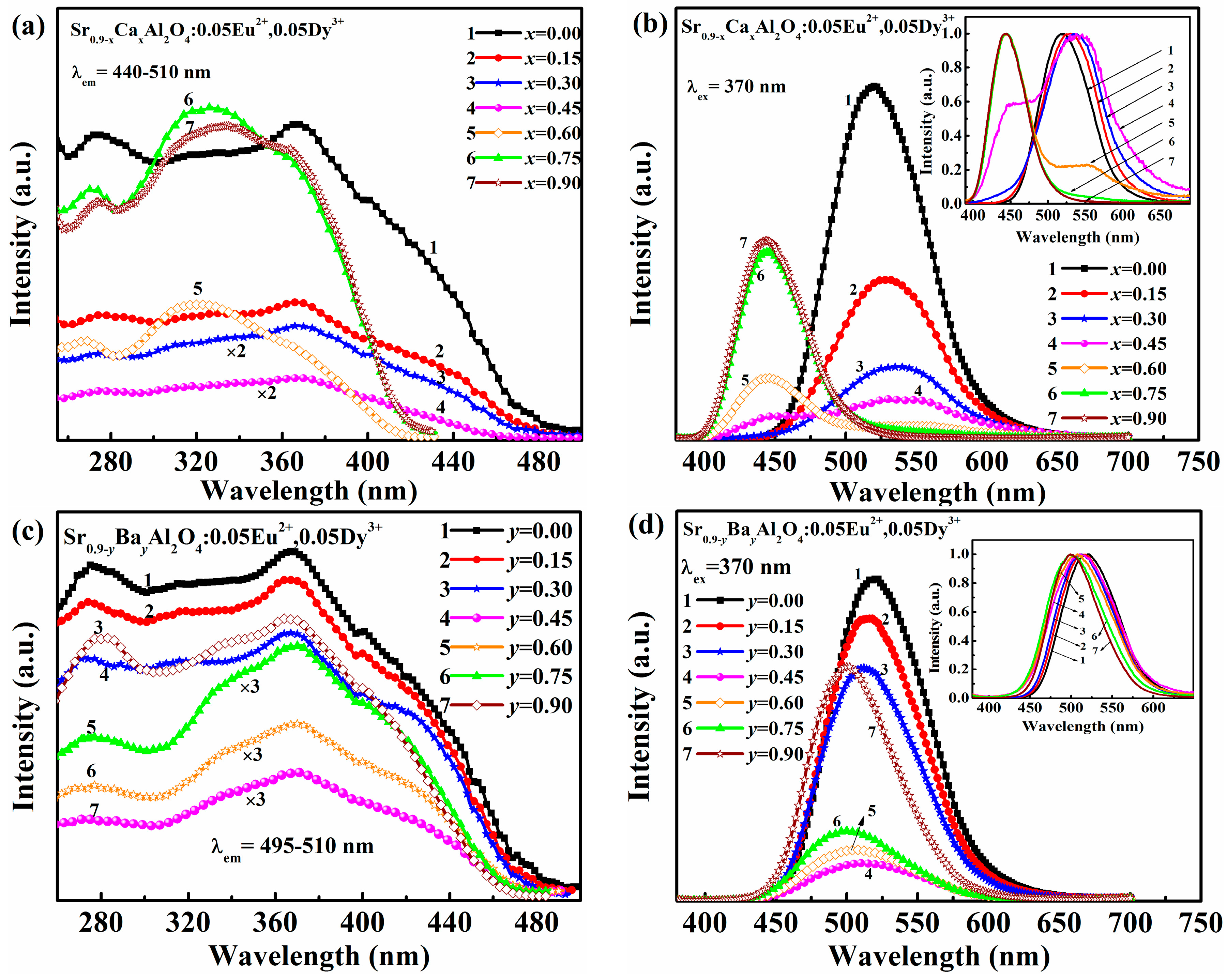
3.2. Luminescence Properties of Sr0.09−xCaxAl2O4:0.05Eu2+, 0.05Dy3+ and Sr0.90−yBayAl2O4:0.05Eu2+, 0.05Dy3+ Phosphors
Figure 2 illustrates the emission and excitation spectra of Sr
0.90−xCa
xAl
2O
4:0.05Eu
2+, 0.05Dy
3+ and Sr
0.90−yBa
yAl
2O
4:0.05 Eu
2+, 0.05Dy
3+ phosphors with
x(
y) ranged from 0.00 to 0.90. The excitation spectra of Sr
0.90−xCa
xAl
2O
4:0.05Eu
2+, 0.05Dy
3+ phosphors are depicted in
Figure 2a with monitoring the emission of Eu
2+ in the range of 440–510 nm. All samples present broad bands from 250 nm to 450 nm due to 4
f7→4
f65
d1 transitions of Eu
2+ ions [
18]. The shape of excitation bands almost remain unchanged while the intensity decreases when
x value (
x= 0.00–0.45) increases. However, it should be noted that excitation bands are different from those of
x < 0.60. The excitation bands of Sr
0.90−xCa
xAl
2O
4:0.05Eu
2+, 0.05Dy
3+ (
x = 0.60–0.90) are similar and blue-shift, which is ascribed to the phase variation as discussed in XRD analysis.
Figure 2b shows the emission spectra of Sr
0.90−xCa
xAl
2O
4:0.05Eu
2+, 0.05Dy
3+ phosphors. It can be divided into three sections (
x = 0–0.30,
x = 0.45–0.60,
x = 0.75–0.90). Interestingly, these results are related to the phase transition. First, the luminescence intensity decreases and the emission peak shifts from 520 nm to 538 nm as
x varied from 0.00 to 0.30. Second, two emission peaks around 450 nm, 540 nm at
x = 0.45–0.60 are observed due to the coexistence of heterogeneous mixtures of two structures. Finally, the emission spectrum presents a single peak around 444 nm when
x value increased from 0.75 to 0.90, while the dominant phase is monoclinic CaAl
2O
4.
The excitation and emission spectra of Sr
0.90−yBa
yAl
2O
4:0.05Eu
2+, 0.05Dy
3+ phosphors are displayed in
Figure 2c,d. The excitation spectra show similar outlines with two broad bands around 275 nm, 375 nm and a shoulder around 420 nm. The broad emission bands originate from the typical transitions of 4
f7 → 4
f65
d1 of Eu
2+ ions, which are affected by the crystal field of host lattice [
19]. The emission band shifts to a shorter wavelength from 520 nm to 500 nm as
y(Ba
2+) increased. It can be explained that the crystal field splitting energy is decreased due to the phase change of the substation of Sr
2+ by the larger Ba
2+ ions. However, the luminescence intensity decreases when Ba
2+ content is 0.00–0.45, while it increases when Ba
2+ content is 0.60–0.90.These changes match very well with the XRD results mentioned above and can be ascribed to Eu
2+ emission in different phase.
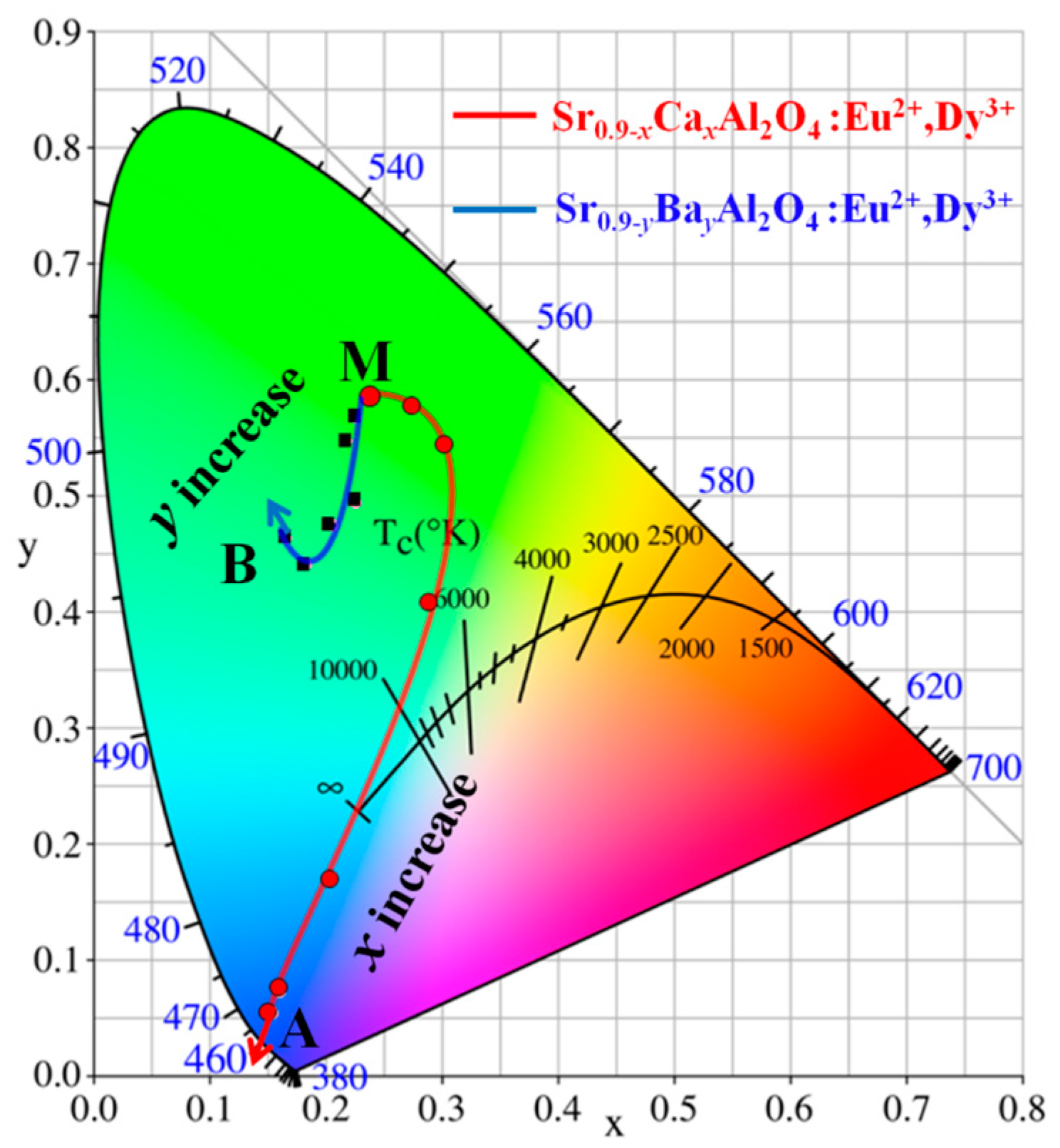
The CIE chromaticity coordinates calculated from the spectra of Sr
0.90−xCa
xAl
2O
4:0.05Eu
2+, 0.05Dy
3+ and Sr
0.9-yBa
yAl
2O
4:0.05Eu
2+, 0.05Dy
3+ phosphors with varied
x (or
y) values are presented in
Figure 3 and
Table 1. These results show that the CIE color coordinates of SrAl
2O
4:Eu
2+, Dy
3+ phosphors can be tuned by Ca
2+ or Sr
2+ content. For Sr
0.90−xCa
xAl
2O
4:0.05Eu
2+, 0.05Dy
3+ phosphors, the color coordinates can be changed from green (0.2393, 0.5874, point M) to blue (0.1531, 0.0528, point A) area with
x increased. In addition, the color coordinates of Sr
0.9−yBa
yAl
2O
4:0.05Eu
2+, 0.05Dy
3+ phosphors have a little shift from green (0.2399, 0.5867, point M) to bluish green (0.1652, 0.4645, point B) area. The above discussion indicates that the emission color of SrAl
2O
4:Eu
2+, Dy
3+ can be tuned due to the phase change of the substitution of Sr
2+ with Ca
2+ or Ba
2+ ions. It is an important challenge to tune the color of phosphors for improving the color rendering index of w-LEDs [
20,
21,
22].
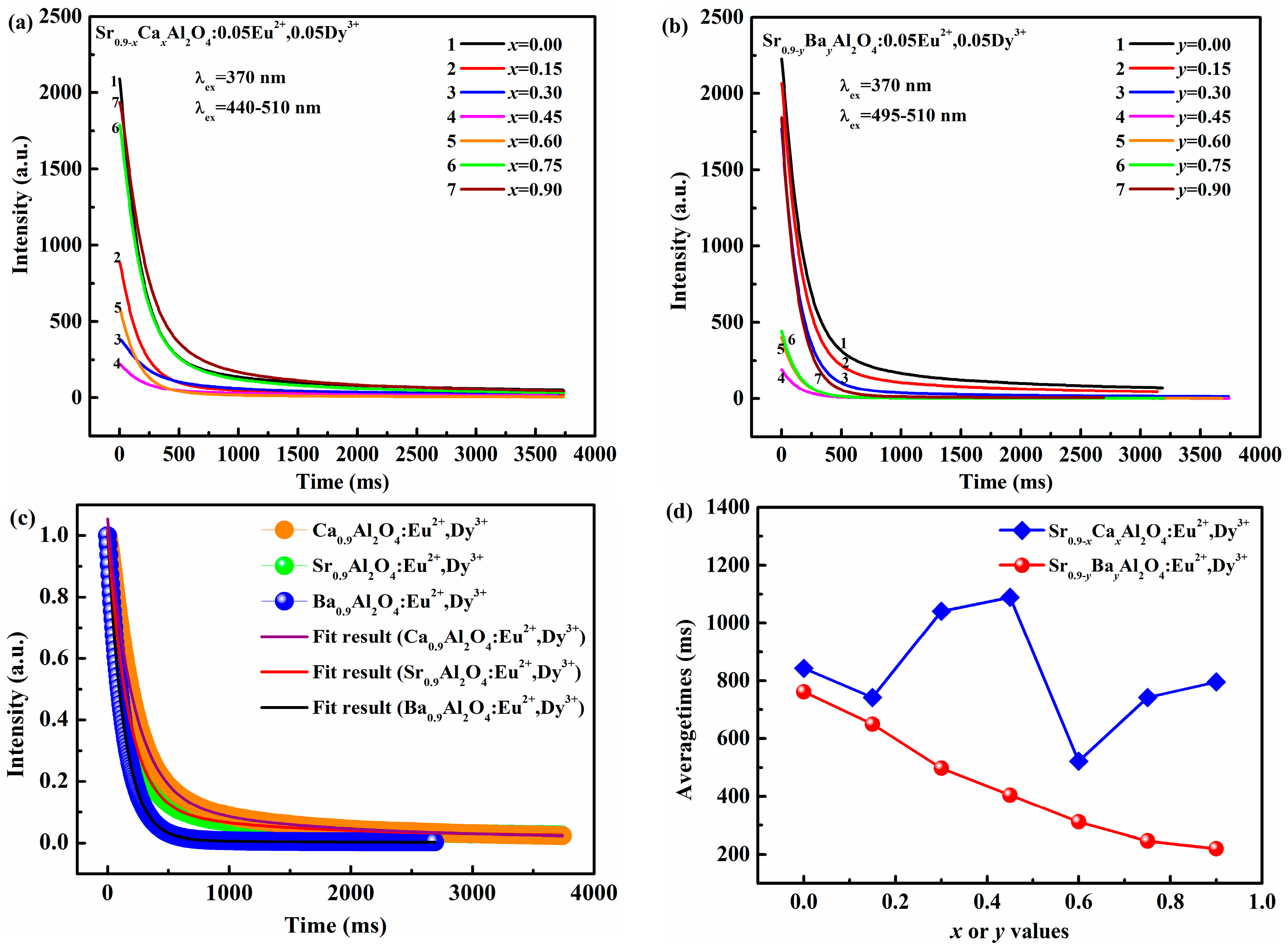
3.3. Decay Characteristics
The photoluminescence decay curves of SrAl
2O
4:Eu
2+, Dy
3+ phosphors with varied Ca
2+ and Ba
2+ content are shown in
Figure 4a,b, respectively. The figures reveal the initial luminous intensity is different when Sr
2+ is replaced by either Ca
2+ or Ba
2+ in SrAl
2O
4:Eu
2+, Dy
3+.Among these phosphors, Sr
0.90Al
2O
4:0.05Eu
2+, 0.05Dy
3+ has the highest initial luminous intensity. The initial luminous intensity gradually decreases when the content of Ca
2+(Ba
2+) increased to 0.45. By further increasing the Ca
2+ or Ba
2+ concentration, the initial luminous intensity enhances. The variation of initial luminous intensity is consistent with the emission spectra, which can be explained by the change of crystal structure.
Moreover, the decay process of all samples is composed of fast decay, medium decay and subsequent of slow decay, which is consistent with our previous report. The decay curves of Ca
0.90Al
2O
4:0.05Eu
2+, 0.05Dy
3+, Sr
0.90Al
2O
4:0.05Eu
2+, 0.05Dy
3+, and Ba
0.90Al
2O
4:0.05Eu
2+, 0.05Dy
3+ phosphors are fitted as representatives, which are presented in
Figure 4c. The decay curves are well fitted with the following triple exponential functions:
where
t is the time,
I0 and
I is the luminescence intensity at initial time and
t, respectively.
A,
B and
C are constants.
1,
2, and
3 are the decay time for the exponential components, respectively. The average lifetimes can be obtained as follows. The detail parameters are listed in
Table 2.
On the basis of the Equation (2), the average lifetime (
average) of SrAl
2O
4:Eu
2+, Dy
3+ phosphors with varied Ca
2+ and Ba
2+ contents are determined and shown in
Figure 4d, respectively. The order of the average lifetimes is Sr
0.90Al
2O
4:0.05Eu
2+, 0.05Dy
3+ > Ca
0.90Al
2O
4:0.05Eu
2+, 0.05Dy
3+ > Ba
0.90Al
2O
4:0.05Eu
2+, 0.05Dy
3+. The average lifetimes of Sr
0.90−xCa
xAl
2O
4:0.05Eu
2+, 0.05Dy
3+ phosphors vary with the change of Ca
2+ content, and the maximum is about 1088.5 ms at
x = 0.45. It could be ascribed to the complicate formation of the phase in
Figure 1. Furthermore, the average lifetimes of Sr
0.90−yBa
yAl
2O
4:0.05Eu
2+, 0.05Dy
3+ phosphors gradually decrease with Ba
2+ content increased due to the different phase formation and reach the shortest time of 219.91 ms at
y = 0.90, demonstrating that the average lifetime or decay characteristics of SrAl
2O
4:Eu
2+. Dy
3+ could be adjusted by the partial substitution of Ba
2+, Ca
2+. The results also indicate that the tuned lifetimes are more suitable for the application in AC-LEDs as the global widely utilize 50/60 Hz alternating current with a 5–20 ms dark duration.
3.4. Application of SrAl2O4:Eu2+, Dy3+ Phosphors with Varied Contents of Ca2+ and Ba2+ in LEDs
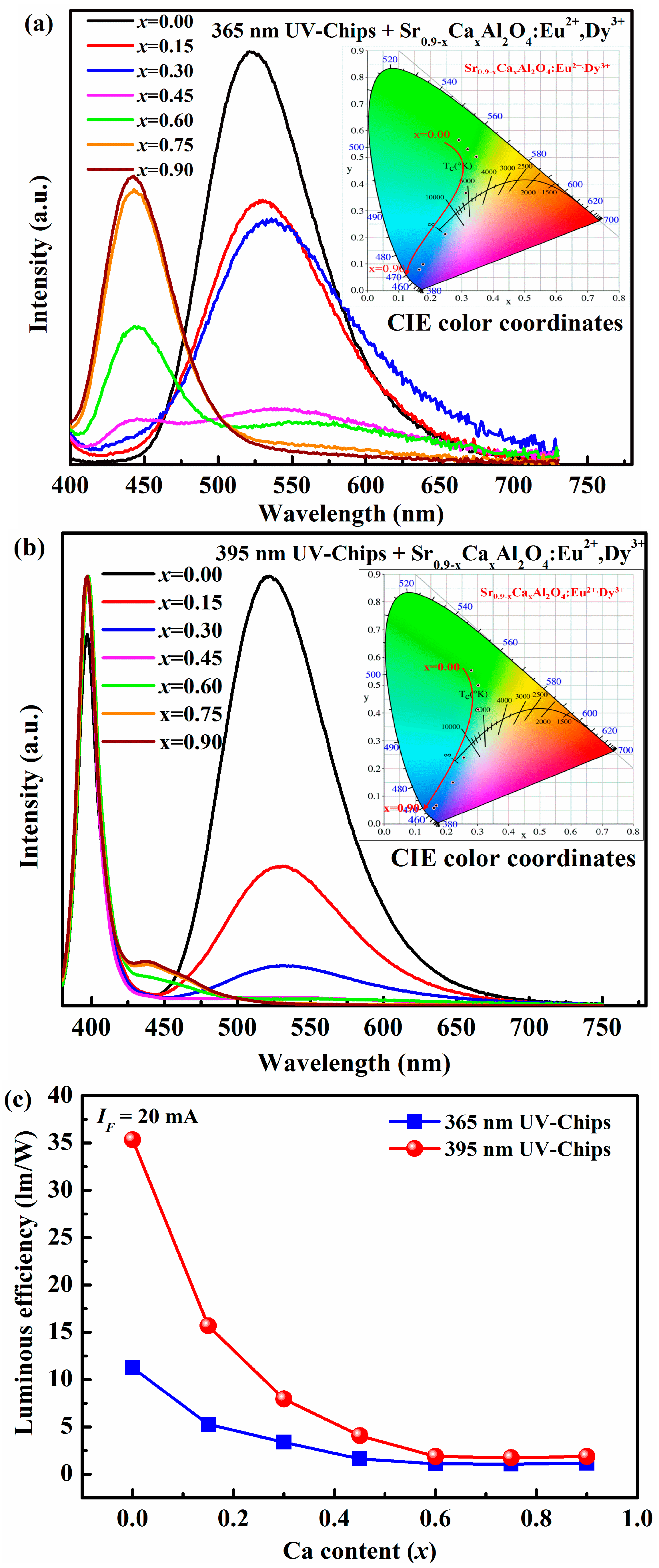
Finally, phosphors converted LEDs were fabricated with silicone, near UV-chips (~365 nm, 395 nm), Sr0.90−xCaxAl2O4:0.05Eu2+, 0.05Dy3+, and Sr0.90−yBayAl2O4:0.05Eu2+, 0.05Dy3+ phosphors. The phosphors account for 20% of the mass of silicone.
Figure 5 is the electroluminescence spectra of the as-fabricated LEDs based on Sr
0.90−xCa
xAl
2O
4:0.05Eu
2+, 0.05Dy
3+ phosphors and near UV Ga(In)N chips. In the
Figure 4a, the shapes of all electroluminescence spectra in the as-fabricated LEDs are similar with the photoluminescence emission spectra of Sr
0.90−xCa
xAl
2O
4:0.05Eu
2+, 0.05Dy
3+ phosphors because the near UV around 365 nm of chips is completely absorbed by phosphors.
Figure 5b depicts that the emission band located at 395 nm belongs to the UV-chips. And other emission bands are ascribed to Eu
2+ emissions excited by 395 nm UV-chips. The results prove that the Sr
0.90−xCa
xAl
2O
4:0.05Eu
2+, 0.05Dy
3+ fluorescence materials can absorb the light with wavelength of 365 nm or 395 nm, and convert it into green or blue visible-light. The CIE color coordinates of as-fabricated LEDs are calculated and listed in
Table 3. The inset of
Figure 5 presents the chromaticity diagram of the as-fabricated LEDs based on SrAl
2O
4:Eu
2+, Dy
3+ phosphors, which can be tuned from green to blue area and are similar with that of phosphors. In addition, the luminous efficiency is also systematically investigated under 20 mA forward-bias current in this work, as indicated in
Figure 5c. The luminous efficiency of the as-fabricated LEDs with 395 nm is higher than in the case of 365 nm UV chips. However, for both kinds of LEDs fabricated by 365 nm and 395 nm UV-chips, the luminous efficiency decreases strongly, then increases slowly with Ca
2+ content elevated. The maximum luminous efficiency is 11.24 lm/W for 365 nm LEDs and 35.35 lm/W for 395 nm LEDs (
x = 0).
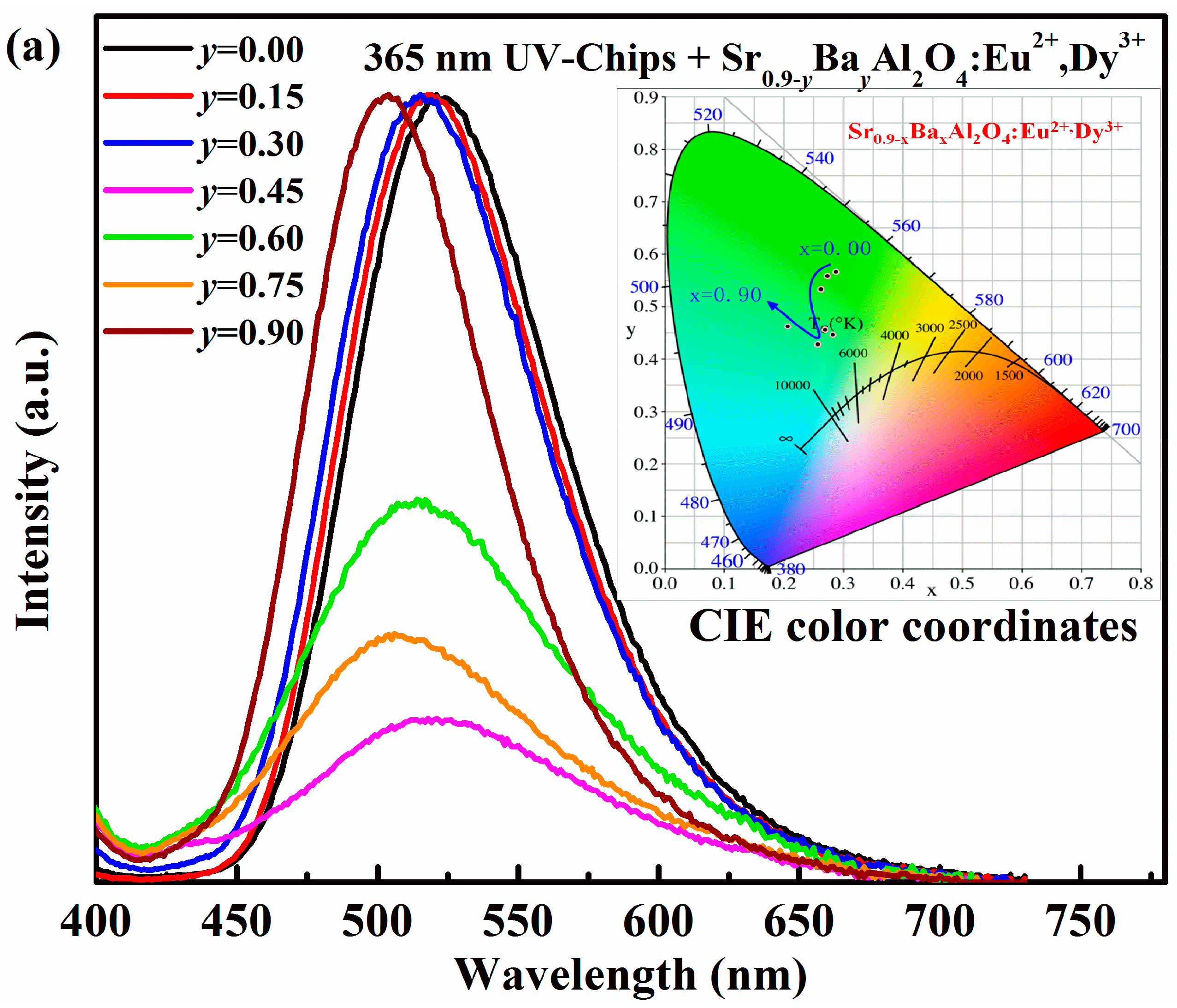
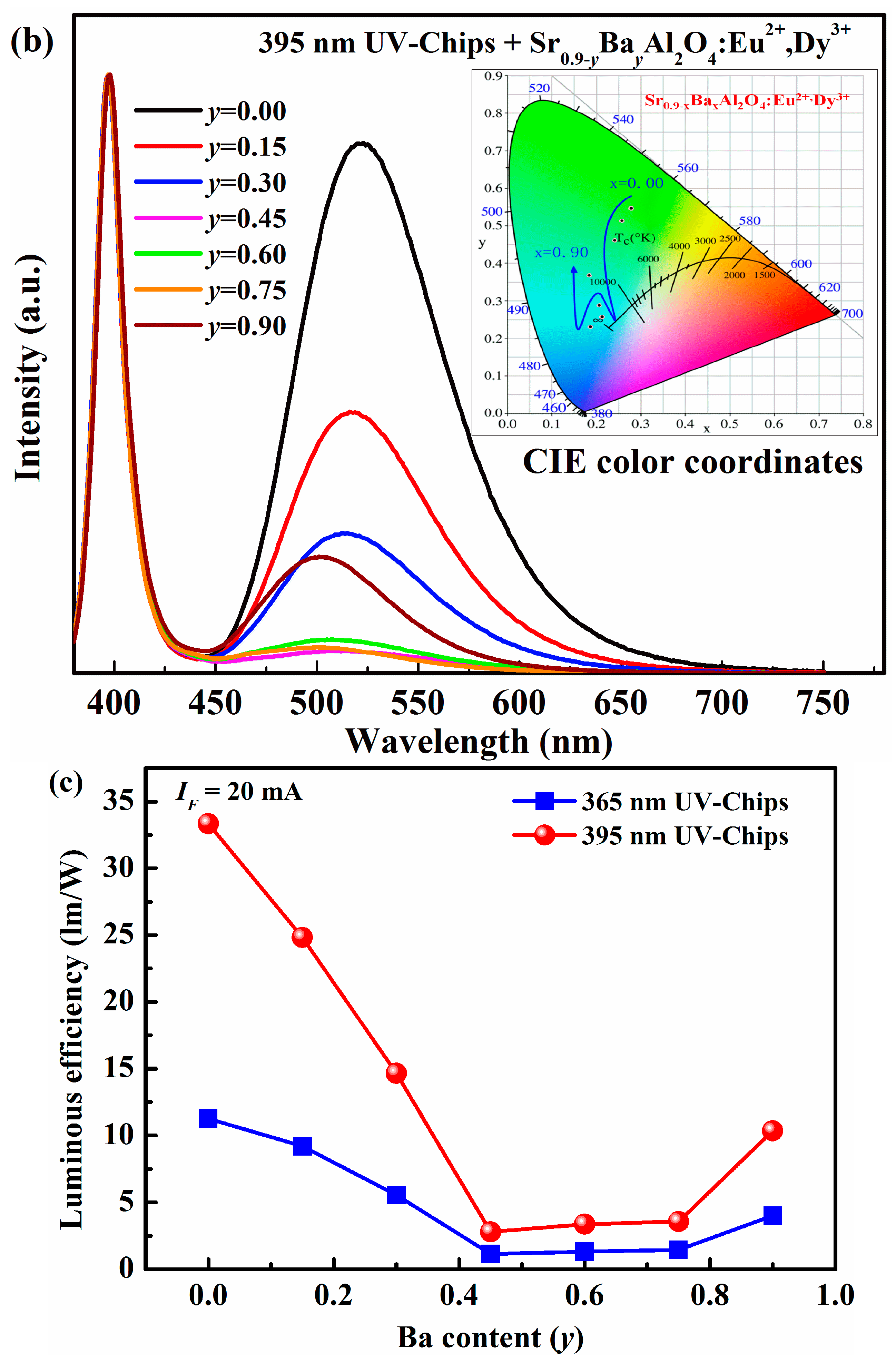
The electroluminescence spectra of LEDs based on Sr
0.90−xBa
xAl
2O
4:0.05Eu
2+, 0.05Dy
3+ phosphors are also investigated, as shown in
Figure 6.Except emission peaks of the UV chip itself, all of the electroluminescence spectra of Sr
0.90−yBa
yAl
2O
4:0.05Eu
2+, 0.05Dy
3+ excited by both 365 nm and 395 nm UV-chips are similar. It indicates that the Sr
0.9-yBa
yAl
2O
4:Eu
2+, Dy
3+ phosphors can absorb 365 nm or 395 nm emission from Ga(N)In chips, and convert it into green or blue-green visible-light. The CIE color coordinates of the as-fabricated LEDs are calculated and listed in
Table 4. By varying Ba
2+ concentrations, the color of the as-fabricated LEDs can be tuned, but the adjustable zone is narrower than in the case of Ca
2+. It suggests that the color coordinates of pc-LEDs cannot be influenced by near UV Ga(In)N chips. However, the luminous efficiency decreases at first, and then increases with Ba
2+ content enhanced. The maximum luminous efficiency is 11.26 lm/W for 365 nm LEDs and 33.34 lm/W for 395 nm LEDs (
y = 0). The variation of luminous efficiency of the as-fabricated LED is also consistent with the emission spectra due to the different phase formation.
4. Conclusions
Sr0.90−xCaxAl2O4:0.05Eu2+, 0.05Dy3+ and Sr0.90−yBayAl2O4:0.05Eu2+, 0.05Dy3+ (x or y = 0.00, 0.15, 0.30, 0.45, 0.60, 0.75, 0.90) phosphors were synthesized. The variation of optical properties for the phosphors is ascribed to the phase structures. For the series of Sr0.90−xCaxAl2O4:0.05Eu2+, 0.05Dy3+ phosphors, two heterogeneous structures coexist, except the samples with x(Ca2+) = 0.00 and 0.90. The emission color of SrAl2O4:Eu2+, Dy3+ phosphor can be adjusted largely from green to blue, and the lifetime can also be tuned largely from 1088.5 ms to 521.6 ms. For Sr0.90−yBayAl2O4:0.05Eu2+, 0.05Dy3+ phosphors, the phase transforms from monoclinic SrAl2O4 to hexagonal BaAl2O4 phase. Meanwhile, the emission wavelength shifts from 520 nm to 500 nm, and the color slightly changes from green to blue-green. More importantly, the average lifetime of SrAl2O4:Eu2+, Dy3+ can be shortened from 842.04 ms to 219.91 ms, which can appropriately compensate for the AC time gap. Finally, the LEDs are successfully fabricated by combining the phosphors with Ga(In)N UV-chips. The maximum of luminous efficiency reaches 33.34 lm/W based on SrAl2O4:Eu2+, Dy3+ phosphors.