Investigating “Egusi” (Citrullus Colocynthis L.) Seed Oil as Potential Biodiesel Feedstock
Abstract
:1. Introduction
2. Experimental Section
2.1. Materials
2.2. Analysis of egusi melon seed oil
2.3. Transesterification
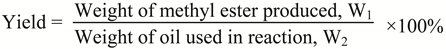
2.4. Gas chromatography analysis
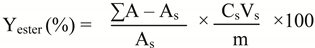
2.5. Biodiesel property determination
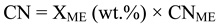
3. Results and Discussion
3.1. Physicochemical properties of egusi melon seed oil
3.2. Fatty ester composition
| Parameter | Egusi |
|---|---|
| Iodine value (g I2/100g) | 114.46 |
| Density at 15 °C (kg/m3) | 905.3 |
| Kinematic viscosity at 40 °C (mm2/s) | 31.52 |
| Saponification value (mg KOH/g) | 204.44 |
| Acid value (mg KOH/g) | 0.98 |
| Free fatty acid (%) | 0.49 |
| Caloric value (MJ/kg) | 39.37 |
| Colour | 5Y + 0.4R |
| Ave. Molecular weight (g) | 874 |
3.3. Biodiesel yield
3.4. Fuel properties of egusi melon oil methyl ester
| Fatty acid | Class | Palma | Jatrophab | Rapea | Soybeana | Safflowera | Sunflowera | Egusi | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Caprylic | C8:0 | 0.1 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | ||||
| Capric | C10:0 | 0.1 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | ||||
| Lauric | C12:0 | 0.9 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | ||||
| Myristic | C14:0 | 1.3 | 0.0 | 0.0 | 0.1 | 0.1 | 0.1 | 0.0 | ||||
| Palmitic | C16:0 | 43.9 | 18.22 | 2.7 | 10.3 | 6.6 | 6.0 | 10.48 | ||||
| Palmitoleic | C16:1 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.06 | ||||
| Stearic | C18:0 | 4.9 | 5.14 | 2.8 | 4.7 | 3.3 | 5.9 | 9.72 | ||||
| Oleic | C18:1 | 39.0 | 28.46 | 21.9 | 22.5 | 14.4 | 16.0 | 17.95 | ||||
| Linoleic | C18:2 | 9.5 | 48.18 | 13.1 | 54.1 | 75.5 | 71.4 | 61.41 | ||||
| Linolenic | C18:3 | 0.3 | 0.0 | 8.6 | 8.3 | 0.1 | 0.6 | 0.38 | ||||
| Erucic | C22:1 | 0.0 | 0.0 | 50.9 | 0.0 | 0.0 | 0.0 | 0.0 | ||||
| Total saturated | 51.20 | 23.36 | 5.50 | 15.10 | 10.00 | 12.00 | 20.20 | |||||
| Total unsaturated | 48.80 | 76.64 | 94.50 | 84.90 | 90.00 | 88.00 | 79.80 | |||||
| Property | Unit | Limits | SOME | SUOME | SAOME | EMOME | |
|---|---|---|---|---|---|---|---|
| ASTM D6751 | EN 14214 | ||||||
| Ester content | % (mol/mol) | - | 96.5 min | 96.9a | 97.2a | 97.67c | 96.78 |
| Density; 15 °C | kg/m3 | - | 860–900 | 885d | 884b | 874c | 883 |
| Kinematic viscosity; 40 °C | mm2/s | 1.9–6.0 | 3.5–5.0 | 4.2a | 4.85b | 4.29c | 3.83 |
| Flash point | ºC | 130 min | 120 min | 171a | 168b | 176c | 142 |
| Cloud point | °C | Report | - | 1d | 1b | 2c | 0.5 |
| Acid value | mg KOH/g | 0.5 max | 0.50 max | 0.14a | 0.4b | 0.28c | 0.19 |
| Linolenic acid content | % (mol/mol) | - | 12.0 max | 6.3a | 0.2a | - | 0.38 |
| Higher heating value | MJ/kg | - | - | 41.28e | 45.5b | 45.21c | 39.97 |
| Oxidation number | h | 3 min | 6 min | 1.3a | 1.96b | - | 1.41 |
| Cetane number | 47 min | 51 min | 49a | 55b | 52.32c | 53.66* | |
| Free glycerol | wt.% | 0.02 max | 0.02 max | 0.07a | 0.015f | 0.016 | 0.011 |
| Total glycerol | wt.% | 0.24 max | 0.25 max | 0.00a | 0.201f | 0.225c | 0.192 |
4. Conclusions
Acknowledgements
References and Notes
- Murugesan, A.; Umarani, C.; Chinnusamy, T.; Krishnan, M.; Subramanian, R.; Neduzchezhain, N. Production and analysis of bio-diesel from non-edible oils–A review. Renew. Sustain. Energy Rev. 2009, 13, 825–834. [Google Scholar] [CrossRef]
- Balat, M.; Balat, H. A critical review of biodiesel as vehicular fuel. Energ. Conv. Mgmt. 2009, 49, 2727–2741. [Google Scholar] [CrossRef]
- Demirbas, A. Progress and recent trends in biodiesel fuels. Renew. Sustain. Energy Rev. 2009, 50, 14–34. [Google Scholar]
- Rashid, U.; Answar, F.; Moser, B.R.; Knothe, G. Moringa oleifera oil: A possible source ofbiodiesel. Bioresour. Technol. 2008, 99, 8175–8179. [Google Scholar] [CrossRef] [PubMed]
- Hosamani, K.M.; Hiremath, V.B.; Keri, R.S. Renewable energy sources from Michelia champaca and Garcinia indica seed oils: A rich source of oil. Biom. Bioenerg. 2009, 33, 267–270. [Google Scholar] [CrossRef]
- Schinas, P.; Karavalakis, G.; Davaris, C.; Anastopoulos, G.; Karonis, D.; Zannikos, F.; Stournas, S.; Lois, E. Pumpkin (Cucurbita pepo L.) seed oil as an alternative feedstock for the production of biodiesel in Greece. Biomass Bioenerg. 2009, 33, 44–49. [Google Scholar] [CrossRef]
- Kansedo, J.; Lee, K.T.; Bhatia, S. Cerbera odollam (sea mango) oil as a promising non-edible feedstock for biodiesel production. Fuel 2009, 88, 1148–1150. [Google Scholar] [CrossRef]
- Chapagain, B.P.; Yehoshua, Y.; Wiesman, Z. Desert date (balanites aegypytiaca) as an arid sustainable bioresource for biodiesel. Bioresour. Technol. 2009, 100, 1221–1226. [Google Scholar] [CrossRef] [PubMed]
- Ng, T.J. New opportunities in the cucurbitaceae. In New Crops, 1st ed.; Janick, J., Simon, J.E., Eds.; Wiley: New York, NY, USA, 1993; pp. 538–546. [Google Scholar]
- Oluba, M.O.; Ogunlowo, Y.R.; Ojieh, G.C.; Adebisi, K.E.; Eidangbe, G.O.; Isiosio, I.O. Physicochemical properties and fatty acid composition of citrullus lanatus (Egusi melon) seed oil. J. Biol. Sci. 2008, 8, 814–817. [Google Scholar] [CrossRef]
- Achu, M.B.; Fokou, E.; Tchiegang, C.; Fotso, M.; Tchouanguep, F.M. Nutritive value of some cucurbitaceae oilseeds from different regions in Cameroon. Afr. J. Biotechnol. 2005, 11, 1329–1334. [Google Scholar]
- Mabaleha, M.B.; Mitei, Y.C.; Yeboah, S.O. A comparative study of the properties of selected melon seed oils as potential candidates for development into commercial edible vegetable oil. J. Am. Oil Chem. Soc. 2007, 84, 31–36. [Google Scholar] [CrossRef]
- Ntui, V.O.; Thirukkumaran, G.; Iioka, S.; Mii, M. Efficient plant regeneration via organogenesis in ‘‘egusi’’ melon (Colocynthis citrullus L.). Scientia Hort. 2009, 119, 397–402. [Google Scholar] [CrossRef]
- Uruakpa, F.O.; Aluko, R.E. Heat-induced gelation of whole egusi (Colocynthis citrullus L.) seeds. Food Chem. 2004, 87, 349–354. [Google Scholar] [CrossRef]
- Federal Ministry of Agriculture and Rural Development. Agricultural outlook in Nigeria. In Proceedings of the Nigeria National Crop Outlook Conference, Kano State, Nigeria, May 2005; Available online: http://www.mistowa.org/files/CAFSTON/AGRICULTURAL%20OUTLOOK%20-%20PRSD.pdf (accessed on 26 December 2009).
- Yaniv, Z.; Shabelsky, E.; Schafferman, D. Colocynth: Potential arid land oilseed from an ancient cucurbit. In Perspectives on New Crops and New Uses, 1st Edition; Janick, J., Ed.; ASHS Press: Alexandria, VA, USA, 1999; pp. 257–261. [Google Scholar]
- Patil, P.D.; Deng, S. Optimization of biodiesel production from edible and non-edible vegetable oils. Fuel 2009, 88, 1302–1306. [Google Scholar] [CrossRef]
- Akgün, N.; Iscan, E. Effects of process variables for biodiesel production by transesterification. Eur. J. Lipid Sci. Technol. 2007, 109, 486–492. [Google Scholar] [CrossRef]
- Ramos, M.J.; Fernandez, C.M.; Casas, A.; Rodriguez, L.; Perez, A. Influence of fatty acid composition of raw materials on biodiesel properties. Bioresour. Technol. 2009, 100, 261–268. [Google Scholar] [CrossRef] [PubMed]
- Moser, B.R. Biodiesel production, properties, and feedstocks. In Vitro Cell. Dev. Biol.-Plant 2009, 45, 229–266. [Google Scholar] [CrossRef]
- Srivastava, A.; Prasad, R. Triglycerides-based diesel fuels. Renew. Sustain. Energy Rev. 2000, 4, 111–33. [Google Scholar] [CrossRef]
- Milovanović, M.; Pićurić-Jovanović, K. Characteristics and composition of melon seed oil. J. Agric. Sci. 2005, 50, 41–49. [Google Scholar]
- Al-Khalifa, A.S. Physicochemical characteristics, fatty acid composition, and lipoxygenase activity of crude pumpkin and melon seeds. J. Agric. Food Chem. 1996, 44, 964–966. [Google Scholar] [CrossRef]
- Kamel, B.S.; Dawson, H.; Kakuda, Y. Characteristics and composition of melon and grape seed oils and cakes. J. Am. Oil Chem. Soc. 1995, 62, 881–883. [Google Scholar] [CrossRef]
- Akoh, C.C.; Nwosu, C.V. Fatty acid composition of melon seed oil lipids and phospholipids. J. Am. Oil Chem. Soc. 1992, 69, 314–316. [Google Scholar] [CrossRef]
- Usta, N. Use of tobacco seed oil methyl ester in a turbocharged indirect injection diesel engine. Biomass Bioenerg. 2005, 28, 77–86. [Google Scholar] [CrossRef]
- Freedman, B.; Pryde, E.; Mounts, T. Variables affecting the yields of fatty esters from transesterified vegetable oils. J. Am. Oil Chem. Soc. 1984, 61, 1638–1643. [Google Scholar] [CrossRef]
- Srivastava, P.K.; Verma, M. Methyl ester of karanja oil as alternative renewable source energy. Fuel 2008, 87, 1673–1677. [Google Scholar] [CrossRef]
- Rashid, U.; Anwar, F.; Arif, M. Optimization of base catalytic methanolysis of sunflower (Helianthus annuus) seed oil for biodiesel production by using response surface methodology. Ind. Eng. Chem. Res. 2009, 48, 1719–1726. [Google Scholar] [CrossRef]
- Saydut, A.; Duz, M.Z.; Kaya, C.; Kafadar, A.B.; Hamamci, C. Transesterified sesame (Sesamum indicum L.) seed oil as a biodiesel fuel. Bioresour. Technol. 2008, 99, 6656–6660. [Google Scholar] [CrossRef] [PubMed]
- Sarin, R.; Sharma, M.; Sinharay, S.; Malhotra, R.K. Jatropha-palm biodiesel blends: An optimum mix for Asia. Fuel 2007, 86, 1365–1371. [Google Scholar] [CrossRef]
- Chakraborty, M.; Baruah, D.C.; Konwer, D. Investigation of terminalia (Terminalia belerica Robx.) seed oil as prospective biodiesel source for North-East India. Fuel Process. Technol. 2009, 90, 1435–1441. [Google Scholar] [CrossRef]
- Knothe, G.; Matheaus, A.C.; Ryan, T.W. Cetane numbers of branched and straight-chain fatty esters determined in an ignition quality tester. Fuel 2003, 82, 971–975. [Google Scholar] [CrossRef]
- Imahara, H.; Minami, E.; Saka, S. Thermodynamic study on cloud point of biodiesel with its fatty acid composition. Fuel 2006, 85, 1666–1670. [Google Scholar] [CrossRef]
- McCormick, R.L.; Ratcliff, M.; Moens, L.; Lawrence, R. Several factors affecting the stability of biodiesel in standard accelerated tests. Fuel Process. Technol. 2007, 88, 651–657. [Google Scholar] [CrossRef]
- Rashid, U.; Anwar, F. Production of biodiesel through base-catalyzed transesterification of safflower oil using an optimized protocol. Energy Fuels 2008, 22, 1306–1312. [Google Scholar] [CrossRef]
- Allen, C.A.W.; Watts, K.C.; Ackman, R.G.; Pegg, M.J. Predicting the viscosity of biodiesel fuels from their fatty acid ester composition. Fuel 1999, 78, 1319–1326. [Google Scholar] [CrossRef]
- Bajpai, D.; Tyagi, V.K. Biodiesel: Source, production, composition, properties and its benefits. J. Oleo Sci. 2006, 55, 487–502. [Google Scholar] [CrossRef]
- Demirbas, A. Relationships derived from physical properties of vegetable oil and biodiesel fuels. Fuel 2008, 87, 1743–1748. [Google Scholar] [CrossRef]
- Rashid, U.; Anwar, F.; Moser, B.R.; Ashraf, S. Production of sunflower oil methyl esters by optimized alkali-catalyzed methanolysis. Biomass Bioenergy 2008, 32, 1202–1205. [Google Scholar] [CrossRef]
- El-Diwani, G.; Attia, N.K.; Hawash, S.I. Development and evaluation of biodiesel fuel and by-products from jatropha oil. Int. J. Environ. Sci. Technol. 2009, 6, 219–224. [Google Scholar] [CrossRef]
- Rashid, U.; Anwar, F.; Ansari, T.M.; Arif, M.; Ahmad, M. Optimization of alkaline transesterification of rice bran oil for biodiesel production using response surface methodology. J. Chem. Technol. Biotechnol. 2009, 84, 1364–1370. [Google Scholar] [CrossRef]
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Giwa, S.; Abdullah, L.C.; Adam, N.M. Investigating “Egusi” (Citrullus Colocynthis L.) Seed Oil as Potential Biodiesel Feedstock. Energies 2010, 3, 607-618. https://doi.org/10.3390/en3040607
Giwa S, Abdullah LC, Adam NM. Investigating “Egusi” (Citrullus Colocynthis L.) Seed Oil as Potential Biodiesel Feedstock. Energies. 2010; 3(4):607-618. https://doi.org/10.3390/en3040607
Chicago/Turabian StyleGiwa, Solomon, Luqman Chuah Abdullah, and Nor Mariah Adam. 2010. "Investigating “Egusi” (Citrullus Colocynthis L.) Seed Oil as Potential Biodiesel Feedstock" Energies 3, no. 4: 607-618. https://doi.org/10.3390/en3040607
APA StyleGiwa, S., Abdullah, L. C., & Adam, N. M. (2010). Investigating “Egusi” (Citrullus Colocynthis L.) Seed Oil as Potential Biodiesel Feedstock. Energies, 3(4), 607-618. https://doi.org/10.3390/en3040607




