Progress and Prospects of Air Water Harvesting System for Remote Areas: A Comprehensive Review
Abstract
1. Introduction
Background
2. Materials and Methods
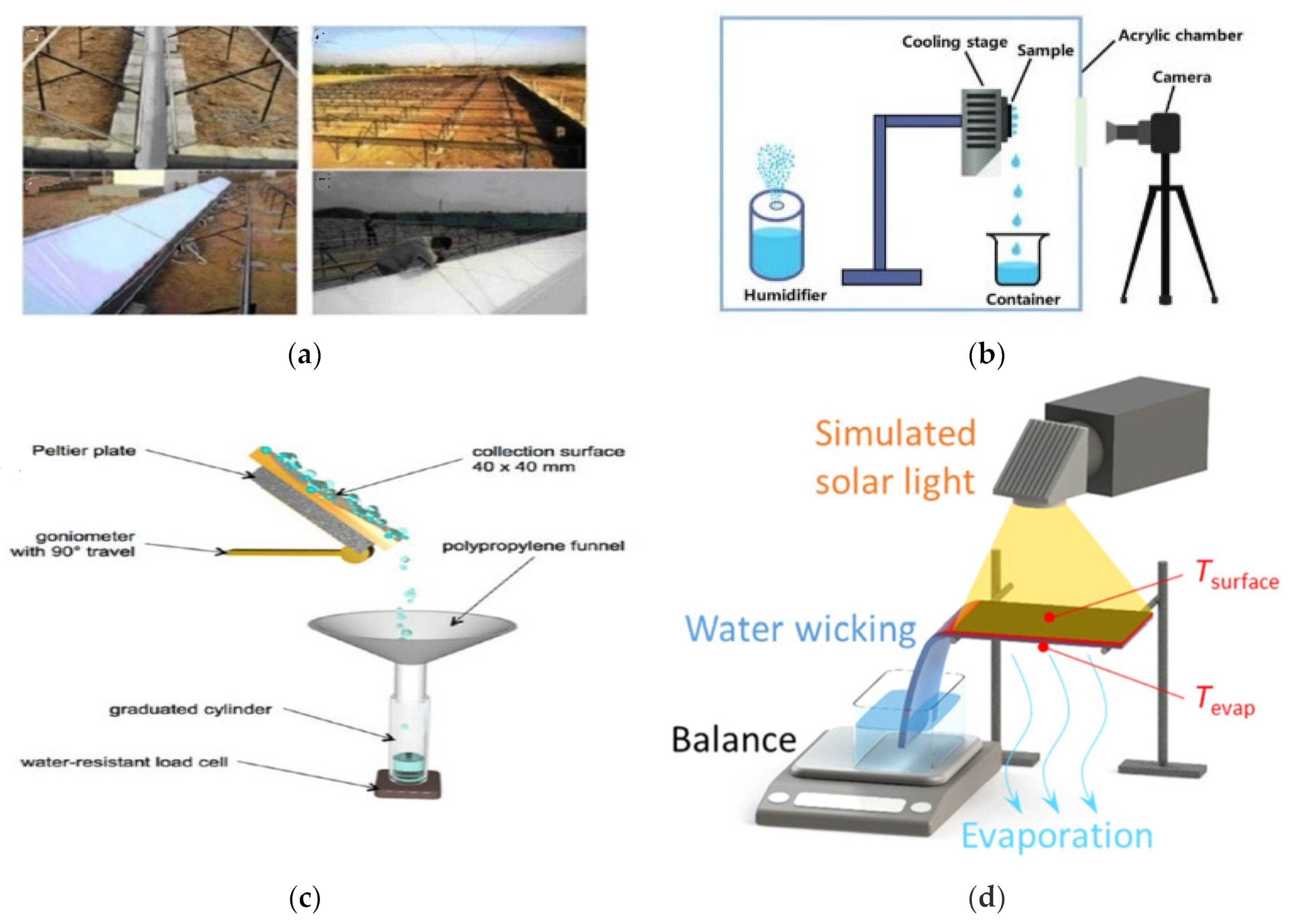
2.1. Dew Water Harvesting
| SN | Surface Material | Relative Humidity RH (%) | Production (L/m2 h) | Literature |
|---|---|---|---|---|
| 1 | Styrene foam panels (Figure 2a) | - | 0.6 | [27] |
| 2 | Epoxy/SiO2 grooves | - | 0.2 | [19] |
| 3 | Polymer flat surfaces | 100 | 0.2 | [28] |
| 4 | Patterned copper (Figure 2b) | 50 | 1.1 | [29] |
| 5 | Nanostructured Teflon AF and PTFE surfaces (Figure 2c) | 98 | 0.72 | [22] |
| 6 | Porous coating layer (Figure 2d) | 0.13 | [30] | |
| 7 | Rough Duralumin Alloy Plates | 70 | 0.15 | [31] |
| 8 | Needle-Shaped Black Silicon | 70 | 0.1–0.27 | [32] |
2.2. Fog Water Harvesting
- (1)
- There should be continuous fog throughout the year and the duration should be long.
- (2)
- High-attitude fogs and a high amount of water content are the major criteria for fog water harvesting in arid lands.
- (3)
- Fog collection is more efficient when the wind is present.
| SN | Material | Condition | Production | Literature |
|---|---|---|---|---|
| 1 | 35 LFCs | Winter dry season | 6300 L of water on average per day throughout the course of 4–6 months during the winter dry season. | [40] |
| 2 | Four LFCs were added in the 2000s, and four more in 2011. | - | - | [41] |
| 3 | 60 fog nets | 100 L of water each day, a reduction in water use of over 60%. | [42] | |
| 4 | Locally created collector for fog | 100 L/day | [43] | |
| 5 | SFC-fog collector | 6 L/day | [43] | |
| 6 | 35 large fog collectors Figure 3c | Winter | 200 L | [44] |

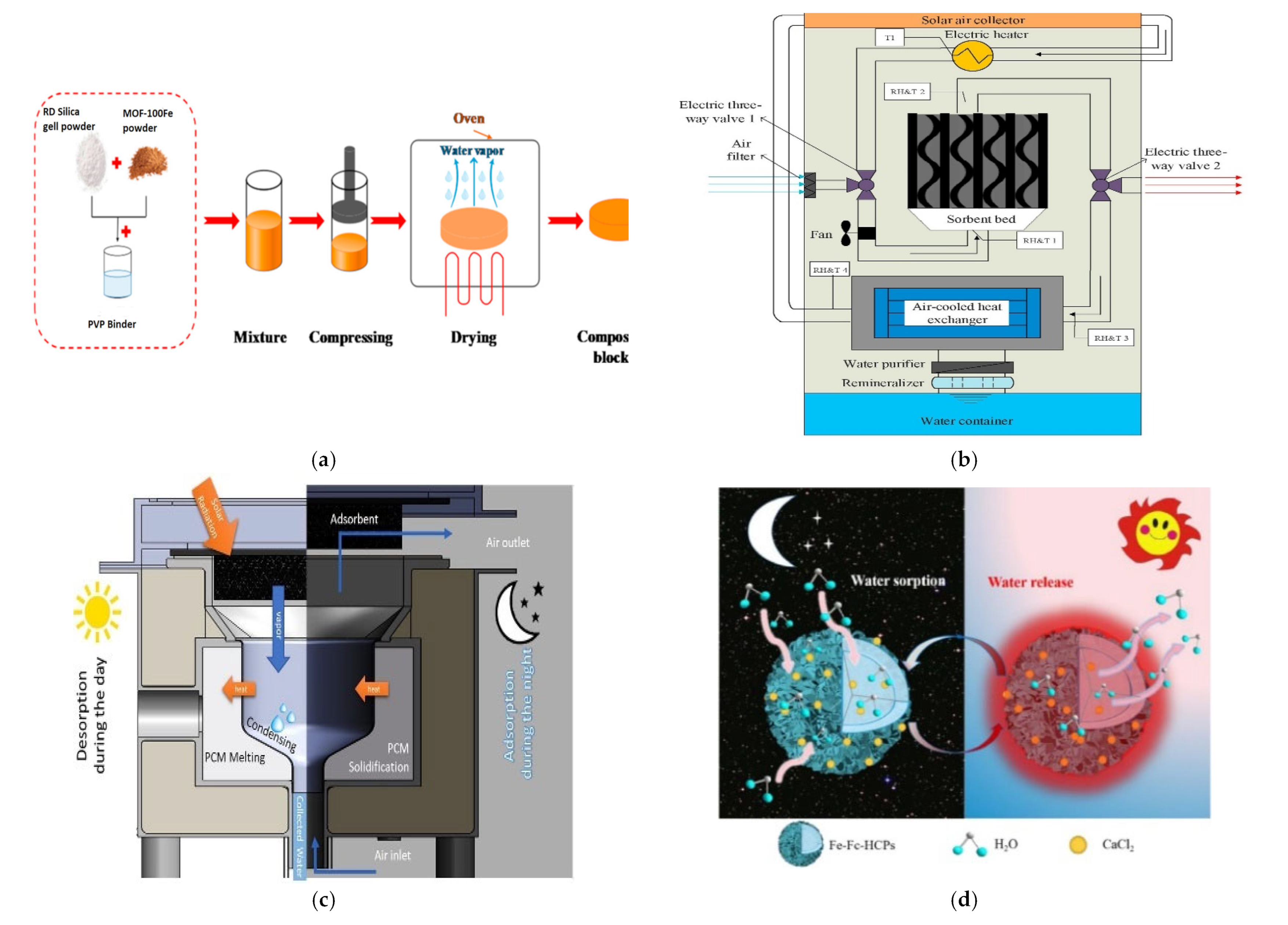
2.3. Sorption Based
2.3.1. Physical Adsorbents
2.3.2. Polymeric Adsorbents
2.3.3. Chemical Sorbent
| S No. | Composite Material | Conditions | Production | Literature |
|---|---|---|---|---|
| 1 | Silica gel-MIL 100(Fe) (Figure 4a) | Temperature 70 and 50 °C | - | [66] |
| 2 | MIL-100(Fe) | Temperature 423 K | 86.8 L/day. | [67] |
| 3 | P-SG and P-SG-L | Temperature 40 degree RH 10% | 1.70 g/g | [68] |
| 4 | carbon fibre felt (ACFF)–silica sol–LiCl3 (Figure 4b) | Temperature 31 degree RH 63% | 7.7 kg | [69] |
| 5 | MgSO4 and LiCl (Figure 4c) | RH 35% | 0.92 g/water/adsorbent | [70] |
| 6 | CaCl2 nanocrystals (Figure 4d) | 80% RH, 15 °C | 2.685 gH2O/gCaCl2 | [71] |
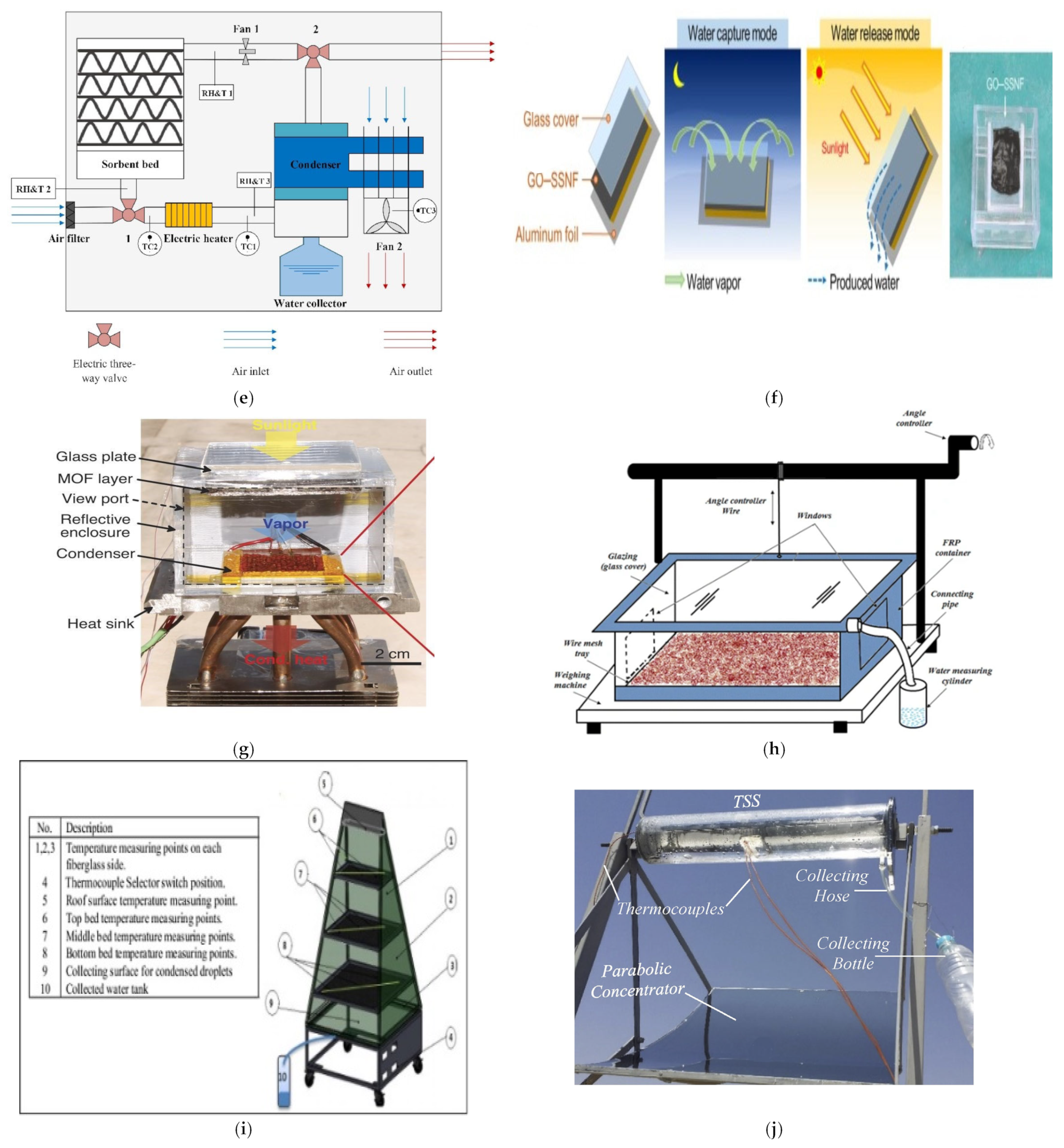
| 7 | ASLi30 (Figure 4e) | Temperature 51 degree | 0.42 kgwater/kg adsorbent | [72] |
| 8 | GO–SSNF (Figure 4f) | RH 30% | 0.96 g/g | [73] |
| 9 | MIL-125(Ti)_NH2 | Temperature range between 283–323 K | 320 L/day/ton. | [74] |
| 10 | Zr6O4(OH)4(fumarate)6 (Figure 4g) | RH 20% | 2.8 L | [75] |
| 11 | Cr-soc-MOF-1 | RH 70% | 1.95 g/g | [76] |
| 12 | Aluminum Fumarate | Temperature 25% | 0.33 g/g | [77] |
| 13 | AQSOA-Z01 zeolites | Temperature 298 K and 333 K | 0.23 g/g | [78] |
| 14 | AlPO4-LTA | Temperature 10 to 15 degree | 0.37 g/g | [57] |
| 15 | Calciumchloride + vermiculite- saw wood (Figure 4h) | - | 195 mL/kg/day | [79] |
| 16 | Calcium chloride and cloth (Figure 4i) | - | 3.02 L/day/m2 | [80] |
| 17 | Calcium chloride and black cloth | 12%RH | Minimum 230 mL/m2/day | [81] |
| 18 | 30% Calcium chloride and black cloth (Figure 4j) | RH 16% | 0.51 L/kg | [82] |
| 19 | LITHIUM BROMIDE ANHYDROUS (LiBr) (Figure 4k) | - | 73 mL/day | [83] |
| 20 | Calcium chloride and black cloth (Figure 4l) | - | 272–750 g/day | [84] |
| 21 | Calcium chloride and cloth (Figure 4m) | - | 0.3295 kg/m2/day to 0.6310 kg/m2/day | [85] |
| 22 | carbon felt + LiCl (Figure 4n) | 85% | 14.7 kg | [86] |
| 23 | Layered structure MnO2 (Figure 4o) | RH 23% Dew point temperature 11 °C. | 0.07 kgwater/kgsorbant | [87] |
| 24 | Functionalized carbon nanotube (FCNT) | RH 40% | 5.6 gwater/gsorbent | [88] |
| 25 | LiCl is contained inside a hollow nanocarbon capsule’s void core. (Figure 4p) | RH 60% | 1.6 kgwater/kgsorbant | [89] |
| 26 | ACF (LiCl, CaCl2, and LiNO3) (Figure 4q) | Temperature 25 °C RH 20% | 1.18 gwater/gsorbant-LiCl | [90] |
| 28 | PDMAPS (Figure 4r) | Temperature 65 °C | 28 g | [91] |
| 29 | Alginate-chained hydrogel that has been altered using binary salts | - | 5.6 g | [88] |
| 30 | pyramidal solar still for CaCl2 (4-shelves) (Figure 4s) | Temperature 15 degree | 2-1 L | [92] |
| 31 | CaCl2 single-basin solar still (Figure 4t) | RH 40% | 1.0 L/m2/day | [93] |
| 32 | CaCl2 ACF sorbent (Figure 4u) | 70–80 °C | 0.32 kg water, 2.25 kgACFCA, and a surface area of 0.77 m2 | [94] |
| 33 | [ZrO4(OH)2(fumarate)6] air-cooled sorbent-based device- (MOF)-801(Figure 4v) | RH 40% | 0.25 L | [95] |
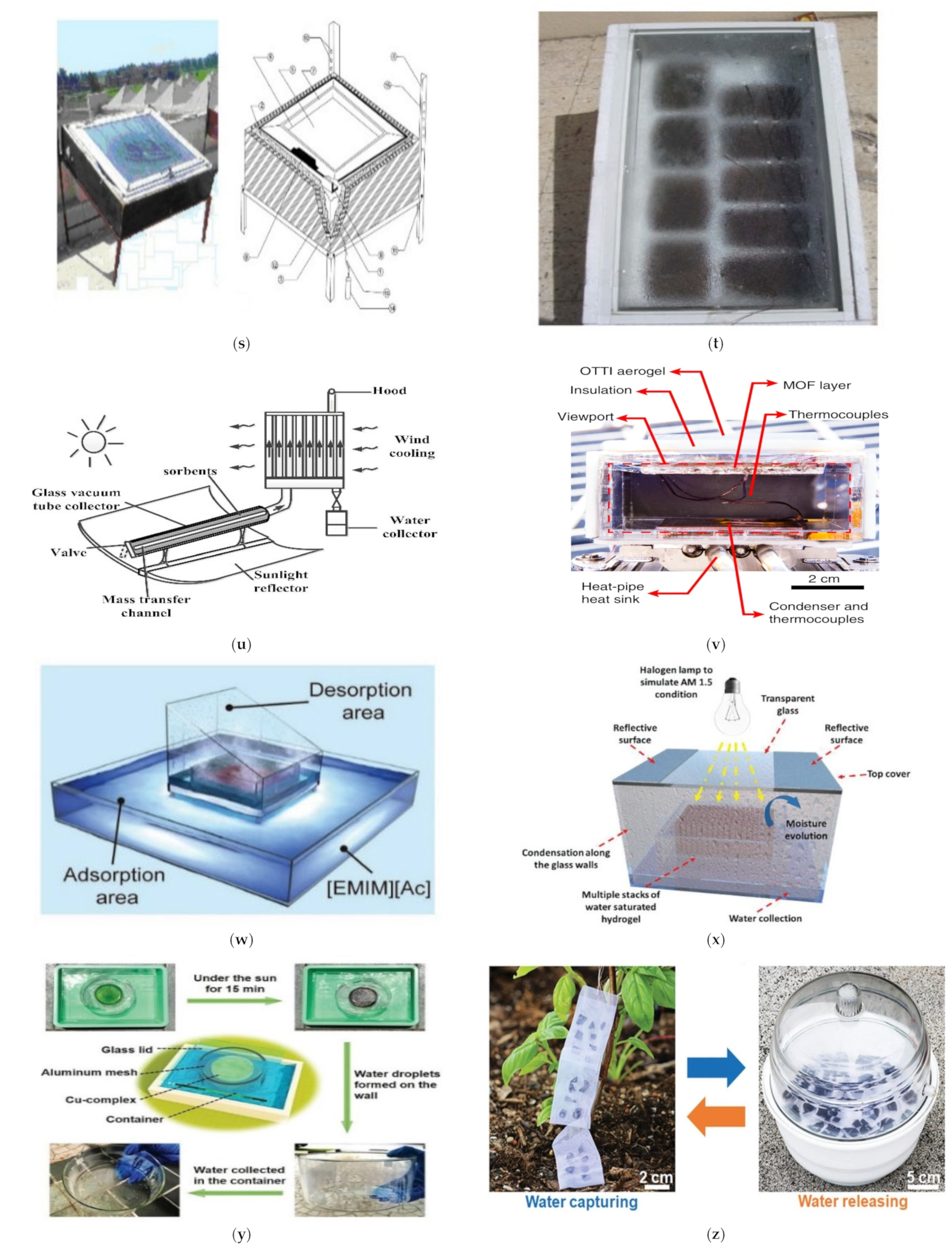
| 34 | Adsorption-Desorption in Liquid Sorbent (Figure 4w) | RH 80% | 2.8 L/m2/day | [57] |
| 35 | ALPO4-LTA | 15% RH | 0.37 g/g | [57] |
| 36 | COF-432 | At 30 °C (40% relative humidity, RH) and 35 °C (30% RH), respectively, adsorption and desorption are carried out. | 0.33 g/g | [96] |
| 37 | Cr-soc-MOF-1 | T 25 °C, 70% RH | 1.95 g/g | [76] |
| 38 | A hydrogel that is super hygroscopic (Figure 4x) | T 25 °C, 90% RH | 4.20 g/g | [97] |
| 39 | Cu (II)-ethanolamine hydrogel (Figure 4y) | T 25 °C, 30% RH | 0.14 g/g | [98] |
| 40 | Co-SHM | T 25 °C, 30% RH | 0.11 g/g | [99] |
| 41 | IPN- interpenetrating polymer network | T 25 °C, 30% RH | 0.18 g/g | [100] |
| 42 | SMAG (Figure 4z) | T 25 °C, 30% RH | 0.70 g/g | [101] |
| 43 | PC-MOF (Figure 4Aa) | T 25 °C, 30% RH | 0.76 g/g | [102] |
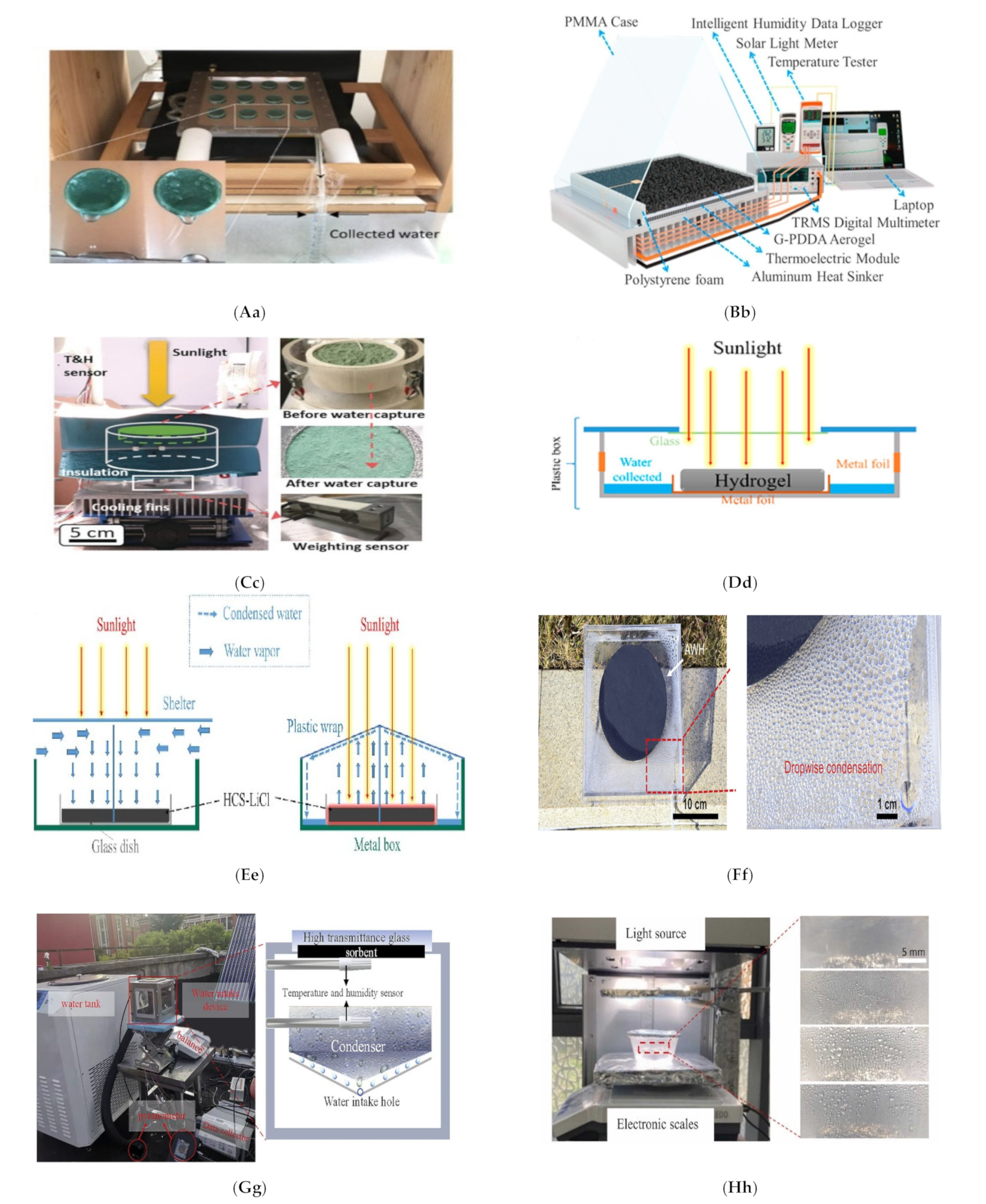
| 44 | G-PDDA (Figure 4Bb) | T 25 °C, 30% RH | 0.13 g/g | [103] |
| 45 | LiCl@MIL-101(Cr)_51 (Figure 4Cc) | T 30 °C, 30% RH | 0.82 g/g | [104] |
| 46 | LiCl@HS-200 | T 25 °C, 30% RH | 0.76 g/g | [105] |
| 47 | PAM-CNT-CaCl2 (Figure 4Dd) | T 25 °C, 35% RH | 0.69 g/g | [106] |
| 48 | HCS-LiCl (Figure 4Ee) | T 25 °C, 35% RH | 0.69 g/g | [89] |
| 49 | Alg-CaCl | T 28 °C, 33% RH | 1.18 g/g | [48,49] |
| 50 | ACFP30 | T 25 °C, 70% RH | 1.62 g/g | [107] |
| 51 | saturated LiBr solution | T 25 °C, 30% RH | 0.34 g/g | [108] |
| 52 | saturated LiCl solution | T 25 °C, 60% RH | 1.79 g/g | [105] |
| 53 | [Pyrrol][Ac] [DEA][Fo] [DEA][Ac] | T 29 °C, 57% RH T 29 °C, 57% RH T 29 °C, 57% RH | 0.45 g/g 0.4945 g/g 0.6545 g/g | [109] |
| 54 | Effective atmospheric water collection with deliquescent sorbents requires heterogeneous wettability and radiative cooling. (Figure 4Ff) | 60% RH | 2.62 g/g/day | [110] |
| 55 | PCLG (Figure 4Gg) | RH 55% | 2.9 L/m2 | [111] |
| 56 | ILCA (Figure 4Hh) | 23 °C and 50% RH | 2.791 g | [112] |
| 57 | multicyclic sorption | T 51 °C | 0.42 kgwater/kgsorbant | [72] |
| 58 | Three-phase sorption | RH 70% | 3.18 g | [90] |
| 59 | CoCl2 (BTDD) | RH 30% | 0.82 gH2O/gMOF | [54,55] |
| 60 | Universal scalable sorption-base (Figure 4Ii) | RH 75% | 38.5 kg/day | [113] |
| 61 | Hygroscopic nanostructured biopolymer aerogels | T 26.8–36.3 °C | 88.3 g | [114] |
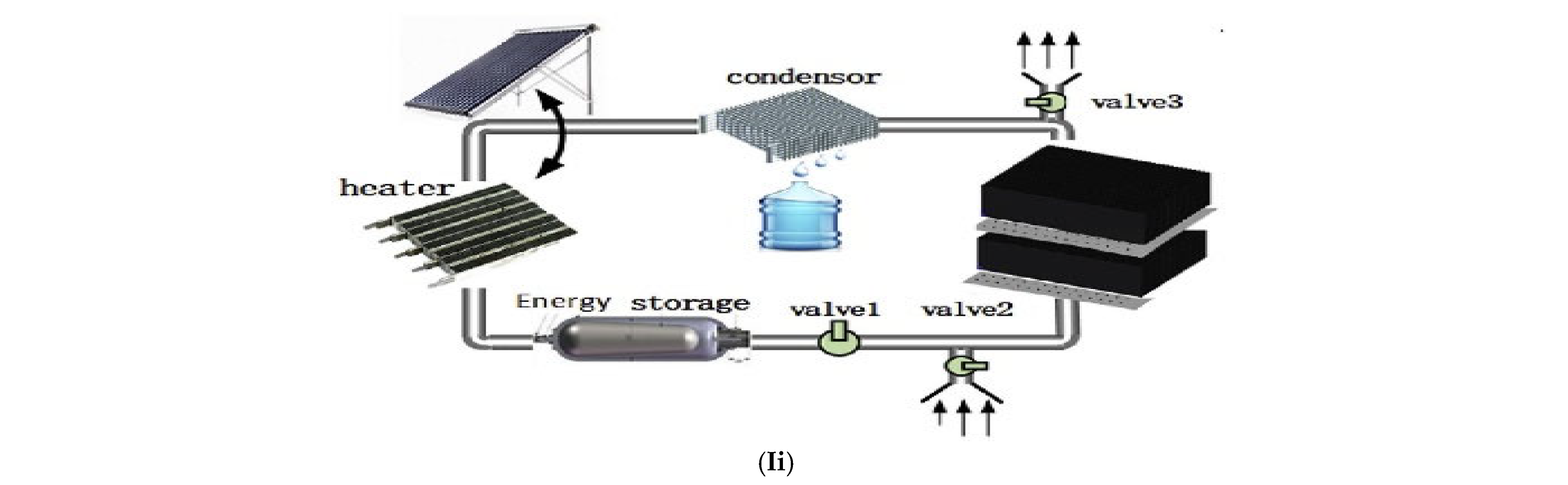
2.4. Condensation Based
2.4.1. Condensation-Based VCC (Vapor Compression Cycle) Technologies
2.4.2. Condensation Technologies Using Thermo-Electric Cooler (TEC)
| SN | System Information | Condition | Production | Reference |
|---|---|---|---|---|
| 1 | The rated air flow rate is 578 m3 h−1, and the VCC-Rated compressor power is 1035 W. | dry bulb wet 26.7 °C wet bulb temperature: 19.4 °C | 1.5 kg/h | [124] |
| 2 | VCC- Uses R22, weighs 39 kg, and has a rated compressor output of 370 W in addition to 1.5 kW of rated cooling capacity. | ambient condition: 35 °C 20–40% RH | 0.13 kg/h | [125] |
| 3 | VCC-AFR ranges between 300 and 1000 m3 per hour. | - | 1.5 kg/h to 4.2 kg/h. | [118] |
| 4 | VCC | Velocity 2–5 ms−1 | 22-26 L/day | [120] |
| 5 | HVAC system (Figure 5a) | RH = 60%, Temperature= 35 °C | 425 L/h | [124,126] |
| 6 | HVAC | Temperature = 25~50 °C, RH = 15~90%, | 2.33 L/h | [117] |
| 7 | HVAC condensate recovery (Figure 5b) | T = 27.3 °C RH = 86% | 78.3 L/h | [127] |
| 8 | TEC (portable water generator) | T = 22 °C, RH = 100% | 24 mL/h | [128] |
| 9 | TEC (Figure 5c) | T = 24.29 °C, RH = 67.8%, | 20 mL/h | [129] |
| 10 | TEC (solar energy is used to power water collection) | T = 24~31 °C, RH = 60~80%, | 20 mL/h | [130] |
| 11 | TEC (thermoelectric dehumidifier system for air ducts) | T = 35 °C, RH = 60–90% | 0.82 L/h | [131] |
| 12 | VCC-Frontal AFR is around 650 m3 h−1 and area is 0.04 m2 (Figure 5d) | - | 0.92 kg/h to 1.08 kg/h. | [117] |
3. Performance Assessment Indicators
- temperature of the cooling coil.
- temperature of the air flow into the cooling coil.
- heat transfer coefficient for dry air.
- A surface area of the cooling coil.
- outdoor dry bulb temperature.
- m mass flow rate of the air through the cooling coil.
- Cp specific heat capacity of the air.
4. Parameters of Atmospheric Water Vapor
- ➢
- Relative humidity (Φ)
- ➢
- Absolute humidity (ω)
- ➢
- Dew-point temperature (Td)
- Φ is the relative humidity.
- Pw is the partial pressure of water vapor in the air.
- Ps is the saturation vapor pressure of water.
- Dry air—Ha
- Water vapor—Hwv
- Heat capacity—Cp,a
5. Conclusions
Funding
Conflicts of Interest
Nomenclature
| AWH | Atmospheric water harvesting |
| HVAC | heating, ventilation, and air conditioning |
| MOF | metal organic frame |
| SVG | Solar vapor generators |
| TEC | Thermo-Electric Cooler |
| UNCCD | United Nations Convention to Combat Desertification |
| UN | United Nations |
| VCC | vapor compression cycle |
| WHR | water harvesting rate |
| Subscripts | |
| temperature of the cooling coil | |
| temperature of air flow into the cooling coil | |
| heat transfer coefficient for dry air | |
| A | surface area of the cooling coil |
| outdoor dry bulb temperature | |
| m | mass flow rate of air through the cooling coil |
| Cp | specific heat capacity of air |
| Qc | heat transported from the cold side |
| Tin | air’s inlet temperature (C) |
| Tout | outflow air’s temperature |
| hwet | enthalpy of moist air |
| ℎDry | enthalpy of dry air |
| C | specific humidity ratio of the air |
| hsurface | Enthalpy of the surface of the evaporator (J/kg) |
| hert | Enthalpy of the refrigerant flow into the evaporator (J/kg) |
| hin | Enthalpy of the refrigerant entering the condenser (J/kg) |
| hwet | Enthalpy of the air at the evaporator inlet (J/kg) |
| A | Area of the heat transfer surface in m2 |
| ṁ | Mass flow rate of air (kg/s) |
| Cp | Specific heat capacity of air at constant pressure (J/(kg.K)) |
| e | Effectiveness of the heat exchanger |
| Qc | Cooling capacity in watts (W) |
| ṁ air | Mass flow rate of air in kilograms per second (kg/s) |
| hin | Enthalpy of the air entering the cooling coil in joules per kilogram (J/kg) |
| hout | Enthalpy of the air leaving the cooling coil in joules per kilogram (J/kg) |
| win | Humidity ratio of the air entering the cooling coil in kilograms of water vapor per kilogram of dry air (kg/kg) |
| wout | Humidity ratio of the air leaving the cooling coil in kilograms of water vapor per kilogram of dry air (kg/kg) |
| Hf | Latent heat of vaporization of water at the coil temperature in joules per kilogram (J/kg) |
| Qh | Heating capacity in watts (W) |
| hh | Enthalpy of the hot fluidin joules per kilogram (J/kg) |
| Th | Temperature of the hot fluid (°C) |
| Tamb | Ambient temperature (°C) |
| amount of water produced | |
| intake air’s absolute humidity | |
| output air’s absolute humidity, and t for the passage of time (s) | |
| Φ | Relative humidity |
| ω | Absolute humidity |
| Td | Dew-point temperature |
| Pw | partial pressure of water vapor in the air |
| Ps | saturation vapor pressure of water |
| P | total air pressure |
References
- Salehi, M. Global Water Shortage and Potable Water Safety; Today’s Concern and Tomorrow’s Crisis. Env. Int. 2022, 158, 106936. [Google Scholar] [CrossRef] [PubMed]
- Lund Schlamovitz, J.; Becker, P. Differentiated Vulnerabilities and Capacities for Adaptation to Water Shortage in Gaborone, Botswana. Int. J. Water Resour. Dev. 2020, 37, 278–299. [Google Scholar] [CrossRef]
- Water Facts—Worldwide Water Supply. ARWEC|CCAO|Area Offices|California-Great Basin|Bureau of Reclamation. Available online: https://www.usbr.gov/mp/arwec/water-facts-ww-water-sup.html (accessed on 27 September 2022).
- Al-Shalabi, H.; Andraws, S.; Alrabea, A.I.; Kumar, A.V.S. V Model of E-Learning Using Gagne Nine Steps of Education. J. Softw. Eng. Appl. 2012, 5, 850–854. [Google Scholar] [CrossRef]
- Progress on Household Drinking Water, Sanitation and Hygiene, 2000–2017—UNICEF DATA. Available online: https://data.unicef.org/resources/progress-drinking-water-sanitation-hygiene-2019/ (accessed on 4 May 2022).
- Impacts of Water Scarcity on the Social Welfare of Citizens in the Middle East. Middle East Institute. Available online: https://www.mei.edu/publications/impacts-water-scarcity-social-welfare-citizens-middle-east (accessed on 10 May 2022).
- UNCCD. Desertification the Invisible Frontline; UNCCD: Bonn, Germany, 2014. [Google Scholar]
- Zhu, X.; Zhang, C.; Yin, J.; Zhou, H. Optimization of Water Diversion Based on Reservoir Operating Rules: Analysis of the Biliu River Reservoir, China. J. Hydrol. Eng. 2014, 19, 411–421. [Google Scholar] [CrossRef]
- Al-Sahlawi, M.A. Seawater Desalination in Saudi Arabia: Economic Review and Demand Projections. In Desalination; Elsevier: Amsterdam, The Netherlands, 1999; Volume 123, pp. 143–147. [Google Scholar]
- Rodriguez, C.A.; Flessa, K.W.; Dettman, D.L. Effects of Upstream Diversion of Colorado River Water on the Estuarine Bivalve Mollusc Mulinia Coloradoensis. Conserv. Biol. 2001, 15, 249–258. [Google Scholar] [CrossRef]
- Micale, G.; Cipollina, A.; Rizzuti, L. Seawater Desalination for Freshwater Production. In Green Energy and Technology; Springer Science and Business Media Deutschland GmbH: Berlin/Heidelberg, Germany, 2009; pp. 1–15. [Google Scholar]
- Karagiannis, I.C.; Soldatos, P.G. Water Desalination Cost Literature: Review and Assessment. Desalination 2008, 223, 448–456. [Google Scholar] [CrossRef]
- Wahlgren, R.V. Atmospheric Water Vapour Processor Designs for Potable Water Production: A Review. Water Res. 2001, 35, 101–108. [Google Scholar] [CrossRef]
- Hao, L.; Liu, N.; Bai, H.; He, P.; Niu, R.; Gong, J. High-Performance Solar-Driven Interfacial Evaporation through Molecular Design of Antibacterial, Biomass-Derived Hydrogels. J. Colloid Interface Sci. 2022, 608, 840–852. [Google Scholar] [CrossRef]
- Li, R.; Wu, M.; Shi, Y.; Aleid, S.; Wang, W.; Zhang, C.; Wang, P. Hybrid Water Vapor Sorbent Design with Pollution Shielding Properties: Extracting Clean Water from Polluted Bulk Water Sources. J. Mater. Chem. A Mater. 2021, 9, 14731–14740. [Google Scholar] [CrossRef]
- Sultan Irshad, M.; Wang, X.; Sehar Abbasi, M.; Arshad, N.; Chen, Z.; Guo, Z.; Yu, L.; Qian, J.; You, J.; Mei, T. Semiconductive, Flexible MnO2 NWs/Chitosan Hydrogels for Efficient Solar Steam Generation. ACS Publ. 2021, 9, 3887–3900. [Google Scholar] [CrossRef]
- Ju, J.; Bai, H.; Zheng, Y.; Zhao, T.; Fang, R.; Jiang, L. A Multi-Structural and Multi-Functional Integrated Fog Collection System in Cactus. Nat. Commun. 2012, 3, 1247. [Google Scholar] [CrossRef] [PubMed]
- Gido, B.; Friedler, E.; Broday, D.M. Assessment of Atmospheric Moisture Harvesting by Direct Cooling. Atmos Res. 2016, 182, 156–162. [Google Scholar] [CrossRef]
- Bintein, P.B.; Lhuissier, H.; Mongruel, A.; Royon, L.; Beysens, D. Grooves Accelerate Dew Shedding. Phys. Rev. Lett. 2019, 122, 098005. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.Y.; Liu, Y.; Jiang, H.B.; Li, S.Y.; Kaya, C.; Stegmaier, T.; Han, Z.W.; Ren, L.Q. A Bioinspired Structured Graphene Surface with Tunable Wetting and High Wearable Properties for Efficient Fog Collection. Nanoscale 2018, 10, 16127–16137. [Google Scholar] [CrossRef] [PubMed]
- Yaghi, O.M. Research UC Berkeley. Available online: https://vcresearch.berkeley.edu/faculty/omar-m-yaghi (accessed on 29 September 2022).
- Gerasopoulos, K.; Luedeman, W.L.; Ölçeroglu, E.; McCarthy, M.; Benkoski, J.J. Effects of Engineered Wettability on the Efficiency of Dew Collection. ACS Appl. Mater. Interfaces 2018, 10, 4066–4076. [Google Scholar] [CrossRef]
- Water Abundance XPRIZE | XPRIZE Foundation. Available online: https://www.xprize.org/prizes/water-abundance (accessed on 5 September 2022).
- Watergen|Water from Air. Available online: https://www.watergen.com/ (accessed on 5 September 2022).
- Khalil, B.; Adamowski, J.; Shabbir, A.; Jang, C.; Rojas, M.; Reilly, K.; Ozga-Zielinski, B. A Review: Dew Water Collection from Radiative Passive Collectors to Recent Developments of Active Collectors. Sustain. Water Resour. Manag. 2016, 2, 71–86. [Google Scholar] [CrossRef]
- LaPotin, A.; Kim, H.; Rao, S.R.; Wang, E.N. Adsorption-Based Atmospheric Water Harvesting: Impact of Material and Component Properties on System-Level Performance. Acc. Chem. Res. 2019, 52, 1588–1597. [Google Scholar] [CrossRef]
- Sharan, G.; Roy, A.K.; Royon, L.; Mongruel, A.; Beysens, D. Dew Plant for Bottling Water. J. Clean Prod. 2017, 155, 83–92. [Google Scholar] [CrossRef]
- Jin, Y.; Zhang, L.; Wang, P.; Jin, Y.; Wang, P.; Zhang, L. Atmospheric Water Harvesting: Role of Surface Wettability and Edge Effect. Glob. Chall. 2017, 1, 1700019. [Google Scholar] [CrossRef]
- Hou, K.; Li, X.; Li, Q.; Chen, X. Tunable Wetting Patterns on Superhydrophilic/Superhydrophobic Hybrid Surfaces for Enhanced Dew-Harvesting Efficacy. Adv. Mater. Interfaces 2020, 7, 1901683. [Google Scholar] [CrossRef]
- Xu, J.; Zhang, J.; Fu, B.; Song, C.; Shang, W.; Tao, P.; Deng, T. All-Day Freshwater Harvesting through Combined Solar-Driven Interfacial Desalination and Passive Radiative Cooling. ACS Appl. Mater. Interfaces 2020, 12, 47612–47622. [Google Scholar] [CrossRef] [PubMed]
- Trosseille, J.; Mongruel, A.; Royon, L.; Medici, M.G.; Beysens, D. Roughness-Enhanced Collection of Condensed Droplets. Eur. Phys. J. 2019, 42, 144. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Trosseille, J.; Mongruel, A.; Marty, F.; Basset, P.; Laurent, J.; Royon, L.; Cui, T.; Beysens, D.; Bourouina, T. Tailoring Silicon for Dew Water Harvesting Panels. iScience 2021, 24, 102814. [Google Scholar] [CrossRef]
- Gultepe, I.; Tardif, R.; Michaelides, S.C.; Cermak, J.; Bott, A.; Bendix, J.; Müller, M.D.; Pagowski, M.; Hansen, B.; Ellrod, G.; et al. Fog Research: A Review of Past Achievements and Future Perspectives. Pure Appl. Geophys. 2007, 164, 1121–1159. [Google Scholar] [CrossRef]
- Klemm, O.; Schemenauer, R.S.; Lummerich, A.; Cereceda, P.; Marzol, V.; Corell, D.; van Heerden, J.; Reinhard, D.; Gherezghiher, T.; Olivier, J.; et al. Fog as a Fresh-Water Resource: Overview and Perspectives. Ambio 2012, 41, 221–234. [Google Scholar] [CrossRef] [PubMed]
- Batisha, A.F. Feasibility and Sustainability of Fog Harvesting. Sustain. Water Qual. Ecol. 2015, 6, 1–10. [Google Scholar] [CrossRef]
- Montecinos, S.; Carvajal, D.; Cereceda, P.; Concha, M. Collection Efficiency of Fog Events. Atmos. Res. 2018, 209, 163–169. [Google Scholar] [CrossRef]
- Abdul-Wahab, S.A.; Al-Hinai, H.; Al-Najar, K.A.; Al-Kalbani, M.S. Feasibility of Fog Water Collection: A Case Study from Oman. J. Water Supply Res. Technol. Aqua 2007, 56, 275–280. [Google Scholar] [CrossRef]
- Schemenauer, R.S.; Cereceda, P.; Osses, P. The complementary aspects of projects to collect rain, fog and dew. In Proceedings of the XI th International Conference on Rainwater Catchment System, Mexico City, Mexico, 25–29 August 2003. [Google Scholar]
- Tu, Y.; Wang, R.; Zhang, Y.; Wang, J. Progress and Expectation of Atmospheric Water Harvesting. Joule 2018, 2, 1452–1475. [Google Scholar] [CrossRef]
- Hu, Y.; Fang, Z.; Wan, X.; Ma, X.; Wang, Y.; Dong, M.; Ye, Z.; Peng, X. Ferrocene Dicarboxylic Acid Ligand-Exchanged Hollow MIL-101(Cr) Nanospheres for Solar-Driven Atmospheric Water Harvesting. ACS Sustain. Chem. Eng. 2022, 10, 6446–6455. [Google Scholar] [CrossRef]
- Marzol Jaén, M.V. Fog Water Collection in a Rural Park in the Canary Islands (Spain). Atmos. Res. 2002, 64, 239–250. [Google Scholar] [CrossRef]
- The Fog Harvesters of Lima, Peru–NaturPhilosophie. Available online: https://www.naturphilosophie.co.uk/fog-harvesters-lima-peru/ (accessed on 4 July 2022).
- Larrain, H.; Velásquez, F.; Cereceda, P.; Espejo, R.; Pinto, R.; Osses, P.; Schemenauer, R.S. Fog Measurements at the Site “Falda Verde” North of Chañaral Compared with Other Fog Stations of Chile. Atmos. Res. 2002, 64, 273–284. [Google Scholar] [CrossRef]
- Latest News. FogQuest: Sustainable Water Solutions. Fog Collection|Rainwater Collection|Rural Water Projects. Available online: http://www.fogquest.org/latest-news/ (accessed on 21 September 2022).
- Butt, H.-J.; Graf, K.; Kappl, M. Physics and Chemistry of Interfaces; Wiley-VCH: Weinheim, Germany, 2003. [Google Scholar]
- Shan, H.; Pan, Q.; Xiang, C.; Poredoš, P.; Ma, Q.; Ye, Z.; Hou, G.; Wang, R. High-Yield Solar-Driven Atmospheric Water Harvesting with Ultra-High Salt Content Composites Encapsulated in Porous Membrane. Cell Rep. Phys. Sci. 2021, 2, 100664. [Google Scholar] [CrossRef]
- Kalmutzki, M.J.; Diercks, C.S.; Yaghi, O.M. Metal–Organic Frameworks for Water Harvesting from Air. Adv. Mater. 2018, 30, 1704304. [Google Scholar] [CrossRef] [PubMed]
- Jia, C.X.; Dai, Y.J.; Wu, J.Y.; Wang, R.Z. Experimental Comparison of Two Honeycombed Desiccant Wheels Fabricated with Silica Gel and Composite Desiccant Material. Energy Convers. Manag. 2006, 47, 2523–2534. [Google Scholar] [CrossRef]
- Kallenberger, P.A.; Fröba, M. Water Harvesting from Air with a Hygroscopic Salt in a Hydrogel–Derived Matrix. Commun. Chem. 2018, 1, 28. [Google Scholar] [CrossRef]
- Wang, W.; Wu, L.; Li, Z.; Fang, Y.; Ding, J.; Xiao, J. An Overview of Adsorbents in the Rotary Desiccant Dehumidifier for Air Dehumidification. Dry. Technol. 2013, 31, 1334–1345. [Google Scholar] [CrossRef]
- La, D.; Dai, Y.; Li, Y.; Ge, T.; Wang, R. Case Study and Theoretical Analysis of a Solar Driven Two-Stage Rotary Desiccant Cooling System Assisted by Vapor Compression Air-Conditioning. Sol. Energy 2011, 85, 2997–3009. [Google Scholar] [CrossRef]
- Gordeeva, L.G.; Tokarev, M.M.; Parmon, V.N.; Aristov, Y.I. Selective Water Sorbents for Multiple Application, 6. Freshwater Production from the Atmosphere. React. Kinet. Catal. Lett. 1998, 65, 153–159. [Google Scholar] [CrossRef]
- Wang, J.Y.; Liu, J.Y.; Wang, R.Z.; Wang, L.W. Experimental Research of Composite Solid Sorbents for Fresh Water Production Driven by Solar Energy. Appl. Eng. 2017, 121, 941–950. [Google Scholar] [CrossRef]
- Rieth, A.J.; Yang, S.; Wang, E.N.; Dincǎ, M. Record Atmospheric Fresh Water Capture and Heat Transfer with a Material Operating at the Water Uptake Reversibility Limit. ACS Cent. Sci. 2017, 3, 668–672. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Rao, S.R.; Kapustin, E.A.; Narayanan, S.; Yang, S.; Furukawa, H.; Umans, A.S.; Yaghi, O.M.; Wang, E.N. Response to Comment on “Water Harvesting from Air with Metal-Organic Frameworks Powered by Natural Sunlight. Science 2017, 358, eaao3139. [Google Scholar] [CrossRef]
- Heidari, A.; Roshandel, R.; Vakiloroaya, V. An Innovative Solar Assisted Desiccant-Based Evaporative Cooling System for Co-Production of Water and Cooling in Hot and Humid Climates. Energy Convers. Manag. 2019, 185, 396–409. [Google Scholar] [CrossRef]
- Krajnc, A.; Varlec, J.; Mazaj, M.; Ristić, A.; Logar, N.Z.; Mali, G. Superior Performance of Microporous Aluminophosphate with LTA Topology in Solar-Energy Storage and Heat Reallocation. Adv. Energy Mater. 2017, 7, 1601815. [Google Scholar] [CrossRef]
- Furukawa, H.; Gándara, F.; Zhang, Y.B.; Jiang, J.; Queen, W.L.; Hudson, M.R.; Yaghi, O.M. Water Adsorption in Porous Metal-Organic Frameworks and Related Materials. J. Am. Chem. Soc. 2014, 136, 4369–4381. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.; Yaghi, O.M. Metal-Organic Frameworks for Water Harvesting from Air, Anywhere, Anytime. ACS Cent. Sci. 2020, 6, 1348–1354. [Google Scholar] [CrossRef] [PubMed]
- Fathieh, F.; Kalmutzki, M.J.; Kapustin, E.A.; Waller, P.J.; Yang, J.; Yaghi, O.M. Practical Water Production from Desert Air. Sci. Adv. 2018, 4, eaat3198. [Google Scholar] [CrossRef]
- Wang, X.; Li, X.; Liu, G.; Li, J.; Hu, X.; Xu, N.; Zhao, W.; Zhu, B.; Zhu, J. An Interfacial Solar Heating Assisted Liquid Sorbent Atmospheric Water Generator. Angew. Chem. Int. Ed. 2019, 58, 12054–12058. [Google Scholar] [CrossRef]
- Feng, A.; Akther, N.; Duan, X.; Peng, S.; Onggowarsito, C.; Mao, S.; Fu, Q.; Kolev, S.D. Recent Development of Atmospheric Water Harvesting Materials: A Review. ACS Mater. Au 2022, 2, 576–595. [Google Scholar] [CrossRef]
- Wang, R.; Wang, L.; Wu, J. Adsorption Refrigeration Technology: Theory and Application; John Wiley & Sons: New York, NY, USA, 2014; ISBN 9781118197479. [Google Scholar]
- Li, R.; Shi, Y.; Shi, L.; Alsaedi, M.; Wang, P. Harvesting Water from Air: Using Anhydrous Salt with Sunlight. Envon. Sci. Technol. 2018, 52, 5398–5406. [Google Scholar] [CrossRef]
- Salehi, A.A.; Ghannadi-Maragheh, M.; Torab-Mostaedi, M.; Torkaman, R.; Asadollahzadeh, M. A Review on the Water-Energy Nexus for Drinking Water Production from Humid Air. Renew. Sustain. Energy Rev. 2020, 120, 109627. [Google Scholar] [CrossRef]
- Maher, H.; Rupam, T.H.; Rocky, K.A.; Bassiouny, R.; Saha, B.B. Silica Gel-MIL 100(Fe) Composite Adsorbents for Ultra-Low Heat-Driven Atmospheric Water Harvester. Energy 2022, 238, 121741. [Google Scholar] [CrossRef]
- Silva, M.P.; Ribeiro, A.M.; Silva, C.G.; Ho Cho, K.; Lee, U.H.; Faria, J.L.; Loureiro, J.M.; Chang, J.S.; Rodrigues, A.E.; Ferreira, A. Atmospheric Water Harvesting on MIL-100(Fe) upon a Cyclic Adsorption Process. Sep. Purif. Technol. 2022, 290, 120803. [Google Scholar] [CrossRef]
- Ma, Q.; Zheng, X. Preparation and Characterization of Thermo-Responsive Composite for Adsorption-Based Dehumidification and Water Harvesting. Chem. Eng. J. 2022, 429, 132498. [Google Scholar] [CrossRef]
- Wang, W.; Xie, S.; Pan, Q.; Dai, Y.; Wang, R.; Ge, T. Air-Cooled Adsorption-Based Device for Harvesting Water from Island Air. Renew. Sustain. Energy Rev. 2021, 141, 110802. [Google Scholar] [CrossRef]
- Ejeian, M.; Entezari, A.; Wang, R.Z. Solar Powered Atmospheric Water Harvesting with Enhanced LiCl /MgSO4/ACF Composite. Appl. Eng. 2020, 176, 115396. [Google Scholar] [CrossRef]
- Hu, Y.; Fang, Z.; Ma, X.; Wan, X.; Wang, S.; Fan, S.; Ye, Z.; Peng, X. CaCl2 Nanocrystals Decorated Photothermal Fe-Ferrocene MOFs Hollow Microspheres for Atmospheric Water Harvesting. Appl. Mater. Today 2021, 23, 101076. [Google Scholar] [CrossRef]
- Wang, W.; Pan, Q.; Xing, Z.; Liu, X.; Dai, Y.; Wang, R.; Ge, T. Viability of a Practical Multicyclic Sorption-Based Water Harvester with Improved Water Yield. Water Res. 2022, 211, 118029. [Google Scholar] [CrossRef]
- Kim, S.; Liang, Y.; Kang, S.; Choi, H. Solar-Assisted Smart Nanofibrous Membranes for Atmospheric Water Harvesting. Chem. Eng. J. 2021, 425, 131601. [Google Scholar] [CrossRef]
- Silva, M.P.; Ribeiro, A.M.; Silva, C.G.; Narin, G.; Nogueira, I.B.R.; Lee, U.H.; Faria, J.L.; Loureiro, J.M.; Chang, J.S.; Rodrigues, A.E.; et al. Water Vapor Harvesting by a (P)TSA Process with MIL-125(Ti)_NH2 as Adsorbent. Sep. Purif. Technol. 2020, 237, 116336. [Google Scholar] [CrossRef]
- Kim, H.; Yang, S.; Rao, S.R.; Narayanan, S.; Kapustin, E.A.; Furukawa, H.; Umans, A.S.; Yaghi, O.M.; Wang, E.N. Water Harvesting from Air with Metal-Organic Frameworks Powered by Natural Sunlight. Science 2017, 356, 430–434. [Google Scholar] [CrossRef] [PubMed]
- Towsif Abtab, S.M.; Alezi, D.; Bhatt, P.M.; Shkurenko, A.; Belmabkhout, Y.; Aggarwal, H.; Weseliński, Ł.J.; Alsadun, N.; Samin, U.; Hedhili, M.N.; et al. Reticular Chemistry in Action: A Hydrolytically Stable MOF Capturing Twice Its Weight in Adsorbed Water. Chem 2018, 4, 94–105. [Google Scholar] [CrossRef]
- Teo, H.W.B.; Chakraborty, A.; Kitagawa, Y.; Kayal, S. Experimental Study of Isotherms and Kinetics for Adsorption of Water on Aluminium Fumarate. Int. J. Heat Mass. Transf. 2017, 114, 621–627. [Google Scholar] [CrossRef]
- Kayal, S.; Baichuan, S.; Saha, B.B. Adsorption Characteristics of AQSOA Zeolites and Water for Adsorption Chillers. Int. J. Heat Mass Transf. 2016, 92, 1120–1127. [Google Scholar] [CrossRef]
- Kumar, M.; Yadav, A. Composite Desiccant Material “CaCl2/Vermiculite/Saw Wood”: A New Material for Fresh Water Production from Atmospheric Air. Appl. Water Sci. 2017, 7, 2103–2111. [Google Scholar] [CrossRef]
- Mohamed, M.H.; William, G.E.; Fatouh, M. Solar Energy Utilization in Water Production from Humid Air. Sol. Energy 2017, 148, 98–109. [Google Scholar] [CrossRef]
- Elashmawy, M. Experimental Study on Water Extraction from Atmospheric Air Using Tubular Solar Still. J. Clean Prod. 2020, 249, 119322. [Google Scholar] [CrossRef]
- Elashmawy, M.; Alshammari, F. Atmospheric Water Harvesting from Low Humid Regions Using Tubular Solar Still Powered by a Parabolic Concentrator System. J. Clean Prod. 2020, 256, 120329. [Google Scholar] [CrossRef]
- Srivastava, S.; Yadav, A. Water Generation from Atmospheric Air by Using Composite Desiccant Material through Fixed Focus Concentrating Solar Thermal Power. Sol. Energy 2018, 169, 302–315. [Google Scholar] [CrossRef]
- Fathy, M.H.; Awad, M.M.; Zeidan, E.S.B.; Hamed, A.M. Solar Powered Foldable Apparatus for Extracting Water from Atmospheric Air. Renew. Energy 2020, 162, 1462–1489. [Google Scholar] [CrossRef]
- Talaat, M.A.; Awad, M.M.; Zeidan, E.B.; Hamed, A.M. Solar-Powered Portable Apparatus for Extracting Water from Air Using Desiccant Solution. Renew. Energy 2018, 119, 662–674. [Google Scholar] [CrossRef]
- Wang, J.Y.; Wang, R.Z.; Wang, L.W.; Liu, J.Y. A High Efficient Semi-Open System for Fresh Water Production from Atmosphere. Energy 2017, 138, 542–551. [Google Scholar] [CrossRef]
- Wang, J.; Dang, Y.; Meguerdichian, A.G.; Dissanayake, S.; Kankanam-Kapuge, T.; Bamonte, S.; Tobin, Z.M.; Achola, L.A.; Suib, S.L. Water Harvesting from the Atmosphere in Arid Areas with Manganese Dioxide. Environ. Sci. Technol. Lett. 2019, 7, 48–53. [Google Scholar] [CrossRef]
- Entezari, A.; Ejeian, M.; Wang, R. Super Atmospheric Water Harvesting Hydrogel with Alginate Chains Modified with Binary Salts. ACS Mater. Lett. 2020, 2, 471–477. [Google Scholar] [CrossRef]
- Li, R.; Shi, Y.; Wu, M.; Hong, S.; Wang, P. Improving Atmospheric Water Production Yield: Enabling Multiple Water Harvesting Cycles with Nano Sorbent. Nano Energy 2020, 67, 104255. [Google Scholar] [CrossRef]
- Entezari, A.; Ejeian, M.; Wang, R.Z. Extraordinary Air Water Harvesting Performance with Three Phase Sorption. Mater. Today Energy 2019, 13, 362–373. [Google Scholar] [CrossRef]
- Aleid, S.; Wu, M.; Li, R.; Wang, W.; Zhang, C.; Zhang, L.; Wang, P. Salting-in Effect of Zwitterionic Polymer Hydrogel Facilitates Atmospheric Water Harvesting. ACS Mater. Lett. 2022, 4, 511–520. [Google Scholar] [CrossRef]
- Kabeel, A.E. Application of Sandy Bed Solar Collector System for Water Extraction from Air. Int. J. Energy Res. 2006, 30, 381–394. [Google Scholar] [CrossRef]
- Hamed, A.M.; Aly, A.A.; Zeidan, E.-S.B. Application of Solar Energy for Recovery of Water from Atmospheric Air in Climatic Zones of Saudi Arabia. Nat. Resour. 2011, 2, 8–17. [Google Scholar] [CrossRef]
- Wang, J.Y.; Liu, J.Y.; Wang, R.Z.; Wang, L.W. Experimental Investigation on Two Solar-Driven Sorption Based Devices to Extract Fresh Water from Atmosphere. Appl. Eng. 2017, 127, 1608–1616. [Google Scholar] [CrossRef]
- Kim, H.; Rao, S.R.; Kapustin, E.A.; Zhao, L.; Yang, S.; Yaghi, O.M.; Wang, E.N. Adsorption-Based Atmospheric Water Harvesting Device for Arid Climates. Nat. Commun. 2018, 9, 1191. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, H.L.; Hanikel, N.; Lyle, S.J.; Zhu, C.; Proserpio, D.M.; Yaghi, O.M. A Porous Covalent Organic Framework with Voided Square Grid Topology for Atmospheric Water Harvesting. J. Am. Chem. Soc. 2020, 142, 2218–2221. [Google Scholar] [CrossRef] [PubMed]
- Krishna Nandakumar, D.; Zhang, Y.; Kishore Ravi, S.; Guo, N.; Zhang, C.; Ching Tan, S.; Nandakumar, D.K.; Zhang, Y.; Ravi, S.K.; Tan, S.C.; et al. Solar Energy Triggered Clean Water Harvesting from Humid Air Existing above Sea Surface Enabled by a Hydrogel with Ultrahigh Hygroscopicity. Adv. Mater. 2019, 31, 1806730. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Zhang, X.; Qu, H.; Gen Yu, Z.; Zhang, Y.; Jie Eey, T.; Zhang, Y.-W.; Ching Tan, S.; Yang, J.; Zhang, X.; et al. A Moisture-Hungry Copper Complex Harvesting Air Moisture for Potable Water and Autonomous Urban Agriculture. Adv. Mater. 2020, 32, 2002936. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Yang, J.; Borayek, R.; Qu, H.; Nandakumar, D.K.; Zhang, Q.; Ding, J.; Tan, S.C. Super-Hygroscopic Film for Wearables with Dual Functions of Expediting Sweat Evaporation and Energy Harvesting. Nano Energy 2020, 75, 104873. [Google Scholar] [CrossRef]
- Matsumoto, K.; Sakikawa, N.; Miyata, T. Thermo-Responsive Gels That Absorb Moisture and Ooze Water. Nat. Commun. 2018, 9, 1–7. [Google Scholar] [CrossRef]
- Zhao, F.; Zhou, X.; Liu, Y.; Shi, Y.; Dai, Y.; Yu, G. Super Moisture-Absorbent Gels for All-Weather Atmospheric Water Harvesting. Adv. Mater. 2019, 31, 1806446. [Google Scholar] [CrossRef]
- Yilmaz, G.; Meng, F.L.; Lu, W.; Abed, J.; Peh, C.K.N.; Gao, M.; Sargent, E.H.; Ho, G.W. Autonomous Atmospheric Water Seeping MOF Matrix. Sci. Adv. 2020, 6, 8605–8621. [Google Scholar] [CrossRef]
- Yang, K.; Pan, T.; Pinnau, I.; Shi, Z.; Han, Y. Simultaneous Generation of Atmospheric Water and Electricity Using a Hygroscopic Aerogel with Fast Sorption Kinetics. Nano Energy 2020, 78, 105326. [Google Scholar] [CrossRef]
- Xu, J.; Li, T.; Chao, J.; Wu, S.; Yan, T.; Li, W.; Cao, B.; Wang, R. Efficient Solar-Driven Water Harvesting from Arid Air with Metal–Organic Frameworks Modified by Hygroscopic Salt. Angew. Chem. Int. Ed. 2020, 59, 5202–5210. [Google Scholar] [CrossRef]
- Yang, K.; Shi, Y.; Wu, M.; Wang, W.; Jin, Y.; Li, R.; Shahzad, M.W.; Ng, K.C.; Wang, P. Hollow Spherical SiO2 Micro-Container Encapsulation of LiCl for High-Performance Simultaneous Heat Reallocation and Seawater Desalination. J. Mater. Chem. A 2020, 8, 1887–1895. [Google Scholar] [CrossRef]
- Li, R.; Shi, Y.; Alsaedi, M.; Wu, M.; Shi, L.; Wang, P. Hybrid Hydrogel with High Water Vapor Harvesting Capacity for Deployable Solar-Driven Atmospheric Water Generator. Envon. Sci. Technol. 2018, 52, 11367–11377. [Google Scholar] [CrossRef]
- Wang, J.Y.; Wang, R.Z.; Wang, L.W. Water Vapor Sorption Performance of ACF-CaCl2 and Silica Gel-CaCl2 Composite Adsorbents. Appl. Eng. 2016, 100, 893–901. [Google Scholar] [CrossRef]
- Gordeeva, L.G.; Restuccia, G.; Freni, A.; Aristov, Y.I. Water Sorption on Composites “LiBr in a Porous Carbon”. Fuel Process. Technol. 2002, 79, 225–231. [Google Scholar] [CrossRef]
- Chen, Y.; Cao, Y.; Lu, X.; Zhao, C.; Yan, C.; Mu, T. Water Sorption in Protic Ionic Liquids: Correlation between Hygroscopicity and Polarity. New J. Chem. 2013, 37, 1959–1967. [Google Scholar] [CrossRef]
- Wang, Y.; Gao, S.; Zhong, H.; Zhang, B.; Cui, M.; Jiang, M.; Wang, S.; Wang, Z. Heterogeneous Wettability and Radiative Cooling for Efficient Deliquescent Sorbents-Based Atmospheric Water Harvesting. Cell Rep. Phys. Sci. 2022, 3, 100879. [Google Scholar] [CrossRef]
- Wang, J.; Deng, C.; Zhong, G.; Ying, W.; Li, C.; Wang, S.; Liu, Y.; Wang, R.; Zhang, H. High-Yield and Scalable Water Harvesting of Honeycomb Hygroscopic Polymer Driven by Natural Sunlight. Cell Rep. Phys. Sci. 2022, 3, 100954. [Google Scholar] [CrossRef]
- Deng, F.; Wang, C.; Xiang, C.; Wang, R. Bioinspired Topological Design of Super Hygroscopic Complex for Cost-Effective Atmospheric Water Harvesting. Nano Energy 2021, 90, 106642. [Google Scholar] [CrossRef]
- Wang, J.Y.; Wang, R.Z.; Tu, Y.D.; Wang, L.W. Universal Scalable Sorption-Based Atmosphere Water Harvesting. Energy 2018, 165, 387–395. [Google Scholar] [CrossRef]
- Wang, M.; Sun, T.; Wan, D.; Dai, M.; Ling, S.; Wang, J.; Liu, Y.; Fang, Y.; Xu, S.; Yeo, J.; et al. Solar-Powered Nanostructured Biopolymer Hygroscopic Aerogels for Atmospheric Water Harvesting. Nano Energy 2021, 80, 105569. [Google Scholar] [CrossRef]
- Qi, H.; Wei, T.; Zhao, W.; Zhu, B.; Liu, G.; Wang, P.; Lin, Z.; Wang, X.; Li, X.; Zhang, X.; et al. An Interfacial Solar-Driven Atmospheric Water Generator Based on a Liquid Sorbent with Simultaneous Adsorption–Desorption. Adv. Mater. 2019, 31, 1903378. [Google Scholar] [CrossRef] [PubMed]
- Harish; Dhanraj, S.; Gopi, R.; Loganathan, G.B. An Efficiency Study on Water Extraction from Air Using Thermophoresis Method. IOP Conf. Ser. Mater. Sci. Eng. 2019, 574, 012003. [Google Scholar] [CrossRef]
- Al-Farayedhi, A.A.; Ibrahim, N.I.; Gandhidasan, P. Condensate as a Water Source from Vapor Compression Systems in Hot and Humid Regions. Desalination 2014, 349, 60–67. [Google Scholar] [CrossRef]
- Elattar, H.F.; Fouda, A.; Nada, S.A. Performance Investigation of a Novel Solar Hybrid Air Conditioning and Humidification–Dehumidification Water Desalination System. Desalination 2016, 382, 28–42. [Google Scholar] [CrossRef]
- Groendijk, L.; de Vries, H.E. Development of a Mobile Water Maker, a Sustainable Way to Produce Safe Drinking Water in Developing Countries. Desalination 2009, 248, 106–113. [Google Scholar] [CrossRef]
- Zolfagharkhani, S.; Zamen, M.; Shahmardan, M.M. Thermodynamic Analysis and Evaluation of a Gas Compression Refrigeration Cycle for Fresh Water Production from Atmospheric Air. Energy Convers. Manag. 2018, 170, 97–107. [Google Scholar] [CrossRef]
- Van Wylen, G.J.; Sonntag, R.E.; Borgnakke, C. Fundamentals of Classical Thermodynamics; Wiley: New York, NY, USA, 1985; ISBN 9780471829331. [Google Scholar]
- Zhang, T.; Wan, R.H.; Yan, T.; Wang, L.; Lin, C.H. Development of Portable Air-Based Water Collector for Field Operations under Conditions of Severe Shortage. Chin. Med. Equip. 2010, 8, 70–72. [Google Scholar]
- Cao, D.; Zou, Y. System Optimization of Water Exaction from Air by Semiconductor Cooling Air. Build. Energy Environ. 2022, 35, 71–73. [Google Scholar]
- Magrini, A.; Cattani, L.; Cartesegna, M.; Magnani, L. Integrated Systems for Air Conditioning and Production of Drinking Water—Preliminary Considerations. Energy Procedia 2015, 75, 1659–1665. [Google Scholar] [CrossRef]
- Habeebullah, B.A. Potential Use of Evaporator Coils for Water Extraction in Hot and Humid Areas. Desalination 2009, 237, 330–345. [Google Scholar] [CrossRef]
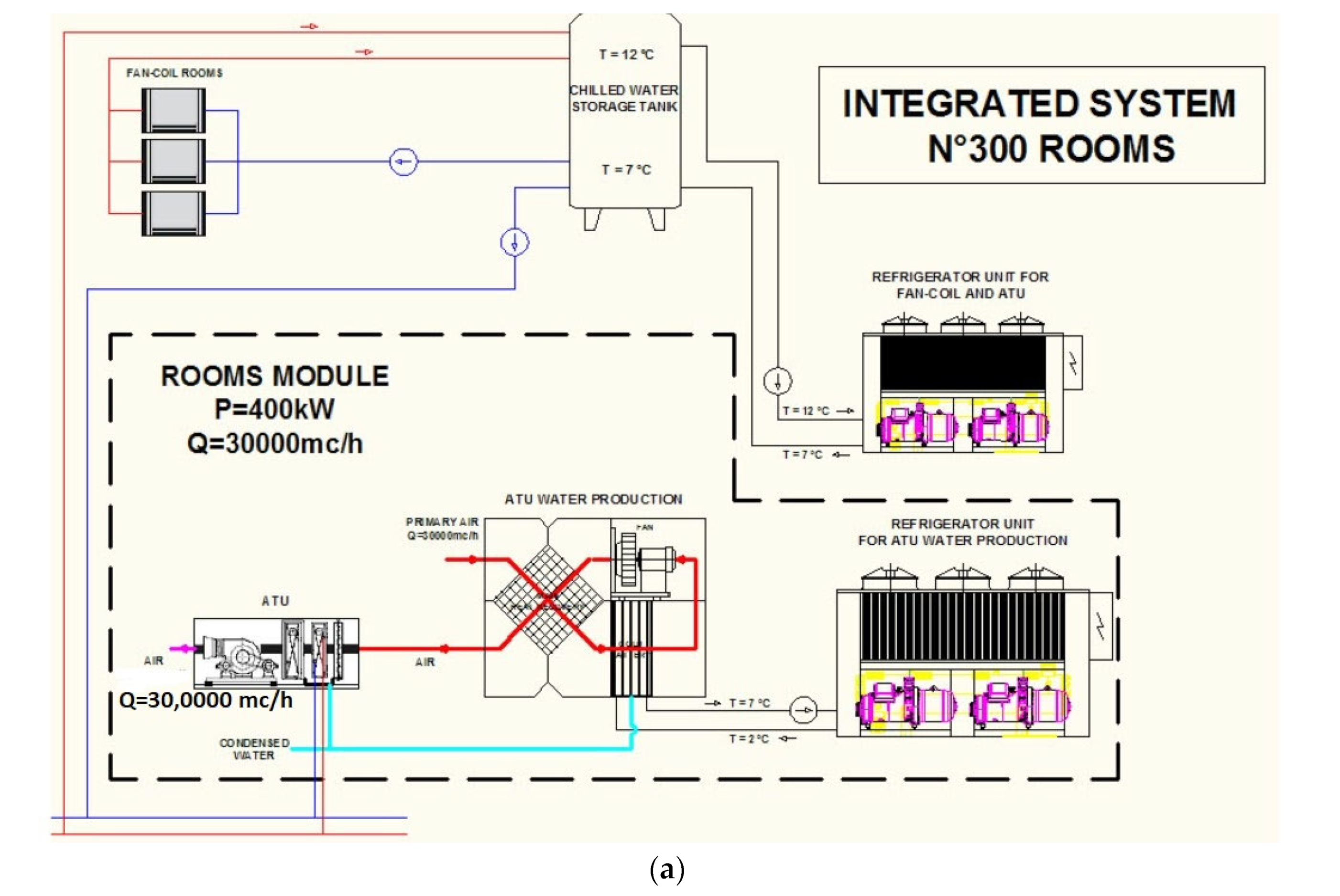
- Magrini, A.; Cattani, L.; Cartesegna, M.; Magnani, L. Water Production from Air Conditioning Systems: Some Evaluations about a Sustainable Use of Resources. Sustainability 2017, 9, 1309. [Google Scholar] [CrossRef]
- Cattani, L.; Magrini, A.; Cattani, P. Water Extraction from Air by Refrigeration—Experimental Results from an Integrated System Application. Appl. Sci. 2018, 8, 2262. [Google Scholar] [CrossRef]
- He, W.; Yu, P.; Hu, Z.; Lv, S.; Qin, M.; Yu, C. Experimental Study and Performance Analysis of a Portable Atmospheric Water Generator. Energies 2019, 13, 73. [Google Scholar] [CrossRef]
- Liu, S.; He, W.; Hu, D.; Lv, S.; Chen, D.; Wu, X.; Xu, F.; Li, S. Experimental Analysis of a Portable Atmospheric Water Generator by Thermoelectric Cooling Method. Energy Procedia 2017, 142, 1609–1614. [Google Scholar] [CrossRef]
- Robertson, B.I.; Shin, Y.-H.; Choi, J.-W.; Kadhim, T.J.; Abbas, A.K.; Kadhim, H.J. Experimental Study of Atmospheric Water Collection Powered by Solar Energy Using the Peltier Effect. IOP Conf. Ser. Mater. Sci. Eng. 2019, 671, 012155. [Google Scholar] [CrossRef]
- Irshad, K.; Almalawi, A.; Habib, K.; Zahir, M.H.; Ali, A.; Islam, S.; Saha, B.B. Experimental Study of a Thermoelectric Air Duct Dehumidification System for Tropical Climate. Heat Transf. Eng. 2020, 42, 1159–1171. [Google Scholar] [CrossRef]
- Çengel, Y.A.; Cimbala, J.M. Fluids Mechanics—Fundaments and Applications. J. Chem. Inf. Model 2017, 53, 1024–1051. [Google Scholar]
- Gabler, R.E. Physical Geography; Wiley: New York, NY, USA, 2017; ISBN 9780495555063. [Google Scholar]
- Peeters, R.; Vanderschaeghe, H.; Rongé, J.; Martens, J.A. Energy Performance and Climate Dependency of Technologies for Fresh Water Production from Atmospheric Water Vapour. Env. Sci. 2020, 6, 2016–2034. [Google Scholar] [CrossRef]
- Huang, J. A Simple Accurate Formula for Calculating Saturation Vapor Pressure of Water and Ice. J. Appl. Meteorol. Clim. 2018, 57, 1265–1272. [Google Scholar] [CrossRef]
- Moonaliza, M. Introduction and Basic Concepts. In Thermodynamics an Engineering Approach, 5th ed.; McGraw-Hill: New York, NY, USA, 2011; Available online: https://www.academia.edu/41069675/Introduction_and_Basic_Concepts_Thermodynamics_An_Engineering_Approach_5th_edition_ (accessed on 21 September 2022).
- Incropera, F.; DeWitt, D.; Bergman, T.; Lavine, A. The Lumped Capacitance Method. In Principles of Heat and Mass Transfer; John Wiley & Sons: Hoboken, NJ, USA, 2003; pp. 279–286. [Google Scholar]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Thavalengal, M.S.; Jamil, M.A.; Mehroz, M.; Xu, B.B.; Yaqoob, H.; Sultan, M.; Imtiaz, N.; Shahzad, M.W. Progress and Prospects of Air Water Harvesting System for Remote Areas: A Comprehensive Review. Energies 2023, 16, 2686. https://doi.org/10.3390/en16062686
Thavalengal MS, Jamil MA, Mehroz M, Xu BB, Yaqoob H, Sultan M, Imtiaz N, Shahzad MW. Progress and Prospects of Air Water Harvesting System for Remote Areas: A Comprehensive Review. Energies. 2023; 16(6):2686. https://doi.org/10.3390/en16062686
Chicago/Turabian StyleThavalengal, Mohammed Sanjid, Muhammad Ahmad Jamil, Muhammad Mehroz, Ben Bin Xu, Haseeb Yaqoob, Muhammad Sultan, Nida Imtiaz, and Muhammad Wakil Shahzad. 2023. "Progress and Prospects of Air Water Harvesting System for Remote Areas: A Comprehensive Review" Energies 16, no. 6: 2686. https://doi.org/10.3390/en16062686
APA StyleThavalengal, M. S., Jamil, M. A., Mehroz, M., Xu, B. B., Yaqoob, H., Sultan, M., Imtiaz, N., & Shahzad, M. W. (2023). Progress and Prospects of Air Water Harvesting System for Remote Areas: A Comprehensive Review. Energies, 16(6), 2686. https://doi.org/10.3390/en16062686










