Waterborne LiNi0.5Mn1.5O4 Cathode Formulation Optimization through Design of Experiments and Upscaling to 1 Ah Li-Ion Pouch Cells
Abstract
:1. Introduction
2. Materials and Methods
2.1. Electrode Manufacturing
2.2. Physicochemical Characterization
2.3. Cell Assembly
2.4. Electrochemical Testing
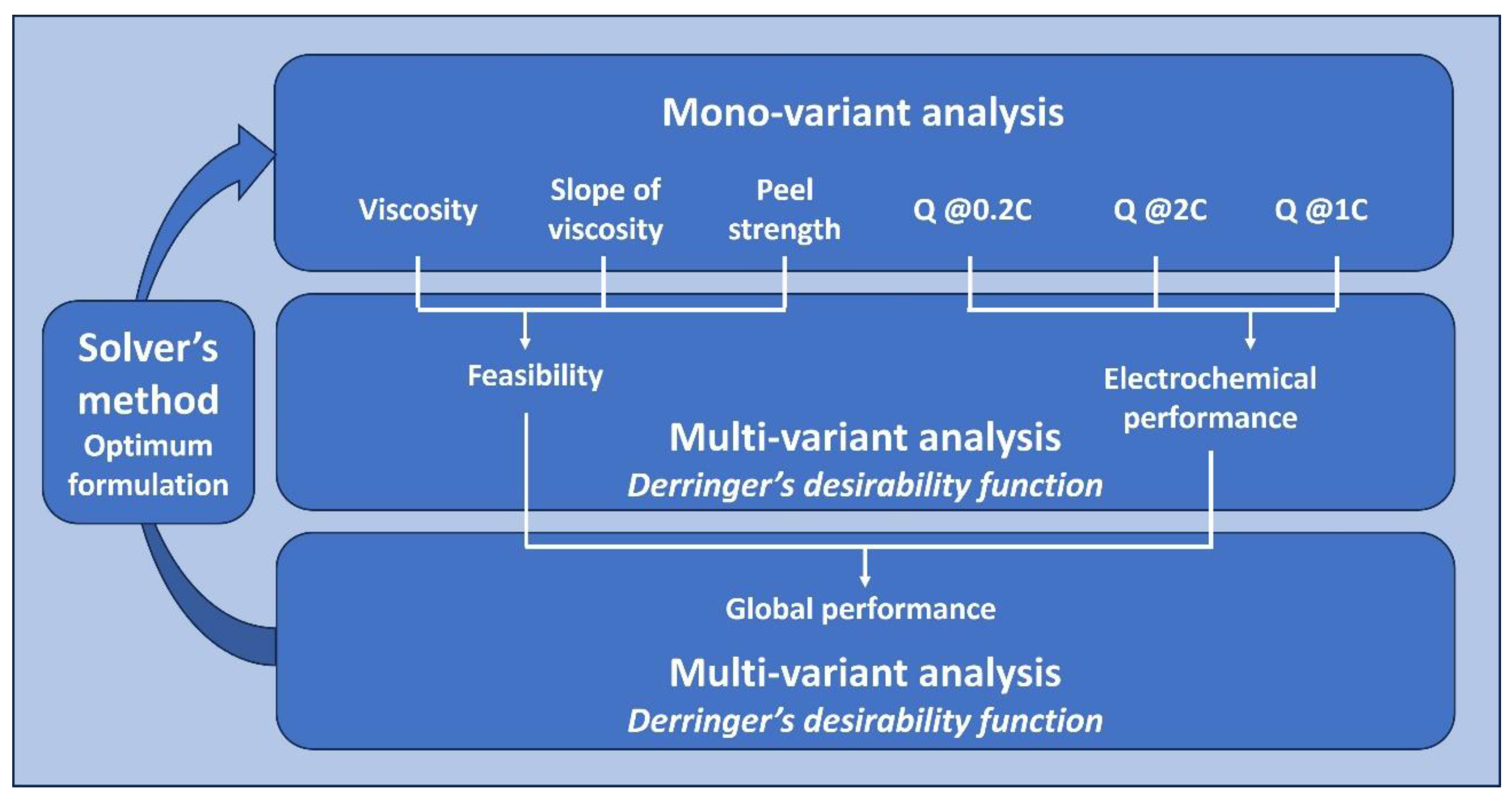
2.5. Definition of the Formulations and the Parameters for Their Evaluation
3. Results and Discussion
3.1. Mono-Variant Analysis
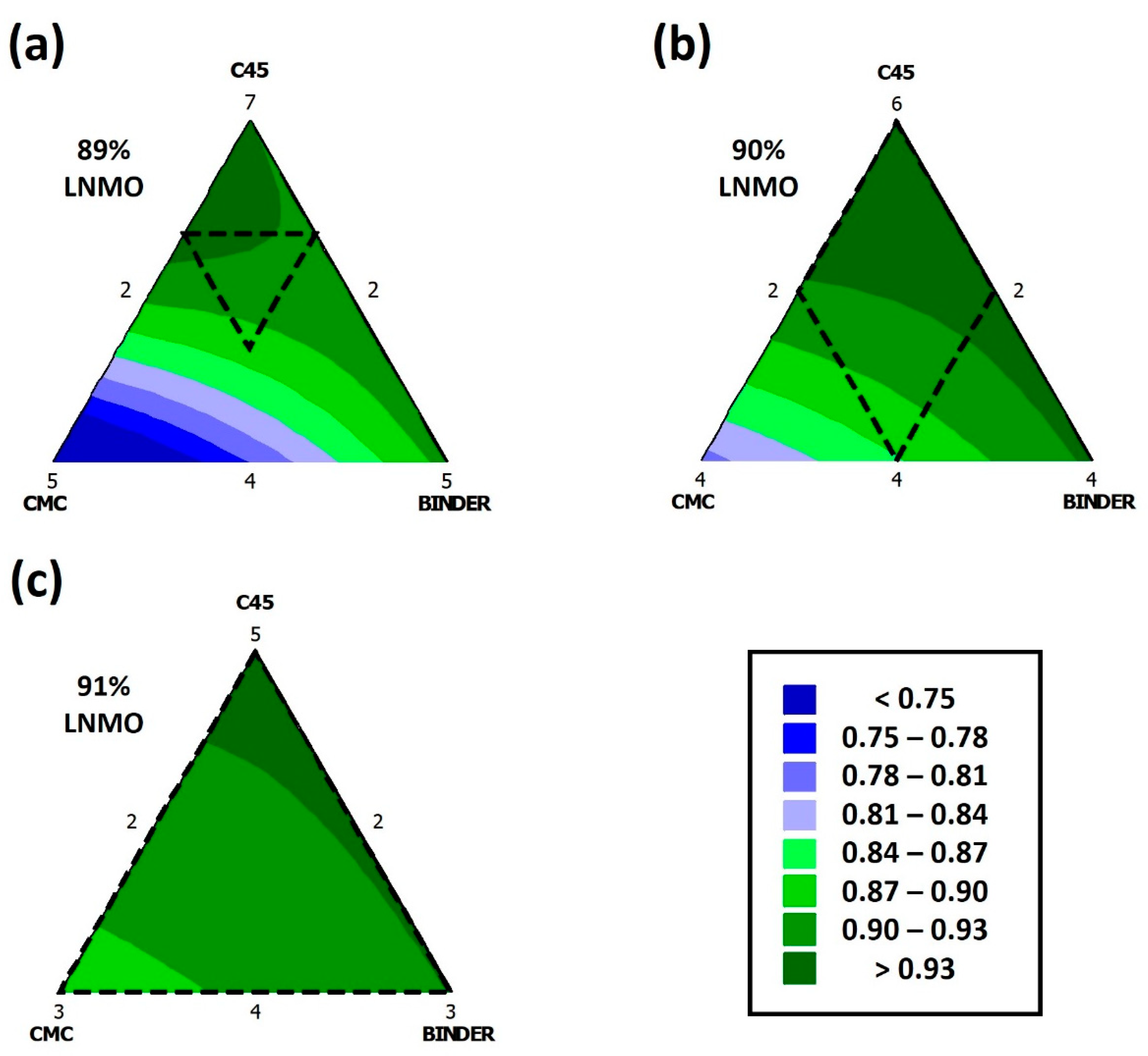
3.2. Multi-Variant Analysis
0.01 [LNMO] [C45] + 0.03 [LNMO] [CMC] + 0.01 [LNMO] [Binder] +
0.05 [C45] [Binder]
3.3. SOLVER’s Method to Obtain the Cathode with the Best Formulation
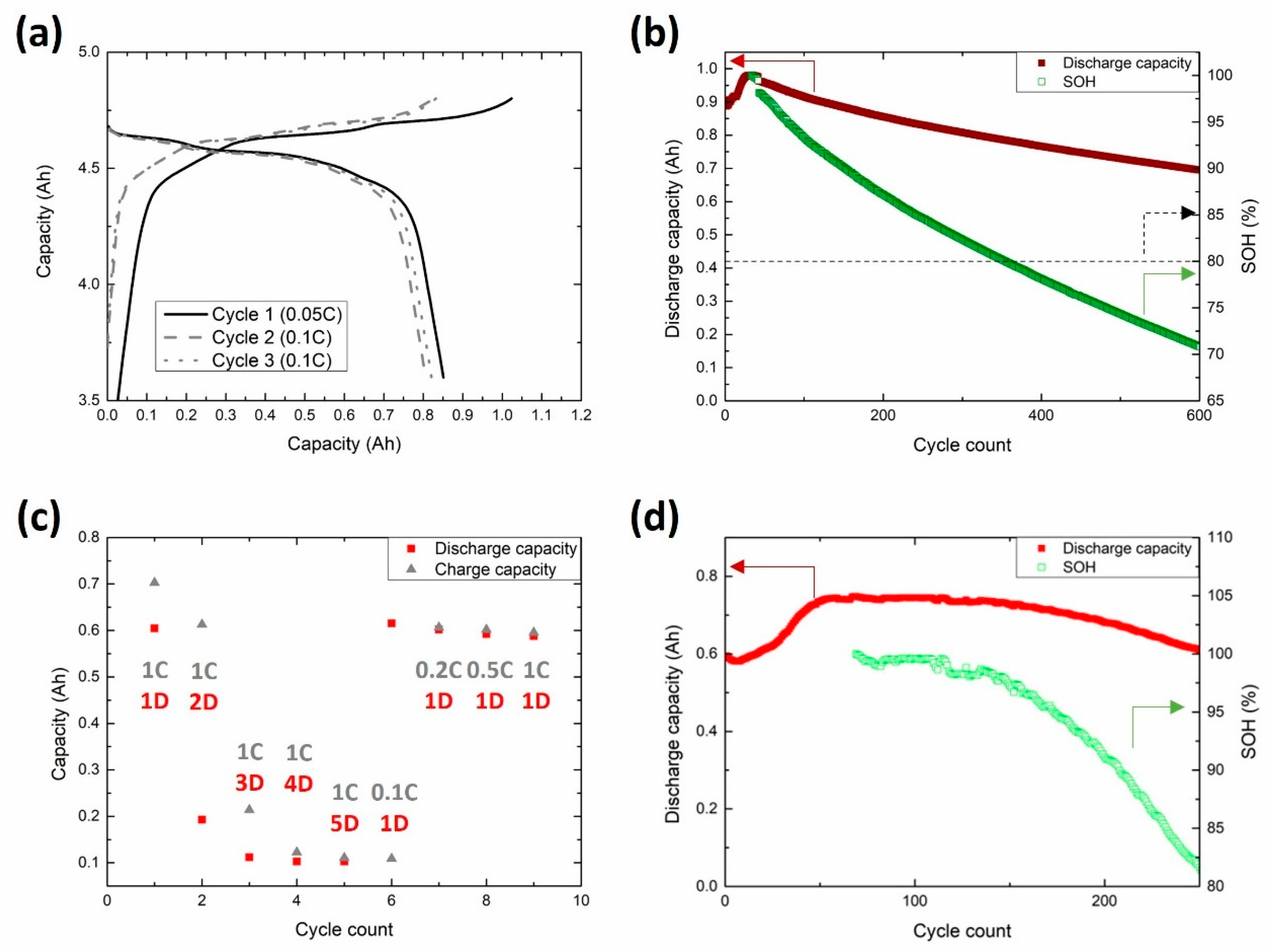
3.4. Upscaling and Pouch Cell Testing
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Winter, M.; Barnett, B.; Xu, K. Before Li Ion Batteries. Chem. Rev. 2018, 118, 11433–11456. [Google Scholar] [CrossRef]
- International Energy Agency. Global EV Outlook 2021 Accelerating Ambitions despite the Pandemic. 2021. Available online: https://www.iea.org/reports/global-ev-outlook-2021 (accessed on 24 October 2023).
- Li, M.; Lu, J.; Chen, Z.; Amine, K. 30 Years of Lithium-Ion Batteries. Adv. Mater. 2018, 30, 1800561. [Google Scholar] [CrossRef]
- Volta Foundation The Battery Report. 2022. Available online: https://intercalationstation.substack.com/p/the-battery-report-2022 (accessed on 24 October 2023).
- Marinaro, M.; Bresser, D.; Beyer, E.; Faguy, P.; Hosoi, K.; Li, H.; Sakovica, J.; Amine, K.; Wohlfahrt-Mehrens, M.; Passerini, S. Bringing Forward the Development of Battery Cells for Automotive Applications: Perspective of R&D Activities in China, Japan, the EU and the USA. J. Power Sources 2020, 459, 228073. [Google Scholar] [CrossRef]
- Tidblad, A.A.; Edström, K.; Hernández, G.; de Meatza, I.; Landa-Medrano, I.; Jacas Biendicho, J.; Trilla, L.; Buysse, M.; Ierides, M.; Horno, B.P.; et al. Future Material Developments for Electric Vehicle Battery Cells Answering Growing Demands from an End-User Perspective. Energies 2021, 14, 4223. [Google Scholar] [CrossRef]
- Qin, Z.; Zhang, Y.; Luo, W.; Zhang, T.; Wang, T.; Ni, L.; Wang, H.; Zhang, N.; Liu, X.; Zhou, J.; et al. A Universal Molten Salt Method for Direct Upcycling of Spent Ni-Rich Cathode towards Single-Crystalline Li-Rich Cathode. Angew. Chem. Int. Ed. 2023, 62, e202218672. [Google Scholar] [CrossRef]
- Fichtner, M. Recent Research and Progress in Batteries for Electric Vehicles. Batter. Supercaps 2022, 5, e202100224. [Google Scholar] [CrossRef]
- Ruffo, G.H. Tesla Slide Reveals $25,000 Electric Car Will Have LFP Batteries. Available online: https://insideevs.com/news/446245/tesla-slide-25000-car-lfp-batteries/ (accessed on 15 September 2023).
- Moretti, A.; Sharova, V.; Carvalho, D.V.; Boulineau, A.; Porcher, W.; de Meatza, I.; Passerini, S. A Comparison of Formation Methods for Graphite//LiFePO4 Cells. Batter. Supercaps 2019, 2, 240–247. [Google Scholar] [CrossRef]
- Aiken, C.P.; Logan, E.R.; Eldesoky, A.; Hebecker, H.; Oxner, J.M.; Harlow, J.E.; Metzger, M.; Dahn, J.R. Li[Ni0.5Mn0.3Co0.2]O2 as a Superior Alternative to LiFePO4 for Long-Lived Low Voltage Li-Ion Cells. J. Electrochem. Soc. 2022, 169, 050512. [Google Scholar] [CrossRef]
- Editorial: The Journey of an Electrifying (r)Evolution. Nat. Commun. 2021, 12, 4153. [CrossRef]
- Su, L.; Jarvis, K.; Charalambous, H.; Dolocan, A.; Manthiram, A. Stabilizing High-Nickel Cathodes with High-Voltage Electrolytes. Adv. Funct. Mater. 2023, 33, 2213675. [Google Scholar] [CrossRef]
- WTO. Panel Rules against Indonesia’s Export Limitations on Raw Material. Available online: https://ec.europa.eu/commission/presscorner/detail/en/ip_22_7314 (accessed on 15 September 2023).
- Indonesia’s Uncertain Climb up the Nickel Value Chain. Available online: https://www.lowyinstitute.org/the-interpreter/indonesia-s-uncertain-climb-nickel-value-chain (accessed on 15 September 2023).
- Xu, W.; Li, Q.; Sui, F.; Guo, S.; Qi, R.; Yan, C.; Chen, L.; Xia, S.; Guo, J.; Li, Z.; et al. Unveiling the Role of Ni Doping in the Electrochemical Performance Improvement of the LiMn2O4 Cathodes. Appl. Surf. Sci. 2023, 624, 157142. [Google Scholar] [CrossRef]
- Capsoni, D.; Bini, M.; Chiodelli, G.; Massarotti, V.; Mustarelli, P.; Linati, L.; Mozzati, M.C.; Azzoni, C.B. Jahn-Teller Transition in Al3+ Doped LiMn2O4 Spinel. Solid. State Commun. 2003, 126, 169–174. [Google Scholar] [CrossRef]
- Fey, G.T.K.; Lu, C.Z.; Prem Kumar, T. Preparation and Electrochemical Properties of High-Voltage Cathode Materials, LiMyNi0.5-YMn1.5O4 (M = Fe, Cu, Al, Mg; y = 0.0–0.4). J. Power Sources 2003, 115, 332–345. [Google Scholar] [CrossRef]
- Liu, G.; Zhang, J.; Zhang, X.; Du, Y.; Zhang, K.; Li, G.; Yu, H.; Li, C.; Li, Z.; Sun, Q.; et al. Study on Oxygen Deficiency in Spinel LiNi0.5Mn1.5O4 and Its Fe and Cr-Doped Compounds. J. Alloys Compd. 2017, 725, 580–586. [Google Scholar] [CrossRef]
- Hu, L.; Amine, K.; Zhang, Z. Fluorinated Electrolytes for 5-V Li-Ion Chemistry: Dramatic Enhancement of LiNi0.5Mn1.5O4/Graphite Cell Performance by a Lithium Reservoir. Electrochem. Commun. 2014, 44, 34–37. [Google Scholar] [CrossRef]
- Lim, G.; Shin, D.; Chae, K.H.; Cho, M.K.; Kim, C.; Sohn, S.S.; Lee, M.; Hong, J. Regulating Dynamic Electrochemical Interface of LiNi0.5Mn1.5O4 Spinel Cathode for Realizing Simultaneous Mn and Ni Redox in Rechargeable Lithium Batteries. Adv. Energy Mater. 2022, 12, 2202049. [Google Scholar] [CrossRef]
- Jusys, Z.; Binder, M.; Schnaidt, J.; Behm, R.J. A Novel DEMS Approach for Studying Gas Evolution at Battery-Type Electrode|electrolyte Interfaces: High-Voltage LiNi0.5Mn1.5O4 Cathode in Ethylene and Dimethyl Carbonate Electrolytes. Electrochim. Acta 2019, 314, 188–201. [Google Scholar] [CrossRef]
- Kim, J.H.; Pieczonka, N.P.W.; Lu, P.; Liu, Z.; Qiao, R.; Yang, W.; Tessema, M.M.; Sun, Y.K.; Powell, B.R. In Situ Formation of a Cathode-Electrolyte Interface with Enhanced Stability by Titanium Substitution for High Voltage Spinel Lithium-Ion Batteries. Adv. Mater. Interfaces 2015, 2, 1500109. [Google Scholar] [CrossRef]
- Huang, X.; Chen, K.; Liu, Y. Enhancement of LiNi0.5Mn1.5O4 Cathode Materials through Interfacial Modification of Amorphous Al2O3 in Lithium Ion Batteries. J. Electrochem. Soc. 2019, 166, A5081–A5089. [Google Scholar] [CrossRef]
- Sun, P.; Ma, Y.; Zhai, T.; Li, H. High Performance LiNi0.5Mn1.5O4 Cathode by Al-Coating and Al3+-Doping through a Physical Vapor Deposition Method. Electrochim. Acta 2016, 191, 237–246. [Google Scholar] [CrossRef]
- Deng, H.; Nie, P.; Luo, H.; Zhang, Y.; Wang, J.; Zhang, X. Highly Enhanced Lithium Storage Capability of LiNi0.5Mn1.5O4 by Coating with Li2TiO3 for Li-Ion Batteries. J. Mater. Chem. A Mater. 2014, 2, 18256–18262. [Google Scholar] [CrossRef]
- Yang, X.; Yang, T.; Liang, S.; Wu, X.; Zhang, H. Modification of LiNi0.5Mn1.5O4 High Potential Cathode from the Inner Lattice to the Outer Surface with Cr3+-Doping and Li+-Conductor Coating. J. Mater. Chem. A Mater. 2014, 2, 10359–10364. [Google Scholar] [CrossRef]
- Hagh, N.M.; Amatucci, G.G. Effect of Cation and Anion Doping on Microstructure and Electrochemical Properties of the LiMn1.5Ni0.5O4-δ Spinel. J. Power Sources 2014, 256, 457–469. [Google Scholar] [CrossRef]
- Höweling, A.; Glatthaar, S.; Nötzel, D.; Binder, J.R. Evidence of Loss of Active Lithium in Titanium-Doped LiNi0.5Mn1.5O4/Graphite Cells. J. Power Sources 2015, 274, 1267–1275. [Google Scholar] [CrossRef]
- Lim, J.M.; Oh, R.G.; Kim, D.; Cho, W.; Cho, K.; Cho, M.; Park, M.S. Design of Surface Doping for Mitigating Transition Metal Dissolution in LiNi0.5Mn1.5O4 Nanoparticles. ChemSusChem 2016, 9, 2967–2973. [Google Scholar] [CrossRef]
- Liu, M.H.; Huang, H.T.; Lin, C.M.; Chen, J.M.; Liao, S.C. Mg Gradient-Doped LiNi0.5Mn1.5O4 as the Cathode Material for Li-Ion Batteries. Electrochim. Acta 2014, 120, 133–139. [Google Scholar] [CrossRef]
- Kim, J.-H.; Myung, S.-T.; Yoon, C.S.; Oh, I.-H.; Sun, Y.-K. Effect of Ti Substitution for Mn on the Structure of LiNi0.5Mn1.5−xTixO4 and Their Electrochemical Properties as Lithium Insertion Material. J. Electrochem. Soc. 2004, 151, A1911. [Google Scholar] [CrossRef]
- Höweling, A.; Stenzel, D.; Gesswein, H.; Kaus, M.; Indris, S.; Bergfeldt, T.; Binder, J.R. Variations in Structure and Electrochemistry of Iron- and Titanium-Doped Lithium Nickel Manganese Oxyfluoride Spinels. J. Power Sources 2016, 315, 269–276. [Google Scholar] [CrossRef]
- Sun, H.; Kong, X.; Wang, B.; Luo, T.; Liu, G. LiNi0.5Mn1.45Zn0.05O4 with Excellent Electrochemical Performance for Lithium Ion Batteries. Int. J. Electrochem. Sci. 2017, 12, 8609–8621. [Google Scholar] [CrossRef]
- Hofmann, A.; Höweling, A.; Bohn, N.; Müller, M.; Binder, J.R.; Hanemann, T. Additives for Cycle Life Improvement of High-Voltage LNMO-Based Li-Ion Cells. ChemElectroChem 2019, 6, 5255–5263. [Google Scholar] [CrossRef]
- Milien, M.S.; Beyer, H.; Beichel, W.; Klose, P.; Gasteiger, H.A.; Lucht, B.L.; Krossing, I. Lithium Bis(2,2,2-Trifluoroethyl)Phosphate Li[O2P(OCH2CF3)2]: A High Voltage Additive for LNMO/Graphite Cells. J. Electrochem. Soc. 2018, 165, A2569–A2576. [Google Scholar] [CrossRef]
- Solchenbach, S.; Wetjen, M.; Pritzl, D.; Schwenke, K.U.; Gasteiger, H.A. Lithium Oxalate as Capacity and Cycle-Life Enhancer in LNMO/Graphite and LNMO/SiG Full Cells. J. Electrochem. Soc. 2018, 165, A512–A524. [Google Scholar] [CrossRef]
- Li, Y.; Veith, G.M.; Browning, K.L.; Chen, J.; Hensley, D.K.; Paranthaman, M.P.; Dai, S.; Sun, X.G. Lithium Malonatoborate Additives Enabled Stable Cycling of 5 V Lithium Metal and Lithium Ion Batteries. Nano Energy 2017, 40, 9–19. [Google Scholar] [CrossRef]
- Lee, T.J.; Lee, J.B.; Yoon, T.; Kim, D.; Chae, O.B.; Jung, J.; Soon, J.; Ryu, J.H.; Kim, J.J.; Oh, S.M. Tris(Pentafluorophenyl)Silane as an Electrolyte Additive for 5 V LiNi0.5Mn1.5O4 Positive Electrode. J. Electrochem. Soc. 2016, 163, A898–A903. [Google Scholar] [CrossRef]
- Feng, T.; Yang, G.; Zhang, S.; Xu, Z.; Zhou, H.; Wu, M. Low-Temperature and High-Voltage Lithium-Ion Battery Enabled by Localized High-Concentration Carboxylate Electrolytes. Chem. Eng. J. 2022, 433, 134138. [Google Scholar] [CrossRef]
- Arbizzani, C.; De Giorgio, F.; Mastragostino, M. Characterization Tests for Plug-in Hybrid Electric Vehicle Application of Graphite/LiNi0.4Mn1.6O4 Cells with Two Different Separators and Electrolytes. J. Power Sources 2014, 266, 170–174. [Google Scholar] [CrossRef]
- Banerjee, A.; Ziv, B.; Shilina, Y.; Ziegelbauer, J.M.; Liu, H.; Harris, K.J.; Botton, G.; Goward, G.R.; Luski, S.; Aurbach, D.; et al. Review—Multifunctional Separators: A Promising Approach for Improving the Durability and Performance of Li-Ion Batteries. J. Electrochem. Soc. 2019, 166, A5369–A5377. [Google Scholar] [CrossRef]
- De Giorgio, F.; Laszczynski, N.; von Zamory, J.; Mastragostino, M.; Arbizzani, C.; Passerini, S. Graphite//LiNi0.5Mn1.5O4 Cells Based on Environmentally Friendly Made-in-Water Electrodes. ChemSusChem 2017, 10, 379–386. [Google Scholar] [CrossRef]
- Zhang, K.; Tian, Y.; Wei, C.; An, Y.; Feng, J. Building Stable Solid Electrolyte Interphases (SEI) for Microsized Silicon Anode and 5V-Class Cathode with Salt Engineered Nonflammable Phosphate-Based Lithium-Ion Battery Electrolyte. Appl. Surf. Sci. 2021, 553, 149566. [Google Scholar] [CrossRef]
- Li, W.; Cho, Y.G.; Yao, W.; Li, Y.; Cronk, A.; Shimizu, R.; Schroeder, M.A.; Fu, Y.; Zou, F.; Battaglia, V.; et al. Enabling High Areal Capacity for Co-Free High Voltage Spinel Materials in next-Generation Li-Ion Batteries. J. Power Sources 2020, 473, 228579. [Google Scholar] [CrossRef]
- Frith, J.T.; Lacey, M.J.; Ulissi, U. A Non-Academic Perspective on the Future of Lithium-Based Batteries. Nat. Commun. 2023, 14, 420. [Google Scholar] [CrossRef]
- Ue, M.; Sakaushi, K.; Uosaki, K. Basic Knowledge in Battery Research Bridging the Gap between Academia and Industry. Mater. Horiz. 2020, 7, 1937–1954. [Google Scholar] [CrossRef]
- Yao, W.; Chouchane, M.; Li, W.; Bai, S.; Liu, Z.; Li, L.; Chen, A.X.; Sayahpour, B.; Shimizu, R.; Raghavendran, G.; et al. A 5 V-Class Cobalt-Free Battery Cathode with High Loading Enabled by Dry Coating. Energy Environ. Sci. 2023, 16, 1620–1630. [Google Scholar] [CrossRef]
- Colombo, R.; Garino, N.; Versaci, D.; Amici, J.; Para, M.L.; Quartarone, E.; Francia, C.; Bella, F.; Bodoardo, S. Designing a Double-Coated Cathode with High Entropy Oxides by Microwave-Assisted Hydrothermal Synthesis for Highly Stable Li–S Batteries. J. Mater. Sci. 2022, 57, 15690–15704. [Google Scholar] [CrossRef]
- Wang, T.; Zhang, Q.; Zhong, J.; Chen, M.; Deng, H.; Cao, J.; Wang, L.; Peng, L.; Zhu, J.; Lu, B. 3D Holey Graphene/Polyacrylonitrile Sulfur Composite Architecture for High Loading Lithium Sulfur Batteries. Adv. Energy Mater. 2021, 11, 2100448. [Google Scholar] [CrossRef]
- Medenbach, L.; Adelhelm, P. Cell Concepts of Metal–Sulfur Batteries (Metal = Li, Na, K, Mg): Strategies for Using Sulfur in Energy Storage Applications. Top. Curr. Chem. 2017, 375, 81. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.; Li, J.; Li, T.; Gao, X.; Yuan, C. Life Cycle Assessment of Lithium Sulfur Battery for Electric Vehicles. J. Power Sources 2017, 343, 284–295. [Google Scholar] [CrossRef]
- Wang, T.; He, J.; Zhu, Z.; Cheng, X.; Zhu, J.; Lu, B.; Wu, Y. Heterostructures Regulating Lithium Polysulfides for Advanced Lithium-Sulfur Batteries. Adv. Mater. 2023, 2303520. [Google Scholar] [CrossRef]
- Wang, T.; He, J.; Cheng, X.B.; Zhu, J.; Lu, B.; Wu, Y. Strategies toward High-Loading Lithium-Sulfur Batteries. ACS Energy Lett. 2023, 8, 116–150. [Google Scholar] [CrossRef]
- Colombo, R.; Versaci, D.; Amici, J.; Bella, F.; Para, M.L.; Garino, N.; Laurenti, M.; Bodoardo, S.; Francia, C. Reduced Graphene Oxide Embedded with ZnS Nanoparticles as Catalytic Cathodic Material for Li-S Batteries. Nanomaterials 2023, 13, 2149. [Google Scholar] [CrossRef]
- Leonet, O.; Landa-Medrano, I.; Shafique, A.; Yari, S.; Rangasamy, V.; Vanhulsel, A.; Safari, M.; Van Bael, M.K.; Hardy, A.; Urdampilleta, I.; et al. Electrochemical Investigation of the Ageing of a 400 Wh/kg Li-S Pouch-Cell. Electrochim. Acta 2023, 464, 142889. [Google Scholar] [CrossRef]
- Muchuweni, E.; Mombeshora, E.T.; Muiva, C.M.; Sathiaraj, T.S. Lithium-Ion Batteries: Recent Progress in Improving the Cycling and Rate Performances of Transition Metal Oxide Anodes by Incorporating Graphene-Based Materials. J. Energy Storage 2023, 73, 109013. [Google Scholar] [CrossRef]
- Chen, S.; Dai, F.; Cai, M. Opportunities and Challenges of High-Energy Lithium Metal Batteries for Electric Vehicle Applications. ACS Energy Lett. 2020, 5, 3140–3151. [Google Scholar] [CrossRef]
- Jetybayeva, A.; Aaron, D.S.; Belharouak, I.; Mench, M.M. Critical Review on Recently Developed Lithium and Non-Lithium Anode-Based Solid-State Lithium-Ion Batteries. J. Power Sources 2023, 566, 232914. [Google Scholar] [CrossRef]
- Niri, M.F.; Apachitei, G.; Lain, M.; Copley, M.; Marco, J. Machine Learning for Investigating the Relative Importance of Electrodes’ N:P Areal Capacity Ratio in the Manufacturing of Lithium-Ion Battery Cells. J. Power Sources 2022, 549, 232124. [Google Scholar] [CrossRef]
- Teo, J.H.; Strauss, F.; Tripković, Đ.; Schweidler, S.; Ma, Y.; Bianchini, M.; Janek, J.; Brezesinski, T. Design-of-Experiments-Guided Optimization of Slurry-Cast Cathodes for Solid-State Batteries. Cell Rep. Phys. Sci. 2021, 2, 100465. [Google Scholar] [CrossRef]
- Rynne, O.; Dubarry, M.; Molson, C.; Lepage, D.; Prébé, A.; Aymé-Perrot, D.; Rochefort, D.; Dollé, M. Designs of Experiments for Beginners—A Quick Start Guide for Application to Electrode Formulation. Batteries 2019, 5, 72. [Google Scholar] [CrossRef]
- Rodríguez-Carvajal, J. Recent Advances in Magnetic Structure Determination by Neutron Powder Diffraction. Phys. B Phys. Condens. Matter 1993, 192, 55–69. [Google Scholar] [CrossRef]
- Kusano, T.; Ishii, M.; Tani, M.; Hiruta, O.; Matsunaga, T.; Nakamura, H. Rheological Behavior of Concentrated Slurry and Wet Granules for Lithium Ion Battery Electrodes. Adv. Powder Technol. 2020, 31, 4491–4499. [Google Scholar] [CrossRef]
- Wood, D.L.; Quass, J.D.; Li, J.; Ahmed, S.; Ventola, D.; Daniel, C. Technical and Economic Analysis of Solvent-Based Lithium-Ion Electrode Drying with Water and NMP. Dry. Technol. 2018, 36, 234–244. [Google Scholar] [CrossRef]
- Sahore, R.; Wood, D.L.; Kukay, A.; Grady, K.M.; Li, J.; Belharouak, I. Towards Understanding of Cracking during Drying of Thick Aqueous-Processed LiNi0.8Mn0.1Co0.1O2 Cathodes. ACS Sustain. Chem. Eng. 2020, 8, 3162–3169. [Google Scholar] [CrossRef]
- Guo, S.; Li, H.; Li, Y.; Han, Y.; Chen, K.; Xu, G.; Zhu, Y.; Hu, X. SiO2-Enhanced Structural Stability and Strong Adhesion with a New Binder of Konjac Glucomannan Enables Stable Cycling of Silicon Anodes for Lithium-Ion Batteries. Adv. Energy Mater. 2018, 8, 1800434. [Google Scholar] [CrossRef]
- Escudier, M.P.; Gouldson, I.W.; Pereira, A.S.; Pinho, F.T.; Poole, R.J. On the Reproducibility of the Rheology of Shear-Thinning Liquids. J. Non-Newton. Fluid. Mech. 2001, 97, 99–124. [Google Scholar] [CrossRef]
- De Meatza, I.; Urdampilleta, I.; Boyano, I.; Castrillo, I.; Landa-Medrano, I.; Sananes-Israel, S.; Eguia-Barrio, A.; Palomares, V. From Lab to Manufacturing Line: Guidelines for the Development and Upscaling of Aqueous Processed NMC622 Electrodes. J. Electrochem. Soc. 2023, 170, 010527. [Google Scholar] [CrossRef]
- Harrington, D.A. Electrochemical Impedance Spectroscopy; 2004; ISBN 9781118684030. Available online: https://www.ntnu.edu/documents/140124/0/EIS+Literature/cd116109-3fbf-4db5-9f99-ff5ab68cf950 (accessed on 24 October 2023).
- Derringer, G.; Suich, R. Simultaneous Optimization of Several Response Variables. J. Qual. Technol. 1980, 12, 214–219. [Google Scholar] [CrossRef]
- Zheng, J.; Xiao, J.; Nie, Z.; Zhang, J.-G. Lattice Mn 3+ Behaviors in Li4Ti5O12/LiNi0.5Mn1.5O4 Full Cells. J. Electrochem. Soc. 2013, 160, A1264–A1268. [Google Scholar] [CrossRef]
- Jehnichen, P.; Wedlich, K.; Korte, C. Degradation of High-Voltage Cathodes for Advanced Lithium-Ion Batteries–Differential Capacity Study on Differently Balanced Cells. Sci. Technol. Adv. Mater. 2019, 20, 1–9. [Google Scholar] [CrossRef]
- Mereacre, V.; Stüble, P.; Ghamlouche, A.; Binder, J.R. Enhancing the Stability of LiNi0.5Mn1.5O4 by Coating with LiNbO3 Solid-State Electrolyte: Novel Chemically Activated Coating Process versus Sol-Gel Method. Nanomaterials 2021, 11, 548. [Google Scholar] [CrossRef]
- Stüble, P.; Mereacre, V.; Geßwein, H.; Binder, J.R. On the Composition of LiNi0.5Mn1.5O4 Cathode Active Materials. Adv. Energy Mater. 2023, 13, 2203778. [Google Scholar] [CrossRef]
- Houache, M.S.E.; Yim, C.H.; Karkar, Z.; Abu-Lebdeh, Y. On the Current and Future Outlook of Battery Chemistries for Electric Vehicles—Mini Review. Batteries 2022, 8, 70. [Google Scholar] [CrossRef]
- Li, L.; Zhao, R.; Xu, T.; Wang, D.; Pan, D.; Zhang, K.; Yu, C.; Lu, X.; He, G.; Bai, Y. Stabilizing a High-Voltage LiNi0.5Mn1.5O4 Cathode towards All Solid State Batteries: A Li-Al-Ti-P-O Solid Electrolyte Nano-Shell with a Host Material. Nanoscale 2019, 11, 8967–8977. [Google Scholar] [CrossRef] [PubMed]
| Point Type | Formulation Number | LNMO wt.% | Carbon Black wt.% | CMC wt.% | Binder wt.% |
|---|---|---|---|---|---|
| Corners | 1 | 91 | 4 | 2 | 3 |
| 2 | 91 | 4 | 3 | 2 | |
| 3 | 90 | 4 | 3 | 3 | |
| 4 | 91 | 5 | 2 | 2 | |
| 5 | 89 | 5 | 3 | 3 | |
| 6 | 90 | 6 | 2 | 2 | |
| 7 | 89 | 6 | 2 | 3 | |
| 8 | 89 | 6 | 3 | 2 | |
| Centroid | 9 | 90 | 5 | 2.5 | 2.5 |
| Parameter Group | Parameter | Unit | Sample | Optimization Criteria |
|---|---|---|---|---|
| Feasibility | Viscosity at shear rate = 10 s−1 | Pa·s | Slurry | Nominal-better |
| Slope of the viscosity with the shear rate | Pa·s2 | Slurry | Higher-better | |
| Peel resistance | N·m−1 | Electrode | Higher-better | |
| Electrochemical performance | Discharge capacity at 0.2 C | mAh·g−1 | Half coin cell | Higher-better |
| Discharge capacity at 2 C | mAh·g−1 | Half coin cell | Higher-better | |
| Discharge capacity after fast cycles (at 1 C) | mAh·g−1 | Half coin cell | Higher-better |
| Formulation | Feasibility | Electrochemical Performance | ||||
|---|---|---|---|---|---|---|
| Viscosity (Pa·s) | Slope Viscosity (Pa·s2) | Peel (N·m−1) | 0.2 C (mAh·g−1) | 2 C (mAh·g−1) | 1 C (mAh·g−1) | |
| 1 | 9.60 | 0.36 | 36.8 | 128.28 | 116.47 | 114.64 |
| 2 | 27.14 | 0.36 | 20.9 | 127.22 | 108.36 | 108.39 |
| 3 | 20.94 | 0.33 | 17.4 | 128.02 | 110.32 | 107.72 |
| 4 | 13.01 | 0.45 | 24.9 | 128.99 | 120.93 | 117.04 |
| 5 | 20.09 | 0.40 | 32.4 | 126.58 | 112.07 | 97.36 |
| 6 | 15.93 | 0.57 | 29.5 | 125.93 | 116.76 | 117.89 |
| 7 | 13.54 | 0.54 | 44.6 | 128.25 | 117.10 | 101.81 |
| 8 | 19.61 | 0.44 | 31.9 | 128.62 | 118.96 | 110.05 |
| 9 | 24.10 | 0.49 | 30.9 | 129.29 | 120.10 | 114.31 |
| Parameter | Input | Output | R2 |
|---|---|---|---|
| Viscosity at shear rate = 10 s−1 | CMC ↑ | ↑ | 99.79 |
| Slope of the viscosity with the shear rate | CB ↑ | ↑ | 99.57 |
| Peel resistance | CB ↓ | ↑ | 95.64 |
| Discharge capacity at 0.2 C | LNMO + CB ↑ | ↑ | 39.11 |
| Discharge capacity at 2 C | LNMO + CB ↑ | ↑ | 90.57 |
| Discharge capacity after fast cycles (at 1 C) | LNMO + CB ↑ | ↑ | 98.10 |
| Formulation | #10: 89.5% LNMO, 6% C45, 2.5% CMC and 2% Binder | #11: 89.5% LNMO, 6% C45, 3% CMC, and 1.5% Binder | ||
|---|---|---|---|---|
| Output | Desirability | Output | Desirability | |
| Viscosity (Pa·s) | 16.82 | 1 | 34.53 | 0.000 |
| Viscosity slope (Pa·s2) | 0.530 | 0.767 | 0.582 | 0.940 |
| Peel strength (N·m−1) | 29.6 | 0.490 | 28.9 | 0.473 |
| Specific capacity at 0.2 C (mAh·g−1) | 128.31 | 0.610 | 124.73 | 0.490 |
| Specific capacity at 2 C (mAh·g−1) | 120.84 | 1.000 | 115.58 | 0.780 |
| Specific capacity at 1 C (mAh·g−1) | 117.83 | 0.930 | 118.06 | 0.940 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lizaso, L.; Urdampilleta, I.; Bengoechea, M.; Boyano, I.; Grande, H.-J.; Landa-Medrano, I.; Eguia-Barrio, A.; de Meatza, I. Waterborne LiNi0.5Mn1.5O4 Cathode Formulation Optimization through Design of Experiments and Upscaling to 1 Ah Li-Ion Pouch Cells. Energies 2023, 16, 7327. https://doi.org/10.3390/en16217327
Lizaso L, Urdampilleta I, Bengoechea M, Boyano I, Grande H-J, Landa-Medrano I, Eguia-Barrio A, de Meatza I. Waterborne LiNi0.5Mn1.5O4 Cathode Formulation Optimization through Design of Experiments and Upscaling to 1 Ah Li-Ion Pouch Cells. Energies. 2023; 16(21):7327. https://doi.org/10.3390/en16217327
Chicago/Turabian StyleLizaso, Lander, Idoia Urdampilleta, Miguel Bengoechea, Iker Boyano, Hans-Jürgen Grande, Imanol Landa-Medrano, Aitor Eguia-Barrio, and Iratxe de Meatza. 2023. "Waterborne LiNi0.5Mn1.5O4 Cathode Formulation Optimization through Design of Experiments and Upscaling to 1 Ah Li-Ion Pouch Cells" Energies 16, no. 21: 7327. https://doi.org/10.3390/en16217327
APA StyleLizaso, L., Urdampilleta, I., Bengoechea, M., Boyano, I., Grande, H.-J., Landa-Medrano, I., Eguia-Barrio, A., & de Meatza, I. (2023). Waterborne LiNi0.5Mn1.5O4 Cathode Formulation Optimization through Design of Experiments and Upscaling to 1 Ah Li-Ion Pouch Cells. Energies, 16(21), 7327. https://doi.org/10.3390/en16217327







