Synergistic Effect of Water-Soluble Hydroxylated Multi-Wall Carbon Nanotubes and Graphene Nanoribbons Coupled with Tetra Butyl Ammonium Bromide on Kinetics of Carbon Dioxide Hydrate Formation
Abstract
:1. Introduction
2. Materials and Methods
2.1. Experimental Materials
2.2. Experimental Apparatus
2.3. Experimental Procedures
3. Results and Discussions
3.1. Phase Equilibrium of Hydrate Formation
3.2. Hydrate Formation Kinetics
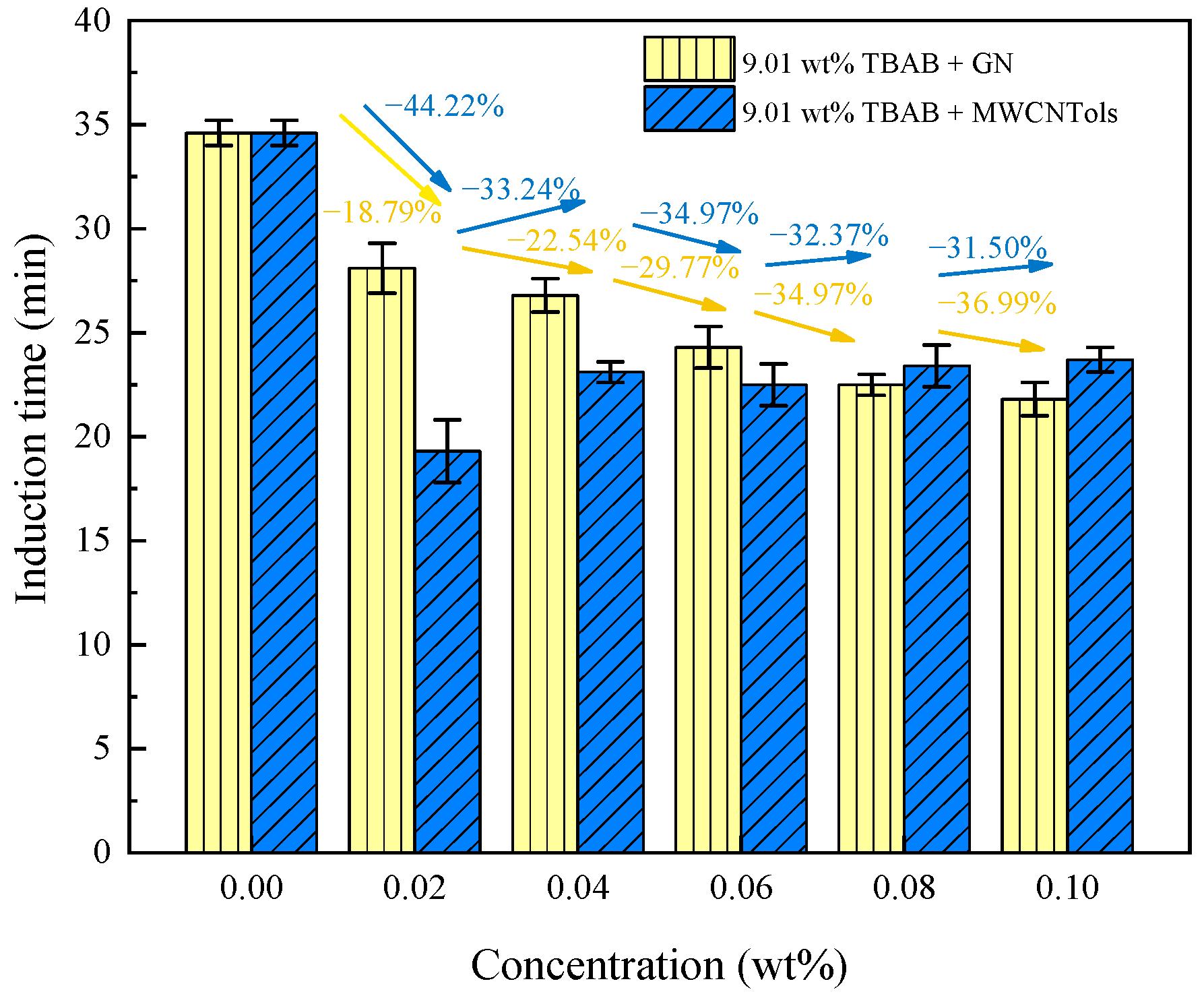
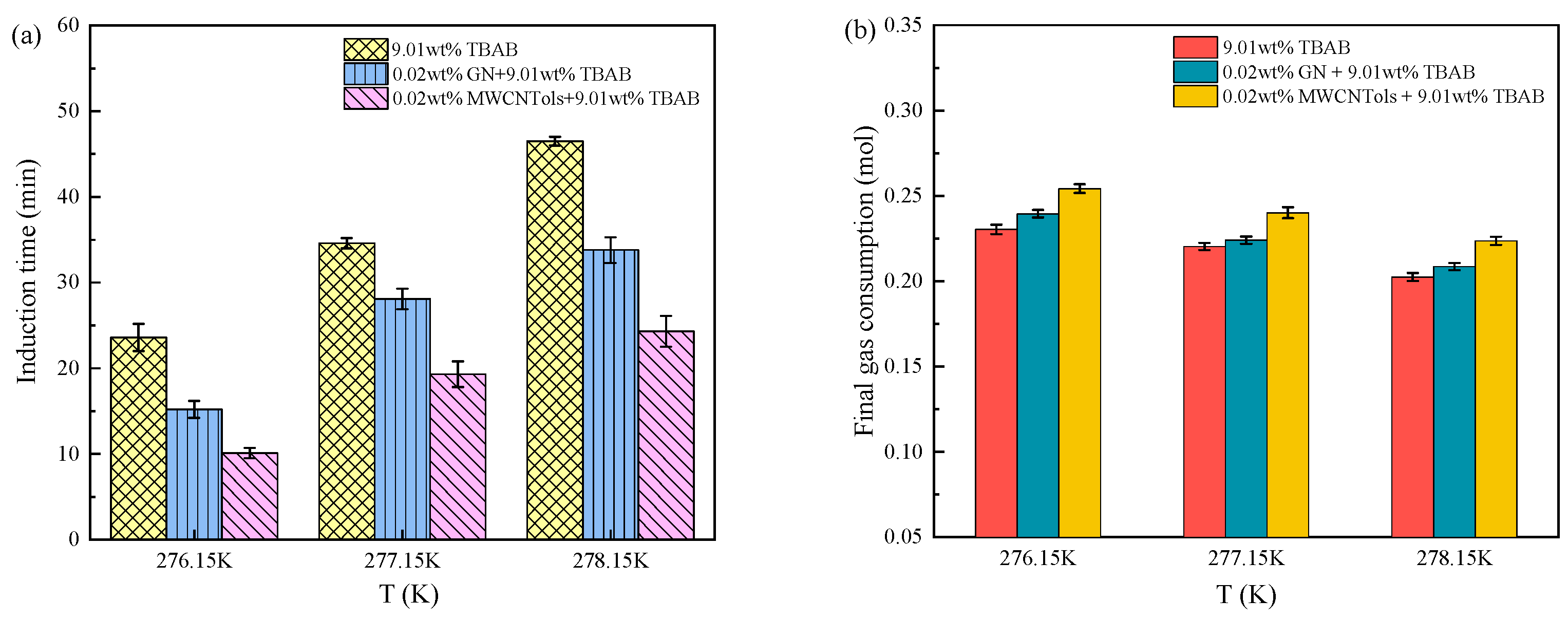
3.2.1. Induction Time
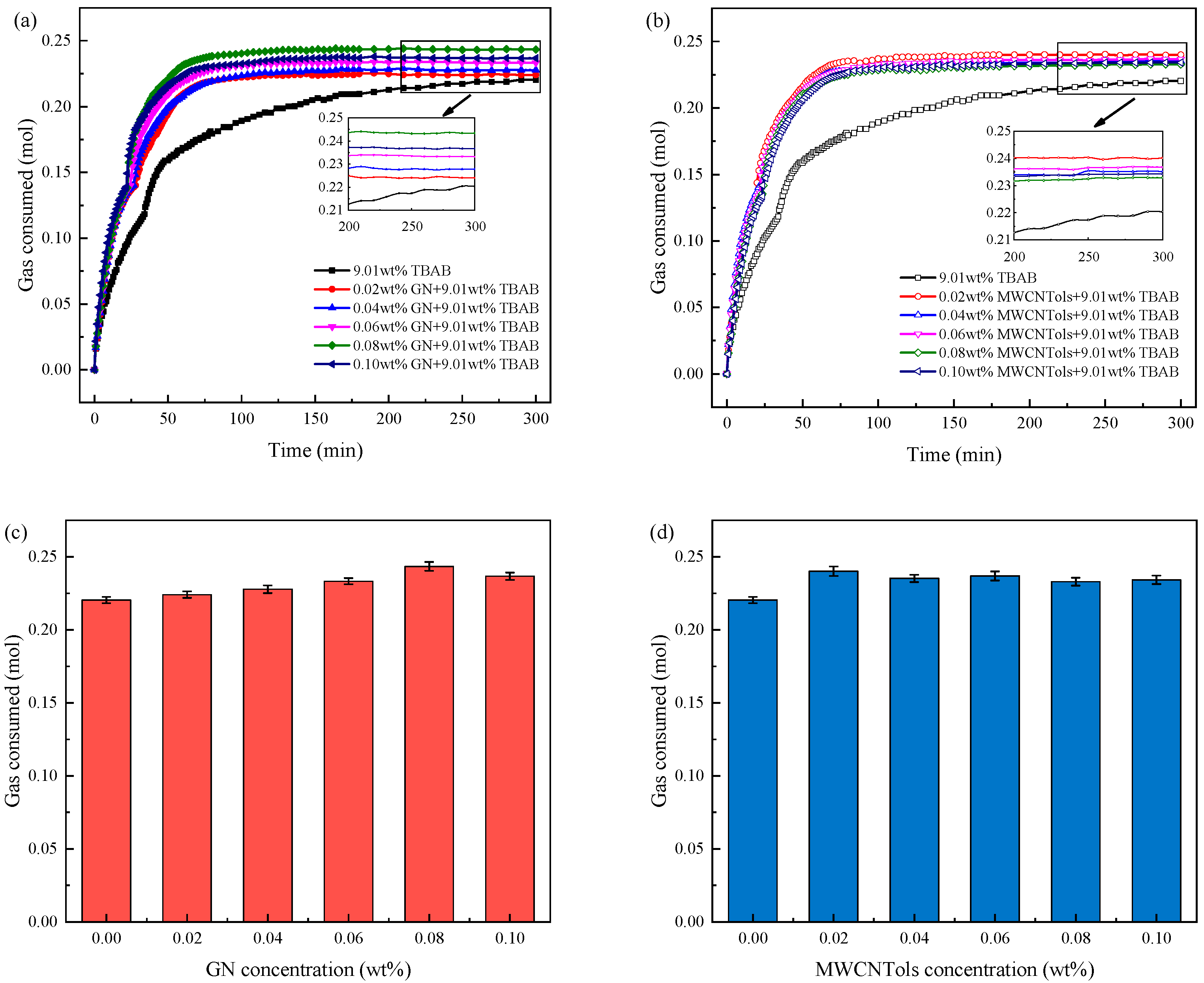
3.2.2. Gas Consumption
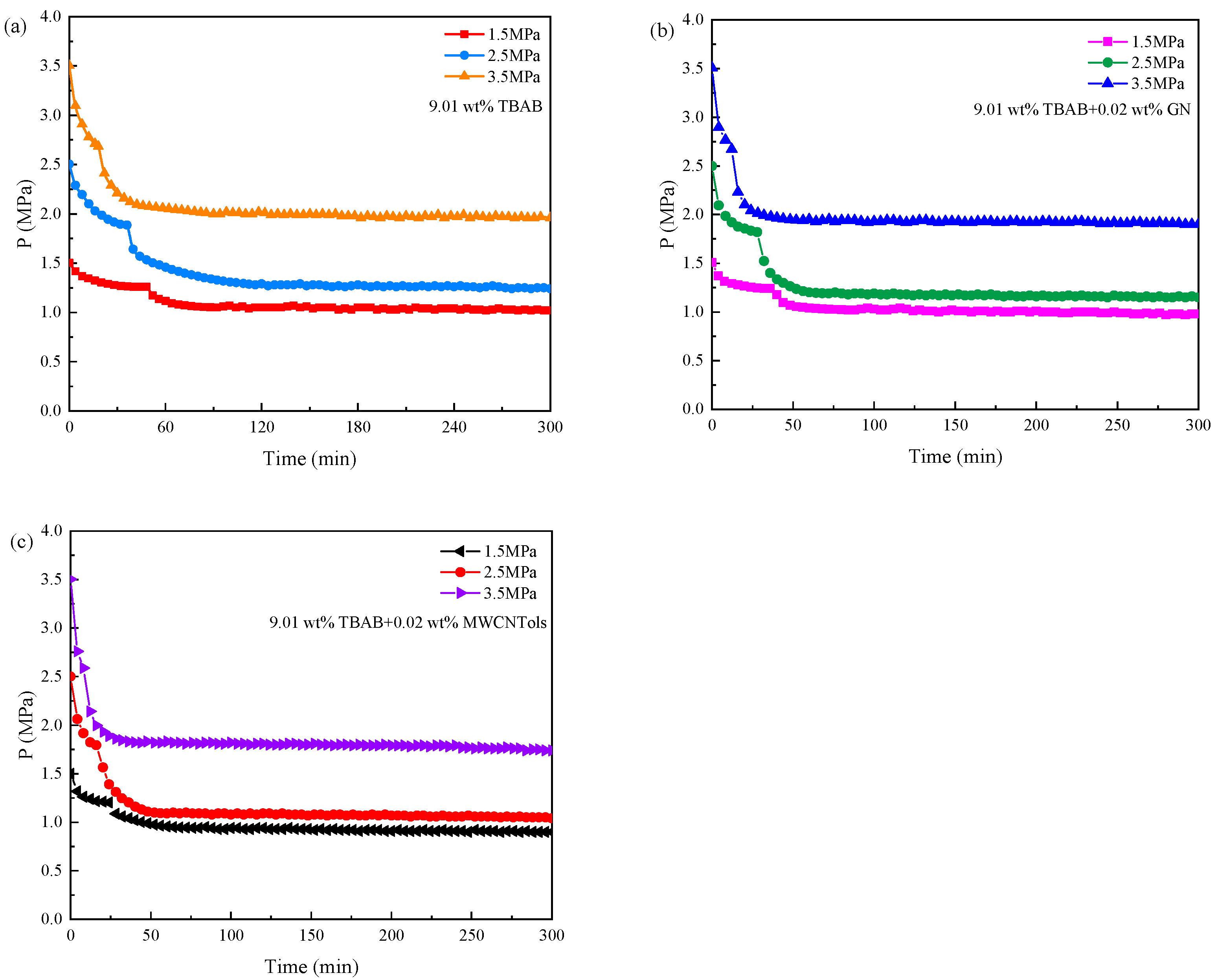
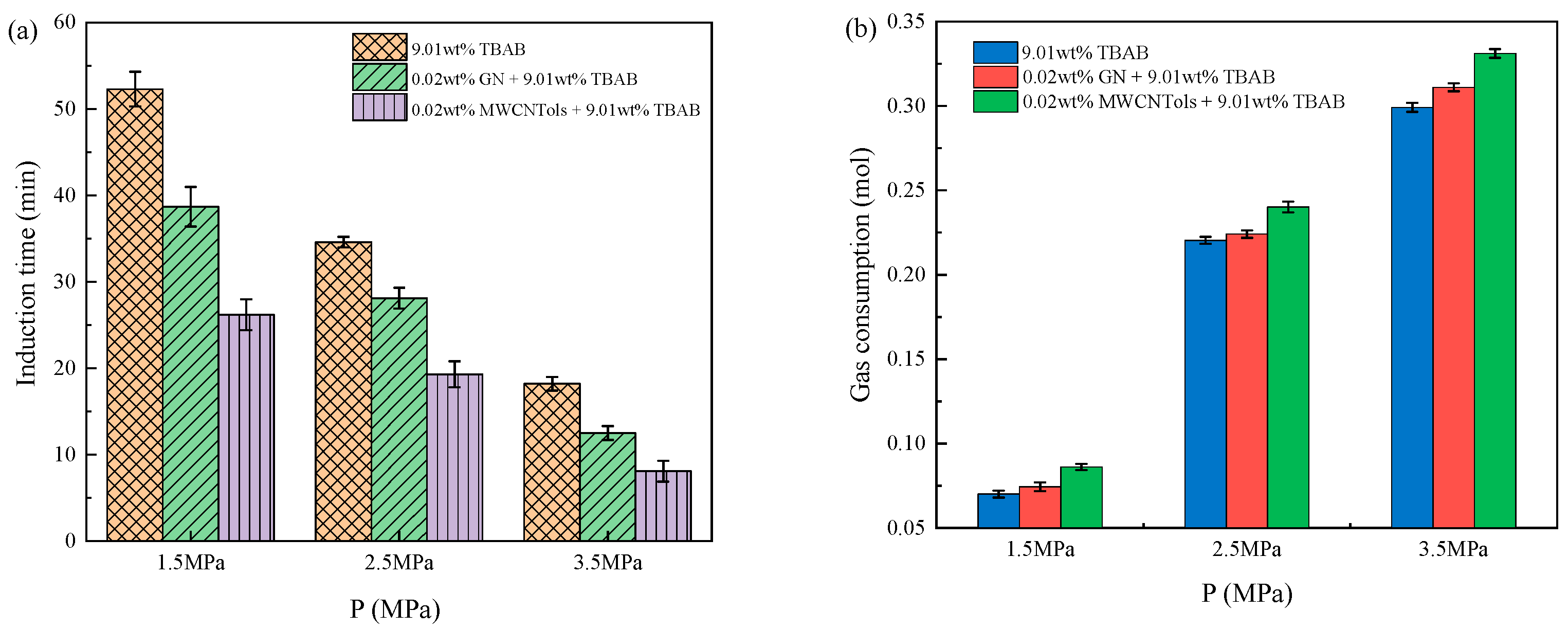
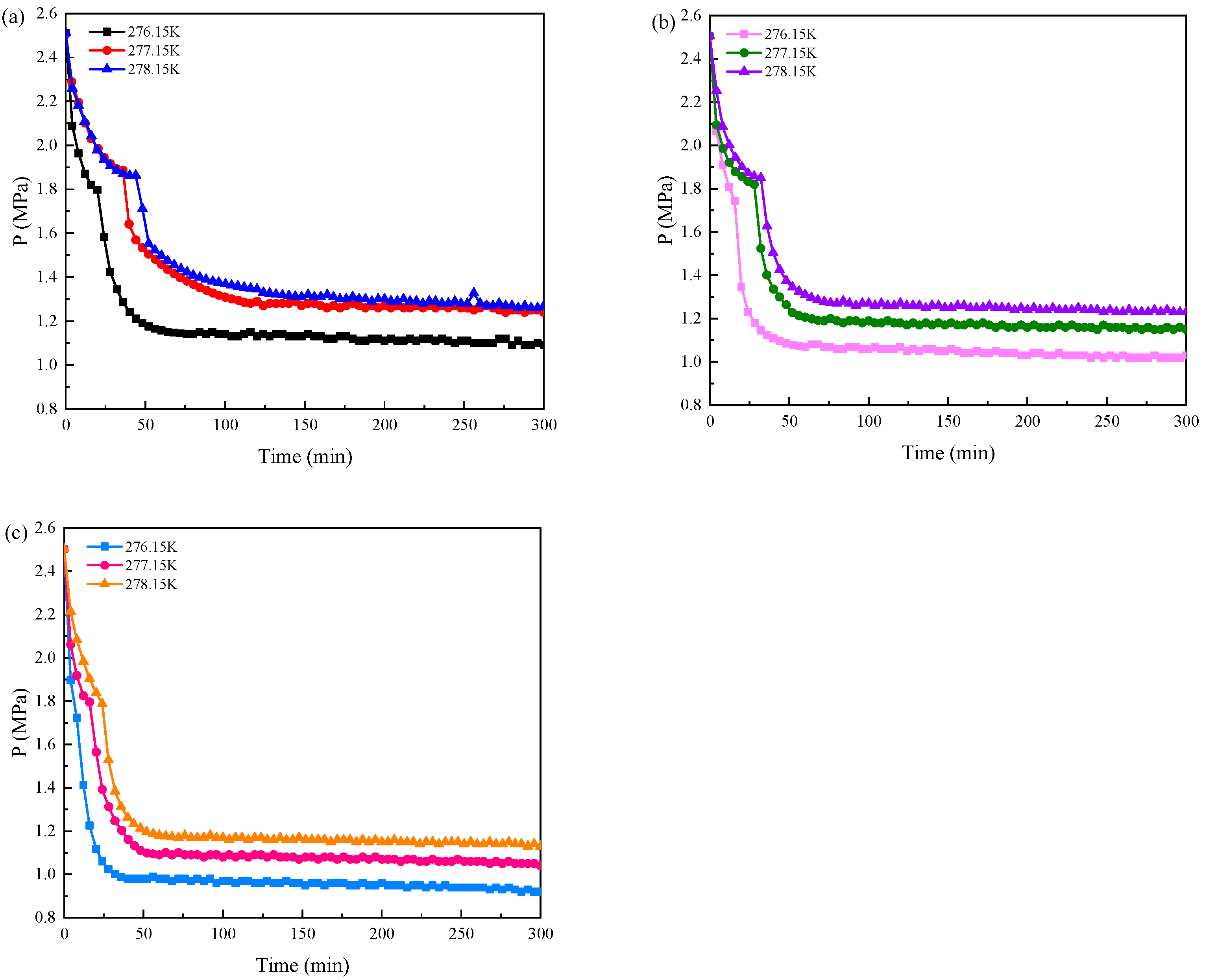
3.3. Pressure and Temperature Conditions
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Kazemifar, F. A review of technologies for carbon capture, sequestration, and utilization: Cost, capacity, and technology readiness. Greenh. Gases Sci. Technol. 2023, 12, 200–230. [Google Scholar] [CrossRef]
- Lang, X.; Fan, S.; Wang, Y. Intensification of methane and hydrogen storage in clathrate hydrate and future prospect. J. Nat. Gas Chem. 2010, 19, 203–209. [Google Scholar] [CrossRef]
- Ge, B.-B.; Li, X.-Y.; Zhong, D.-L.; Lu, Y.-Y. Investigation of natural gas storage and transportation by gas hydrate formation in the presence of bio-surfactant sulfonated lignin. Energy 2022, 244, 122665. [Google Scholar] [CrossRef]
- Chi, Y.; Sarica, C.; Daraboina, N. Experimental investigation of two-phase gas-oil stratified flow wax deposition in pipeline. Fuel 2019, 247, 113–125. [Google Scholar] [CrossRef]
- Ayaz, M.; Namazi, M.A.; Din, M.A.; Ershath, M.I.M.; Mansour, A.; Aggoune, e.-H.M. Sustainable seawater desalination: Current status, environmental implications and future expectations. Desalination 2022, 540, 116022. [Google Scholar] [CrossRef]
- Zhou, S.; Yan, H.; Su, D.; Navaneethakannan, S.; Chi, Y. Investigation on the kinetics of carbon dioxide hydrate formation using flow loop testing. J. Nat. Gas Ence Eng. 2017, 49, 385–392. [Google Scholar] [CrossRef]
- Kang, K.C.; Linga, P.; Park, K.N.; Choi, S.J.; Ju, D.L. Seawater desalination by gas hydrate process and removal characteristics of dissolved ions (Na+, K+, Mg2+, Ca2+, B3+, Cl−, SO42−). Desalination 2014, 353, 84–90. [Google Scholar] [CrossRef]
- Choi, S.; Park, J.; Park, J.H.; Kim, S.-C.; Won, S.O.; Kang, Y.T. Study on CO2 hydrate formation characteristics with promoters for CO2 capture and cold thermal energy transportation. J. Clean. Prod. 2021, 295, 126392. [Google Scholar] [CrossRef]
- Chi, Y.D.; Daraboina, N.; Sarica, C. Investigation of inhibitors efficacy in wax deposition mitigation using a laboratory scale flow loop. Aiche J. 2016, 62, 4131–4139. [Google Scholar] [CrossRef]
- Chi, Y.D.; Daraboina, N.; Sarica, C. Effect of the Flow Field on the Wax Deposition and Performance of Wax Inhibitors: Cold Finger and Flow Loop Testing. Energy Fuels 2017, 31, 4915–4924. [Google Scholar] [CrossRef]
- Herzog, H.; Dan, G. Carbon Capture and Storage from Fossil Fuel Use. Encycl. Energy 2004, 51, 277–287. [Google Scholar]
- Dubey, A.; Chi, Y.; Daraboina, N. Investigating the Performance of Paraffin Inhibitors under Different Operating Conditions. In Proceedings of the Spe Technical Conference & Exhibition, San Antonio, TX, USA, 9–11 October 2017. [Google Scholar]
- Koh, D.-Y.; Kang, H.; Kim, D.-O.; Park, J.; Cha, M.; Lee, H. Recovery of Methane from Gas Hydrates Intercalated within Natural Sediments Using CO2 and a CO2/N2 Gas Mixture. Chemsuschem 2012, 5, 1443–1448. [Google Scholar] [CrossRef]
- Lee, H.; Lee, J.W.; Kim, D.Y.; Park, J.; Seo, Y.T.; Zeng, H.; Moudrakovski, I.L.; Ratcliffe, C.I.; Ripmeester, J.A. Tuning clathrate hydrates for hydrogen storage. Nature 2005, 434, 743–746. [Google Scholar] [CrossRef]
- Nogami, T.; Oya, N.; Ishida, H.; Matsumoto, H. Development of natural gas ocean transportation chain by means of natural gas hydrate (NGH). Drug Metab. Pharmacokinet. 2008, 9, 184–187. [Google Scholar]
- Zhou, S.; Jiang, K.; Zhao, Y.; Chi, Y.; Wang, S.; Zhang, G. Experimental Investigation of CO2 Hydrate Formation in the Water Containing Graphite Nanoparticles and Tetra-n-butyl Ammonium Bromide. J. Chem. Eng. Data ACS J. Data 2018, 2, 63. [Google Scholar] [CrossRef]
- Qin, Y.; Bao, R.; Zhou, L.; Yang, X.; Liu, C.; Wan, W.; Chen, Y.; Guo, Y. Enhanced formation kinetics of hydrates using 1,3-dioxolane, l-leucine, and β-cyclodextrin. Fuel 2023, 332, 126165. [Google Scholar] [CrossRef]
- Du, S.; Han, X.; Cai, W.; Zhu, J.; Ma, X.; Han, S.; Chen, D.; Zhao, Y.; Li, H.; Lu, H.; et al. Formation of the structure-II gas hydrate from low-concentration propane mixed with methane. Chin. J. Chem. Eng. 2023, 58, 306–314. [Google Scholar] [CrossRef]
- Shirota, H.; Aya, I.; Namie, S. Measurement of methane hydrate dissociation for application to natural gas storage and transportation. In Proceedings of the Fourth International Conference on Gas Hydrates, Yokohama, Japan, 19–23 May 2002. [Google Scholar]
- Sun, X.; Liu, D. Advances in carbon dioxide hydrate kinetic additives. Chem. Ind. Eng. Prog. 2018, 37, 517–524. [Google Scholar]
- Liu, X.; Ren, J.; Chen, D.; Yin, Z. Comparison of SDS and L-Methionine in promoting CO2 hydrate kinetics: Implication for hydrate-based CO2 storage. Chem. Eng. J. 2022, 438, 135504. [Google Scholar] [CrossRef]
- Meshram, S.B.; Sardar, H.; Kushwaha, O.S.; Sangwai, J.; Kumar, R. A systematic molecular investigation on Sodium Dodecyl Benzene Sulphonate (SDBS) as a Low Dosage Hydrate Inhibitor (LDHI) and the role of Benzene Ring in the structure. J. Mol. Liq. 2021, 337, 116374. [Google Scholar] [CrossRef]
- Sun, D.; Englezos, P. Storage of CO2 in a partially water saturated porous medium at gas hydrate formation conditions. Int. J. Greenh. Gas Control. 2014, 25, 1–8. [Google Scholar] [CrossRef]
- Kalogerakis, N.; Jamaluddin, A.; Dholabhai, P.D.; Bishnoi, P.R. Effect of Surfactants on Hydrate Formation Kinetics. In Proceedings of the SPE International Conference on Oilfield Chemistry, New Orleans, LA, USA, 2–5 March 1993. [Google Scholar]
- Okutani, K.; Kuwabara, Y.; Mori, Y.H. Surfactant effects on hydrate formation in an unstirred gas/liquid system: An experimental study using methane and sodium alkyl sulfates. Chem. Eng. Sci. 2008, 63, 183–194. [Google Scholar] [CrossRef]
- Liu, N.; Lai, X.; Bai, F. CO2 Separation From Gas Mixtures by Clathrate Crystallization. Proc. Csee 2017, 37, 2312–2318. [Google Scholar]
- Hawkins, R.E.; Davidson, D.W. Dielectric relaxation in the clathrate hydrates of some cyclic ethers. Phys. Chem 1966, 70, 1889–1894. [Google Scholar] [CrossRef]
- Lee, Y.-J.; Kawamura, T.; Yamamoto, Y.; Yoon, J.-H. Phase Equilibrium Studies of Tetrahydrofuran (THF) + CH4, THF + CO2, CH4 + CO2, and THF + CO2 + CH4 Hydrates. J. Chem. Eng. Data 2012, 57, 3543–3548. [Google Scholar] [CrossRef]
- Yang, M.; Jing, W.; Wang, P.; Jiang, L.; Song, Y. Effects of an additive mixture (THF+TBAB) on CO2 hydrate phase equilibrium. Fluid Phase Equilibria 2015, 401, 27–33. [Google Scholar] [CrossRef]
- Seo, Y.T.; Moudrakovski, I.L.; Ripmeester, J.A.; Lee, J.W.; Lee, H. Efficient recovery of CO2 from flue gas by clathrate hydrate formation in porous silica gels. Environ. Sci. Technol. 2005, 39, 2315–2319. [Google Scholar] [CrossRef] [Green Version]
- Zhang, X.; Li, J.; Wu, Q.; Wang, C.; Nan, J. Experimental Study on the Formation Process of Carbon Dioxide Hydrate in Porous Media. J. Basic Ence Eng. 2016. [Google Scholar]
- Wang, J.; Wang, R.; Yoon, R.H.; Seol, Y. Use of Hydrophobic Particles as Kinetic Promoters for Gas Hydrate Formation. J. Chem. Eng. Data 2015, 60, 383–388. [Google Scholar] [CrossRef]
- Shi, B.H.; Fan, S.S.; Lou, X. Application of the shrinking-core model to the kinetics of repeated formation of methane hydrates in a system of mixed dry-water and porous hydrogel particulates. Chem. Eng. Sci. 2014, 109, 315–325. [Google Scholar] [CrossRef]
- Xuan, Y.M. An overview on nanofluids and applications. Sci. Sin. Technol. 2014, 44, 269–279. [Google Scholar] [CrossRef]
- Park, S.S.; Lee, S.B.; Kim, N.J. Effect of multi-walled carbon nanotubes on methane hydrate formation. J. Ind. Eng. Chem. 2010, 16, 551–555. [Google Scholar] [CrossRef]
- Renault-Crispo, J.S.; Coulombe, S.; Servio, P. Kinetics of carbon dioxide gas hydrates with tetrabutylammonium bromide and functionalized multi-walled carbon nanotubes. Energy 2017, 128, 414–420. [Google Scholar] [CrossRef]
- Aliabadi, M.; Rasoolzadeh, A.; Esmaeilzadeh, F.; Alamdari, A.M. Experimental study of using CuO nanoparticles as a methane hydrate promoter. J. Nat. Gas Sci. Eng. 2015, 27, 1518–1522. [Google Scholar] [CrossRef]
- Nesterov, A.N.; Reshetnikov, A.M.; Manakov, A.Y.; Rodionova, T.V.; Paukshtis, E.A.; Asanov, I.P.; Bardakhanov, S.P.; Bulavchenko, A.I. Promotion and inhibition of gas hydrate formation by oxide powders. J. Mol. Liq. 2015, 204, 118–125. [Google Scholar] [CrossRef]
- Mohammadi, A.; Manteghian, M.; Haghtalab, A.; Mohammadi, A.H.; Rahmati-Abkenar, M. Kinetic study of carbon dioxide hydrate formation in presence of silver nanoparticles and SDS. Chem. Eng. J. 2014, 237, 387–395. [Google Scholar] [CrossRef]
- Lee, J.D.; Kim, H.C.; Kim, Y.S.; Kim, Y.D.; Lee, M.S. Synthesis of Nanosized TiO2-Ag-SiO2 Sols by Modified Sol-Gel Method and their Application for Methane Hydrate Formation. Solid State Phenom. 2007, 124–126, 1059–1062. [Google Scholar] [CrossRef]
- Said, S.; Govindaraj, V.; Herri, J.M.; Ouabbas, Y.; Khodja, M.; Belloum, M.; Sangwai, J.S.; Nagarajan, R. A study on the influence of nanofluids on gas hydrate formation kinetics and their potential: Application to the CO2 capture process. J. Nat. Gas Sci. Eng. 2016, 32, 95–108. [Google Scholar] [CrossRef]
- Yu, Y.; Zhou, S.; Li, X.; Wang, S. Effect of graphite nanoparticles on CO2 hydrate phase equilibrium. Fluid Phase Equilibria 2016, 414, 23–28. [Google Scholar] [CrossRef]
- Zhou, S.D.; Yu, Y.S.; Zhao, M.M.; Wang, S.L.; Zhang, G.Z. Effect of Graphite Nanoparticles on Promoting CO2 Hydrate Formation. Energy Fuels 2014, 28, 4694–4698. [Google Scholar] [CrossRef]
- Mohammadi, A.H.; Eslamimanesh, A.; Richon, D. Semi-clathrate hydrate phase equilibrium measurements for the CO2+H2/CH4+tetra-n-butylammonium bromide aqueous solution system. Chem. Eng. Sci. 2013, 94, 284–290. [Google Scholar] [CrossRef]
- Holloway, S.; Darling, G. The structure of water on the (0001) surface of graphite. Surf. Sci. 2003, 532, 166–172. [Google Scholar]
- Agrawal, K.V.; Shimizu, S.; Drahushuk, L.W.; Kilcoyne, D.; Strano, M.S. Observation of extreme phase transition temperatures of water confined inside isolated carbon nanotubes. Nat. Nanotechnol. 2016, 12, 267–273. [Google Scholar] [CrossRef]
- Shu-Mei, S.; Ye, Z.; Dong-Liang, L.I.; De-Qing, L.; Xiao-Ping, W.U. Promotion effect of multi-walled carbon nanotubes on CH4-CO2-TBAB hydrates formation. Nat. Gas Chem. Ind. 2019. [Google Scholar]
- Kashchiev, D.; Firoozabadi, A. Induction time in crystallization of gas hydrates. J. Cryst. Growth 2003, 250, 499–515. [Google Scholar] [CrossRef]
- Tao, J.; Rui-Xiang, W. Model for predicting thermal conductivity of CNT water-based nanofluids. J. Therm. Sci. Technol. 2014, 1, 381–390. [Google Scholar]
- Smith, J.M. Introduction to chemical engineering thermodynamics. J. Chem. Educ. 1950, 27, 584. [Google Scholar] [CrossRef] [Green Version]
- Peng, D.Y.; Robinson, D.B. New Two-Constant Equation of State. Ind. Eng. Chem. Fundam. 1976, 15, 3069–3078. [Google Scholar] [CrossRef]
- Song, Y.; Wang, F.; Liu, G.; Luo, S.; Guo, R. Promotion Effect of Carbon Nanotubes-Doped SDS on Methane Hydrate Formation. Energy Fuels 2017, 31, 1850–1857. [Google Scholar] [CrossRef]
- Xia, Z.; Li, Z.; Chen, Z.; Li, X.; Zhang, Y.; Yan, K.; Lv, Q. CO2/H2/H2O Hydrate Formation with TBAB and Nanoporous Materials. Energy Procedia 2019, 158, 5866–5871. [Google Scholar] [CrossRef]
- Englezos, P.; Kalogerakis, N.; Dholabhai, P.D.; Bishnoi, P.R. Kinetics of formation of methane and ethane gas hydrates. Chem. Eng. Sci. 1987, 42, 2647–2658. [Google Scholar] [CrossRef]
- Li, X.G. Studies on the Formation Affecting Factor and Stability of Natural Gas Hydrates; China University of Petroleum (East China): Qingdao, China, 2008. [Google Scholar]


| Runs | Concentration (wt%) | Gas Consumption (mol) | Induction Time (min) |
|---|---|---|---|
| Run 1 | 0.00 | 0.2204 ± 0.0021 | 34.6 ± 0.6 |
| Run 2 | 0.02 MWCNTols | 0.2401 ± 0.0032 | 19.3 ± 1.5 |
| Run 3 | 0.04 MWCNTols | 0.2352 ± 0.0025 | 23.1 ± 0.5 |
| Run 4 | 0.06 MWCNTols | 0.2368 ± 0.0031 | 22.5 ± 1.0 |
| Run 5 | 0.08 MWCNTols | 0.2329 ± 0.0027 | 23.4 ± 1.0 |
| Run 6 | 0.10 MWCNTols | 0.2342 ± 0.0029 | 23.7 ± 0.6 |
| Run 7 | 0.02 GN | 0.2241 ± 0.0022 | 28.1 ± 1.2 |
| Run 8 | 0.04 GN | 0.2278 ± 0.0026 | 26.8 ± 0.8 |
| Run 9 | 0.06 GN | 0.2333 ± 0.0021 | 24.3 ± 1.0 |
| Run 10 | 0.08 GN | 0.2434 ± 0.0030 | 22.5 ± 0.5 |
| Run 11 | 0.10 GN | 0.2367 ± 0.0025 | 21.8 ± 0.8 |
| Runs | Systems | P (MPa) | T (K) | Gas Consumption (mol) | Induction Time (min) |
|---|---|---|---|---|---|
| Run 12 | 9.01 wt% TBAB | 1.5 | 277.15 | 0.0701 ± 0.0021 | 52.3 ± 2.0 |
| Run 13 | 9.01 wt% TBAB + 0.02 wt% MWCNTols | 1.5 | 277.15 | 0.0862 ± 0.0019 | 26.2 ± 1.8 |
| Run 14 | 9.01 wt% TBAB + 0.02 wt% GN | 1.5 | 277.15 | 0.0745 ± 0.0026 | 38.7 ± 2.3 |
| Run 15 | 9.01 wt% TBAB | 3.5 | 277.15 | 0.2991 ± 0.0027 | 18.2 ± 0.8 |
| Run 16 | 9.01 wt% TBAB + 0.02 wt% MWCNTols | 3.5 | 277.15 | 0.3311 ± 0.0026 | 8.1 ± 1.2 |
| Run 17 | 9.01 wt% TBAB + 0.02 wt% GN | 3.5 | 277.15 | 0.3110 ± 0.0023 | 12.5 ± 0.8 |
| Run 18 | 9.01 wt% TBAB | 2.5 | 276.15 | 0.2304 ± 0.0028 | 23.6 ± 1.6 |
| Run 19 | 9.01 wt% TBAB + 0.02 wt% MWCNTols | 2.5 | 276.15 | 0.2543 ± 0.0026 | 10.1 ± 0.6 |
| Run 20 | 9.01 wt% TBAB + 0.02 wt% GN | 2.5 | 276.15 | 0.2395 ± 0.0022 | 15.2 ± 1.0 |
| Run 21 | 9.01 wt% TBAB | 2.5 | 278.15 | 0.2025 ± 0.0023 | 46.5 ± 0.5 |
| Run 22 | 9.01 wt% TBAB + 0.02 wt% MWCNTols | 2.5 | 278.15 | 0.2237 ± 0.0024 | 24.3 ± 1.8 |
| Run 23 | 9.01 wt% TBAB + 0.02 wt% GN | 2.5 | 278.15 | 0.2086 ± 0.0021 | 33.8 ± 1.5 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, S.-L.; Xiao, Y.-Y.; Zhou, S.-D.; Jiang, K.; Yu, Y.-S.; Rao, Y.-C. Synergistic Effect of Water-Soluble Hydroxylated Multi-Wall Carbon Nanotubes and Graphene Nanoribbons Coupled with Tetra Butyl Ammonium Bromide on Kinetics of Carbon Dioxide Hydrate Formation. Energies 2023, 16, 5831. https://doi.org/10.3390/en16155831
Wang S-L, Xiao Y-Y, Zhou S-D, Jiang K, Yu Y-S, Rao Y-C. Synergistic Effect of Water-Soluble Hydroxylated Multi-Wall Carbon Nanotubes and Graphene Nanoribbons Coupled with Tetra Butyl Ammonium Bromide on Kinetics of Carbon Dioxide Hydrate Formation. Energies. 2023; 16(15):5831. https://doi.org/10.3390/en16155831
Chicago/Turabian StyleWang, Shu-Li, Yan-Yun Xiao, Shi-Dong Zhou, Kun Jiang, Yi-Song Yu, and Yong-Chao Rao. 2023. "Synergistic Effect of Water-Soluble Hydroxylated Multi-Wall Carbon Nanotubes and Graphene Nanoribbons Coupled with Tetra Butyl Ammonium Bromide on Kinetics of Carbon Dioxide Hydrate Formation" Energies 16, no. 15: 5831. https://doi.org/10.3390/en16155831
APA StyleWang, S.-L., Xiao, Y.-Y., Zhou, S.-D., Jiang, K., Yu, Y.-S., & Rao, Y.-C. (2023). Synergistic Effect of Water-Soluble Hydroxylated Multi-Wall Carbon Nanotubes and Graphene Nanoribbons Coupled with Tetra Butyl Ammonium Bromide on Kinetics of Carbon Dioxide Hydrate Formation. Energies, 16(15), 5831. https://doi.org/10.3390/en16155831






