Feasibility Study of the Solar-Promoted Photoreduction of CO2 to Liquid Fuels with Direct or Indirect Use of Renewable Energy Sources
Abstract
1. Introduction
2. Experimental
2.1. Photocatalysts
2.2. Photoreactor
3. Results and Discussion
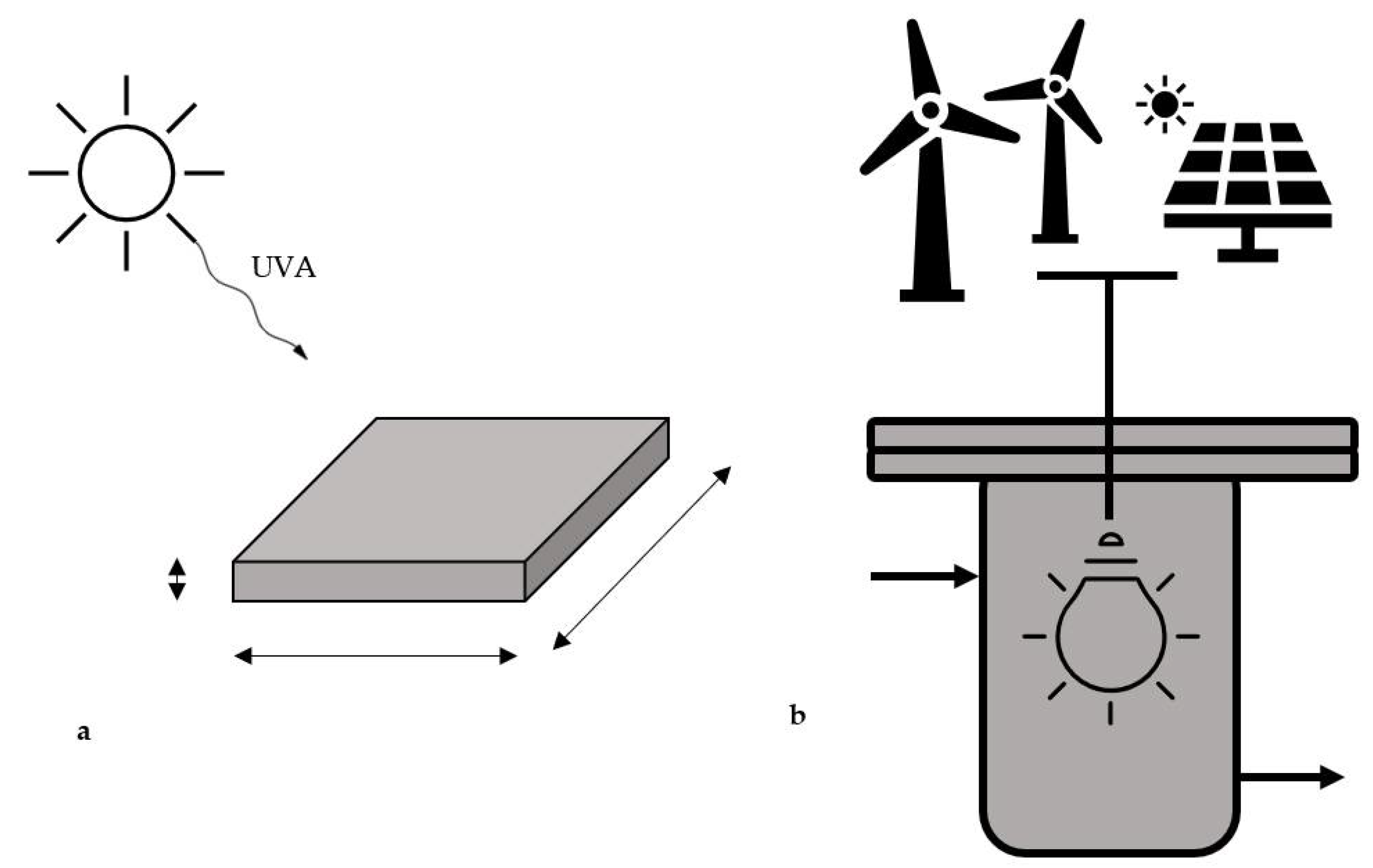
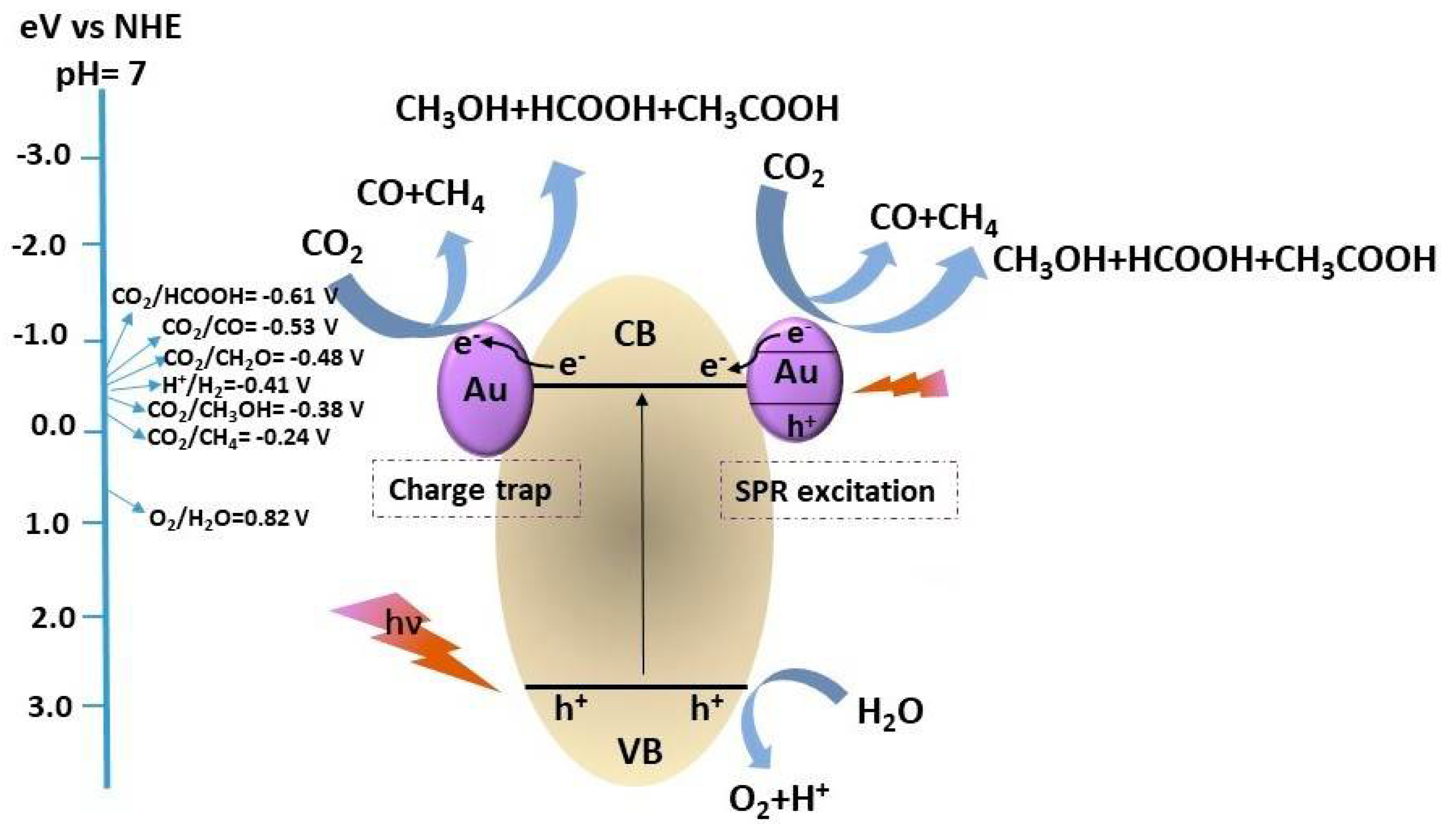
3.1. Photoreduction of CO2 in a Direct Solar Photoreactor (Case A)
- CO2 + 2H+ + 2e− → CO + H2O
- CO2 + 2H+ + 2e− → HCOOH
- CO2 + 4H+ + 4e− → HCHO + H2O
- CO2 + 6H+ + 6e− → CH3OH + H2O
- CO2 + 8 H+ + 8e− → CH4 + 2H2O
- 2H+ + 2e− → H2
3.2. Indirect Exploitation of Electric Energy from Renewable Sources
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Energy Information Administration Office of Integrated Analysis and Forecasting U.S. Department of Energy. International Energy Outlook; Energy Information Administration Office of Integrated Analysis and Forecasting U.S. Department of Energy: Washington, DC, USA, 2005.
- Kabir, E.; Kumar, P.; Kumar, S.; Adelodun, A.A.; Kim, K.H. Solar energy: Potential and future prospects. Renew. Sustain. Energy Rev. 2018. [CrossRef]
- Diffey, B.L. Solar Ultraviolet Radiation Effects on Biological Systems. Phys. Med. Biol. 1991, 36, 299. [Google Scholar] [CrossRef]
- Fioletov, V.E.; Bodeker, G.E.; Miller, A.J.; McPeters, R.D.; Stolarski, R. Global and zonal total ozone variations estimated from ground-based and satellite measurements: 1964-2000. J. Geophys. Res. Atmos. 2002, 107, 2615–2621. [Google Scholar] [CrossRef]
- Lincoln, S.F. Fossil Fuels in the 21st Century. Ambio 2005, 34, 621–627. [Google Scholar] [CrossRef] [PubMed]
- Turns, S.R. An Introduction to Combustion: Concepts and Applications; McGraw-Hill Higher Education: New York, NY, USA, 2000; ISBN 0-07-230096-5. [Google Scholar]
- Olajire, A.A. Valorization of greenhouse carbon dioxide emissions into value-added products by catalytic processes. J. CO2 Util. 2013, 3–4, 74–92. [Google Scholar] [CrossRef]
- Lee, S.Y.; Lee, J.U.; Lee, I.B.; Han, J. Design under uncertainty of carbon capture and storage infrastructure considering cost, environmental impact, and preference on risk. Appl. Energy 2017. [CrossRef]
- Lee, B.J.; Lee, J.I.; Yun, S.Y.; Lim, C.S.; Park, Y.K. Economic evaluation of carbon capture and utilization applying the technology of mineral carbonation at coal-fired power plant. Sustainability 2020, 12, 6175. [Google Scholar] [CrossRef]
- Pilorgé, H.; McQueen, N.; Maynard, D.; Psarras, P.; He, J.; Rufael, T.; Wilcox, J. Cost Analysis of Carbon Capture and Sequestration of Process Emissions from the U.S. Industrial Sector. Environ. Sci. Technol. 2020, 54, 7524–7532. [Google Scholar] [CrossRef]
- Indrakanti, V.P.; Kubicki, J.D.; Schobert, H.H. Photoinduced activation of CO2 on Ti-based heterogeneous catalysts: Current state, chemical physics-based insights and outlook. Energy Environ. Sci. 2009, 2, 745–758. [Google Scholar] [CrossRef]
- Liu, L.; Li, Y. Understanding the reaction mechanism of photocatalytic reduction of CO2 with H2O on TiO2-based photocatalysts: A review. Aerosol Air Qual. Res. 2014, 14, 453–469. [Google Scholar] [CrossRef]
- Schneider, J.; Matsuoka, M.; Takeuchi, M.; Zhang, J.; Horiuchi, Y.; Anpo, M.; Bahnemann, D.W. Understanding TiO2 Photocatalysis: Mechanisms and Materials. Chem. Rev. 2014, 114, 9919–9986. [Google Scholar] [CrossRef]
- Bahadori, E.; Tripodi, A.; Villa, A.; Pirola, C.; Prati, L.; Ramis, G.; Rossetti, I. High pressure photoreduction of CO2: Effect of catalyst formulation, hole scavenger addition and operating conditions. Catalysts 2018, 8, 430. [Google Scholar] [CrossRef]
- Kim, J.; Kwon, E.E. Photoconversion of carbon dioxide into fuels using semiconductors. J. CO2 Util. 2019, 33, 72–82. [Google Scholar] [CrossRef]
- Ulmer, U.; Dingle, T.; Duchesne, P.N.; Morris, R.H.; Tavasoli, A.; Wood, T.; Ozin, G.A. Fundamentals and applications of photocatalytic CO2 methanation. Nat. Commun. 2019, 10. [Google Scholar] [CrossRef] [PubMed]
- Murcia Valderrama, M.A.; van Putten, R.-J.; Gruter, G.-J.M. The potential of oxalic-and glycolic acid based polyesters (review). Towards CO2 as a feedstock (Carbon Capture and Utilization-CCU). Eur. Polym. J. 2019, 119, 445–468. [Google Scholar] [CrossRef]
- Wang, C.; Sun, Z.; Zheng, Y.; Hu, Y.H. Recent Progress in Visible Light Photocatalytic Conversion of Carbon Dioxide. J. Mater. Chem. A 2019, 7, 865–887. [Google Scholar] [CrossRef]
- Pomilla, F.R.; Brunetti, A.; Marcì, G.; Garcĺa-López, E.I.; Fontananova, E.; Palmisano, L.; Barbieri, G. CO2 to Liquid Fuels: Photocatalytic Conversion in a Continuous Membrane Reactor. ACS Sustain. Chem. Eng. 2018, 6, 8743–8753. [Google Scholar] [CrossRef]
- Rafiee, A.; Rajab Khalilpour, K.; Milani, D.; Panahi, M. Trends in CO2 conversion and utilization: A review from process systems perspective. J. Environ. Chem. Eng. 2018, 6, 5771–5794. [Google Scholar] [CrossRef]
- Richter, A.; Hermle, M.; Glunz, S.W. Reassessment of the limiting efficiency for crystalline silicon solar cells. IEEE J. Photovoltaics 2013, 3, 1184–1191. [Google Scholar] [CrossRef]
- Chaudhery Mustansar, H.; Ajay Kumar, M. Handbook of Smart Photocatalytic Materials; Elsevier: Amsterdam, The Netherlands, 2020; ISBN 9780128190517. [Google Scholar]
- Wang, P.; Yin, G.; Bi, Q.; Huang, X.; Du, X.; Zhao, W.; Huang, F. Efficient Photocatalytic Reduction of CO2 Using Carbon-Doped Amorphous Titanium Oxide. ChemCatChem 2018, 10, 3854–3861. [Google Scholar] [CrossRef]
- Meng, A.; Wu, S.; Cheng, B.; Yu, J.; Xu, J. Hierarchical TiO2/Ni(OH)2composite fibers with enhanced photocatalytic CO2reduction performance. J. Mater. Chem. A 2018, 6, 4729–4736. [Google Scholar] [CrossRef]
- Wei, L.; Yu, C.; Zhang, Q.; Liu, H.; Wang, Y. TiO2-based heterojunction photocatalysts for photocatalytic reduction of CO2 into solar fuels. J. Mater. Chem. A 2018, 22411–22436. [Google Scholar] [CrossRef]
- Zhang, S.; Yin, X.; Zheng, Y. Enhanced photocatalytic reduction of CO2to methanol by ZnO nanoparticles deposited on ZnSe nanosheet. Chem. Phys. Lett. 2018, 693, 170–175. [Google Scholar] [CrossRef]
- Wang, W.; Xu, D.; Cheng, B.; Yu, J.; Jiang, C. Hybrid carbon@TiO2hollow spheres with enhanced photocatalytic CO2 reduction activity. J. Mater. Chem. A 2017, 5, 5020–5029. [Google Scholar] [CrossRef]
- Gao, S.; Gu, B.; Jiao, X.; Sun, Y.; Zu, X.; Yang, F.; Zhu, W.; Wang, C.; Feng, Z.; Ye, B.; et al. Highly Efficient and Exceptionally Durable CO2Photoreduction to Methanol over Freestanding Defective Single-Unit-Cell Bismuth Vanadate Layers. J. Am. Chem. Soc. 2017, 139, 3438–3445. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Mohamed, H.H.; Dillert, R.; Bahnemann, D. Kinetics and mechanisms of charge transfer processes in photocatalytic systems: A review. J. Photochem. Photobiol. C Photochem. Rev. 2012, 13, 263–276. [Google Scholar] [CrossRef]
- Mills, A.; Le Hunte, S. An overview of semiconductor photocatalysis. J. Photochem. Photobiol. A Chem. 1997, 108, 1–35. [Google Scholar] [CrossRef]
- Bahadori, E.; Tripodi, A.; Villa, A.; Pirola, C.; Prati, L.; Ramis, G.; Dimitratos, N.; Wang, D.; Rossetti, I. High pressure CO2 photoreduction using Au/TiO2: Unravelling the effect of co-catalysts and of titania polymorphs. Catal. Sci. Technol. 2019, 9, 2253–2265. [Google Scholar] [CrossRef]
- Galli, F.; Compagnoni, M.; Vitali, D.; Pirola, C.; Bianchi, C.L.; Villa, A.; Prati, L.; Rossetti, I. CO2 photoreduction at high pressure to both gas and liquid products over titanium dioxide. Appl. Catal. B Environ. 2017, 200, 386–391. [Google Scholar] [CrossRef]
- Bahadori, E.; Ramis, G.; Zanardo, D.; Menegazzo, F.; Signoretto, M.; Gazzoli, D.; Pietrogiacomi, D.; Di Michele, A.; Rossetti, I. Photoreforming of glucose over CuO/TiO2. Catalysts 2020, 10, 477. [Google Scholar] [CrossRef]
- Ramis, G.; Bahadori, E.; Rossetti, I. Design of efficient photocatalytic processes for the production of hydrogen from biomass derived substrates. Int. J. H2 Energy 2021, 46, 12105–12116. [Google Scholar] [CrossRef]
- P25-EVONIK. Available online: https://products-re.evonik.com/lpa-productfinder/page/productsbytext/detail.html?xd_co_f=MGRkYTFiM2MtMWNiYi00ZTA1LTg5ZTktNjMyNTgyYWI3ODUz&channel=aerosil&pid=1822&lang=en (accessed on 1 February 2021).
- Chiarello, G.L.; Rossetti, I.; Forni, L. Flame-spray pyrolysis preparation of perovskites for methane catalytic combustion. J. Catal. 2005, 236, 251–261. [Google Scholar] [CrossRef]
- Sander, R. Compilation of Henry’s law constants (version 4.0) for water as solvent. Atmos. Chem. Phys. 2015, 15, 4399–4981. [Google Scholar] [CrossRef]
- Haimei, L.; Imanishi, A.; Nakato, Y. Mechanisms for photooxidation reactions of water and organic compounds on carbon-doped titanium dioxide, as studied by photocurrent measurements. J. Phys. Chem. C 2007, 111, 8603–8610. [Google Scholar] [CrossRef]
- Ola, O.; Maroto-Valer, M.M. Review of material design and reactor engineering on TiO2 photocatalysis for CO2 reduction. J. Photochem. Photobiol. C Photochem. Rev. 2015, 24, 16–42. [Google Scholar] [CrossRef]
- Schneider, J.T.; Firak, D.S.; Ribeiro, R.R.; Peralta-Zamora, P. Use of scavenger agents in heterogeneous photocatalysis: Truths, half-truths, and misinterpretations. Phys. Chem. Chem. Phys. 2020, 22, 15723–15733. [Google Scholar] [CrossRef]
- Boxwell, M. Solar Electricity Handbook: A Simple, Practical Guide to Solar Energy : How to Design and Install Photovoltaic Solar Electric Systems, 2012th ed.; Greenstream Publishing: Ryton-on-Dunsmore, UK, 2012; ISBN 1907670181. [Google Scholar]
- ARPA Environmental Measurements. Available online: http://www.arpalombardia.it/Pages/Metereologia/Richiesta-dati-misurati.aspx (accessed on 1 February 2021).
- Yan, H.; Wang, X.; Yao, M.; Yao, X. Band structure design of semiconductors for enhanced photocatalytic activity: The case of TiO2. Prog. Nat. Sci. Mater. Int. 2013, 23, 402–407. [Google Scholar] [CrossRef]
- Available online: https://www.engineeringtoolbox.com/fuels-higher-calorific-values-d_169.html (accessed on 1 February 2021).
- Rossetti, I.; Bahadori, E.; Tripodi, A.; Villa, A.; Prati, L.; Ramis, G. Conceptual design and feasibility assessment of photoreactors for solar energy storage. Sol. Energy 2018, 172, 225–231. [Google Scholar] [CrossRef]
- Shockley, W.; Queisser, H.J. Detailed balance limit of efficiency of p-n junction solar cells. J. Appl. Phys. 1961, 32, 510–519. [Google Scholar] [CrossRef]
- IEA-Energy Related CO2 Emissions. Available online: https://www.iea.org/data-and-statistics/charts/global-energy-related-co2-emissions-1990-2020 (accessed on 1 February 2021).
- Interactive Wind Atlas. Available online: https://www.mise.gov.it/index.php/itenergia/efficienza-energetica (accessed on 1 February 2021).
| Period | Mean Irradiance (W/m2) | UV Fraction (W/m2) | Monthly Energy (MJ/m2) | Monthly UV Energy (MJ/m2) |
|---|---|---|---|---|
| JAN | 53.1 | 3.2 | 142.2 | 8.5 |
| FEB | 77.6 | 4.7 | 207.8 | 11.3 |
| MAR | 109.0 | 6.5 | 291.9 | 17.5 |
| APR | 201.0 | 12.1 | 538.4 | 31.3 |
| MAY | 211.0 | 12.7 | 565.1 | 33.9 |
| JUN | 295.0 | 17.7 | 790.1 | 45.9 |
| JUL | 286.0 | 17.2 | 766.0 | 46.0 |
| AUG | 248.0 | 14.9 | 664.2 | 39.9 |
| SEP | 195.0 | 11.7 | 522.3 | 30.3 |
| OCT | 108.0 | 6.5 | 289.3 | 17.4 |
| NOV | 43.5 | 2.6 | 116.5 | 6.8 |
| DEC | 57.5 | 3.5 | 154.0 | 9.2 |
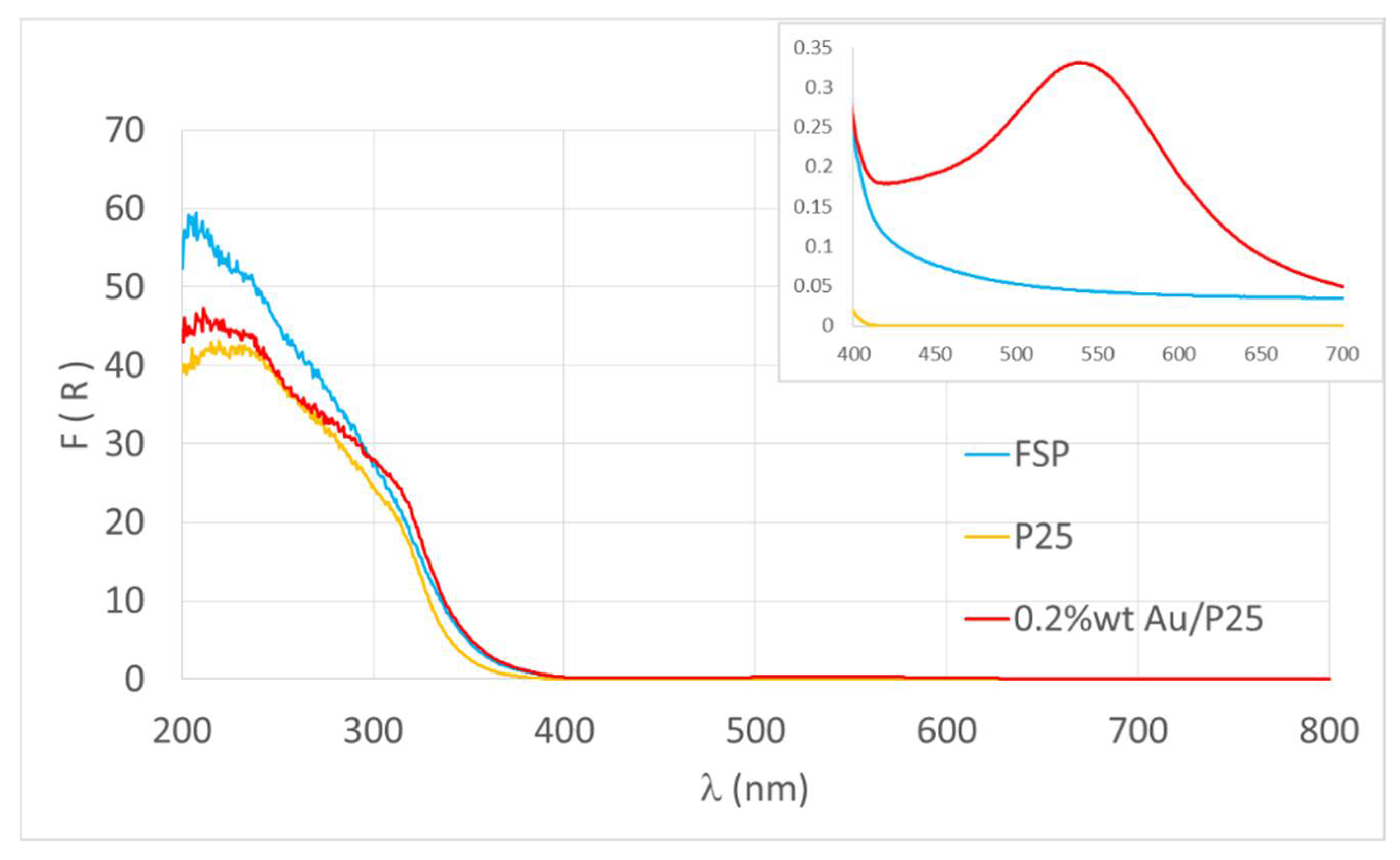
| Photocatalyst | BG (eV) | λmax (nm) | % UVA Absorbed |
|---|---|---|---|
| P25 | 3.41 | 364 | 64% |
| FSP | 3.31 | 375 | 75% |
| 0.2%wt/wt Au/P25 | 3.12 | 397 | 97% |
| Compound | Catalyst | Production Rate (mol/h kgcat) | Production Rate (kg/h kgcat) |
|---|---|---|---|
| H2 | P25 | 4.0000 | 0.0080 |
| FSP | 1.6100 | 0.0032 | |
| Au/P25 | 2.0200 | 0.0040 | |
| CO | P25 | 0.2290 | 0.0064 |
| FSP | 0.0660 | 0.0018 | |
| Au/P25 | 0.5410 | 0.0151 | |
| HCOOH | P25 | 39.4000 | 1.8124 |
| FSP | 7.4300 | 0.3418 | |
| Au/P25 | 6.9800 | 0.3211 | |
| HCHO | P25 | 0.0008 | 0.0000 |
| FSP | 0.0481 | 0.0014 | |
| Au/P25 | 0.0148 | 0.0004 | |
| CH3OH | P25 | 0.0006 | 0.0000 |
| FSP | 0.0000 | 0.0000 | |
| Au/P25 | 0.0802 | 0.0026 |
| Compound | Catalyst | Productivity (kg m2/MJ kgcat) | Yearly Productivity (kg/kgcat) |
|---|---|---|---|
| H2 | P25 | 0.015 | 2.82 |
| FSP | 0.0060 | 1.33 | |
| Au/P25 | 0.0075 | 2.16 | |
| CO | P25 | 0.012 | 2.26 |
| FSP | 0.0034 | 0.76 | |
| Au/P25 | 0.028 | 8.1 | |
| HCOOH | P25 | 3.36 | 639.8 |
| FSP | 0.63 | 141.4 | |
| Au/P25 | 0.59 | 171.8 | |
| HCHO | P25 | 0.00004 | 0.008 |
| FSP | 0.0027 | 0.60 | |
| Au/P25 | 0.00082 | 0.24 | |
| CH3OH | P25 | 0.00004 | 0.007 |
| FSP | 0.00000 | 0.000 | |
| Au/P25 | 0.0048 | 1.37 |
| Total Solar Energy | |||||
|---|---|---|---|---|---|
| ESCH2 LHV (MJ/kgcat) | ESCH2 HHV (MJ/kgcat) | ηH2 LHV | ηH2 HHV | ESCHCOOH (MJ/kgcat) | ηHCOOH |
| 338.6 | 400.6 | 0.10% | 0.12% | 3519 | 1.1% |
| UV solar energy | |||||
| 1.76% | 2.08% | 18.3% | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Conte, F.; Tripodi, A.; Rossetti, I.; Ramis, G. Feasibility Study of the Solar-Promoted Photoreduction of CO2 to Liquid Fuels with Direct or Indirect Use of Renewable Energy Sources. Energies 2021, 14, 2804. https://doi.org/10.3390/en14102804
Conte F, Tripodi A, Rossetti I, Ramis G. Feasibility Study of the Solar-Promoted Photoreduction of CO2 to Liquid Fuels with Direct or Indirect Use of Renewable Energy Sources. Energies. 2021; 14(10):2804. https://doi.org/10.3390/en14102804
Chicago/Turabian StyleConte, Francesco, Antonio Tripodi, Ilenia Rossetti, and Gianguido Ramis. 2021. "Feasibility Study of the Solar-Promoted Photoreduction of CO2 to Liquid Fuels with Direct or Indirect Use of Renewable Energy Sources" Energies 14, no. 10: 2804. https://doi.org/10.3390/en14102804
APA StyleConte, F., Tripodi, A., Rossetti, I., & Ramis, G. (2021). Feasibility Study of the Solar-Promoted Photoreduction of CO2 to Liquid Fuels with Direct or Indirect Use of Renewable Energy Sources. Energies, 14(10), 2804. https://doi.org/10.3390/en14102804











