Catalytic Steam Reforming of Toluene: Understanding the Influence of the Main Reaction Parameters over a Reference Catalyst
Abstract
1. Introduction
2. Experimental
2.1. Catalyst Preparation
2.2. Characterisation
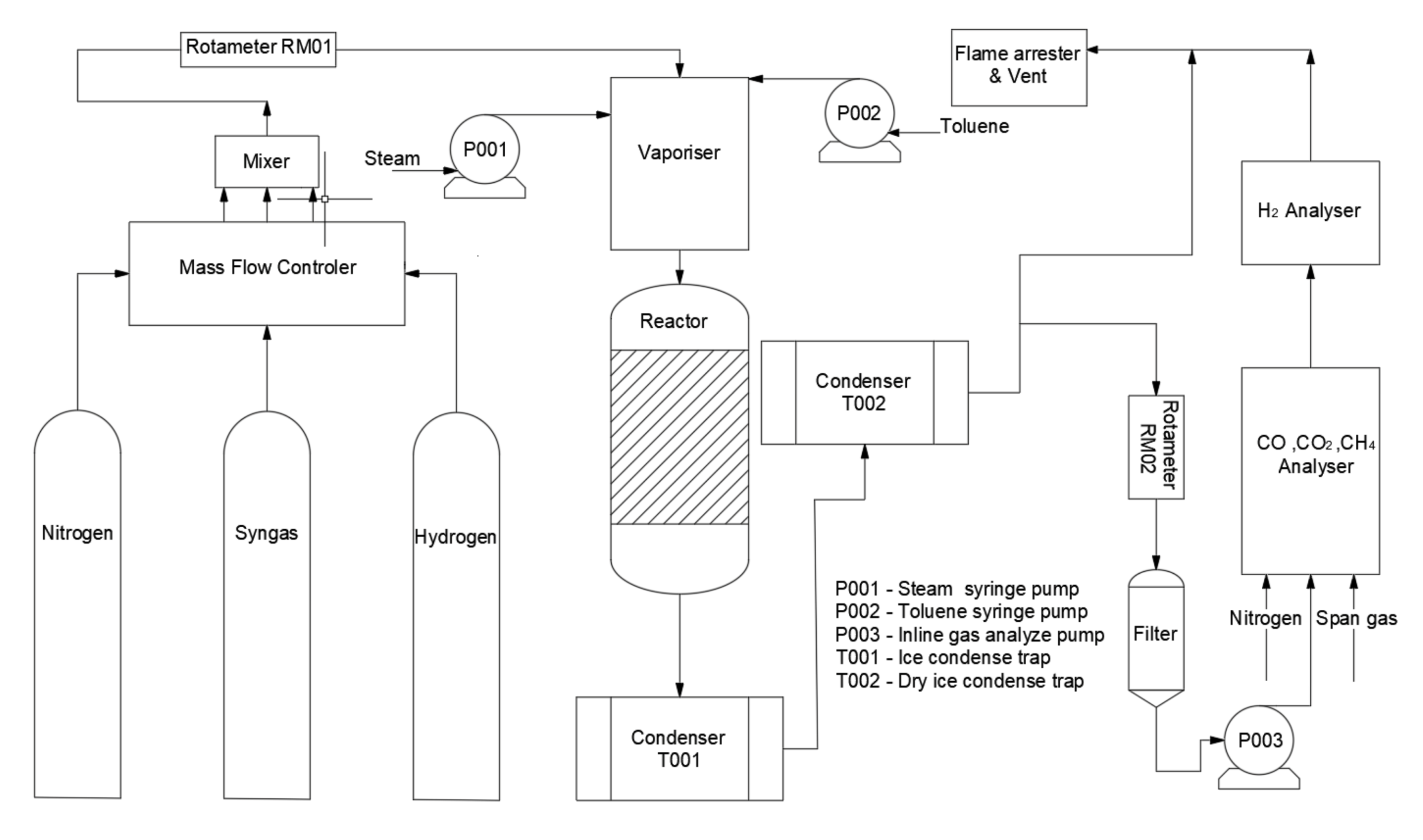
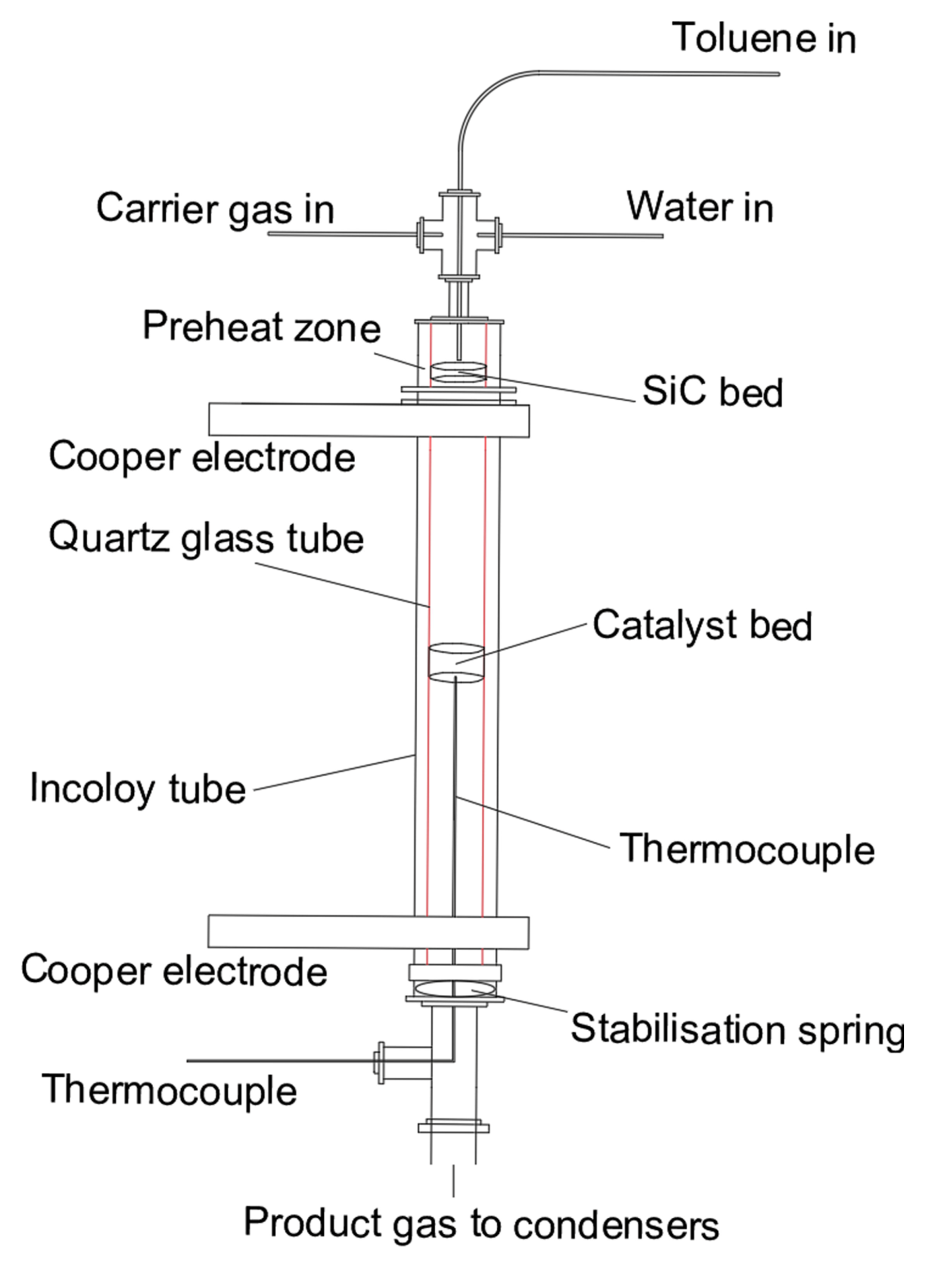
2.3. Catalytic Toluene Steam Reforming Tests
2.4. Thermodynamic Simulation
3. Results and Discussion
3.1. Textural Properties of the Synthetised Catalyst
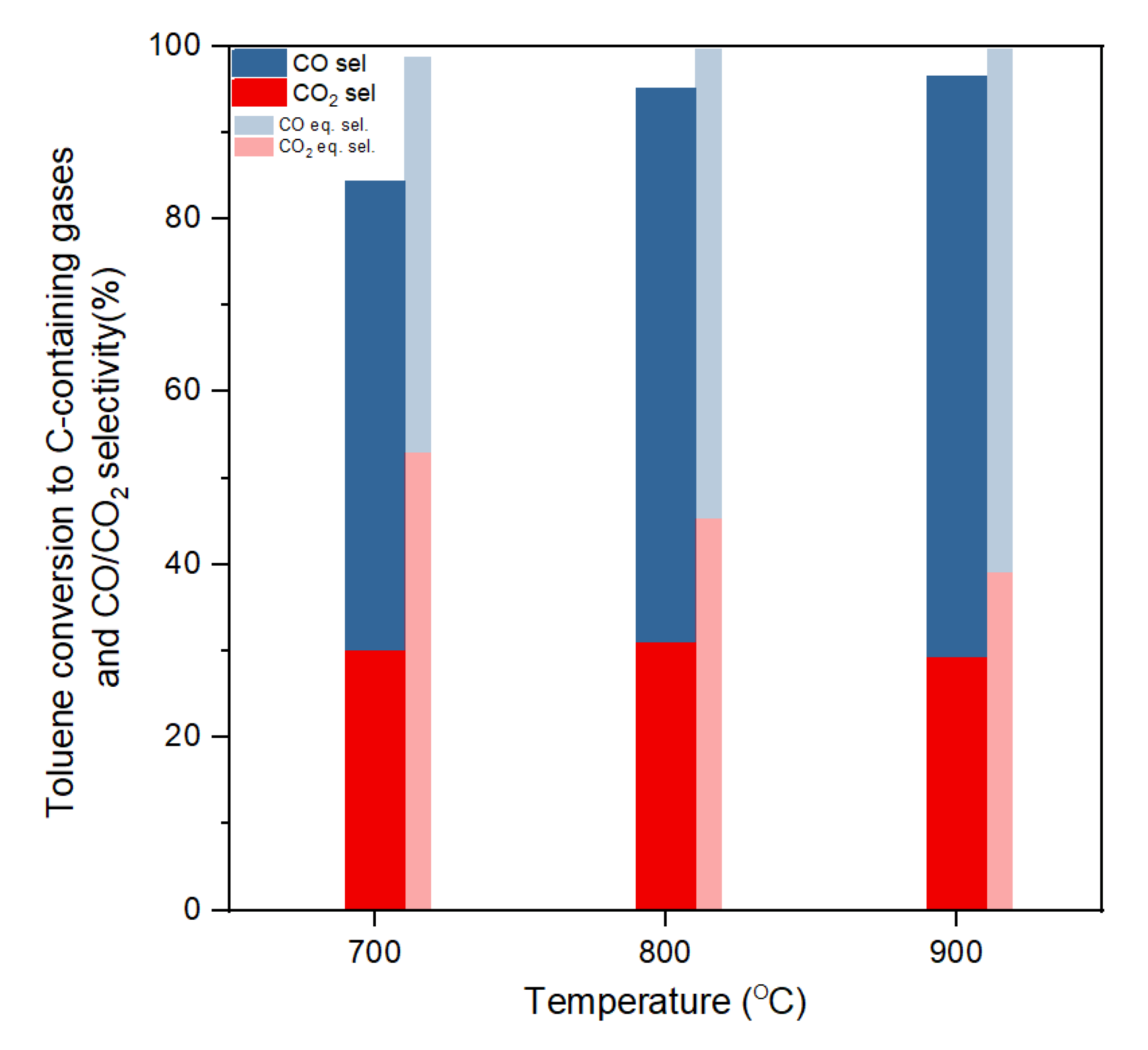
3.2. Influence of Temperature on Toluene Steam Reforming
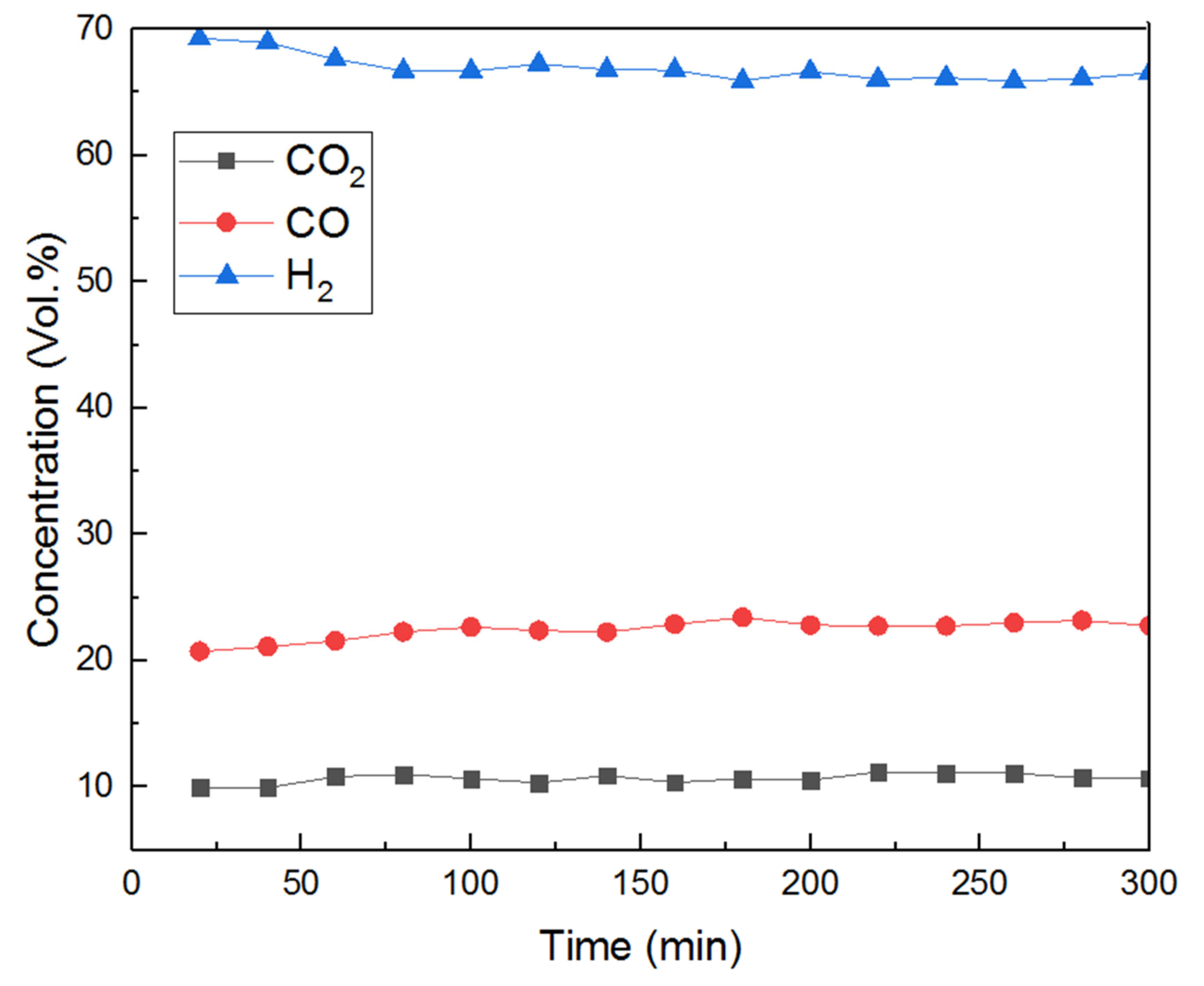
3.3. Influence of GHSV in Toluene Steam Reforming
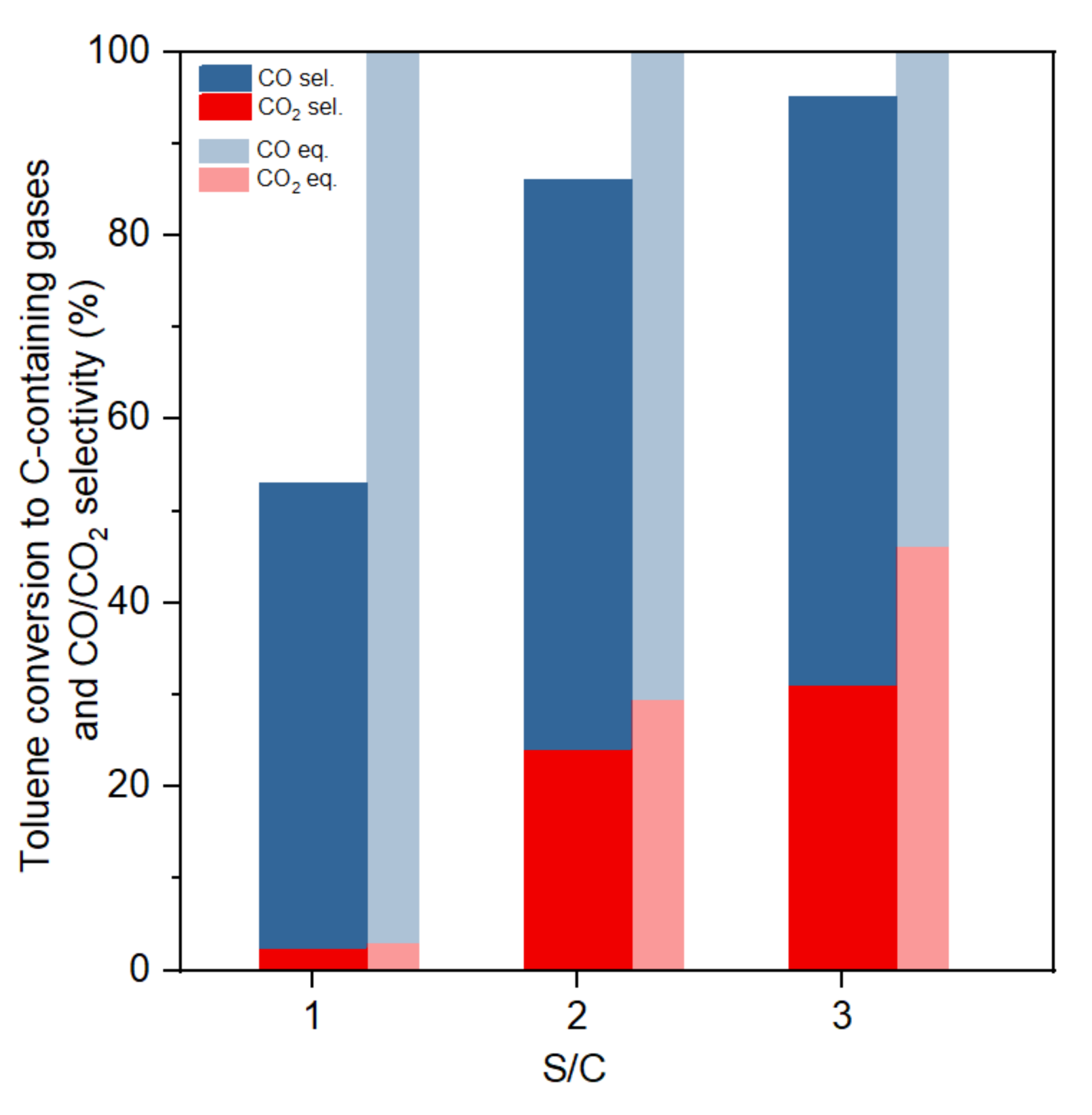
3.4. Influence of S/C Ratio in Toluene Steam Reforming
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Farzad, S.; Mandegari, M.A.; Görgens, J.F. A critical review on biomass gasification, co-gasification, and their environmental assessments. Biofuel Res. J. 2016, 3, 483–495. [Google Scholar] [CrossRef]
- Göransson, K.; Söderlind, U.; He, J.; Zhang, W. Review of syngas production via biomass DFBGs. Renew. Sustain. Energy Rev. 2011, 15, 482–492. [Google Scholar] [CrossRef]
- International Energy Agency. International Energy Outlook 2017; International Energy Agency: Paris, France, 2017. [Google Scholar]
- Balat, H.; Kırtay, E. Hydrogen from biomass–present scenario and future prospects. Int. J. Hydrogen Energy 2010, 35, 7416–7426. [Google Scholar] [CrossRef]
- Świerczyński, D.; Libs, S.; Courson, C.; Kiennemann, A. Steam reforming of tar from a biomass gasification process over Ni/olivine catalyst using toluene as a model compound. Appl. Catal. B-Environ. 2007, 74, 211–222. [Google Scholar] [CrossRef]
- Claude, V.; Mahy, J.G.; Douven, S.; Pirard, S.L.; Courson, C.; Lambert, S.D. Ni-and Fe-doped γ-Al2O3 or olivine as primary catalyst for toluene reforming. Mater. Today Chem. 2019, 14, 100197. [Google Scholar] [CrossRef]
- Devi, L.; Ptasinski, K.J.; Janssen, F.J. A review of the primary measures for tar elimination in biomass gasification processes. Biomass Bioenergy 2003, 24, 125–140. [Google Scholar] [CrossRef]
- Anis, S.; Zainal, Z.A. Tar reduction in biomass producer gas via mechanical, catalytic and thermal methods: A review. Renew. Sustain. Energy Rev. 2011, 15, 2355–2377. [Google Scholar] [CrossRef]
- Noichi, H.; Uddin, A.; Sasaoka, E. Steam reforming of naphthalene as model biomass tar over iron–aluminum and iron–zirconium oxide catalyst catalysts. Fuel Process. Technol. 2010, 91, 1609–1616. [Google Scholar] [CrossRef]
- Guan, G.; Kaewpanha, M.; Hao, X.; Abudula, A. Catalytic steam reforming of biomass tar: Prospects and challenges. Renew. Sustain. Energy Rev. 2016, 58, 450–461. [Google Scholar] [CrossRef]
- Zhao, B.; Zhang, X.; Chen, L.; Qu, R.; Meng, G.; Yi, X.; Sun, L. Steam reforming of toluene as model compound of biomass pyrolysis tar for hydrogen. Biomass Bioenergy 2010, 34, 140–144. [Google Scholar] [CrossRef]
- Virginie, M.; Courson, C.; Kiennemann, A. Toluene steam reforming as tar model molecule produced during biomass gasification with an iron/olivine catalyst. Comptes Rendus Chimie 2010, 13, 1319–1325. [Google Scholar] [CrossRef]
- Park, H.J.; Park, S.H.; Sohn, J.M.; Park, J.; Jeon, J.K.; Kim, S.S.; Park, Y.K. Steam reforming of biomass gasification tar using benzene as a model compound over various Ni supported metal oxide catalysts. Bioresour. Technol. 2010, 101, S101–S103. [Google Scholar] [CrossRef] [PubMed]
- Lorente, E.; Millan, M.; Brandon, N.P. Use of gasification syngas in SOFC: Impact of real tar on anode materials. Int. J. Hydrogen Energy 2012, 37, 7271–7278. [Google Scholar] [CrossRef]
- Lorente, E.; Berrueco, C.; Millan, M.; Brandon, N.P. Effect of tar fractions from coal gasification on nickel-yttria stabilized zirconia and nickel-gadolinium doped ceria solid oxide fuel cell anode materials. J. Power Sources 2013, 242, 824–831. [Google Scholar] [CrossRef]
- Coll, R.; Salvado, J.; Farriol, X.; Montane, D. Steam reforming model compounds of biomass gasification tars: Conversion at different operating conditions and tendency towards coke formation. Fuel Process. Technol. 2001, 74, 19–31. [Google Scholar] [CrossRef]
- Matsumura, Y.; Nakamori, T. Steam reforming of methane over nickel catalysts at low reaction temperature. Appl. Catal. A-Gen. 2004, 258, 107–114. [Google Scholar] [CrossRef]
- Lertwittayanon, K.; Youravong, W.; Lau, W.J. Enhanced catalytic performance of Ni/α-Al2O3 catalyst modified with CaZrO3 nanoparticles in steam-methane reforming. Int. J. Hydrogen Energy 2017, 42, 28254–28265. [Google Scholar] [CrossRef]
- Ahmed, T.; Xiu, S.; Wang, L.; Shahbazi, A. Investigation of Ni/Fe/Mg zeolite-supported catalysts in steam reforming of tar using simulated-toluene as model compound. Fuel 2018, 211, 566–571. [Google Scholar] [CrossRef]
- Mermelstein, J.; Millan, M.; Brandon, N.P. The impact of carbon formation on Ni–YSZ anodes from biomass gasification model tars operating in dry conditions. Chem. Eng. Sci. 2009, 64, 492–500. [Google Scholar] [CrossRef]
- Xu, H.; Liu, Y.; Sun, G.; Kang, S.; Wang, Y.; Zheng, Z.; Li, X. Synthesis of graphitic mesoporous carbon supported Ce-doped nickel catalyst for steam reforming of toluene. Mater. Lett. 2019, 244, 123–125. [Google Scholar] [CrossRef]
- Li, Z.; Li, M.; Ashok, J.; Kawi, S. NiCo@ NiCo phyllosilicate@ CeO2 hollow core shell catalysts for steam reforming of toluene as biomass tar model compound. Energy Convers. Manag. 2019, 180, 822–830. [Google Scholar] [CrossRef]
- Ashok, J.; Dewangan, N.; Das, S.; Hongmanorom, P.; Wai, M.H.; Tomishige, K.; Kawi, S. Recent progress in the development of catalysts for steam reforming of biomass tar model reaction. Fuel Process. Technol. 2020, 199, 106252. [Google Scholar] [CrossRef]
- Bizkarra, K.; Bermudez, J.M.; Arcelus-Arrillaga, P.; Barrio, V.L.; Cambra, J.F.; Millan, M. Nickel based monometallic and bimetallic catalysts for synthetic and real bio-oil steam reforming. Int. J. Hydrogen Energy 2018, 43, 11706–11718. [Google Scholar] [CrossRef]
- Meng, J.; Zhao, Z.; Wang, X.; Wu, X.; Zheng, A.; Huang, Z.; Li, H. Effects of catalyst preparation parameters and reaction operating conditions on the activity and stability of thermally fused Fe-olivine catalyst in the steam reforming of toluene. Int. J. Hydrogen Energy 2018, 43, 127–138. [Google Scholar] [CrossRef]
- Oh, G.; Park, S.Y.; Seo, M.W.; Kim, Y.K.; Ra, H.W.; Lee, J.G.; Yoon, S.J. Ni/Ru–Mn/Al2O3 catalysts for steam reforming of toluene as model biomass tar. Renew. Energy 2016, 86, 841–847. [Google Scholar] [CrossRef]
- Meidanshahi, V.; Bahmanpour, A.M.; Iranshahi, D.; Rahimpour, M.R. Theoretical investigation of aromatics production enhancement in thermal coupling of naphtha reforming and hydrodealkylation of toluene. Chem. Eng. Process 2011, 50, 893–903. [Google Scholar] [CrossRef]
- Tang, Y.; Liu, J. Effect of anode and Boudouard reaction catalysts on the performance of direct carbon solid oxide fuel cells. Int. J. Hydrogen Energy 2010, 35, 11188–11193. [Google Scholar] [CrossRef]
- Zhang, Y.; Williams, P.T. Carbon nanotubes and hydrogen production from the pyrolysis catalysis or catalytic-steam reforming of waste tyres. J. Anal. Appl. Pyrol. 2016, 122, 490–501. [Google Scholar] [CrossRef]
- Yue, B.; Wang, X.; Ai, X.; Yang, J.; Li, L.; Lu, X.; Ding, W. Catalytic reforming of model tar compounds from hot coke oven gas with low steam/carbon ratio over Ni/MgO–Al2O3 catalysts. Fuel Process. Technol. 2010, 91, 1098–1104. [Google Scholar] [CrossRef]
- Dieuzeide, M.L.; Iannibelli, V.; Jobbagy, M.; Amadeo, N. Steam reforming of glycerol over Ni/Mg/γ-Al2O3 catalysts. Effect of calcination temperatures. Int. J. Hydrogen Energy 2012, 37, 14926–14930. [Google Scholar] [CrossRef]
- Bona, S.; Guillén, P.; Alcalde, J.G.; García, L.; Bilbao, R. Toluene steam reforming using coprecipitated Ni/Al catalysts modified with lanthanum or cobalt. Chem. Eng. J. 2008, 137, 587–597. [Google Scholar] [CrossRef]
- Quitete, C.P.; Bittencourt, R.C.P.; Souza, M.M. Steam reforming of tar using toluene as a model compound with nickel catalysts supported on hexaaluminates. Appl. Catal. A-Gen. 2014, 478, 234–240. [Google Scholar] [CrossRef]
- Yang, L.; Pastor-Pérez, L.; Gu, S.; Sepúlveda-Escribano, A.; Reina, T.R. Highly efficient Ni/CeO2-Al2O3 catalysts for CO2 upgrading via reverse water-gas shift: Effect of selected transition metal promoters. Appl. Catal. B-Environ. 2018, 232, 464–471. [Google Scholar] [CrossRef]
- Zhu, H.L.; Zhang, Y.S.; Materazzi, M.; Aranda, G.; Brett, D.J.; Shearing, P.R.; Manos, G. Co-gasification of beech-wood and polyethylene in a fluidized-bed reactor. Fuel Process. Technol. 2019, 190, 29–37. [Google Scholar] [CrossRef]
- Stroud, T.; Smith, T.J.; Le Saché, E.; Santos, J.L.; Centeno, M.A.; Arellano-Garcia, H.; Odriozola, J.A.; Reina, T.R. Chemical CO2 recycling via dry and bi reforming of methane using Ni-Sn/Al2O3 and Ni-Sn/CeO2-Al2O3 catalysts. Appl. Catal. B-Environ. 2018, 224, 125–135. [Google Scholar] [CrossRef]
- Mukai, D.; Tochiya, S.; Murai, Y.; Imori, M.; Hashimoto, T.; Sugiura, Y.; Sekine, Y. Role of support lattice oxygen on steam reforming of toluene for hydrogen production over Ni/La0.7Sr0.3AlO3− δ catalyst. Appl. Catal. A-Gen. 2013, 453, 60–70. [Google Scholar] [CrossRef]
- Frusteri, F.; Freni, S.; Spadaro, L.; Chiodo, V.; Bonura, G.; Donato, S.; Cavallaro, S. H2 production for MC fuel cell by steam reforming of ethanol over MgO supported Pd, Rh, Ni and Co catalysts. Catal. Commun. 2004, 5, 611–615. [Google Scholar] [CrossRef]
- He, L.; Hu, S.; Jiang, L.; Liao, G.; Chen, X.; Han, H.; Xiao, L.; Ren, Q.; Wang, Y.; Su, S.; et al. Carbon nanotubes formation and its influence on steam reforming of toluene over Ni/Al2O3 catalysts: Roles of catalyst supports. Fuel Process. Technol. 2018, 176, 7–14. [Google Scholar] [CrossRef]
- Li, Q.; Wang, Q.; Kayamori, A.; Zhang, J. Experimental study and modeling of heavy tar steam reforming. Fuel Process. Technol. 2018, 178, 180–188. [Google Scholar] [CrossRef]
- Li, C.; Hirabayashi, D.; Suzuki, K. Development of new nickel based catalyst for biomass tar steam reforming producing H2-rich syngas. Fuel Process. Technol. 2009, 90, 790–796. [Google Scholar] [CrossRef]
- Le Saché, E.; Pastor-Pérez, L.; Garcilaso, V.; Watson, D.; Centeno, M.A.; Odriozola, J.A.; Reina, T.R. Flexible syngas production using a La2Zr2-xNixO7-δ pyrochlore-double perovskite catalyst: Towards a direct route for gas phase CO2 recycling. Catal. Today 2019. [Google Scholar] [CrossRef]

| Temperature (°C) | CO2 (mol/mol Toluene) | CO (mol/mol Toluene) | H2 (mol/mol Toluene) | CH4 (mol/mol Toluene) |
|---|---|---|---|---|
| 700 | 2.1 | 3.8 | 10.3 | 0 |
| (equilibrium) | (3.7) | (3.2) | (14.5) | (0.1) |
| 800 | 2.2 | 4.5 | 13.0 | 0 |
| (equilibrium) | (3.2) | (3.8) | (14.3) | (0) |
| 900 | 2.1 | 4.7 | 11.8 | 0 |
| (equilibrium) | (2.8) | (4.2) | (13.8) | (0) |
| Reforming Temperature | 700 °C | 800 °C | 900 °C |
|---|---|---|---|
| Coke/C in toluene | 0.43% | 0.61% | 0.77% |
| Coke/Catalyst (gC/gcat) | 0.118 | 0.167 | 0.211 |
| GHSV (h−1) | CO2 (mol/mol Toluene) | CO (mol/mol Toluene) | H2 (mol/mol Toluene) |
|---|---|---|---|
| 30,600 | 2.4 | 4.3 | 13.2 |
| 61,200 | 2.2 | 4.5 | 13.0 |
| 91,800 | 2.1 | 4.2 | 12.8 |
| 122,400 | 2.1 | 3.9 | 10.8 |
| (Equilibrium) | (3.2) | (3.8) | (14.3) |
| GHSV(h−1) | 30,600 | 61,200 | 91,800 | 122,400 |
|---|---|---|---|---|
| Coke/C in toluene | 0.38% | 0.31% | 0.23% | 0.22% |
| Coke/Catalyst (gC/gcat) | 0.105 | 0.167 | 0.188 | 0.242 |
| S/C ratio | CO2 (mol/mol Toluene) | CO (mol/mol Toluene) | H2 (mol/mol Toluene) |
|---|---|---|---|
| 1 | 0.2 | 3.5 | 5.1 |
| (Equilibrium) | (0.2) | (6.8) | (9.8) |
| 2 | 1.7 | 4.3 | 10.5 |
| (Equilibrium) | (2.1) | (4.9) | (13.0) |
| 3 | 2.2 | 4.5 | 13.0 |
| (Equilibrium) | (3.2) | (3.8) | (14.3) |
| S/C ratio | 1 | 2 | 3 |
|---|---|---|---|
| Coke/C in toluene | 1.07% | 0.82% | 0.61% |
| Coke/Catalyst (gC/gcat) | 0.289 | 0.222 | 0.167 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhu, H.L.; Pastor-Pérez, L.; Millan, M. Catalytic Steam Reforming of Toluene: Understanding the Influence of the Main Reaction Parameters over a Reference Catalyst. Energies 2020, 13, 813. https://doi.org/10.3390/en13040813
Zhu HL, Pastor-Pérez L, Millan M. Catalytic Steam Reforming of Toluene: Understanding the Influence of the Main Reaction Parameters over a Reference Catalyst. Energies. 2020; 13(4):813. https://doi.org/10.3390/en13040813
Chicago/Turabian StyleZhu, Hua Lun, Laura Pastor-Pérez, and Marcos Millan. 2020. "Catalytic Steam Reforming of Toluene: Understanding the Influence of the Main Reaction Parameters over a Reference Catalyst" Energies 13, no. 4: 813. https://doi.org/10.3390/en13040813
APA StyleZhu, H. L., Pastor-Pérez, L., & Millan, M. (2020). Catalytic Steam Reforming of Toluene: Understanding the Influence of the Main Reaction Parameters over a Reference Catalyst. Energies, 13(4), 813. https://doi.org/10.3390/en13040813





