Quantification of Minor Chemical Elements in Particulate Matter Collected from In-Use Diesel Engine Passenger Vehicles by Laser-Induced Breakdown Spectroscopy
Abstract
1. Introduction
2. Methodology
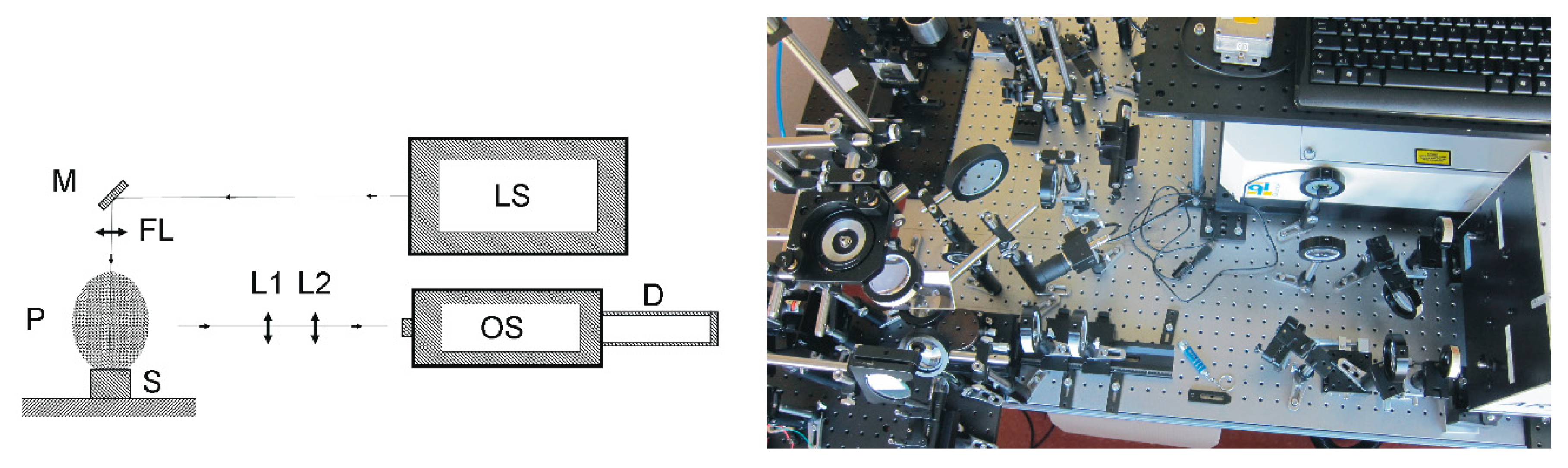
2.1. Experimental LIBS Setup
2.2. Particulate Matter Collection and Sample Preparation
3. Results and Discussion
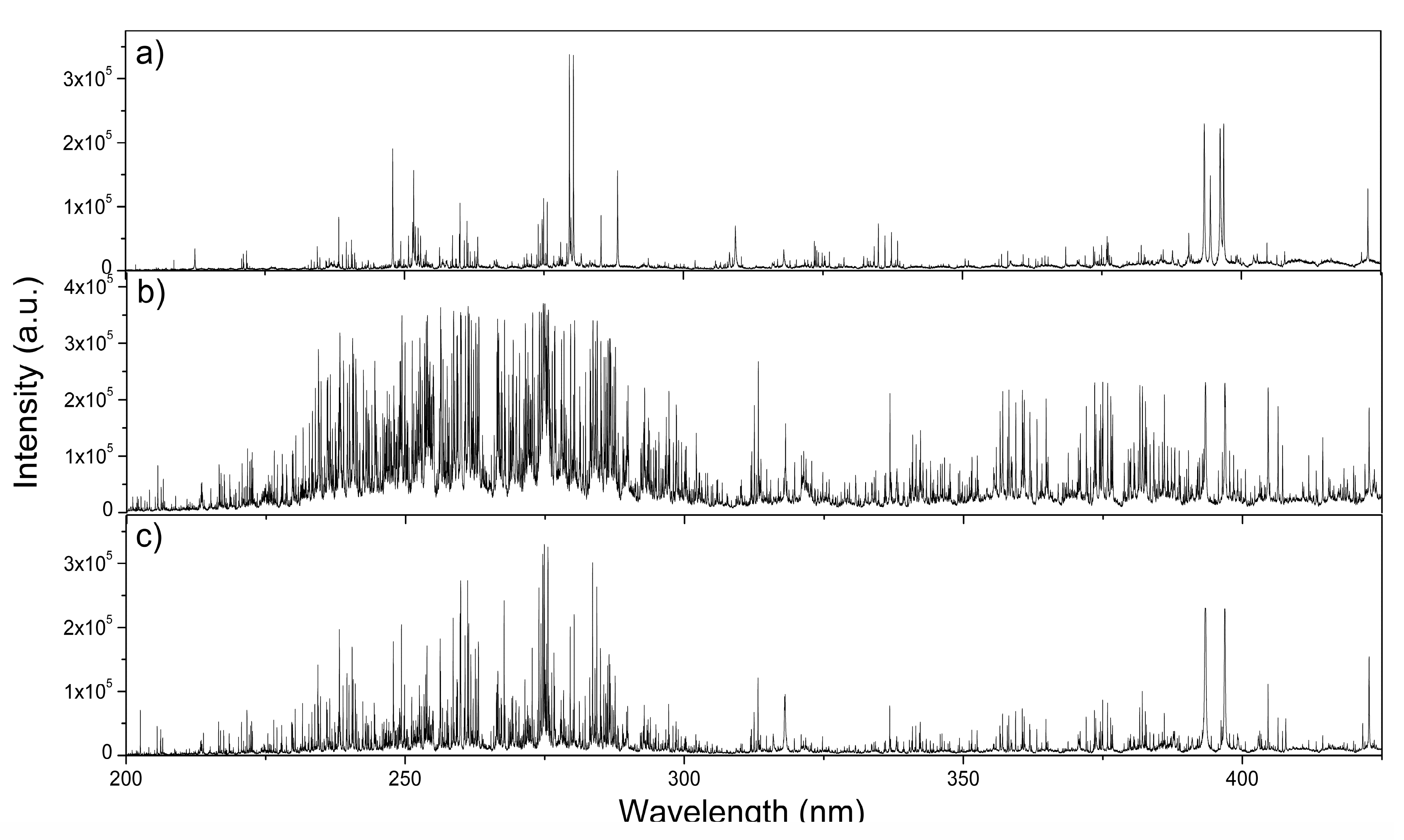
3.1. Diesel Particulate Matter Measured by LIBS
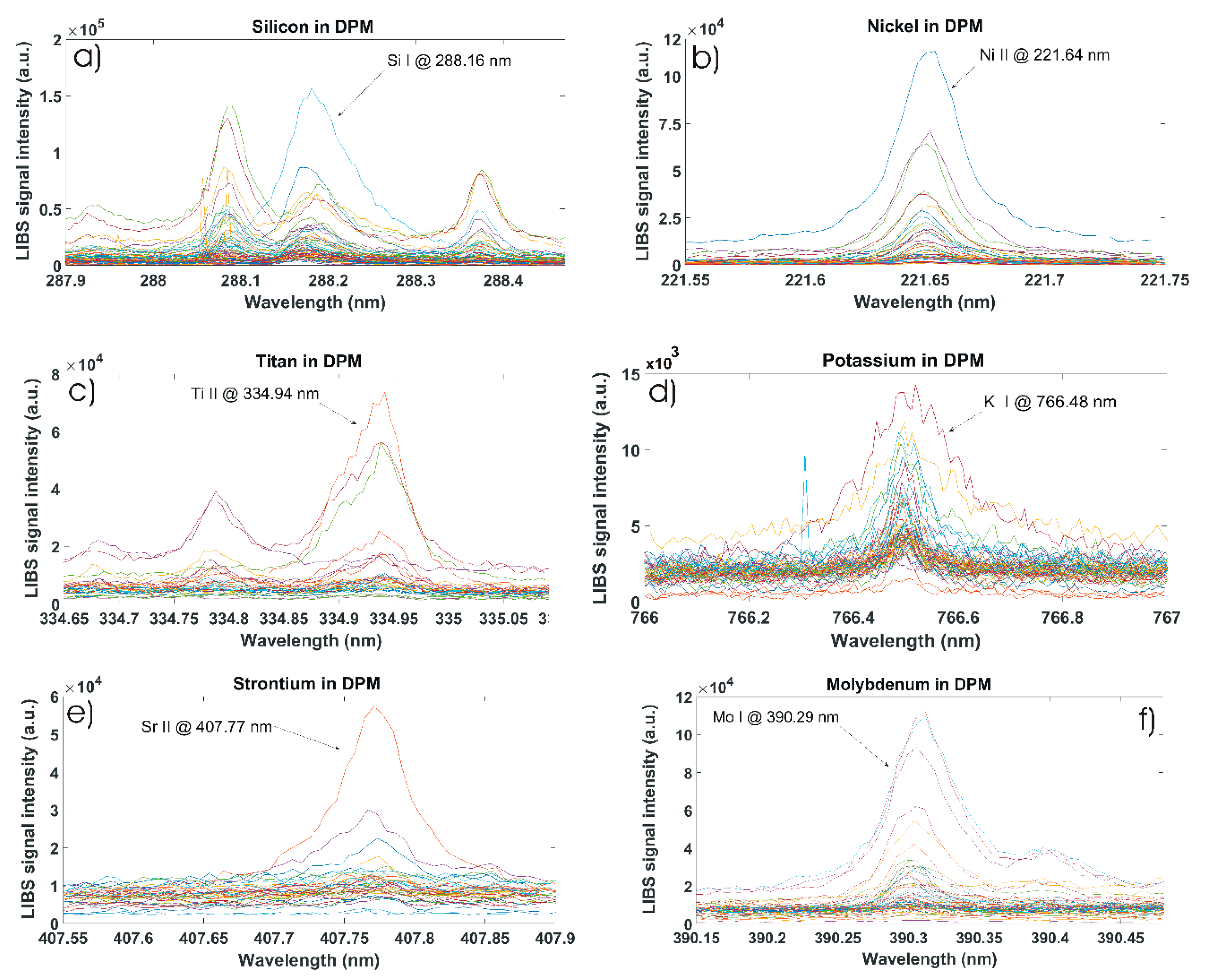
3.2. Qualitative Measurements of Minor Chemical Elements in Diesel Particulate Matter
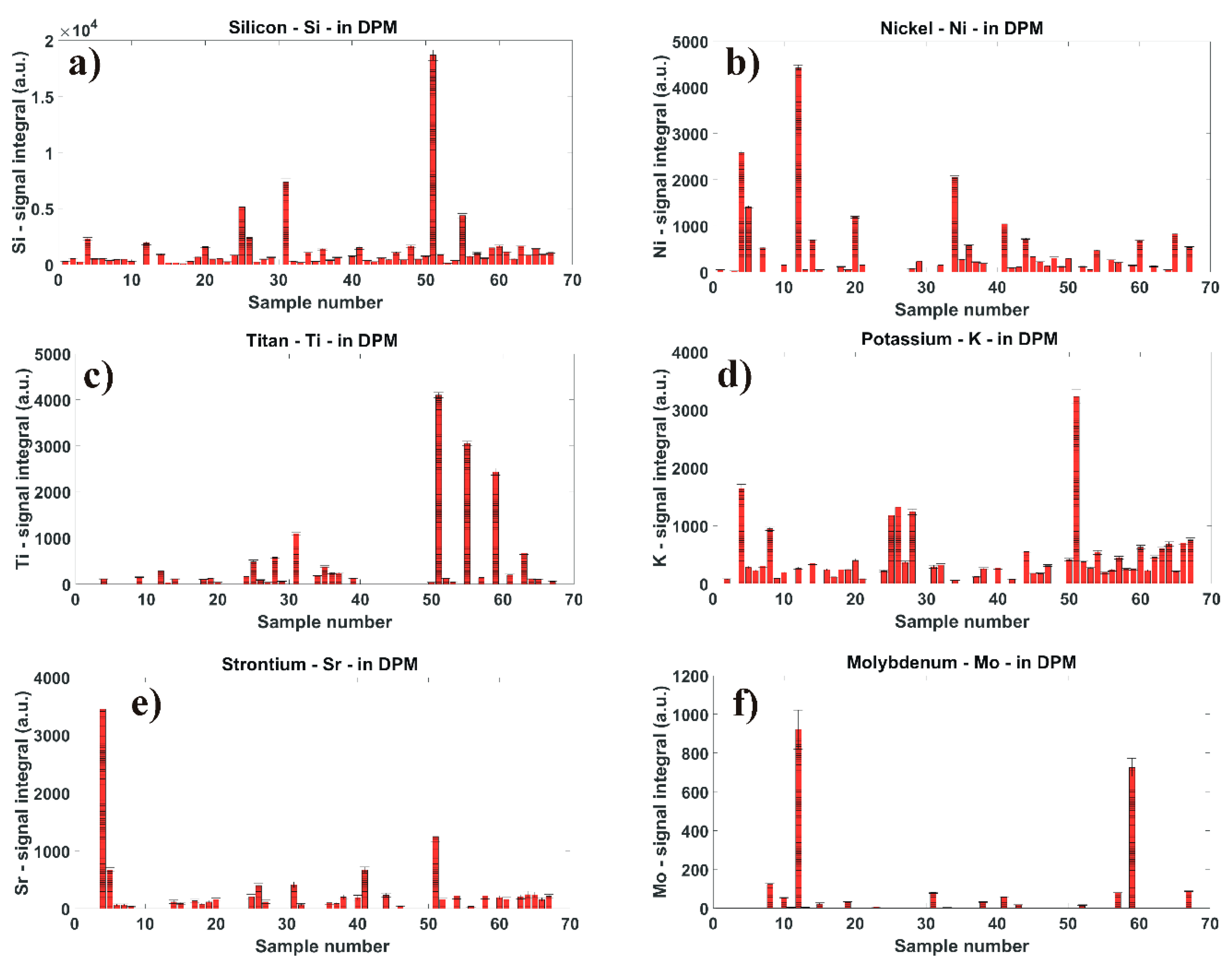
3.3. Quantitative Analyses of LIBS Signal
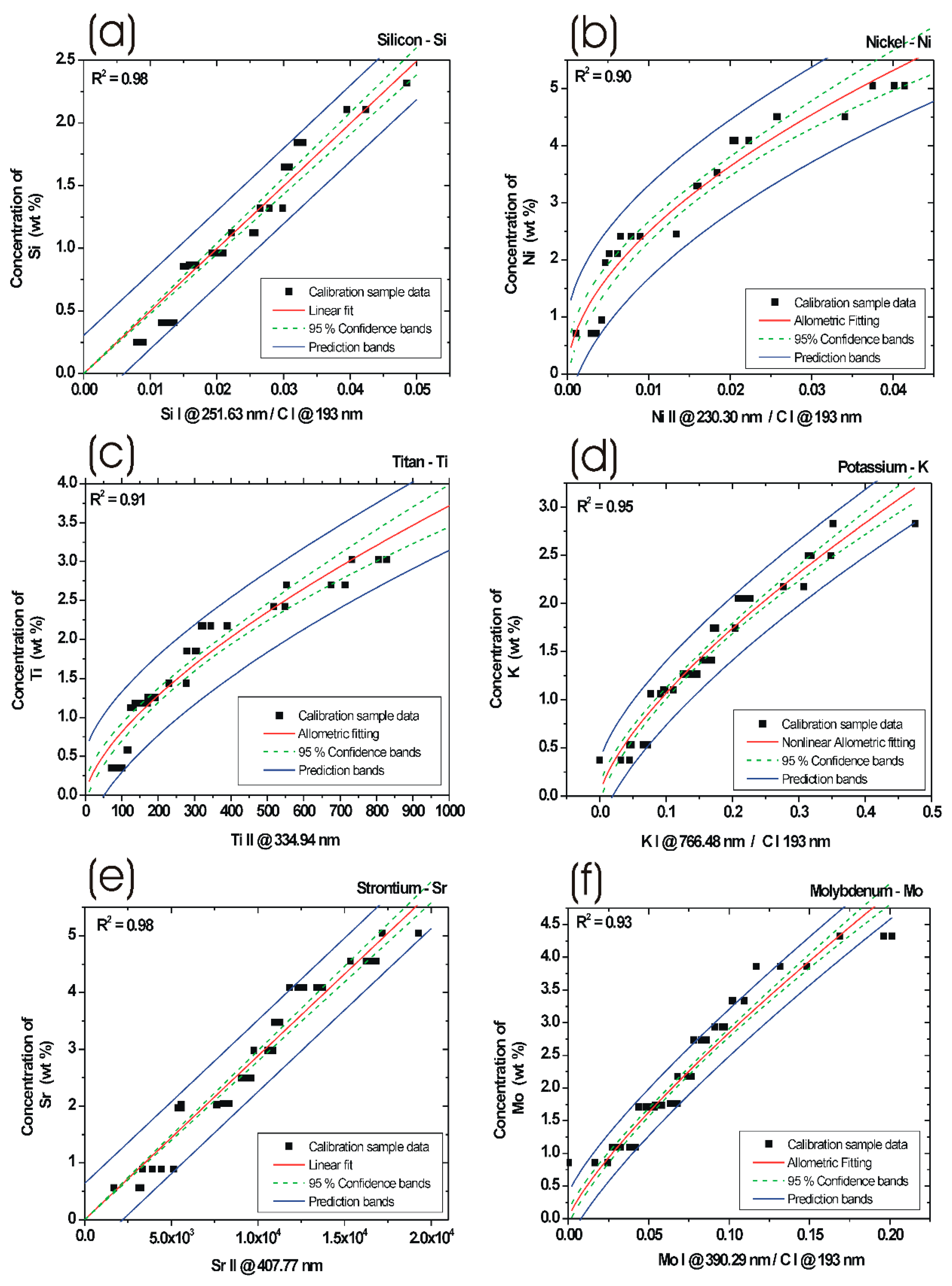
3.4. LIBS Measurements of Calibration Samples
3.4.1. Silicon Calibration Curve
3.4.2. Nickel Calibration Curve
3.4.3. Titan Calibration Curve
3.4.4. Potassium Calibration Curve
3.4.5. Strontium Calibration Curve
3.4.6. Molybdenum Calibration Curve
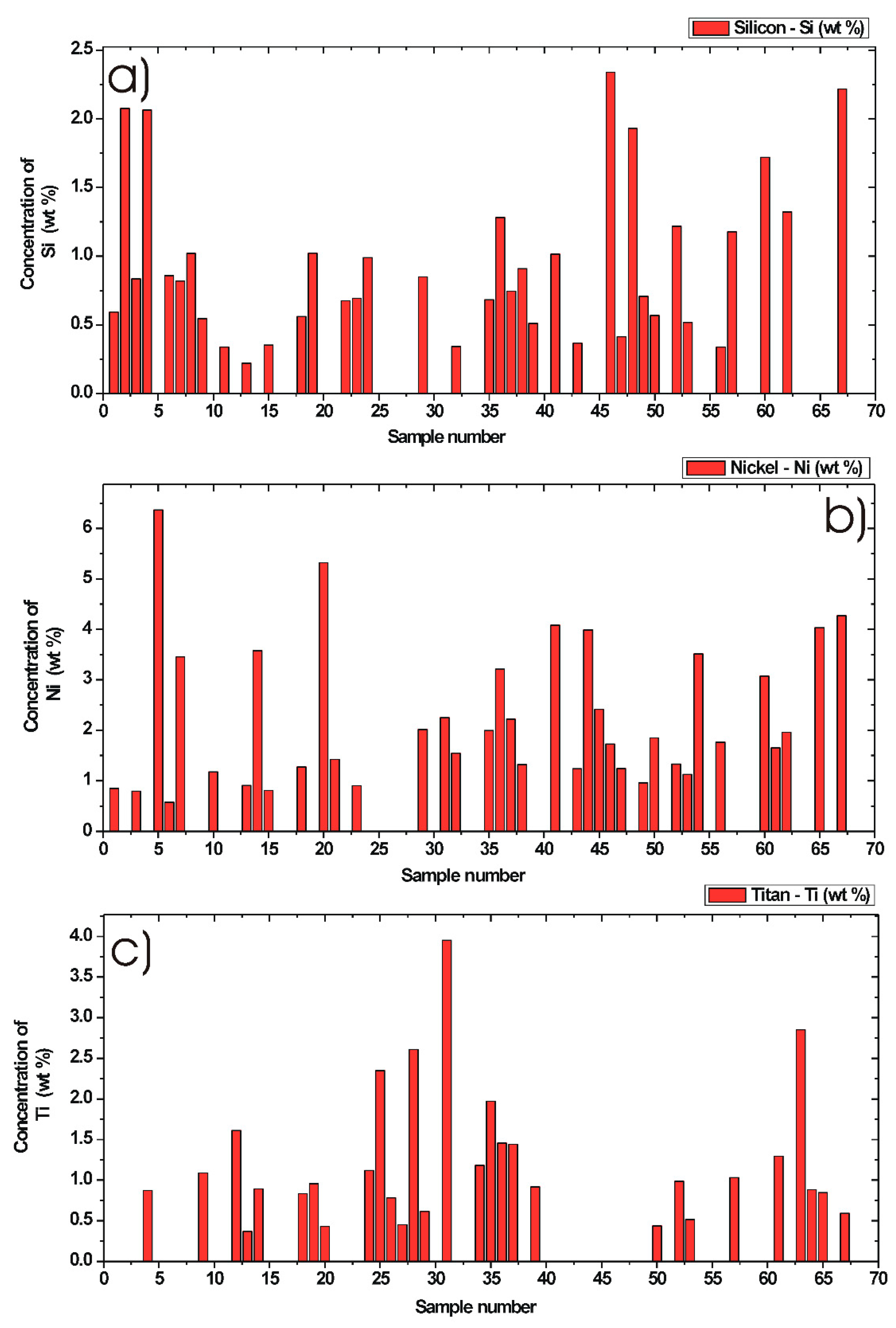
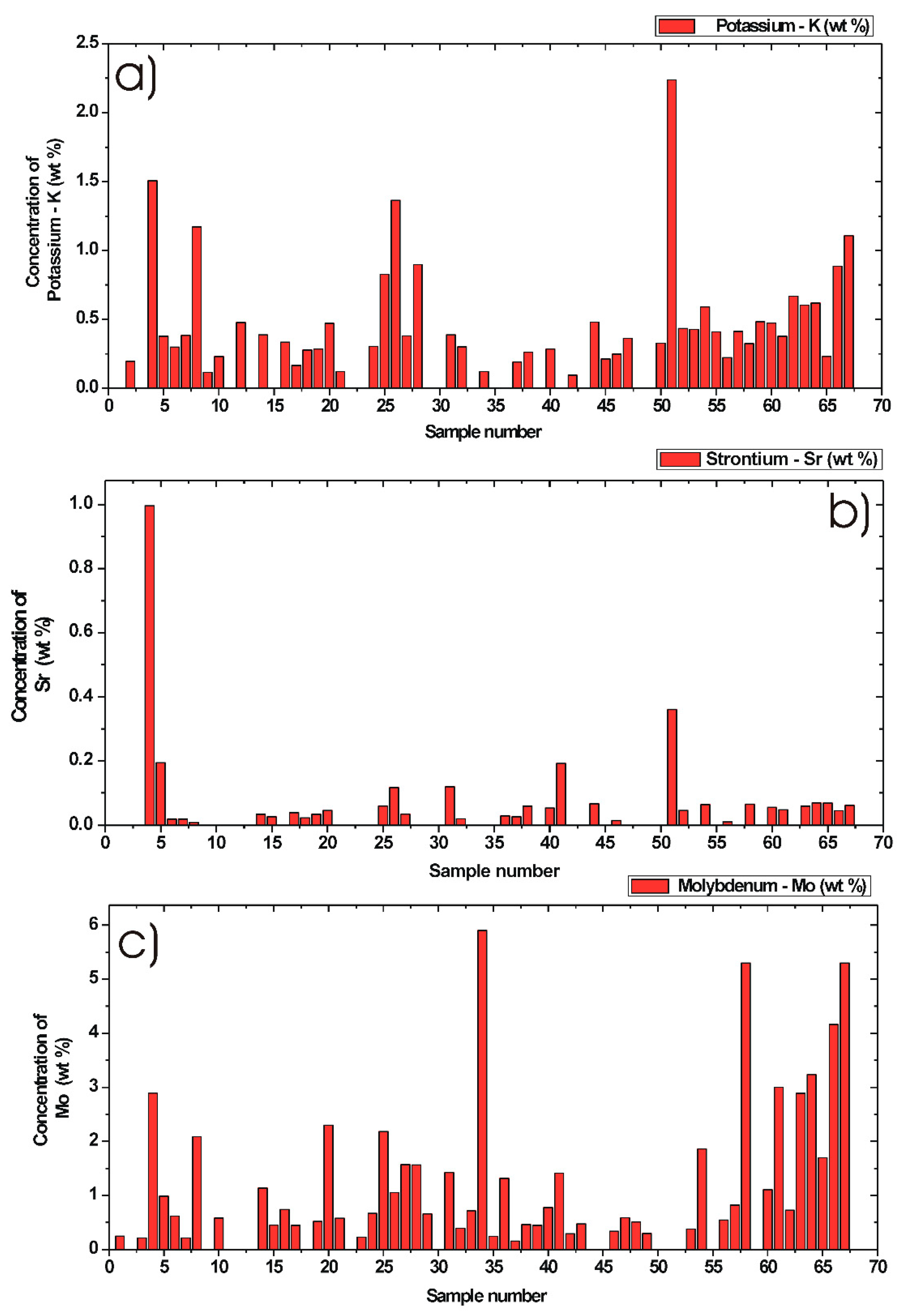
3.5. Quantification of Minor Chemical Elements in Diesel Particulate Matter
3.5.1. Silicon Concentration in DPM
3.5.2. Nickel Concentration in DPM
3.5.3. Titan Concentration in DPM
3.5.4. Potassium Concentration in DPM
3.5.5. Strontium Concentration in DPM
3.5.6. Molybdenum Concentration in DPM
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ntziachristos, L.; Papadimitriou, G.; Ligterink, N.; Hausberger, S. Implications of Diesel emissions control failures to emission factors and road transport NOx evolution. Atmos. Environ. 2016, 141, 542–551. [Google Scholar] [CrossRef]
- Zacharof, N.; Tietge, U.; Franco, V.; Mock, P. Type approval and real-world CO2 and NOx emissions from EU light commercial vehicles. Energy Policy 2016, 97, 540–548. [Google Scholar] [CrossRef]
- Commission Regulation (EU) 2016/646. Commission Regulation (EU) 2016/646 of 20 April 2016 Amending Regulation (EC) (No 692/2008) as Regards Emissions from Light Passenger and Commercial Vehicles (Euro 6). Available online: http://eur-lex.europa.eu/eli/reg/2016/646/oj (accessed on 9 January 2020).
- Calderón-Garcidueñas, L.; Mora-Tiscareño, A.; Ontiveros, E.; Gómez-Garza, G.; Barragán-Mejía, G.; Broadway, J.; Chapman, S.; Valencia-Salazar, G.; Jewells, V.; Maronpot, R.R.; et al. Air pollution, cognitive deficits and brain abnormalities: A pilot study with children and dogs. Brain Cogn. 2008, 68, 117–127. [Google Scholar] [CrossRef] [PubMed]
- Block, M.L.; Calderon-Garciduenas, L. Air pollution: Mechanisms of neuroinflammation and CNS disease. Trends Neurosci. 2009, 32, 506–516. [Google Scholar] [CrossRef] [PubMed]
- Noll, R. Laser-Induced Breakdown Spectroscopy, Fundamentals and Applications; Springer: Berlin, Germany, 2012. [Google Scholar]
- Viskup, R.; Wolf, C.; Baumgartner, W. Qualitative and quantitative characterisation of major elements in particulate matter from in-use diesel engine passenger vehicles by LIBS. Energies 2020, 13, 368. [Google Scholar] [CrossRef]
- Viskup, R.; Wolf, C.; Baumgartner, W. Major Chemical Elements in Soot and Particulate Matter Exhaust Emissions Generated from In-Use Diesel Engine Passenger Vehicles, Introduction to Diesel Emissions, Richard Viskup. In Introduction to Diesel Emissions; IntechOpen: London, UK, 2020. [Google Scholar] [CrossRef]
- Miziolek, A.W.; Palleschi, V.; Schechter, I. Laser-Induced Breakdown Spectroscopy (LIBS): Fundamentals and Application; Cambridge University Press: New York, NY, USA, 2006. [Google Scholar]
- Cremers, D.A.; Radziemski, L.J. Handbook of Laser-Induced Breakdown Spectroscopy; John Wiley & Sons Inc.: New Delhi, India, 2013. [Google Scholar]
- Viskup, R. Single and Double Laser Pulse Interaction with Solid State–Application to Plasma Spectroscopy. In Nd YAG Laser; Dumitras, D.C., Ed.; InTech: Rijeka, Croatia, 2012; ISBN 978-953-51-0105-5. [Google Scholar] [CrossRef]
- Kramida, A.; Ralchenko, Y.; Reader, J.; NIST ASD Team National Institute of Standards and Technology NIST. Atomic Spectra Database. Available online: http://physics.nist.gov/asd (accessed on 9 January 2020).
| Type of Equipment | Model | Company | Specifications |
|---|---|---|---|
| Nd:YAG laser | Yasmin | Quantel, France | τp ≈ 8.5 ns; λ ≈ 1064 nm; Ep ≈ 300 mJ |
| Echelle spectrograph | Aryelle Butterfly | LTB, Berlin, Germany | UV: 190–440 nm, Res: 3–7 pm VIS: 440–800 nm, Res: 4–8 pm |
| ICCD detector | PI-Max 4 | Princeton Instruments, USA | delay time = 1 μs; gate width = 2 μs |
| Analyte | Spectral Line | Wavelength (nm) | Detected in/Total Number of Samples |
|---|---|---|---|
| Si | Si I | 288.16 | 63/67 |
| Ni | Ni II | 221.64 | 43/67 |
| Ti | Ti II | 334.94 | 32/67 |
| K | K I | 766.48 | 50/67 |
| Sr | Sr II | 407.77 | 35/67 |
| Mo | Mo I | 390.29 | 55/67 |
| Calibration for Analyte | Regression Parameter R2 | Calibration for Concentrations wt % | Optical Spectral Lines and Integrated Peak Ratio for Calculation |
|---|---|---|---|
| Si | 0.98 | 0–3 | Si I @ 251.63 nm, C I @ 193.02 nm |
| Ni | 0.90 | 0–7 | Ni II @ 230.30 nm, C I @ 193.02 nm |
| Ti | 0.91 | 0–4 | Ti II @ 334.94 nm |
| K | 0.95 | 0–3 | K I @ 766.48nm, C I @ 193.02 nm |
| Sr | 0.98 | 0–5 | Sr II @ 407.77 nm |
| Mo | 0.93 | 0–6 | Mo I @ 390.29 nm, C I @ 193.02 nm |
| Analyte | Max. Concentration (wt %) | Min. Concentration (wt %) | Mean Value (wt %) | Median Value (wt %) | Number of DPM Samples Used for Statistics |
|---|---|---|---|---|---|
| Si | 2.33 | 0.22 | 0.94 | 0.81 | 37 |
| Ni | 6.36 | 0.57 | 2.22 | 1.76 | 37 |
| Ti | 3.95 | 0.36 | 1.21 | 0.95 | 29 |
| K | 2.23 | 0.1 | 0.48 | 0.37 | 50 |
| Sr | 0.99 | 0.01 | 0.09 | 0.04 | 35 |
| Mo | 5.9 | 0.16 | 1.32 | 0.72 | 52 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Viskup, R.; Wolf, C.; Baumgartner, W. Quantification of Minor Chemical Elements in Particulate Matter Collected from In-Use Diesel Engine Passenger Vehicles by Laser-Induced Breakdown Spectroscopy. Energies 2020, 13, 6113. https://doi.org/10.3390/en13226113
Viskup R, Wolf C, Baumgartner W. Quantification of Minor Chemical Elements in Particulate Matter Collected from In-Use Diesel Engine Passenger Vehicles by Laser-Induced Breakdown Spectroscopy. Energies. 2020; 13(22):6113. https://doi.org/10.3390/en13226113
Chicago/Turabian StyleViskup, Richard, Christoph Wolf, and Werner Baumgartner. 2020. "Quantification of Minor Chemical Elements in Particulate Matter Collected from In-Use Diesel Engine Passenger Vehicles by Laser-Induced Breakdown Spectroscopy" Energies 13, no. 22: 6113. https://doi.org/10.3390/en13226113
APA StyleViskup, R., Wolf, C., & Baumgartner, W. (2020). Quantification of Minor Chemical Elements in Particulate Matter Collected from In-Use Diesel Engine Passenger Vehicles by Laser-Induced Breakdown Spectroscopy. Energies, 13(22), 6113. https://doi.org/10.3390/en13226113





