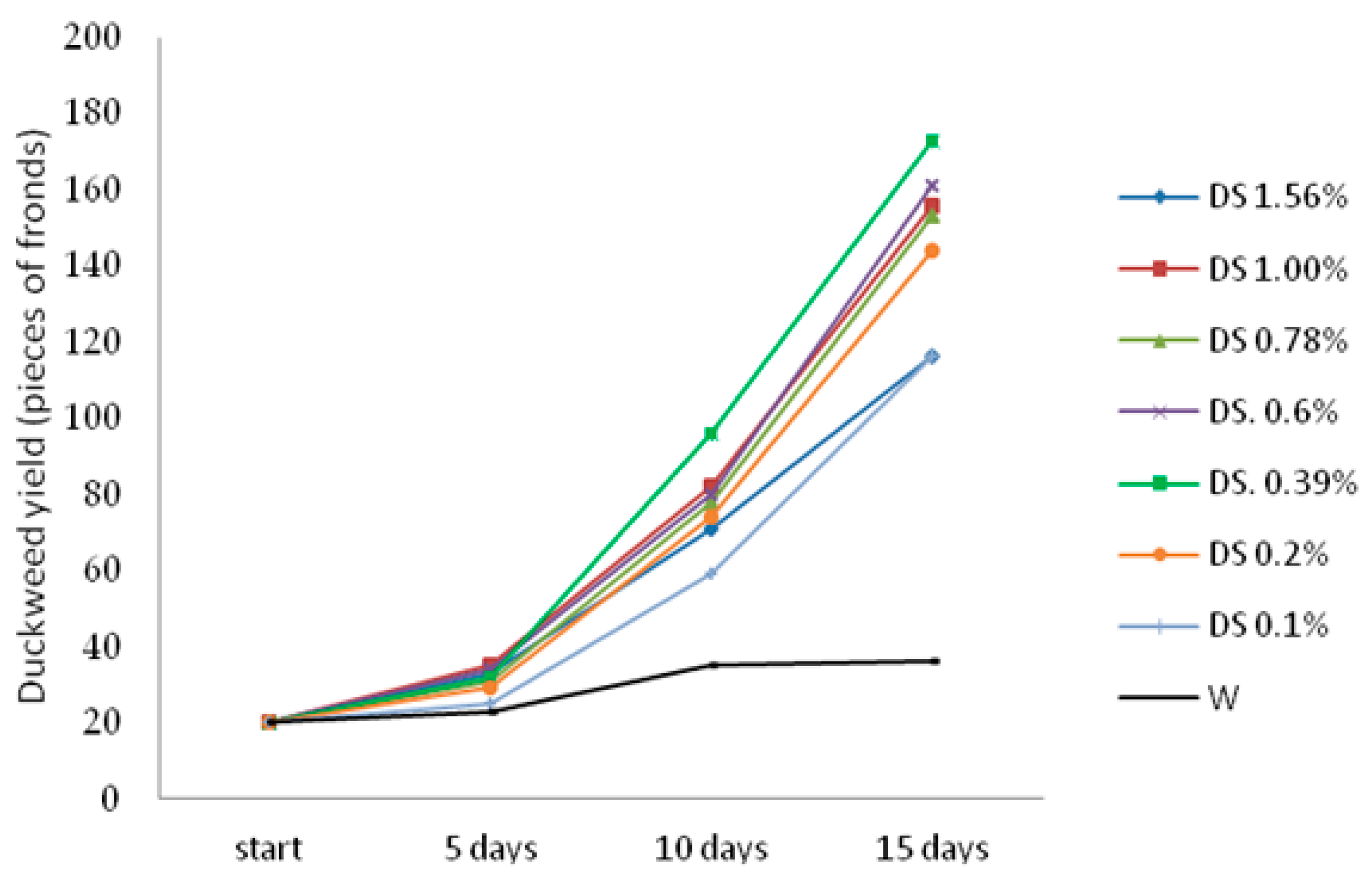
3.1. Duckweed Yields
Under optimal conditions of access to light, carbon dioxide and nutrients, the growth of
Lemnaceae can be exponential [
36]. Among the nutrients, the content of nitrogen and phosphorus have the greatest influence on duckweed growth [
16]. The organic waste analyzed in the present study seems to be a suitable growth medium for duckweed due to the high proportion of ammonium nitrogen in total nitrogen (88% and 78% for DS and EFL, respectively,
Table 1). Duckweed prefers an ammoniacal form of nitrogen because NH
4+ is easily converted into amino acids which form organic N compound [
37]. In our study, the type of growth medium (
Table 2 and
Table 3) used in the experiment affected duckweed growth (
Figure 1 and
Figure 2). Duckweed culture on growth media with high contents of the digestate from a biogas plant or an effluent from a biorefinery was impossible. After 15 days of culture, the highest duckweed yield was obtained on the media with DS or EFL content at 0.39% (173 and 77 pieces of fronds, i.e., 37.8 g FM and 16.8 g FM per box, respectively,
Figure 1 and
Figure 2). The yield of duckweed cultured on the growth media with DS
0.39% and EFL
0.39% was almost 5-fold and 2.5-fold higher, respectively, as compared with the yield obtained from the control box (W). After 15 days of culture, approximately 144–161 pieces of fronds were harvested from each box, i.e., 31.5–35.2 g FM per box for DS
0.2% and DS
0.6%, respectively. In the case of boxes with DS added to the culture medium, the lowest duckweed yields were obtained in those with DS
0.1% and DS
1.56% (116 pieces of fronds, i.e., 25.4 g FM per box). In the box with DS
0.1%, duckweed growth could be inhibited by nitrogen deficiency (3.5 mg N
tot L
−1), whereas in the box with DS
1.56% it could be inhibited by excess nitrogen (54.6 mg N
tot L
−1) and potassium (75.5 mg K
tot L
−1) (
Table 2). Bergman et al. [
30] and Caicedo et al. [
31] proved that the best duckweed growth was obtained when the total nitrogen concentrations ranged from 3.5 to 40 mg N L
−1. A higher concentration of nitrogen than 60 mg N L
−1 resulted in inhibition of duckweed growth which was probably associated with the presence of free ammonia in the growth medium [
38]. According to Körner et al. [
39], the NH
3 toxicity on duckweed occurred with an NH
3-N concentration greater than 1 mg L
−1; the maximum tolerance for unionized ammonia was observed at 8 mg NH
3-N L
−1. Ammonia is the main nutrient limiting duckweed growth, but further research is needed to determine its optimal level for duckweed growth [
38].
In the present experiment, the 5% and higher EFL addition to the growth medium had a negative impact on duckweed yield. In boxes with these media, duckweed decay was observed from the 10th day of culture (
Figure 2), while no duckweed growth was observed in the boxes with growth media containing 2.5% and 3.5% of EFL. In contrast, in the boxes with 0.78% and 0.6% of EFL in the media, duckweed yield reached approximately 50 pieces of fronds, i.e., 10.8 g FM per box. Although in the growth media with EFL
0.78% and EFL
0.6% the contents of nitrogen and phosphorus approximated those optimal for duckweed growth (10–40 mg N L
−1 and 1.5–2.2 mg P L
−1 [
38,
39]), duckweed yields in these boxes were ca. 36% lower than in those with EFL
0.39%. The above results indicate that high concentrations of the effluent from the biorefinery inhibit duckweed growth. The growth limiting factors could be the excessive contents of potassium and sodium ions in the growth medium (23.3–30.2 mg K
tot L
−1 and 56.3–73.2 mg Na
tot L
−1 in EFL
0.6% and EFL
0.78%, respectively). The high potassium content of EFL (3.8 g K
tot kg
−1 FM,
Table 1) was because it was obtained from the liquid fraction of the digestate (featuring a high potassium concentration) subjected to the processes of nitrogen and phosphorus recovery. During separation, a large part of potassium ions contained in the digestate migrates to its liquid fraction because, unlike nitrogen and phosphorus, they are not bound with the organic matter [
4]. In turn, the high sodium content of EFL was due to NaOH addition during earlier struvite precipitation and ammonia stripping from the digestate’s liquid fraction. Study results demonstrate that potassium content (approximately 48 mg L
−1) and, simultaneously, a high sodium content (approximately 117 mg Na L
−1) in the medium inhibited duckweed growth, and that approximately 291 mg K L
−1 and 704 mg Na L
−1 made it impossible (
Figure 2). However, literature data have indicated the feasibility of duckweed culture on media with a high salt content [
38]. Sea salt has been applied in pilot studies of duckweed farming in Bangladesh. Leng [
38] showed, however, that duckweeds tolerated high salt levels when additional nutrients were added into the growth medium.
The literature provides considerable data on the potential growth rates of duckweeds. Landolt and Kandeler [
40] reported that, under optimal conditions, 73 tons dry matter (DM) of duckweed biomass ha
−1 yr
−1 could be produced, i.e., 20 g DM m
−2 d
−1. Under less than optimum conditions, it was possible to obtain between five and 20 tons DM of duckweed biomass ha
−1 per year. In this experiment, the duckweed yield produced was approximately 1.5 and 3.1 g DM m
−2 d
−1 (i.e., 31 and 70 g FM m
−2 d
−1) on the media with EFL
0.39% and DS
0.39%, respectively. Similar yields of duckweed (i.e., 52–96.2 g FM m
−2 d
−1) were obtained by Stadtlander et al. [
41] on household sewage and diluted cattle slurry, respectively. In the present study, assuming that the duckweed production cycle was 15 days, it would be feasible to achieve yields approximating 5.4 and 11.1 tons DM ha
−1 per year on a growth media with EFL
0.39% and DS
0.39%, respectively.
3.2. Nutritional Value of Duckweed
Duckweed produced had a low content of dry matter, i.e., 43.98 g kg
−1 and 47.10 g kg
−1 when grown on the media with DS
0.39% and EFL
0.39%, respectively. Investigations conducted by other authors with various duckweed species showed their dry matter contents ranged from 3 to almost 8% [
18]. Considering the low dry matter content in the fresh matter of duckweed (approximately 4%), its addition to a diet will not significantly increase the energy or protein value of the diet. This is an important factor limiting the applicability of high amounts of duckweed fresh matter in feed ratios for animals from the intensive production systems. The nutritional value and usability of duckweed might be increased when it is dried and added in a dried form to complete feed mixtures. The potential of biomass to be used for feedstuff purposes is determined by protein content in its dry matter. In this study, the protein content of duckweed reached 360.7 g kg
−1 DM and 353.7 g kg
−1 DM when it was grown in the media with EFL
0.39% and DS
0.39%, respectively (
Table 4). Dried duckweed with a protein content of approximately 350 g kg
−1 DM can be considered to be a potential source of protein for farm animals and aquaculture. The content of protein in the studied duckweed was lower than in extracted soybean (46%), but higher than in rapeseed cake (30%), maize DDGS (25–30%), or legume seeds (21–35%), except for yellow lupine (38%) [
42,
43]. Investigations conducted by Rusoff et al. [
44] and Appenroth et al. [
18] pointed to a good amino acid composition of duckweed protein, resembling that of legume seed protein. However, Appenroth et al. [
18] emphasized that its culture conditions significantly determined the total protein content of duckweed. In their study addressing the feasibility of duckweed culture on swine wastewater, Zhao et al. [
23] demonstrated protein content in the biomass which ranged from 280 to 320 g kg
−1 DM. In studies carried out at the University of New England, the crude protein content of duckweed, growing on a diluted effluent from housed pigs, increased with effluent levels of N which increased from about 150 g kg
−1 DM crude protein with low levels of N (1–4 mg N l
−1) to 370 g kg
−1 DM at between 10–15 mg N l
−1 [
38].
The high protein content and biological value of dried duckweed make it an interesting high-protein feedstuff on the condition that costs of its drying are low. The broad utilization of this feedstuff in animal feeding may be limited by its high fiber content (86.92 g kg
−1 DM and 94.87 g kg
−1 DM when duckweed was cultured on the media with EFL
0.39% and DS
0.39%, respectively,
Table 4), which is higher than in such high-protein raw materials as extracted soybean, pea meal, or lupine meal. Therefore, dried duckweed can be used in higher amounts in the feeding of ruminants, such as Equidae or Leporidae.
Regardless of the growth medium type, the crude fat content of duckweed biomass was low and approximated 29.8 g kg
−1 DM for EFL
0.39% and DS
0.39% (
Table 4). In contrast, the analyzed duckweed had a high content of crude ash (224.8 g kg
−1 DM and 220.3 g kg
−1 DM when grown on the media with EFL
0.39% and DS
0.39%, respectively,
Table 4) and, consequently, high contents of minerals. The mineral composition of duckweeds tested is presented in
Table 5. The duckweeds differed significantly in the contents of Ca (
p ≤ 0.05) and P (
p ≤ 0.01), which were significantly higher in the plants cultured in the medium with DS
0.39% as compared with EFL
0.39%, i.e., 4.32 g vs. 3.08 g Ca kg
−1 and 4.54 g vs. 3.82 g P kg
−1, respectively (
Table 5). Appenroth et al. [
45] obtained higher contents of these elements in various species of
Wolffia genus duckweed, i.e., approximately 20.4 g Ca kg
−1 and 16.2 g P kg
−1 DM on average.
Among all determined macroelements, duckweed featured the highest contents of potassium (23.08 and 18.98 g K kg
−1 DM when grown with EFL
0.39% and DS
0.39%, respectively) and sodium (10.58 and 7.87 g Na kg
−1 when grown with EFL
0.39% and DS
0.39%, respectively). These values differ from the literature data. According to Appenroth et al. [
45], the potassium content of duckweed reached approximately 67.8 mg kg
−1 DM and that of sodium reached 0.25 g kg
−1 DM. Considering microelements and heavy metals analyzed in duckweed in the present study, worthy of attention are high contents of zinc (226.66 mg Zn kg
−1 DM and 186.98 mg Zn kg
−1 DM when duckweed was grown in the media with EFL
0.39% and DS
0.39%, respectively) and aluminum (189.94 mg Al kg
−1 DM and 161.28 mg Al kg
−1 DM for EFL
0.39% and DS
0.39%, respectively). The determined content of zinc was nearly four times higher than its average content reported by Appenroth et al. [
45], i.e., 53 mg kg
−1 DM. The contents of minerals in duckweed are significantly influenced by their contents in the medium it grows in [
46]. This is due to the high ability of this aquatic plant to absorb and accumulate metal ions, including heavy metals, from water [
47]. Therefore, duckweed is used to remove minerals from water [
48]. This may explain differences between the contents of elements (Ca, P, K, Na, and Zn) noted in this study and those reported by other authors [
18,
45].
Table 6 presents the contents of α-tocopherol and carotenoids in duckweed cultured in different growth media. Significant differences between duckweeds were demonstrated in their contents of β-carotene and zeaxanthin (
p ≤ 0.05). Higher contents of these components were determined in duckweed grown on the medium with EFL
0.39% than with DS
0.39%. In turn, β-carotene content was higher by 43.4 mg kg
−1 DM, and zeaxanthin content by 4.7 mg kg
−1 DM. Regardless of the growth medium type, the major carotenoid of duckweed proved to be lutein, followed by β-carotene and violaxanthin. The concentrations of the other carotenoids tested were significantly lower.
Investigations conducted by Appenroth et al. [
18,
45] demonstrated similar contents of individual carotenoids and α-tocopherol in duckweed. The present study proved that duckweed, and in particular dried duckweed, could be a rich source of these compounds for animals. Alpha-tocopherol and carotenoids are both valuable antioxidants and serve important functions in the immune system of animals [
49,
50,
51]. A comparison of α-tocopherol contents in dry matter of duckweed, grasses, silages, or hay has shown it was similar or higher [
52,
53,
54]. In turn, contents of β-carotene, lutein, or zeaxanthin in duckweed dry matter were higher than in a dry matter of green forage, as well as grass and hay silages [
55,
56,
57,
58]. Due to low contents of α-tocopherol and carotenoids in maize silage, the addition of fresh or dried duckweed can complete contents of these bioactive compounds and improve feedstuff quality [
53,
56].
3.3. Nutrients Uptake
Depending on the species, N and P uptake by duckweed could vary between 150–1670 mg N m
−2 d
−1 and 15–300 g P m
−2 d
−1 [
59]. Other authors reported that nutrient uptake rates of duckweed vary between 45 and 1670 mg N m
−2 d
−1 and between 8 and 220 mg P m
−2 d
−1 [
39]. In this study, N uptake by
Lemna minuta was 45.74 and 94.16 mg per box for EFL
0.39% and DS
0.39%, respectively (i.e., 84.7 and 174.4 mg N m
−2 d
−1) (
Table 7). At the same time, the P uptake was 3.03 and 7.55 mg P per box (i.e., 5.61 and 13.98 mg P m
−2 d
−1). Due to higher duckweed yields obtained at DS
0.39%, nutrient uptake by
Lemna minuta from DS
0.39% was higher than from EFL
0.39%, i.e., by approximately 73, 98, 44, 73, and 437% for K, Mg, Ca, Zn, and Cu, respectively (
Table 7). Landolt [
16] demonstrated that the uptake of nutrients by duckweeds increased with their increased concentrations in the medium. Therefore, the
Lemna minuta uptook larger amounts of nutrients from DS
0.39% than from EFL
0.39%. However, EFL
0.39% contained more sodium than DS
0.39% (36.6 and 2.8 mg Na
tot L
−1, respectively,
Table 2 and
Table 3), which explains the 15% higher uptake of this nutrient from EFL
0.39%.
3.4. Nutrient Recovery
The usefulness of duckweed for the purification of numerous wastewaters was analyzed by Körner et al. [
60]. After 12–24 days of growth, duckweed removed 40 to 60% of the nitrogen from a solution. Goopy and Murray [
36] reported that
Lemnaceae usually used from 30 to 50% of dissolved phosphorus. Lower P recovery than N from organic waste may be because P can be absorbed by organic matter or precipitated with Ca
2+, Fe
3+, and Al
3+ [
61]. Alaerts at al. [
62] showed that duckweed utilized 74% nitrogen and 77% phosphorus from stabilized sewage. Zhou et al. [
24] reported that over 93% of nitrogen and phosphorus was removed from municipal wastewater during 15 days of cultivation of four species of duckweed [
24]. Similar data were presented by Yu et al. [
63] and Cheng and Stomp [
14] who studied duckweed growth on sewage water and wastewater from a hog farm, respectively. A higher level of nitrogen and phosphorus removal (98%) was recorded by Mohedano et al. [
64] after duckweed cultivation on a pig-farm effluent. In the present study, N recovery reached approximately 75% and 81%, whereas P recovery reached approximately 45% and 55% from EFL
0.39% and DS
0.39%, respectively (
Table 8). The recovery of other components was lower, except for zinc, whose recovery level was similar to that of P. The tested
Lemna minuta was able to remove more than 14% and 19% potassium, and 18% and 11% calcium from EFL
0.39% and DS
0.39%, respectively. Magnesium recovery was similar for both growth media (i.e., approximately 35%). The lowest recovery was obtained for copper, reaching only 1.6% on average. Sodium recovery from DS
0.39% reached 33%, whereas it was only 2.9% from EFL
0.39% (
Table 8). The results obtained show that after duckweed harvest, there was still a significant amount of sodium in the growth medium. Therefore, research should be continued to find a different species of duckweed that will absorb more sodium from the growth medium.
3.5. Land Requirement
Due to the relatively high yield of duckweed with a proven forage value and the high degree of nitrogen and phosphorus removal from a growth medium, it can be concluded that this treatment of anaerobic digestate and effluent from biorefinery can be successfully carried out on a large scale. Pond depth intended for duckweed cultivation should be less than 0.5 m. Such ponds ensure that the plants grown there will have full access to the nutrients [
65]. Assuming that the depth of the pond for duckweed cultivation is 0.5 m, the share of DS or EFL will be 0.39%, the duckweed cultivation cycle will last 15 days, and it will be possible to utilize about 468 m
3 of DS or EFL on 1 ha per year. This indicates that the proposed solution can be implemented preferably by small size biogas plants or biorefineries. The use of such low DS or EFL concentrations (0.39%) in the growth medium requires significant amounts of freshwater, and significant area, which may be a problem in large scale installations. In order to reduce this problem, it is necessary to find another species of duckweed that can tolerate higher concentrations of potassium and sodium in the growth medium than that of the tested
Lemna minuta. A duckweed pond can produce approximately 5.4 and 11.1 ton DM ha
−1 per year on EFL
0.39% and DS
0.39% ponds, respectively. With a protein content in duckweed of approximately 354–361 g kg
−1 (
Table 4), it is possible to obtain 2–4 t of protein per 1 ha yearly from EFL
0.39% and DS
0.39% ponds, respectively. For comparison, with an average alfalfa yield of approximately 8–15 t DM ha
−1, the protein yield is approximately 2–2.5 t [
66,
67,
68]. The conducted research indicates that duckweed culture may be a promising source of protein.