Solar-hybrid Thermochemical Gasification of Wood Particles and Solid Recovered Fuel in a Continuously-Fed Prototype Reactor
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
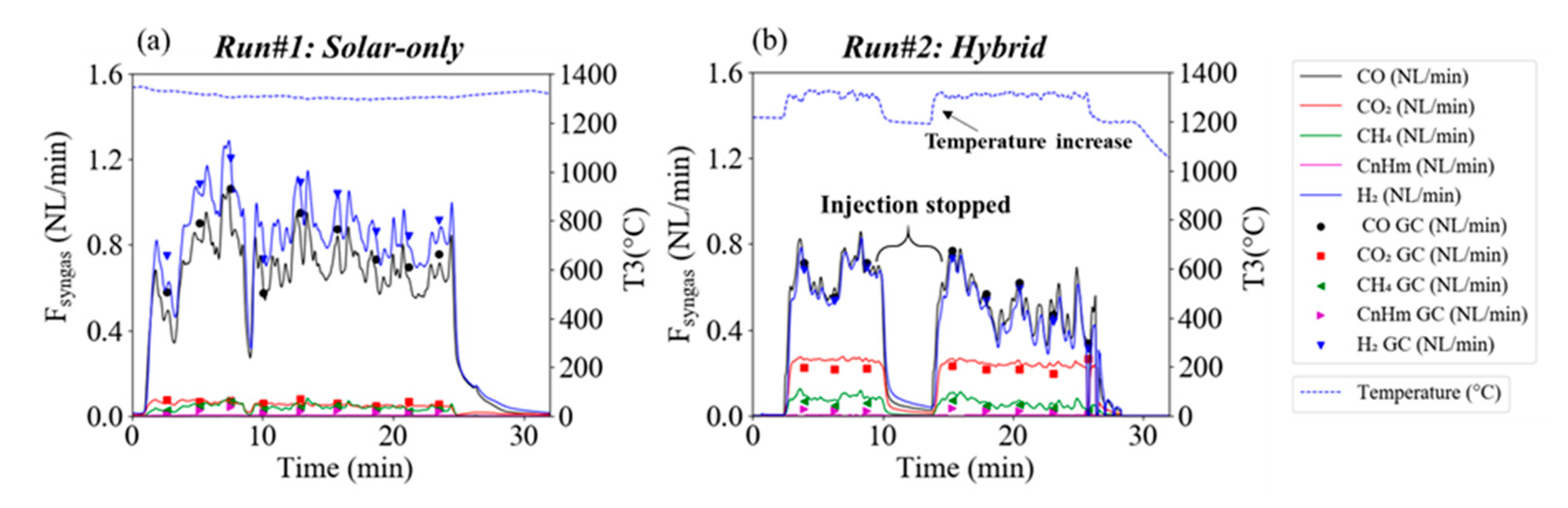
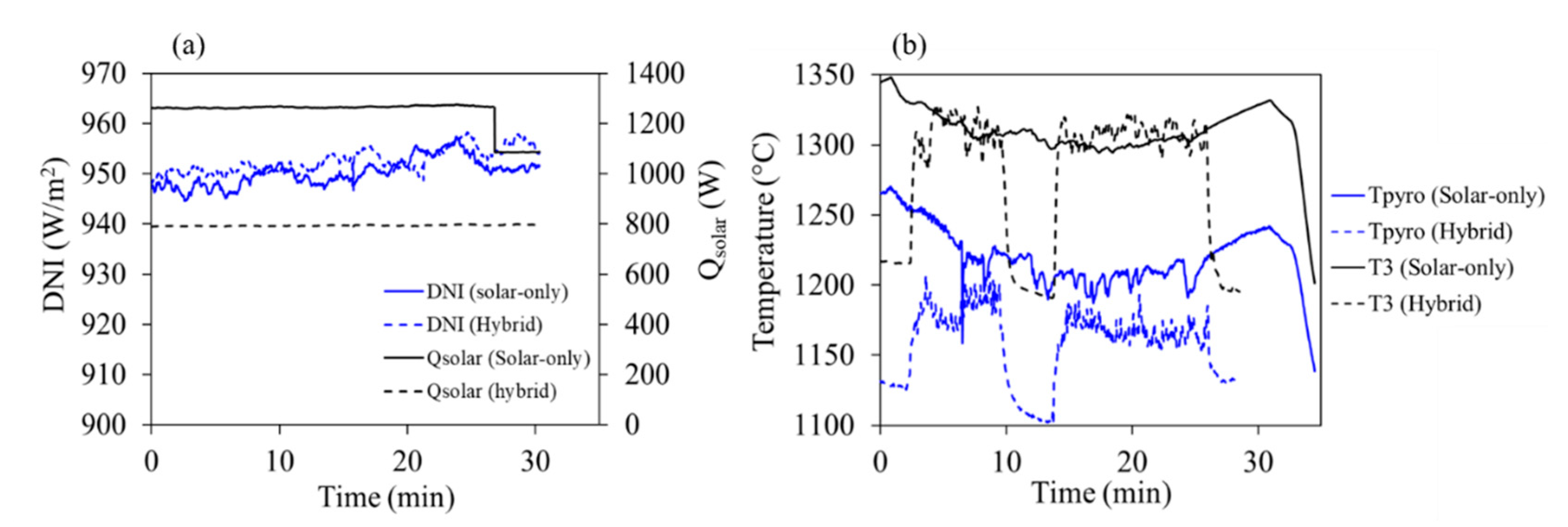
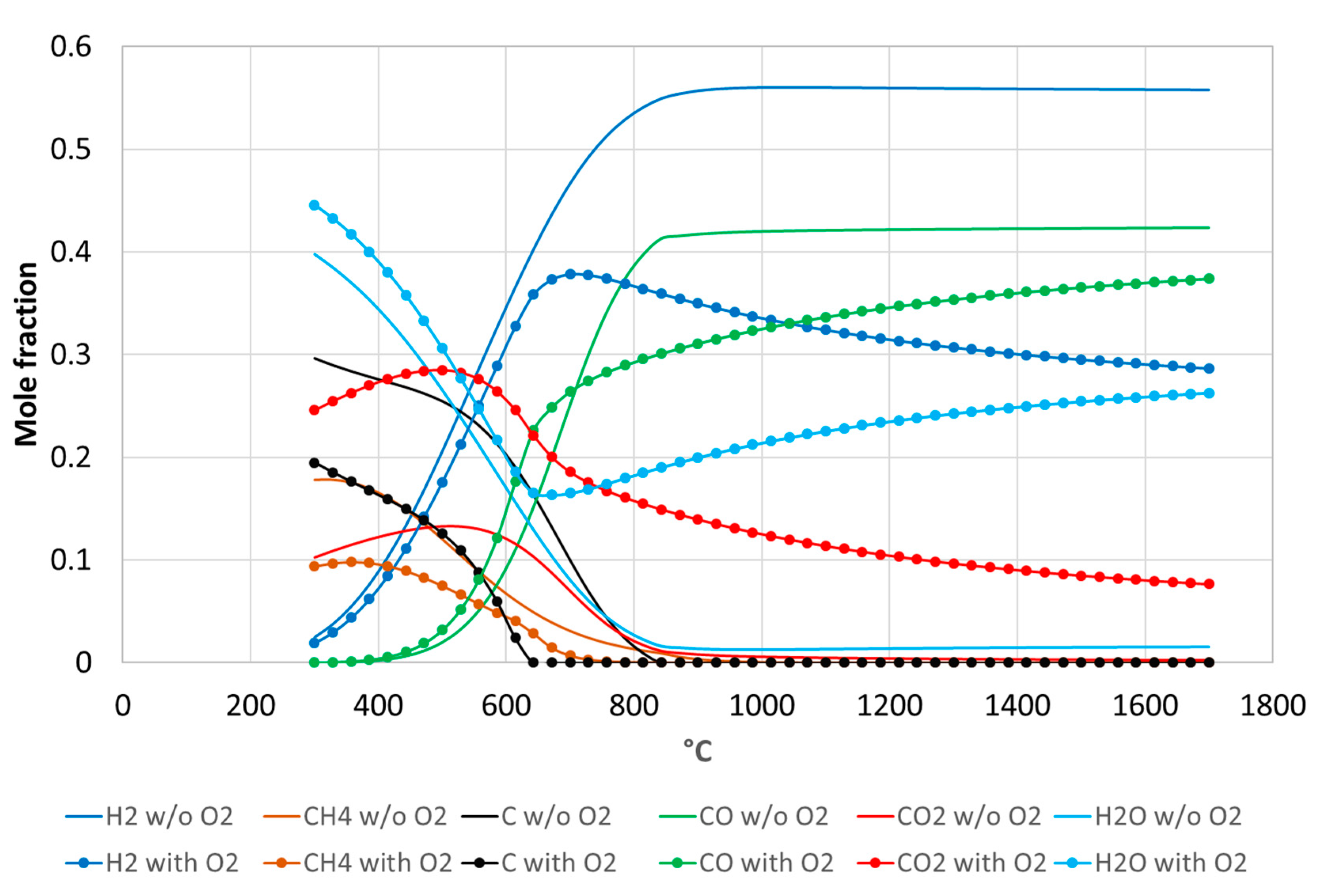
3.1. Beechwood Gasification
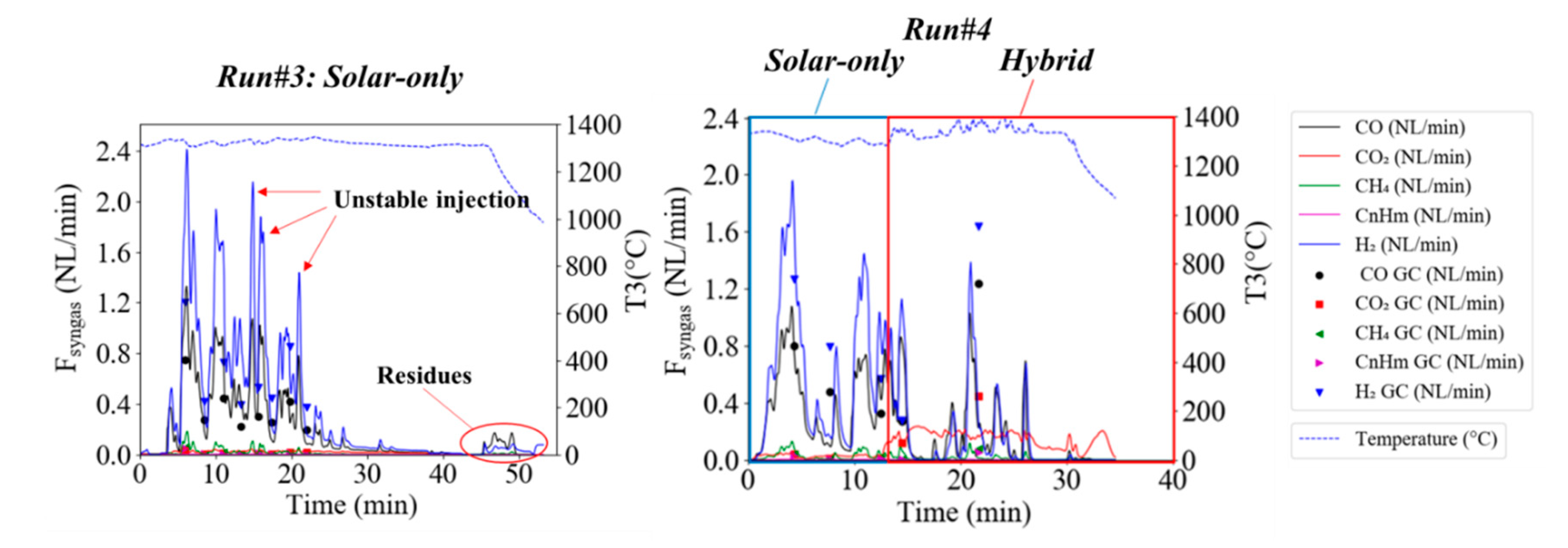
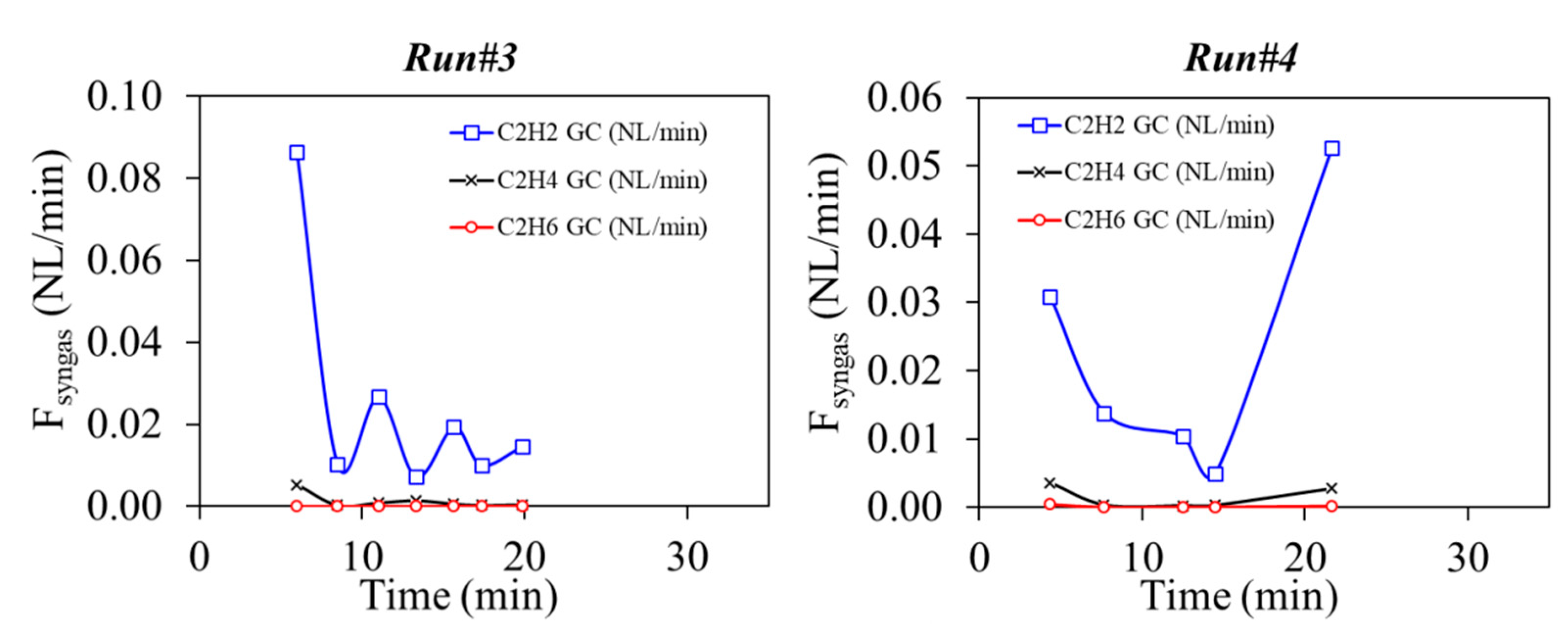
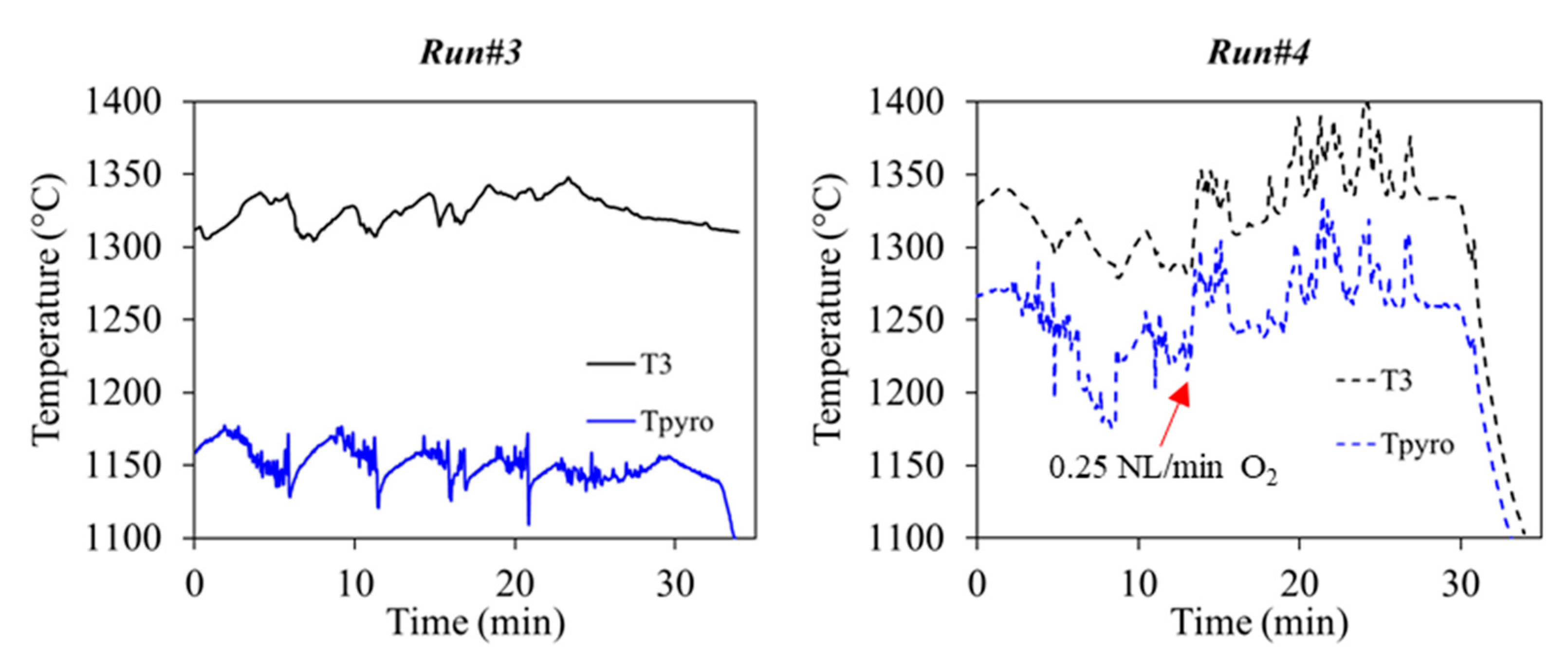
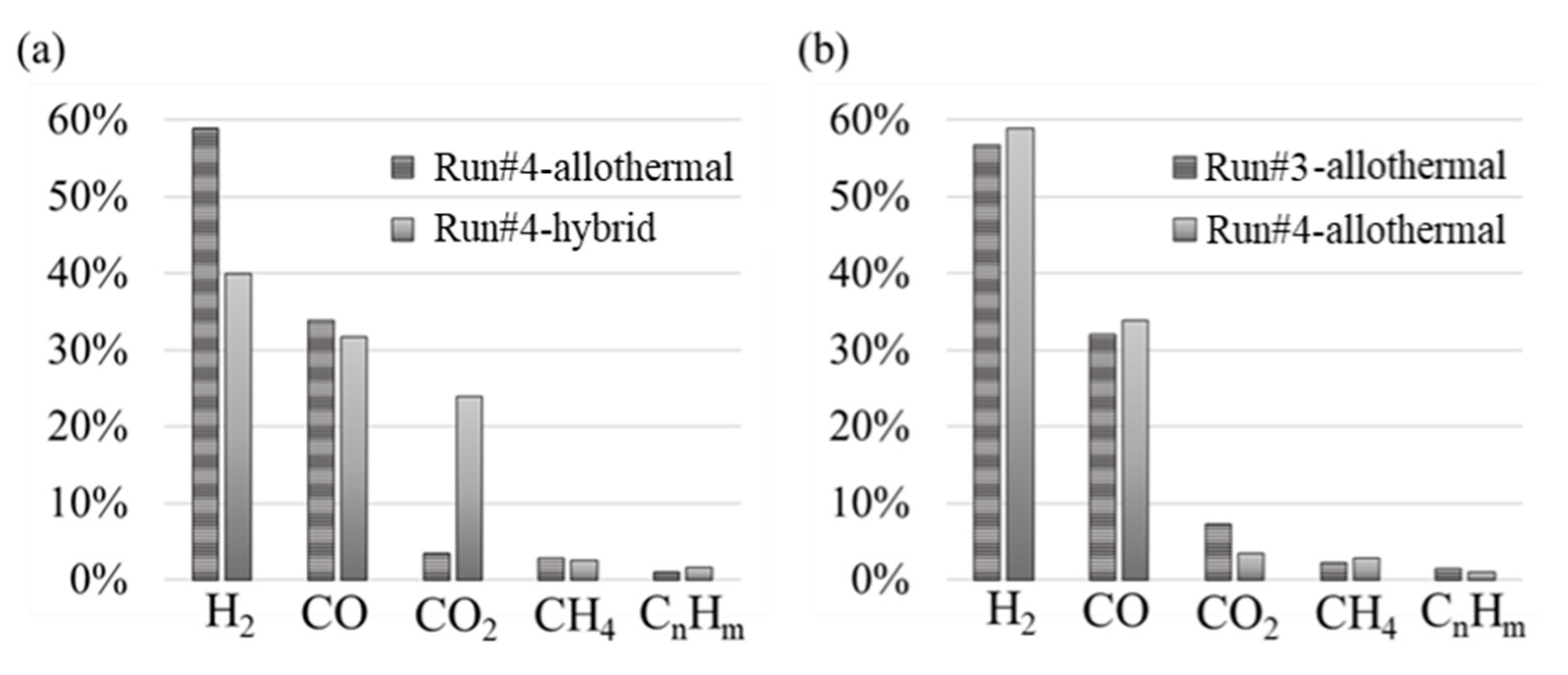
3.2. Solid Recovered Fuels Gasification
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Probstein, R.F.; Hicks, E.H. Synthetic Fuels; Dover Publications Inc.: Mineola, NY, USA, 2006; p. 11501. [Google Scholar]
- Breault, R.W. Gasification processes old and new: A basic review of the major technologies. Energies 2010, 3, 216–240. [Google Scholar] [CrossRef]
- Jafri, Y.; Furusjö, E.; Kirtania, K.; Gebart, B.R. Performance of a pilot-scale entrained-flow black liquor gasifier. Energy Fuels 2016, 30, 3175–3185. [Google Scholar] [CrossRef]
- Larsson, A.; Seemann, M.; Neves, D.; Thunman, H. Evaluation of performance of industrial-scale dual fluidized bed gasifiers using the chalmers 2–4-MWth gasifier. Energy Fuels 2013, 27, 6665–6680. [Google Scholar] [CrossRef]
- Pfeifer, C.; Koppatz, S.; Hofbauer, H. Steam gasification of various feedstocks at a dual fluidised bed gasifier: Impacts of operation conditions and bed materials. Biomass Convers. Biorefinery 2011, 1, 39–53. [Google Scholar] [CrossRef]
- Valin, S.; Ravel, S.; De Vincent, P.P.; Thiery, S.; Miller, H. Fluidized bed air gasification of solid recovered fuel and woody biomass: Influence of experimental conditions on product gas and pollutant release. Fuel 2019, 242, 664–672. [Google Scholar] [CrossRef]
- Wu, H.; Liu, Q.; Bai, Z.; Xie, G.; Zheng, J.; Su, B. Thermodynamics analysis of a novel steam/air biomass gasification combined cooling, heating and power system with solar energy. Appl. Therm. Eng. 2020, 164, 114494. [Google Scholar] [CrossRef]
- Guo, P.; Saw, W.L.; Van Eyk, P.; Ashman, P.; Nathan, G.; Stechel, E. Fischer-tropschliquid fuel production by co-gasification of coal and biomass in a solar hybrid dual fluidized bed gasifier. Energy Procedia 2015, 69, 1770–1779. [Google Scholar] [CrossRef]
- Bruckner, A.P. Continuous duty solar coal gasification system using molten slag and direct-contact heat exchange. Sol. Energy 1985, 34, 239–247. [Google Scholar] [CrossRef]
- Hathaway, B.J.; Davidson, J.H. Demonstration of a prototype molten salt solar gasification reactor. Sol. Energy 2017, 142, 224–230. [Google Scholar] [CrossRef]
- Loutzenhiser, P.G.; Muroyama, A.P. A review of the state-of-the-art in solar-driven gasification processes with carbonaceous materials. Sol. Energy 2017, 156, 93–100. [Google Scholar] [CrossRef]
- Arnavat, M.P.; Tora, E.; Bruno, J.C.; Coronas, A. State of the art on reactor designs for solar gasification of carbonaceous feedstock. Sol. Energy 2013, 97, 67–84. [Google Scholar] [CrossRef]
- Yadav, D.; Banerjee, R. A review of solar thermochemical processes. Renew. Sustain. Energy Rev. 2016, 54, 497–532. [Google Scholar] [CrossRef]
- Chuayboon, S.; Abanades, S. An overview of solar decarbonization processes, reacting oxide materials, and thermochemical reactors for hydrogen and syngas production. Int. J. Hydrog. Energy 2020, 45, 25783–25810. [Google Scholar] [CrossRef]
- Bellouard, Q.; Rodat, S.; Abanades, S.; Ravel, S.; Frayssines, P.-É. Design, simulation and experimental study of a directly-irradiated solar chemical reactor for hydrogen and syngas production from continuous solar-driven wood biomass gasification. Int. J. Hydrog. Energy 2019, 44, 19193–19205. [Google Scholar] [CrossRef]
- Bellouard, Q.; Rodat, S.; Dupassieux, N.; Abanades, S. A high temperature drop-tube and packed-bed solar reactor for continuous biomass gasification. AIP Conf. Proc. 2017, 1850, 100001. [Google Scholar] [CrossRef]
- Kodama, T.; Kondoh, Y.; Tamagawa, T.; Funatoh, A.; Shimizu, K.-I.; Kitayama, Y. Fluidized bed coal gasification with CO2 under direct irradiation with concentrated visible light. Energy Fuels 2002, 16, 1264–1270. [Google Scholar] [CrossRef]
- Gokon, N.; Ono, R.; Hatamachi, T.; Liuyun, L.; Kim, H.-J.; Kodama, T. CO2 gasification of coal cokes using internally circulating fluidized bed reactor by concentrated Xe-light irradiation for solar gasification. Int. J. Hydrog. Energy 2012, 37, 12128–12137. [Google Scholar] [CrossRef]
- Zgraggen, A.; Haueter, P.; Trommer, D.; Romero, M.; DeJesus, J.; Steinfeld, A. Hydrogen production by steam-gasification of petroleum coke using concentrated solar power—II Reactor design, testing, and modeling. Int. J. Hydrog. Energy 2006, 31, 797–811. [Google Scholar] [CrossRef]
- Piatkowski, N.; Wieckert, C.; Steinfeld, A. Experimental investigation of a packed-bed solar reactor for the steam-gasification of carbonaceous feedstocks. Fuel Process. Technol. 2009, 90, 360–366. [Google Scholar] [CrossRef]
- Müller, F.; Poživil, P.; Van Eyk, P.; Villarrazo, A.; Haueter, P.; Wieckert, C.; Nathan, G.; Steinfeld, A. A pressurized high-flux solar reactor for the efficient thermochemical gasification of carbonaceous feedstock. Fuel 2017, 193, 432–443. [Google Scholar] [CrossRef]
- Lichty, P.; Perkins, C.; Woodruff, B.; Bingham, C.; Weimer, A. Rapid high temperature solar thermal biomass gasification in a prototype cavity reactor. J. Sol. Energy Eng. 2010, 132, 011012. [Google Scholar] [CrossRef]
- Kruesi, M.; Jovanovic, Z.R.; Santos, E.C.D.; Yoon, H.C.; Steinfeld, A. Solar-driven steam-based gasification of sugarcane bagasse in a combined drop-tube and fixed-bed reactor—Thermodynamic, kinetic, and experimental analyses. Biomass Bioenergy 2013, 52, 173–183. [Google Scholar] [CrossRef]
- Gregg, D.; Taylor, R.; Campbell, J.; Taylor, J.; Cotton, A. Solar gasification of coal, activated carbon, coke and coal and biomass mixtures. Sol. Energy 1980, 25, 353–364. [Google Scholar] [CrossRef]
- Piatkowski, N.; Steinfeld, A. Solar-driven coal gasification in a thermally irradiated packed-bed reactor. Energy Fuels 2008, 22, 2043–2052. [Google Scholar] [CrossRef]
- Abe, T.; Gokon, N.; Izawa, T.; Kodama, T. Internally-circulating fluidized bed reactor using thermal storage material for solar coal coke gasification. Energy Procedia 2015, 69, 1722–1730. [Google Scholar] [CrossRef][Green Version]
- Boujjat, H.; Rodat, S.; Chuayboon, S.; Abanades, S. Experimental and CFD investigation of inert bed materials effects in a high-temperature conical cavity-type reactor for continuous solar-driven steam gasification of biomass. Chem. Eng. Sci. 2020, 228, 115970. [Google Scholar] [CrossRef]
- Z’Graggen, A.; Steinfeld, A. A two-phase reactor model for the steam-gasification of carbonaceous materials under concentrated thermal radiation. Chem. Eng. Process. Process. Intensif. 2008, 47, 655–662. [Google Scholar] [CrossRef]
- Bellouard, Q.; Abanades, S.; Rodat, S.; Dupassieux, N. Solar thermochemical gasification of wood biomass for syngas production in a high-temperature continuously-fed tubular reactor. Int. J. Hydrog. Energy 2017, 42, 13486–13497. [Google Scholar] [CrossRef]
- Hathaway, B.J.; Kittelson, D.; Davidson, J.H. Development of a molten salt reactor for solar gasification of biomass. Energy Procedia 2014, 49, 1950–1959. [Google Scholar] [CrossRef]
- Muroyama, A.P.; Guscetti, I.; Schieber, G.L.; Haussener, S.; Loutzenhiser, P.G. Design and demonstration of a prototype 1.5 kWth hybrid solar/autothermal steam gasifier. Fuel 2018, 211, 331–340. [Google Scholar] [CrossRef]
- Boujjat, H.; Rodat, S.; Chuayboon, S.; Abanades, S. Experimental and numerical study of a directly irradiated hybrid solar/combustion spouted bed reactor for continuous steam gasification of biomass. Energy 2019, 189, 116118. [Google Scholar] [CrossRef]
- Boujjat, H.; Junior, G.M.Y.; Rodat, S.; Abanades, S. Dynamic simulation and control of solar biomass gasification for hydrogen-rich syngas production during allothermal and hybrid solar/autothermal operation. Int. J. Hydrog. Energy 2020, 45, 25827–25837. [Google Scholar] [CrossRef]
- Rodat, S.; Abanades, S.; Boujjat, H.; Chuayboon, S. On the path toward day and night continuous solar high temperature thermochemical processes: A review. Renew. Sustain. Energy Rev. 2020, 132, 110061. [Google Scholar] [CrossRef]
- Ischia, G.; Castello, D.; Orlandi, M.; Miotello, A.; Rosendahl, L.A.; Fiori, L. Waste to biofuels through zero-energy hydrothermal solar plants: Process design. Chem. Eng. Trans. 2020, 80, 7–12. [Google Scholar]
- Ischia, G.; Orlandi, M.; Fendrich, M.; Bettonte, M.; Merzari, F.; Miotello, A.; Fiori, L. Realization of a solar hydrothermal carbonization reactor: A zero-energy technology for waste biomass valorization. J. Environ. Manag. 2020, 259, 110067. [Google Scholar] [CrossRef]
- Arena, U.; Di Gregorio, F. Gasification of a solid recovered fuel in a pilot scale fluidized bed reactor. Fuel 2014, 117, 528–536. [Google Scholar] [CrossRef]
- Fryda, L.; Panopoulos, K.; Kakaras, E. Agglomeration in fluidised bed gasification of biomass. Powder Technol. 2008, 181, 307–320. [Google Scholar] [CrossRef]
- Kirnbauer, F.; Hofbauer, H. Investigations on bed material changes in a dual fluidized bed steam gasification plant in Gussing, Austria. Energy Fuels 2011, 25, 3793–3798. [Google Scholar] [CrossRef]
- ADEME. État de l’art de la Production et de l’utilisation de Combustibles Solides de. 2012. Available online: https://www.ademe.fr/etat-lart-production-lutilisation-combustibles-solides-recuperation (accessed on 20 September 2020).
- Bellouard, Q.; Abanades, S.; Rodat, S. Biomass gasification in an innovative spouted-bed solar reactor: Experimental proof of concept and parametric study. Energy Fuels 2017, 31, 10933–10945. [Google Scholar] [CrossRef]
- Boujjat, H.; Rodat, S.; Chuayboon, S.; Abanades, S. Numerical simulation of reactive gas-particle flow in a solar jet spouted bed reactor for continuous biomass gasification. Int. J. Heat Mass Transf. 2019, 144, 118572. [Google Scholar] [CrossRef]
- Speight, J.G. Combustion of Hydrocarbons, in Handbook of Industrial Hydrocarbon Processes; Elsevier: Amsterdam, The Nederland, 2011. [Google Scholar]
- Chuayboon, S.; Abanades, S.; Rodat, S. Insights into the influence of biomass feedstock type, particle size and feeding rate on thermochemical performances of a continuous solar gasification reactor. Renew. Energy 2019, 130, 360–370. [Google Scholar] [CrossRef]
- Chuayboon, S.; Abanades, S.; Rodat, S. Experimental analysis of continuous steam gasification of wood biomass for syngas production in a high-temperature particle-fed solar reactor. Chem. Eng. Process. Process. Intensif. 2018, 125, 253–265. [Google Scholar] [CrossRef]
- Chuayboon, S.; Abanades, S.; Rodat, S. Comprehensive performance assessment of a continuous solar-driven biomass gasifier. Fuel Process. Technol. 2018, 182, 1–14. [Google Scholar] [CrossRef]
- Glassman, I. Combustion, 3rd ed.; Academic Press: New York, NY, USA, 1996; p. 631. [Google Scholar]
- Goodwin, D.G.; Speth, R.L.; Moffat, H.K.; Weber, B.W. Cantera: An Object-Oriented Software Toolkit for Chemical Kinetics, Thermodynamics, and Transport Processes. 2017. Available online: https://zenodo.org/record/1174508#.X3KE1-0RVEY (accessed on 24 August 2018).
- Kwon, E.; Westby, K.J.; Castaldi, M.J. Transforming Municipal Solid Waste (MSW) into fuel via the gasification/pyrolysis process. In North American Waste-to-Energy Conference, Proceedings of the 18th Annual North American Waste-to-Energy Conference, Orlando, FL, USA, 11–13 May 2010; ASME International: New York, NY, USA, 2010; pp. 53–60. [Google Scholar]
- Khosasaeng, T.; Suntivarakorn, R. Effect of equivalence ratio on an efficiency of single throat downdraft gasifier using RDF from municipal solid waste. Energy Procedia 2017, 138, 784–788. [Google Scholar] [CrossRef]
- Materazzi, M.; Lettieri, P.; Taylor, R.; Chapman, C. Performance analysis of RDF gasification in a two stage fluidized bed–plasma process. Waste Manag. 2016, 47, 256–266. [Google Scholar] [CrossRef]
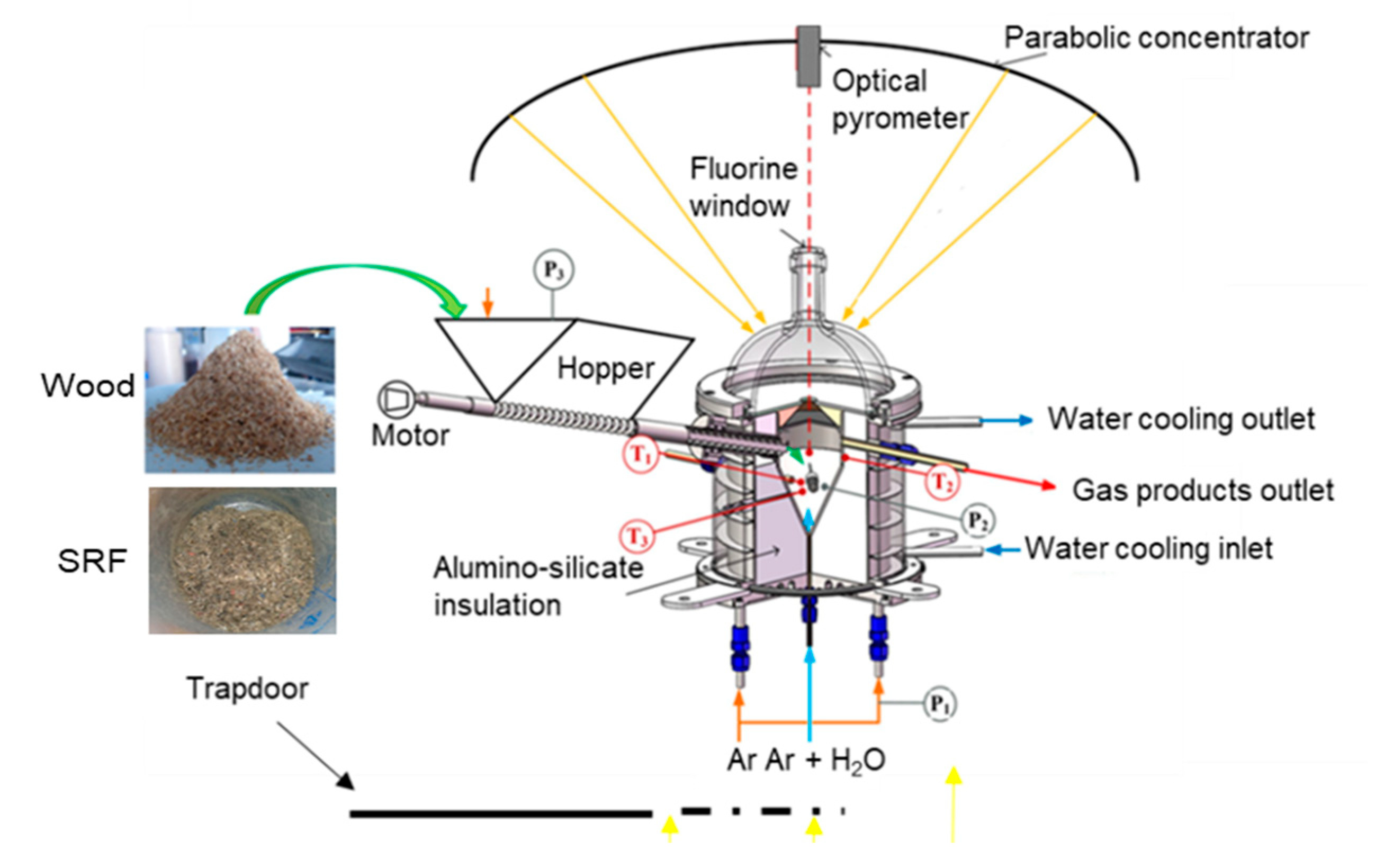



| C (wt.%) | H (wt.%) | O (wt.%) | N (wt.%) | S (wt.%) | Ash (wt.%) | Cl (wt.%) | Moisture (wt.%) | LHV (MJ/kg) | |
|---|---|---|---|---|---|---|---|---|---|
| SRF | 48.6 | 5.7 | 25.8 | 2.9 | 0.9 | 15.0 | 1.1 | 8.9 | 20.6 |
| wood | 48.3 | 6.7 | 44.4 | 0.1 | <0.1% | 0.4 | <0.1% | 8.9 | 16.8 |
| Beechwood Particles | Solid Recovered Fuels | ||||
|---|---|---|---|---|---|
| Runs | #1 | #2 | #3 | #4 | |
| Temperature, Toperating (°C) | 1300 | 1300 | 1300 | 1300–1350 | |
| Feedstock mass, mfeedstock (g) | 30.00 | 27.90 | 20.00 | 20.00 | |
| Voltage to feeder motor, Umotor (V) | 9.5 | 10.5 | 12.0 | 12.0 | |
| Operating mode | Allothermal | Hybrid | Allothermal | Allothermal | Hybrid |
| Qsolar (kWthermal) | 1.2 | 0.8 | 1.0 | 1.0 | 1.0 |
| Ffeedstock (g.min−1) | 1.20 | 1.40 | 0.57 | 0.58 | 0.58 |
| Fsteam (g.min−1) | 0.20 | 0.20 | 0.25 | 0.20 | 0.20 |
| Foxygen (NL.min−1) | 0 | 0.25 | 0 | 0 | 0.25 |
| (S/B)/(S/B)st | 1.24 | 1.13 | 1.06 | 0.87 | 0.87 |
| Run | Gas Production (mmol/gbiomass,dry) | Mass Balance (g) | Closure | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| H2 | CO | CO2 | CH4 | CnHm | Reactants | Products | |||||
| Biomass | H2O | O2 | Gas | Residues | % | ||||||
| #1 | 33.88 | 27.49 | 2.26 | 1.67 | 0.86 | 30.00 | 6.70 | 0.00 | 26.95 | 7.66 | 94.8 |
| #2 | 19.26 | 20.46 | 8.73 | 2.47 | 1.04 | 27.86 | 5.19 | 7.00 | 26.97 | 12.05 | 96.0 |
| Run | Energy breakdown (kJ) | CCE | CGE | SFE | ||
|---|---|---|---|---|---|---|
| Biomass | Syngas | Solar | ||||
| #1 | 460 | 520 | 2498 | 83.3% | 112.9% | 17.6% |
| #2 | 427 | 362 | 1457 | 84.6% | 84.2% | 17.6% |
| Run | Gas Production (mmol/gSRF) | Mass Balance (g) | Closure | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| H2 | CO | CO2 | CH4 | CnHm | Reactants | Products | |||||
| SRF | H2O | O2 | Gas | Residues | % | ||||||
| #3 | 44.73 | 25.25 | 5.76 | 1.79 | 1.19 | 20.00 | 8.85 | 0.00 | 20.23 | 4.95 | 87.7 |
| #4 | 30.98 | 20.22 | 7.87 | 1.70 | 0.80 | 20.00 | 6.54 | 8.51 | 18.63 | 11.87 | 87.3 |
| Run | Energy Breakdown (kJ) | CCE | CGE | SFE | ||
|---|---|---|---|---|---|---|
| SRF | Syngas | Solar | ||||
| #3 | 375 | 391 | 2102 | 88.1% | 104.5% | 15.8% |
| #4 | 375 | 292 | 2086 | 78.5% | 78.0% | 11.9% |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Boujjat, H.; Rodat, S.; Abanades, S. Solar-hybrid Thermochemical Gasification of Wood Particles and Solid Recovered Fuel in a Continuously-Fed Prototype Reactor. Energies 2020, 13, 5217. https://doi.org/10.3390/en13195217
Boujjat H, Rodat S, Abanades S. Solar-hybrid Thermochemical Gasification of Wood Particles and Solid Recovered Fuel in a Continuously-Fed Prototype Reactor. Energies. 2020; 13(19):5217. https://doi.org/10.3390/en13195217
Chicago/Turabian StyleBoujjat, Houssame, Sylvain Rodat, and Stéphane Abanades. 2020. "Solar-hybrid Thermochemical Gasification of Wood Particles and Solid Recovered Fuel in a Continuously-Fed Prototype Reactor" Energies 13, no. 19: 5217. https://doi.org/10.3390/en13195217
APA StyleBoujjat, H., Rodat, S., & Abanades, S. (2020). Solar-hybrid Thermochemical Gasification of Wood Particles and Solid Recovered Fuel in a Continuously-Fed Prototype Reactor. Energies, 13(19), 5217. https://doi.org/10.3390/en13195217





