Use of Carbon Additives towards Rechargeable Zinc Slurry Air Flow Batteries
Abstract
1. Introduction
2. Materials and Methods
2.1. Air Electrode
2.2. Zinc Slurry Preparation
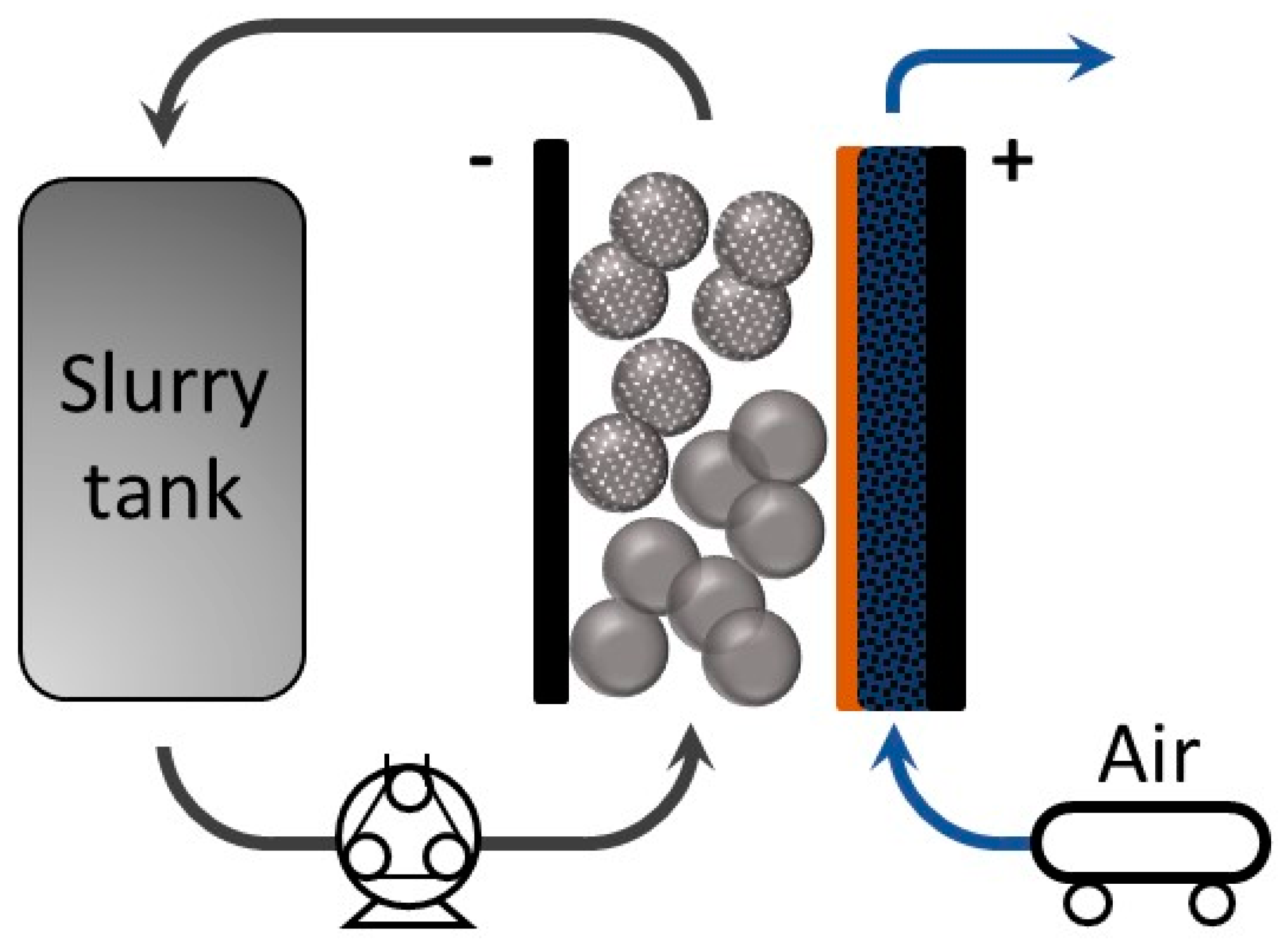
2.3. Cell Design and Electrochemical Characterization
2.4. Rheometry
3. Results
3.1. Oscillatory and Shear Rheometry
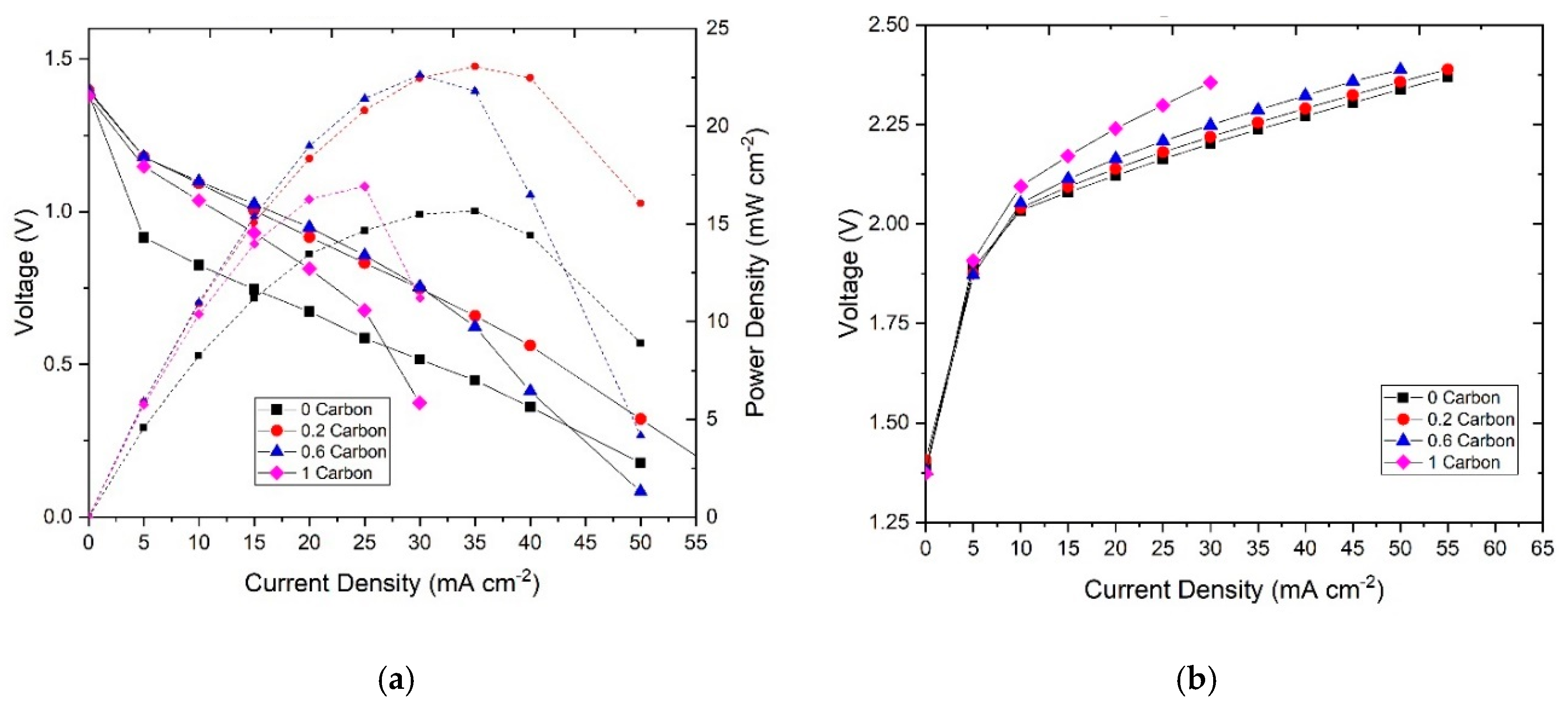
3.2. Polarization Curve Analysis
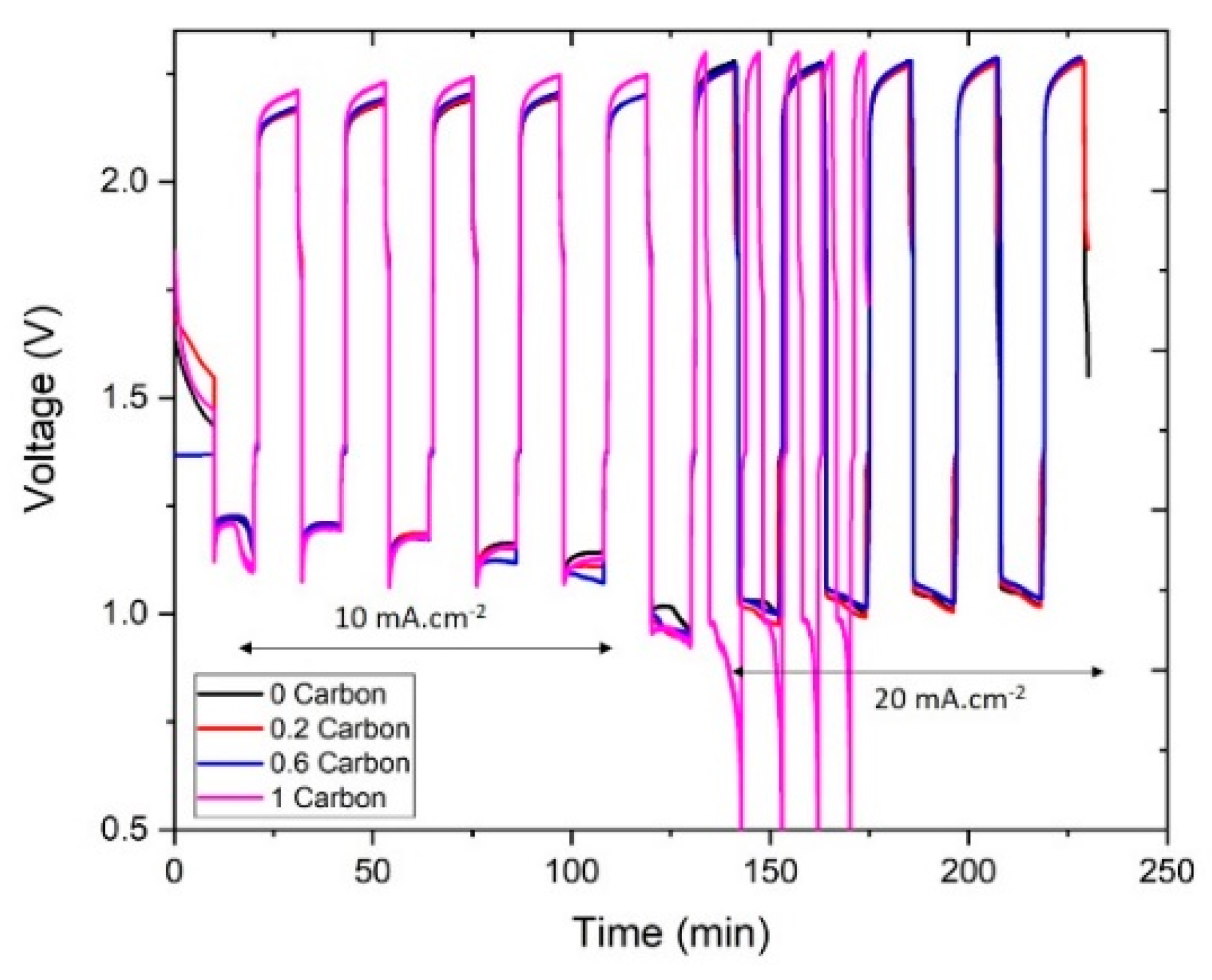
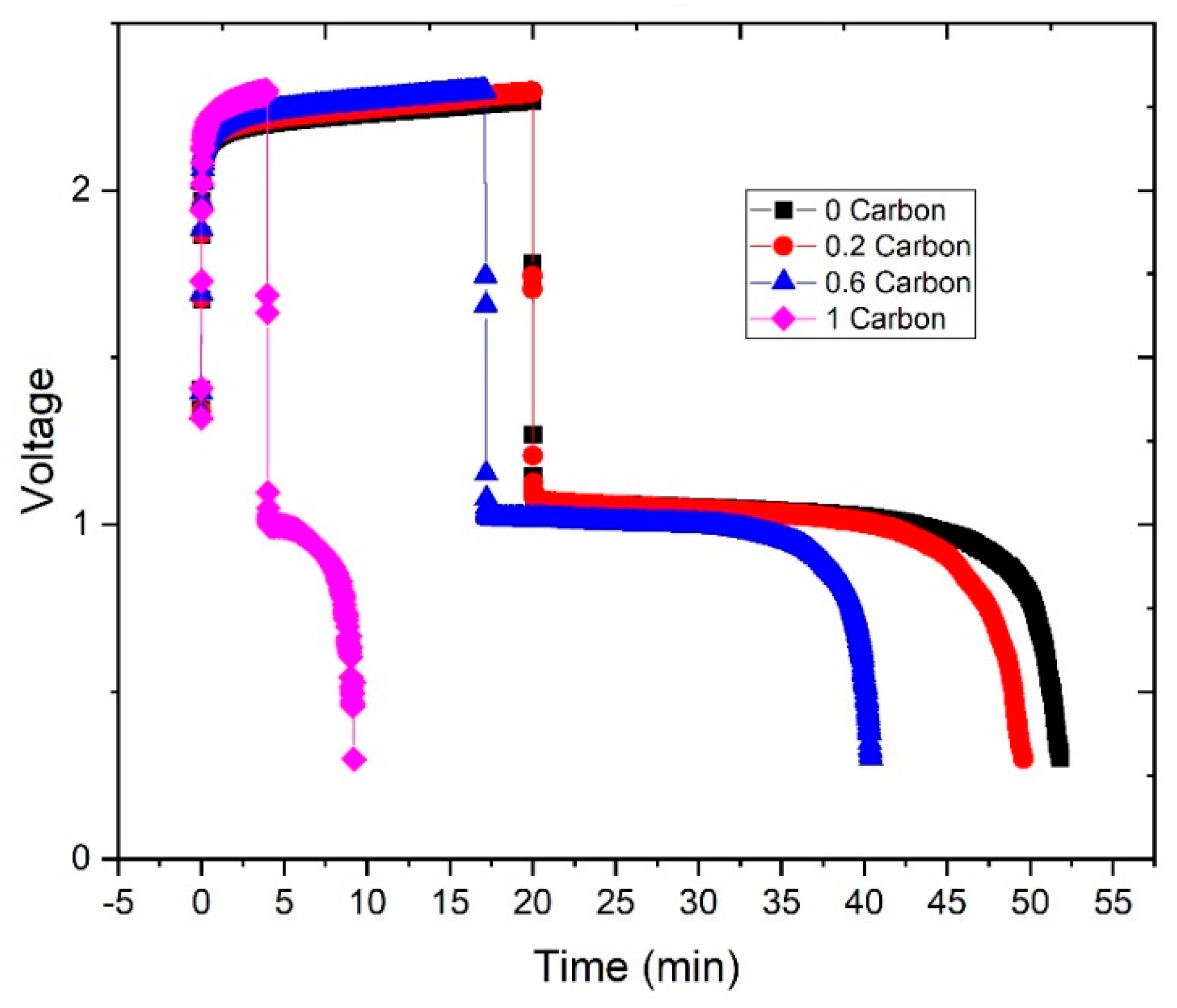
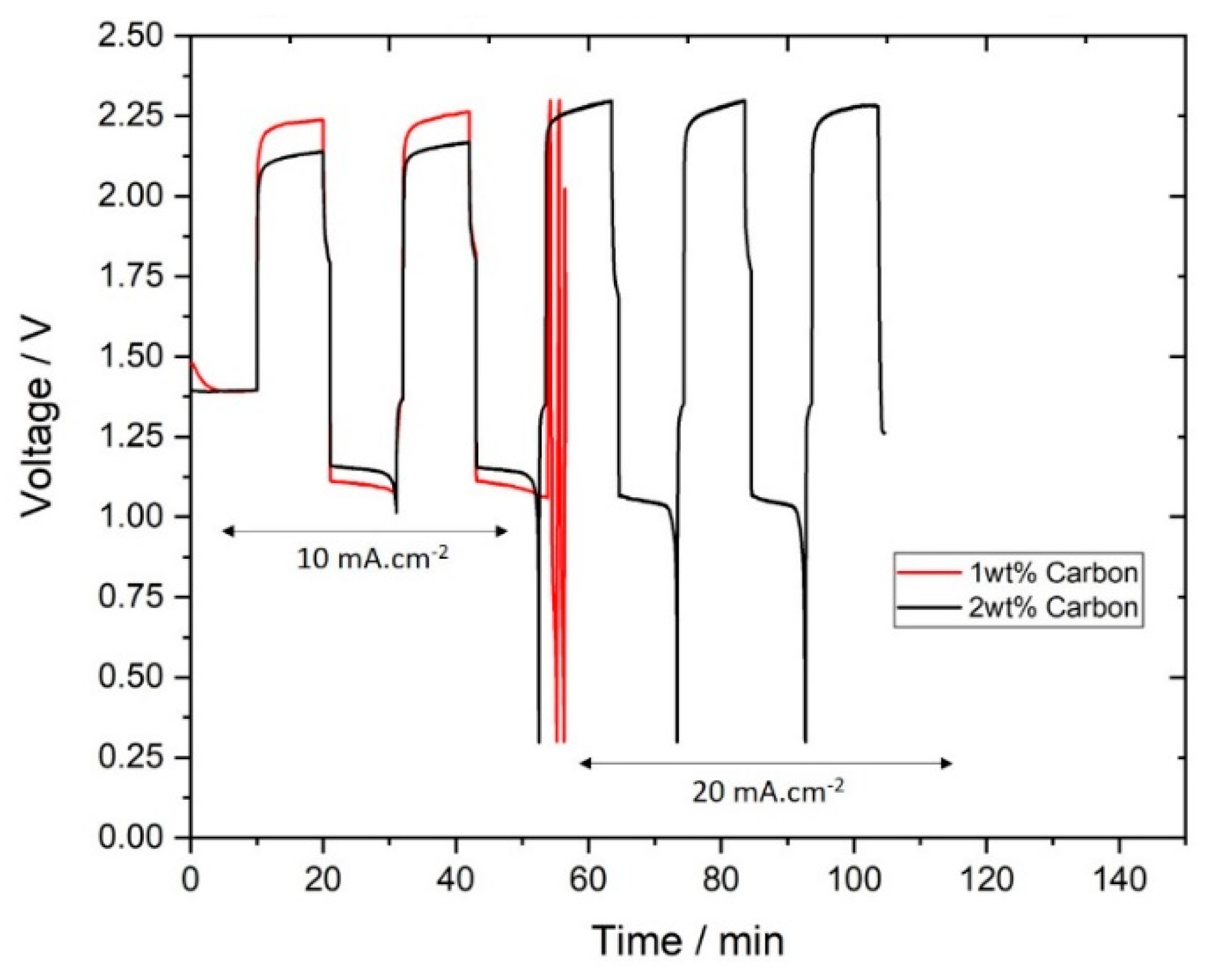
3.3. Cycling Performance
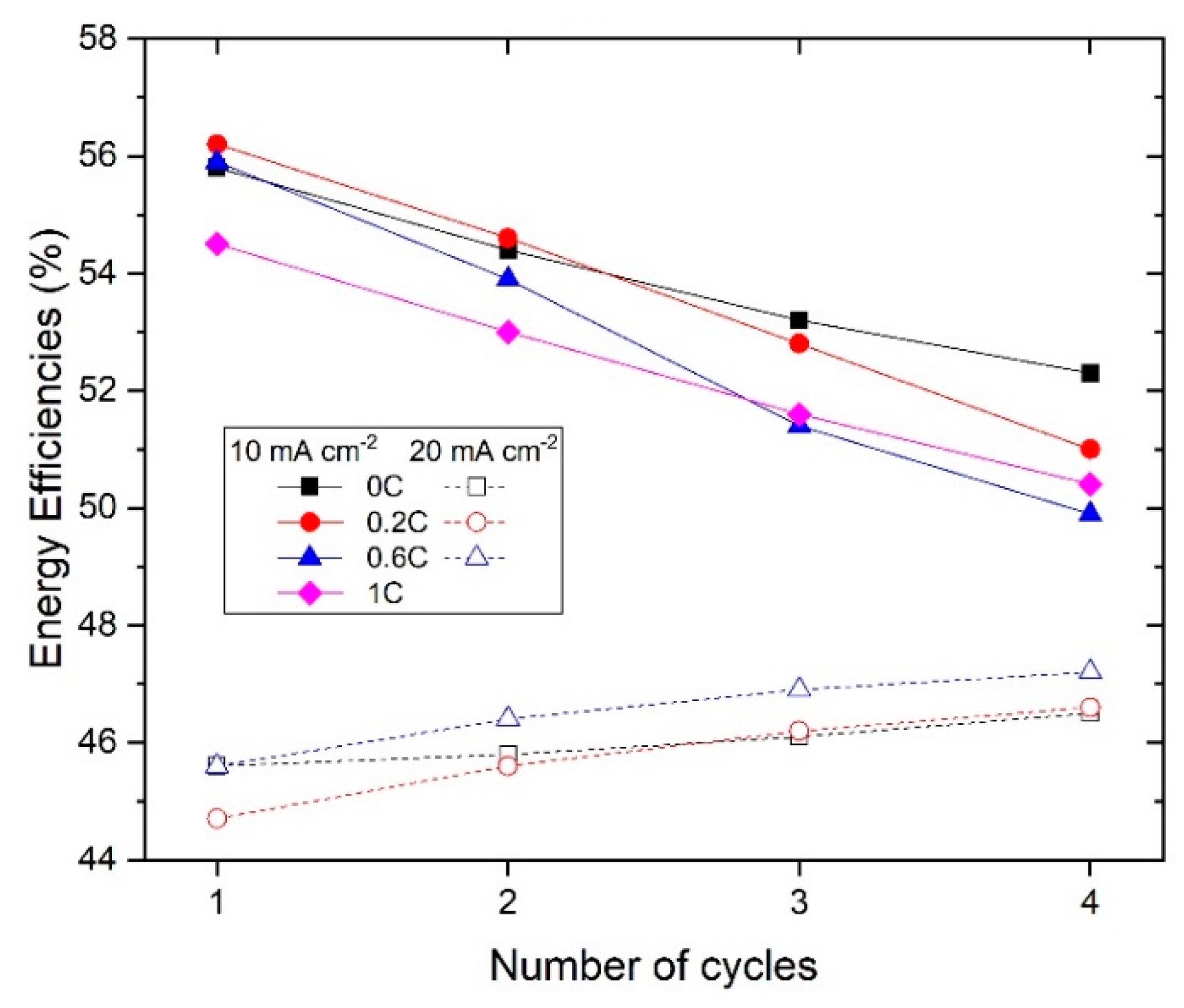
3.4. Estimation of Charging Efficiency
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Sperstad, I.B.; Korpås, M. Energy Storage Scheduling in Distribution Systems Considering Wind and Photovoltaic Generation Uncertainties. Energies 2019, 12, 1231. [Google Scholar] [CrossRef]
- Ding, Y.; Zhang, C.; Zhang, L.; Zhou, Y.; Yu, G. Pathways to Widespread Applications: Development of Redox Flow Batteries Based on New Chemistries. Chem 2019, 5, 1964–1987. [Google Scholar] [CrossRef]
- Caramia, V.; Bozzini, B. Materials science aspects of zinc-air batteries: A review. Mater. Renew. Sustain. Energy 2014, 3, 28. [Google Scholar] [CrossRef]
- Gentil, S.; Reynard, D.; Girault, H.H. Aqueous organic and redox-mediated redox flow batteries: A review. Curr. Opin. Electrochem. 2020, 21, 7–13. [Google Scholar] [CrossRef]
- Choi, N.H.; del Olmo, D.; Fischer, P.; Karsten, P.; Tübke, J. Development of flow fields for Zinc Slurry Air Flow Batteries. Batteries 2020, 6, 15. [Google Scholar] [CrossRef]
- Bockelmann, M.; Kunz, U.; Turek, T. Electrically rechargeable zinc-oxygen flow battery with high power density. Electrochem. Commun. 2016, 69, 24–27. [Google Scholar] [CrossRef]
- Jiratchayamaethasakul, C.; Srijaroenpramong, N.; Bunyangyuen, T.; Arpavate, W.; Wongyao, N.; Therdthianwong, A.; Therdthianwong, S. Effects of anode orientation and flow channel design on performance of refuelable zinc-air fuel cells. J. Appl. Electrochem. 2014, 44, 1205–1218. [Google Scholar] [CrossRef]
- Li, Y.; Gong, M.; Liang, Y.; Feng, J.; Kim, J.-E.; Wang, H.; Hong, G.; Zhang, B.; Dai, H. Advanced zinc-air batteries based on high-performance hybrid electrocatalysts. Nat. Commun. 2013, 4, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Zelger, C.; Süßenbacher, M.; Laskos, A.; Gollas, B. State of charge indicators for alkaline zinc-air redox flow batteries. J. Power Sources 2019, 424, 76–81. [Google Scholar] [CrossRef]
- Wang, K.; Yu, J. Lifetime simulation of rechargeable zinc-air battery based on electrode aging. J. Energy Storage 2020, 28, 101191. [Google Scholar] [CrossRef]
- Mainar, A.; Iruin, E.; Colmenares, L.; Blázquez, J.; Grande, H.-J. Systematic cycle life assessment of a secondary zinc–air battery as a function of the alkaline electrolyte composition. Energy Sci. Eng. 2018, 6, 174–186. [Google Scholar] [CrossRef]
- Ma, H.; Wang, B.; Fan, Y.; Hong, W. Development and Characterization of an Electrically Rechargeable Zinc-Air Battery Stack. Energies 2014, 7, 6548–6557. [Google Scholar] [CrossRef]
- Fu, J.; Cano, Z.P.; Park, M.G.; Yu, A.; Fowler, M.; Chen, Z. Electrically Rechargeable Zinc-Air Batteries: Progress Challenges and Perspectives. Adv. Mater. 2016, 29, 1604685. [Google Scholar] [CrossRef] [PubMed]
- Aremu, E.O.; Park, D.J.; Ryu, K.S. The effects of anode additives towards suppressing dendrite growth and hydrogen gas evolution reaction in Zn-air secondary batteries. Ionics 2019, 25, 4197–4207. [Google Scholar] [CrossRef]
- Zhao, Z.; Fan, X.; Ding, J.; Hu, W.; Zhong, C.; Lu, J. Challengis in Zinc Electrodes for Alkaline Zinc-Air Batteries: Obstacles to commercialization. ACS Energy Lett. 2019, 4, 2259–2270. [Google Scholar] [CrossRef]
- Wang, K.; Pei, P.; Ma, Z.; Chen, H.; Xu, H.; Chen, D.; Wang, X. Dendrite growth in the recharging process of zinc-air batteries. J. Mater. Chem. A 2015, 3, 22648–22655. [Google Scholar] [CrossRef]
- Liang, S.; Yan, W.; Wu, X.; Zhang, Y.; Zhu, Y.; Wang, H.; Wu, Y. Gel polymer electrolytes for lithium ion batteries: Fabrication, characterization and performance. Solid State Ion. 2018, 318, 2–18. [Google Scholar] [CrossRef]
- Smith, K.C.; Chiang, Y.-M.; Carter, W.C. Maximizing Energetic Efficiency in Flow Batteries Utilizing Non-Newtonian Fluids. J. Electrochem. Soc. 2014, 161, A486–A496. [Google Scholar] [CrossRef]
- Cheng, X.; Pan, J.; Zhao, Y.; Liao, M.; Peng, H. Gel Polymer Electrolytes for Electrochemical Energy Storage. Adv. Energy Mater. 2018, 8, 1702184. [Google Scholar] [CrossRef]
- Zhang, Z.; Zuo, C.; Liu, Z.; Yu, Y.; Zuo, Y.; Song, Y. All-solid-state Al-air batteries with polymer alkaline gel electrolyte. J. Power Sources 2014, 251, 470–475. [Google Scholar] [CrossRef]
- Lee, S.M.; Kim, Y.J.; Eom, S.W.; Choi, N.S.; Kim, K.W.; Cho, S.B. Improvement in self-discharge of Zn anode by applying surface modification for Zn-air batteries with high energy density. J. Power Sources 2013, 227, 177–184. [Google Scholar] [CrossRef]
- Tran, T.N.T.; Chung, H.J.; Ivey, D.G. A study of alkaline gel polymer electrolytes for rechargeable zinc–air batteries. Electrochim. Acta 2019, 327, 135021. [Google Scholar] [CrossRef]
- Piau, J.M. Carbopol gels: Elastoviscoplastic and slippery glasses made of individual swollen sponges. Meso- and macroscopic properties, constitutive equations and scaling laws. J. Nonnewton. Fluid Mech. 2007, 144, 1–29. [Google Scholar] [CrossRef]
- Mainar, A.R.; Leonet, O.; Bengoechea, M.; Boyano, I.; de Meatza, I.; Kvasha, A.; Alberto Blázquez, J. Alkaline aqueous electrolytes for secondary zinc-air batteries: An overview. Int. J. Energy Res. 2016, 40, 1032–1049. [Google Scholar] [CrossRef]
- Othman, R.; Yahaya, A.H.; Arof, A.K. A zinc-air cell employing a porous zinc electrode fabricated from zinc-graphite-natural biodegradable polymer paste. J. Appl. Electrochem. 2002, 32, 1347–1353. [Google Scholar] [CrossRef]
- Masri, M.N.; Mohamad, A.A. Effect of adding carbon black to a porous zinc anode in a zinc-air battery. J. Electrochem. Soc. 2013, 160, A715. [Google Scholar] [CrossRef]
- Tao, H.; Tong, X.; Gan, L.; Zhang, S.; Zhang, X.; Liu, X. Effect of adding various carbon additives to porous zinc anode in rechargeable hybrid aqueous battery. J. Alloys Compd. 2016, 658, 119–124. [Google Scholar] [CrossRef]
- Chotipanich, J.; Arpornwichanop, A.; Yonezawa, T.; Kheawhom, S. Electronic and ionic conductivities enhancement of zinc anode for flexible printed zinc-air battery. Eng. J. 2018, 22, 47–57. [Google Scholar] [CrossRef]
- Duduta, M.; Ho, B.; Wood, V.C.; Limthongkul, P.; Brunini, V.E.; Carter, W.C.; Chiang, Y.-M. Semi-Solid Lithium Rechargeable Flow Battery. Adv. Energy Mater. 2011, 1, 511–516. [Google Scholar] [CrossRef]
- Youssry, M.; Madec, L.; Soudan, P.; Cerbelaud, M.; Guyomard, D.; Lestriez, B. Non-aqueous carbon black suspensions for lithium-based redox flow batteries: Rheology and simultaneous rheo-electrical behavior. Phys. Chem. Chem. Phys. 2013, 15, 14476–14486. [Google Scholar] [CrossRef]
- Youssry, M.; Madec, L.; Soudan, P.; Cerbelaud, M.; Guyomard, D.; Lestriez, B. Formulation of flowable anolyte for redox flow batteries: Rheo-electrical study. J. Power Sources 2015, 274, 424–431. [Google Scholar] [CrossRef]
- Shukla, G.; del Olmo Diaz, D.; Thangavel, V.; Franco, A.A. Self-Organization of Electroactive Suspensions in Discharging Slurry Batteries: A Mesoscale Modeling Investigation. ACS Appl. Mater. Interfaces 2017, 9, 17882–17889. [Google Scholar] [CrossRef] [PubMed]
- Shukla, G.; Franco, A.A. Handling Complexity of Semi-solid Redox Flow Battery Operation Principles Through Mechanistic Simulations. J. Phys. Chem. 2018, 122, 23867–23877. [Google Scholar]
- Hoyt, N.C.; Wainright, J.S.; Savinell, R.F. Current Density Scaling in Electrochemical Flow Capacitors. J. Electrochem. Soc. 2015, 162, A1102–A1110. [Google Scholar] [CrossRef]
- Akuzum, B.; Singh, P.; Eichfeld, D.A.; Agartan, L.; Uzun, S.; Gogotsi, Y.; Kumbur, E.C. Percolation Characteristics of Conductive Additives for Capacitive Flowable (Semi-Solid) Electrodes. ACS Appl. Mater. Interfaces 2020, 12, 5866–5875. [Google Scholar] [CrossRef]
- Kisdarjono, H.; Lu, Y.; Lee, J.-J.; Evans, D.; Wang, L. Air Cathode Battery Using Zinc Slurry Anode with Carbon Additive. Available online: https://patents.google.com/patent/US20140370401A1/en (accessed on 1 July 2020).
- Hreiz, R.; Adouani, N.; Fünfschilling, D.; Marchal, P.; Pons, M.N. Rheological characterization of raw and anaerobically digested cow slurry. Chem. Eng. Res. Des. 2017, 119, 47–57. [Google Scholar] [CrossRef]
- Hermoso, J.; Jofore, B.D.; Martínez-Boza, F.J.; Gallegos, C. High pressure mixing rheology of drilling fluids. Ind. Eng. Chem. Res. 2012, 51, 14399–14407. [Google Scholar] [CrossRef]
- Owens, C.E.; Hart, A.J.; McKinley, G.H. Improved rheometry of yield stress fluids using bespoke fractal 3D printed vanes. J. Rheol. 2020, 64, 643–662. [Google Scholar] [CrossRef]
- Baravian, C.; Lalante, A.; Parker, A. Vane rheometry with a large, finite gap. Appl. Rheol. 2002, 12, 81–87. [Google Scholar] [CrossRef]
- Helal, A.; Divoux, T.; McKinley, G.H. Simultaneous Rheoelectric Measurements of Strongly Conductive Complex Fluids. Phys. Rev. Appl. 2016, 6, 064004. [Google Scholar] [CrossRef]
- Ito, Y.; Nyce, M.; Plivelich, R.; Klein, M.; Steingart, D.; Banerjee, S. Zinc morphology in zinc–nickel flow assisted batteries and impact on performance. J. Power Sources 2011, 196, 2340–2345. [Google Scholar] [CrossRef]
- Wei, T.S.; Fan, F.Y.; Helal, A.; Smith, K.C.; McKinley, G.H.; Chiang, Y.M.; Lewis, J.A. Biphasic Electrode Suspensions for Li-Ion Semi-solid Flow Cells with High Energy Density, Fast Charge Transport, and Low-Dissipation Flow. Adv. Energy Mater. 2015, 5, 1500535. [Google Scholar] [CrossRef]
- Dinkgreve, M.; Paredes, J.; Denn, M.M.; Bonn, D. On different ways of measuring ‘the’ yield stress. J. Nonnewton. Fluid Mech. 2016, 238, 233–241. [Google Scholar] [CrossRef]
- Gallier, S.; Lemaire, E.; Peters, F.; Lobry, L. Rheology of sheared suspensions of rough frictional particles. J. Fluid Mech. 2014, 757, 514–549. [Google Scholar] [CrossRef]
- Tang, A.; Bao, J.; Skyllas-Kazacos, M. Studies on pressure losses and flow rate optimization in vanadium redox flow battery. J. Power Sources 2014, 248, 154–162. [Google Scholar] [CrossRef]
- Petek, T.J.; Hoyt, N.C.; Savinell, R.F.; Wainright, J.S. Characterizing Slurry Electrodes Using Electrochemical Impedance Spectroscopy. J. Electrochem. Soc. 2015, 163, A5001–A5009. [Google Scholar] [CrossRef]



| Mass Fraction (wt.%) | |||||
|---|---|---|---|---|---|
| Zinc | ZnO | Carbopol | Carbon Black | KOH + Water | |
| 0 Carbon (0%CB) | 33.8 | 4 | 0.7 | 0 | 61.5 |
| 0.2 Carbon (0.2%CB) | 0.2 | 61.3 | |||
| 0.6 Carbon (0.6%CB) | 0.6 | 60.9 | |||
| 1 Carbon (1%CB) | 1 | 60.5 | |||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Choi, N.H.; del Olmo, D.; Milian, D.; El Kissi, N.; Fischer, P.; Pinkwart, K.; Tübke, J. Use of Carbon Additives towards Rechargeable Zinc Slurry Air Flow Batteries. Energies 2020, 13, 4482. https://doi.org/10.3390/en13174482
Choi NH, del Olmo D, Milian D, El Kissi N, Fischer P, Pinkwart K, Tübke J. Use of Carbon Additives towards Rechargeable Zinc Slurry Air Flow Batteries. Energies. 2020; 13(17):4482. https://doi.org/10.3390/en13174482
Chicago/Turabian StyleChoi, Nak Heon, Diego del Olmo, Diego Milian, Nadia El Kissi, Peter Fischer, Karsten Pinkwart, and Jens Tübke. 2020. "Use of Carbon Additives towards Rechargeable Zinc Slurry Air Flow Batteries" Energies 13, no. 17: 4482. https://doi.org/10.3390/en13174482
APA StyleChoi, N. H., del Olmo, D., Milian, D., El Kissi, N., Fischer, P., Pinkwart, K., & Tübke, J. (2020). Use of Carbon Additives towards Rechargeable Zinc Slurry Air Flow Batteries. Energies, 13(17), 4482. https://doi.org/10.3390/en13174482






