Electrochemical Evaluation of Surface Modified Free-Standing CNT Electrode for Li–O2 Battery Cathode
Abstract
:1. Introduction
2. Materials and Methods
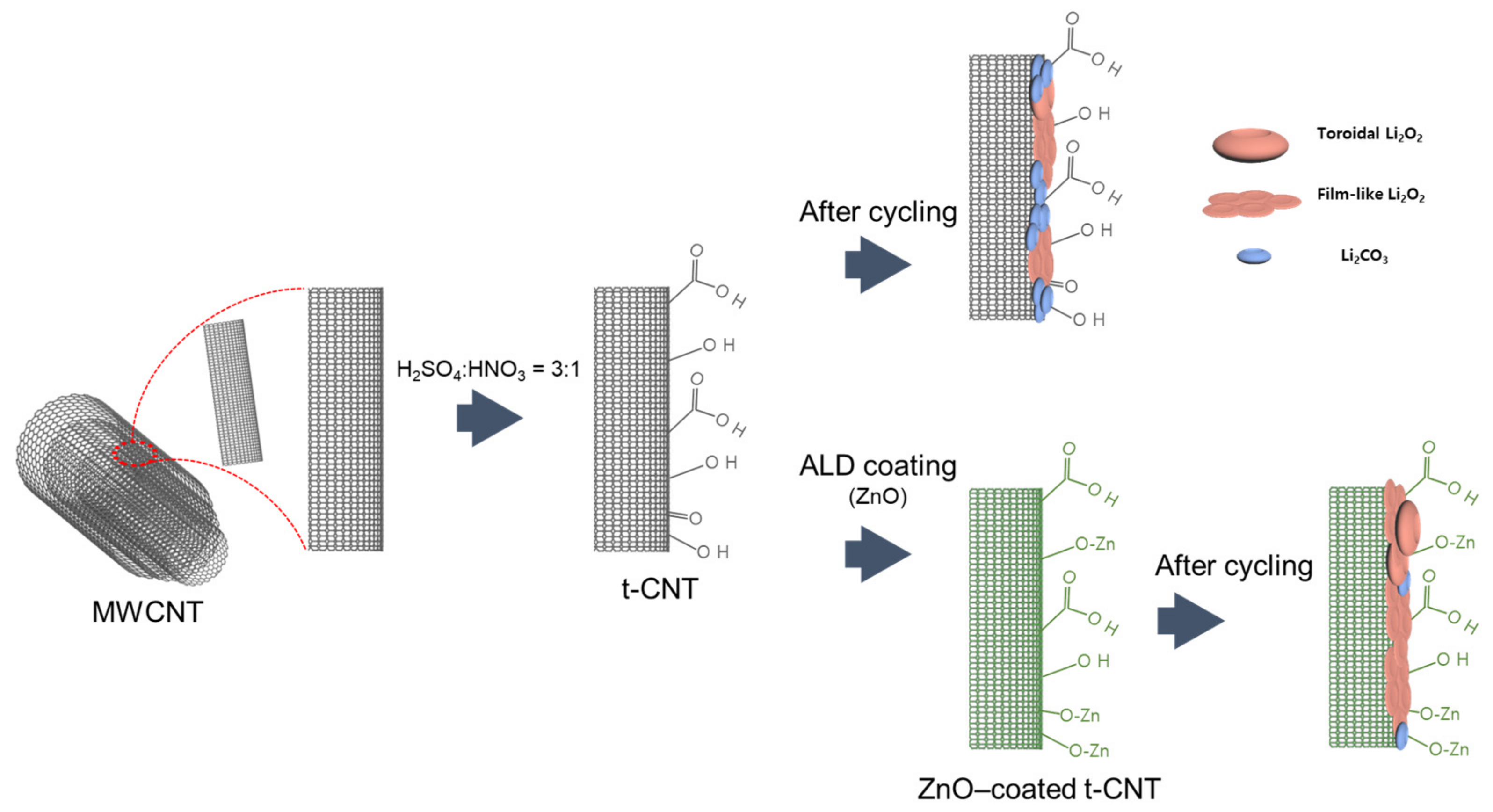
2.1. Preparation of CNT Electrode with Controlled Oxygen Functional Groups
2.2. Characterization of CNTs with Controlled Oxygen Functional Groups
2.3. Electrochemical Characterization
2.4. Iodometric Titration
3. Results
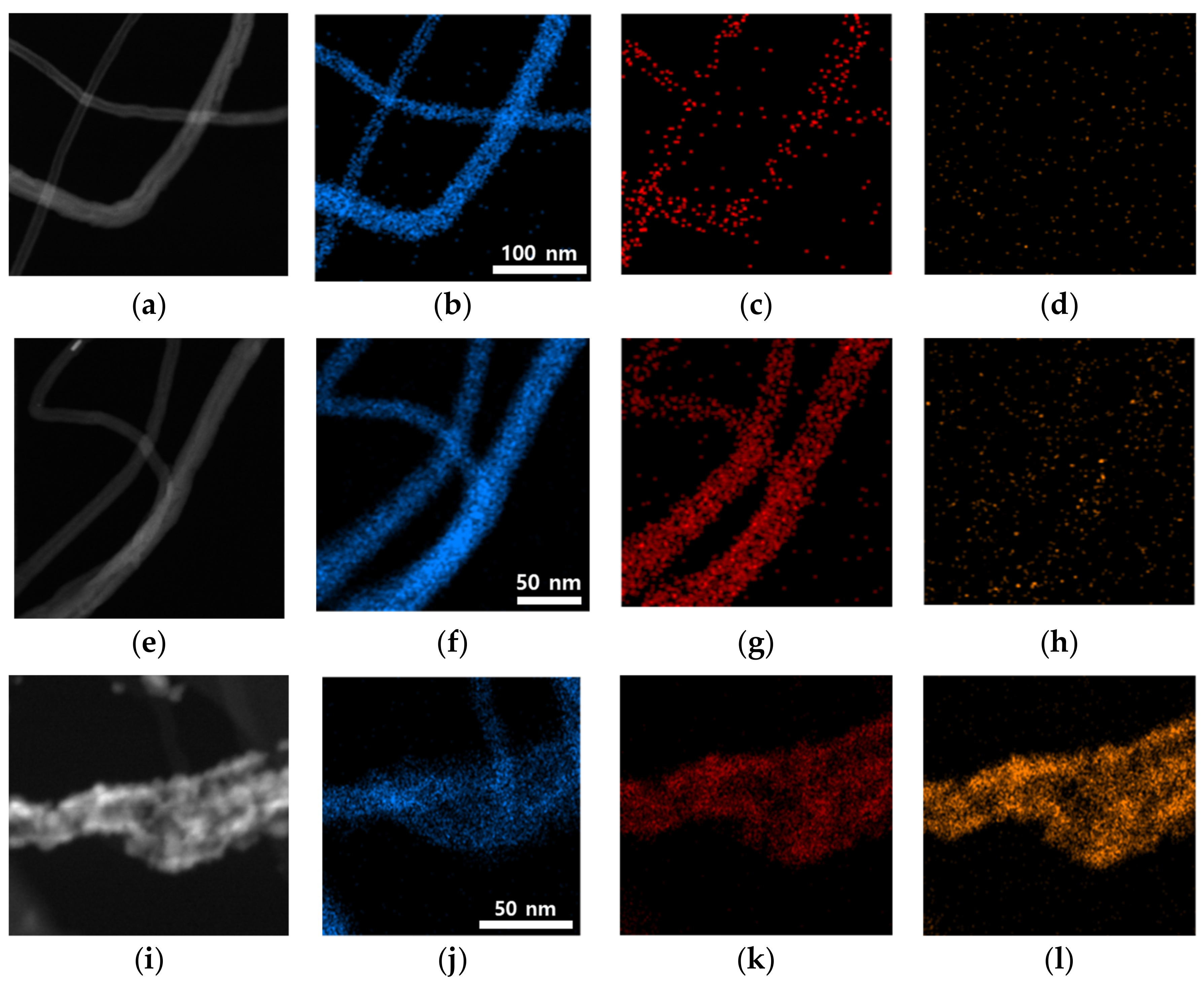
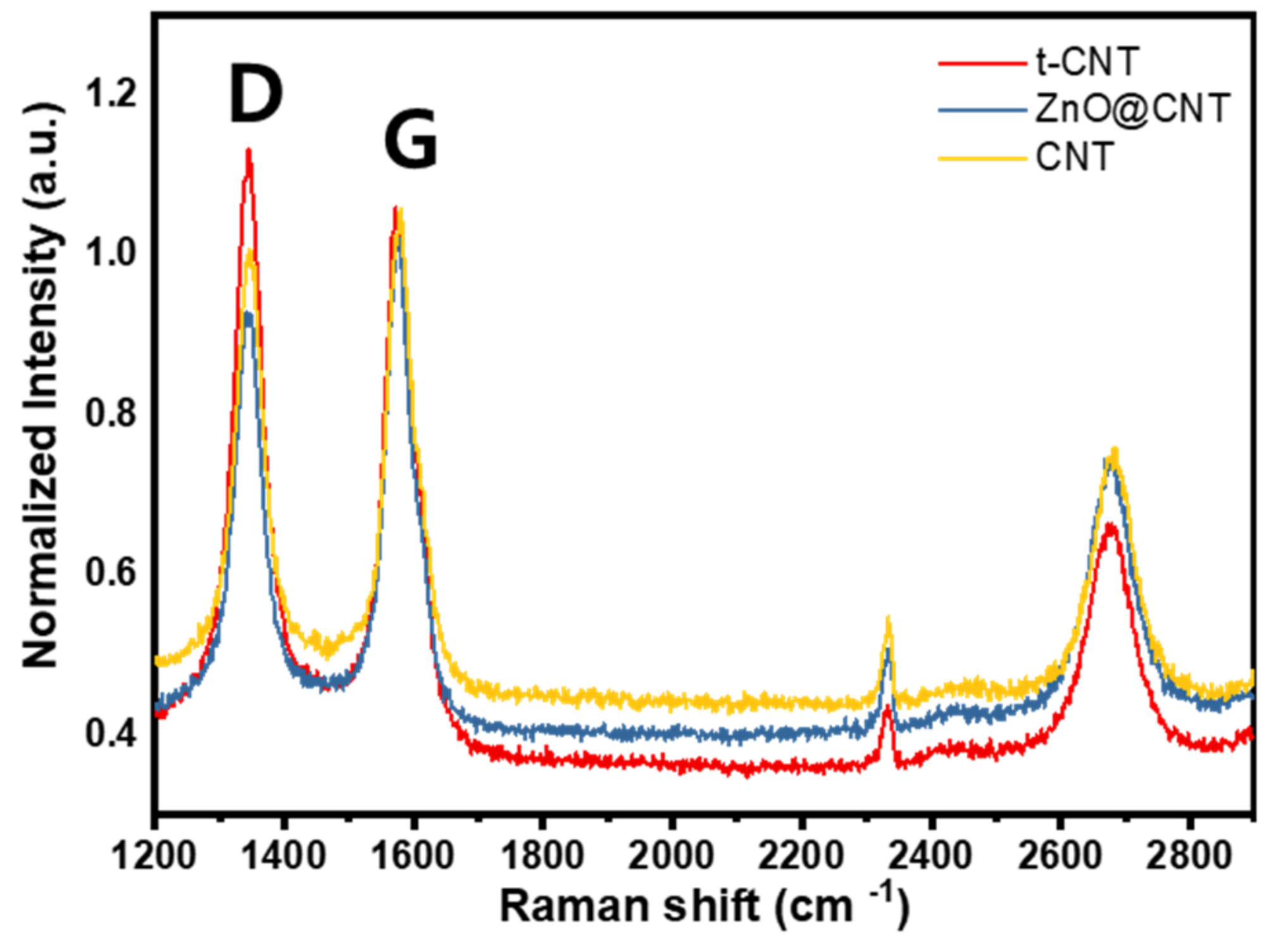
3.1. Material Characterization of Prepared CNT Materials
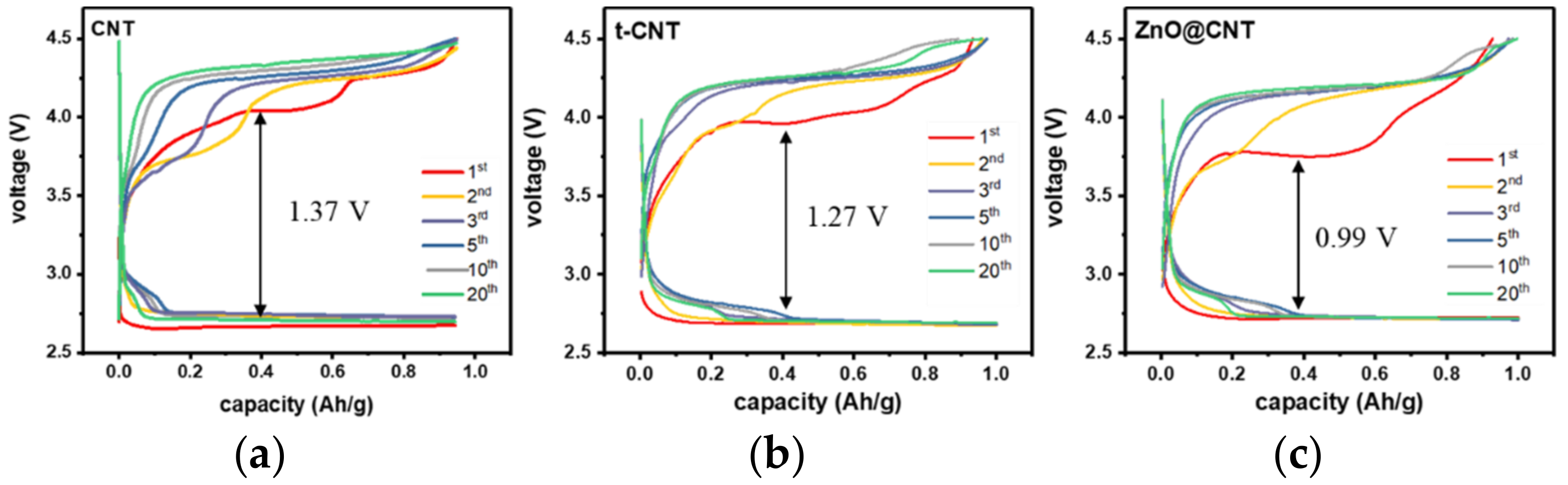
3.2. Electrochemical Properties of Prepared CNT Electrodes
3.3. The Yield Calculation for Li2O2 Formation by Iodine-Metric Titration
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Park, M.; Sun, H.; Lee, H.; Lee, J.; Cho, J. Lithium-air batteries: Survey on the current status and perspectives towards automotive applications from a battery industry standpoint. Adv. Energy Mater. 2012, 2, 780–800. [Google Scholar] [CrossRef]
- Girishkumar, G.; McCloskey, B.; Luntz, A.C.; Swanson, S.; Wilcke, W. Lithium−air battery: Promise and challenges. J. Phys. Chem. Lett. 2010, 1, 2193–2203. [Google Scholar] [CrossRef]
- Christensen, J.; Albertus, P.; Sanchez-Carrera, R.S.; Lohmann, T.; Kozinsky, B.; Liedtke, R.; Ahmed, J.; Kojic, A. A critical review of Li/air batteries. J. Electrochem. Soc. 2011, 159, R1. [Google Scholar] [CrossRef]
- Lim, H.D.; Lee, B.; Bae, Y.; Park, H.; Ko, Y.; Kim, H.; Kim, J.; Kang, K. Reaction chemistry in rechargeable Li-O2 batteries. Chem. Soc. Rev. 2017, 46, 2873–2888. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Geng, D.; Ding, N.; Hor, T.A.; Chien, S.W.; Liu, Z.; Wuu, D.; Sun, X.; Zong, Y. From Lithium-Oxygen to Lithium-Air Batteries: Challenges and Opportunities. Adv. Energy Mater. 2016, 6, 1502164. [Google Scholar] [CrossRef]
- Bruce, P.G.; Freunberger, S.A.; Hardwick, L.J.; Tarascon, J.-M. Li-O2 and Li-S batteries with high energy storage. Nat. Mater. 2012, 11, 19. [Google Scholar] [CrossRef] [PubMed]
- Bryantsev, V.S.; Uddin, J.; Giordani, V.; Walker, W.; Addison, D.; Chase, G.V. The identification of stable solvents for nonaqueous rechargeable Li-air batteries. J. Electrochem. Soc. 2012, 160, A160. [Google Scholar] [CrossRef]
- Freunberger, S.A.; Chen, Y.; Peng, Z.; Griffin, J.M.; Hardwick, L.J.; Bardé, F.; Novák, P.; Bruce, P.G. Reactions in the rechargeable lithium-O2 battery with alkyl carbonate electrolytes. J. Am. Chem. Soc. 2011, 133, 8040–8047. [Google Scholar] [CrossRef]
- McCloskey, B.; Speidel, A.; Scheffler, R.; Miller, D.; Viswanathan, V.; Hummelshøj, J.; Nørskov, J.; Luntz, A. Twin problems of interfacial carbonate formation in nonaqueous Li-O2 batteries. J. Phys. Chem. Lett. 2012, 3, 997–1001. [Google Scholar] [CrossRef] [PubMed]
- Ogasawara, T.; Débart, A.; Holzapfel, M.; Novák, P.; Bruce, P.G. Rechargeable Li2O2 electrode for lithium batteries. J. Am. Chem. Soc. 2006, 128, 1390–1393. [Google Scholar] [CrossRef] [PubMed]
- Read, J. Characterization of the lithium/oxygen organic electrolyte battery. J. Electrochem. Soc. 2002, 149, A1190. [Google Scholar] [CrossRef]
- Abraham, K.; Jiang, Z. A polymer electrolyte-based rechargeable lithium/oxygen battery. J. Electrochem. Soc. 1996, 143, 1. [Google Scholar] [CrossRef]
- Itkis, D.M.; Semenenko, D.A.; Kataev, E.Y.; Belova, A.I.; Neudachina, V.S.; Sirotina, A.P.; Hävecker, M.; Teschner, D.; Knop-Gericke, A.; Dudin, P. Reactivity of carbon in lithium–oxygen battery positive electrodes. Nano Lett. 2013, 13, 4697–4701. [Google Scholar] [CrossRef] [PubMed]
- Aetukuri, N.B.; McCloskey, B.D.; García, J.M.; Krupp, L.E.; Viswanathan, V.; Luntz, A.C. Solvating additives drive solution-mediated electrochemistry and enhance toroid growth in non-aqueous Li-O2 batteries. Nat. Chem. 2015, 7, 50–56. [Google Scholar] [CrossRef]
- Oh, D.; Qi, J.; Lu, Y.-C.; Zhang, Y.; Shao-Horn, Y.; Belcher, A.M. Biologically enhanced cathode design for improved capacity and cycle life for lithium-oxygen batteries. Nat. Commun. 2013, 4, 2756. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, Q.; Cao, R.; Cho, J.; Wu, G. Nanostructured carbon-based cathode catalysts for nonaqueous lithium–oxygen batteries. Phys. Chem. Chem. Phys. 2014, 16, 13568–13582. [Google Scholar] [CrossRef] [PubMed]
- Ottakam Thotiyl, M.M.; Freunberger, S.A.; Peng, Z.; Bruce, P.G. The carbon electrode in nonaqueous Li-O2 cells. J. Am. Chem. Soc. 2013, 135, 494–500. [Google Scholar] [CrossRef]
- Andrieux, C.P.; Hapiot, P.; Saveant, J.M. Mechanism of superoxide ion disproportionation in aprotic solvents. J. Am. Chem. Soc. 1987, 109, 3768–3775. [Google Scholar] [CrossRef]
- Frimer, A.A.; Farkash-Solomon, T.; Aljadeff, G. Mechanism of the superoxide anion radical (O2-) mediated oxidation of diarylmethanes. J. Org. Chem. 1986, 51, 2093–2098. [Google Scholar] [CrossRef]
- McCloskey, B.D.; Bethune, D.; Shelby, R.; Mori, T.; Scheffler, R.; Speidel, A.; Sherwood, M.; Luntz, A. Limitations in rechargeability of Li-O2 batteries and possible origins. J. Phys. Chem. Lett. 2012, 3, 3043–3047. [Google Scholar] [CrossRef]
- Luo, N.; Ji, G.-J.; Wang, H.-F.; Li, F.; Liu, Q.-C.; Xu, J.-J. Process for a Free-Standing and Stable All-Metal Structure for Symmetrical Lithium–Oxygen Batteries. ACS Nano 2020, 14, 3281–3289. [Google Scholar] [CrossRef]
- Peng, Z.; Freunberger, S.A.; Chen, Y.; Bruce, P.G. A reversible and higher-rate Li-O2 battery. Science 2012, 337, 563–566. [Google Scholar] [CrossRef]
- Xia, G.; Shen, S.; Zhu, F.; Xie, J.; Hu, Y.; Zhu, K.; Zhang, J. Effect of oxygen-containing functional groups of carbon materials on the performance of Li-O2 batteries. Electrochem. Commun. 2015, 60, 26–29. [Google Scholar] [CrossRef]
- Lee, M.; Yoo, Y.; Kwak, J.H.; Yun, Y.S.; Jung, H.-G.; Byun, D.; Oh, S.H.; Lim, H.-D. Effect of surface characteristics of carbon host on electrochemical performance of nonaqueous Li-O2 batteries. Chem. Eng. J. 2021, 412, 128549. [Google Scholar] [CrossRef]
- Débart, A.; Paterson, A.J.; Bao, J.; Bruce, P.G. α-MnO2 nanowires: A catalyst for the O2 electrode in rechargeable lithium batteries. Angew. Chem. Int. Ed. 2008, 47, 4521–4524. [Google Scholar] [CrossRef]
- De Menezes, B.; Ferreira, F.; Silva, B.; Simonetti, E.; Bastos, T.; Cividanes, L.; Thim, G. Effects of octadecylamine functionalization of carbon nanotubes on dispersion, polarity, and mechanical properties of CNT/HDPE nanocomposites. J. Mater. Sci. 2018, 53, 14311–14327. [Google Scholar] [CrossRef]
- Xiao, J.; Mei, D.; Li, X.; Xu, W.; Wang, D.; Graff, G.L.; Bennett, W.D.; Nie, Z.; Saraf, L.V.; Aksay, I.A. Hierarchically porous graphene as a lithium–air battery electrode. Nano Lett. 2011, 11, 5071–5078. [Google Scholar] [CrossRef]
- McCloskey, B.D.; Valery, A.; Luntz, A.C.; Gowda, S.R.; Wallraff, G.M.; Garcia, J.M.; Mori, T.; Krupp, L.E. Combining accurate O2 and Li2O2 assays to separate discharge and charge stability limitations in nonaqueous Li-O2 batteries. J. Phys. Chem. Lett. 2013, 4, 2989–2993. [Google Scholar] [CrossRef]
- Ferreira, F.; Francisco, W.; Cividanes, L.; Coutinho, A.; Thim, G. Dodecylamine Functionalization of Cnt: Thermal Stability And Dispersion. Blucher Chem. Eng. Proc. 2015, 1, 2421–2426. [Google Scholar]
- Lu, Y.-C.; Gasteiger, H.A.; Shao-Horn, Y. Catalytic activity trends of oxygen reduction reaction for nonaqueous Li-air batteries. J. Am. Chem. Soc. 2011, 133, 19048–19051. [Google Scholar] [CrossRef]
- Yang, C.; Wong, R.A.; Hong, M.; Yamanaka, K.; Ohta, T.; Byon, H.R. Unexpected Li2O2 film growth on carbon nanotube electrodes with CeO2 nanoparticles in Li-O2 batteries. Nano Lett. 2016, 16, 2969–2974. [Google Scholar] [CrossRef]
- Biniak, S.; Szymański, G.; Siedlewski, J.; Świątkowski, A. The characterization of activated carbons with oxygen and nitrogen surface groups. Carbon 1997, 35, 1799–1810. [Google Scholar] [CrossRef]
- Largani, S.H.; Pasha, M.A. The effect of concentration ratio and type of functional group on synthesis of CNT–ZnO hybrid nanomaterial by an in situ sol–gel process. Int. Nano Lett. 2017, 7, 25–33. [Google Scholar] [CrossRef] [Green Version]
- Sadri, R.; Hosseini, M.; Kazi, S.; Bagheri, S.; Zubir, N.; Solangi, K.; Zaharinie, T.; Badarudin, A. A bio-based, facile approach for the preparation of covalently functionalized carbon nanotubes aqueous suspensions and their potential as heat transfer fluids. J. Colloid Interface Sci. 2017, 504, 115–123. [Google Scholar] [CrossRef]
- Sadri, R.; Hosseini, M.; Kazi, S.; Bagheri, S.; Zubir, N.; Ahmadi, G.; Dahari, M.; Zaharinie, T. A novel, eco-friendly technique for covalent functionalization of graphene nanoplatelets and the potential of their nanofluids for heat transfer applications. Chem. Phys. Lett. 2017, 675, 92–97. [Google Scholar] [CrossRef]
- Lu, J.; Lei, Y.; Lau, K.C.; Luo, X.; Du, P.; Wen, J.; Assary, R.S.; Das, U.; Miller, D.J.; Elam, J.W. A nanostructured cathode architecture for low charge overpotential in lithium-oxygen batteries. Nat. Commun. 2013, 4, 2383. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ferrari, A.C.; Meyer, J.; Scardaci, V.; Casiraghi, C.; Lazzeri, M.; Mauri, F.; Piscanec, S.; Jiang, D.; Novoselov, K.; Roth, S. Raman spectrum of graphene and graphene layers. Phys. Rev. Lett. 2006, 97, 187401. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Das, A.; Pisana, S.; Chakraborty, B.; Piscanec, S.; Saha, S.K.; Waghmare, U.V.; Novoselov, K.S.; Krishnamurthy, H.R.; Geim, A.K.; Ferrari, A.C. Monitoring dopants by Raman scattering in an electrochemically top-gated graphene transistor. Nat. Nanotechnol. 2008, 3, 210–215. [Google Scholar] [CrossRef] [Green Version]
- Deboever, J. Characterization of Lithium Peroxide Formation in Lithium Air Battery Electrode Via Titration Techniques and EIS; Oregon Institute of Technology: Klamath Falls, OR, USA, 2014. [Google Scholar]
- Jia, H.; Li, X.; Song, J.; Zhang, X.; Luo, L.; He, Y.; Li, B.; Cai, Y.; Hu, S.; Xiao, X. Hierarchical porous silicon structures with extraordinary mechanical strength as high-performance lithium-ion battery anodes. Nat. Commun. 2020, 11, 1474. [Google Scholar] [CrossRef] [Green Version]
- Kim, K.i.; Guo, Q.; Tang, L.; Zhu, L.; Pan, C.; Chang, C.h.; Razink, J.; Lerner, M.M.; Fang, C.; Ji, X. Reversible Insertion of Mg-Cl Superhalides in Graphite as a Cathode for Aqueous Dual-Ion Batteries. Angew. Chem. 2020, 132, 20096–20100. [Google Scholar] [CrossRef]
- Lim, H.-D.; Park, K.-Y.; Gwon, H.; Hong, J.; Kim, H.; Kang, K. The potential for long-term operation of a lithium–oxygen battery using a non-carbonate-based electrolyte. Chem. Commun. 2012, 48, 8374–8376. [Google Scholar] [CrossRef]
- Elizabeth, I.; Mathur, R.; Maheshwari, P.; Singh, B.; Gopukumar, S. Development of SnO2/multiwalled carbon nanotube paper as free standing anode for lithium ion batteries (LIB). Electrochim. Acta 2015, 176, 735–742. [Google Scholar] [CrossRef]
- Yue, L.; Zhong, H.; Zhang, L. Enhanced reversible lithium storage in a nano-Si/MWCNT free-standing paper electrode prepared by a simple filtration and post sintering process. Electrochim. Acta 2012, 76, 326–332. [Google Scholar] [CrossRef]

| 0.1 mA cm−2 | ZnO@CNT | t-CNT | CNT |
|---|---|---|---|
| 1 | 53.93% | 45.56% | 41.36% |
| 2 | 53.26% | 47.86% | 45.67% |
| 3 | 54.38% | 49.88% | 44.38% |
| Average | 53.86% | 47.81% | 43.80% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, J.H.; Jung, H.W.; Kim, I.S.; Park, M.; Kim, H.-S. Electrochemical Evaluation of Surface Modified Free-Standing CNT Electrode for Li–O2 Battery Cathode. Energies 2021, 14, 4196. https://doi.org/10.3390/en14144196
Lee JH, Jung HW, Kim IS, Park M, Kim H-S. Electrochemical Evaluation of Surface Modified Free-Standing CNT Electrode for Li–O2 Battery Cathode. Energies. 2021; 14(14):4196. https://doi.org/10.3390/en14144196
Chicago/Turabian StyleLee, Ji Hyeon, Hyun Wook Jung, In Soo Kim, Min Park, and Hyung-Seok Kim. 2021. "Electrochemical Evaluation of Surface Modified Free-Standing CNT Electrode for Li–O2 Battery Cathode" Energies 14, no. 14: 4196. https://doi.org/10.3390/en14144196
APA StyleLee, J. H., Jung, H. W., Kim, I. S., Park, M., & Kim, H.-S. (2021). Electrochemical Evaluation of Surface Modified Free-Standing CNT Electrode for Li–O2 Battery Cathode. Energies, 14(14), 4196. https://doi.org/10.3390/en14144196










