Absorption of Cu(II) and Zn(II) from Aqueous Solutions onto Biochars Derived from Apple Tree Branches
Abstract
:1. Introduction
2. Materials and Methods
2.1. Biochar Preparation
2.2. Batch Adsorption Experiments
2.2.1. Chemical Reagents
2.2.2. Adsorption Experiments
2.3. Mathematical Models
2.3.1. Kinetics Test
2.3.2. Adsorption Isotherm
2.3.3. Thermodynamics Study
2.4. Statistical Analysis
3. Results and Discussion
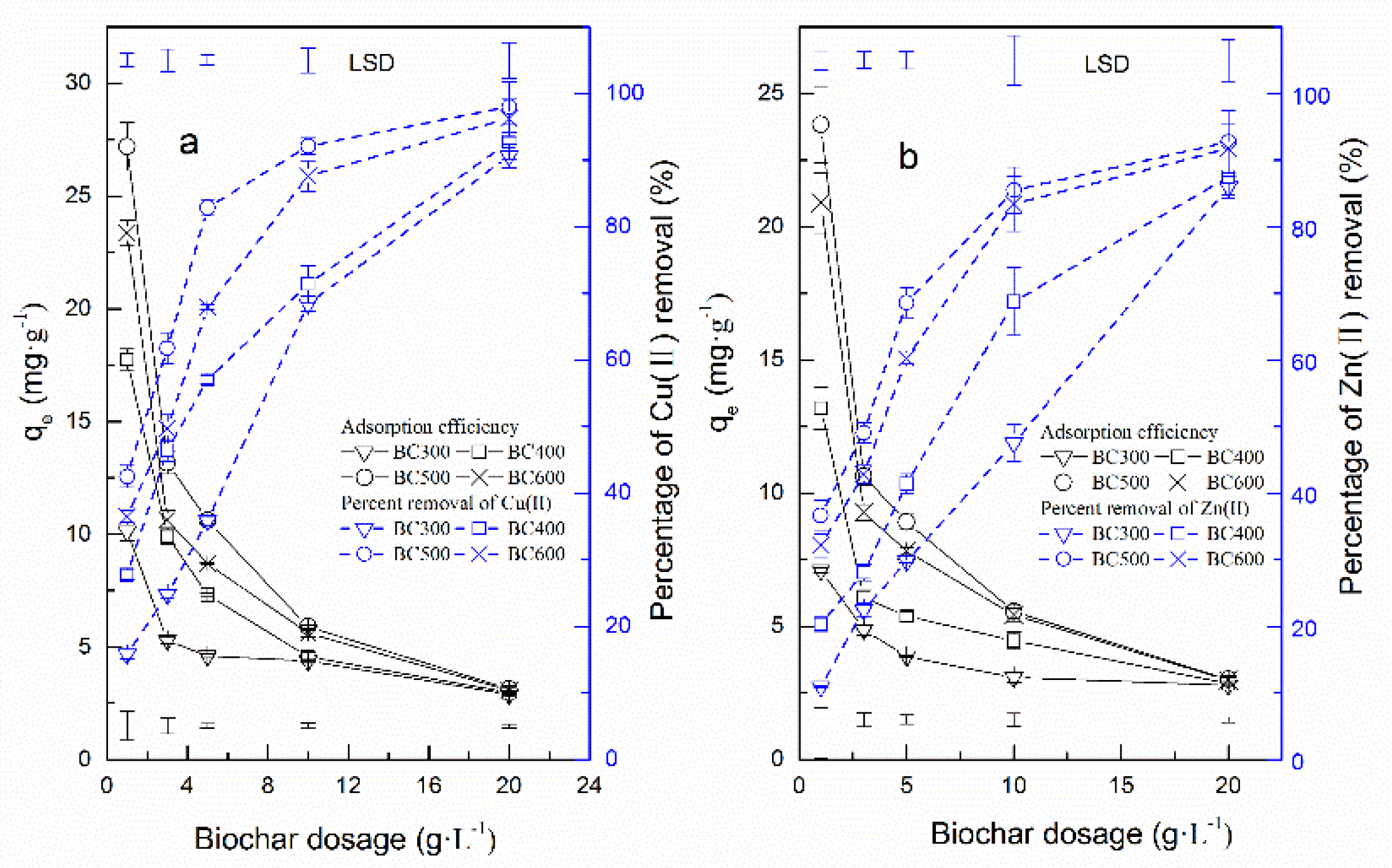
3.1. Influence of BC Dosage
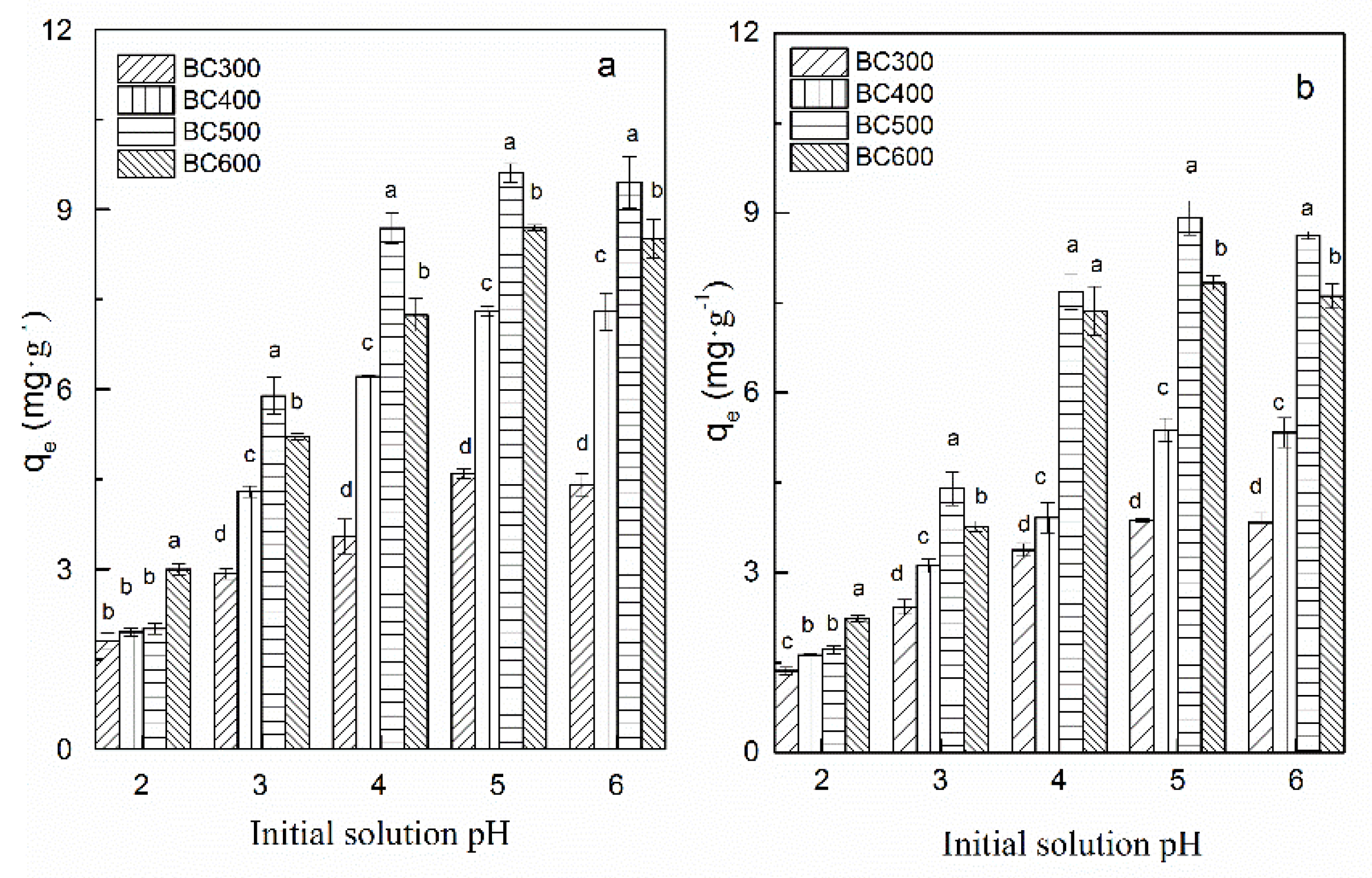
3.2. Influence of Solution pH
3.3. Influence of Contact Time and Adsorption Kinetics
3.4. Adsorption Isotherm Studies
3.5. Influence of Temperature and Thermodynamics Study
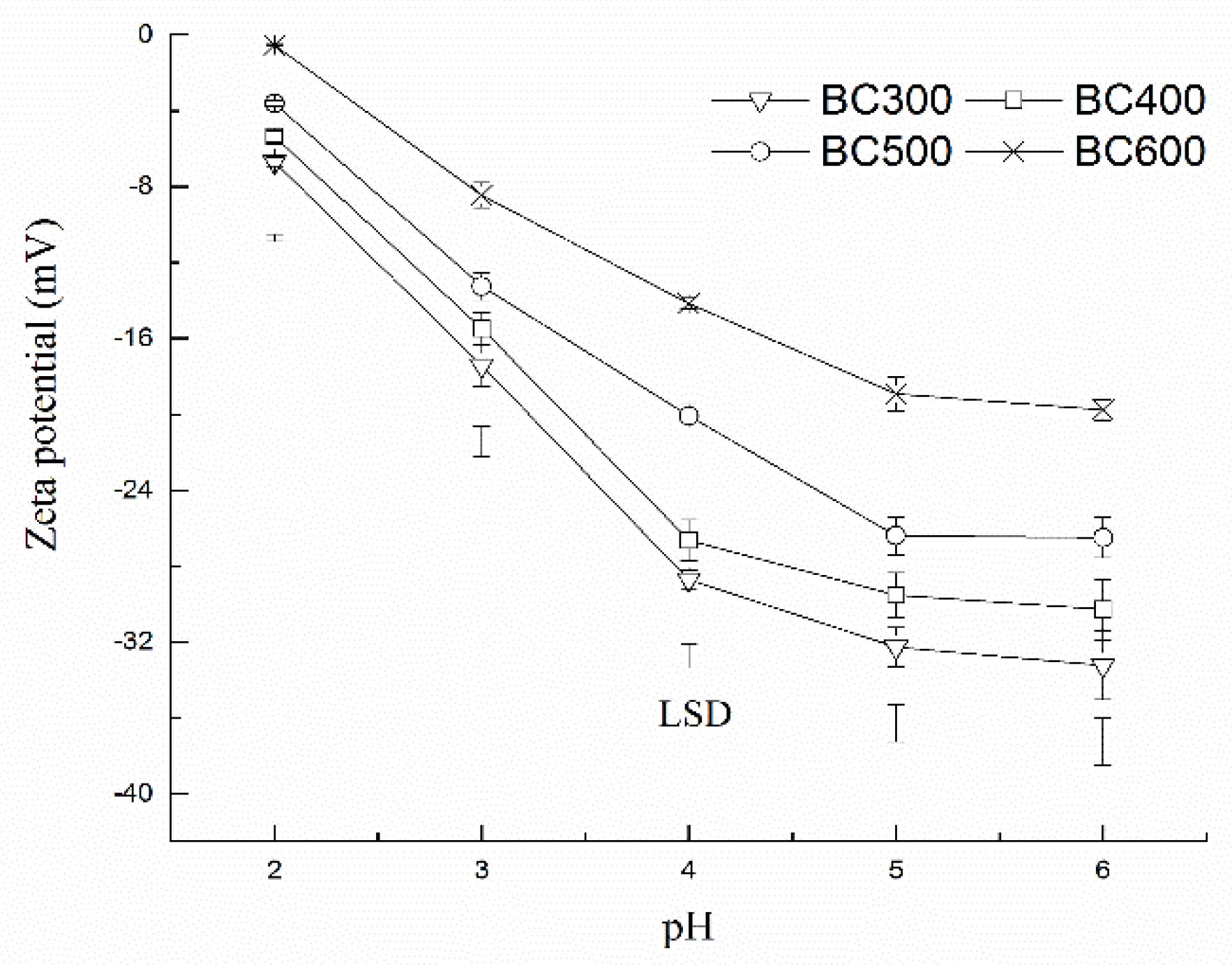
3.6. Mechanism of Absorption
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Li, M.; Liu, Q.; Guo, L.; Zhang, Y.; Lou, Z.; Wang, Y.; Qian, G. Cu(II) removal from aqueous solution by Spartina alterniflora derived biochar. Bioresour. Technol. 2013, 141, 83–88. [Google Scholar] [CrossRef] [PubMed]
- Hadjittofi, L.; Prodromou, M.; Pashalidis, I. Activated biochar derived from cactus fibres--preparation, characterization and application on Cu(II) removal from aqueous solutions. Bioresour. Technol. 2014, 159, 460–464. [Google Scholar] [CrossRef] [PubMed]
- Pellera, F.M.; Giannis, A.; Kalderis, D.; Anastasiadou, K.; Stegmann, R.; Wang, J.Y.; Gidarakos, E. Absorption of Cu(II) ions from aqueous solutions on biochars prepared from agricultural by-products. J. Environ. Manag. 2012, 96, 35–42. [Google Scholar] [CrossRef]
- Chen, T.; Zhou, Z.; Han, R.; Meng, R.; Wang, H.; Lu, W. Adsorption of cadmium by biochar derived from municipal sewage sludge: Impact factors and adsorption mechanism. Chemosphere 2015, 134, 286–293. [Google Scholar] [CrossRef]
- Han, Y.; Boateng, A.A.; Qi, P.X.; Lima, I.M.; Chang, J. Heavy metal and phenol adsorptive properties of biochars from pyrolyzed switchgrass and woody biomass in correlation with surface properties. J. Environ. Manag. 2013, 118, 196–204. [Google Scholar] [CrossRef] [PubMed]
- Tong, X.J.; Li, J.Y.; Yuan, J.H.; Xu, R.K. Adsorption of Cu(II) by biochars generated from three crop straws. Chem. Eng. J. 2011, 172, 828–834. [Google Scholar] [CrossRef]
- Jin, H.; Hanif, M.U.; Capared, S.; Chang, Z.; Huang, H.; Ai, Y. Copper(II) removal potential from aqueous solution by pyrolysis biochar derived from anaerobically digested algae-dairy-manure and effect of KOH activation. J. Environ. Chem. Eng. 2016, 4, 365–372. [Google Scholar] [CrossRef]
- Zhao, R.; Coles, N.; Wu, J. Carbon mineralization following additions of fresh and aged biochar to an infertile soil. Catena 2015, 125, 183–189. [Google Scholar] [CrossRef]
- Suliman, W.; Harsh, J.B.; Abu-Lail, N.I.; Fortuna, A.M.; Dallmeyer, I.; Garcia-Perez, M. Influence of feedstock source and pyrolysis temperature on biochar bulk and surface properties. Biomass Bioenergy 2016, 84, 37–48. [Google Scholar] [CrossRef]
- Hu, Y.; Oduro, I.N.; Huang, Y.; Fang, Y. Structural characterization and pyrolysis behavior of holocellulose obtained from lignin-first biorefinery. J. Anal. Appl. Pyrolysis 2016, 120, 416–422. [Google Scholar] [CrossRef]
- Kim, B.S.; Lee, H.W.; Park, S.H.; Baek, K.; Jeon, J.K.; Cho, H.J.; Jung, S.C.; Kim, S.C.; Park, Y.K. Removal of Cu2+ by biochars derived from green macroalgae. Environ. Sci. Pollut. Res. 2016, 23, 985–994. [Google Scholar] [CrossRef] [PubMed]
- Melo, L.C.A.; Puga, A.P.; Coscione, A.R.; Beesley, L.; Abreu, C.A.; Camargo, O.A. Sorption and desorption of cadmium and zinc in two tropical soils amended with sugarcane-straw-derived biochar. J. Soils Sediments 2016, 16, 226–234. [Google Scholar] [CrossRef]
- Boni, M.R.; Chiavola, A.; Marzeddu, S. Application of Biochar to the Remediation of Pb-Contaminated Solutions. Sustainability 2018, 10, 4440. [Google Scholar] [CrossRef] [Green Version]
- Mireles, S.; Parsons, J.; Trad, T.; Cheng, C.L.; Kang, J. Lead removal from aqueous solutions using biochars derived from corn stover, orange peel, and pistachio shell. Int. J. Environ. Sci. Technol. 2019, 16, 5817–5826. [Google Scholar] [CrossRef]
- Park, J.H.; Ok, Y.S.; Kim, S.H.; Cho, J.S.; Heo, J.S.; Delaune, R.D.; Seo, D.C. Competitive adsorption of heavy metals onto sesame straw biochar in aqueous solutions. Chemosphere 2016, 142, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Lou, K.Y.; Rajapaksha, A.U.; Ok, Y.S.; Chang, S.X. Sorption of copper(II) from synthetic oil sands process-affected water (OSPW) by pine sawdust biochars: Effects of pyrolysis temperature and steam activation. J. Soils Sediments 2016, 16, 2081–2089. [Google Scholar] [CrossRef]
- Jiang, S.; Huang, L.; Nguyen, T.A.; Ok, Y.S.; Rudolph, V.; Yang, H.; Zhang, D. Copper and zinc adsorption by softwood and hardwood biochars under elevated sulphate-induced salinity and acidic pH conditions. Chemosphere 2016, 142, 64–71. [Google Scholar] [CrossRef]
- Ding, Z.H.; Hu, X.; Wan, Y.S.; Wang, S.S.; Gao, B. Removal of lead, copper, cadmium, zinc, and nickel from aqueous solutions by alkali-modified biochar: Batch and column tests. J. Ind. Eng. Chem. 2016, 33, 239–245. [Google Scholar] [CrossRef] [Green Version]
- Komnitsas, K.; Zaharaki, D.; Pyliotis, I.; Vamvuka, D.; Bartzas, G. Assessment of Pistachio Shell Biochar Quality and Its Potential for Adsorption of Heavy Metals. Waste Biomass Valorization 2015, 6, 805–816. [Google Scholar] [CrossRef]
- Chen, T.; Zhang, Y.; Wang, H.; Lu, W.; Zhou, Z.; Zhang, Y.; Ren, L. Influence of pyrolysis temperature on characteristics and heavy metal adsorptive performance of biochar derived from municipal sewage sludge. Bioresour. Technol. 2014, 164, 47–54. [Google Scholar] [CrossRef]
- Kołodyńska, D.; Wnętrzak, R.; Leahy, J.J.; Hayes, M.H.B.; Kwapiński, W.; Hubicki, Z. Kinetic and adsorptive characterization of biochar in metal ions removal. Chem. Eng. J. 2012, 197, 295–305. [Google Scholar] [CrossRef]
- Xu, X.; Cao, X.; Zhao, L.; Wang, H.; Yu, H.; Gao, B. Removal of Cu, Zn, and Cd from aqueous solutions by the dairy manure-derived biochar. Environ. Sci. Pollut. Res. Int. 2013, 20, 358–368. [Google Scholar] [CrossRef]
- Tan, X.; Liu, Y.; Zeng, G.; Wang, X.; Hu, X.; Gu, Y.; Yang, Z. Application of biochar for the removal of pollutants from aqueous solutions. Chemosphere 2015, 125, 70–85. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Liu, J.; Liu, R. Effects of pyrolysis temperature and heating time on biochar obtained from the pyrolysis of straw and lignosulfonate. Bioresour. Technol. 2015, 176, 288–291. [Google Scholar] [CrossRef] [PubMed]
- Claoston, N.; Samsuri, A.W.; Ahmad Husni, M.H.; Mohd Amran, M.S. Effects of pyrolysis temperature on the physicochemical properties of empty fruit bunch and rice husk biochars. Waste management & research: The journal of the International Solid Wastes and Public Cleansing Association. ISWA 2014, 32, 331–339. [Google Scholar] [CrossRef]
- Fang, K.; Li, H.; Wang, Z.; Du, Y.; Wang, J. Comparative analysis on spatial variability of soil moisture under different land use types in orchard. Sci. Hortic. 2016, 207, 65–72. [Google Scholar] [CrossRef]
- Zhao, S.X.; Ta, N.; Wang, X.D. Effect of Temperature on the Structural and Physicochemical Properties of Biochar with Apple Tree Branches as Feedstock Material. Energies 2017, 10, 1293. [Google Scholar] [CrossRef] [Green Version]
- Meng, J.; Feng, X.; Dai, Z.; Liu, X.; Wu, J.; Xu, J. Adsorption characteristics of Cu(II) from aqueous solution onto biochar derived from swine manure. Environ. Sci. Pollut. Res. Int. 2014, 21, 7035–7046. [Google Scholar] [CrossRef]
- Cao, X.; Harris, W. Properties of dairy-manure-derived biochar pertinent to its potential use in remediation. Bioresour. Technol. 2010, 101, 5222–5228. [Google Scholar] [CrossRef]
- Kołodyńska, D.; Krukowska, J.; Thomas, P. Comparison of Sorption and Desorption Studies of Heavy Metal Ions from Biochar and Commercial Active Carbon. Chem. Eng. J. 2016, 307, 353–363. [Google Scholar] [CrossRef]
- Ho, Y.S.; Mckay, G. Sorption of Dye from Aqueous Solution by Peat. Chem. Eng. J. 1998, 70, 115–124. [Google Scholar] [CrossRef]
- Cui, X.; Fang, S.; Yao, Y.; Li, T.; Ni, Q.; Yang, X.; He, Z. Potential mechanisms of cadmium removal from aqueous solution by Canna indica derived biochar. Sci. Total Environ. 2016, 562, 517–525. [Google Scholar] [CrossRef] [PubMed]
- Weber, W.J.; Morris, J.C. Kinetics of Adsorption on Carbon from Solution. J. Sanit. Eng. Div. 1963, 89, 31–60. [Google Scholar]
- Langmuir, I. The constitution and fundamental properties of solids and liquids. J. Am. Chem. Soc. 1917, 38, 102–105. [Google Scholar] [CrossRef]
- Fan, S.; Tang, J.; Wang, Y.; Li, H.; Zhang, H.; Tang, J.; Wang, Z.; Li, X. Biochar prepared from co-pyrolysis of municipal sewage sludge and tea waste for the adsorption of methylene blue from aqueous solutions: Kinetics, isotherm, thermodynamic and mechanism. J. Mol. Liq. 2016, 220, 432–441. [Google Scholar] [CrossRef]
- Wang, L.; Xu, Y.M.; Liang, X.F.; Sun, Y.B.; Lin, D.S.; Dong, R.Y. Effects of biochar and chicken manure on cadmium uptake in pakchoi cultivars with low cadmium accumulation. Zhongguo Huanjing Kexue/China Environ. Sci. 2014, 34, 2851–2858. [Google Scholar]
- Bogusz, A.; Oleszczuk, P.; Dobrowolski, R. Application of laboratory prepared and commercially available biochars to adsorption of cadmium, copper and zinc ions from water. Bioresour. Technol. 2015, 196, 540–549. [Google Scholar] [CrossRef]
- Freundlich, H. Über die Adsorption in Lösungen, Zeitschrift für physikalische Chemie. J. Am. Chem. Soc. 1906, 62, 121–125. [Google Scholar]
- Chen, X.; Chen, G.; Chen, L.; Chen, Y.; Lehmann, J.; McBride, M.B.; Hay, A.G. Adsorption of copper and zinc by biochars produced from pyrolysis of hardwood and corn straw in aqueous solution. Bioresour. Technol. 2011, 102, 8877–8884. [Google Scholar] [CrossRef]
- Tong, X.; Xu, R. Removal of Cu(II) from acidic electroplating effluent by biochars generated from crop straws. J. Environ. Sci. 2013, 25, 652–658. [Google Scholar] [CrossRef]
- Ahmad, M.; Rajapaksha, A.U.; Lim, J.E.; Zhang, M.; Bolan, N.; Mohan, D.; Vithanage, M.; Lee, S.S.; Ok, Y.S. Biochar as a sorbent for contaminant management in soil and water: A review. Chemosphere 2014, 99, 19–33. [Google Scholar] [CrossRef]
- Trakal, L.; Šigut, R.; Šillerová, H.; Faturíková, D.; Komárek, M. Copper removal from aqueous solution using biochar: Effect of chemical activation. Arab. J. Chem. 2014, 7, 43–52. [Google Scholar] [CrossRef] [Green Version]
- Zheng, W.; Li, X.M.; Wang, F.; Yang, Q.; Deng, P.; Zeng, G.M. Adsorption removal of cadmium and copper from aqueous solution by areca—A food waste. J. Hazard. Mater. 2008, 157, 490–495. [Google Scholar] [CrossRef] [PubMed]
- El-Ashtoukhy, E.S.Z.; Amin, N.K.; Abdelwahab, O. Removal of lead (II) and copper (II) from aqueous solution using pomegranate peel as a new adsorbent. Desalination 2008, 223, 162–173. [Google Scholar] [CrossRef]
- Zhang, J.; Fu, H.; Lv, X.; Tang, J.; Xu, X. Removal of Cu(II) from aqueous solution using the rice husk carbons prepared by the physical activation process. Biomass Bioenergy 2011, 35, 464–472. [Google Scholar] [CrossRef]
- Liu, Z.G.; Zhang, F.S. Removal of lead from water using biochars prepared from hydrothermal liquefaction of biomass. J. Hazard. Mater. 2009, 167, 933–939. [Google Scholar] [CrossRef] [PubMed]
- Parshetti, G.K.; Chowdhury, S.; Balasubramanian, R. Hydrothermal conversion of urban food waste to chars for removal of textile dyes from contaminated waters. Bioresour. Technol. 2014, 161, 310–319. [Google Scholar] [CrossRef]
- Banerjee, S.; Mukherjee, S.; LaminKa-ot, A.; Joshi, S.R.; Mandal, T.; Halder, G. Biosorptive uptake of Fe2+, Cu2+ and As5+ by activated biochar derived from Colocasia esculenta: Isotherm, kinetics, thermodynamics, and cost estimation. J. Adv. Res. 2016, 7, 597–610. [Google Scholar] [CrossRef] [Green Version]
- Cao, X.D.; Ma, L.; Gao, B.; Harris, W. Dairy-manure derived biochar effectively sorbs lead and atrazine. Environ. Sci. Technol. 2009, 43, 3285. [Google Scholar] [CrossRef]
- Karami, N.; Clemente, R.; Moreno-Jimnez, E.; Lepp, N.W.; Beesley, L. Efficiency of Green Waste Compost and Biochar Soil Amendments for Reducing Lead and Copper Mobility and Uptake to Ryegrass. J. Hazard. Mater. 2011, 191, 41–48. [Google Scholar] [CrossRef]
- Caporale, A.G.; Pigna, M.; Sommella, A.; Conte, P. Effect of pruning-derived biochar on heavy metals removal and water dynamics. Biol. Fertil. Soils 2014, 50, 1211–1222. [Google Scholar] [CrossRef] [Green Version]







| Sample | BC300 | BC400 | BC500 | BC600 |
|---|---|---|---|---|
| C (%) | 62.20 | 71.13 | 74.88 | 80.01 |
| H (%) | 5.18 | 4.03 | 2.88 | 2.72 |
| N (%) | 1.69 | 1.94 | 1.77 | 1.28 |
| O (%) | 24.21 | 15.05 | 10.41 | 6.59 |
| H/C | 1.00 | 0.68 | 0.46 | 0.41 |
| O/C | 0.29 | 0.16 | 0.11 | 0.06 |
| Surface area (m2·g−1) | 2.39 | 7.00 | 37.25 | 108.59 |
| Ash (%) | 6.72 | 7.85 | 10.06 | 9.41 |
| pH (5:1) | 7.48 | 11.42 | 11.62 | 10.60 |
| Cation exchange capacity (cmol·g−1) | 44.72 | 64.33 | 66.70 | 18.53 |
| Total acidic functional groups (mmol·g−1) | 0.54 | 0.39 | 0.30 | 0.22 |
| Surface carboxyl groups (mmol·g−1) | 0.30 | 0.24 | 0.17 | 0.12 |
| Surface phenol groups (mmol·g−1) | 0.12 | 0.08 | 0.07 | 0.06 |
| Surface lactone groups (mmol·g−1) | 0.12 | 0.06 | 0.05 | 0.04 |
| Conditions | Experiment 1 | Experiment 2 | Experiment 3 | Experiment 4 | Experiment 5 |
|---|---|---|---|---|---|
| Biochar dosage (g L−1) | 1, 3, 5, 10, 20 | 5 | 5 | 5 | 5 |
| Initial solution pH | 5 | 2, 3, 4, 5, 6 | 5 | 5 | 5 |
| Contact time (min) | 1440 | 1440 | 10, 20, 40, 60, 90, 120, 180, 240, 360,480, 600, 720, 960, 1200, 1440, 1680, 1920, 2400, 2880 | 1440 | 1440 |
| Reaction temperature | 25 °C | 25 °C | 25 °C | 25 °C | 25, 30, 35, and 40 °C |
| Initial Cu (II) concentration (mg L−1) | 64 | 64 | 64 | 6.4, 12.8, 38.4, 64, 128, 256, 384 | 64 |
| Initial Zu (II) concentration (mg L−1) | 65 | 65 | 65 | 6.5, 13, 39, 65, 130, 260, 390 | 65 |
| Rotation speed | 180 | 180 | 180 | 180 | 180 |
| Metal | Cu | Zn | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Biochar | BC300 | BC400 | BC500 | BC600 | BC300 | BC400 | BC500 | BC600 | |
| qe,exp | 4.96 ± 0.18d | 7.56 ± 0.27c | 9.70 ± 0.33a | 8.87 ± 0.42b | 4.30 ± 0.10d | 5.69 ± 0.13c | 9.11 ± 0.28a | 7.99 ± 0.22b | |
| PF order model | qe (mg g−1) | 4.63 ± 0.07d | 7.14 ± 0.10c | 9.23 ± 0.13a | 8.47 ± 0.12b | 4.05 ± 0.06c | 5.30 ± 0.09b | 8.77 ± 0.13a | 7.75 ± 0.11a |
| k1 × 10−3 (L·min−1) | 3.49 ± 0.20c | 7.77 ± 0.49b | 13.5 ± 0.94a | 14.36 ± 1.06a | 2.91 ± 0.16c | 5.26 ± 0.37b | 9.14 ± 0.61a | 10.33 ± 0.69a | |
| R2 | 0.93 | 0.91 | 0.90 | 0.89 | 0.93 | 0.93 | 0.92 | 0.91 | |
| PS order model | qe (mg g−1) | 5.33 ± 0.04d | 7.80 ± 0.02c | 9.86 ± 0.08a | 9.02 ± 0.02b | 4.74 ± 0.03d | 5.93 ± 0.03c | 9.54 ± 0.06a | 8.34 ± 0.04b |
| k2 × 10−3(g·mg−1·min−1) | 0.79 ± 0.15c | 1.35 ± 0.14b | 1.99 ± 0.24a | 2.37 ± 0.28a | 0.72 ± 0.06c | 1.14 ± 0.15b | 1.31 ± 0.37ab | 1.79 ± 0.0.34a | |
| h (mg·g−1·min−1) | 0.02 ± 0.00c | 0.08 ± 0.01b | 0.19 ± 0.03a | 0.19 ± 0.05a | 0.02 ± 0.00c | 0.04 ± 0.00b | 0.12 ± 0.01a | 0.12 ± 0.02a | |
| R2 | 1.00 | 1.00 | 0.99 | 1.00 | 1.00 | 1.00 | 1.00 | 0.99 | |
| IPD model | ki (mg·g−1·min−0.5) | 0.10 ± 0.01a | 0.12 ± 0.01a | 0.13 ± 0.02a | 0.11 ± 0.02a | 0.10 ± 0.20ab | 0.10 ± 0.01b | 0.14 ± 0.02a | 0.12 ± 0.02ab |
| C | 0.72 ± 0.25c | 2.56 ± 0.48b | 4.59 ± 0.64a | 4.41 ± 0.58a | 0.46 ± 0.02c | 1.35 ± 0.33b | 3.50 ± 0.61a | 3.42 ± 0.55a | |
| R2 | 0.87 | 0.73 | 0.64 | 0.65 | 0.90 | 0.81 | 0.71 | 0.67 | |
| Metal | Biochar | Langmuir Isotherm Model | Freundlich Isotherm Model | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Qmax (mg·g−1) | b (L·g−1) | R2 | RL, max | RL, min | n | Kf (mg·g−1) | R2 | ||
| Cu | BC300 | 5.20 ± 0.23d | 0.08 ± 0.02b | 0.97 | 0.66 ± 0.06a | 0.03 ± 0.01a | 4.35 ± 0.07b | 1.41 ± 0.47b | 0.74 |
| BC400 | 9.58 ± 0.18c | 0.10 ± 0.01b | 0.99 | 0.60 ± 0.02a | 0.03 ± 0.01a | 4.17 ± 0.06b | 2.58 ± 0.76ab | 0.80 | |
| BC500 | 11.41 ± 0.23a | 0.23 ± 0.02a | 0.99 | 0.41 ± 0.11b | 0.01 ± 0.01a | 5.00 ± 0.06a | 3.94 ± 1.08a | 0.76 | |
| BC600 | 10.31 ± 0.50b | 0.12 ± 0.03b | 0.96 | 0.57 ± 0.04a | 0.02 ± 0.01a | 4.55 ± 0.07b | 2.99 ± 0.99a | 0.73 | |
| Zn | BC300 | 4.98 ± 0.32c | 0.04 ± 0.01c | 0.95 | 0.79 ± 0.03a | 0.06 ± 0.01a | 3.45 ± 0.08d | 0.93 ± 0.37c | 0.74 |
| BC400 | 7.22 ± 0.36b | 0.05 ± 0.01bc | 0.97 | 0.75 ± 0.02a | 0.05 ± 0.01a | 3.70 ± 0.07c | 1.48 ± 0.52bc | 0.80 | |
| BC500 | 10.22 ± 0.52a | 0.13 ± 0.03a | 0.96 | 0.54 ± 0.07c | 0.02 ± 0.00c | 4.55 ± 0.07a | 3.10 ± 1.00a | 0.76 | |
| BC600 | 9.15 ± 0.63a | 0.09 ± 0.03ab | 0.95 | 0.63 ± 0.01b | 0.03 ± 0.00b | 4.17 ± 0.08b | 2.35 ± 0.88ab | 0.73 | |
| Heavy Metal | Biochar | ΔG (KJ·mol−1) | ΔH (KJ·mol−1) | ΔS (J·mol−1·K−1) | |||
|---|---|---|---|---|---|---|---|
| Temperature (°C) | |||||||
| 25 | 30 | 35 | 40 | ||||
| Cu | BC300 | −0.48 ± 0.06a | −0.60 ± 0.01a | −0.72 ± 0.05a | −0.83 ± 0.05a | 6.45 ± 0.43d | 23.28 ± 1.92d |
| BC400 | −1.05 ± 0.05b | −1.23 ± 0.09b | −1.41 ± 0.01b | −1.60 ± 0.04b | 9.72 ± 0.86c | 36.16 ± 0.59c | |
| BC500 | −1.27 ± 0.04c | −1.54 ± 0.08d | −1.81 ± 0.09c | −2.08 ± 0.06d | 14.78 ± 1.12a | 53.87 ± 2.72a | |
| BC600 | −1.13 ± 0.02c | −1.35 ± 0.04c | −1.57 ± 0.05c | −1.79 ± 0.02c | 11.91 ± 0.91b | 43.77 ± 1.21b | |
| Zn | BC300 | −0.30 ± 0.06a | −0.40 ± 0.02a | −0.51 ± 0.07a | −0.62 ± 0.03a | 5.93 ± 0.02b | 20.95 ± 0.28c |
| BC400 | −0.50 ± 0.07b | −0.66 ± 0.09b | −0.83 ± 0.01b | −0.99 ± 0.03b | 9.28 ± 0.81a | 32.82 ± 0.67b | |
| BC500 | −1.14 ± 0.08d | −1.32 ± 0.05d | −1.51 ± 0.02d | −1.70 ± 0.08d | 9.91 ± 0.82a | 37.11 ± 1.37a | |
| BC600 | −0.92 ± 0.06c | −1.09 ± 0.06c | −1.26 ± 0.04c | −1.43 ± 0.05c | 9.42 ± 0.76a | 34.72 ± 2.06ab | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, S.; Ta, N.; Wang, X. Absorption of Cu(II) and Zn(II) from Aqueous Solutions onto Biochars Derived from Apple Tree Branches. Energies 2020, 13, 3498. https://doi.org/10.3390/en13133498
Zhao S, Ta N, Wang X. Absorption of Cu(II) and Zn(II) from Aqueous Solutions onto Biochars Derived from Apple Tree Branches. Energies. 2020; 13(13):3498. https://doi.org/10.3390/en13133498
Chicago/Turabian StyleZhao, Shixiang, Na Ta, and Xudong Wang. 2020. "Absorption of Cu(II) and Zn(II) from Aqueous Solutions onto Biochars Derived from Apple Tree Branches" Energies 13, no. 13: 3498. https://doi.org/10.3390/en13133498




